Conservative Management for Retained Products of Conception in Late Pregnancy
Abstract
1. Introduction
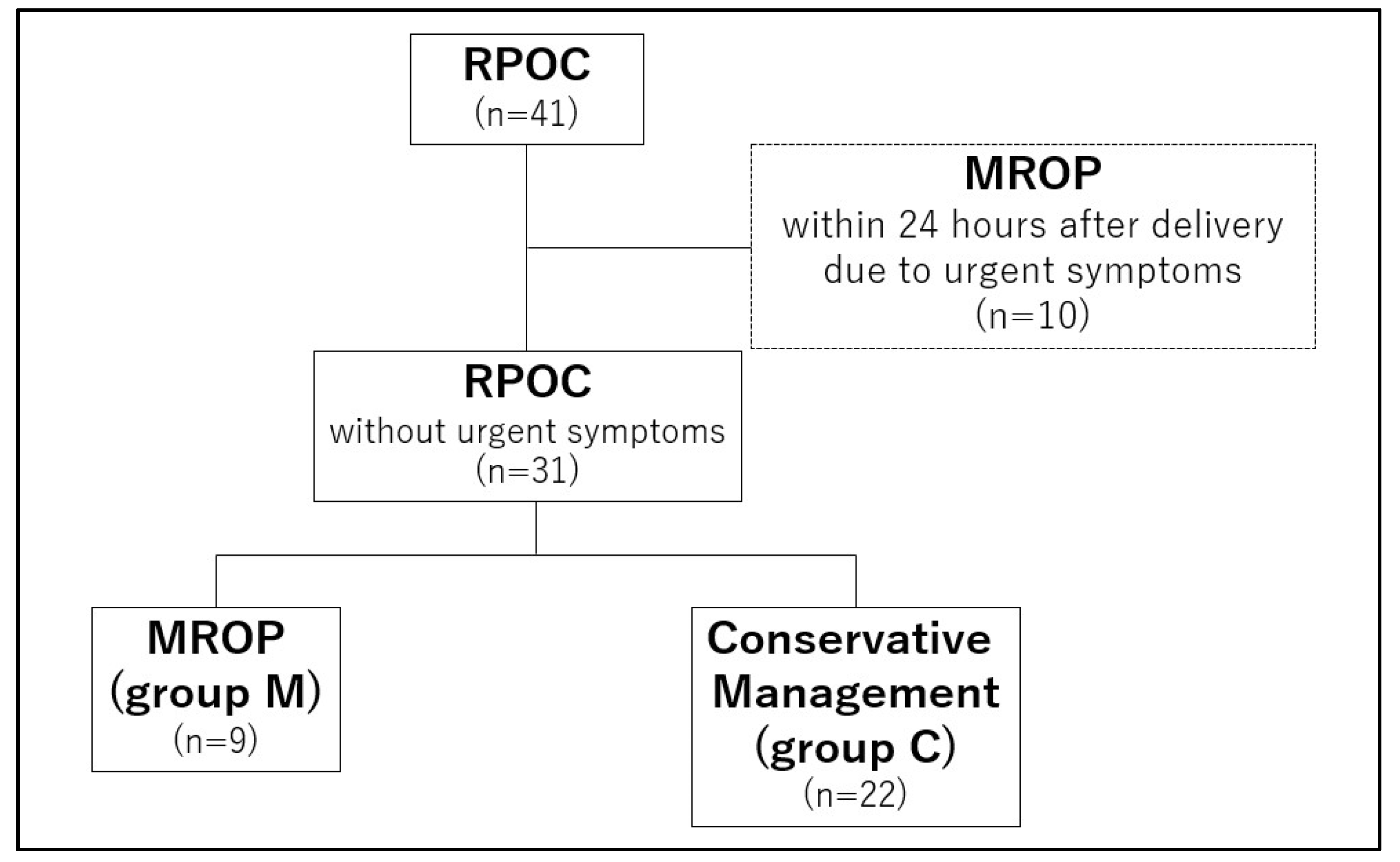
2. Materials and Methods
2.1. Patients
2.2. Endpoint
2.3. Statistical Analysis
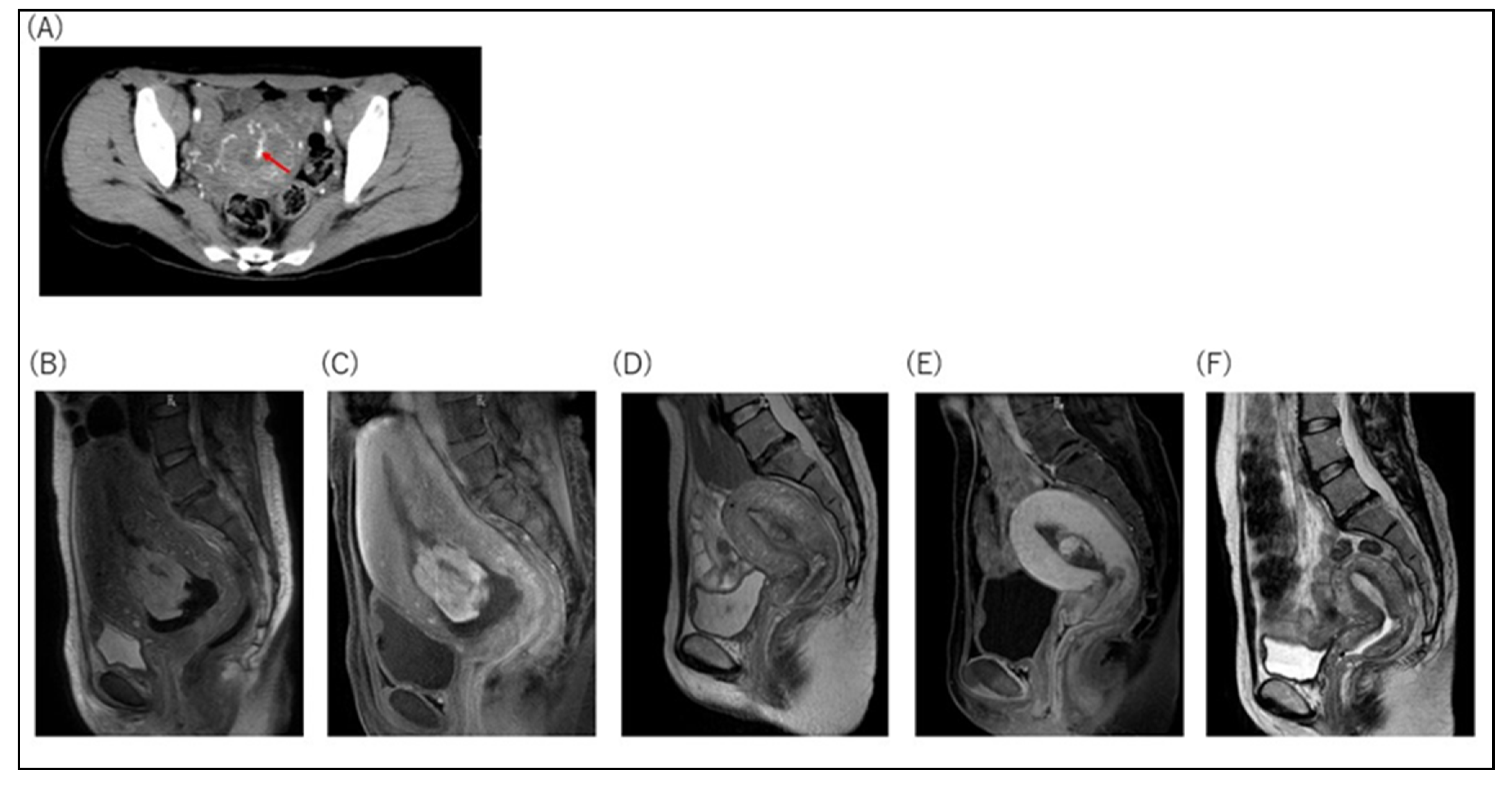
3. Results
3.1. Nineteen Cases with MROP within 24 Hours of Delivery
3.2. Twenty-Two Cases with Conservative Management
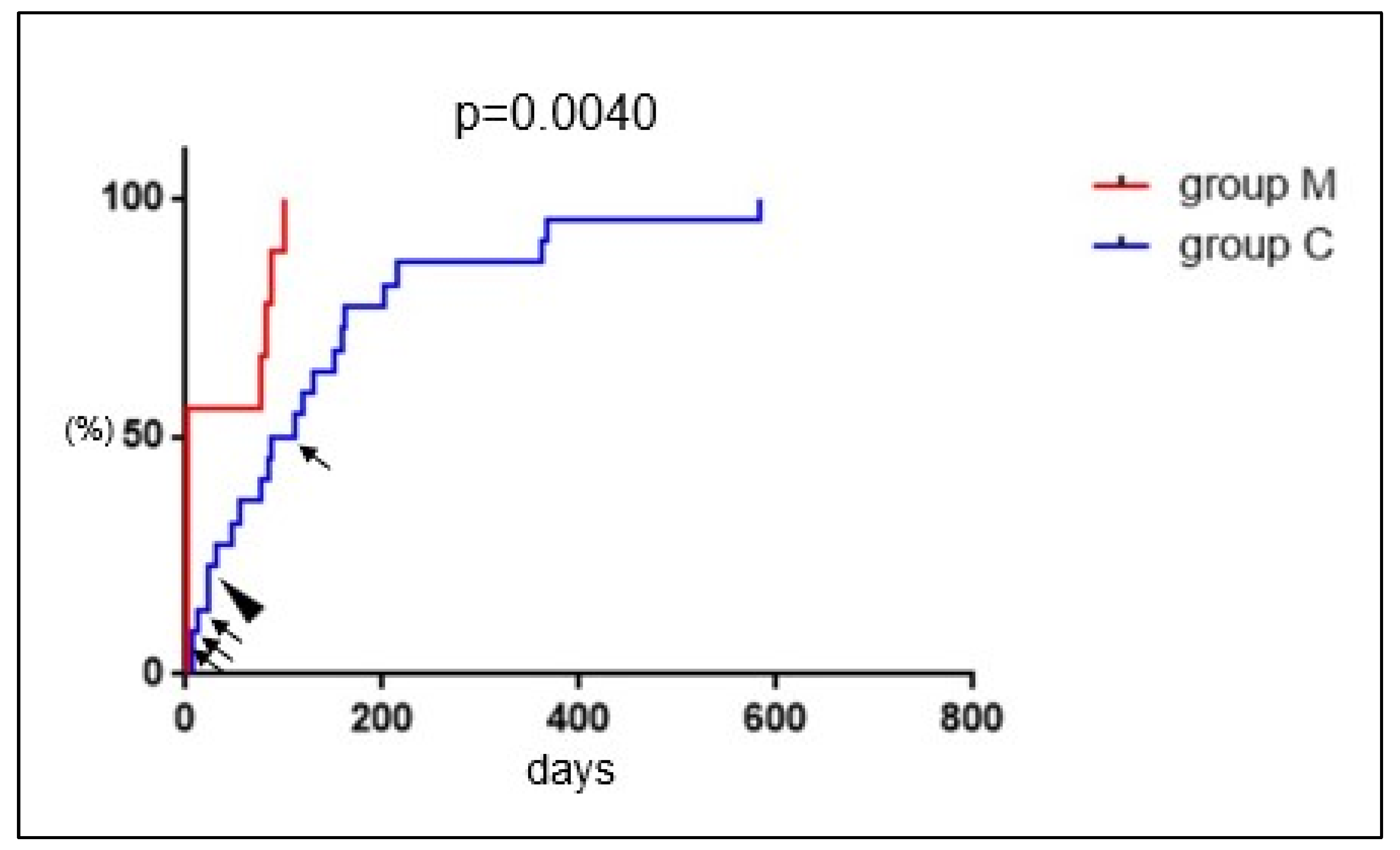
3.3. Comparison between Group M and Group C
3.4. Serial Changes in Serum hCG
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Linde, L.E.; Rasmussen, S.; Moster, D.; Kessler, J.; Baghestan, E.; Gissler, M.; Ebbing, C. Risk factors and recurrence of cause-specific postpartum hemorrhage: A population-based study. PLoS ONE 2022, 17, e0275879. [Google Scholar] [CrossRef] [PubMed]
- Tunçalp, O.; Souza, J.P.; Gülmezoglu, M. New WHO recommendations on prevention and treatment of postpartum hemorrhage. Int. J. Gynaecol. Obstet. 2013, 123, 254–256. [Google Scholar] [CrossRef]
- Weissbach, T.; Haikin-Herzberger, E.; Bacci-Hugger, K.; Shechter-Maor, G.; Fejgin, M.; Biron-Shental, T. Immediate postpartum ultrasound evaluation for suspected retained placental tissue in patients undergoing manual removal of placenta. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 192, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Hoveyda, F.; MacKenzie, I.Z. Secondary postpartum haemorrhage: Incidence, morbidity and current management. Bjog 2001, 108, 927–930. [Google Scholar] [PubMed]
- National Collaborating Centre for Women’s and Children’s Health (UK). Intrapartum Care: Care of Healthy Women and Their Babies During Childbirth; National Institute for Health and Care Excellence (UK): London, UK, 2014. [Google Scholar]
- World Health Organization. Pregnancy, Childbirth, Postpartum and Newborn Care: A Guide for Essential Practice, 3rd ed.; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Urner, F.; Zimmermann, R.; Krafft, A. Manual removal of the placenta after vaginal delivery: An unsolved problem in obstetrics. J. Pregnancy 2014, 2014, 274651. [Google Scholar] [CrossRef] [PubMed]
- Anteby, M.; Many, A.; Ashwal, E.; Yogev, Y.; Shinar, S. Risk factors and complications of manual placental removal after vaginal delivery—How common are additional invasive procedures? J. Matern. Fetal Neonatal. Med. 2019, 32, 384–388. [Google Scholar] [CrossRef]
- Takahashi, H.; Ohhashi, M.; Baba, Y.; Nagayama, S.; Ogoyama, M.; Horie, K.; Suzuki, H.; Usui, R.; Ohkuchi, A.; Matsubara, S. Conservative management of retained products of conception in the normal placental position: A retrospective observational study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2019, 240, 87–92. [Google Scholar] [CrossRef]
- Wada, Y.; Takahashi, H.; Suzuki, H.; Ohashi, M.; Ogoyama, M.; Nagayama, S.; Baba, Y.; Usui, R.; Suzuki, T.; Ohkuchi, A.; et al. Expectant management of retained products of conception following abortion: A retrospective cohort study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 260, 1–5. [Google Scholar] [CrossRef]
- Kobayashi, M.; Nakagawa, S.; Kawanishi, Y.; Masuda, T.; Maenaka, T.; Toda, A.; Miyake, T.; Hiramatsu, K.; Miyoshi, A.; Mimura, K.; et al. The RPOC long axis is a simple indicator for predicting the need of invasive strategies for secondary postpartum hemorrhage in either post-abortion or post-partum women: A retrospective case control study. BMC Pregnancy Childbirth 2021, 21, 653. [Google Scholar] [CrossRef]
- Sentilhes, L.; Ambroselli, C.; Kayem, G.; Provansal, M.; Fernandez, H.; Perrotin, F.; Winer, N.; Pierre, F.; Benachi, A.; Dreyfus, M.; et al. Maternal outcome after conservative treatment of placenta accreta. Obstet. Gynecol. 2010, 115, 526–534. [Google Scholar] [CrossRef]
- Sardo, A.D.S.; Foreste, V.; Gallo, A.; Manzi, A.; Riccardi, C.; Carugno, J. Hysteroscopy and retained products of conception: An update. Gynecol. Minim. Invasive Ther. 2021, 10, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Vitale, S.G.; Parry, J.P.; Carugno, J.; Cholkeri-Singh, A.; Della Corte, L.; Cianci, S.; Schiattarella, A.; Riemma, G.; De Franciscis, P. Surgical and Reproductive Outcomes after Hysteroscopic Removal of Retained Products of Conception: A Systematic Review and Meta-analysis. J. Minim. Invasive Gynecol. 2021, 28, 204–217. [Google Scholar] [CrossRef] [PubMed]
- Pather, S.; Ford, M.; Reid, R.; Sykes, P. Postpartum curettage: An audit of 200 cases. Aust. N. Z. J. Obstet. Gynaecol. 2005, 45, 368–371. [Google Scholar] [CrossRef]
- Imai, S.; Kondoh, E.; Kawasaki, K.; Mogami, H.; Ueda, A.; Umeoka, S.; Konishi, I. Placental blood flow disappears coincident with a fall in human chorionic gonadotropin to undetectable levels in conservative management of placenta accreta. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 180, 199–201. [Google Scholar] [CrossRef]
- Miyakoshi, K.; Otani, T.; Kondoh, E.; Makino, S.; Tanaka, M.; Takeda, S. Retrospective multicenter study of leaving the placenta in situ for patients with placenta previa on a cesarean scar. Int. J. Gynecol. Obstet. 2018, 140, 345–351. [Google Scholar] [CrossRef]
- Kamaya, A.; Krishnarao, P.M.; Nayak, N.; Jeffrey, R.B.; Maturen, K.E. Clinical and imaging predictors of management in retained products of conception. Abdom. Radiol. 2016, 41, 2429–2434. [Google Scholar] [CrossRef] [PubMed]
- Hooker, A.B.; Aydin, H.; Brölmann, H.A.; Huirne, J.A. Long-term complications and reproductive outcome after the management of retained products of conception: A systematic review. Fertil. Steril. 2016, 105, 156–164.e2. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Ushida, T.; Imai, K.; Nakano-Kobayashi, T.; Iitani, Y.; Matsuo, S.; Yoshida, S.; Yamashita, M.; Kajiyama, H.; Kotani, T. Manual removal of the placenta and postpartum hemorrhage: A multicenter retrospective study. J. Obstet. Gynaecol. Res. 2021, 47, 3867–3874. [Google Scholar] [CrossRef]
- van Ast, M.; Goedhart, M.M.; Luttmer, R.; Orelio, C.; Deurloo, K.L.; Veerbeek, J. The duration of the third stage in relation to postpartum hemorrhage. Birth 2019, 46, 602–607. [Google Scholar] [CrossRef]
- Ikeda, A.; Kondoh, E.; Chigusa, Y.; Mogami, H.; Nakao, K.K.; Kido, A.; Horie, A.; Baba, T.; Hamanishi, J.; Mandai, M. Novel subtype of atonic postpartum hemorrhage: Dynamic computed tomography evaluation of bleeding characteristics and the uterine cavity. J. Matern. Fetal Neonatal Med. 2020, 33, 3286–3292. [Google Scholar] [CrossRef]
- Furukawa, S.; Fujisaki, M.; Maki, Y.; Oohashi, M.; Doi, K.; Sameshima, H. Manual removal of placenta in women having unpredictable adherent placenta. J. Obstet. Gynaecol. Res. 2019, 45, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Tandberg, A.; Albrechtsen, S.; Iversen, O.E. Manual removal of the placenta. Incidence and clinical significance. Acta Obstet. Gynecol. Scand. 1999, 78, 33–36. [Google Scholar] [PubMed]
- Ely, J.W.; Rijhsinghani, A.; Bowdler, N.C.; Dawson, J.D. The association between manual removal of the placenta and postpartum endometritis following vaginal delivery. Obstet. Gynecol. 1995, 86, 1002–1006. [Google Scholar] [CrossRef] [PubMed]
- Akiba, N.; Iriyama, T.; Nakayama, T.; Seyama, T.; Sayama, S.; Kumasawa, K.; Komatsu, A.; Yabe, S.; Nagamatsu, T.; Osuga, Y.; et al. Ultrasonographic vascularity assessment for predicting future severe hemorrhage in retained products of conception after second-trimester abortion. J. Matern. Fetal Neonatal Med. 2021, 34, 562–568. [Google Scholar] [CrossRef]
- Lockwood, C.J.; Moore, T.; Copel, J.; Silver, R.M.; Resnik, R. Creasy & Resnik’s Maternal-Fetal Medicine Principles and Practice, 8th ed.; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Shitanaka, S.; Chigusa, Y.; Kawahara, S.; Kawasaki, K.; Mogami, H.; Mandai, M.; Kondoh, E. Conservative management for retained products of conception after less than 22 weeks of gestation. J. Obstet. Gynaecol. Res. 2020, 46, 1982–1987. [Google Scholar] [CrossRef]
- Grewal, K.; Al-Memar, M.; Fourie, H.; Stalder, C.; Timmerman, D.; Bourne, T. Natural history of pregnancy-related enhanced myometrial vascularity following miscarriage. Ultrasound Obstet. Gynecol. 2020, 55, 676–682. [Google Scholar] [CrossRef]
- Midgley, A.R., Jr.; Jaffe, R.B. Regulation of human gonadotropins. II. Disappearance of human chorionic gonadotropin following delivery. J. Clin. Endocrinol. Metab. 1968, 28, 1712–1718. [Google Scholar] [CrossRef]
- Matsumura, N.; Inoue, T.; Fukuoka, M.; Sagawa, N.; Fujii, S. Changes in the serum levels of human chorionic gonadotropin and the pulsatility index of uterine arteries during conservative management of retained adherent placenta. J. Obstet. Gynaecol. Res. 2000, 26, 81–87. [Google Scholar] [CrossRef]
- van Mello, N.M.; Mol, F.; Ankum, W.M.; Mol, B.W.; van der Veen, F.; Hajenius, P.J. Ectopic pregnancy: How the diagnostic and therapeutic management has changed. Fertil. Steril. 2012, 98, 1066–1073. [Google Scholar] [CrossRef]
- D’Hoore, E.; D’Hoore, L.; Van den Berghe, S.; Roets, E.; van Wessel, S.; Hamerlynck, T. Operative hysteroscopy in the minimally invasive management of interstitial pregnancy and interstitially retained products of conception: A case report and systematic literature review. Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 265, 54–59. [Google Scholar] [CrossRef]





| Heavy Bleeding (n = 12) | Light Bleeding (n = 9) | p Value | |
|---|---|---|---|
| Gestational age at delivery (weeks), median (range) | 39.9 (35.1–41.4) | 38.1 (36.0–41.1) | 0.2881 |
| Mode of delivery, n (%) | |||
| vaginal delivery | 12 (100) | 6 (66.7) | 0.0632 |
| cesarean section | 0 (0) | 3 (33.3) | |
| RPOC length (mm) at diagnosis, median (range) | 47.5 (17.0–112.0) | 45.0 (20.0–131) | 0.6511 |
| RPOC with flow on MRI or CT, n (%) | 9/12 (75.0) | 8/9 (88.9) | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fujishima, R.; Kawasaki, K.; Moriuchi, K.; Shiro, R.; Yo, Y.; Matsumura, N. Conservative Management for Retained Products of Conception in Late Pregnancy. Healthcare 2023, 11, 168. https://doi.org/10.3390/healthcare11020168
Fujishima R, Kawasaki K, Moriuchi K, Shiro R, Yo Y, Matsumura N. Conservative Management for Retained Products of Conception in Late Pregnancy. Healthcare. 2023; 11(2):168. https://doi.org/10.3390/healthcare11020168
Chicago/Turabian StyleFujishima, Risa, Kaoru Kawasaki, Kaori Moriuchi, Reona Shiro, Yoshie Yo, and Noriomi Matsumura. 2023. "Conservative Management for Retained Products of Conception in Late Pregnancy" Healthcare 11, no. 2: 168. https://doi.org/10.3390/healthcare11020168
APA StyleFujishima, R., Kawasaki, K., Moriuchi, K., Shiro, R., Yo, Y., & Matsumura, N. (2023). Conservative Management for Retained Products of Conception in Late Pregnancy. Healthcare, 11(2), 168. https://doi.org/10.3390/healthcare11020168







