Implementation, Feasibility, and Acceptability of MATCH to Prevent Iatrogenic Disability in Hospitalized Older Adults: A Question of Geriatric Care Program?
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Participants
2.2.1. Changes due to the COVID-19 Pandemic
2.2.2. Participants’ Consent
2.2.3. Inclusion and Exclusion Criteria
2.3. MATCH Tool and Intervention
2.4. MATCH Implementation Procedures (Figure 1)
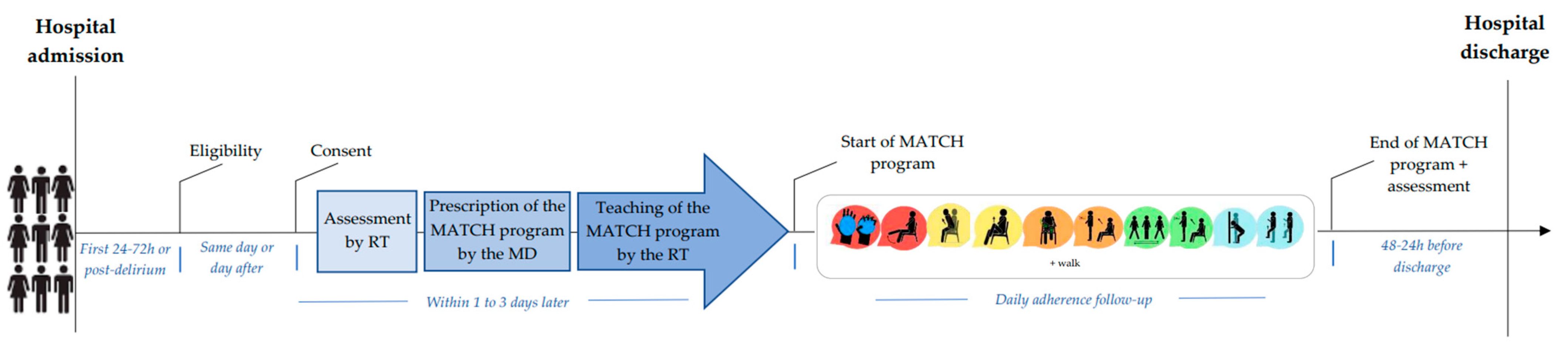
2.5. Measures
- -
- Healthcare team acceptability was assessed by asking them if, according to their clinical judgement, they felt that the program prescribed was appropriate for the patient (“yes” or “no”);
- -
- Patient acceptability was determined before discharge using the System Usability Scale (SUS) questionnaire and the enjoyment Likert scales. The SUS questionnaire included 10 questions with scores between 0 (not satisfied) and 100 (very satisfied) [27]. The score must be >71.4/100 to be considered acceptable [28]. The enjoyment Likert scale included four answers from “strongly disagree” to “strongly agree”.
2.6. Sample Size
2.6.1. Recruitment Timeline
2.6.2. End of the Study
2.7. Statistical Analysis
3. Results
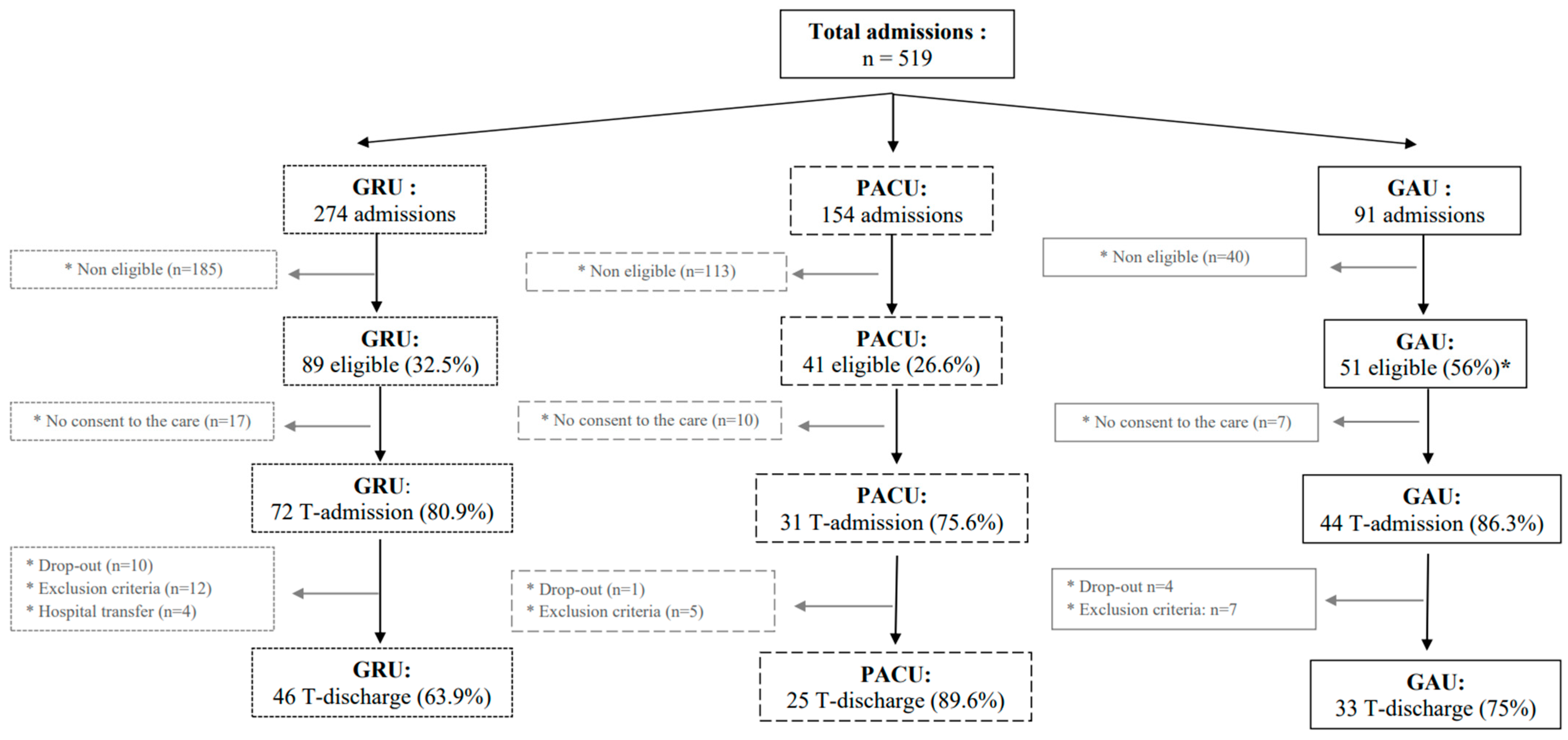
3.1. Participants
3.2. Participant Characteristics
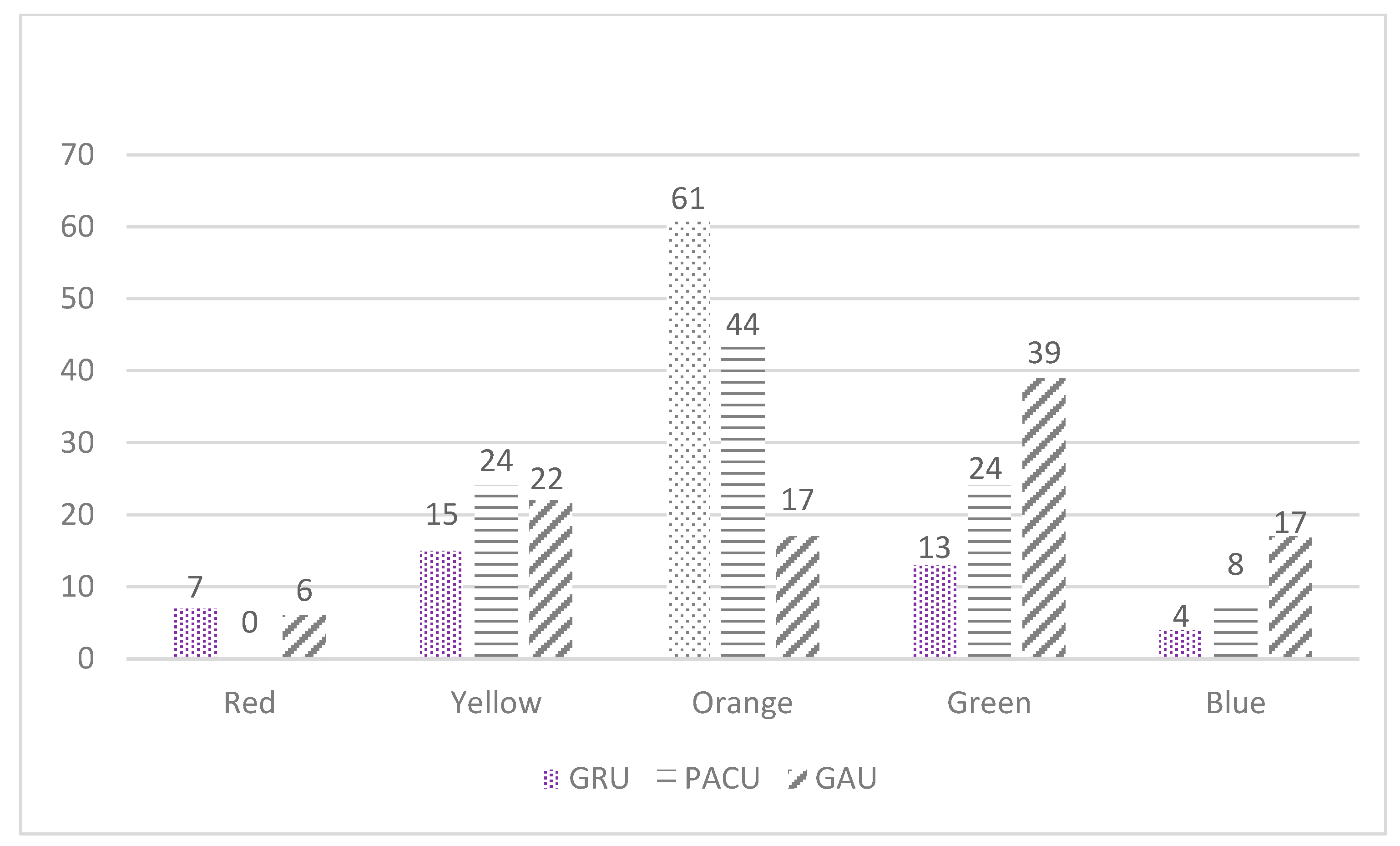
3.3. MATCH Tool Implementation
3.4. Feasibility of MATCH Implementation
3.5. Acceptability of MATCH Implementation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Coker, R.H.; Hays, N.P.; Williams, R.H.; Wolfe, R.R.; Evans, W.J. Bed Rest Promotes Reductions in Walking Speed, Functional Parameters, and Aerobic Fitness in Older, Healthy Adults. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Covinsky, K.E.; Palmer, R.M.; Fortinsky, R.H.; Counsell, S.R.; Stewart, A.L.; Kresevic, D.; Burant, C.J.; Landefeld, C.S. Loss of Independence in Activities of Daily Living in Older Adults Hospitalized with Medical Illnesses: Increased Vulnerability with Age. J. Am. Geriatr. Soc. 2003, 51, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Sourdet, S.; Lafont, C.; Rolland, Y.; Nourhashemi, F.; Andrieu, S.; Vellas, B. Preventable Iatrogenic Disability in Elderly Patients During Hospitalization. J. Am. Med. Dir. Assoc. 2015, 16, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Sherrington, C.; Lord, S.R.; Close, J.C.T.; Barraclough, E.; Taylor, M.; O’Rourke, S.; Kurrle, S.; Tiedemann, A.; Cumming, R.G.; Herbert, R.D. A Simple Tool Predicted Probability of Falling after Aged Care Inpatient Rehabilitation. J. Clin. Epidemiol. 2011, 64, 779–786. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, J.S.; Howrey, B.; Zhang, D.D.; Kuo, Y.-F. Risk of Continued Institutionalization After Hospitalization in Older Adults. J. Gerontol. A Biol. Sci. Med. Sci. 2011, 66, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Fisher, S.R.; Graham, J.E.; Ottenbacher, K.J.; Deer, R.; Ostir, G.V. Inpatient Walking Activity to Predict Readmission in Older Adults. Arch. Phys. Med. Rehabil. 2016, 97, S226–S231. [Google Scholar] [CrossRef]
- Biswas, A.; Oh, P.I.; Faulkner, G.E.; Bajaj, R.R.; Silver, M.A.; Mitchell, M.S.; Alter, D.A. Sedentary Time and Its Association with Risk for Disease Incidence, Mortality, and Hospitalization in Adults: A Systematic Review and Meta-Analysis. Ann. Intern. Med. 2015, 162, 123–132. [Google Scholar] [CrossRef]
- Institut Canadien D’information sur La Santé: Réadmission en Soins de Courte Durée et Retour au Service D’urgence, Toutes Causes Confondues. Ottawa (Ontario, Canada): ICIS. 2012. Available online: https://secure.cihi.ca/freeproducts/Readmissiontoacutecarefr.pdf (accessed on 5 October 2022).
- Fortinsky, R.H.; Covinsky, K.E.; Palmer, R.M.; Landefeld, C.S. Effects of Functional Status Changes before and during Hospitalization on Nursing Home Admission of Older Adults. J. Gerontol. A Biol. Sci. Med. Sci. 1999, 54, M521–M526. [Google Scholar] [CrossRef]
- Hauer, K.; Ullrich, P.; Dutzi, I.; Beurskens, R.; Kern, S.; Bauer, J.; Schwenk, M. Effects of Standardized Home Training in Patients with Cognitive Impairment Following Geriatric Rehabilitation: A Randomized Controlled Pilot Study. Gerontology 2017, 63, 495–506. [Google Scholar] [CrossRef]
- Cortes, O.L.; Delgado, S.; Esparza, M. Systematic Review and Meta-Analysis of Experimental Studies: In-Hospital Mobilization for Patients Admitted for Medical Treatment. J. Adv. Nurs. 2019, 75, 1823–1837. [Google Scholar] [CrossRef]
- Martínez-Velilla, N.; Casas-Herrero, A.; Zambom-Ferraresi, F.; Sáez de Asteasu, M.L.; Lucia, A.; Galbete, A.; García-Baztán, A.; Alonso-Renedo, J.; González-Glaría, B.; Gonzalo-Lázaro, M.; et al. Effect of Exercise Intervention on Functional Decline in Very Elderly Patients During Acute Hospitalization: A Randomized Clinical Trial. JAMA Intern. Med. 2019, 179, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Sáez de Asteasu, M.L.; Martínez-Velilla, N.; Zambom-Ferraresi, F.; Ramírez-Vélez, R.; García-Hermoso, A.; Cadore, E.L.; Casas-Herrero, Á.; Galbete, A.; Izquierdo, M. Changes in Muscle Power after Usual Care or Early Structured Exercise Intervention in Acutely Hospitalized Older Adults. J. Cachexia Sarcopenia Muscle 2020, 11, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Alonso, J.; Bustamante-Ara, N.; Valenzuela, P.L.; Vidán-Astiz, M.; Rodríguez-Romo, G.; Mayordomo-Cava, J.; Javier-González, M.; Hidalgo-Gamarra, M.; Lopéz-Tatis, M.; Valades-Malagón, M.I.; et al. Effect of a Simple Exercise Program on Hospitalization-Associated Disability in Older Patients: A Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2020, 21, 531–537.e1. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, P.L.; Ortiz-Alonso, J.; Bustamante-Ara, N.; Vidán, M.T.; Rodríguez-Romo, G.; Mayordomo-Cava, J.; Javier-González, M.; Hidalgo-Gamarra, M.; López-Tatis, M.; Valadés-Malagón, M.I.; et al. Individual Responsiveness to Physical Exercise Intervention in Acutely Hospitalized Older Adults. J. Clin. Med. 2020, 9, 797. [Google Scholar] [CrossRef]
- Dijkstra, F.; van der Sluis, G.; Jager-Wittenaar, H.; Hempenius, L.; Hobbelen, J.S.M.; Finnema, E. Facilitators and Barriers to Enhancing Physical Activity in Older Patients during Acute Hospital Stay: A Systematic Review. Int. J. Behav. Nutr. Phys. Act. 2022, 19, 99. [Google Scholar] [CrossRef]
- Aubertin-Leheudre, M.; Rolland, Y. The Importance of Physical Activity to Care for Frail Older Adults During the COVID-19 Pandemic. J. Am. Med. Dir. Assoc. 2020, 21, 973–976. [Google Scholar] [CrossRef]
- Peyrusqué, E.; Kergoat, M.-J.; Bolduc, A.; Buckinx, F.; Law, C.; Veillette, N.; Fonseca, R.; Aubertin-Leheudre, M. Maintenance of Autonomy Through ExerCise in Hospital Setting (MATCH): A Feasibility Study. J. Am. Med. Dir. Assoc. 2021, 22, 873–875. [Google Scholar] [CrossRef]
- Laurine, A.; Rolland, Y.; Gerard, S.; Kergoat, M.-J.; Peyrusqué, E.; Aubertin-Leheudre, M. Feasibility and Effect of Implementing Physical Activity Program Implementation During COVID-19 in Hospitalized Positive COVID-19 Older Adults. J. Nutr. Health Aging 2021, 25, 724–726. [Google Scholar] [CrossRef]
- Population Agée de 65 Ans et Plus|Santéscope. Available online: https://www.inspq.qc.ca/santescope/syntheses/population-agee-65-ans-plus (accessed on 5 October 2022).
- Le Commissaire à la Santé et au Bien-Etre (CSBE). Les Urgences au Québec: Évolution de 2003–2004 à 2012–2013; CSBE: Quebec, QC, Canada, 2014.
- Jones, C.J.; Rikli, R.E.; Beam, W.C. A 30-s Chair-Stand Test as a Measure of Lower Body Strength in Community-Residing Older Adults. Res. Q. Exerc. Sport 1999, 70, 113–119. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A Short Physical Performance Battery Assessing Lower Extremity Function: Association with Self-Reported Disability and Prediction of Mortality and Nursing Home Admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef]
- Peters, D.M.; Fritz, S.L.; Krotish, D.E. Assessing the Reliability and Validity of a Shorter Walk Test Compared with the 10-Meter Walk Test for Measurements of Gait Speed in Healthy, Older Adults. J. Geriatr. Phys. Ther. 2001 2013, 36, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-Mental State”: A Practical Method for Grading the Cognitive State of Patients for the Clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Clément, J.P.; Nassif, R.F.; Léger, J.M.; Marchan, F. Development and contribution to the validation of a brief French version of the Yesavage Geriatric Depression Scale. L’Encephale 1997, 23, 91–99. [Google Scholar] [PubMed]
- Gronier, G.; Baudet, A. Psychometric Evaluation of the F-SUS: Creation and Validation of the French Version of the System Usability Scale. Int. J. Hum.–Comput. Interact. 2021, 37, 1571–1582. [Google Scholar] [CrossRef]
- Bangor, A.; Kortum, P.; Miller, J. Determining What Individual SUS Scores Mean: Adding an Adjective Rating Scale. J. Usability Stud. 2009, 4, 114–123. [Google Scholar]
- Lewis, M.; Bromley, K.; Sutton, C.J.; McCray, G.; Myers, H.L.; Lancaster, G.A. Determining Sample Size for Progression Criteria for Pragmatic Pilot RCTs: The Hypothesis Test Strikes Back! Pilot Feasibility Stud. 2021, 7, 40. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; L. Erlbaum Associates: Hillsdale, NJ, USA, 1988; ISBN 978-0-8058-0283-2. [Google Scholar]
- Latour, J.; Lebel, P.; Leclerc, B.-S.; Leduc, N.; Berg, K.; Bolduc, A.; Kergoat, M.-J. Short-Term Geriatric Assessment Units: 30 Years Later. BMC Geriatr. 2010, 10, 41. [Google Scholar] [CrossRef]
- McCullagh, R.; O’Connell, E.; O’Meara, S.; Dahly, D.; O’Reilly, E.; O’Connor, K.; Horgan, N.F.; Timmons, S. Augmented Exercise in Hospital Improves Physical Performance and Reduces Negative Post Hospitalization Events: A Randomized Controlled Trial. BMC Geriatr. 2020, 20, 46. [Google Scholar] [CrossRef]
- Jones, C.T.; Lowe, A.J.; MacGregor, L.; Brand, C.A.; Tweddle, N.; Russell, D.M. A Randomised Controlled Trial of an Exercise Intervention to Reduce Functional Decline and Health Service Utilisation in the Hospitalised Elderly. Australas. J. Ageing 2006, 25, 126–133. [Google Scholar] [CrossRef]
- Dermody, G.; Kovach, C.R. Barriers to Promoting Mobility in Hospitalized Older Adults. Res. Gerontol. Nurs. 2018, 11, 17–27. [Google Scholar] [CrossRef]
- Scheerman, K.; Schoenmakers, A.H.C.; Meskers, C.G.M.; Maier, A.B. Physical, Motivational and Environmental Factors Influencing Physical Activity Promotion during Hospitalization: Older Patients’ Perspective. Geriatr. Nurs. N. Y. N 2021, 42, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Hoyer, E.H.; Brotman, D.J.; Chan, K.S.; Needham, D.M. Barriers to Early Mobility of Hospitalized General Medicine Patients: Survey Development and Results. Am. J. Phys. Med. Rehabil. 2015, 94, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Koenders, N.; van Oorsouw, R.; Seeger, J.P.H.; Nijhuis-van der Sanden, M.W.G.; van de Glind, I.; Hoogeboom, T.J. “I’m Not Going to Walk, Just for the Sake of Walking…”: A Qualitative, Phenomenological Study on Physical Activity during Hospital Stay. Disabil. Rehabil. 2020, 42, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Boltz, M.; Capezuti, E.; Shabbat, N.; Hall, K. Going Home Better Not Worse: Older Adults’ Views on Physical Function during Hospitalization. Int. J. Nurs. Pract. 2010, 16, 381–388. [Google Scholar] [CrossRef] [PubMed]
- American College of Sports Medicine; Chodzko-Zajko, W.J.; Proctor, D.N.; Fiatarone Singh, M.A.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. American College of Sports Medicine Position Stand. Exercise and Physical Activity for Older Adults. Med. Sci. Sport. Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, C.; van Kessel, G.; Phillips, A.; Johnston, K. Accelerometry Shows Inpatients with Acute Medical or Surgical Conditions Spend Little Time Upright and Are Highly Sedentary: Systematic Review. Phys. Ther. 2017, 97, 1044–1065. [Google Scholar] [CrossRef]
- Pedersen, M.M.; Bodilsen, A.C.; Petersen, J.; Beyer, N.; Andersen, O.; Lawson-Smith, L.; Kehlet, H.; Bandholm, T. Twenty-Four-Hour Mobility during Acute Hospitalization in Older Medical Patients. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 331–337. [Google Scholar] [CrossRef]
- Juneau, A.; Bolduc, A.; Nguyen, P.; Leclerc, B.-S.; Rousseau, J.; Dubé, F.; Ringuet, M.-È.; Kergoat, M.-J. Feasibility of Implementing an Exercise Program in a Geriatric Assessment Unit: The SPRINT Program. Can. Geriatr. J. CGJ 2018, 21, 284–289. [Google Scholar] [CrossRef]
- Oesch, P.; Kool, J.; Fernandez-Luque, L.; Brox, E.; Evertsen, G.; Civit, A.; Hilfiker, R.; Bachmann, S. Exergames versus Self-Regulated Exercises with Instruction Leaflets to Improve Adherence during Geriatric Rehabilitation: A Randomized Controlled Trial. BMC Geriatr. 2017, 17, 77. [Google Scholar] [CrossRef]
- Geller, K.; Renneke, K.; Custer, S.; Tigue, G. Intrinsic and Extrinsic Motives Support Adults’ Regular Physical Activity Maintenance. Sport. Med. Int. Open 2018, 2, E62–E66. [Google Scholar] [CrossRef]
| Variables | GRU | PACU | GAU | p-Value (ES) |
|---|---|---|---|---|
| Age (years) [95% CI] | 82.4 ± 7.9 [80.1–84.8] | 83.8 ± 7.2 [80.8–86.8] | 79.5 ± 7.6 [75.8–83.3] | 0.24 † (0.03 a) |
| Women (n; (%)) | 23 (50%) | 15 (60%) | 10 (55.5%) | 0.75 § (0.09 b) |
| Cognitive Status (n; (%)) | 0.34 § (0.2 b) | |||
| Good cognition (Good self-criticism or MMSE ≥ 22) | 38 (82.6%) | 22 (88.0%) | 18 (100%) | |
| Slightly impaired cognition (Slightly impaired self-criticism or MMSE = 18–21) | 7 (15.2%) | 3 (12.0%) | 0 (0%) | |
| Impaired cognition (Impaired self-criticism or MMSE < 18) | 1 (2.2%) | 0 (0%) | 0 (0%) | |
| Geriatric Depression Scale-4 (n; % of depression) | 2 (4.4%) $ | 5 (20.0%) & | 9 (50.0%) $& | <0.001 § (0.45 b) |
| Body mass index (kg/m2) [95% CI] | 25.9 ± 4.6 [24.6–27.3] | 23.5 ± 4.3 [21.8–25.3] | 24.6 ± 5.3 [21.9–27.2] | 0.054 † (0.08 a) |
| Walking speed (m/s) [95% CI] | 0.44 ± 0.16 $ [0.39–0.49] | 0.45 ± 0.18 & [0.37–0.53] | 0.64 ± 0.29 $& [0.50–0.78] | <0.05 † (0.12 a) |
| Diagnosis (n; (%)) | <0.001 § (0.54 b) | |||
| Neurological | 18 (39%) $# | 1 (4%) &# | 1 (5.5%) $& | |
| Traumatology | 4 (9.0%) | 10 (40.0%) | 1 (5.5%) | |
| Deconditioning | 24 (52.0%) | 14 (56.0%) | 16 (89%) | |
| Rehabilitation care time (min/working day) [95% CI] | 33.0 ± 9.0 $# [30.3–35.8] | 26.3 ± 9.7 &# [22.3–30.3] | 12.6 ± 10.1 $& [6.4–18.7] | <0.001 † (0.27 a) |
| Rehabilitation care time (min/working and weekend days) [95% CI] | 24.6 ± 6.8 $# [22.6–26.7] | 19.8 ± 7.4 &# [16.7–22.8] | 9.6 ± 7.6 $& [4.9–14.2] | <0.001 † (0.28 a) |
| Length of stay (working days) [95% CI] | 29.2 ± 12.8 [25.4–33.0] | 28.1 ± 11.1 [23.5–32.7] | 22.9 ± 9.3 [18.3–27.6] | 0.05 † (0.07 a) |
| Length of stay (working and weekend days) [95% CI] | 39.4 ± 17.9 $ [34.1–44.7] | 37.6 ± 15.4 [31.2–43.9] | 30.3 ± 13.3 $ [23.7–36.9] | 0.043 † (0.06 a) |
| Variables | GRU | PACU | GAU | p-Value (ES) |
|---|---|---|---|---|
| Implementation time (weekdays) [95% CI] | 5.9 ± 2.3 [5.78–7.74] | 5.9 ± 3.3 [4.52–8.36] | 4.8 ± 1.2 [3.76–6.02] | 0.22 (0.024) |
| Implementation time (weekdays and weekends) [95% CI] | 6.8 ± 3.0 [5.22–6.60] | 6.4 ± 4.6 [4.5–7.26] | 4.9 ± 2.3 [4.17–5.38] | 0.14 (0.018) |
| % of LOS with MATCH (weekdays) [95% CI] | 76.2 [72.3–80.0] | 78.8 [74.9–82.6] | 77.3 [73.6–81.0] | 0.87 (0.017) |
| % of LOS with MATCH (weekdays and weekends) [95% CI] | 79.4 [75.6–83.1] | 83.1 [79.0–87.2] | 82.8 [78.8–86.9] | 0.55 (−0.020) |
| 0-min | 10 min | 15 min | 20 min | 30 min | |
|---|---|---|---|---|---|
| GRU $ | 4 | 30 | 46 | 9 | 11 |
| PACU & | 0 | 20 | 48 | 16 | 16 |
| GAU &$ | 0 | 17 | 17 | 0 | 66 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peyrusqué, E.; Kergoat, M.-J.; Sirois, M.-J.; Veillette, N.; Fonseca, R.; Aubertin-Leheudre, M. Implementation, Feasibility, and Acceptability of MATCH to Prevent Iatrogenic Disability in Hospitalized Older Adults: A Question of Geriatric Care Program? Healthcare 2023, 11, 1186. https://doi.org/10.3390/healthcare11081186
Peyrusqué E, Kergoat M-J, Sirois M-J, Veillette N, Fonseca R, Aubertin-Leheudre M. Implementation, Feasibility, and Acceptability of MATCH to Prevent Iatrogenic Disability in Hospitalized Older Adults: A Question of Geriatric Care Program? Healthcare. 2023; 11(8):1186. https://doi.org/10.3390/healthcare11081186
Chicago/Turabian StylePeyrusqué, Eva, Marie-Jeanne Kergoat, Marie-Josée Sirois, Nathalie Veillette, Raquel Fonseca, and Mylène Aubertin-Leheudre. 2023. "Implementation, Feasibility, and Acceptability of MATCH to Prevent Iatrogenic Disability in Hospitalized Older Adults: A Question of Geriatric Care Program?" Healthcare 11, no. 8: 1186. https://doi.org/10.3390/healthcare11081186
APA StylePeyrusqué, E., Kergoat, M.-J., Sirois, M.-J., Veillette, N., Fonseca, R., & Aubertin-Leheudre, M. (2023). Implementation, Feasibility, and Acceptability of MATCH to Prevent Iatrogenic Disability in Hospitalized Older Adults: A Question of Geriatric Care Program? Healthcare, 11(8), 1186. https://doi.org/10.3390/healthcare11081186






