Treatment and Improvement of Healing after Surgical Intervention
Abstract
1. Introduction
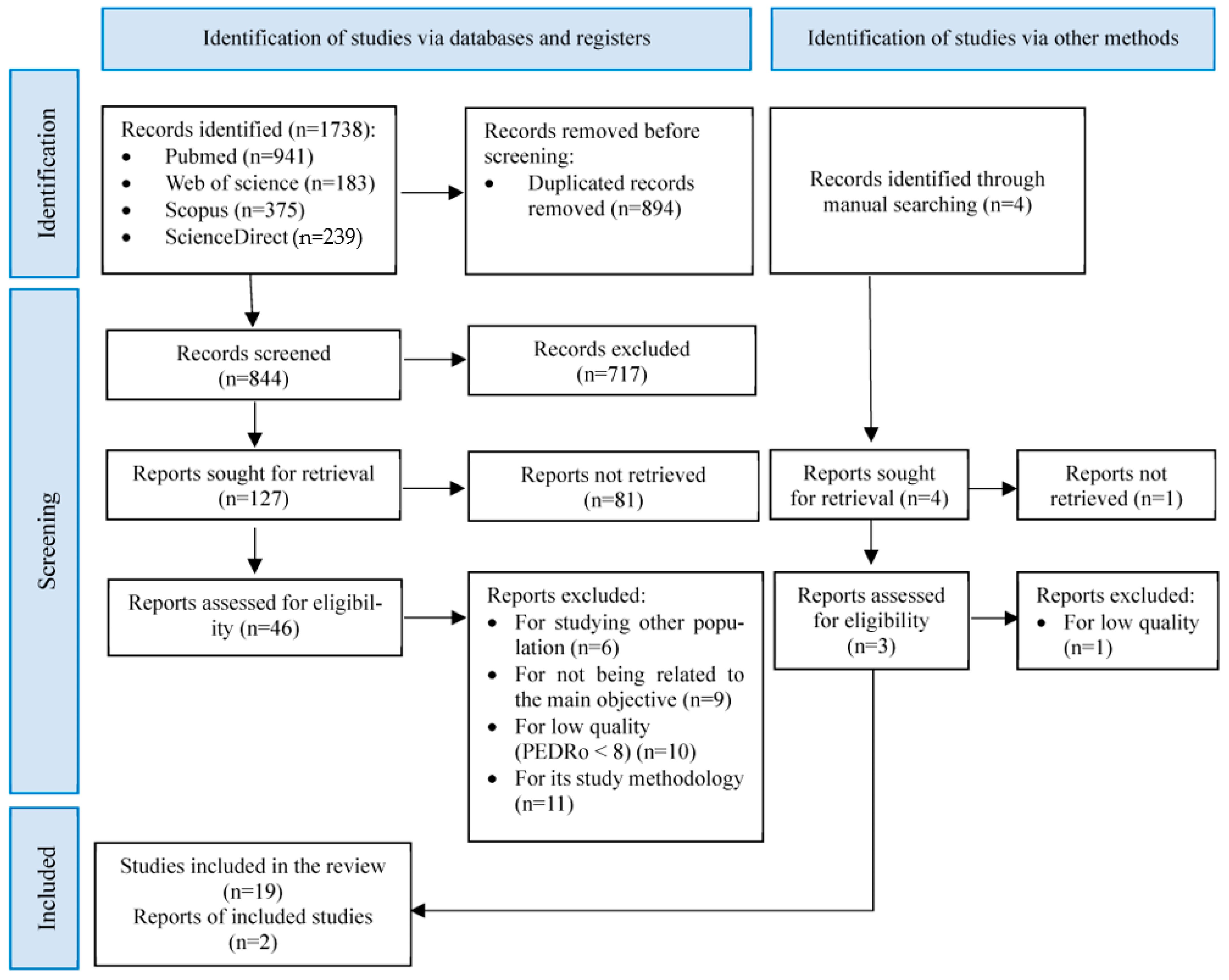
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lingzhi, Z.; Meirong, L.; Xiaobing, F. Biological Approaches for Hypertrophic Scars. Int. Wound J. 2020, 17, 405–418. [Google Scholar] [CrossRef] [PubMed]
- Dunphy, J.E. Wound Healing. Surg. Clin. N. Am. 1978, 58, 907–916. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, E.L.; Roberts, J.L.; Moseley, R.; Griffiths, P.C.; Thomas, D.W. Evaluation of the Physical and Biological Properties of Hyaluronan and Hyaluronan Fragments. Int. J. Pharm. 2011, 420, 84–92. [Google Scholar] [CrossRef]
- Son, D.; Harijan, A. Overview of Surgical Scar Prevention and Management. J. Korean Med. Sci. 2014, 29, 751–757. [Google Scholar] [CrossRef]
- Almine, J.F.; Wise, S.G.; Weiss, A.S. Elastin Signaling in Wound Repair. Birth Defects Res. Part C Embryo Today 2012, 96, 248–257. [Google Scholar] [CrossRef]
- Monslow, J.; Sato, N.; MacK, J.A.; Maytin, E.V. Wounding-Induced Synthesis of Hyaluronic Acid in Organotypic Epidermal Cultures Requires the Release of Heparin-Binding EGF and Activation of the EGFR. J. Investig. Dermatol. 2009, 129, 2046–2058. [Google Scholar] [CrossRef] [PubMed]
- Slemp, A.E.; Kirschner, R.E. Keloids and Scars: A Review of Keloids and Scars, Their Pathogenesis, Risk Factors, and Management. Curr. Opin. Pediatr. 2006, 18, 396–402. [Google Scholar] [CrossRef]
- Freedman, B.R.; Hwang, C.; Talbot, S.; Hibler, B.; Matoori, S.; Mooney, D.J. Breakthrough Treatments for Accelerated Wound Healing. Sci. Adv. 2023, 9, eade7007. [Google Scholar] [CrossRef]
- Watson, D.; Reuther, M. Scar Revision Techniquespearls and Pitfalls. Facial Plast. Surg. 2012, 28, 487–491. [Google Scholar] [CrossRef]
- Gantwerker, E.A.; Hom, D.B. Skin: Histology and Physiology of Wound Healing. Facial Plast. Surg. Clin. N. Am. 2011, 19, 441–453. [Google Scholar] [CrossRef]
- David Chang, C.W.; Russell Ries, W. Nonoperative Techniques for Scar Management and Revision. Facial Plast. Surg. 2001, 17, 283–287. [Google Scholar] [CrossRef] [PubMed]
- González, N.; Goldberg, D.J. Update on the Treatment of Scars. J. Drugs Dermatol. 2019, 18, 550–555. [Google Scholar] [PubMed]
- Ogawa, R. The Most Current Algorithms for the Treatment and Prevention of Hypertrophic Scars and Keloids: A 2020 Update of the Algorithms Published 10 Years Ago. Plast. Reconstr. Surg. 2022, 149, 79E–94E. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, U.E. Internationale Klinische Empfehlungen Zur Narbenbehandlung. Zentralbl. Chir. 2004, 129, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Riccio, M.; Marchesini, A.; Senesi, L.; Skrami, E.; Gesuita, R.; De Francesco, F. Managing Pathologic Scars by Injecting Auto-Cross-Linked Hyaluronic Acid: A Preliminary Prospective Clinical Study. Aesthet. Plast. Surg. 2019, 43, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Garg, S.; Dahiya, N. Surgical Scar Revision: An Overview. J. Cutan. Aesthet. Surg. 2014, 7, 3. [Google Scholar] [CrossRef]
- Kerwin, L.Y.; El Tal, A.K.; Stiff, M.A.; Fakhouri, T.M. Scar Prevention and Remodeling: A Review of the Medical, Surgical, Topical and Light Treatment Approaches. Int. J. Dermatol. 2014, 53, 922–936. [Google Scholar] [CrossRef]
- Lee Peng, G.; Kerolus, J.L. Management of Surgical Scars. Facial Plast. Surg. Clin. N. Am. 2019, 27, 513–517. [Google Scholar] [CrossRef]
- Urrútia, G.; Bonfill, X. PRISMA Declaration: A Proposal to Improve the Publication of Systematic Reviews and Meta-Analyses. Med. Clin. 2010, 135, 507–511. [Google Scholar] [CrossRef]
- Sackett, D.L.; Rosenberg, W.M.C.; Gray, J.A.M.; Haynes, R.B.; Richardson, W.S. Evidence Based Medicine: What It Is and What It Isn’t. 1996. Clin. Orthop. Relat. Res. 2007, 455, 3–5. [Google Scholar]
- Sherrington, C.; Herbert, R.D.; Maher, C.G.; Moseley, A.M. PEDro. A Database of Randomized Trials and Systematic Reviews in Physiotherapy. Man. Ther. 2000, 5, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Abedini, R.; Mehdizade Rayeni, N.; Haddady Abianeh, S.; Rahmati, J.; Teymourpour, A.; Nasimi, M. Botulinum Toxin Type A Injection for Mammoplasty and Abdominoplasty Scar Management: A Split-Scar Double-Blinded Randomized Controlled Study. Aesthet. Plast. Surg. 2020, 44, 2270–2276. [Google Scholar] [CrossRef]
- Chen, Z.; Jin, Y.; Zou, Y.; Qiu, Y.; Hu, L.; Chang, L.; Chen, H.; Lin, X. Scar Prevention with Prolonged Use of Tissue Adhesive Zipper Immediately after Facial Surgery: A Randomized Controlled Trial. Aesthet. Surg. J. 2022, 42, NP265–NP272. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, Z.; Pang, R.; Wei, Z.; Zhang, H.; Liu, W.; Li, G. The Effect of Botulinum Toxin Injection Dose on the Appearance of Surgical Scar. Sci. Rep. 2021, 11, 13670. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.H.; Kim, D.S.; Cheon, J.H.; Yoon, J.M.; Baek, S.K.; Jung, K.Y.; Yoon, E.S.; Park, S.H. Current Protocol for Aesthetic Scar Management in Thyroid Surgery. Laryngoscope 2021, 131, E2188–E2195. [Google Scholar] [CrossRef] [PubMed]
- Dolynchuk, K.N.; Tredget, E.E. A Preliminary Report of the Biochemical and Clinical Effects of 1,4-Diaminobutane on Prevention of Human Hypertrophic Scars. Plast. Reconstr. Surg. 2020, 145, 76e–84e. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.L.; Ho, C.K.; Tremp, M.; Xie, Y.; Li, Q.; Zan, T. Early Postoperative Application of Botulinum Toxin Type A Prevents Hypertrophic Scarring after Epicanthoplasty: A Split-Face, Double-Blind, Randomized Trial. Plast. Reconstr. Surg. 2019, 144, 835–844. [Google Scholar] [CrossRef]
- Ilori, O.S.; Oladele, A.O.; Ilori, O.R.; Onilede, D.A. Efficacy of Microporous Tape in the Prevention of Abnormal Post-Surgical Scars among a Black Population. J. Ayub Med. Coll. Abbottabad 2022, 34, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.; Gentzkow, G.; Berman, G.; Senne, L.; Jewell, M.; Connall, T.P.; Miller, S.R.; Galiano, R.D.; Young, L. Anti-CTGF Oligonucleotide Reduces Severity of Postsurgical Hypertrophic Scars in a Randomized, Double-Blind, within-Subject, Placebo-Controlled Study. Plast. Reconstr. Surg. 2018, 142, 192E–201E. [Google Scholar] [CrossRef]
- Karmisholt, K.E.; Banzhaf, C.A.; Glud, M.; Yeung, K.; Paasch, U.; Nast, A.; Haedersdal, M. Laser Treatments in Early Wound Healing Improve Scar Appearance: A Randomized Split-Wound Trial with Nonablative Fractional Laser Exposures vs. Untreated Controls. Br. J. Dermatol. 2018, 179, 1307–1314. [Google Scholar] [CrossRef]
- Kong, X.; Yang, M.; Cao, Z.; Chen, J.; Chai, W.; Wang, Y. Tissue Adhesive for Wound Closure in Enhanced-Recovery Total Hip Arthroplasty: A Prospective, Randomized and Controlled Study. BMC Musculoskelet. Disord. 2020, 21, 178. [Google Scholar] [CrossRef]
- Lin, M.J.; Dubin, D.P.; Torbeck, R.L.; Bernstein, D.M.; Nabatian, A.; Dolan, C.K.; Bacigalupi, R.; Zade, J.; Zheng, Z.; Desman, G.; et al. Early Fractional Ablative Laser for Skin Cancer Excision Scars: A Randomized Split-Scar Study. Dermatol. Surg. 2023, 49, 338–342. [Google Scholar] [CrossRef]
- Musham, A.; Samuel, E.M.K.; Sahoo, A.K.; Elamurugan, T.P.; Manwar, A.S. Comparison of Tissue Adhesive Glue with Subcuticular Absorbable Suture for Skin Closure Following Thyroid Surgery A Single-Blinded Randomised Controlled Trial. Sultan Qaboos Univ. Med. J. 2023, 23, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Pangkanon, W.; Yenbutra, P.; Kamanamool, N.; Tannirandorn, A.; Udompataikul, M. A Comparison of the Efficacy of Silicone Gel Containing Onion Extract and Aloe Vera to Silicone Gel Sheets to Prevent Postoperative Hypertrophic Scars and Keloids. J. Cosmet. Dermatol. 2021, 20, 1146–1153. [Google Scholar] [CrossRef]
- Phillips, T.J.; Fung, E.; Rigby, M.H.; Burke, E.; Hart, R.D.; Trites, J.R.B.; Gassner, H.G.; Taylor, S.M. The Use of Botulinum Toxin Type A in the Healing of Thyroidectomy Wounds. Plast. Reconstr. Surg. 2019, 143, 375e–381e. [Google Scholar] [CrossRef] [PubMed]
- Ramos, R.M.; Burland, M.; Silva, J.B.; Burman, L.M.; Gelain, M.S.; Debom, L.M.; Bec, J.M.; Alirezai, M.; Uebel, C.O.; Valmier, J.; et al. Photobiomodulation Improved the First Stages of Wound Healing Process After Abdominoplasty: An Experimental, Double-Blinded, Non-Randomized Clinical Trial. Aesthet. Plast. Surg. 2019, 43, 147–154. [Google Scholar] [CrossRef]
- Safra, T.; Shehadeh, W.; Koren, A.; Salameh, F.; Friedman, O.; Sprecher, E.; Artzi, O. Early Intervention with Pulse Dye and CO2 Ablative Fractional Lasers to Improve Cutaneous Scarring Post-Lumpectomy: A Randomized Controlled Trial on the Impact of Intervention on Final Cosmesis. Lasers Med. Sci. 2019, 34, 1881–1887. [Google Scholar] [CrossRef]
- Surakunprapha, P.; Winaikosol, K.; Chowchuen, B.; Jenwithaesuk, K.; Jenwitheesuk, K. Adding Herbal Extracts to Silicone Gel on Post-Sternotomy Scar: A Prospective Randomised Double-Blind Study. J. Wound Care 2020, 29, S36–S42. [Google Scholar] [CrossRef] [PubMed]
- Suwannaphisit, S.; Aonsong, W.; Suwanno, P.; Yuenyongviwat, V. Comparing the Running Subcuticular Technique versus the Donati Technique in Open Carpal Tunnel Release: A Randomized Controlled Trial. J. Orthop. Surg. Res. 2021, 16, 565. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, F.W.; Mokken, S.E.; Smit, J.M.; Bouman, M.B.; van de Grift, T.C.; Mullender, M.G.; Middelkoop, E. The Impact of Incisional Negative Pressure Wound Therapy on Scar Quality and Patient-Reported Outcomes: A within-Patient-Controlled, Randomised Trial. Wound Repair Regen. 2022, 30, 210–221. [Google Scholar] [CrossRef]
- Zhang, S.; Nabi, O.; Jiang, X. New Strategy of Modulating Incision Tension: A Wound Tension Offloading Device Applied before Surgery. Dermatol. Ther. 2021, 34, e14797. [Google Scholar] [CrossRef] [PubMed]
- Zoumalan, C.I.; Tadayon, S.C.; Roostaeian, J.; Rossi, A.M.; Gabriel, A. Safety and Efficacy of a Scar Cream Consisting of Highly Selective Growth Factors within a Silicone Cream Matrix: A Double-Blinded, Randomized, Multicenter Study. Aesthet. Surg. J. 2019, 39, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Kemaloğlu, C.A.; Özyazgan, İ.; Gönen, Z.B. Immediate Fat and Nanofat-Enriched Fat Grafting in Breast Reduction for Scar Management. J. Plast. Surg. Hand Surg. 2021, 55, 173–180. [Google Scholar] [CrossRef]
- Yuan, X.; Shen, J.; Chen, L.; Wang, L.; Yan, Q.; Zhang, J. Onion Extract Gel Is Not Better than Other Topical Treatments in Scar Management: A Meta-Analysis from Randomised Controlled Trails. Int. Wound J. 2021, 18, 396–409. [Google Scholar] [CrossRef]
- Januszyk, M.; Wong, V.W.; Bhatt, K.A.; Vial, I.N.; Paterno, J.; Longaker, M.T.; Gurtner, G.C. Mechanical Offloading of Incisional Wounds Is Associated with Transcriptional Downregulation of Inflammatory Pathways in a Large Animal Model. Organogenesis 2014, 10, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Wong, V.W.; Beasley, B.; Zepeda, J.; Dauskardt, R.H.; Yock, P.G.; Longaker, M.T.; Gurtner, G.C. A Mechanomodulatory Device to Minimize Incisional Scar Formation. Adv. Wound Care 2013, 2, 185–194. [Google Scholar] [CrossRef]
- Wang, D.; Ding, J.; Jiang, Y.; Liu, Y.; Chen, B. Continuous Tension Reduction Technique in Facial Scar Management: A Comparison of W-Plasty and Straight-Line Closure on Aesthetic Effects in Asian Patients. Int. Wound J. 2022, 19, 1064–1070. [Google Scholar] [CrossRef] [PubMed]
- Scalise, A.; Calamita, R.; Tartaglione, C.; Pierangeli, M.; Bolletta, E.; Gioacchini, M.; Gesuita, R.; Di Benedetto, G. Improving Wound Healing and Preventing Surgical Site Complications of Closed Surgical Incisions: A Possible Role of Incisional Negative Pressure Wound Therapy. A Systematic Review of the Literature. Int. Wound J. 2016, 13, 1260–1281. [Google Scholar] [CrossRef] [PubMed]
- Armour, A.; Scott, P.G.; Tredget, E.E. Cellular and Molecular Pathology of HTS: Basis for Treatment. Wound Repair Regen. 2007, 15, S6–S17. [Google Scholar] [CrossRef]
- Klotz, T. The Effect of Moisturisers on Scars: A Systematic Review. Ph.D. Thesis, University of Adelaide, Adelaide, Australia, 2018. [Google Scholar]
- Cheng, J.W.; Cheng, S.W.; Wei, R.L.; Lu, G.C. Anti-Vascular Endothelial Growth Factor for Control of Wound Healing in Glaucoma Surgery. Cochrane Database Syst. Rev. 2016, 2016, CD009782. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, T.; He, J.; Dong, J. Growth Factor Therapy in Patients with Partial-Thickness Burns: A Systematic Review and Meta-Analysis. Int. Wound J. 2016, 13, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Santamato, A.; Cinone, N.; Panza, F.; Letizia, S.; Santoro, L.; Lozupone, M.; Daniele, A.; Picelli, A.; Baricich, A.; Intiso, D.; et al. Botulinum Toxin Type A for the Treatment of Lower Limb Spasticity after Stroke. Drugs 2019, 79, 143–160. [Google Scholar] [CrossRef] [PubMed]
- Picelli, A.; Santamato, A.; Chemello, E.; Cinone, N.; Cisari, C.; Gandolfi, M.; Ranieri, M.; Smania, N.; Baricich, A. Adjuvant Treatments Associated with Botulinum Toxin Injection for Managing Spasticity: An Overview of the Literature. Ann. Phys. Rehabil. Med. 2019, 62, 291–296. [Google Scholar] [CrossRef]
- Park, J.H.; Park, H.J. Botulinum Toxin for the Treatment of Neuropathic Pain. Toxins 2017, 9, 260. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.S.; Lee, B.H.; Sung, H.M.; Park, S.Y.; Ahn, D.K.; Jung, M.S.; Suh, I.S. Effect of Botulinum Toxin Type A on Differentiation of Fibroblasts Derived from Scar Tissue. Plast. Reconstr. Surg. 2015, 136, 171e–178e. [Google Scholar] [CrossRef] [PubMed]
- Scala, J.; Vojvodic, A.; Vojvodic, P.; Vlaskovic-Jovicevic, T.; Peric-Hajzler, Z.; Matovic, D.; Dimitrijevic, S.; Vojvodic, J.; Sijan, G.; Stepic, N.; et al. Botulin Toxin Use in Scars/Keloids Treatment. Open Access Maced. J. Med. Sci. 2019, 7, 2979–2981. [Google Scholar] [CrossRef]
- Arias-Rodríguez, C. Toxina Botulínica: Usos Novedosos y Su Evidencia Botulinum Toxin: Novel Uses and Evidence. Dermatol. Cosmét. Med. Quir. 2022, 20, 448–452. [Google Scholar]
- Ashraf, I.; Butt, E.; Veitch, D.; Wernham, A. Dermatological Surgery: An Update on Suture Materials and Techniques: Part 1. Clin. Exp. Dermatol. 2021, 46, 1400–1410. [Google Scholar] [CrossRef]
- Butt, E.; Ashraf, I.; Veitch, D.; Wernham, A. Dermatological Surgery: An Update on Suture Materials and Techniques: Part 2. Clin. Exp. Dermatol. 2021, 46, 1411–1419. [Google Scholar] [CrossRef]
- Byrne, M.; Aly, A. The Surgical Suture. Aesthet. Surg. J. 2019, 39, S67–S72. [Google Scholar] [CrossRef]
- Chowdhury, B.; Kassir, M.; Salas-Alanis, J.; Nistico, S.; Galadari, H.; Fritz, K.; Salavastru, C.; Blicharz, L.; Goldust, M. Laser in Surgical Scar Clearance: An Update Review. J. Cosmet. Dermatol. 2021, 20, 3808–3811. [Google Scholar] [CrossRef] [PubMed]
| Population | Surgical Procedures Population |
| Intervention | Recent approaches of cicatrization process |
| Outcomes | Quality of scars |
| Research question | What are the current approaches to improve the quality of scars in surgical procedures population? |
| Database | Search Strategy |
|---|---|
| PubMed | ((“cicatrix” [MeSH Terms] OR “cicatrix” [All Fields] OR “cicatrization” [All Fields] OR “cicatrize” [All Fields] OR “cicatrized” [All Fields] OR “cicatrizing” [All Fields]) AND “Cicatrix, Hypertrophic” [Mesh] AND (prevention) AND (treatment) AND (surgery [Title/Abstract]) OR (surgical scar [Title/Abstract]) NOT (burn* [Title/Abstract])) |
| Web of Science | ((((AB=(cicatrization)) OR AB=(“hypertrophic cicatrix” OR “surgical scar”)) AND AB=(treatment OR therapy)) AND AB=(surgery)) NOT ALL=(conjunctivitis OR burn) |
| Scopus | TITLE-ABS-KEY (cicatrization) AND TITLE-ABS-KEY (treatment OR therapy) AND TITLE-ABS-KEY (surgery) OR TITLE-ABS-KEY (surgical AND scar) |
| ScienceDirect | (cicatrization) OR (hypertrophic cicatrix AND prevention) AND (treatment) AND (“surgery” OR “surgical scar” OR) NOT (conjunctivitis OR burn) |
| Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Q9 | Q10 | Q11 | Score | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abedini et al., 2020 [22] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | 11 |
| Chen Z et al., 2022 [23] | Y | Y | Y | Y | N | N | N | Y | Y | Y | Y | 8 |
| Chen et al., 2021 [24] | Y | Y | Y | N | Y | Y | Y | Y | Y | Y | N | 9 |
| Chung et al., 2021 [25] | Y | Y | Y | Y | N | Y | Y | Y | Y | Y | Y | 10 |
| Dolynchuk et al., 2020 [26] | Y | Y | Y | N | Y | Y | Y | N | Y | Y | Y | 9 |
| Huang et al., 2019 [27] | Y | Y | Y | Y | Y | Y | Y | N | Y | Y | Y | 10 |
| Ilori et al., 2022 [28] | Y | Y | Y | Y | N | N | N | Y | Y | Y | Y | 8 |
| Jensen et al., 2018 [29] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | 11 |
| Karmisholt et al., 2018 [30] | Y | Y | Y | Y | N | N | Y | Y | Y | Y | Y | 9 |
| Kong et al., 2020 [31] | Y | Y | Y | Y | N | Y | N | Y | Y | Y | Y | 9 |
| Lin et al., 2023 [32] | Y | Y | N | Y | N | N | Y | Y | Y | Y | Y | 8 |
| Musham et al., 2023 [33] | Y | Y | Y | Y | N | N | Y | Y | Y | Y | Y | 9 |
| Pangkanon et al., 2021 [34] | Y | Y | Y | Y | N | N | Y | Y | Y | Y | Y | 9 |
| Phillips et al., 2018 [35] | Y | Y | Y | N | Y | N | Y | N | Y | Y | Y | 8 |
| Ramos et al., 2019 [36] | Y | N | Y | Y | Y | N | Y | Y | Y | Y | Y | 9 |
| Safra et al., 2019 [37] | Y | N | Y | Y | N | N | Y | Y | Y | Y | Y | 8 |
| Surakunprapha et al., 2020 [38] | Y | Y | Y | Y | Y | N | Y | Y | Y | N | Y | 9 |
| Suwannaphisit et al., 2021 [39] | Y | Y | Y | N | Y | N | Y | Y | Y | Y | Y | 9 |
| Timmermans et al., 2022 [40] | Y | Y | Y | Y | N | N | Y | Y | Y | Y | Y | 9 |
| Zhang et al., 2021 [41] | Y | Y | Y | Y | N | N | Y | Y | Y | Y | N | 8 |
| Zoumalan et al., 2019 [42] | Y | Y | Y | N | Y | N | Y | N | Y | Y | Y | 8 |
| Study/Author | Typology/Main Objective | Participants | Interventions | Evaluation | Main Findings | PEDro |
|---|---|---|---|---|---|---|
| Abedini et al., 2020 [22] | Randomized, split-scar, double- blinded, prospective, controlled trial. To investigate the role of botulinum toxin on the prevention of scar formation in comparison with control in cosmetic plastic surgeries including mammoplasty and abdominoplasty. | n = 19 Patients of mammoplasty and abdominoplasty. 26–54 years old. Sex (f/m): 19/0 | Botulinum toxin (EG) or saline solution (CG) on each side of scar. | mSBSES Assessments at 3 and 6 months | Significant improvements in EG compared to CG at 3 and 6 months (p < 0.001 each). Also, in the subset analysis, there were significant differences between groups in width, height, color, and scar visibility at months 3 and 6. In EG, scores of mSBSES, height, visibility, and redness (p < 0.001; p = 0.002; p = 0.002; p = 0.008) increased significantly from month 3 to 6, but the scar width did not change significantly (p = 0.051). In CG scores of mSBSES, height, and redness (p = 0.0015; p = 0.038; p = 0.019) improved significantly over time, but scar width and visibility (p = 0.34; p = 0.24) did not change significantly. | 11 |
| Chen et al., 2022 [23] | Randomized, controlled, prospective, rated blinded trial. To determinate efficacy and safety of tissue adhesive zippers in post-surgical scar prevention among patients undergoing surgical excision of the face. | n = 53 Patients with surgical excision on the face. <14 years old. Sex (f/m): 30/23 | EG participants used a tissue adhesive zipper for three months. CG no intervention. | Scar Width. POSAS Assessments at 1, 3, 6, 12 months. | In EG group the scar width was significantly smaller than CG (p = 0.0025) at 12 months. EG and CG differed significantly in POSAS scores for Scar irregularity (p = 0.0145). No differences between groups in the other scores and observer score. | 8 |
| Chen et al., 2021 [24] | Randomized, prospective, double-blind, split-scar trial. To investigate the effect of different doses of botulinum toxin administered early after surgery on scar improvement through a split-scar experiment. | n = 22 Tumor resection (tumor did not invade the muscle) 18–52 years old. Sex (f/m): 9/11 | High and low doses of botulinum toxin into each half of the surgical wound closure. | mSBSES. VAS. Assessment at 6 months | The high-dose sides had significantly better mSBSES score compared with low-dose in terms of width (p < 0.01), incision visibility line (p < 0.01). No significant differences between groups in height and color. High-dose sides had significantly higher VAS scores than low-dose sides (p < 0.01). | 9 |
| Chung et al. 2021 [25] | Randomized, blinded, prospective trial. To compare the scar quality when different protocols were applied, and eventually aim to find the optimal scar management protocol | N = 126 Patients undergoing thyroidectomy. >18 years old Sex (f/m): 105/21 | Tissue adhesive (group A), or subcuticular suturing and early NAFL (group B), or skin closure with tissue adhesive and early NAFL (group C) | POSAS. Assessment at 6 months. | No significant differences between groups at baseline. At 6 months, B group showed a narrower width scar, with no differences between A and C (p > 0.017). According to the patients, groups B and C showed significantly higher satisfaction in all sub-scales than A except for the pigmentation. According to physicians, B showed better thickness, relief, pliability, surface area, and overall cosmesis (p < 0.017) with no differences between A and C (p > 0.017). | 10 |
| Dolynchuk et al., 2020 [26] | Randomized, prospective, double-blind trial. To analyze the biochemical and clinical effects of 1,4-Diaminobutane (DAB)on prevention of human hypertrophic scars. | Total n = 78 patients of breast reduction. Biochemical evaluation n = 30 Clinical evaluation n = 48 5–53 years old. Sex (f/m): 50/10 | Topical 1,4-DAB or control treatment on each side (EG or CG respectively). | Biochemical: Analytical assessment (biopsy) At 2 months Clinical: Durometer test POSAS At 6 and 12 weeks | The biopsies registered more 1,4DAB in treated scars than control group (p < 0.05). Durometer test was significantly better (p < 0.05) in EG. POSAS score was significantly better in EG than CG (p < 0.05) | 9 |
| Huang et al., 2019 [27] | Randomized, prospective, double blind, split-face trial. To investigate the safety and efficacy of early botulinum toxin A injection in preventing hypertrophic scarring in the medial cantal area after epicanthoplasty. | n = 43 Patients of epicanthoplasty. 18–45 years old. Sex (f/m): 43/0 | Botulinum toxin A (EG) or saline treatment (placebo side) into each side of surgery. | VSS VAS. Patient Satisfaction Assessment al 1, 3, 6 months | 13 patients were lost to follow-up. The botulinum toxin A side had significantly better scores at VSS 1 month (p = 0.034), 3 months (p < 0.001), and 6 months (p < 0.001) after administration. The same was found at VAS (p = 0.017; p < 0.001; p = 0.032 respectively). Patient satisfaction was better with botulinum toxin A (p < 0.001) | 10 |
| Ilori et al., 2022 [28] | Randomized, controlled, prospective trial. To determinate the efficacy of microporous tape in the prevention of abnormal post-surgical scars. | n = 72 patients with 92 scars EG n = 36 CG n = 36 Benign tumors excision, open reduction fixation of fractures, osteotomies, arthroplasties. 15–65 years old. Sex (f/m): 25/38 | Microporous tape directly over the scar for 6 months (EG) or standard care. | POSAS Scar types (normal, hypertrophic, or atrophic). Assessments at 6 weeks, 3 months, and 6 months. | No significant differences between groups at the baseline. At 6 months, the scar height and width were significantly better in EG than CG (p < 0.0001 each), and scar types were significantly better in EG than CG (p = < 0.0001). | 8 |
| Jensen et al., 2018 [29] | Randomized, double-blinded, within-subject, placebo-controlled, prospective trial. To determinate the effect of anti-CTFG (EXC001) on the severity of surgical scars. | n = 23 Mammoplasty. Bilateral, symmetric hypertrophic scars of the breast. 28–55 years old. Sex (f/m): 23/0 | EXC001 (EG) or placebo (CG) injected intradermally at post-surgery weeks 2, 5, 8, and 11 | POSAS Assessment at 12 and 24 weeks. | At 24 weeks, EG reduced scar severity significantly compared to CG, by physician (vascularity p < 0.001; pigmentation p < 0.001; thickness p = 0.001; relief p < 0.001; Pliability p = 0.005; surface area p < 0.001; overall opinion p < 0.001) and subjects (pigmentation p = 0.01; stiffness p = 0.003; thickness p = 0.005; surface area p = 0.032 and overall opinion p = 0.003) | 11 |
| Karmisholt et al., 2018 [30] | Randomized split-wound, prospective trial. To assess scar formation clinically after three nonablative fractional laser (NAFL) exposures, targeting the inflammation, proliferation, and remodeling wound healing phases in patients vs. untreated controls. | n = 32 Patients undergoing surgical excision. >18 years old Sex (f/m): 15/17 | 1540-nm NAFL (3 exposures: before surgery, at suture removal and 6 weeks after surgery) (EG) or no laser treatment on each side of the scar (CG). | POSAS VSS Assessment at 3 months | 30 patients completed follow-up. At 3 months, EG improved scores compared with the control in POSAS, vascularity, relief, pliability, surface area, and overall opinion (p < 0.001; p = 0.005; p = 0.023; p = 0.037; p = 0.016; p = 0.003), but pigmentation and thickness did not (p = 0.13 each). The greater improvements were located in thorax area (p < 0.001). Patients older and younger than 50 years answered similarly (p = 0.015; p = 0.008) | 9 |
| Kong et al., 2020 [31] | Randomized, prospective, controlled trial. To present the experience of adopting tissue adhesive as adjunct to standard wound closure in total hip arthroplasty and evaluate its role and cost performance. | n = 30 Patients with bilateral total hip arthroplasty. 18–60 years old. Sex (f/m): 13/17 | Standard wound closure (GC) or additional tissue adhesive (EG). | PSAS HWES VSS Assessment at 1 month | PSAS showed that, from the view of patients, hips with tissue adhesive were significantly better than sutured hips (p = 0.004). Most patients preferred the tissue adhesive. From view of evaluators, there were not significant differences between groups in HWES or VSS. | 9 |
| Lin et al., 2023 [32] | Randomized, prospective, single blinded, split-scar trial. To compare surgical scars treated with fractional carbon dioxide (CO2) laser performed on Day 0 and Day 14. | n = 30 Patients of skin cancer excision on limbs. 34–82 years old Sex (f/m): 13/17 | 2 passes of CO2 laser before the cutaneous suture (day 0) or when sutures were removed (day 14) on each side. | MSS Analytical assessment (biopsy) Assessment at 6 months | 26 subjects completed follow-up. There were no differences between groups for patients (p = 0.058) or physicians (p = 0.028). Fractal dimensions and lacunarity were similar (p = 0.80; p = 0.44). | 8 |
| Musham et al., 2023 [33] | Randomized, prospective, controlled, single-blind trial. To compare the skin closure time, postoperative pain, and the scar outcome between tissue adhesive and sub-cuticular sutures in thyroid surgery. | n = 124 Patients of thyroidectomy. 30–55 years old Sex (f/m): 96/28 | Subcuticular suture (CG) or tissue adhesive (EG) | VAS MSS Assessment at post operative, 1, and 3 months. | VAS showed better post operative results in EG (p < 0.01). However, there were not significant differences between groups in MSS at 1 and 3 months. | 9 |
| Pangkanon et al., 2021 [34] | Randomized, assessor-blind, prospective controlled trial. To compare the efficacy of silicone gel containing onion extract and aloe vera (SGOA) to silicone gel sheets (SGS) to prevent postoperative hypertrophic scars and keloids. | n = 40 Patients underwent surgery. 18–60 years old Sex (f/m): 36/4 | SGOA twice a day or SGS wound 24 h/day. | POSAS Mexameter Cutometer Assessments at 1, 2, and 3 months. | No differences between groups in number of hypertrophic scars (p = 0.465), melanin (p = 0.571), erythema (p = 0.863), and POSAS by physicians and patients. SGOA group had significantly greater pliability (p = 0.009). | 9 |
| Phillips et al., 2018 [35] | Randomized, double blind, prospective controlled trial. To assess the effects of botulinum toxin type A on scar formation after thyroid surgery | n = 40 Patients of total thyroidectomy, hemithyroidectomy, or parathyroidectomy. Sex (f/m): 36/4 | Botulinum toxin A (EG) or saline solution (CG) on each half of the scar. | POSAS VSS Preoperative evaluation. Assessments at 1, 6, and 12 months. | 23 patients completed the last follow-up. There were not significant differences between groups at 1, 6, and 12 months. However, at 6 months, better results were found in EG compared to CG in patients with poor cicatrization history in POSAS total score (p = 0.012), overall (p = 0.010), and VSS (p = 0.021) | 8 |
| Ramos et al., 2019 [36] | Non-randomized, double-blinded split-scar prospective trial. To evaluate the influence of photobiomodulation on the post abdominoplasty healing process. | n = 17 Patients of abdominoplasty 18–55 years old Sex (f/m): 17/0 | Right side of scars: 10 sessions of photobiomodulation (experimental side). Left side of scars: any treatment (used as control). | POSAS VSS Assessments at 1, 6, and 12 months. | The treated side of scars was significantly better after 1 and 6 months on VSS (p = 0.0065). The scores of POSAS were better on treated side for observers (p = 0.0034) and patients (p = 0.0047). | 9 |
| Safra et al., 2019 [37] | Randomized, controlled, single blinded, split-scar, prospective trial. To study the safety and efficacy of a combination of pulsed dye laser (PDL) and fractional ablative CO2 laser (FACL) for attenuation of post-lumpectomy scarring. | n = 18 Patients of lumpectomy. >18 years old Sex (f/m): 18/0 | Treated side received 3 sessions at 1-month intervals of PDL and FACL 6 weeks after suture removal. The other half of scars did not receive any treatment | POSAS Assessment at 6 months. | The improvements in scar parameters were significantly greater in treated side (overall, pigmentation, and relief p < 0.001; vascularity and pliability p = 0.001; thickness p = 0.002). | 8 |
| Surakunprapha et al., 2020 [38] | Randomized, controlled, double-blind, prospective trial. To determine whether adding herbal extracts to the gel would augment the healing effect. | n = 46 Patients of sternotomy 32–61 years old. Sex (f/m): 19/27 | EG: topical silicone gel plus herbal extract gel (Allium cepa, Centella Asiatica, Aloe vera and Paper Mulberry). CG: topical silicone gel | POSAS Assessments at 6 months | At 6 months, EG had significantly greater scores than baseline: vascularity (p = 0.013), pigmentation (p = 0.000), overall (p = 0.018). CG also had improvements in pigmentation (p = 0.000) and vascularity (p = 0.046). | 9 |
| Suwannaphisit et al., 2021 [39] | Randomized, prospective controlled trial. To compare the Donati suture technique and running subcuticular technique in terms of surgical scar, pain, and functional outcome. | n = 142 Patients of open carpal tunel release. 48–70 years old. Sex (f/m): 120/18 | Donati or running subcuticular technique. | POSAS Pain (verbal numerical rating score) Assessments at 2, 6, and 12 weeks | At 2 weeks, POSAS showed that subcuticular running technique had lower scores than Donati by the patients (p < 0.05) but not by physicians (p = 0.15). At 6 and 12 weeks, there was no difference between groups in any parameter. | 9 |
| Timmermans et al., 2022 [40] | Randomized, prospective, within- subject controlled trial. To establish if incisional negative pressure wound results in improved scar outcomes in comparison to the standard of care. | n = 85 Transgender men undergoing gender-affirming mastectomies. 18–63 years old Sex (f/m): 0/85 | Incisional negative pressure wound (EG) or standard care (CG) on each side. | Cutometer. POSAS Assessments at 1, 3, and 12 months. | 80 patients completed follow-up. At 12 months, there were not significant differences between groups for Cutometer subdomains (p > 0.05). Significant improvements were found in EG compared to CG at 3 months in vascularity, POSAS total score, and overall cosmesis (p = 0.022; p = 0.003; p = 0.004), but these differences were not found at 12 months. By patients, at 1 and 3 months, thickness was better in EG (p = 0.027; p = 0.042), and at 12 months color, pliability, thickness, total score, and overall opinion were better in EG (p = 0.003; p < 0.001; p = 0.003; p = 0.039; p = 0.008). | 9 |
| Zhang et al., 2021 [41] | Randomized, single blind, prospective controlled trial. To determine whether the application of a tension offloading device preoperatively would result in superior attenuation of scar genesis in comparison to traditional methods. | n = 12 Patients with a history of hypertrophic scar formation, who underwent surgical excision of benign cutaneous lesions located over buttocks and truncal region. 11–33 years old Sex (f/m): 8/4 | Application of device before (2 weeks) and after surgery (for 3 months, starting from the suture removal) (EG1); application after surgery (for 3 months) (EG2); or no tension offloading (CG) | Skin elasticity coefficient Assessment Before surgery POSAS Assessment at 6 months | The median skin elasticity coefficient was 27.5% in the pre-op group in comparison to 15% in both the post-op group and the control group (p = 0.0286). EG1 and EG2 did not show significant differences. There were differences compared to CG in terms of width and color. Overall Score was significantly better between 3 groups by patients but only between offloaded groups and CG by physicians. | 8 |
| Zoumalan et al., 2019 [42] | Randomized, double-blinded, multicenter, prospective trial. To compare the efficacy and safety of a scar cream consisting on highly selective human growth factors (SKN2017B) and hyaluronic acid within a silicone matrix. | n = 45 (49 bilateral and 12 unilateral scars) Surgical patients with unilateral or bilateral scars on their face. >18 years old. Sex (f/m): 43/2 | SKN2017B or silicone cream twice for 3 months. Unilateral scars were randomly assigned in split-scar and bilateral scars were assigned by sides. | VSS Assessment at 12 weeks. (Independent Assessment) | Investigators rated 74% improvements in SKN2017B group and 54% in silicone group (p < 0.0001), patients rated 85% and 51% improvements respectively (p < 0.001). Independent reviewers rated 87% and 1% (p < 0.0001). | 8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bueno, A.; Nevado-Sanchez, E.; Pardo-Hernández, R.; de la Fuente-Anuncibay, R.; González-Bernal, J.J. Treatment and Improvement of Healing after Surgical Intervention. Healthcare 2023, 11, 2213. https://doi.org/10.3390/healthcare11152213
Bueno A, Nevado-Sanchez E, Pardo-Hernández R, de la Fuente-Anuncibay R, González-Bernal JJ. Treatment and Improvement of Healing after Surgical Intervention. Healthcare. 2023; 11(15):2213. https://doi.org/10.3390/healthcare11152213
Chicago/Turabian StyleBueno, Andrea, Endika Nevado-Sanchez, Rocío Pardo-Hernández, Raquel de la Fuente-Anuncibay, and Jerónimo J. González-Bernal. 2023. "Treatment and Improvement of Healing after Surgical Intervention" Healthcare 11, no. 15: 2213. https://doi.org/10.3390/healthcare11152213
APA StyleBueno, A., Nevado-Sanchez, E., Pardo-Hernández, R., de la Fuente-Anuncibay, R., & González-Bernal, J. J. (2023). Treatment and Improvement of Healing after Surgical Intervention. Healthcare, 11(15), 2213. https://doi.org/10.3390/healthcare11152213







