Attitudes towards Human Papilloma Virus Vaccination in the Latin American Andean Region
Abstract
:1. Introduction
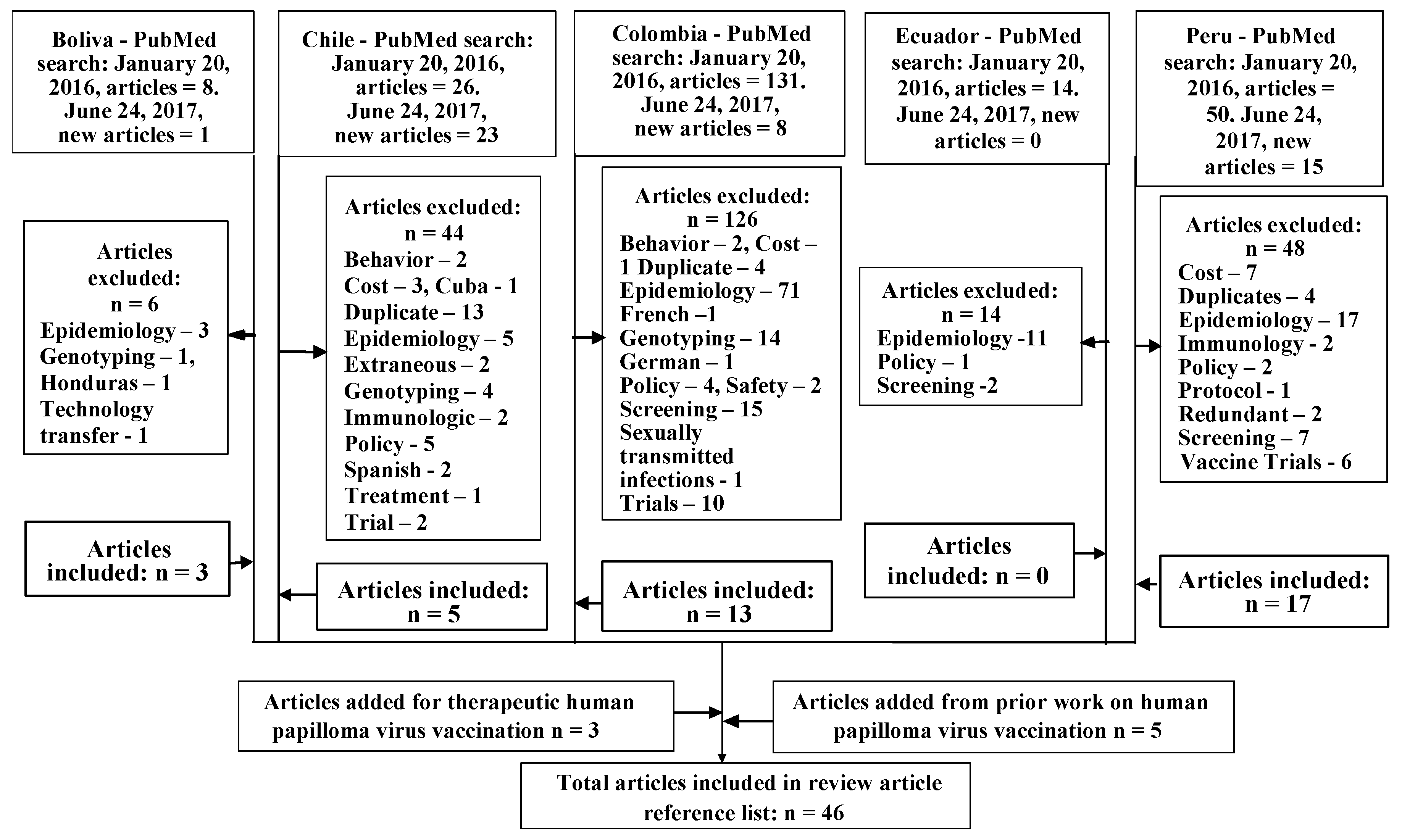
2. Materials and Methods
3. Results
3.1. Population Specific Prevalence and Sequela of HPV in the Andean Region
3.2. Chilean Attitudes towards HPV Vaccination
3.3. Colombian Attitudes towards HPV, HPV-Related Diseases, and HPV Vaccination
3.4. Peruvian Attitudes towards HPV, HPV-Related Diseases, and HPV Vaccination
4. Discussion
4.1. Comparison of Attitudes towards HPV and HPV Vaccination across Andean Nations
4.2. Means for Changing Attitudes towards HPV Vaccination
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Capote Negrin, L.G. Epidemiology of cervical cancer in Latin America. eCancerMedicalScience 2015, 9, 577. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Scalabrino, A.; Almonte, M.; Dos-Santos-Silva, I. Country-level correlates of cervical cancer mortality in Latin America and the Caribbean. Salud Publica Mex. 2013, 55, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Bartolini, R.M.; Drake, J.K.; Creed-Kanashiro, H.M.; Díaz-Otoya, M.M.; Mosqueira-Lovón, N.R.; Penny, M.E.; Winkler, J.L.; LaMontagne, D.S.; Bingham, A. Formative research to shape HPV vaccine introduction strategies in Peru. Salud Publica Mex. 2010, 52, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Goldie, S.J.; Levin, C.; Mosqueira-Lovón, N.R.; Ortendahl, J.; Kim, J.; O’Shea, M.; Sanchez, M.D.; Araujo, M.A.M. Health and economic impact of human papillomavirus 16 and 18 vaccination of preadolescent girls and cervical cancer screening of adult women in Peru. Rev. Panam. Salud Publica 2012, 32, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Penny, M.; Bartolini, R.; Mosqueira, N.R.; LaMontagne, D.S.; Mendoza, M.A.; Ramos, I.; Winkler, J.L.; Villafana, J.; Janmohamed, A.; Jumaan, A.O. Strategies to vaccinate against cancer of the cervix: Feasibility of a school-based HPV vaccination program in Peru. Vaccine 2011, 29, 5022–5030. [Google Scholar] [CrossRef] [PubMed]
- Joura, E.A.; Giuliano, A.R.; Iverson, O.E.; Bouchard, C.; Mao, C.; Mehlsen, J.; Moreira, E.D.; Ngan, Y.; Petersen, L.K.; Lazcano-Ponce, E.; et al. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N. Engl. J. Med. 2015, 372, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Quintero, K.; Giraldo, G.A.; Uribe, M.L.; Baena, A.; Lopez, C.; Alvarez, E.; Sanchez, G.I. Human papillomavirus types in cases of squamous cell carcinoma of head and neck in Colombia. Braz. J. Otorhinolaryngol. 2013, 79, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Castellsagué, X.; Alemany, L.; Quer, M.; Halec, G.; Quirós, B.; Tous, S.; Clavero, O.; Alòs, L.; Biegner, T.; Szafarowski, T.; et al. HPV involvement in head and neck cancers: Comprehensive assessment of biomarkers in 3680 patients. J. Natl. Cancer Inst. 2016, 108, djv403. [Google Scholar] [CrossRef]
- Alemany, L.; Cubilla, A.; Halec, G.; Kasamatsu, E.; Quirós, B.; Masferrer, E.; Tous, S.; Lloveras, B.; Hernández-Suarez, G.; Lonsdale, R.; et al. Role of human papillomavirus in penile carcinomas worldwide. Eur. Urol. 2016, 69, 953–961. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, G.L. Parental attitudes towards vaccinating sons with human papillomavirus vaccine. Dan. Med. Bull. 2010, 57, A4230. [Google Scholar] [PubMed]
- Piñeros, M.; Hernández-Suárez, G.; Orjuela, L.; Vargas, J.C.; Pérez, G. HPV knowledge and impact of genital warts on self-esteem and sexual life in Colombian patients. BMC Public Health 2013, 13, 272. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, G.I.; Jaramillo, R.; Cuello, G.; Quintero, K.; Baena, A.; O’Byrne, A.; Reyes, A.J.; Santamaria, C.; Cuello, H.; Arrunategui, A.; et al. Human papillomavirus genotype detection in recurrent respiratory papillomatosis (RRP) in Colombia. Head Neck 2013, 35, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Soohoo, M.; Blas, M.; Byraiah, G.; Carcamo, C.; Brown, B. Cervical HPV Infection in Female Sex Workers: A Global Perspective. Open AIDS J. 2013, 7, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Perez, G.; Lazcano-Ponce, E.; Hernandez-Avila, M.; García, P.J.; Muñoz, N.; Villa, L.L.; Bryan, J.; Taddeo, F.J.; Lu, S.; Esser, M.T.; et al. Safety, immunogenicity, and efficacy of quadrivalent human papillomavirus (types 6, 11, 16, 18) L1 virus-like-particle vaccine in Latin American women. Int. J. Cancer 2008, 122, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Bruni, L.; Diaz, M.; Barrionuevo-Rosas, L.; Herrero, R.; Bray, F.; Bosch, F.X.; de Sanjosé, S.; Castellsagué, X. Global estimates of human papillomavirus vaccination coverage by region and income level: A pooled analysis. Lancet Glob. Health 2016, 4, e453–e463. [Google Scholar] [CrossRef]
- Brown, B.; Carcamo, C.; Blas, M.M.; Valderrama, M.; Halsey, N. Peruvian FSWs: Understanding HPV and barriers to vaccination. Vaccine 2010, 28, 7743–7747. [Google Scholar] [CrossRef] [PubMed]
- Cerda, A.A.; García, L.Y.; Gaete, F.I.; Pizarro, T.H. Disposición a pagar por vacunas contra el virus papiloma humano en la Región Metropolitana de Santiago de Chile. Rev. Med. Chile 2013, 141, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Cerda, A.A.; García, L.Y.; Albornoz, D.V. Disposición de los padres a pagar por una vacuna contra el virus del papiloma humano para sus hijas adolescentes. Región del Maule, Chile. Salud Pública Méx. 2014, 56, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Lee, F.H.; Paz-Soldan, V.A.; Carcamo, C.; Garcia, P.J. Knowledge and attitudes of adult Peruvian women vis-a-vis Human Papillomavirus (HPV), cervical cancer, and the HPV vaccine. J. Low Genit. Tract Dis. 2010, 14, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Sternberg, A.M.; Pinzón-Rondón, A.M. Risk perception and sexual behavior in HPV-vaccinated and unvaccinated young Colombian women. Int. J. Gynecol. Obstet. 2014, 126, 205–208. [Google Scholar] [CrossRef] [PubMed]
- Han, C.S.; Ferris, D.G.; Waller, J.; Tharp, P.; Walter, J.; Allmond, L. Comparison of knowledge and attitudes toward human papillomavirus, HPV vaccine, pap tests, and cervical cancer between US and Peruvian Women. J. Low Genit. Tract Dis. 2012, 16, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Murillo, R.; Molano, M.; Martínez, G.; Mejía, J.C.; Gamboa, O. HPV prevalence in Colombian women with cervical cancer: Implications for vaccination in a developing country. Infect. Dis. Obstet. Gynecol. 2009, 2009, 653598. [Google Scholar] [CrossRef] [PubMed]
- Bedoya, A.M.; Gaviria, A.M.; Baena, A.; Borrero, M.; Duarte, D.F.; Combita, A.L.; Castaño, J.; Grisales, H.; Sánchez, G.I. Age-specific seroprevalence of human papillomavirus 16, 18, 31, and 58 in women of a rural town of Colombia. Int. J. Gynecol. Cancer 2012, 22, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Camargo, M.; Soto-De Leon, S.C.; Sanchez, R.; Perez-Prados, A.; Patarroyo, M.A. Frequency of human papillomavirus infection, coinfection, and association with different risk factors in Colombia. Ann. Epidemiol. 2011, 21, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, J.; Lema, C.; Hurtado, L.; Andrade, R.; Quiroga, G.; Garcia, G.; Torricos, L.; Zegarra, L.; Vera, V.; Panoso, W.; et al. Prevalence of human papillomavirus infection in rural villages of the Bolivian Amazon. Rev. Inst. Med. Trop. Sao Paulo 2003, 45, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.; Blas, M.M.; Cabral, A.; Carcamo, C.; Gravitt, P.E.; Halsey, N. Oral sex practices, oral human papillomavirus and correlations between oral and cervical human papillomavirus prevalence among female sex workers in Lima, Peru. Int. J. STD AIDS 2011, 22, 655–658. [Google Scholar] [CrossRef] [PubMed]
- Ferreccio, C.; van de Wyngard, V.; Olcay, F.; Domínguez, M.A.; Puschel, K.; Corvalán, A.H.; Franceschi, S.; Snijders, P.J.F. High-risk HPV infection after five years in a population-based cohort of Chilean women. Infect. Agents Cancer 2011, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Balanda, M.; Quiero, A.; Vergara, N.; Espinoza, G.; San Martín, H.; Rojas, G.; Ramírez, E. Prevalence of human papillomavirus infection among women presenting for cervical cancer screening in Chile, 2014–2015. Med. Microiol. Immunol. 2016, 205, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Vergara, N.; Espinoza, G.; Balanda, M.; Quiero, A.; Hidalgo, W.; San Martín, H.; Ramírez, A.; Ramírez, E. Prevalence of human papillomavirus infection among Chilean women from 2012 to 2016. J. Med. Virol. 2017, 89, 1646–1653. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.M.; Ruiz, J.E.; Gavilanes, A.V.; Eriksson, T.; Lehtinen, M.; Pérez, G.; Sings, H.L.; James, M.K.; Haupt, R.M.; for the FUTURE I and II Study Group. Proximity of first sexual intercourse to menarche and risk of high-grade cervical disease. J. Infect. Dis. 2012, 206, 1887–1896. [Google Scholar] [CrossRef] [PubMed]
- Urrutia, M.T.; Concha, X.; Riquelme, G.; Padilla, O. Conocimientos y conductas preventivas sobre cáncer cérvico-uterino y virus papiloma humano en un grupo de adolescentes chilenas. Rev. Chilena Infectol. 2012, 29, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, R.; Gustat, J.; Hagensee, M.E.; Baena, A.; Salazar, J.E.; Castro, M.V.; Gaviria, A.M.; Sánchez, G.I. Knowledge of pap screening and human papillomavirus among women attending clinics in Medellín, Colombia. Int. J. Gynecol. Cancer 2008, 18, 1020–1026. [Google Scholar] [CrossRef] [PubMed]
- Luque, J.S.; Maupin, J.N.; Ferris, D.G.; Condorhuaman, G.W.S. Reaching women in the Peruvian Andes through cervical cancer screening campaigns: Assessing attitudes of stakeholders and patients. Patient Prefer. Adherence 2016, 10, 2107–2116. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.; Blas, M.M.; Heidari, O.; Carcamo, C.; Halsey, N.A. Reported changes in sexual behavior and human papillomavirus knowledge in Peruvian female sex workers following participation in a human papillomavirus vaccine trial. Int. J. STD AIDS 2013, 23, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Abuelo, C.E.; Levinson, K.L.; Salmeron, J.; Sologuren, C.V.; Fernandez, M.J.V.; Belinson, J.L. The Peru Cervical Cancer Screening Study (PERCAPS): The design and implementation of a mother/daughter screen, treat, and vaccinate program in the Peruvian Jungle. J. Community Health 2014, 39, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Bartolini, R.M.; Winkler, J.L.; Penny, M.E.; LaMontagne, D.S. Parental acceptance of HPV vaccine in Peru: A decision framework. PLoS ONE 2012, 7, e48017. [Google Scholar] [CrossRef] [PubMed]
- Bingham, A.; Drake, J.K.; LaMontagne, D.S. Sociocultural issues in the introduction of human papillomavirus vaccine in low-resource settings. Arch. Pediatr. Adolesc. Med. 2009, 163, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Wiesner, C.; Piñeros, M.; Trujillo, L.M.; Ardila, J. Aceptabilidad de la vacuna contra el Virus Papiloma Humano en padres de adolescentes, en Colombia. Rev. Salud Pública 2010, 12, 961–973. [Google Scholar] [PubMed]
- Piñeros, M.; Wiesner, C.; Cortés, C.; Trujillo, L.M. HPV vaccine introduction at the local level in a developing country: Attitudes and criteria among key actors. Cad. Saude Publica 2010, 26, 900–908. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.; Monsour, E.; Klausner, J.D.; Galea, J.T. Sociodemographic and behavioral correlates of anogenital warts and human papillomavirus-related knowledge among men who have sex with men and transwomen in Lima, Peru. Sex. Transm. Dis. 2015, 42, 198–201. [Google Scholar] [CrossRef] [PubMed]
- Nureña, C.R.; Brown, B.; Galea, J.T.; Sánchez, H.; Blas, M.M. HPV and genital warts among Peruvian men who have sex with men and transgender people: Knowledge, attitudes and treatment experiences. PLoS ONE 2013, 8, e58684. [Google Scholar] [CrossRef] [PubMed]
- Galea, J.T.; Monsour, E.; Nureña, C.R.; Blas, M.M.; Brown, B. HPV vaccine knowledge and acceptability among Peruvian men who have sex with men and transgender women: A pilot, qualitative study. PLoS ONE 2017, 12, e0172964. [Google Scholar] [CrossRef] [PubMed]
- Trimble, C.L.; Morrow, M.P.; Kraynyak, K.A.; Shen, X.; Dallas, M.; Yan, J.; Edwards, L.; Parker, R.L.; Denny, L.; Giffear, M.; et al. Safety, efficacy, and immuunogenicity of VGX-3100, a therapeutic synthetic DNA vaccine targeting human papillomavirus 16 and 18 E6 and E7 proteins for cervical intraepithelial neoplasia 2/3: A randomized, double-blind, placebo controlled phase 2b trial. Lancet 2015, 386, 2078–2088. [Google Scholar] [CrossRef]
- Alvarez, R.D.; Huh, W.K.; Bae, S.; Lamb, L.S.; Conner, M.G.; Boyer, J.; Wang, C.; Hung, C.F.; Sauter, E.; Paradis, M.; et al. A pilot study of pNGVL4a-CRT/E7 (detox) for the treatment of patients with HPV16+ cervical intraepithelial neoplasia 2/3 (CIN2/3). Gynecol. Oncol. 2016, 140, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Stevanović, S.; Draper, L.M.; Langham, M.M.; Campbell, T.E.; Kwong, M.L.; Wunderlich, J.R.; Dudley, M.E.; Yang, J.C.; Sherry, R.M.; Kammula, U.S.; et al. Complete regression of metastatic cervical cancer after treatment with human papillomavirus-targeting tumor-infiltrating T cells. J. Clin. Oncol. 2015, 33, 1543–1550. [Google Scholar] [CrossRef] [PubMed]
- LaMontagne, D.S.; Barge, S.; Le, N.T.; Mugisha, E.; Penny, M.E.; Gandhi, S.; Janmohamed, A.; Kumakech, E.; Mosqueira, N.R.; Nguyen, N.Q.; et al. Human papillomavirus vaccine delivery strategies that achieved high coverage in low- and middle-income countries. Bull. World Health Organ. 2011, 89, 821–830. [Google Scholar] [CrossRef] [PubMed]
| Bolivia | Chile | Colombia | Ecuador | Peru | |
|---|---|---|---|---|---|
| 2012 New CC cases | 2029 | 1441 | 4661 | 2094 | 4636 |
| 2012 Standardized CC incidence per 100,000 women | 47.7 | 2.8 | 18.7 | 29.0 | 32.7 |
| 2012 CC deaths | 845 | 734 | 1986 | 1026 | 1715 |
| 2012 Standardized CC mortality per 100,000 women | 21 | 6.0 | 8.0 | 14.0 | 12.0 |
| Life expectancy (years) | 69 | 82 | 78 | 79 | 77 |
| 2012 Potential years of life lost due to CC | 185,108 | 19,818 | 45,678 | 24,624 | 37,730 |
| Cytological cover | 12.00 | 60.00 | 79.00 | 64.30 | 51.30 |
| Estimated mortality under five years old per 1000 | 67.6 | 9.3 | 30.9 | 28.2 | 49.5 |
| Per capita total health expenditure at International dollar rate | 176 | 707 | 522 | 220 | 233 |
| Proportion of population with access to improved sanitation | 45 | 92 | 86 | 72 | 62 |
| % of women screened | - | 51.4 | 67 | 31.3 | 22.7 |
| % false-negative Pap tests | <5 | 27 | |||
| % abnormal Pap test follow-up | 59 | >90 | 25–34 |
| Epidemiology | ||
| Reference | Nation(s) | Primary Results |
| [1] | - | Epidemiology of cervical cancer (CC) in Latin America using GLOBOCAN data from 2000, 2008, and 2012 is summarized in Table 1. |
| [2] | Latin America | GLOBOCAN 2008 CC data with 2000–2008 demographic, socioeconomic, and public health correlates. |
| [7] | Colombia | Human papilloma virus (HPV)-16 and -18 are present in almost 20% cases of HNSCC. |
| [8] | - | HPV may be less involved than previously believed in HNSCC other than oropharyngeal cancer. Gender, global location, and cancer site affect HPV contribution to HNSCC. |
| [9] | - | Globally, HPV may be more prevalent in high grade squamous intraepithelial lesions of the penis than in invasive penile cancer. |
| [12] | Colombia | HPV-6, -11, and -16 were found in 69%, 27.1%, and 7.8% of HPV positive recurrent respiratory papillomatosis. Quadrivalent HPV vaccine (4vHPV) would be a more effective vaccine than bivalent HPV vaccine (2vHPV). |
| [13] | Peru | In Lima, HPV prevalence in female sex workers (FSW) ranges from 50.6 to 66.8%. |
| [22] | Colombia | HPV-31, -33, -45, and -58 may be more prevalent than HPV-18 in CC. Colombia would benefit from an HPV vaccine other than 2vHPV or 4vHPV. |
| [23] | Colombia | Age-specific seroprevalence of HPV-16, -18, -31, and -58 in women of a rural town was associated with a greater number of lifetime sexual partners (OR = 3.05, 95% confidence interval [CI] 1.26–7.37), and having >2 regular sexual partners (odds ratio [OR] 3, 95% CI 1.21–7.45). In women >45 years old, oral contraceptive use and tobacco smoking were associated with reduced HPV prevalence. |
| [24] | Colombia | Geographic region, ethnicity, parity, and lifetime number of sexual partners affect HPV infection. |
| [25] | Bolivia | HPV prevalence in 4 Amazonian villages ranged from 0 to 16.6%, related to enetic milieu or lifestyle factors limiting HPV exposure or effect. |
| [26] | Peru | In Lima, FSW oral HPV prevalence (7.6%) is associated with cervical HPV prevalence (71.4% if oral HPV positive). |
| [27] | Chile | From 2001 to 2005 female high-risk (HR) HPV infection rose by 43%. Dual peak HR HPV prevalence in women <20 years old and 45–55 years old. HR HPV infections in women <30 years old cleared in the five-year interval; 4.7% were HR HPV positive in 2001 and 2005. |
| [28] | Chile | From 2014 to 2015 in Santiago HPV prevalance in CC screening was 11.1%, of which 9.7% were high risk. HPV-66 (1.4%), -51 (1.2%), and -59 (1.2%), were more prevalent than HPV-18. |
| [29] | Chile | From 2012 to 2016 in Santiago, pap tests had a 12% HPV prevalence. Rates varied between primary care and referral centers. |
| [30] | Colombia, Finland | Coitarche within three years of menarche (12.4 and 16 years) increases cytologic abnormalities (OR 1.65, 95% CI 1.02–2.68, p = 0.04 and cervical intraepithelial neoplasia (CIN)2-3/adenocarcinoma in situ (AIS) (OR 3.56, 95% CI 1.02–12.47, p = 0.05). |
| Knowledge and Knowledge Gaps | ||
| Reference | Nation(s) | Primary Results |
| [5] | Peru | The Piura region school-based HPV vaccination project required extensive vaccinator training, coordination with schools and education authories, sensitization of parents, adolescent daughters, teachers, and the community. Isolated, small schools were difficult and expensive (time and resources) to reach. Three-dose HPV vaccine administration tracking for each recipient was difficult. |
| [11] | Colombia | Men and women with external genital warts (EGW) had little HPV knowledge. EGW adversely affected women’s self esteem more than men’s self esteem (90.3% versus 60.4%, p < 0.001). EGW adversely affected women’s sexual life more than men’s (83% versus 66%, p = 0.05). |
| [16] | Peru | FSW in Lima have little HPV, HPV vaccine, and CC knowledge. |
| [31] | Chile | Chilean adolescents were aware that multiple sexual partners is a CC risk factor (70.8%) and a HPV infection risk factor (78.3%). While 68.2% were aware that unprotected sex facilitates HPV transmission, only 31.1% used condoms. |
| [32] | Colombia | Women attending clinics in Medellín were aware of CC screening (76.3%), but only 7.8% had HPV knowledge. Education, marital status, household income, and insurance affected CC screening knowledge. Education, age, and household income affected HPV knowledge. |
| [33] | Peru | Forty-six participant, mixed method, descriptive case study of CerviCusco programs. Explored HPV knowledge and CC attitudes and beliefs. Half of participants were unaware that CC is preventable. |
| [34] | Peru | FSW participation in a HPV vaccine trial was associated with increased HPV and HPV-related disease knowledge, increased awareness of HPV prevention strategies, and with a reduction in new and total clients, p = 0.001. |
| Finance and Cost | ||
| Reference | Nation(s) | Primary Results |
| [4] | Peru | 2vHPV vaccination of preadolescent girls with an 82% three-dose completion and 10% coverage CC screening of adult women would reduce the lifetime risk of CC by 58% at a cost of less than US$500 per year of life saved. Annual program cost ranges from US$5 million at US$5/dose to US$16 million at US$20/dose. |
| [17] | Chile | Parents of teenage daughters in Santiago were willing to pay US$758 for HPV vaccination series. But, 25% of parents would not pay for HPV vaccine. However, at half the price, 96% of parents would pay for HPV vaccine. Household income and size affect willingness to pay. |
| [18] | Chile | Double blinded format contingent valuation ascertained that parents of teenage daughters were willing to pay US$252.71 for HPV vaccination series in Maule region. Cost per dose and number of daughters lower the amount parents will pay. Household income raises the amount parents will pay. Shared funding between government and parents increases access of teenage daughters to HPV vaccination. |
| [19] | Peru | In Puente Piedra, Los Olivos, and Comas, Lima, although 59–71% of low-income 25–65 year old women were unaware of HPV, HPV vaccine, and CC, 90% would accept HPV vaccination, and 58% were willing to pay “something” for HPV vaccination. |
| Policy | ||
| Reference | Nation(s) | Primary Results |
| [35] | Peru | A mother/daughter screen, treat, and vaccinate program around Iquitos, in the Peruvian jungle, achieved an 88% two-dose and a 65% three-dose 4vHPV vaccination rate. |
| [36] | Peru | If enough credible sources, including health workers and teachers promote HPV vaccine, parents will accept HPV vaccine. Health promotion, CC prevention, and trust in vaccines drive acceptance. Accessibility via free, school-based administration, and media campaigns motivate parental acceptance of HPV vaccination. Problems with the health system drive HPV vaccine refusal. |
| Vaccination Barriers | ||
| Reference | Nation(s) | Primary Results |
| [20] | Colombia | HPV vaccination was not associated with sexual risk behavior young women in Bogotá. HPV vaccination was associated with other preventive health measures: Routine contraception and condom use, and CC screening. HPV vaccinated women percieved their risks of CC, EGW, and HPV infection to be less than did unvaccinated women. |
| [21] | Peru, United States | Spanish-speaking Peruvian women were most embarrassed and afraid to have pap tests (OR = 17.25, 14.43 respectively), less likely to know that the HPV vaccine is safe and effective (OR = 0.11), and less likely to know that HPV causes CC (OR = 0.03). |
| [37] | Peru | Adolescents’ mothers are vaccination decision-makers. Girls can choose for themselves. Teachers and principals are involved in school-based vaccination program decision-making. The Peruvian government has a role in determining what vaccines are available. Peruvian administrators, health care workers, and school staff have greater awareness of CC than do adolescents and their parents, but all had limited knowledge that CC is a HPV-related disease. Awareness that vaccines may be preventive or curative. Peruvians have concerns about vaccinators’ effective interactions with female HPV vaccine recipients and vaccination administration safety practices. Fear of vaccine-induced sterilization due to coercive sterilizations in the 1990s. Fear of vaccine induced precocious puberty and experimental vaccines. |
| [38] | Colombia | In Arauca, Bogotá, Cartagena, and Manizales parents of daughters younger than 12 years old were resistant to HPV vaccination to prevent sexually-transmitted infection (STI), and were concerned of sexual disinhibition, but would accept HPV vaccination to prevent CC. Parents of higher socioeconomic status were less accepting of HPV vaccination. |
| [39] | Colombia | Qualitative study. Interviews with HPV vaccine program decision-makers 1–2 years after introduction of 2vHPV and 4vHPV. Interest in CC and EGW prevention. Need for information sources other than pharmaceutical representatives, to reduce HPV vaccine cost. Lack of resources to vaccinate, screen, and treat. Ethics of economic inequity. |
| [40] | Peru | An HPV-related knowledge gap exists among men who have sex with men (MSM) and transwomen in Lima, that may be due to sex work stigmatization, STI underreporting, and lack of access to HPV vaccine. |
| [41] | Peru | MSM and transgender people in Lima had limited HPV knowledge, but were aware that EGW are a STI. EGW were stigmatizing, negatively affecting access to care, which was less used when EGW could not be effectively treated. |
| [42] | Peru | Focus group and individual interviews with 36 MSM and transgender women in Lima found mostly positive attitudes towards HPV vaccination, but concern for being stigmatized if positive HPV vaccination status was disclosed. |
| Vaccination Efficacy | ||
| Reference | Nation(s) | Primary Results |
| [6] | - | The nonavalent HPV (9vHPV) vaccine generated antibody response to HPV 6,11, 16, and 18 is noninferior to the 4vHPV vaccine. 9vHPV vaccine is also prophylactic against infection and intraepithelial neoplasia in women from HPV-31, -33, -45, -52, and -58. |
| [14] | Colombia, Peru | 4vHPV vaccine had 92.8% and 100% efficacy in preventing CIN and EGW, respectively, in Latin American women. |
| [43] | - | Phase 2b trial of VGX-3100 therapeutic synthetic DNA vaccine targeting HPV 16 and 18 E6 and E7 proteins for CIN 2/3 achieved 18.2 percentage point improved regression than placebo, p = 0.034. |
| [44] | - | Thirty percent histologic regression from CIN 2/3 to CIN 1 at 15 weeks following initial vaccination dose, in patients who received three doses of pNGVL4a-CRT-E7 (detox) vaccination. |
| [45] | - | Complete and partial regression of metastatic CC occurred in two of nine and one of nine patients, respectively, who received a single adoptive T-cell therapy infusion comprised of tumor-infiltrating T cells with possible HPV E6 and E7 reactivity. |
| Vaccination Safety | ||
| Reference | Nation(s) | Primary Results |
| [3] | Peru | Policymakers and parents support HPV vaccine introduction, but communities have vaccination safety and quality concerns. Vaccines are seen as preventive not curative. A few parents were concerned about vaccine-related promiscuity and precocious puberty. |
| [46] | Peru | Piura region school-based HPV vaccination achieved a 82.6% three-dose completion rate. HPV vaccination was accepted for CC prevention and disease prevention due to positive beliefs concerning vaccination and free vaccination provision. HPV vaccination refusal was due to fear of experimentation, being dissuaded against HPV vaccination, and “allergies”. School absenteeism resulted in non-vaccination. |
| Chile | Colombia | Peru | |||
|---|---|---|---|---|---|
| Aware that cervical cancer risk increases with more sexual partners | 70.8% of adolescents | ||||
| Aware of HPV | 44–69%. Higher with higher socioeconomic class | ||||
| Aware that HPV causes CC | Quechua women—42.4%; Bilingual women—36.2%; Spanish speaking women—29.9%; Urban women—38% | ||||
| Aware of HPV vaccination | 25–59% | ||||
| Aware that HPV vaccination prevents CC and external genital warts | Quechua women—81%; Bilingual women—87.8%; Spanish speaking women—78.6% | ||||
| Aware that HPV is transmitted by unprotected sex (HPV is a sexually transmitted infection (STI)) | 68.2% of adolescents | Quechua women—86.7%; Bilingual women—84.9%; Spanish speaking women—80.7% | |||
| Normally use condoms | 31.1% of adolescents | 28% of HIV negative MSM and TW use condoms for anal or oral sex | |||
| Best time to receive HPV vaccination | After coitarche Quechua women—61%; Bilingual women—76.7%; Spanish speaking women—71.3% | ||||
| Parents approve of HPV vaccinations for their daughters to prevent STI | Resistant | ||||
| Fear of post-vaccination sexual disinhibition | Concern does not override choice to prevent CC | Limited | |||
| Parents approve of HPV vaccinations for their daughters to prevent CC | Higher socioeconomic class parents are less supportive than lower socioeconomic class parents | Joint decision between parents, children, teachers, and school principles | |||
| Parental HPV vaccination acceptance decision | Rural parents are quicker to accept than urban parents | ||||
| Actual HPV non-vaccination rate and reason | 30%—lack of information | MSM | |||
| 28.9%—access barriers | Lack of a male HPV vaccination program | ||||
| 21%—unknown | Non-realization of cancer prevention need | ||||
| 20%—neglect | Societal and healthcare provision homophobia | ||||
| External genital warts negative effect | Women | Men | MSMTraumatic, become the passive partner, start to have sex in darkness. | ||
| Self-esteem | 90% | 62% | |||
| Sexuality | 77% | 46% | |||
| External genital warts positive effect | Prompts MSM HPV vaccine acceptance | ||||
| HPV knowledge sources | Media—82.25%; Social—72.4%; Educational—77.8% | ||||
| HPV vaccination knowledge sources | Media—59.8%; Social—44.8%; Educational—50% | ||||
| HPV vaccination concerns | Covert reproductive sterilization | ||||
| Covert cancer dissemination | |||||
| Experimental, unsafe vaccine | |||||
| Fear from previous vaccination adverse events | |||||
| Post-expiration vaccine administration | |||||
| Vaccine initiated reproductive development | |||||
| Vaccinator training inadequacy, non-sterile vaccination | |||||
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nwanodi, O. Attitudes towards Human Papilloma Virus Vaccination in the Latin American Andean Region. Healthcare 2017, 5, 55. https://doi.org/10.3390/healthcare5030055
Nwanodi O. Attitudes towards Human Papilloma Virus Vaccination in the Latin American Andean Region. Healthcare. 2017; 5(3):55. https://doi.org/10.3390/healthcare5030055
Chicago/Turabian StyleNwanodi, Oroma. 2017. "Attitudes towards Human Papilloma Virus Vaccination in the Latin American Andean Region" Healthcare 5, no. 3: 55. https://doi.org/10.3390/healthcare5030055
APA StyleNwanodi, O. (2017). Attitudes towards Human Papilloma Virus Vaccination in the Latin American Andean Region. Healthcare, 5(3), 55. https://doi.org/10.3390/healthcare5030055






