Pharmacological Support for the Treatment of Obesity—Present and Future
Abstract
1. Introduction
2. Drugs Registered for Obesity Treatment
2.1. Orlistat
2.2. Bupropion with Naltrexone
2.3. Phentermine with Topiramate
2.4. Lorcaserin
2.5. Liraglutide
2.6. Semaglutide
3. Drugs Registered in Other Disease Entities Demonstrating a Weight-Reducing Effect
3.1. Exenatide
3.2. Dulaglutide
3.3. Tirzepatide
3.4. Glucose-Sodium Cotransporter Inhibitors (SGLT-2i)
4. Drugs in Clinical Trials
4.1. Cotadutide
4.2. Triple GLP-1/Glucagon/GIP Receptor Agonist
4.3. Setmelanotide
4.4. Tesofensine
4.5. Methylphenidate
4.6. Zonisamide with Bupropion
4.7. Cetilistat
4.8. Type 1 Cannabinoid Receptor Antagonist
4.9. Sildenafil
4.10. Oxytocin
4.11. Velneperit
4.12. Amylin Analogues
4.13. Peptide Tyrosine-Tyrosine
4.14. Fibroblast Growth Factor Analogue 21
4.15. Mirabegron
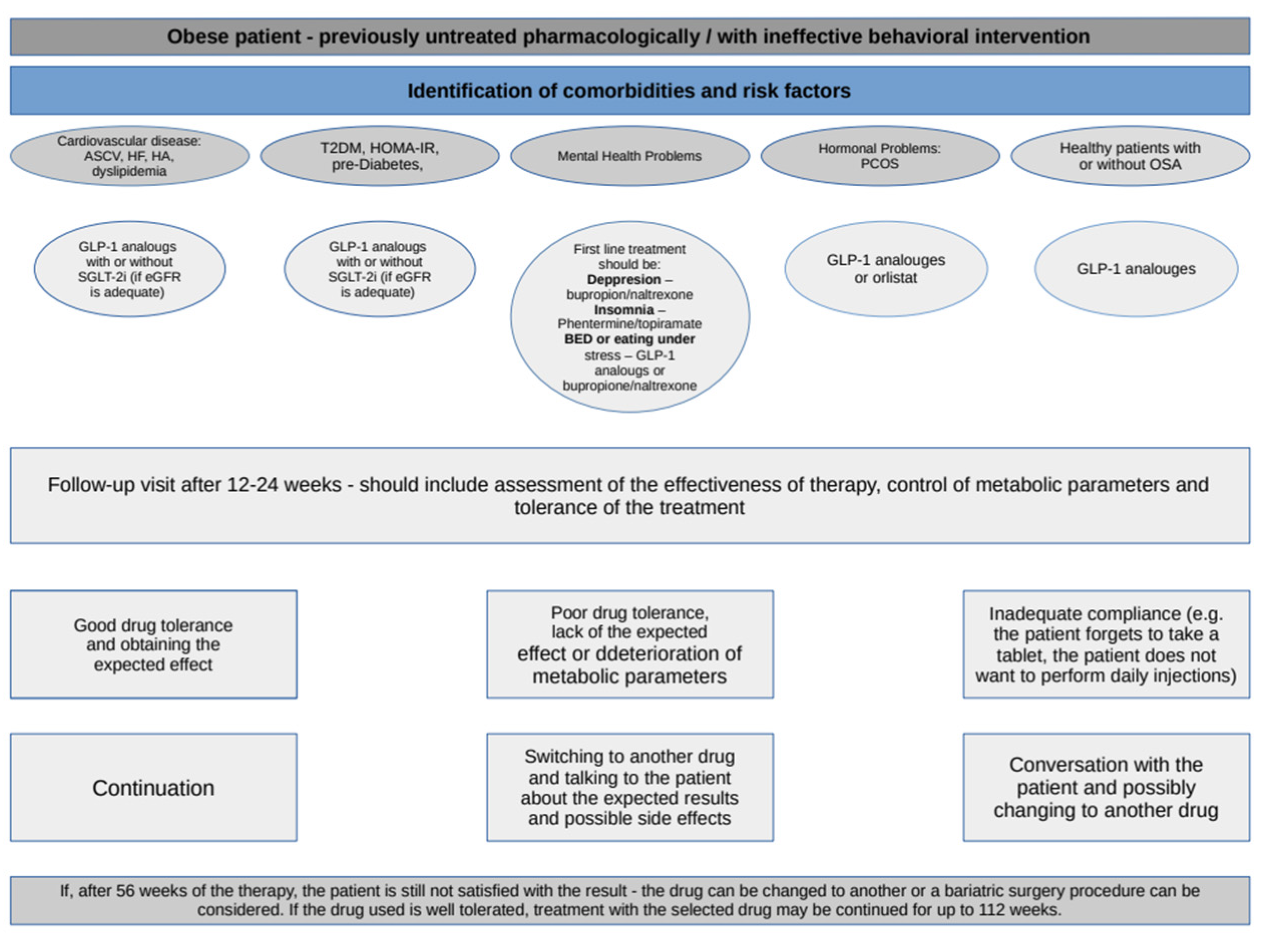
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chooi, Y.C.; Ding, C.; Magkos, F. The Epidemiology of Obesity. Metabolism 2019, 92, 6–10. [Google Scholar] [CrossRef]
- Sørensen, T.I.A.; Martinez, A.R.; Jørgensen, T.S.H. Epidemiology of Obesity. Handb. Exp. Pharmacol. 2022, 274, 3–27. [Google Scholar] [CrossRef]
- Lin, X.; Li, H. Obesity: Epidemiology, Pathophysiology, and Therapeutics. Front. Endocrinol. 2021, 12, 706978. [Google Scholar] [CrossRef]
- Blüher, M. Obesity: Global Epidemiology and Pathogenesis. Nat. Rev. Endocrinol. 2019, 15, 288–298. [Google Scholar] [CrossRef]
- Baio, R.; Napodano, G.; Caruana, C.; Molisso, G.; Di Mauro, U.; Intilla, O.; Pane, U.; D’Angelo, C.; Francavilla, A.B.; Guarnaccia, C.; et al. Association between Obesity and Frequency of High-Grade Prostate Cancer on Biopsy in Men: A Single-Center Retrospective Study. Mol. Clin. Oncol. 2022, 17, 127. [Google Scholar] [CrossRef]
- Bardou, M.; Rouland, A.; Martel, M.; Loffroy, R.; Barkun, A.N.; Chapelle, N. Review Article: Obesity and Colorectal Cancer. Aliment. Pharmacol. Ther. 2022, 56, 407–418. [Google Scholar] [CrossRef]
- Burkbauer, L.; Goldbach, M.; Huang, C.; Lewandowski, J.; Krouse, R.; Allison, K.; Tchou, J. Awareness of Link between Obesity and Breast Cancer Risk Is Associated with Willingness to Participate in Weight Loss Intervention. Breast Cancer Res. Treat. 2022, 194, 541–550. [Google Scholar] [CrossRef]
- Kosmalski, M.; Ziółkowska, S.; Czarny, P.; Szemraj, J.; Pietras, T. The Coexistence of Nonalcoholic Fatty Liver Disease and Type 2 Diabetes Mellitus. J. Clin. Med. 2022, 11, 1375. [Google Scholar] [CrossRef]
- Rhee, E.-J. The Influence of Obesity and Metabolic Health on Vascular Health. Endocrinol. Metab. 2022, 37, 1–8. [Google Scholar] [CrossRef]
- Wen, Y.; Zhang, H.; Tang, Y.; Yan, R. Research on the Association between Obstructive Sleep Apnea Hypopnea Syndrome Complicated with Coronary Heart Disease and Inflammatory Factors, Glycolipid Metabolism, Obesity, and Insulin Resistance. Front. Endocrinol. 2022, 13, 854142. [Google Scholar] [CrossRef]
- Cooper, A.J.; Gupta, S.R.; Moustafa, A.F.; Chao, A.M. Sex/Gender Differences in Obesity Prevalence, Comorbidities, and Treatment. Curr. Obes. Rep. 2021, 10, 458–466. [Google Scholar] [CrossRef]
- Lingvay, I.; Sumithran, P.; Cohen, R.V.; le Roux, C.W. Obesity Management as a Primary Treatment Goal for Type 2 Diabetes: Time to Reframe the Conversation. Lancet Lond. Engl. 2022, 399, 394–405. [Google Scholar] [CrossRef]
- Zeinalian, R.; Ahmadikhatir, S.; Esfahani, E.N.; Namazi, N.; Larijani, B. The Roles of Personalized Nutrition in Obesity and Diabetes Management: A Review. J. Diabetes Metab. Disord. 2022, 21, 1119–1127. [Google Scholar] [CrossRef]
- Wiechert, M.; Holzapfel, C. Nutrition Concepts for the Treatment of Obesity in Adults. Nutrients 2021, 14, 169. [Google Scholar] [CrossRef]
- Chao, A.M.; Quigley, K.M.; Wadden, T.A. Dietary Interventions for Obesity: Clinical and Mechanistic Findings. J. Clin. Investig. 2021, 131, 140065. [Google Scholar] [CrossRef]
- Wadden, T.A.; Crerand, C.E.; Brock, J. Behavioral Treatment of Obesity. Psychiatr. Clin. North Am. 2005, 28, 151–170. [Google Scholar] [CrossRef]
- Chacon, D.; Bernardino, T.; Geraghty, F.; Carrion Rodriguez, A.; Fiani, B.; Chadhaury, A.; Pierre-Louis, M. Bariatric Surgery with Roux-En-Y Gastric Bypass or Sleeve Gastrectomy for Treatment of Obesity and Comorbidities: Current Evidence and Practice. Cureus 2022, 14, e25762. [Google Scholar] [CrossRef]
- Fu, R.; Zhang, Y.; Yu, K.; Mao, D.; Su, H. Bariatric Surgery Alleviates Depression in Obese Patients: A Systematic Review and Meta-Analysis. Obes. Res. Clin. Pract. 2022, 16, 10–16. [Google Scholar] [CrossRef]
- Saad, R.K.; Ghezzawi, M.; Habli, D.; Alami, R.S.; Chakhtoura, M. Fracture Risk Following Bariatric Surgery: A Systematic Review and Meta-Analysis. Osteoporos. Int. J. Establ. Result Coop. Eur. Found. Osteoporos. Natl. Osteoporos. Found. USA 2022, 33, 511–526. [Google Scholar] [CrossRef]
- Sakran, N.; Parmar, C.; Ahmed, S.; Singhal, R.; Madhok, B.; Stier, C.; Chiappetta, S.; Mahawar, K.; Pouwels, S.; Global Bariatric Research Collaborative. Chyloperitoneum and Chylothorax Following Bariatric Surgery: A Systematic Review. Obes. Surg. 2022, 32, 2764–2771. [Google Scholar] [CrossRef]
- Thaher, O.; Croner, R.S.; Hukauf, M.; Driouch, J.; Stroh, C. The Effect of Roux-En-Y Gastric Bypass Compared with Omega-Loop Gastric Bypass on Weight Loss, Perioperative Surgical Events, and Comorbidities. Minerva Surg. 2022, 5. [Google Scholar] [CrossRef]
- Stenberg, E.; dos Reis Falcão, L.F.; O’Kane, M.; Liem, R.; Pournaras, D.J.; Salminen, P.; Urman, R.D.; Wadhwa, A.; Gustafsson, U.O.; Thorell, A. Guidelines for Perioperative Care in Bariatric Surgery: Enhanced Recovery After Surgery (ERAS) Society Recommendations: A 2021 Update. World J. Surg. 2022, 46, 729–751. [Google Scholar] [CrossRef]
- Bansal, A.B.; Al Khalili, Y. Orlistat. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Heck, A.M.; Yanovski, J.A.; Calis, K.A. Orlistat, a New Lipase Inhibitor for the Management of Obesity. Pharmacotherapy 2000, 20, 270–279. [Google Scholar] [CrossRef]
- Sumithran, P.; Proietto, J. Benefit-Risk Assessment of Orlistat in the Treatment of Obesity. Drug Saf. 2014, 37, 597–608. [Google Scholar] [CrossRef]
- Sjöström, L.; Rissanen, A.; Andersen, T.; Boldrin, M.; Golay, A.; Koppeschaar, H.P.; Krempf, M. Randomised Placebo-Controlled Trial of Orlistat for Weight Loss and Prevention of Weight Regain in Obese Patients. Lancet 1998, 352, 167–172. [Google Scholar] [CrossRef]
- Davidson, M.H.; Hauptman, J.; DiGirolamo, M.; Foreyt, J.P.; Halsted, C.H.; Heber, D.; Heimburger, D.C.; Lucas, C.P.; Robbins, D.C.; Chung, J.; et al. Weight Control and Risk Factor Reduction in Obese Subjects Treated for 2 Years with Orlistat: A Randomized Controlled Trial. JAMA 1999, 281, 235. [Google Scholar] [CrossRef]
- Gorgojo-Martínez, J.J.; Basagoiti-Carreño, B.; Sanz-Velasco, A.; Serrano-Moreno, C.; Almodóvar-Ruiz, F. Effectiveness and Tolerability of Orlistat and Liraglutide in Patients with Obesity in a Real-world Setting: The XENSOR Study. Int. J. Clin. Pract. 2019, 73, e13399. [Google Scholar] [CrossRef]
- Wasta Esmail, V.A.; Al-Nimer, M.S.M.; Mohammed, M.O. Effects of Orlistat or Telmisartan on the Serum Free Fatty Acids in Non-Alcoholic Fatty Liver Disease Patients: An Open-Labeled Randomized Controlled Study. Turk. J. Gastroenterol. Off. J. Turk. Soc. Gastroenterol. 2022, 33, 421–426. [Google Scholar] [CrossRef]
- Esmail, V.A.W.; Mohammed, M.O.; Al-Nimer, M.S.M. Short-Term Orlistat Therapy Improves Fatty Infiltration Indices and Liver Fibrosis Scores in Patients with Non-Alcoholic Fatty Liver Disease and Metabolic Syndrome. Arab J. Gastroenterol. Off. Publ. Pan-Arab Assoc. Gastroenterol. 2021, 22, 1–5. [Google Scholar] [CrossRef]
- Gu, M.; Ruan, X.; Li, Y.; Li, T.; Yin, C.; Mueck, A.O. Effect on the Cardiovascular Independent Risk Factor Lipoprotein(a) in Overweight or Obese PCOS Patients with Ethinyl-Estradiol/Drospirenone Alone or plus Orlistat. Gynecol. Endocrinol. Off. J. Int. Soc. Gynecol. Endocrinol. 2022, 38, 598–602. [Google Scholar] [CrossRef]
- Zhao, Y.X.; Wang, L.J.; Gong, F.Y.; Pan, H.; Miao, H.; Duan, L.; Yang, H.B.; Zhu, H.J. Effects of orlistat and metformin on metabolism and gonadal function in overweight or obese patients with polycystic ovary syndrome. Zhonghua Nei Ke Za Zhi 2021, 60, 1165–1168. [Google Scholar] [CrossRef]
- Min, M.; Ruan, X.; Wang, H.; Cheng, J.; Luo, S.; Xu, Z.; Li, M.; Mueck, A.O. Effect of Orlistat during Individualized Comprehensive Life-Style Intervention on Visceral Fat in Overweight or Obese PCOS Patients. Gynecol. Endocrinol. Off. J. Int. Soc. Gynecol. Endocrinol. 2022, 38, 676–680. [Google Scholar] [CrossRef]
- Chen, Z.; Cai, Z. Effects of Oral Contraceptives plus Orlistat in Patients with Polycystic Ovary Syndrome and Overweight/Obesity: A Meta-Analysis. J. Obstet. Gynaecol. Res. 2022, 48, 1399–1408. [Google Scholar] [CrossRef]
- Suleiman, J.B.; Nna, V.U.; Zakaria, Z.; Othman, Z.A.; Bakar, A.B.A.; Mohamed, M. Obesity-Induced Testicular Oxidative Stress, Inflammation and Apoptosis: Protective and Therapeutic Effects of Orlistat. Reprod. Toxicol. Elmsford N 2020, 95, 113–122. [Google Scholar] [CrossRef]
- Othman, Z.A.; Zakaria, Z.; Suleiman, J.B.; Ghazali, W.S.W.; Mohamed, M. Anti-Atherogenic Effects of Orlistat on Obesity-Induced Vascular Oxidative Stress Rat Model. Antioxidants 2021, 10, 251. [Google Scholar] [CrossRef]
- Kadri, S.; El Ayed, M.; Kadri, A.; Limam, F.; Aouani, E.; Mokni, M. Protective Effect of Grape Seed Extract and Orlistat Co-Treatment against Stroke: Effect on Oxidative Stress and Energy Failure. Biomed. Pharmacother. Biomed. Pharmacother. 2021, 136, 111282. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; Segal, K.R.; Hauptman, J.; Lucas, C.P.; Boldrin, M.N.; Rissanen, A.; Wilding, J.P.; Sjöström, L. Effects of Weight Loss with Orlistat on Glucose Tolerance and Progression to Type 2 Diabetes in Obese Adults. Arch. Intern. Med. 2000, 160, 1321–1326. [Google Scholar] [CrossRef]
- Torgerson, J.S.; Hauptman, J.; Boldrin, M.N.; Sjöström, L. XENical in the Prevention of Diabetes in Obese Subjects (XENDOS) Study: A Randomized Study of Orlistat as an Adjunct to Lifestyle Changes for the Prevention of Type 2 Diabetes in Obese Patients. Diabetes Care 2004, 27, 155–161. [Google Scholar] [CrossRef]
- Jarrett, Z.S.; Kou, C.-T.J.; Wan, W.; Colburn, J.A. The Use of Orlistat in an Adult with Lipoprotein Lipase Deficiency: A Case Report. AACE Clin. Case Rep. 2022, 8, 93–95. [Google Scholar] [CrossRef]
- Nascimento, J.; Mariot, C.; Vianna, D.R.B.; Kliemann, L.M.; Chaves, P.S.; Loda, M.; Buffon, A.; Beck, R.C.R.; Pilger, D.A. Fatty Acid Synthase as a Potential New Therapeutic Target for Cervical Cancer. An. Acad. Bras. Cienc. 2022, 94, e20210670. [Google Scholar] [CrossRef]
- Shueng, P.-W.; Chan, H.-W.; Lin, W.-C.; Kuo, D.-Y.; Chuang, H.-Y. Orlistat Resensitizes Sorafenib-Resistance in Hepatocellular Carcinoma Cells through Modulating Metabolism. Int. J. Mol. Sci. 2022, 23, 6501. [Google Scholar] [CrossRef]
- Ardissino, M.; Vincent, M.; Hines, O.; Amin, R.; Eichhorn, C.; Tang, A.R.; Collins, P.; Moussa, O.; Purkayastha, S. Long-Term Cardiovascular Outcomes after Orlistat Therapy in Patients with Obesity: A Nationwide, Propensity-Score Matched Cohort Study. Eur. Heart J. Cardiovasc. Pharmacother. 2022, 8, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, N.N.; Robinson, S.; Kennedy-Martin, T.; Poon, J.L.; Kan, H. Clinical Outcomes Associated with Anti-obesity Medications in Real-world Practice: A Systematic Literature Review. Obes. Rev. 2021, 22, e13326. [Google Scholar] [CrossRef]
- Feighner, J.P. Mechanism of Action of Antidepressant Medications. J. Clin. Psychiatry 1999, 60, 4–11, discussion 12–13. [Google Scholar] [PubMed]
- Mercer, S.L. ACS Chemical Neuroscience Molecule Spotlight on Contrave. ACS Chem. Neurosci. 2011, 2, 484–486. [Google Scholar] [CrossRef]
- Apovian, C.M. Naltrexone/Bupropion for the Treatment of Obesity and Obesity with Type 2 Diabetes. Future Cardiol. 2016, 12, 129–138. [Google Scholar] [CrossRef]
- Jepma, M.; Roy, M.; Ramlakhan, K.; van Velzen, M.; Dahan, A. Different Brain Systems Support Learning from Received and Avoided Pain during Human Pain-Avoidance Learning. eLife 2022, 11, e74149. [Google Scholar] [CrossRef]
- Soyka, M.; Rösner, S. Opioid Antagonists for Pharmacological Treatment of Alcohol Dependence—A Critical Review. Curr. Drug Abuse Rev. 2008, 1, 280–291. [Google Scholar] [CrossRef]
- Skolnick, P. Treatment of Overdose in the Synthetic Opioid Era. Pharmacol. Ther. 2022, 233, 108019. [Google Scholar] [CrossRef]
- Roberto da Silva, G.; Carneiro, M.G.; Barbosa, M.P.; Costa, J.d.A.; de Souza, I.A.; Dos Santos Oliveira, L.; de Vasconcelos, D.A.A.; do Nascimento, E.; Matos, R.J.B.; Lopes de Souza, S.; et al. Naltrexone/Bupropion Modifies Weight, Food Intake, and Drd2 Gene Expression in Rats. J. Endocrinol. 2022, 253, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Greenway, F.L.; Fujioka, K.; Plodkowski, R.A.; Mudaliar, S.; Guttadauria, M.; Erickson, J.; Kim, D.D.; Dunayevich, E. Effect of Naltrexone plus Bupropion on Weight Loss in Overweight and Obese Adults (COR-I): A Multicentre, Randomised, Double-Blind, Placebo-Controlled, Phase 3 Trial. Lancet 2010, 376, 595–605. [Google Scholar] [CrossRef]
- Apovian, C.M.; Aronne, L.; Rubino, D.; Still, C.; Wyatt, H.; Burns, C.; Kim, D.; Dunayevich, E.; COR-II Study Group. A Randomized, Phase 3 Trial of Naltrexone SR/Bupropion SR on Weight and Obesity-Related Risk Factors (COR-II). Obes. Silver Spring Md 2013, 21, 935–943. [Google Scholar] [CrossRef]
- Wadden, T.A.; Foreyt, J.P.; Foster, G.D.; Hill, J.O.; Klein, S.; O’Neil, P.M.; Perri, M.G.; Pi-Sunyer, F.X.; Rock, C.L.; Erickson, J.S.; et al. Weight Loss with Naltrexone SR/Bupropion SR Combination Therapy as an Adjunct to Behavior Modification: The COR-BMOD Trial. Obesity 2011, 19, 110–120. [Google Scholar] [CrossRef]
- Hollander, P.; Gupta, A.K.; Plodkowski, R.; Greenway, F.; Bays, H.; Burns, C.; Klassen, P.; Fujioka, K.; for the COR-Diabetes Study Group. Effects of Naltrexone Sustained- Release/Bupropion Sustained-Release Combination Therapy on Body Weight and Glycemic Parameters in Overweight and Obese Patients With Type 2 Diabetes. Diabetes Care 2013, 36, 4022–4029. [Google Scholar] [CrossRef]
- Fujioka, K.; Plodkowski, R.; O’Neil, P.M.; Gilder, K.; Walsh, B.; Greenway, F.L. The Relationship between Early Weight Loss and Weight Loss at 1 Year with Naltrexone ER/Bupropion ER Combination Therapy. Int. J. Obes. 2016, 40, 1369–1375. [Google Scholar] [CrossRef]
- Bajaj, H.S.; Burrows, M.; Blavignac, J.; Paron, E.; Camacho, F.; Gould, E.; Barakat, M. Extended-release Naltrexone/Bupropion and Liver Health: Pooled, Post Hoc Analysis from Four Randomized Controlled Trials. Diabetes Obes. Metab. 2021, 23, 861–865. [Google Scholar] [CrossRef]
- Carbone, E.A.; Caroleo, M.; Rania, M.; Calabrò, G.; Staltari, F.A.; de Filippis, R.; Aloi, M.; Condoleo, F.; Arturi, F.; Segura-Garcia, C. An Open-Label Trial on the Efficacy and Tolerability of Naltrexone/Bupropion SR for Treating Altered Eating Behaviours and Weight Loss in Binge Eating Disorder. Eat. Weight Disord.-Stud. Anorex. Bulim. Obes. 2021, 26, 779–788. [Google Scholar] [CrossRef]
- Foley, K.F.; DeSanty, K.P.; Kast, R.E. Bupropion: Pharmacology and Therapeutic Applications. Expert Rev. Neurother. 2006, 6, 1249–1265. [Google Scholar] [CrossRef]
- Theriot, J.; Sabir, S.; Azadfard, M. Opioid Antagonists. Pharmacol. Rev. 2019, 19, 463–521. [Google Scholar]
- Woloshin, S.; Schwartz, L.M. The New Weight-Loss Drugs, Lorcaserin and Phentermine-Topiramate: Slim Pickings? JAMA Intern. Med. 2014, 174, 615. [Google Scholar] [CrossRef]
- Allison, D.B.; Gadde, K.M.; Garvey, W.T.; Peterson, C.A.; Schwiers, M.L.; Najarian, T.; Tam, P.Y.; Troupin, B.; Day, W.W. Controlled-Release Phentermine/Topiramate in Severely Obese Adults: A Randomized Controlled Trial (EQUIP). Obes. Silver Spring Md 2012, 20, 330–342. [Google Scholar] [CrossRef]
- Gadde, K.M.; Allison, D.B.; Ryan, D.H.; Peterson, C.A.; Troupin, B.; Schwiers, M.L.; Day, W.W. Effects of Low-Dose, Controlled-Release, Phentermine plus Topiramate Combination on Weight and Associated Comorbidities in Overweight and Obese Adults (CONQUER): A Randomised, Placebo-Controlled, Phase 3 Trial. Lancet 2011, 377, 1341–1352. [Google Scholar] [CrossRef]
- Garvey, W.T.; Ryan, D.H.; Look, M.; Gadde, K.M.; Allison, D.B.; Peterson, C.A.; Schwiers, M.; Day, W.W.; Bowden, C.H. Two-Year Sustained Weight Loss and Metabolic Benefits with Controlled-Release Phentermine/Topiramate in Obese and Overweight Adults (SEQUEL): A Randomized, Placebo-Controlled, Phase 3 Extension Study. Am. J. Clin. Nutr. 2012, 95, 297–308. [Google Scholar] [CrossRef]
- Safer, D.L.; Adler, S.; Dalai, S.S.; Bentley, J.P.; Toyama, H.; Pajarito, S.; Najarian, T. A Randomized, Placebo-controlled Crossover Trial of Phentermine-topiramate ER in Patients with Binge-eating Disorder and Bulimia Nervosa. Int. J. Eat. Disord. 2020, 53, 266–277. [Google Scholar] [CrossRef]
- Grunvald, E.; DeConde, J. Phentermine-Topiramate Extended Release for the Dual Treatment of Obesity and Sleep-Related Eating Disorder: A Case Report. J. Med. Case Rep. 2022, 16, 34. [Google Scholar] [CrossRef]
- Mauer, Y.; Parker, M.; Kashyap, S.R. Antiobesity Drug Therapy: An Individualized and Comprehensive Approach. Cleve. Clin. J. Med. 2021, 88, 440–448. [Google Scholar] [CrossRef]
- Salari, N.; Jafari, S.; Darvishi, N.; Valipour, E.; Mohammadi, M.; Mansouri, K.; Shohaimi, S. The Best Drug Supplement for Obesity Treatment: A Systematic Review and Network Meta-Analysis. Diabetol. Metab. Syndr. 2021, 13, 110. [Google Scholar] [CrossRef]
- Dunican, K.C.; Adams, N.M.; Desilets, A.R. The Role of Pramlintide for Weight Loss. Ann. Pharmacother. 2010, 44, 538–545. [Google Scholar] [CrossRef]
- Davidson, M.H.; Tonstad, S.; Oparil, S.; Schwiers, M.; Day, W.W.; Bowden, C.H. Changes in Cardiovascular Risk Associated with Phentermine and Topiramate Extended-Release in Participants with Comorbidities and a Body Mass Index ≥ 27 Kg/m2. Am. J. Cardiol. 2013, 111, 1131–1138. [Google Scholar] [CrossRef]
- Burke, L.K.; Heisler, L.K. 5-Hydroxytryptamine Medications for the Treatment of Obesity. J. Neuroendocrinol. 2015, 27, 389–398. [Google Scholar] [CrossRef]
- Valencia-Torres, L.; Olarte-Sánchez, C.M.; Lyons, D.J.; Georgescu, T.; Greenwald-Yarnell, M.; Myers, M.G.; Bradshaw, C.M.; Heisler, L.K. Activation of Ventral Tegmental Area 5-HT2C Receptors Reduces Incentive Motivation. Neuropsychopharmacology 2017, 42, 1511–1521. [Google Scholar] [CrossRef] [PubMed]
- Fidler, M.C.; Sanchez, M.; Raether, B.; Weissman, N.J.; Smith, S.R.; Shanahan, W.R.; Anderson, C.M.; BLOSSOM Clinical Trial Group. A One-Year Randomized Trial of Lorcaserin for Weight Loss in Obese and Overweight Adults: The BLOSSOM Trial. J. Clin. Endocrinol. Metab. 2011, 96, 3067–3077. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, P.M.; Smith, S.R.; Weissman, N.J.; Fidler, M.C.; Sanchez, M.; Zhang, J.; Raether, B.; Anderson, C.M.; Shanahan, W.R. Randomized Placebo-Controlled Clinical Trial of Lorcaserin for Weight Loss in Type 2 Diabetes Mellitus: The BLOOM-DM Study. Obes. Silver Spring Md 2012, 20, 1426–1436. [Google Scholar] [CrossRef] [PubMed]
- Bohula, E.A.; Scirica, B.M.; Inzucchi, S.E.; McGuire, D.K.; Keech, A.C.; Smith, S.R.; Kanevsky, E.; Murphy, S.A.; Leiter, L.A.; Dwyer, J.P.; et al. Effect of Lorcaserin on Prevention and Remission of Type 2 Diabetes in Overweight and Obese Patients (CAMELLIA-TIMI 61): A Randomised, Placebo-Controlled Trial. Lancet Lond. Engl. 2018, 392, 2269–2279. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.R.; Weissman, N.J.; Anderson, C.M.; Sanchez, M.; Chuang, E.; Stubbe, S.; Bays, H.; Shanahan, W.R.; Behavioral Modification and Lorcaserin for Overweight and Obesity Management (BLOOM) Study Group. Multicenter, Placebo-Controlled Trial of Lorcaserin for Weight Management. N. Engl. J. Med. 2010, 363, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Singh, R. Efficacy and Safety of Lorcaserin in Obesity: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Expert Rev. Clin. Pharmacol. 2020, 13, 183–190. [Google Scholar] [CrossRef]
- Shukla, A.P.; Kumar, R.B.; Aronne, L.J. Lorcaserin Hcl for the Treatment of Obesity. Expert Opin. Pharmacother. 2015, 16, 2531–2538. [Google Scholar] [CrossRef]
- Fujioka, K. Safety and Tolerability of Medications Approved for Chronic Weight Management. Obes. Silver Spring Md 2015, 23, S7–S11. [Google Scholar] [CrossRef]
- Kelly, E.M.; Tungol, A.A.; Wesolowicz, L.A. Formulary Management of 2 New Agents: Lorcaserin and Phentermine/Topiramate for Weight Loss. J. Manag. Care Pharm. JMCP 2013, 19, 642–654. [Google Scholar] [CrossRef]
- Andreasen, C.R.; Andersen, A.; Knop, F.K.; Vilsbøll, T. How Glucagon-like Peptide 1 Receptor Agonists Work. Endocr. Connect. 2021, 10, R200–R212. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, M.; Wen, Z.; Lu, Z.; Cui, L.; Fu, C.; Xue, H.; Liu, Y.; Zhang, Y. GLP-1 Receptor Agonists: Beyond Their Pancreatic Effects. Front. Endocrinol. 2021, 12, 721135. [Google Scholar] [CrossRef]
- Drucker, D.J. GLP-1 Physiology Informs the Pharmacotherapy of Obesity. Mol. Metab. 2022, 57, 101351. [Google Scholar] [CrossRef]
- Goyal, R.K.; Cristofaro, V.; Sullivan, M.P. Rapid Gastric Emptying in Diabetes Mellitus: Pathophysiology and Clinical Importance. J. Diabetes Complicat. 2019, 33, 107414. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.; Vella, A. Effects of GLP-1 on Appetite and Weight. Rev. Endocr. Metab. Disord. 2014, 15, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Beiroa, D.; Imbernon, M.; Gallego, R.; Senra, A.; Herranz, D.; Villarroya, F.; Serrano, M.; Fernø, J.; Salvador, J.; Escalada, J.; et al. GLP-1 Agonism Stimulates Brown Adipose Tissue Thermogenesis and Browning through Hypothalamic AMPK. Diabetes 2014, 63, 3346–3358. [Google Scholar] [CrossRef] [PubMed]
- Brunton, S.A.; Mosenzon, O.; Wright, E.E. Integrating Oral Semaglutide into Clinical Practice in Primary Care: For Whom, When, and How? Postgrad. Med. 2020, 132, 48–60. [Google Scholar] [CrossRef]
- Nuffer, W.A.; Trujillo, J.M. Liraglutide: A New Option for the Treatment of Obesity. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2015, 35, 926–934. [Google Scholar] [CrossRef]
- Pi-Sunyer, X.; Astrup, A.; Fujioka, K.; Greenway, F.; Halpern, A.; Krempf, M.; Lau, D.C.W.; le Roux, C.W.; Violante Ortiz, R.; Jensen, C.B.; et al. A Randomized, Controlled Trial of 3.0 mg of Liraglutide in Weight Management. N. Engl. J. Med. 2015, 373, 11–22. [Google Scholar] [CrossRef]
- Davies, M.J.; Bergenstal, R.; Bode, B.; Kushner, R.F.; Lewin, A.; Skjøth, T.V.; Andreasen, A.H.; Jensen, C.B.; DeFronzo, R.A.; for the NN8022-1922 Study Group. Efficacy of Liraglutide for Weight Loss among Patients with Type 2 Diabetes: The SCALE Diabetes Randomized Clinical Trial. JAMA 2015, 314, 687. [Google Scholar] [CrossRef] [PubMed]
- Blackman, A.; Foster, G.D.; Zammit, G.; Rosenberg, R.; Aronne, L.; Wadden, T.; Claudius, B.; Jensen, C.B.; Mignot, E. Effect of Liraglutide 3.0 mg in Individuals with Obesity and Moderate or Severe Obstructive Sleep Apnea: The SCALE Sleep Apnea Randomized Clinical Trial. Int. J. Obes. 2016, 40, 1310–1319. [Google Scholar] [CrossRef]
- Wadden, T.A.; Hollander, P.; Klein, S.; Niswender, K.; Woo, V.; Hale, P.M.; Aronne, L. Weight Maintenance and Additional Weight Loss with Liraglutide after Low-Calorie-Diet-Induced Weight Loss: The SCALE Maintenance Randomized Study. Int. J. Obes. 2013, 37, 1443–1451. [Google Scholar] [CrossRef]
- Elkind-Hirsch, K.E.; Chappell, N.; Shaler, D.; Storment, J.; Bellanger, D. Liraglutide 3 mg on Weight, Body Composition, and Hormonal and Metabolic Parameters in Women with Obesity and Polycystic Ovary Syndrome: A Randomized Placebo-Controlled-Phase 3 Study. Fertil. Steril. 2022, 118, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.J.; Wang, D.J.; Song, W.; Shen, S.M.; Ge, W.H. The Effectiveness and Safety of Liraglutide in Treating Overweight/Obese Patients with Polycystic Ovary Syndrome: A Meta-Analysis. J. Endocrinol. Investig. 2022, 45, 261–273. [Google Scholar] [CrossRef] [PubMed]
- Nolan, B.J.; Proietto, J.; Sumithran, P. Intensive Management of Obesity in People with Prader-Willi Syndrome. Endocrine 2022, 77, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Pharmacological Treatment of Nonresponders Following Bariatric Surgery: A Narrative Review of the Current Evidence. Available online: https://europepmc.org/article/med/33792233 (accessed on 1 November 2022).
- Elhag, W.; El Ansari, W. Effectiveness and Safety of Liraglutide in Managing Inadequate Weight Loss and Weight Regain after Primary and Revisional Bariatric Surgery: Anthropometric and Cardiometabolic Outcomes. Obes. Surg. 2022, 32, 1005–1015. [Google Scholar] [CrossRef]
- Muratori, F.; Vignati, F.; Di Sacco, G.; Gavazzi, L.; Pellegrino, D.; Del Prete, M. Efficacy of Liraglutide 3.0 mg Treatment on Weight Loss in Patients with Weight Regain after Bariatric Surgery. Eat. Weight Disord. EWD 2022, 27, 2775–2781. [Google Scholar] [CrossRef]
- Simeone, P.; Tripaldi, R.; Michelsen, A.; Ueland, T.; Liani, R.; Ciotti, S.; Birkeland, K.I.; Gulseth, H.L.; Di Castelnuovo, A.; Cipollone, F.; et al. Effects of Liraglutide vs. Lifestyle Changes on Soluble Suppression of Tumorigenesis-2 (SST2) and Galectin-3 in Obese Subjects with Prediabetes or Type 2 Diabetes after Comparable Weight Loss. Cardiovasc. Diabetol. 2022, 21, 36. [Google Scholar] [CrossRef]
- Elliott, W.; Chan, J. Semaglutide Injection (Wegovy). Intern. Med. Alert 2021, 43. [Google Scholar]
- Wilding, J.P.H.; Batterham, R.L.; Davies, M.; Van Gaal, L.F.; Kandler, K.; Konakli, K.; Lingvay, I.; McGowan, B.M.; Oral, T.K.; Rosenstock, J.; et al. Weight Regain and Cardiometabolic Effects after Withdrawal of Semaglutide: The STEP 1 Trial Extension. Diabetes Obes. Metab. 2022, 24, 1553–1564. [Google Scholar] [CrossRef]
- Davies, M.; Færch, L.; Jeppesen, O.K.; Pakseresht, A.; Pedersen, S.D.; Perreault, L.; Rosenstock, J.; Shimomura, I.; Viljoen, A.; Wadden, T.A.; et al. Semaglutide 2·4 Mg Once a Week in Adults with Overweight or Obesity, and Type 2 Diabetes (STEP 2): A Randomised, Double-Blind, Double-Dummy, Placebo-Controlled, Phase 3 Trial. Lancet Lond. Engl. 2021, 397, 971–984. [Google Scholar] [CrossRef] [PubMed]
- Wadden, T.A.; Bailey, T.S.; Billings, L.K.; Davies, M.; Frias, J.P.; Koroleva, A.; Lingvay, I.; O’Neil, P.M.; Rubino, D.M.; Skovgaard, D.; et al. Effect of Subcutaneous Semaglutide vs. Placebo as an Adjunct to Intensive Behavioral Therapy on Body Weight in Adults with Overweight or Obesity: The STEP 3 Randomized Clinical Trial. JAMA 2021, 325, 1403–1413. [Google Scholar] [CrossRef]
- Rubino, D.; Abrahamsson, N.; Davies, M.; Hesse, D.; Greenway, F.L.; Jensen, C.; Lingvay, I.; Mosenzon, O.; Rosenstock, J.; Rubio, M.A.; et al. Effect of Continued Weekly Subcutaneous Semaglutide vs. Placebo on Weight Loss Maintenance in Adults with Overweight or Obesity: The STEP 4 Randomized Clinical Trial. JAMA 2021, 325, 1414–1425. [Google Scholar] [CrossRef]
- Kadowaki, T.; Isendahl, J.; Khalid, U.; Lee, S.Y.; Nishida, T.; Ogawa, W.; Tobe, K.; Yamauchi, T.; Lim, S.; STEP 6 Investigators. Semaglutide Once a Week in Adults with Overweight or Obesity, with or without Type 2 Diabetes in an East Asian Population (STEP 6): A Randomised, Double-Blind, Double-Dummy, Placebo-Controlled, Phase 3a Trial. Lancet Diabetes Endocrinol. 2022, 10, 193–206. [Google Scholar] [CrossRef]
- Wilding, J.P.H.; Batterham, R.L.; Calanna, S.; Davies, M.; Van Gaal, L.F.; Lingvay, I.; McGowan, B.M.; Rosenstock, J.; Tran, M.T.D.; Wadden, T.A.; et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N. Engl. J. Med. 2021, 384, 989–1002. [Google Scholar] [CrossRef]
- Newsome, P.N.; Buchholtz, K.; Cusi, K.; Linder, M.; Okanoue, T.; Ratziu, V.; Sanyal, A.J.; Sejling, A.-S.; Harrison, S.A. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. N. Engl. J. Med. 2021, 384, 1113–1124. [Google Scholar] [CrossRef]
- Harrison, S.A.; Calanna, S.; Cusi, K.; Linder, M.; Okanoue, T.; Ratziu, V.; Sanyal, A.; Sejling, A.-S.; Newsome, P.N. Semaglutide for the Treatment of Non-Alcoholic Steatohepatitis: Trial Design and Comparison of Non-Invasive Biomarkers. Contemp. Clin. Trials 2020, 97, 106174. [Google Scholar] [CrossRef]
- Goldman, V.E.; Naguib, M.N.; Vidmar, A.P. Anti-Obesity Medication Use in Children and Adolescents with Prader-Willi Syndrome: Case Review and Literature Search. J. Clin. Med. 2021, 10, 4540. [Google Scholar] [CrossRef]
- Sani, E.; Prato, G.D.; Zenti, M.G.; Bordugo, A.; Trombetta, M.; Bonora, E. Effects of Semaglutide on Glycemic Control and Weight Loss in a Patient with Prader-Willi Syndrome: A Case Report. Endocr. Metab. Immune Disord. Drug Targets 2022, 22, 1053–1057. [Google Scholar] [CrossRef]
- Gibbons, C.; Blundell, J.; Tetens Hoff, S.; Dahl, K.; Bauer, R.; Baekdal, T. Effects of Oral Semaglutide on Energy Intake, Food Preference, Appetite, Control of Eating and Body Weight in Subjects with Type 2 Diabetes. Diabetes Obes. Metab. 2021, 23, 581–588. [Google Scholar] [CrossRef]
- Shoemaker, A.H.; Silver, H.J.; Buchowski, M.; Slaughter, J.C.; Yanovski, J.A.; Elfers, C.; Roth, C.L.; Abuzzahab, M.J. Energy Balance in Hypothalamic Obesity in Response to Treatment with a Once-Weekly GLP-1 Receptor Agonist. Int. J. Obes. 2022, 46, 623–629. [Google Scholar] [CrossRef]
- Jabbour, S.A.; Frías, J.P.; Guja, C.; Hardy, E.; Ahmed, A.; Öhman, P. Effects of Exenatide Once Weekly plus Dapagliflozin, Exenatide Once Weekly, or Dapagliflozin, Added to Metformin Monotherapy, on Body Weight, Systolic Blood Pressure, and Triglycerides in Patients with Type 2 Diabetes in the DURATION-8 Study. Diabetes Obes. Metab. 2018, 20, 1515–1519. [Google Scholar] [CrossRef]
- van Ruiten, C.C.; Veltman, D.J.; Nieuwdorp, M.; IJzerman, R.G. Brain Activation in Response to Low-Calorie Food Pictures: An Explorative Analysis of a Randomized Trial with Dapagliflozin and Exenatide. Front. Endocrinol. 2022, 13, 863592. [Google Scholar] [CrossRef]
- Ma, R.-L.; Deng, Y.; Wang, Y.-F.; Zhu, S.-Y.; Ding, X.-S.; Sun, A.-J. Short-Term Combined Treatment with Exenatide and Metformin for Overweight/Obese Women with Polycystic Ovary Syndrome. Chin. Med. J. (Engl.) 2021, 134, 2882–2889. [Google Scholar] [CrossRef]
- Tao, T.; Zhang, Y.; Zhu, Y.-C.; Fu, J.-R.; Wang, Y.-Y.; Cai, J.; Ma, J.-Y.; Xu, Y.; Gao, Y.-N.; Sun, Y.; et al. Exenatide, Metformin, or Both for Prediabetes in PCOS: A Randomized, Open-Label, Parallel-Group Controlled Study. J. Clin. Endocrinol. Metab. 2021, 106, e1420–e1432. [Google Scholar] [CrossRef]
- Rodgers, M.; Migdal, A.L.; Rodríguez, T.G.; Chen, Z.-Z.; Nath, A.K.; Gerszten, R.E.; Kasid, N.; Toschi, E.; Tripaldi, J.; Heineman, B.; et al. Weight Loss Outcomes among Early High Responders to Exenatide Treatment: A Randomized, Placebo Controlled Study in Overweight and Obese Women. Front. Endocrinol. 2021, 12, 742873. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, M.; Zhang, Y.; Tong, N. Efficacy and Safety of Dulaglutide in Patients with Type 2 Diabetes: A Meta-Analysis and Systematic Review. Sci. Rep. 2016, 6, 18904. [Google Scholar] [CrossRef]
- Morieri, M.L.; Frison, V.; Rigato, M.; D’Ambrosio, M.; Tadiotto, F.; Paccagnella, A.; Simioni, N.; Lapolla, A.; Avogaro, A.; Fadini, G.P. Effectiveness of Dulaglutide in the Real World and in Special Populations of Type 2 Diabetic Patients. J. Clin. Endocrinol. Metab. 2020, 105, dgaa204. [Google Scholar] [CrossRef]
- Da Porto, A.; Casarsa, V.; Colussi, G.; Catena, C.; Cavarape, A.; Sechi, L. Dulaglutide Reduces Binge Episodes in Type 2 Diabetic Patients with Binge Eating Disorder: A Pilot Study. Diabetes Metab. Syndr. 2020, 14, 289–292. [Google Scholar] [CrossRef]
- Syed, Y.Y. Tirzepatide: First Approval. Drugs 2022, 82, 1213–1220. [Google Scholar] [CrossRef]
- Jastreboff, A.M.; Aronne, L.J.; Ahmad, N.N.; Wharton, S.; Connery, L.; Alves, B.; Kiyosue, A.; Zhang, S.; Liu, B.; Bunck, M.C.; et al. Tirzepatide Once Weekly for the Treatment of Obesity. N. Engl. J. Med. 2022, 387, 205–216. [Google Scholar] [CrossRef]
- Jung, H.N.; Jung, C.H. The Upcoming Weekly Tides (Semaglutide vs. Tirzepatide) against Obesity: STEP or SURPASS? J. Obes. Metab. Syndr. 2022, 31, 28–36. [Google Scholar] [CrossRef]
- Karagiannis, T.; Avgerinos, I.; Liakos, A.; Del Prato, S.; Matthews, D.R.; Tsapas, A.; Bekiari, E. Management of Type 2 Diabetes with the Dual GIP/GLP-1 Receptor Agonist Tirzepatide: A Systematic Review and Meta-Analysis. Diabetologia 2022, 65, 1251–1261. [Google Scholar] [CrossRef] [PubMed]
- Ludvik, B.; Giorgino, F.; Jódar, E.; Frias, J.P.; Fernández Landó, L.; Brown, K.; Bray, R.; Rodríguez, Á. Once-Weekly Tirzepatide versus Once-Daily Insulin Degludec as Add-on to Metformin with or without SGLT2 Inhibitors in Patients with Type 2 Diabetes (SURPASS-3): A Randomised, Open-Label, Parallel-Group, Phase 3 Trial. Lancet Lond. Engl. 2021, 398, 583–598. [Google Scholar] [CrossRef] [PubMed]
- Mathiesen, D.S.; Bagger, J.I.; Bergmann, N.C.; Lund, A.; Christensen, M.B.; Vilsbøll, T.; Knop, F.K. The Effects of Dual GLP-1/GIP Receptor Agonism on Glucagon Secretion—A Review. Int. J. Mol. Sci. 2019, 20, 4092. [Google Scholar] [CrossRef] [PubMed]
- Samms, R.J.; Coghlan, M.P.; Sloop, K.W. How May GIP Enhance the Therapeutic Efficacy of GLP-1? Trends Endocrinol. Metab. 2020, 31, 410–421. [Google Scholar] [CrossRef]
- Frías, J.P.; Davies, M.J.; Rosenstock, J.; Pérez Manghi, F.C.; Fernández Landó, L.; Bergman, B.K.; Liu, B.; Cui, X.; Brown, K.; SURPASS-2 Investigators. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N. Engl. J. Med. 2021, 385, 503–515. [Google Scholar] [CrossRef]
- Min, T.; Bain, S.C. The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials. Diabetes Ther. Res. Treat. Educ. Diabetes Relat. Disord. 2021, 12, 143–157. [Google Scholar] [CrossRef]
- Kroopnick, J.M.; Davis, S.N. The Role of Recent Pharmacotherapeutic Options on the Management of Treatment Resistant Type 2 Diabetes. Expert Opin. Pharmacother. 2022, 23, 1259–1271. [Google Scholar] [CrossRef]
- LiverTox: Clinical and Research Information on Drug-Induced Liver Injury; National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD, USA, 2012.
- Lupsa, B.C.; Inzucchi, S.E. Use of SGLT2 Inhibitors in Type 2 Diabetes: Weighing the Risks and Benefits. Diabetologia 2018, 61, 2118–2125. [Google Scholar] [CrossRef]
- Butler, J.; Usman, M.S.; Khan, M.S.; Greene, S.J.; Friede, T.; Vaduganathan, M.; Filippatos, G.; Coats, A.J.S.; Anker, S.D. Efficacy and Safety of SGLT2 Inhibitors in Heart Failure: Systematic Review and Meta-analysis. ESC Heart Fail. 2020, 7, 3298–3309. [Google Scholar] [CrossRef]
- Tsapas, A.; Karagiannis, T.; Kakotrichi, P.; Avgerinos, I.; Mantsiou, C.; Tousinas, G.; Manolopoulos, A.; Liakos, A.; Malandris, K.; Matthews, D.R.; et al. Comparative Efficacy of Glucose-Lowering Medications on Body Weight and Blood Pressure in Patients with Type 2 Diabetes: A Systematic Review and Network Meta-Analysis. Diabetes Obes. Metab. 2021, 23, 2116–2124. [Google Scholar] [CrossRef]
- Janež, A.; Fioretto, P. SGLT2 Inhibitors and the Clinical Implications of Associated Weight Loss in Type 2 Diabetes: A Narrative Review. Diabetes Ther. Res. Treat. Educ. Diabetes Relat. Disord. 2021, 12, 2249–2261. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.; Liu, M.; Li, S.; Shi, Q.; Zhang, S.; Zhou, Y.; Su, N. Sodium-Glucose Co-Transporter-2 Inhibitors in Non-Diabetic Adults With Overweight or Obesity: A Systematic Review and Meta-Analysis. Front. Endocrinol. 2021, 12, 706914. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.; Chan, K.Y.; Lo, K. Sodium-Glucose Co-Transporter 2 Inhibitors on Weight Change and Cardiometabolic Profiles in Individuals with Overweight or Obesity and without Diabetes: A Meta-Analysis. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2021, 22, e13336. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.; Heerspink, H.J.L.; Cuthbertson, D.J.; Wilding, J.P.H. SGLT2 Inhibitors and GLP-1 Receptor Agonists: Established and Emerging Indications. Lancet Lond. Engl. 2021, 398, 262–276. [Google Scholar] [CrossRef]
- Arnott, C.; Fletcher, R.A.; Neal, B. Sodium Glucose Cotransporter 2 Inhibitors, Amputation Risk, and Fracture Risk. Heart Fail. Clin. 2022, 18, 645–654. [Google Scholar] [CrossRef]
- Bays, H.E.; Kozlovski, P.; Shao, Q.; Proot, P.; Keefe, D. Licogliflozin, a Novel SGLT1 and 2 Inhibitor: Body Weight Effects in a Randomized Trial in Adults with Overweight or Obesity. Obes. Silver Spring Md 2020, 28, 870–881. [Google Scholar] [CrossRef]
- American Diabetes Association Professional Practice Committee; Draznin, B.; Aroda, V.R.; Bakris, G.; Benson, G.; Brown, F.M.; Freeman, R.; Green, J.; Huang, E.; Isaacs, D.; et al. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes-2022. Diabetes Care 2022, 45, S125–S143. [Google Scholar] [CrossRef]
- Pocai, A.; Carrington, P.E.; Adams, J.R.; Wright, M.; Eiermann, G.; Zhu, L.; Du, X.; Petrov, A.; Lassman, M.E.; Jiang, G.; et al. Glucagon-Like Peptide 1/Glucagon Receptor Dual Agonism Reverses Obesity in Mice. Diabetes 2009, 58, 2258–2266. [Google Scholar] [CrossRef]
- Day, J.W.; Gelfanov, V.; Smiley, D.; Carrington, P.E.; Eiermann, G.; Chicchi, G.; Erion, M.D.; Gidda, J.; Thornberry, N.A.; Tschöp, M.H. Optimization of Co-agonism at GLP-1 and Glucagon Receptors to Safely Maximize Weight Reduction in DIO-rodents. Pept. Sci. 2012, 98, 443–450. [Google Scholar] [CrossRef]
- Elvert, R.; Herling, A.W.; Bossart, M.; Weiss, T.; Zhang, B.; Wenski, P.; Wandschneider, J.; Kleutsch, S.; Butty, U.; Kannt, A. Running on Mixed Fuel-dual Agonistic Approach of GLP-1 and GCG Receptors Leads to Beneficial Impact on Body Weight and Blood Glucose Control: A Comparative Study between Mice and Non-human Primates. Diabetes Obes. Metab. 2018, 20, 1836–1851. [Google Scholar] [CrossRef]
- Ambery, P.; Parker, V.E.; Stumvoll, M.; Posch, M.G.; Heise, T.; Plum-Moerschel, L.; Tsai, L.-F.; Robertson, D.; Jain, M.; Petrone, M. MEDI0382, a GLP-1 and Glucagon Receptor Dual Agonist, in Obese or Overweight Patients with Type 2 Diabetes: A Randomised, Controlled, Double-Blind, Ascending Dose and Phase 2a Study. Lancet 2018, 391, 2607–2618. [Google Scholar] [CrossRef] [PubMed]
- Nahra, R.; Wang, T.; Gadde, K.M.; Oscarsson, J.; Stumvoll, M.; Jermutus, L.; Hirshberg, B.; Ambery, P. Effects of Cotadutide on Metabolic and Hepatic Parameters in Adults With Overweight or Obesity and Type 2 Diabetes: A 54-Week Randomized Phase 2b Study. Diabetes Care 2021, 44, 1433–1442. [Google Scholar] [CrossRef] [PubMed]
- Cegla, J.; Troke, R.C.; Jones, B.; Tharakan, G.; Kenkre, J.; McCullough, K.A.; Lim, C.T.; Parvizi, N.; Hussein, M.; Chambers, E.S.; et al. Coinfusion of Low-Dose GLP-1 and Glucagon in Man Results in a Reduction in Food Intake. Diabetes 2014, 63, 3711–3720. [Google Scholar] [CrossRef]
- Knerr, P.J.; Mowery, S.A.; Douros, J.D.; Premdjee, B.; Hjøllund, K.R.; He, Y.; Kruse Hansen, A.M.; Olsen, A.K.; Perez-Tilve, D.; DiMarchi, R.D.; et al. Next Generation GLP-1/GIP/Glucagon Triple Agonists Normalize Body Weight in Obese Mice. Mol. Metab. 2022, 63, 101533. [Google Scholar] [CrossRef]
- Bossart, M.; Wagner, M.; Elvert, R.; Evers, A.; Hübschle, T.; Kloeckener, T.; Lorenz, K.; Moessinger, C.; Eriksson, O.; Velikyan, I.; et al. Effects on Weight Loss and Glycemic Control with SAR441255, a Potent Unimolecular Peptide GLP-1/GIP/GCG Receptor Triagonist. Cell Metab. 2022, 34, 59–74.e10. [Google Scholar] [CrossRef]
- Kannt, A.; Madsen, A.N.; Kammermeier, C.; Elvert, R.; Klöckener, T.; Bossart, M.; Haack, T.; Evers, A.; Lorenz, K.; Hennerici, W. Incretin Combination Therapy for the Treatment of Non-alcoholic Steatohepatitis. Diabetes Obes. Metab. 2020, 22, 1328–1338. [Google Scholar] [CrossRef]
- Del Prato, S.; Gallwitz, B.; Holst, J.J.; Meier, J.J. The Incretin/Glucagon System as a Target for Pharmacotherapy of Obesity. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2022, 23, e13372. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.H. Setmelanotide: What Does It Mean for Clinical Care of Patients with Obesity? Lancet Diabetes Endocrinol. 2020, 8, 933–935. [Google Scholar] [CrossRef]
- Clément, K.; van den Akker, E.; Argente, J.; Bahm, A.; Chung, W.K.; Connors, H.; De Waele, K.; Farooqi, I.S.; Gonneau-Lejeune, J.; Gordon, G.; et al. Efficacy and Safety of Setmelanotide, an MC4R Agonist, in Individuals with Severe Obesity Due to LEPR or POMC Deficiency: Single-Arm, Open-Label, Multicentre, Phase 3 Trials. Lancet Diabetes Endocrinol. 2020, 8, 960–970. [Google Scholar] [CrossRef]
- Haws, R.; Brady, S.; Davis, E.; Fletty, K.; Yuan, G.; Gordon, G.; Stewart, M.; Yanovski, J. Effect of Setmelanotide, a Melanocortin-4 Receptor Agonist, on Obesity in Bardet-Biedl Syndrome. Diabetes Obes. Metab. 2020, 22, 2133–2140. [Google Scholar] [CrossRef] [PubMed]
- Kühnen, P.; Wabitsch, M.; von Schnurbein, J.; Chirila, C.; Mallya, U.G.; Callahan, P.; Gnanasakthy, A.; Poitou, C.; Krabusch, P.M.; Stewart, M.; et al. Quality of Life Outcomes in Two Phase 3 Trials of Setmelanotide in Patients with Obesity Due to LEPR or POMC Deficiency. Orphanet J. Rare Dis. 2022, 17, 38. [Google Scholar] [CrossRef] [PubMed]
- Markham, A. Setmelanotide: First Approval. Drugs 2021, 81, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J.-A.; Gasteyger, C.; Raben, A.; Meier, D.H.; Astrup, A.; Sjödin, A. The Effect of Tesofensine on Appetite Sensations. Obes. Silver Spring Md 2012, 20, 553–561. [Google Scholar] [CrossRef]
- Appel, L.; Bergström, M.; Buus Lassen, J.; Långström, B. Tesofensine, a Novel Triple Monoamine Re-Uptake Inhibitor with Anti-Obesity Effects: Dopamine Transporter Occupancy as Measured by PET. Eur. Neuropsychopharmacol. J. Eur. Coll. Neuropsychopharmacol. 2014, 24, 251–261. [Google Scholar] [CrossRef]
- Sjödin, A.; Gasteyger, C.; Nielsen, A.-L.; Raben, A.; Mikkelsen, J.D.; Jensen, J.K.S.; Meier, D.; Astrup, A. The Effect of the Triple Monoamine Reuptake Inhibitor Tesofensine on Energy Metabolism and Appetite in Overweight and Moderately Obese Men. Int. J. Obes. 2010, 34, 1634–1643. [Google Scholar] [CrossRef]
- Astrup, A.; Madsbad, S.; Breum, L.; Jensen, T.J.; Kroustrup, J.P.; Larsen, T.M. Effect of Tesofensine on Bodyweight Loss, Body Composition, and Quality of Life in Obese Patients: A Randomised, Double-Blind, Placebo-Controlled Trial. Lancet 2008, 372, 1906–1913. [Google Scholar] [CrossRef]
- Huynh, K.; Klose, M.; Krogsgaard, K.; Drejer, J.; Byberg, S.; Madsbad, S.; Magkos, F.; Aharaz, A.; Edsberg, B.; Tfelt-Hansen, J.; et al. Randomized Controlled Trial of Tesomet for Weight Loss in Hypothalamic Obesity. Eur. J. Endocrinol. 2022, 186, 687–700. [Google Scholar] [CrossRef]
- Turan, S.; Ermiş, Ç.; Pereira-Sanchez, V.; Tunctürk, M.; Pekcanlar, A.A. ADHD and Drug Holidays: Effects on Anthropometric Changes during Methylpenidate Treatment. Psychopharmacol. Bull. 2021, 51, 10–26. [Google Scholar]
- El Amine, F.; Heidinger, B.; Cameron, J.D.; Hafizi, K.; BaniFatemi, S.; Robaey, P.; Vaillancourt, R.; Goldfield, G.S.; Doucet, É. Two-Month Administration of Methylphenidate Improves Olfactory Sensitivity and Suppresses Appetite in Individuals with Obesity. Can. J. Physiol. Pharmacol. 2022, 100, 432–440. [Google Scholar] [CrossRef]
- Kadian, R.; Kumar, A. Zonisamide. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Gadde, K.M.; Yonish, G.M.; Foust, M.S.; Wagner, H.R. Combination Therapy of Zonisamide and Bupropion for Weight Reduction in Obese Women: A Preliminary, Randomized, Open-Label Study. J. Clin. Psychiatry 2007, 68, 1226–1229. [Google Scholar] [CrossRef] [PubMed]
- Gadde, K.M.; Kopping, M.F.; Wagner, H.R.; Yonish, G.M.; Allison, D.B.; Bray, G.A. Zonisamide for Weight Reduction in Obese Adults: A 1-Year Randomized Controlled Trial. Arch. Intern. Med. 2012, 172, 1557–1564. [Google Scholar] [CrossRef] [PubMed]
- Jackson, V.M.; Price, D.A.; Carpino, P.A. Investigational Drugs in Phase II Clinical Trials for the Treatment of Obesity: Implications for Future Development of Novel Therapies. Expert Opin. Investig. Drugs 2014, 23, 1055–1066. [Google Scholar] [CrossRef] [PubMed]
- Kopelman, P.; Groot, G.d.H.; Rissanen, A.; Rossner, S.; Toubro, S.; Palmer, R.; Hallam, R.; Bryson, A.; Hickling, R.I. Weight Loss, HbA1c Reduction, and Tolerability of Cetilistat in a Randomized, Placebo-Controlled Phase 2 Trial in Obese Diabetics: Comparison with Orlistat (Xenical). Obes. Silver Spring Md 2010, 18, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Kopelman, P.; Bryson, A.; Hickling, R.; Rissanen, A.; Rossner, S.; Toubro, S.; Valensi, P. Cetilistat (ATL-962), a Novel Lipase Inhibitor: A 12-Week Randomized, Placebo-Controlled Study of Weight Reduction in Obese Patients. Int. J. Obes. 2007, 31, 494–499. [Google Scholar] [CrossRef]
- Bryson, A.; de la Motte, S.; Dunk, C. Reduction of Dietary Fat Absorption by the Novel Gastrointestinal Lipase Inhibitor Cetilistat in Healthy Volunteers. Br. J. Clin. Pharmacol. 2009, 67, 309–315. [Google Scholar] [CrossRef]
- O’Sullivan, S.E.; Yates, A.S.; Porter, R.K. The Peripheral Cannabinoid Receptor Type 1 (CB1) as a Molecular Target for Modulating Body Weight in Man. Molecules 2021, 26, 6178. [Google Scholar] [CrossRef]
- Christensen, R.; Kristensen, P.K.; Bartels, E.M.; Bliddal, H.; Astrup, A. Efficacy and Safety of the Weight-Loss Drug Rimonabant: A Meta-Analysis of Randomised Trials. Lancet 2007, 370, 1706–1713. [Google Scholar] [CrossRef]
- Christopoulou, F.D.; Kiortsis, D.N. An Overview of the Metabolic Effects of Rimonabant in Randomized Controlled Trials: Potential for Other Cannabinoid 1 Receptor Blockers in Obesity: The Metabolic Effects of Rimonabant. J. Clin. Pharm. Ther. 2011, 36, 10–18. [Google Scholar] [CrossRef]
- Nguyen, T.; Thomas, B.F.; Zhang, Y. Overcoming the Psychiatric Side Effects of the Cannabinoid CB1 Receptor Antagonists: Current Approaches for Therapeutics Development. Curr. Top. Med. Chem. 2019, 19, 1418–1435. [Google Scholar] [CrossRef]
- Paszkiewicz, R.L.; Bergman, R.N.; Santos, R.S.; Frank, A.P.; Woolcott, O.O.; Iyer, M.S.; Stefanovski, D.; Clegg, D.J.; Kabir, M. A Peripheral CB1R Antagonist Increases Lipolysis, Oxygen Consumption Rate, and Markers of Beiging in 3T3-L1 Adipocytes Similar to RIM, Suggesting That Central Effects Can Be Avoided. Int. J. Mol. Sci. 2020, 21, 6639. [Google Scholar] [CrossRef] [PubMed]
- Argueta, D.A.; Perez, P.A.; Makriyannis, A.; DiPatrizio, N.V. Cannabinoid CB1 Receptors Inhibit Gut-Brain Satiation Signaling in Diet-Induced Obesity. Front. Physiol. 2019, 10, 704. [Google Scholar] [CrossRef]
- Knani, I.; Earley, B.J.; Udi, S.; Nemirovski, A.; Hadar, R.; Gammal, A.; Cinar, R.; Hirsch, H.J.; Pollak, Y.; Gross, I.; et al. Targeting the Endocannabinoid/CB1 Receptor System for Treating Obesity in Prader-Willi Syndrome. Mol. Metab. 2016, 5, 1187–1199. [Google Scholar] [CrossRef] [PubMed]
- Fiore, D.; Gianfrilli, D.; Cardarelli, S.; Naro, F.; Lenzi, A.; Isidori, A.M.; Venneri, M.A. Chronic Phosphodiesterase Type 5 Inhibition Has Beneficial Effects on Subcutaneous Adipose Tissue Plasticity in Type 2 Diabetic Mice. J. Cell. Physiol. 2018, 233, 8411–8417. [Google Scholar] [CrossRef] [PubMed]
- Johann, K.; Reis, M.C.; Harder, L.; Herrmann, B.; Gachkar, S.; Mittag, J.; Oelkrug, R. Effects of Sildenafil Treatment on Thermogenesis and Glucose Homeostasis in Diet-Induced Obese Mice. Nutr. Diabetes 2018, 8, 9. [Google Scholar] [CrossRef]
- Leite, S.; Moreira-Costa, L.; Cerqueira, R.; Sousa-Mendes, C.; Angélico-Gonçalves, A.; Fontoura, D.; Vasques-Nóvoa, F.; Leite-Moreira, A.F.; Lourenço, A.P. Chronic Sildenafil Therapy in the ZSF1 Obese Rat Model of Metabolic Syndrome and Heart Failure With Preserved Ejection Fraction. J. Cardiovasc. Pharmacol. Ther. 2021, 26, 690–701. [Google Scholar] [CrossRef]
- Li, S.; Li, Y.; Xiang, L.; Dong, J.; Liu, M.; Xiang, G. Sildenafil Induces Browning of Subcutaneous White Adipose Tissue in Overweight Adults. Metabolism. 2018, 78, 106–117. [Google Scholar] [CrossRef]
- Zemel, M.B.; Kolterman, O.; Rinella, M.; Vuppalanchi, R.; Flores, O.; Barritt, A.S.; Siddiqui, M.; Chalasani, N. Randomized Controlled Trial of a Leucine-Metformin-Sildenafil Combination (NS-0200) on Weight and Metabolic Parameters. Obes. Silver Spring Md 2019, 27, 59–67. [Google Scholar] [CrossRef]
- Palanisamy, A.; Toftlund, S.A.; Giri, T.; Strandberg-Larsen, K.; Lønfeldt, N.N. Birth with Synthetic Oxytocin and the Risk of Being Overweight or Obese during Childhood. Pediatr. Obes. 2022, 17, e12871. [Google Scholar] [CrossRef]
- Lawson, E.A.; Marengi, D.A.; DeSanti, R.L.; Holmes, T.M.; Schoenfeld, D.A.; Tolley, C.J. Oxytocin Reduces Caloric Intake in Men. Obes. Silver Spring Md 2015, 23, 950–956. [Google Scholar] [CrossRef]
- Thienel, M.; Fritsche, A.; Heinrichs, M.; Peter, A.; Ewers, M.; Lehnert, H.; Born, J.; Hallschmid, M. Oxytocin’s Inhibitory Effect on Food Intake Is Stronger in Obese than Normal-Weight Men. Int. J. Obes. 2016, 40, 1707–1714. [Google Scholar] [CrossRef]
- Kerem, L.; Hadjikhani, N.; Holsen, L.; Lawson, E.A.; Plessow, F. Oxytocin Reduces the Functional Connectivity between Brain Regions Involved in Eating Behavior in Men with Overweight and Obesity. Int. J. Obes. 2020, 44, 980–989. [Google Scholar] [CrossRef]
- Plessow, F.; Marengi, D.A.; Perry, S.K.; Lawson, E.A. Oxytocin Administration Increases Proactive Control in Men with Overweight or Obesity: A Randomized, Double-Blind, Placebo-Controlled Crossover Study. Obes. Silver Spring Md 2021, 29, 56–61. [Google Scholar] [CrossRef]
- Espinoza, S.E.; Lee, J.L.; Wang, C.-P.; Ganapathy, V.; MacCarthy, D.; Pascucci, C.; Musi, N.; Volpi, E. Intranasal Oxytocin Improves Lean Muscle Mass and Lowers LDL Cholesterol in Older Adults with Sarcopenic Obesity: A Pilot Randomized Controlled Trial. J. Am. Med. Dir. Assoc. 2021, 22, 1877–1882.e2. [Google Scholar] [CrossRef] [PubMed]
- Burmester, V.; Gibson, E.L.; Butler, G.; Bailey, A.; Terry, P. Oxytocin Reduces Post-Stress Sweet Snack Intake in Women without Attenuating Salivary Cortisol. Physiol. Behav. 2019, 212, 112704. [Google Scholar] [CrossRef]
- Damen, L.; Grootjen, L.N.; Juriaans, A.F.; Donze, S.H.; Huisman, T.M.; Visser, J.A.; Delhanty, P.J.D.; Hokken-Koelega, A.C.S. Oxytocin in Young Children with Prader-Willi Syndrome: Results of a Randomized, Double-Blind, Placebo-Controlled, Crossover Trial Investigating 3 Months of Oxytocin. Clin. Endocrinol. 2021, 94, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Kuppens, R.J.; Donze, S.H.; Hokken-Koelega, A.C.S. Promising Effects of Oxytocin on Social and Food-Related Behaviour in Young Children with Prader-Willi Syndrome: A Randomized, Double-Blind, Controlled Crossover Trial. Clin. Endocrinol. 2016, 85, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.L.; Tamura, R.; Butler, M.G.; Kimonis, V.; Sulsona, C.; Gold, J.-A.; Driscoll, D.J. Oxytocin Treatment in Children with Prader-Willi Syndrome: A Double-Blind, Placebo-Controlled, Crossover Study. Am. J. Med. Genet. A. 2017, 173, 1243–1250. [Google Scholar] [CrossRef]
- Tauber, M.; Boulanouar, K.; Diene, G.; Çabal-Berthoumieu, S.; Ehlinger, V.; Fichaux-Bourin, P.; Molinas, C.; Faye, S.; Valette, M.; Pourrinet, J.; et al. The Use of Oxytocin to Improve Feeding and Social Skills in Infants with Prader-Willi Syndrome. Pediatrics 2017, 139, e20162976. [Google Scholar] [CrossRef]
- Fukasaka, Y.; Nambu, H.; Tanioka, H.; Obata, A.; Tonomura, M.; Okuno, T.; Yukioka, H. An Insurmountable NPY Y5 Receptor Antagonist Exhibits Superior Anti-Obesity Effects in High-Fat Diet-Induced Obese Mice. Neuropeptides 2018, 70, 55–63. [Google Scholar] [CrossRef]
- Powell, A.G.; Apovian, C.M.; Aronne, L.J. New Drug Targets for the Treatment of Obesity. Clin. Pharmacol. Ther. 2011, 90, 40–51. [Google Scholar] [CrossRef] [PubMed]
- George, M.; Rajaram, M.; Shanmugam, E. New and Emerging Drug Molecules Against Obesity. J. Cardiovasc. Pharmacol. Ther. 2014, 19, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Lutz, T.A. Creating the Amylin Story. Appetite 2022, 172, 105965. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kromm, G.H.; Barnhill, O.K.; Sperber, J.; Heuer, L.B.; Loomis, S.; Newman, M.C.; Han, K.; Gulamali, F.F.; Legan, T.B.; et al. A Discrete Parasubthalamic Nucleus Subpopulation Plays a Critical Role in Appetite Suppression. eLife 2022, 11, e75470. [Google Scholar] [CrossRef] [PubMed]
- Kawecki, G.E.; King, K.M.; Cramer, N.A.; Bevan, D.R.; Brown, A.M. Simulations of Cross-Amyloid Aggregation of Amyloid-β and Islet Amyloid Polypeptide Fragments. Biophys. J. 2022, 121, 2002–2013. [Google Scholar] [CrossRef]
- Andreassen, K.V.; Feigh, M.; Hjuler, S.T.; Gydesen, S.; Henriksen, J.E.; Beck-Nielsen, H.; Christiansen, C.; Karsdal, M.A.; Henriksen, K. A Novel Oral Dual Amylin and Calcitonin Receptor Agonist (KBP-042) Exerts Antiobesity and Antidiabetic Effects in Rats. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E24–E33. [Google Scholar] [CrossRef]
- Gydesen, S.; Hjuler, S.T.; Freving, Z.; Andreassen, K.V.; Sonne, N.; Hellgren, L.I.; Karsdal, M.A.; Henriksen, K. A Novel Dual Amylin and Calcitonin Receptor Agonist, KBP-089, Induces Weight Loss through a Reduction in Fat, but Not Lean Mass, While Improving Food Preference: Novel DACRA, KBP-089, Reduces Body Weight and Fat Deposition. Br. J. Pharmacol. 2017, 174, 591–602. [Google Scholar] [CrossRef]
- Andreassen, K.V.; Larsen, A.T.; Sonne, N.; Mohamed, K.E.; Karsdal, M.A.; Henriksen, K. KBP-066A, a Long-Acting Dual Amylin and Calcitonin Receptor Agonist, Induces Weight Loss and Improves Glycemic Control in Obese and Diabetic Rats. Mol. Metab. 2021, 53, 101282. [Google Scholar] [CrossRef]
- Baggio, L.L.; Drucker, D.J. Glucagon-like Peptide-1 Receptor Co-Agonists for Treating Metabolic Disease. Mol. Metab. 2021, 46, 101090. [Google Scholar] [CrossRef]
- Patch, R.J.; Zhang, R.; Edavettal, S.; Macielag, M.J.; Eckardt, A.J.; Li, J.; Rives, M.-L.; Edwards, W.; Hinke, S.A.; Qiu, X.; et al. Design, Synthesis and Preclinical Evaluation of Bio-Conjugated Amylinomimetic Peptides as Long-Acting Amylin Receptor Agonists. Eur. J. Med. Chem. 2022, 236, 114330. [Google Scholar] [CrossRef]
- Smith, S.R.; Aronne, L.J.; Burns, C.M.; Kesty, N.C.; Halseth, A.E.; Weyer, C. Sustained Weight Loss Following 12-Month Pramlintide Treatment as an Adjunct to Lifestyle Intervention in Obesity. Diabetes Care 2008, 31, 1816–1823. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.R.; Blundell, J.E.; Burns, C.; Ellero, C.; Schroeder, B.E.; Kesty, N.C.; Chen, K.S.; Halseth, A.E.; Lush, C.W.; Weyer, C. Pramlintide Treatment Reduces 24-h Caloric Intake and Meal Sizes and Improves Control of Eating in Obese Subjects: A 6-Wk Translational Research Study. Am. J. Physiol.-Endocrinol. Metab. 2007, 293, E620–E627. [Google Scholar] [CrossRef] [PubMed]
- Aronne, L.; Fujioka, K.; Aroda, V.; Chen, K.; Halseth, A.; Kesty, N.C.; Burns, C.; Lush, C.W.; Weyer, C. Progressive Reduction in Body Weight after Treatment with the Amylin Analog Pramlintide in Obese Subjects: A Phase 2, Randomized, Placebo-Controlled, Dose-Escalation Study. J. Clin. Endocrinol. Metab. 2007, 92, 2977–2983. [Google Scholar] [CrossRef] [PubMed]
- Aronne, L.J.; Halseth, A.E.; Burns, C.M.; Miller, S.; Shen, L.Z. Enhanced Weight Loss Following Coadministration of Pramlintide With Sibutramine or Phentermine in a Multicenter Trial. Obesity 2010, 18, 1739–1746. [Google Scholar] [CrossRef]
- Lau, D.C.W.; Erichsen, L.; Francisco, A.M.; Satylganova, A.; le Roux, C.W.; McGowan, B.; Pedersen, S.D.; Pietiläinen, K.H.; Rubino, D.; Batterham, R.L. Once-Weekly Cagrilintide for Weight Management in People with Overweight and Obesity: A Multicentre, Randomised, Double-Blind, Placebo-Controlled and Active-Controlled, Dose-Finding Phase 2 Trial. Lancet Lond. Engl. 2021, 398, 2160–2172. [Google Scholar] [CrossRef]
- Novo Nordisk Successfully Completes AM833 Phase 2 Trial and Phase 1 Combination Trial with AM833 and Semaglutide in Obesity. Available online: https://www.novonordisk.com/content/nncorp/global/en/news-and-media/news-and-ir-materials/news-details.html?id=274 (accessed on 10 October 2022).
- Batterham, R.L.; Cowley, M.A.; Small, C.J.; Herzog, H.; Cohen, M.A.; Dakin, C.L.; Wren, A.M.; Brynes, A.E.; Low, M.J.; Ghatei, M.A.; et al. Gut Hormone PYY(3-36) Physiologically Inhibits Food Intake. Nature 2002, 418, 650–654. [Google Scholar] [CrossRef]
- Moris, J.M.; Heinold, C.; Blades, A.; Koh, Y. Nutrient-Based Appetite Regulation. J. Obes. Metab. Syndr. 2022, 31, 161–168. [Google Scholar] [CrossRef]
- Adrian, T.E.; Savage, A.P.; Sagor, G.R.; Allen, J.M.; Bacarese-Hamilton, A.J.; Tatemoto, K.; Polak, J.M.; Bloom, S.R. Effect of Peptide YY on Gastric, Pancreatic, and Biliary Function in Humans. Gastroenterology 1985, 89, 494–499. [Google Scholar] [CrossRef]
- Koda, S.; Date, Y.; Murakami, N.; Shimbara, T.; Hanada, T.; Toshinai, K.; Niijima, A.; Furuya, M.; Inomata, N.; Osuye, K.; et al. The Role of the Vagal Nerve in Peripheral PYY3–36-Induced Feeding Reduction in Rats. Endocrinology 2005, 146, 2369–2375. [Google Scholar] [CrossRef]
- Lumb, K.J.; DeCarr, L.B.; Milardo, L.F.; Mays, M.R.; Buckholz, T.M.; Fisk, S.E.; Pellegrino, C.M.; Ortiz, A.A.; Mahle, C.D. Novel Selective Neuropeptide Y2 Receptor PEGylated Peptide Agonists Reduce Food Intake and Body Weight in Mice. J. Med. Chem. 2007, 50, 2264–2268. [Google Scholar] [CrossRef]
- Gantz, I.; Erondu, N.; Mallick, M.; Musser, B.; Krishna, R.; Tanaka, W.K.; Snyder, K.; Stevens, C.; Stroh, M.A.; Zhu, H.; et al. Efficacy and Safety of Intranasal Peptide YY3-36 for Weight Reduction in Obese Adults. J. Clin. Endocrinol. Metab. 2007, 92, 1754–1757. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, L.; Liu, Q.; Li, F.; Zhang, L.; Zhu, F.; Shao, T.; Barve, S.; Chen, Y.; Li, X.; et al. Fibroblast Growth Factor 21 Is Required for the Therapeutic Effects of Lactobacillus Rhamnosus GG against Fructose-Induced Fatty Liver in Mice. Mol. Metab. 2019, 29, 145–157. [Google Scholar] [CrossRef]
- Mana, M.F.; Parisi, M.C.R.; Correa-Giannella, M.L.; Neto, A.M.; Yamanaka, A.; Cunha-Silva, M.; Cavaleiro, A.M.; Dos Santos, C.R.; Pavan, C.R.; Sevá-Pereira, T.; et al. Non-Alcoholic Fatty Liver Disease in Long-Term Type 2 Diabetes: Role of Rs738409 PNPLA3 and Rs499765 FGF21 Polymorphisms and Serum Biomarkers. Mol. Basel Switz. 2022, 27, 3193. [Google Scholar] [CrossRef]
- Hui, X.; Feng, T.; Liu, Q.; Gao, Y.; Xu, A. The FGF21–Adiponectin Axis in Controlling Energy and Vascular Homeostasis. J. Mol. Cell Biol. 2016, 8, 110–119. [Google Scholar] [CrossRef]
- Talukdar, S.; Zhou, Y.; Li, D.; Rossulek, M.; Dong, J.; Somayaji, V.; Weng, Y.; Clark, R.; Lanba, A.; Owen, B.M.; et al. A Long-Acting FGF21 Molecule, PF-05231023, Decreases Body Weight and Improves Lipid Profile in Non-Human Primates and Type 2 Diabetic Subjects. Cell Metab. 2016, 23, 427–440. [Google Scholar] [CrossRef]
- Yadav, P.; Khurana, A.; Bhatti, J.S.; Weiskirchen, R.; Navik, U. Glucagon-like Peptide 1 and Fibroblast Growth Factor-21 in Non-Alcoholic Steatohepatitis: An Experimental to Clinical Perspective. Pharmacol. Res. 2022, 184, 106426. [Google Scholar] [CrossRef]
- Baruch, A.; Wong, C.; Chinn, L.W.; Vaze, A.; Sonoda, J.; Gelzleichter, T.; Chen, S.; Lewin-Koh, N.; Morrow, L.; Dheerendra, S.; et al. Antibody-Mediated Activation of the FGFR1/Klothoβ Complex Corrects Metabolic Dysfunction and Alters Food Preference in Obese Humans. Proc. Natl. Acad. Sci. USA 2020, 117, 28992–29000. [Google Scholar] [CrossRef]
- Kaufman, A.; Abuqayyas, L.; Denney, W.S.; Tillman, E.J.; Rolph, T. AKR-001, an Fc-FGF21 Analog, Showed Sustained Pharmacodynamic Effects on Insulin Sensitivity and Lipid Metabolism in Type 2 Diabetes Patients. Cell Rep. Med. 2020, 1, 100057. [Google Scholar] [CrossRef]
- Sanyal, A.; Charles, E.D.; Neuschwander-Tetri, B.A.; Loomba, R.; Harrison, S.A.; Abdelmalek, M.F.; Lawitz, E.J.; Halegoua-DeMarzio, D.; Kundu, S.; Noviello, S.; et al. Pegbelfermin (BMS-986036), a PEGylated Fibroblast Growth Factor 21 Analogue, in Patients with Non-Alcoholic Steatohepatitis: A Randomised, Double-Blind, Placebo-Controlled, Phase 2a Trial. Lancet Lond. Engl. 2019, 392, 2705–2717. [Google Scholar] [CrossRef]
- Cero, C.; Lea, H.J.; Zhu, K.Y.; Shamsi, F.; Tseng, Y.-H.; Cypess, A.M. Β3-Adrenergic Receptors Regulate Human Brown/Beige Adipocyte Lipolysis and Thermogenesis. JCI Insight 2021, 6, e139160. [Google Scholar] [CrossRef]
- Nureki, I.; Kobayashi, K.; Tanaka, T.; Demura, K.; Inoue, A.; Shihoya, W.; Nureki, O. Cryo-EM Structures of the Β3 Adrenergic Receptor Bound to Solabegron and Isoproterenol. Biochem. Biophys. Res. Commun. 2022, 611, 158–164. [Google Scholar] [CrossRef]
- Peres Valgas da Silva, C.; Calmasini, F.; Alexandre, E.C.; Raposo, H.F.; Delbin, M.A.; Monica, F.Z.; Zanesco, A. The Effects of Mirabegron on Obesity-Induced Inflammation and Insulin Resistance Are Associated with Brown Adipose Tissue Activation but Not Beiging in the Subcutaneous White Adipose Tissue. Clin. Exp. Pharmacol. Physiol. 2021, 48, 1477–1487. [Google Scholar] [CrossRef]
- Cypess, A.M.; Weiner, L.S.; Roberts-Toler, C.; Elía, E.F.; Kessler, S.H.; Kahn, P.A.; English, J.; Chatman, K.; Trauger, S.A.; Doria, A.; et al. Activation of Human Brown Adipose Tissue by a Β3-Adrenergic Receptor Agonist. Cell Metab. 2015, 21, 33–38. [Google Scholar] [CrossRef]
- Dehvari, N.; Sato, M.; Bokhari, M.H.; Kalinovich, A.; Ham, S.; Gao, J.; Nguyen, H.T.M.; Whiting, L.; Mukaida, S.; Merlin, J.; et al. The Metabolic Effects of Mirabegron Are Mediated Primarily by Β3 -Adrenoceptors. Pharmacol. Res. Perspect. 2020, 8, e00643. [Google Scholar] [CrossRef] [PubMed]
- Finlin, B.S.; Memetimin, H.; Zhu, B.; Confides, A.L.; Vekaria, H.J.; El Khouli, R.H.; Johnson, Z.R.; Westgate, P.M.; Chen, J.; Morris, A.J.; et al. Pioglitazone Does Not Synergize with Mirabegron to Increase Beige Fat or Further Improve Glucose Metabolism. JCI Insight 2021, 6, 143650. [Google Scholar] [CrossRef]
- Calmasini, F.B.; de Oliveira, M.G.; Alexandre, E.C.; da Silva, F.H.; da Silva, C.P.V.; Candido, T.Z.; Antunes, E.; Mónica, F.Z. Long-Term Treatment with the Beta-3 Adrenoceptor Agonist, Mirabegron Ameliorates Detrusor Overactivity and Restores Cyclic Adenosine Monophosphate (CAMP) Levels in Obese Mice. Neurourol. Urodyn. 2017, 36, 1511–1518. [Google Scholar] [CrossRef] [PubMed]
- O’Mara, A.E.; Johnson, J.W.; Linderman, J.D.; Brychta, R.J.; McGehee, S.; Fletcher, L.A.; Fink, Y.A.; Kapuria, D.; Cassimatis, T.M.; Kelsey, N.; et al. Chronic Mirabegron Treatment Increases Human Brown Fat, HDL Cholesterol, and Insulin Sensitivity. J. Clin. Investig. 2020, 130, 2209–2219. [Google Scholar] [CrossRef] [PubMed]
| Study | Study Design | Baseline Sample | Duration | Dose of Bupropion/Naltrexon | Body Mass Reduction (%) | Percent of Patients with 5/10/15% Weight Reduction | Additional Benefits |
|---|---|---|---|---|---|---|---|
| COR I [52] | RCT | 1742 patients ages 18–65 years with a BMI of 30–45 kg/m2 and uncomplicated obesity or BMI 27–45 kg/m2 with dyslipidaemia or AH, or both | 56 weeks | 360 mg/32 mg 360 mg/16 mg | 6.1 5.0 | 48/25/12 39/20/9 | SSR in WC, TG, hs-CRP, FINS, FPG, HOMA-IR, SBP, DBP. SSI in HDL-C. SSR in WC, TG, hsCRP, HOMA-IR, SBP, DBP. SSI in HDL-C. |
| COR II [53] | RCT | 1496 patients ages 18–65 years with a BMI of 30–45 kg/m2, or a BMI of 27–45 kg/m2 and controlled AH and/or dyslipidemia. | 56 weeks | 360 mg/32 mg | 6.5 | 50.5/28.3/13.5 | SSR in WC, TG, LDL-C, hsCRP, FINS, HOMA-IR, SBP. SSI in HDL-C. |
| COR-BMOD [54] | RCT | 793 patients ages 18–65 years with a BMI of 30–45 kg/m2, or a BMI of 27–45 kg/m2 in the presence of controlled AH and/or dyslipidemia | 56 weeks | 360 mg/32 mg | 9.3 | 66.4/41.5/29.1 | SSR in WC, TG, FINS, HOMA-IR. SSI in HDL-C. |
| COR-DM [55] | RCT | 505 patients with T2DM and ages 18–70 years, with a BMI of ≥27 and ≤45 kg/m2, HbA1c between 7% and 10%, and FPG <270 mg/dL | 56 weeks | 360 mg/32 mg | 5.0 | ≥5% weight loss in 44.5% patients | SSR in WC, HbA1c, TG. SSI in HDL-C. |
| Study | Study Design | Baseline Sample | Duration | Dose of Phentermine/Topiramat | Body Mass Reduction (%) | Percent of Patients with 5/10/15% Weight Reduction | Additional Benefits |
|---|---|---|---|---|---|---|---|
| EQUIP [62] | RCT | 1267 patients with a BMI of ≥35 kg/m2, TG ≤ 200 mg/dL with treatment of 0–1 lipid-lowering medication, BP ≤ 140/90 mmHg with treatment of 0–2 antihypertensive medications, FPG ≤ 110 mg/dL | 56 weeks | 15 mg/92 mg 3.73 mg/23 mg | 10.9 5.1 | 66.7/47.2/32.3 44.9 /18.8/7.3 | SSR in WC, TG, FPG, TCH, LDL-C, SBP, DBP, TCH/HDL-C ratio. SSI in HDL-C. SSR in WC, SBP. SSI in HDL-C. |
| CONQUER [63] | RCT | 2487 patients aged 18–70 years, with a BMI of 27–45 kg/m2, ≥2 comorbidities at baseline (SBP 140–160 mmHg or 130–160 mmHg in T2DM patients, DBP 90–100 mmHg or 85–100 mmHg in T2DM patients or taking ≥2 antihypertensive drugs; TG 2.26–4.52 mmol/L or using ≥2 lipid-lowering drugs; FBG > 5.5 mmol/L or 2 h after OGTT > 7.77 mmol/L or diagnosed T2DM managed with lifestyle changes or metformin monotherapy; WC ≥ 102 cm for men or 88 cm for women. | 56-weeks | 15 mg /92 mg 7.5 mg/46 mg | 9.8 7.8 | 70/48/- 62/37/- | SSR in WC, SBP, DBO, TCH, LDL-C, TG, FPG, HbA1c, FINS, Homa-IR, hsCRP. SSI in HDL-C, adiponectin. SSR in WC, SBP, TCH, TG, FPG, HbA1c, FINS, HOMA-IR, hsCRP. SSI in HDL-C, adiponectin. |
| SEQUEL [64] | RCT | 676 patients with a BMI of 27–45 kg/m2 and two or more comorbidities (AH, dyslipidaemia, diabetes or prediabetes, or abdominal obesity), | 56 weeks | 15.0 mg/92 mg 7.5/46 mg | 10.5 9.3 | 79.3/53.9/31.9 75.2/50.3/24.2 | SSR in TG, LDL-C, FPG, FINS, HbA1c, SBP, DBP. SSI in HDL-C. SSR in TG, LDL-C, FINS, HbA1c, SBP, DBP. SSI in HDL-C. |
| EQUATE Clinicaltrials.gov: NCT00563368 | RCT | 756 patients ages 18–70 years of age who had a BMI of ≥30 and ≤45 kg/m2 | 28 weeks | 15 mg/92 mg 7.5 mg/46 mg | 9.2 8.5 | 68/42/- 64/40/- | SSR in WC, SBP, HBA1c. SSR in WC, HbA1c, adiponectin. |
| Study | Study Design | Baseline Sample | Duration | Dose of Lorcaserine | Body Mass Reduction (%) | Percent of Patients with 5/10/15% Weight Reduction | Additional Benefits |
|---|---|---|---|---|---|---|---|
| BLOM [76] | RCT | 3182 patients ages 18–65 years and a BMI of 30–45 kg/m2 or 27–45 kg/m2 with at least 1 coexisting condition (AH, dyslipidemia, cardiovascular disease, impaired glucose tolerance, or sleep apnea). | 52 weeks | 10 mg BID | 5.81 | 47.5/22.6 | SSR in SBP, DBP, TCH, LFL-C, TG, FPG, FINS, HOMA-IR, hsCRP, fibrinogen. |
| BLOOSOM [73] | RCT | 4008 patients ages 16–65 years, with a BMI of 30–45 kg/m2 or 27–29.9 kg/m2 with obesity-related comorbid condition | 52 weeks | 10 mg BID 10 mg QD | 5.8 4.7 | 47.2/22.6/- 40.2/17.4/- | SSR in WC, TG, ApoB, total body fat, lean body mass. SSI in HDL-C. SSR in WC, TG/ SSI in HDL-C. |
| BLOOM-DM [74] | RCT | 604 patients ages 18–65 years with T2DM treated with metformin, a SFU, or both; with HbA1c 7–10%; with a BMI of 27–45 kg/m2 | 52 weeks | 10 mg BID 10 mg QD | 4.7 5.0 | 37.5/16.3/- 44.7/18.1/- | SSR in WC, HC, HR, FPG, HbA1c, HOMA-IR. SSI in HDL-C. SSR in HC, HR, apoA1, FPG, FINS. |
| CAMELLIA -TIMI 61 [75] | RCT | 12,000 patients ages at least 40 years old with a BMI of ≥27 kg/m2 with either established atherosclerotic cardiovascular disease or multiple cardiovascular risk factors | 3.3 years | 10 mg BID | 4.2 | 38.7/14.6 | SSR in HbA1c, FPG. |
| Study | Study Design | Baseline Sample | Duration | Dose of Liraglutide (9 mg) | Body Mass Reduction (%) | Percent of Patients with 5/10/15% Weight Reduction | Additional Benefits |
|---|---|---|---|---|---|---|---|
| SCALE Obesity and Prediabetes [89] | RCT | 3731 patients ages ≥ 18 years with a BMI of ≥30 kg/m2 or ≥27 kg/m2 with treated or untreated dyslipidemia or AH, without DM | 56 weeks | 3.0 | 8.0 | 63.2/33.1/14.4 | SSR in SBP, DBP, HbA1c, FPG, IR, and beta-cell function. SSI in FINS, C-peptide, HDL-C, PAI-1, adiponectin. |
| SCALE Diabetes [90] | RCT | 846 patients ages ≥18 years with a BMI of ≥27 kg/m2 taking 0–3 oral hypoglycemic agents (metformin, thiazolidinedione, sulfonylurea) with stable body weight, and HbA1c 7–10%. | 56 weeks | 1.8 3.0 | 4,7 6,0 | 40.4/15.9/- 54.3/25.2/- | SSR in WC, HbA1c, FPG, PPG, HOMA-B. SSI in glucagon, proinsulin, proinsulin to insulin ratio, hsCRP. SSR in WC, HbA1c, FPG, PPG, HOMA-B, HOMA-IR, SBP, TCH, VLDL, TG. SSI in HDL-C, glucagon, proinsulin, proinsulin to insulin ratio, fibrinogen, PAI-1, hsCRP. |
| SCALE Maintenance [92] | RCT | 422 patients ages ≥ 18 years with a stable BMI of ≥30 kg/m2 or ≥27 kg/m2 with comorbidities of treated or untreated dyslipidemia and/or treated or untreated AH who lost ≥5% of initial weight during a low-calorie diet run-in, without DM | 56 weeks | 3.0 | 6.2 | 50.5/26.1/- | SSR in WC, HbA1c, FPG, FINS, TG, hsCRP, SBP. |
| SCALE Sleep Apnea [91] | RCT | 359 patients ages 18–64 years with a stable BMI (<5% change during the previous 3 months) and a BMI of ≥30 kg/m2 with moderate or severe OSA | 32 weeks | 3.0 | 5.7% | 46.3/23.4/- | SSR in HbA1c, FPG, SBP. |
| Study | Study Design | Baseline Sample | Duration | Dose of Semaglutide (mg) | Body Mass Reduction (%) | Percent of Patients with 5/10/15% Weight Reduction | Additional Benefits |
|---|---|---|---|---|---|---|---|
| STEP 1 [101] | RCT | 1961 patients ages ≥ 18 years old with one or more self-reported unsuccessful dietary efforts to lose weight and either a BMI of ≥30 kg/m2 or ≥27 kg/m2 with ≥1 treated or untreated weight-related coexisting conditions (i.e., AH, dyslipidemia, OSAS, CVD) | 68 weeks | 2.4 | 14.9 | 86.4/69.1/50.5 | SSR in WC, SBP. |
| STEP 2 [102] | RCT | 1210 patients ages ≥ 18 years old, with at least one reported unsuccessful dietary effort to lose weight, a BMI of ≥27 kg/m2, HbA1c 7–10%, T2DM at least 180 days before screening | 68 weeks | 2.4 1.0 | 9.6 6.99 | 68.8/45.6/25.8 57.1/28.7/13.7 | SSR in WC, HbA1c, SBP, FSIN, DBP, TCH, VLDL, free fatty acids, TG, CRP. SSI in HDL-C. SSR in HbA1c. |
| STEP 3 [103] | RCT | 611 patients with ages ≥ 18 years old, reported ≥1 unsuccessful dietary efforts to lose weight and BMI ≥30 kg/m2 or ≥27 with ≥1 weight-related comorbidity (CVD, dyslipidemia, AH, OSAS | 68 weeks | 2.4 | 16 | 86.6/75.3/55.8 | SSR in WC, SBP, DBP, FPG, HbA1c, FSIN, TCH, LDL-CVLDL, free fatty acids, TG, CRP. |
| STEP 4 [104] | RCT | 902 patients with ages ≥ 18 years old, with ≥1 reported unsuccessful dietary efforts to lose weight and a BMI of ≥30 kg/m2 or ≥27 with ≥1 weight-related comorbidity (CVD, dyslipidemia, AH, OSAS) without T2DM. | 68 weeks | 2.4 | 7.9 | 88.7/79/63.4 | SSR in WC, SBP, HfA1c, FSIN, FPG, TCH, LDL-C, VLDL-C, TG. SSI in |
| STEP 6 [105] | RCT | 401 patients with ages ≥ 18 years old, with ≥1 reported unsuccessful dietary efforts to lose weight and a BMI of ≥27 kg/m2 ported ≥2 treated or untreated weight-related comorbidity or ≥35 with ≥1 weight-related comorbidity (dyslipidemia, AH, or, in Japan only, T2DM diagnosis 80 days before screening and HbA1c 7–10%). | 68 | 2.4 1.7 | 13.2 9.6 | 83/61/41 72/42/24 | SSR in WC, HbA1c, FSIN, SBP, DBP, PAI-1, CRP, TCH, LDL-C, TG FFA. SSRI in WC, HbA1c, FSIN, SBP, DBP, PAI-1, CRP, FFA, TG, TCH, LDL-C. |
| Drug | Most Common Side Effects | Serious Side Effects | Interaction | Contraindications | Dosage and Route of Administration |
|---|---|---|---|---|---|
| Orlistat | Abdominal pain and discomfort, loose stools, liquid stools, push to the stools, rectal pain and discomfort, headache, fatigue, anxiety | Hypoglycemia infections | Cyclosporine, Acarbose, Oral antidiabetic drugs, Fat-soluble vitamins, Amiodarone | Hypersensitivity to orlistat or to any of the excipients, chronic malabsorption syndrome, cholestasis, breast-feeding | 3x/day, p.o. |
| Bupropion /naltrexone | Nausea, constipation, vomiting, dizziness, dry mouth, headache | Affective disorder, suicidal ideation, seizure, cholecystitis, hepatitis, erythema multiforme, Stevens-Johnson syndrome | Active substances metabolized by cytochrome P450 isoenzymes; CYP2D6 isoenzyme substrates; inducers, inhibitors and substrates of the CYP2B6 isoenzyme; organic cation transporter 2 inhibitors | Hypersensitivity to bupropion, naltrexone or to any of the excipients; uncontrolled hypertension; epilepsy or history of seizures; cancer tumor in the central nervous system; the period immediately after abrupt withdrawal from alcohol or benzodiazepines in an addicted person; history of bipolar affective disorder; psychic bulimia or anorexia nervosa; dependence on long-term use of opioids or opiate agonists (e.g., methadone) and the period immediately after abrupt opiate withdrawal in an addicted person; taking monoamine oxidase inhibitors; severe hepatic impairment; end-stage renal failure or severe renal impairment | 2x/day, p.o |
| Phentermine/topiramat | Dry mouth, paresthesia, constipation, memory impairment insomnia, depression, anxiety | Metabolic acidosis, hypokalemia, angle-closure glaucoma, transient blindness, deafness, atrial fibrillation, arrhythmias, deep vein thrombosis, falls | Antiepileptic drugs, Hydrochlorothiazide, Hypericum perforatum, oral contraceptives, metformin, pioglitazone, glibenclamide, digoxin, lithium, risperidone, central nervous system depressants, carbonic anhydrase inhibitors, potassium-sparing diuretics, valproic acid | Hypersensitivity to phentermine, topiramate, or to any of the excipients; pregnancy; breast feeding; treatment with monoamine oxidase inhibitors | 2x/day, p.o |
| Liraglutide | Gastrointestinal symptoms (for example: nausea, vomiting, diarrhea, constipation, dry mouth, dyspepsia), insomnia, headache, hives | Hypoglycemia, inflammation of the pancreas, cholecystitis, cholelithiasis, acute renal failure | Paracetamol, atorvastatin, griseofulvin, digoxin, lisinopril, oral contraceptives | Hypersensitivity to liraglutide or to any of the excipients, medullary thyroid cancer, pregnancy, breast feeding | 1x/day, s.c. |
| Semaglutide | Gastrointestinal symptoms (for example: vomiting, diarrhea, constipation, nausea, abdominal pain, dyspepsia, bouncing), headache, dizziness, tiredness | Cholelithiasis, hypoglycemia | Paracetamol, oral contraceptives | Hypersensitivity to semaglutide or to any of the excipients, medullary thyroid cancer, pregnancy, breast feeding | 1x/week, s.c. |
| Lorcanserin | Nausea, vomiting, constipation, diarrhea, fatigue, upper respiratory tract infection, urinary tract infection, back pain, headache, dizziness, rash, attention and memory deficit, priapism, hyperprolactinemia | Infections, serotonin syndrome, hypoglycemia | Drugs that interfere with serotonin neurotransmission, drugs metabolized by cytochrome P450 2D6 | Hypersensitivity to lorcaserin or to any of the excipients, pregnancy, breast feeding | 2x/day, p.o. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kosmalski, M.; Deska, K.; Bąk, B.; Różycka-Kosmalska, M.; Pietras, T. Pharmacological Support for the Treatment of Obesity—Present and Future. Healthcare 2023, 11, 433. https://doi.org/10.3390/healthcare11030433
Kosmalski M, Deska K, Bąk B, Różycka-Kosmalska M, Pietras T. Pharmacological Support for the Treatment of Obesity—Present and Future. Healthcare. 2023; 11(3):433. https://doi.org/10.3390/healthcare11030433
Chicago/Turabian StyleKosmalski, Marcin, Kacper Deska, Bartłomiej Bąk, Monika Różycka-Kosmalska, and Tadeusz Pietras. 2023. "Pharmacological Support for the Treatment of Obesity—Present and Future" Healthcare 11, no. 3: 433. https://doi.org/10.3390/healthcare11030433
APA StyleKosmalski, M., Deska, K., Bąk, B., Różycka-Kosmalska, M., & Pietras, T. (2023). Pharmacological Support for the Treatment of Obesity—Present and Future. Healthcare, 11(3), 433. https://doi.org/10.3390/healthcare11030433








