Preoperative Risk Factors for Periprosthetic Joint Infection: A Narrative Review of the Literature
Abstract
1. Introduction
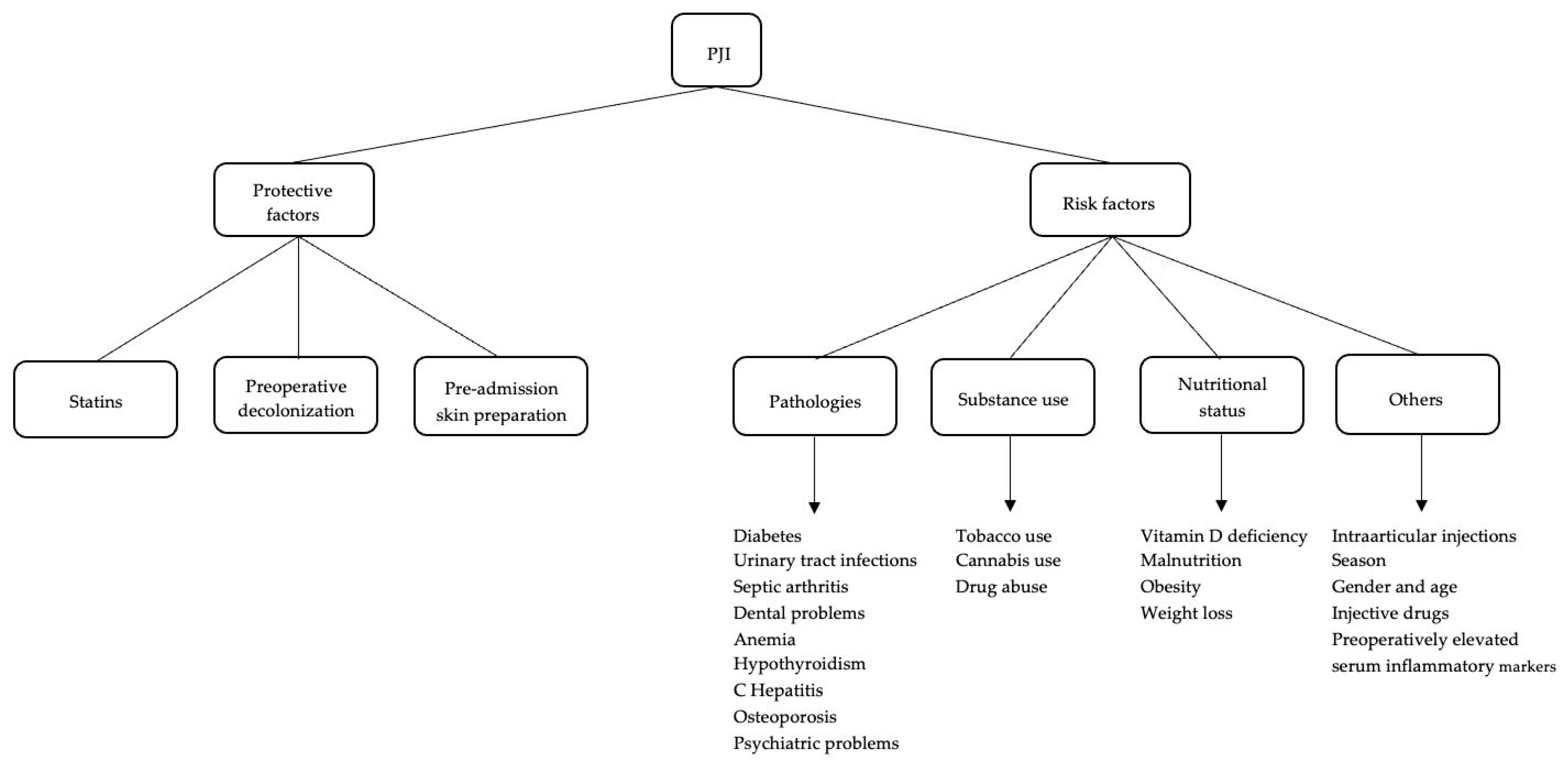
2. Risk Factors
2.1. Pathologies
2.1.1. Diabetes
2.1.2. Urinary Tract Infections
2.1.3. Septic Arthritis
2.1.4. Dental Problems
2.1.5. Anemia
2.1.6. Hypothyroidism
2.1.7. Hepatitis
2.1.8. Osteoporosis
2.1.9. Psychiatric Disorders
2.2. Substance Use
2.2.1. Tobacco Use
2.2.2. Tobacco and Cannabis Concomitant Use
2.2.3. Drug Abuse
2.3. Nutritional Status
2.3.1. Vitamin D Deficiency
2.3.2. Malnutrition
2.3.3. Obesity
2.3.4. Weight Loss
2.4. Other
2.4.1. Injections
2.4.2. Season
2.4.3. Gender and Age
2.4.4. Drugs
2.4.5. Preoperatively Elevated Serum Inflammatory Markers
3. Defensive Factors
3.1. Statins
3.2. Preoperative Decolonization
3.3. Preadmission Skin Preparation
4. Conclusions and Future Recommendations
- Identifying all risk factors reported in the literature is very challenging. However, it is fundamental to know, understand, and contrast all the factors that may higher the risk of PJI. Optimizing preoperative assessment before elective arthroplasty is crucial for identifying and addressing these diverse risk factors;
- The absence of widely accepted and standardized protocols for PJI prevention adds complexity to this issue. Although our comprehension of PJI and its associated risks is substantial, it persists as a significant challenge for both arthroplasty surgeons and patients. A pivotal strategy in overcoming this feared complication is prioritizing prevention. The identification of modifiable risk factors and the implementation of evidence-based preoperative protocols becomes paramount, aiming to maximize patient safety and enhance the overall benefits of undergoing arthroplasties. The main limitation is the subjective selection of the articles that was limited to the most recent studies that seemed related to the topic. Furthermore, several factors have been reported without considering the impact they have on the risk of infection and how it differs between individuals or in associations with each other. By evaluating how each factor influences the risk of periprosthetic joint infection, specific preoperative protocols can be created, particularly for patients with multiple comorbidities and more complex clinical conditions.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kong, L.; Cao, J.; Zhang, Y.; Ding, W.; Shen, Y. Risk Factors for Periprosthetic Joint Infection Following Primary Total Hip or Knee Arthroplasty: A Meta-analysis. Int. Wound J. 2017, 14, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Bozic, K.J.; Lau, E.; Kurtz, S.; Ong, K.; Rubash, H.; Vail, T.P.; Berry, D.J. Patient-Related Risk Factors for Periprosthetic Joint Infection and Postoperative Mortality Following Total Hip Arthroplasty in Medicare Patients. J. Bone Jt. Surg. 2012, 94, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Kunutsor, S.K.; Beswick, A.D.; Whitehouse, M.R.; Blom, A.W.; Lenguerrand, E. Implant Fixation and Risk of Prosthetic Joint Infection Following Primary Total Hip Replacement: Meta-Analysis of Observational Cohort and Randomised Intervention Studies. JCM 2019, 8, 722. [Google Scholar] [CrossRef] [PubMed]
- Zmistowski, B.; Karam, J.A.; Durinka, J.B.; Casper, D.S.; Parvizi, J. Periprosthetic Joint Infection Increases the Risk of One-Year Mortality: J. Bone Jt. Surg. 2013, 95, 2177–2184. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, S.M.; Lau, E.; Watson, H.; Schmier, J.K.; Parvizi, J. Economic Burden of Periprosthetic Joint Infection in the United States. J. Arthroplast. 2012, 27, 61–65.e1. [Google Scholar] [CrossRef]
- Bozic, K.J.; Kurtz, S.M.; Lau, E.; Ong, K.; Chiu, V.; Vail, T.P.; Rubash, H.E.; Berry, D.J. The Epidemiology of Revision Total Knee Arthroplasty in the United States. Clin. Orthop. Relat. Res. 2010, 468, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.S.; Twaij, H.; Haddad, F.S. Two-Stage Revision for the Culture-Negative Infected Total Hip Arthroplasty: A Comparative Study. Bone Jt. J. 2018, 100-B, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Middleton, R.; Khan, T.; Alvand, A. Update on the Diagnosis and Management of Prosthetic Joint Infection in Hip and Knee Arthroplasty. Bone Jt. 360 2019, 8, 5–13. [Google Scholar] [CrossRef]
- McNally, M.; Sousa, R.; Wouthuyzen-Bakker, M.; Chen, A.F.; Soriano, A.; Vogely, H.C.; Clauss, M.; Higuera, C.A.; Trebše, R. The EBJIS Definition of Periprosthetic Joint Infection: A Practical Guide for Clinicians. Bone Jt. J. 2021, 103-B, 18–25. [Google Scholar] [CrossRef]
- Kunutsor, S.K.; Whitehouse, M.R.; Blom, A.W.; Beswick, A.D. INFORM Team Patient-Related Risk Factors for Periprosthetic Joint Infection after Total Joint Arthroplasty: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0150866. [Google Scholar] [CrossRef] [PubMed]
- Resende, V.A.C.; Neto, A.C.; Nunes, C.; Andrade, R.; Espregueira-Mendes, J.; Lopes, S. Higher Age, Female Gender, Osteoarthritis and Blood Transfusion Protect against Periprosthetic Joint Infection in Total Hip or Knee Arthroplasties: A Systematic Review and Meta-Analysis. Knee Surg. Sports Traumatol. Arthrosc. 2021, 29, 8–43. [Google Scholar] [CrossRef] [PubMed]
- Kee, J.R.; Mears, S.C.; Edwards, P.K.; Barnes, C.L. Modifiable Risk Factors Are Common in Early Revision Hip and Knee Arthroplasty. J. Arthroplast. 2017, 32, 3689–3692. [Google Scholar] [CrossRef]
- Chan, V.W.; Chan, P.; Fu, H.; Cheung, M.; Cheung, A.; Yan, C.; Chiu, K. Preoperative Optimization to Prevent Periprosthetic Joint Infection in At-Risk Patients. J. Orthop. Surg. 2020, 28, 230949902094720. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, J.D.; Lepkowsky, E.R.; Callari, M.M.; Jordan, E.T.; Koenig, J.A.; Sirounian, G.H. The Prevalence of Diabetes Mellitus and Routine Hemoglobin A1c Screening in Elective Total Joint Arthroplasty Patients. J. Arthroplast. 2017, 32, 304–308. [Google Scholar] [CrossRef]
- Honkanen, M.; Jämsen, E.; Karppelin, M.; Huttunen, R.; Huhtala, H.; Eskelinen, A.; Syrjänen, J. The Impact of Preoperative Bacteriuria on the Risk of Periprosthetic Joint Infection after Primary Knee or Hip Replacement: A Retrospective Study with a 1-Year Follow Up. Clin. Microbiol. Infect. 2018, 24, 376–380. [Google Scholar] [CrossRef] [PubMed]
- Pancio, S.I.; Sousa, P.L.; Krych, A.J.; Abdel, M.P.; Levy, B.A.; Dahm, D.L.; Stuart, M.J. Increased Risk of Revision, Reoperation, and Implant Constraint in TKA After Multiligament Knee Surgery. Clin. Orthop. Relat. Res. 2017, 475, 1618–1626. [Google Scholar] [CrossRef] [PubMed]
- Fenske, F.; Krause, L.; Meyer, S.; Kujat, B.; Repmann, J.; Neuhaus, M.; Zimmerer, R.; Roth, A.; Lethaus, B.; Ziebolz, D.; et al. Oral Health Screening for Risk Reduction for Early Periprosthetic Joint Infections of Hip and Knee Endoprostheses—Results of a Prospective Cohort Study. JCM 2023, 12, 4451. [Google Scholar] [CrossRef]
- Antonelli, B.; Chen, A.F. Reducing the Risk of Infection after Total Joint Arthroplasty: Preoperative Optimization. Arthroplasty 2019, 1, 4. [Google Scholar] [CrossRef]
- Tan, T.L.; Rajeswaran, H.; Haddad, S.; Shahi, A.; Parvizi, J. Increased Risk of Periprosthetic Joint Infections in Patients with Hypothyroidism Undergoing Total Joint Arthroplasty. J. Arthroplast. 2016, 31, 868–871. [Google Scholar] [CrossRef] [PubMed]
- Bendich, I.; Takemoto, S.; Patterson, J.T.; Monto, A.; Barber, T.C.; Kuo, A.C. Preoperative Treatment of Hepatitis C Is Associated with Lower Prosthetic Joint Infection Rates in US Veterans. J. Arthroplast. 2019, 34, S319–S326.e1. [Google Scholar] [CrossRef] [PubMed]
- Kheir, M.M.; Kheir, Y.N.P.; Tan, T.L.; Ackerman, C.T.; Rondon, A.J.; Chen, A.F. Increased Complications for Schizophrenia and Bipolar Disorder Patients Undergoing Total Joint Arthroplasty. J. Arthroplast. 2018, 33, 1462–1466. [Google Scholar] [CrossRef] [PubMed]
- Gold, P.A.; Garbarino, L.J.; Anis, H.K.; Neufeld, E.V.; Sodhi, N.; Danoff, J.R.; Boraiah, S.; Rasquinha, V.J.; Mont, M.A. The Cumulative Effect of Substance Abuse Disorders and Depression on Postoperative Complications After Primary Total Knee Arthroplasty. J. Arthroplast. 2020, 35, S151–S157. [Google Scholar] [CrossRef]
- Ledford, C.K.; Kumar, A.R.; Guier, C.G.; Fruth, K.M.; Pagnano, M.W.; Berry, D.J.; Abdel, M.P. Does Metabolic Syndrome Impact the Risk of Reoperation, Revision, or Complication After Primary Total Knee Arthroplasty? J. Arthroplast. 2023, 38, 259–265. [Google Scholar] [CrossRef]
- Yang, X.; Li, L.; Ren, X.; Nie, L. Do Preoperative Intra-Articular Injections of Corticosteroids or Hyaluronic Acid Increase the Risk of Infection after Total Knee Arthroplasty? A Meta-Analysis. Bone Jt. Res. 2022, 11, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Albanese, J.; Feltri, P.; Boffa, A.; Werner, B.C.; Traina, F.; Filardo, G. Infection Risk Increases After Total Hip Arthroplasty Within 3 Months Following Intra-Articular Corticosteroid Injection. A Meta-Analysis on Knee and Hip Arthroplasty. J. Arthroplast. 2023, 38, 1184–1193.e2. [Google Scholar] [CrossRef]
- Yang, Z.; Ji, W.; Xia, Y.; Wang, X. Late Summer Is a Risk Factor for Periprosthetic Joint Infection after Total Joint Arthroplasty: A Retrospective Cohort Study. Medicine 2023, 102, e33089. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, T.J.; Tatara, A.M.; Bedair, H.S.; Alpaugh, K.; Melnic, C.M.; Nelson, S.B. Rates and Outcomes of Periprosthetic Joint Infection in Persons Who Inject Drugs. J. Arthroplast. 2023, 38, 152–157. [Google Scholar] [CrossRef]
- Xu, C.; Guo, H.; Qu, P.; Fu, J.; Kuo, F.-C.; Chen, J.-Y. Preoperatively Elevated Serum Inflammatory Markers Increase the Risk of Periprosthetic Joint Infection Following Total Knee Arthroplasty in Patients with Osteoarthritis. Ther. Clin. Risk Manag. 2018, 14, 1719–1724. [Google Scholar] [CrossRef]
- Sax, O.C.; Chen, Z.; Bains, S.S.; Jacobstein, D.A.; Dubin, J.A.; Hameed, D.; Moore, M.C.; Mont, M.A.; Nace, J.; Delanois, R.E. Does Preoperative Statin Exposure Reduce Prosthetic Joint Infections and Revisions Following Total Joint Arthroplasty? Surg. Technol. Int. 2023, 43, sti43/1717. [Google Scholar] [CrossRef]
- Stryker, L.S.; Abdel, M.P.; Morrey, M.E.; Morrow, M.M.; Kor, D.J.; Morrey, B.F. Elevated Postoperative Blood Glucose and Preoperative Hemoglobin A1C Are Associated with Increased Wound Complications Following Total Joint Arthroplasty. J. Bone Jt. Surg. 2013, 95, 808–814. [Google Scholar] [CrossRef] [PubMed]
- Marchant, M.H.; Viens, N.A.; Cook, C.; Vail, T.P.; Bolognesi, M.P. The Impact of Glycemic Control and Diabetes Mellitus on Perioperative Outcomes After Total Joint Arthroplasty: J. Bone Jt. Surg. Am. Vol. 2009, 91, 1621–1629.e1. [Google Scholar] [CrossRef] [PubMed]
- Klonoff, D.C. The Increasing Incidence of Diabetes in the 21st Century. J. Diabetes Sci. Technol. 2009, 3, 1–2. [Google Scholar] [CrossRef]
- Bozic, K.J.; Lau, E.; Kurtz, S.; Ong, K.; Berry, D.J. Patient-Related Risk Factors for Postoperative Mortality and Periprosthetic Joint Infection in Medicare Patients Undergoing TKA. Clin. Orthop. Relat. Res. 2012, 470, 130–137. [Google Scholar] [CrossRef]
- Yang, L.; Sun, Y.; Li, G.; Liu, J. Is Hemoglobin A1c and Perioperative Hyperglycemia Predictive of Periprosthetic Joint Infection Following Total Joint Arthroplasty?: A Systematic Review and Meta-Analysis. Medicine 2017, 96, e8805. [Google Scholar] [CrossRef] [PubMed]
- Tarabichi, M.; Shohat, N.; Kheir, M.M.; Adelani, M.; Brigati, D.; Kearns, S.M.; Patel, P.; Clohisy, J.C.; Higuera, C.A.; Levine, B.R.; et al. Determining the Threshold for HbA1c as a Predictor for Adverse Outcomes After Total Joint Arthroplasty: A Multicenter, Retrospective Study. J. Arthroplast. 2017, 32, S263–S267.e1. [Google Scholar] [CrossRef] [PubMed]
- Mraovic, B.; Suh, D.; Jacovides, C.; Parvizi, J. Perioperative Hyperglycemia and Postoperative Infection after Lower Limb Arthroplasty. J. Diabetes Sci. Technol. 2011, 5, 412–418. [Google Scholar] [CrossRef]
- Reátegui, D.; Sanchez-Etayo, G.; Núñez, E.; Tió, M.; Popescu, D.; Núñez, M.; Lozano, L. Perioperative Hyperglycaemia and Incidence of Post-Operative Complications in Patients Undergoing Total Knee Arthroplasty. Knee Surg. Sports Traumatol. Arthrosc. 2015, 23, 2026–2031. [Google Scholar] [CrossRef]
- Kheir, M.M.; Tan, T.L.; Kheir, M.; Maltenfort, M.G.; Chen, A.F. Postoperative Blood Glucose Levels Predict Infection After Total Joint Arthroplasty. J. Bone Jt. Surg. 2018, 100, 1423–1431. [Google Scholar] [CrossRef]
- Varady, N.H.; Schwab, P.-E.; Jones, T.; Collins, J.E.; Fitz, W.; Chen, A.F. Optimal Timing of Glucose Measurements After Total Joint Arthroplasty. J. Arthroplast. 2019, 34, S152–S158. [Google Scholar] [CrossRef] [PubMed]
- Shohat, N.; Muhsen, K.; Gilat, R.; Rondon, A.J.; Chen, A.F.; Parvizi, J. Inadequate Glycemic Control Is Associated with Increased Surgical Site Infection in Total Joint Arthroplasty: A Systematic Review and Meta-Analysis. J. Arthroplast. 2018, 33, 2312–2321.e3. [Google Scholar] [CrossRef]
- Shohat, N.; Tarabichi, M.; Tan, T.L.; Goswami, K.; Kheir, M.; Malkani, A.L.; Shah, R.P.; Schwarzkopf, R.; Parvizi, J. 2019 John Insall Award: Fructosamine Is a Better Glycaemic Marker Compared with Glycated Haemoglobin (HbA1C) in Predicting Adverse Outcomes Following Total Knee Arthroplasty: A Prospective Multicentre Study. Bone Jt. J. 2019, 101-B, 3–9. [Google Scholar] [CrossRef]
- Mayne, A.I.W.; Davies, P.S.E.; Simpson, J.M. Antibiotic Treatment of Asymptomatic Bacteriuria Prior to Hip and Knee Arthroplasty; a Systematic Review of the Literature. Surgeon 2018, 16, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Barrere, S.; Reina, N.; Peters, O.A.; Rapp, L.; Vergnes, J.-N.; Maret, D. Dental Assessment Prior to Orthopedic Surgery: A Systematic Review. Orthop. Traumatol. Surg. Res. 2019, 105, 761–772. [Google Scholar] [CrossRef]
- Young, H.; Hirsh, J.; Hammerberg, E.M.; Price, C.S. Dental Disease and Periprosthetic Joint Infection. J. Bone Jt. Surg. 2014, 96, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Kwan, S.A.; Lau, V.; Fliegel, B.E.; Baker, C.; Courtney, P.M.; Deirmengian, G.K. Routine Preoperative Dental Clearance for Total Joint Arthroplasty: Is There a Benefit? Cureus 2023, 15, e41352. [Google Scholar] [CrossRef]
- Jeong Bae, K.; Chae, Y.J.; Jae Jung, S.; Sik Gong, H. Incidence and Risk Factors for Periprosthetic Joint Infection: A Common Data Model Analysis. Jt. Dis. Relat. Surg. 2022, 33, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Tella, G.F.; Donadono, C.; Castagnini, F.; Bordini, B.; Cosentino, M.; Di Liddo, M.; Traina, F. Preoperative and Postoperative Risk Factors for Periprosthetic Joint Infection in Primary Total Hip Arthroplasty: A 1-Year Experience. World J. Orthop. 2022, 13, 903–910. [Google Scholar] [CrossRef]
- Seok, H.-G.; Park, J.-J.; Park, S. Risk Factors for Periprosthetic Joint Infection after Shoulder Arthroplasty: Systematic Review and Meta-Analysis. JCM 2022, 11, 4245. [Google Scholar] [CrossRef] [PubMed]
- Gottwald-Hostalek, U.; Schulte, B. Low Awareness and Under-Diagnosis of Hypothyroidism. Curr. Med. Res. Opin. 2022, 38, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Buller, L.T.; Rosas, S.; Sabeh, K.G.; Roche, M.W.; McLawhorn, A.S.; Barsoum, W.K. Hypothyroidism Increases 90-Day Complications and Costs Following Primary Total Knee Arthroplasty. J. Arthroplast. 2018, 33, 1003–1007. [Google Scholar] [CrossRef] [PubMed]
- Bedair, H.S.; Schurko, B.M.; Dwyer, M.K.; Novikov, D.; Anoushiravani, A.A.; Schwarzkopf, R. Treatment for Chronic Hepatitis C Prior to Total Hip Arthroplasty Significantly Reduces Periprosthetic Joint Infection. J. Arthroplast. 2019, 34, 132–135. [Google Scholar] [CrossRef]
- Mangano, G.R.A.; Avola, M.; Blatti, C.; Caldaci, A.; Sapienza, M.; Chiaramonte, R.; Vecchio, M.; Pavone, V.; Testa, G. Non-Adherence to Anti-Osteoporosis Medication: Factors Influencing and Strategies to Overcome It. A Narrative Review. JCM 2022, 12, 14. [Google Scholar] [CrossRef]
- Harris, A.B.; Lantieri, M.A.; Agarwal, A.R.; Golladay, G.J.; Thakkar, S.C. Osteoporosis and Total Knee Arthroplasty: Higher 5-Year Implant-Related Complications. J. Arthroplast. 2023, S0883540323010914. [Google Scholar] [CrossRef] [PubMed]
- Vakharia, R.M.; Sabeh, K.G.; Sodhi, N.; Naziri, Q.; Mont, M.A.; Roche, M.W. Implant-Related Complications Among Patients with Opioid Use Disorder Following Primary Total Hip Arthroplasty: A Matched-Control Analysis of 42,097 Medicare Patients. J. Arthroplast. 2020, 35, 178–181. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.A.; Schleck, C.; Harmsen, W.S.; Jacob, A.K.; Warner, D.O.; Lewallen, D.G. Current Tobacco Use Is Associated with Higher Rates of Implant Revision and Deep Infection after Total Hip or Knee Arthroplasty: A Prospective Cohort Study. BMC Med. 2015, 13, 283. [Google Scholar] [CrossRef]
- Oster, B.; Hameed, D.; Bains, S.S.; Delanois, R.E.; Johnson, A.J.; Nace, J.; Mont, M.A. Tobacco and Cannabis Use Have a Synergistic Association on Infection Risk Following Total Knee Arthroplasty. J. Arthroplast. 2023, 38, 2137–2141. [Google Scholar] [CrossRef] [PubMed]
- Chung, B.C.; Bouz, G.J.; Mayfield, C.K.; Nakata, H.; Christ, A.B.; Oakes, D.A.; Lieberman, J.R.; Heckmann, N.D. Dose-Dependent Early Postoperative Opioid Use Is Associated with Periprosthetic Joint Infection and Other Complications in Primary TJA. J. Bone Jt. Surg. 2021, 103, 1531–1542. [Google Scholar] [CrossRef]
- Kenanidis, E.; Kakoulidis, P.; Karponis, D.; Tsiridis, E. The Effect of Perioperative Vitamin D Levels on the Functional, Patient-Related Outcome Measures and the Risk of Infection Following Hip and Knee Arthroplasty: A Systematic Review. PROM 2020, 11, 161–171. [Google Scholar] [CrossRef]
- Traven, S.A.; Chiaramonti, A.M.; Barfield, W.R.; Kirkland, P.A.; Demos, H.A.; Schutte, H.D.; Drew, J.M. Fewer Complications Following Revision Hip and Knee Arthroplasty in Patients with Normal Vitamin D Levels. J. Arthroplast. 2017, 32, S193–S196. [Google Scholar] [CrossRef]
- Arshi, A.; Shieh, A.; Adams, J.S.; Bernthal, N.M.; Zeegen, E.N.; Sassoon, A.A. Preoperative Vitamin D Repletion in Total Knee Arthroplasty: A Cost-Effectiveness Model. J. Arthroplast. 2020, 35, 1379–1383. [Google Scholar] [CrossRef] [PubMed]
- Zajonz, D.; Daikos, A.; Prager, F.; Edel, M.; Möbius, R.; Fakler, J.K.M.; Roth, A.; Ghanem, M. Significance of Nutritional Status in the Development of Periprosthetic Infections: A Retrospective Analysis of 194 Patients. Orthopäde 2021, 50, 188–197. [Google Scholar] [CrossRef]
- Shi, T.; Chen, Z.; Hu, D.; Wu, D.; Wang, Z.; Liu, W. Concomitant Malnutrition and Frailty Are Significant Risk Factors for Poor Outcome Following Two-Stage Revision for Chronic Periprosthetic Joint Infection. J. Orthop. Surg. Res. 2023, 18, 804. [Google Scholar] [CrossRef]
- Tsantes, A.G.; Papadopoulos, D.V.; Lytras, T.; Tsantes, A.E.; Mavrogenis, A.F.; Korompilias, A.V.; Gelalis, I.D.; Tsantes, C.G.; Bonovas, S. Association of Malnutrition with Periprosthetic Joint and Surgical Site Infections after Total Joint Arthroplasty: A Systematic Review and Meta-Analysis. J. Hosp. Infect. 2019, 103, 69–77. [Google Scholar] [CrossRef]
- Boutari, C.; Mantzoros, C.S. A 2022 Update on the Epidemiology of Obesity and a Call to Action: As Its Twin COVID-19 Pandemic Appears to Be Receding, the Obesity and Dysmetabolism Pandemic Continues to Rage On. Metabolism 2022, 133, 155217. [Google Scholar] [CrossRef] [PubMed]
- Iannotti, F.; Prati, P.; Fidanza, A.; Iorio, R.; Ferretti, A.; Pèrez Prieto, D.; Kort, N.; Violante, B.; Pipino, G.; Schiavone Panni, A.; et al. Prevention of Periprosthetic Joint Infection (PJI): A Clinical Practice Protocol in High-Risk Patients. TropicalMed 2020, 5, 186. [Google Scholar] [CrossRef] [PubMed]
- Girardi, F.M.; Liu, J.; Guo, Z.; Valle, A.G.D.; MacLean, C.; Memtsoudis, S.G. The Impact of Obesity on Resource Utilization among Patients Undergoing Total Joint Arthroplasty. Int. Orthop. 2019, 43, 269–274. [Google Scholar] [CrossRef]
- Xu, C.; Guo, H.; Wang, Q.; Qu, P.; Bell, K.; Chen, J. Interaction of Obesity with Smoking and Inflammatory Arthropathies Increases the Risk of Periprosthetic Joint Infection: A Propensity Score Matched Study in a Chinese Han Population. J. Hosp. Infect. 2019, 101, 222–228. [Google Scholar] [CrossRef]
- Blackburn, A.Z.; Katakam, A.; Roberts, T.; Humphrey, T.J.; Salimy, M.S.; Egan, C.R.; Melnic, C.M.; Bedair, H.S. Visceral Fat as a Risk Factor for Periprosthetic Joint Infection After Total Hip and Knee Arthroplasty. J. Arthroplast. 2023, 38, 1839–1845.e1. [Google Scholar] [CrossRef]
- Heifner, J.J.; Sakalian, P.A.; Rowland, R.J.; Corces, A. Local Adiposity May Be a More Reliable Predictor for Infection than Body Mass Index Following Total Knee Arthroplasty: A Systematic Review. J. Exp. Ortop. 2023, 10, 110. [Google Scholar] [CrossRef] [PubMed]
- Rechenmacher, A.J.; Yancy, W.S.; Bolognesi, M.P.; Jiranek, W.A.; Seyler, T.M.; Horn, M.E. Does Preoperative Weight Loss Within 6 Months or 1 Year Change the Risk of Adverse Outcomes in Total Knee Arthroplasty by Initial Body Mass Index Classification? J. Arthroplast. 2023, 38, 2517–2522.e2. [Google Scholar] [CrossRef] [PubMed]
- Colen, S.; Hoorntje, A.; Maeckelbergh, L.; Van Diemen, M.; Dalemans, A.; Van Den Bekerom, M.P.J.; Mulier, M. Intra-Articular Hyaluronic Acid Injections Less Than 6 Months Before Total Hip Arthroplasty: Is It Safe? A Retrospective Cohort Study in 565 Patients. J. Arthroplast. 2021, 36, 1003–1008. [Google Scholar] [CrossRef]
- Makris, G.; Wright, J.D.; Ingham, E.; Holland, K.T. The hyaluronate lyase of Staphylococcus aureus—A virulence factor? Microbiology 2004, 150 Pt 6, 2005–2013. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Merchan, E.C.; Delgado-Martinez, A.D. Risk Factors for Periprosthetic Joint Infection after Primary Total Knee Arthroplasty. JCM 2022, 11, 6128. [Google Scholar] [CrossRef] [PubMed]
- Papalia, R.; Vespasiani-Gentilucci, U.; Longo, U.G.; Esposito, C.; Zampogna, B.; Antonelli Incalzi, R.; Denaro, V. Advances in Management of Periprosthetic Joint Infections: An Historical Prospective Study. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 129–138. [Google Scholar] [CrossRef]
- Saracco, M.; Ciriello, V.; D’Angelo, F.; Zagra, L.; Solarino, G.; Logroscino, G. Do Prior Intra-Articular Injections Impact on the Risk of Periprosthetic Joint Infection in Patients Undergoing Total Hip Arthroplasty? A Meta-Analysis of the Current Evidences with a Focus on the Timing of Injection before Surgery. EFORT Open Rev. 2023, 8, 459–467. [Google Scholar] [CrossRef]
- Kim, Y.M.; Joo, Y.B.; Song, J.-H. Preoperative Intra-Articular Steroid Injections within 3 Months Increase the Risk of Periprosthetic Joint Infection in Total Knee Arthroplasty: A Systematic Review and Meta-Analysis. J. Orthop. Surg. Res. 2023, 18, 148. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.S.; Schairer, W.W.; Sculco, T.P.; Sculco, P.K. Comparison of Infection Risk with Corticosteroid or Hyaluronic Acid Injection Prior to Total Knee Arthroplasty. J. Bone Joint Surg. Am. 2019, 101, 112–118. [Google Scholar] [CrossRef]
- Forlenza, E.M.; Burnett, R.A.; Korrapati, A.; Yang, J.; Forsythe, B.; Della Valle, C.J. Preoperative Corticosteroid Injections Demonstrate a Temporal and Dose-Dependent Relationship with the Rate of Postoperative Infection Following Total Hip Arthroplasty. J. Arthroplast. 2021, 36, 2033–2037.e1. [Google Scholar] [CrossRef] [PubMed]
- Kokubun, B.A.; Manista, G.C.; Courtney, P.M.; Kearns, S.M.; Levine, B.R. Intra-Articular Knee Injections Before Total Knee Arthroplasty: Outcomes and Complication Rates. J. Arthroplast. 2017, 32, 1798–1802. [Google Scholar] [CrossRef]
- Muffly, B.; Ayeni, A.; Jones, C.; Heo, K.; Guild, G.; Premkumar, A. Periprosthetic Joint Infection Risk after Primary Total Knee Arthroplasty: Are All Preoperative Corticosteroid Injections the Same? J. Arthroplast. 2023, S0883540323010999. [Google Scholar] [CrossRef]
- Ren, X.; Ling, L.; Qi, L.; Liu, Z.; Zhang, W.; Yang, Z.; Wang, W.; Tu, C.; Li, Z. Patients’ Risk Factors for Periprosthetic Joint Infection in Primary Total Hip Arthroplasty: A Meta-Analysis of 40 Studies. BMC Musculoskelet. Disord. 2021, 22, 776. [Google Scholar] [CrossRef]
- Schiffner, E.; Latz, D.; Karbowski, A.; Grassmann, J.P.; Thelen, S.; Gehrmann, S.; Windolf, J.; Schneppendahl, J.; Jungbluth, P. Possible Risk Factors for Acute and Chronic Deep Periprosthetic Joint Infections in Primary Total Knee Arthroplasty. Do BMI, Smoking, Urinary Tract Infections, Gender, and ASA Classification Have an Impact? J. Orthop. 2020, 19, 111–113. [Google Scholar] [CrossRef]
- Bruin, M.M.; Deijkers, R.L.M.; Bus, M.P.A.; Van Elzakker, E.P.M.; Bazuin, R.; Nelissen, R.G.; Pijls, B.G. Inhaled Corticosteroids, Vitamin K Antagonists and Amlodipine Were Associated with an Increased Risk of Acute Periprosthetic Joint Infection in Patients with Total Hip Arthroplasty: A Retrospective Case–Cohort Study. JCM 2022, 11, 1842. [Google Scholar] [CrossRef] [PubMed]
- Parvizi, J.; Ghanem, E.; Joshi, A.; Sharkey, P.F.; Hozack, W.J.; Rothman, R.H. Does “Excessive” Anticoagulation Predispose to Periprosthetic Infection? J. Arthroplast. 2007, 22, 24–28. [Google Scholar] [CrossRef]
- Mazumdar, K.; Asok Kumar, K.; Dutta, N.K. Potential Role of the Cardiovascular Non-Antibiotic (Helper Compound) Amlodipine in the Treatment of Microbial Infections: Scope and Hope for the Future. Int. J. Antimicrob. Agents 2010, 36, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z. Evaluation of Amlodipine Inhibition and Antimicrobial Effects. IJPC 2019, 5, 12. [Google Scholar] [CrossRef]
- Godoy, G.; Sumarriva, G.; Ochsner, J.L.; Chimento, G.; Schmucker, D.; Dasa, V.; Meyer, M. Preoperative Acute Inflammatory Markers as Predictors for Postoperative Complications in Primary Total Knee Arthroplasty. Ochsner J. 2016, 16, 481–485. [Google Scholar]
- Probasco, W.V.; Cefalu, C.; Lee, R.; Lee, D.; Gu, A.; Dasa, V. Prevalence of Idiopathically Elevated ESR and CRP in Patients Undergoing Primary Total Knee Arthroplasty as a Function of Body Mass Index. J. Clin. Orthop. Trauma. 2020, 11, S722–S728. [Google Scholar] [CrossRef]
- Sutton, S.S.; Magagnoli, J.C.; Cummings, T.H.; Hardin, J.W. Statin Exposure and Risk of Prosthetic Joint Infection After Total Knee or Hip Arthroplasty Among U.S. Veterans. J. Arthroplast. 2021, 36, 3584–3588.e1. [Google Scholar] [CrossRef]
- Rohrer, F.; Wendt, M.; Noetzli, H.; Risch, L.; Bodmer, T.; Cottagnoud, P.; Hermann, T.; Limacher, A.; Gahl, B.; Bruegger, J. Preoperative Decolonization and Periprosthetic Joint Infections—A Randomized Controlled Trial with 2-year Follow-up. J. Orthop. Res. 2021, 39, 333–338. [Google Scholar] [CrossRef]
- Dai, W.; Fang, F. Pre-Admission Use of Chlorhexidine-Impregnated Gauze for Skin Preparation Reduces the Incidence of Peri-Prosthetic Joint Infection after Primary Total Knee Arthroplasty: A Prospective Cohort with Retrospective Controls. Surg. Infect. 2022, 23, 717–721. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.S.; Singh, A.M.; Gregori, P.; Horneff, J.G.; Namdari, S.; Lazarus, M.D. Cutibacterium acnes: A threat to shoulder surgery or an orthopedic red herring? J. Shoulder Elb. Surg. 2020, 29, 1920–1927. [Google Scholar] [CrossRef]
| Risk Factors | Prevalence Data | |
|---|---|---|
| Pathologies | Diabetes | 22% of patients undergoing TJA [14] |
| Urinary tract infections | from 3.5% to 36% of patients undergoing TJA [15] | |
| Septic arthritis Dental problems Anemia Hypothyroidism C Hepatitis Osteoporosis Psychiatric disorders | 5.96% of patients undergoing TJA [16] 15% of PJI are caused by dental problems [17] up to 35% of patients undergoing TJA [18] 3.4% of patients develop PJI [19] 14.3% untreated patients, 0% treated patients [20] NA 7.7% of patients undergoing TJA [21] | |
| Substance use | Tobacco and cannabis use Drug abuse | NA 2.38% of patients undergoing TJA [22] |
| Nutritional status | Vitamin D deficiency | NA |
| Malnutrition | NA | |
| Obesity | 10% of prevalence in PJI [23] | |
| Weight loss | NA | |
| Other | Intra-articular injections | from 0.3% to 38.3% according to different studies [24,25] |
| Season | 1.24% late summer vs. 0.36% in winter [26] | |
| Gender and age | NA | |
| Injective drugs | 29.3% of patients undergoing TJA [27] | |
| Preoperatively elevated serum inflammatory markers | 12.5% prevalence of PJI with both high CRP and ESR [28] | |
| Protective factors | ||
| Statins | lower incidence at one year (0.36 vs. 0.39%) TKA lower incidence at 90 days (0.37 vs. 0.45%) and 2 years (2% vs. 2.14%) for THA [29] | |
| Preoperative decolonization | NA | |
| Preadmission skin preparation | NA | |
| Joint | Incidence | Risk Factors |
|---|---|---|
| Hip | 0.5–1% | obesity, malnutrition, hyperglycemia, uncontrolled diabetes mellitus, rheumatoid arthritis, preoperative anemia, cardiovascular disorders, chronic renal failure, smoking, alcohol abuse, and depression |
| Knee | 0.5–2% | male sex, younger age, diabetes mellitus 2, obesity, hypertension, hypoalbuminemia, malnutrition, rheumatoid arthritis, post-traumatic osteoarthritis, preoperative intra-articular injections, previous multiligament knee surgery, previous steroid therapy, tobacco use, procedure type (bilateral), length of stay over 35 days, patellar resurfacing, prolonged operative time, blood transfusions, higher glucose variability in the postoperative phase, congestive heart failure, chronic pulmonary illness, preoperative anemia, depression, renal illness, pulmonary circulation disorders, psychoses, metastatic tumor, peripheral vascular illness, and valvular illness |
| Ankle | 0.2–26.1% | inflammatory arthritis, prior ankle surgery, age < 65 years, BMI < 19, peripheral vascular disease, chronic lung disease, hypothyroidism, low preoperative AOFAS (American Orthopaedic Foot and Ankle Society) hindfoot scores. |
| Shoulder | <1% | male sex, diabetes mellitus, liver disease, alcohol overuse, iron-deficiency anemia, rheumatoid arthritis, operation history, revision arthroplasty, acute trauma, and nonosteoarthritis as a preoperative diagnosis |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lucenti, L.; Testa, G.; Caldaci, A.; Sammartino, F.; Cicio, C.; Ilardo, M.; Sapienza, M.; Pavone, V. Preoperative Risk Factors for Periprosthetic Joint Infection: A Narrative Review of the Literature. Healthcare 2024, 12, 666. https://doi.org/10.3390/healthcare12060666
Lucenti L, Testa G, Caldaci A, Sammartino F, Cicio C, Ilardo M, Sapienza M, Pavone V. Preoperative Risk Factors for Periprosthetic Joint Infection: A Narrative Review of the Literature. Healthcare. 2024; 12(6):666. https://doi.org/10.3390/healthcare12060666
Chicago/Turabian StyleLucenti, Ludovico, Gianluca Testa, Alessia Caldaci, Fabio Sammartino, Calogero Cicio, Martina Ilardo, Marco Sapienza, and Vito Pavone. 2024. "Preoperative Risk Factors for Periprosthetic Joint Infection: A Narrative Review of the Literature" Healthcare 12, no. 6: 666. https://doi.org/10.3390/healthcare12060666
APA StyleLucenti, L., Testa, G., Caldaci, A., Sammartino, F., Cicio, C., Ilardo, M., Sapienza, M., & Pavone, V. (2024). Preoperative Risk Factors for Periprosthetic Joint Infection: A Narrative Review of the Literature. Healthcare, 12(6), 666. https://doi.org/10.3390/healthcare12060666








