Abstract
With the appearance of new viruses and infectious diseases (ID) such as COVID-19 in 2019, as well as the lack of specific pharmacological tools for the management of patients with severe complications or comorbidities, it is important to search for adjuvant treatments that help improve the prognosis of infectious disease patients. It is also important that these treatments limit the oxidative and hyperinflammatory damage caused as a response to pathogenic agents, since, in some cases, an inflammatory syndrome may develop that worsens the patient’s prognosis. The potential benefits of complementary nutrients and dietary interventions in the treatment of pathological processes in which oxidative stress and inflammation play a fundamental role have been widely evaluated. Coenzyme Q10 (CoQ10) is a supplement that has been shown to protect cells and be effective in cardiovascular diseases and obesity. Additionally, some studies have proposed it as a possible adjuvant treatment in viral infections. Preclinical and clinical studies have shown that CoQ10 has anti-inflammatory and antioxidant effects, and effects on mitochondrial dysfunction, which have been linked to the inflammatory response.
1. Introduction
In 1957, a substance was discovered with a structure containing a benzoquinone ring attached to a chain of 10 isoprene units: coenzyme Q10 (CoQ10) [1]. Coenzyme Q10 (CoQ10) is an essential compound in the human body that is synthesized in the inner mitochondrial membrane [1], with a lipophilic character that makes it easy to diffuse through membranes [2]. Like other compounds, CoQ10 has been considered as a possible candidate in the adjunctive treatment of chronic diseases in which oxidative stress (OS) plays an important role in the pathophysiology [3]. The search for nutrients or compounds with antioxidant, immunomodulatory and anti-inflammatory capabilities and evidence for their properties is of great interest in the scientific community, especially for diseases where an unhealthy diet influences their prevention, as is the case for infectious diseases (ID) [4]. ID have become more prevalent in recent decades; causes of this increase include environmental changes due to human activity, increased international mobility, and microbial adaptations and resistance [5]. ID are generally caused by microorganisms. Their clinical importance depends on the damage they cause to the host. Damage to tissues and organs after infection results mainly from the growth and metabolic processes of infectious agents intracellularly or within body fluids, with the production and release of toxins or enzymes that interfere with normal organ functions and/or or systems. Endogenous mechanisms can also contribute to increased damage, including the exacerbated inflammatory response of the host [6]. Depending on the causal agent of disease, the ideal treatment comprises antimicrobials, a large group of compounds with diverse structures and mechanisms of action that interfere with the growth of bacteria, viruses, fungi and parasites. Depending on their use, they are called antibiotics, antivirals, antifungals, antimycotics or antiparasitics [5]. Unfortunately, the number of antimicrobial agents in clinical development is woefully insufficient to keep pace with growing antibiotic resistance [7]. In addition to the use of antimicrobial drugs for the treatment of infectious diseases, nutritional status and diet modulate processes such as inflammation, immune function and oxidative status, which can favour the patient’s prognosis [4,8]. Therefore, the objective of this review is to present scientific evidence from clinical data that demonstrates the beneficial effects of CoQ10 on ID, evaluating its immunomodulatory, anti-inflammatory and antioxidant effects.
2. Data Source
An exhaustive compilation of the relevant literature on the effects of coenzyme Q10 supplementation in infectious diseases was carried out through an electronic search of the PubMed (www.pubmed.ncbi.nlm.nih.gov/, accessed on 8 January 2022), Web of Science (www.webofknowledge.com, accessed on 15 January 2022) and Scopus (www.scopus.com, accessed on 20 January 2022) databases to retrieve studies published up to December 2021. The following search criteria was used: (CoQ10 OR Ubiquinone OR ubiquinol) AND (viral infections OR infectious disease OR bacterial infections) AND (Antioxidants OR Anti-inflammatory effects). Additionally, other relevant references of identified studies were retrieved by cross-references. The research had no geographic area or language restrictions.
3. Coenzyme Q10
CoQ10 is a fundamental element in metabolic processes such as mitochondrial oxidative phosphorylation. It is also considered a powerful fat-soluble antioxidant. CoQ10 is present in all cell membranes and is biosynthesized in all tissues from its precursor, the 4-hydroxybenzoate ring (4HB), a derivative of tyrosine. The isoprene chain is synthesized through the mevalonate pathway, a common pathway for cholesterol synthesis [9,10]. CoQ10 (also known as ubiquinone, because it is ubiquitous) exists in its oxidized (ubiquinone) or reduced form (ubiquinol), the latter being the most predominant in the human body [11]. Although CoQ10 has similar biological functions to vitamins, it differs in that it is biosynthesized in the body and vitamins are obtained only from the diet. About half of the body’s CoQ10 is obtained through the diet and the rest is biosynthesized endogenously; major dietary sources of CoQ10 include meats, fish, salmon, sardines, pork, chicken, nuts, soybeans, vegetable oils, there are other sources that also contain CoQ10 although at much lower levels, dairy products, vegetables, fruits and cereals [12]. The daily requirement of CoQ10, both from endogenous biosynthesis and from dietary sources, is approximately 500 mg [13]. There are no established minimum or maximum effective doses; consumption of 200 mg twice a day is required to achieve a therapeutic blood level of >2.5 µg/mL, although the doses used have varied according to the type of disease [14]. Regarding its modulatory effect on inflammation, supplementation with 100–300 mg/day for 2–3 months reduced pro-inflammatory parameters [15]. There are several processes in which OS plays a fundamental part, including as aging, neuro- 69 degenerative illnesses, diabetes, and autoimmune disorders, where a decreasing level of CoQ10 is evident, and supplementation with CoQ10 has considerable health advantages [16,17,18,19,20].
Experimental data from preclinical and clinical safety studies have shown that CoQ10 does not cause adverse effects in humans and is safe for use as a dietary supplement. Indeed, acute toxicity studies in rodents have shown that the maximum tolerated CoQ10 doses are >4000 mg/kg. In a subacute toxicity study of CoQ10 in rats, doses of 0, 40, 200 or 1000 mg/kg/day were administered orally for 5 weeks, with no signs of toxicity. Chronic toxicity studies have also shown that CoQ10 administration is quite safe: none of the doses produced signs of toxicity based on haematological analysis, biochemical tests on blood or urine samples and post-mortem examinations. CoQ10 was well tolerated at doses up to 600 mg/kg/day by male and female rats [21].
4. Mechanism of Coenzyme Q10 Action Associated Antioxidant and Anti-Inflammatory Activity
During the infectious process, there could be an imbalance between oxidative and antioxidant species. As part of the body’s response to attack pathogens, the expression of myeloperoxidase, NADPH oxidase and nitric oxide synthase (iNOS) is induced in phagocytic cells, leading to increased production of ROS and RNS. In infection there is abundant iNOS activity in macrophages and leukocytes leading to increased production of nitric oxide (NO), which in the presence of superoxide reacts to form highly toxic peroxynitrite. The oxidants generated activate the redox-sensitive NF-κB signalling pathway, which promotes the expression of proinflammatory cytokines, chemokines and cell adhesion receptors, all of which are involved in the radical production and persistence of inflammation [22].
4.1. Antioxidant Functions
The most important function of CoQ10 is as a carrier in the mitochondrial electron transport chain. CoQ10 is responsible for the transport of electrons from complex I (NADH dehydrogenase) to complex II (succinate dehydrogenase); up to this point, CoQ10 is in a permanent balance between a reduced form after receiving two electrons: Ubiquinol, and an oxidized form, Ubiquinone. In the respiratory chain, this redox cycle occurs by a two-step transfer of one electron each, producing the semiquinone intermediate [20]. Likewise, CoQ10 transfers protons to the mitochondrial intermembrane space, favouring the formation of a proton gradient capable of generating the energy to produce adenosine triphosphate (ATP); this means that it has important functions in the supply of cellular energy [23,24].
Due to these electron transport properties, its function as an antioxidant is of great relevance. It has been shown to protect cell membranes from lipoperoxidation caused by free radicals (FR), and being present in all tissues, its function is very important [15]. It is assumed that the reduced form of CoQ10, ubiquinol, is the active antioxidant agent involved in most of its functions. CoQ10 is converted to reduced form of coenzyme Q10 (CoQ10H2) with electrons provided by other redox reactions in the mitochondria, it is reduced by mitochondrial glycerol 3-phosphate dehydrogenase, mitochondrial dihydroorotate dehydrogenase, and electron-carrying flavoprotein dehydrogenases (ETFDHs) [24]. For this reason, the CoQ10 redox cycle takes place in cell membranes, in which different oxidoreductases such as cytochrome b5 reductase (CytB5) and NAD(P)H quinone dehydrogenase 1 (NQO1) participate. This enables the recovery of the active form of CoQ10, and in turn, ubiquinol interacts with α-tocopherol and vitamin C radicals, decreasing OS [25]. Reducing compounds such as ubiquinol can regenerate alpha-tocopherol from the tocopherol radical. This recycling is dependent on the activity of mitochondrial succinate dehydrogenase, which provides the reducing equivalents for the conversion of ubiquinone to ubiquinol. Microsomal NADPH-dependent reduction of the phenoxyl radical in vitamin E achieves maximum efficiency in the presence of CoQ10 [26].
4.2. Anti-Inflammatory and Immune Functions
The antioxidant properties of CoQ10 can protect all cells and tissues, especially cells involved in the innate and adaptive immune response. OS play an important role in immunological cytotoxicity against pathogens, through the production of reactive oxygen species (ROS) by macrophages [27,28]. Research suggests that CoQ10 mediates its beneficial effects through direct and indirect anti-inflammatory mechanisms; CoQ10 has been reported to regulate the gene expression of Interleukin (IL) 1 IL-1 and tumour necrosis factor α (TNF-α) in patients with diabetic nephropathy who received supplementation [29]. There are also studies that support the idea that CoQ10 exerts anti-apoptotic and anti-inflammatory activities, through redox-dependent mechanisms, since it has been shown that supplementation with CoQ10 decreases plasma levels of C-Reactive Protein (CRP), IL-6 and TNF-α [30]. Although the specific mechanism of action of CoQ10 as an anti-inflammatory is not clear, several potential mechanisms could explain it. CoQ10 could play a role in reducing the production of pro-inflammatory cytokines by inhibiting the expression of the gene encoding nuclear factor kappa B (NF-Κb), lipoperoxides present in pathogens and oxidizing agents generated during the infectious process induce the signalling pathway in monocytes that activates the NF-kB factor, resulting in the release of TNF-alpha being favoured. The antioxidant power of CoQ10 can reduce the signalling pathway due to its radical scavenging activity [31]. Other studies suggest that the anti-inflammatory effect of CoQ10 may be associated with adiponectin; by causing an increase in CoQ10 levels, supplementation leads to a rise in adiponectin levels, which then leads to a decrease in the inflammatory response mediated by TNF-α [32]. A randomized placebo-controlled study in which selenium supplementation combined with CoQ10 was administered to healthy elderly subjects with low selenium levels showed that this supplementation reduces the inflammatory response through a decrease in plasma CRP levels [33]. Likewise, another study also evaluated the effect of administration of CoQ10 in elderly subjects on inflammatory response markers such as osteopontin, osteoprotegerin, tumour necrosis factor receptor (TNFr) 1 (TNFr1) and TNFr2, finding a reduction in these biomarkers in patients treated with selenium and CoQ10 compared to those receiving placebo [34].
Another pathology related to oxidative stress and inflammation is cancer. Although there are few studies on CoQ10 supplementation as an adjuvant in cancer treatment, there is evidence of remission after supplementation with this compound. In addition, CoQ10 has demonstrated anti-inflammatory effects and decreased oxidative stress in patients with hepatocellular carcinoma [35,36,37]. It is believed that the anti-inflammatory actions of CoQ10 in cancer may be mediated by inhibition of the activation of NF-κB transcription factors. Likewise, studies in patients with migraine have reported that CoQ10 supplementation at a dose of 400 mg/day for three months significantly reduces the levels of calcitonin gene-related peptide (CGRP) and TNF-α [38].
Most of the available information on the anti-inflammatory effects of CoQ10 regards metabolic diseases such as diabetes, obesity and hypertension. However, many other pathophysiological processes such as infections and the immune response to pathogens also present with redox imbalance. It has been shown that, in patients with endogenous antioxidant deficiency, the prognosis can be serious, so the use of CoQ10 could be beneficial. Given the recent appearance of new viruses such as Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) and the daily increase in cases, as well as the high mortality in some patients with risk factors or previous comorbidities, the scientific population is seeking new pharmacological strategies to support a better prognosis for these patients (Figure 1).
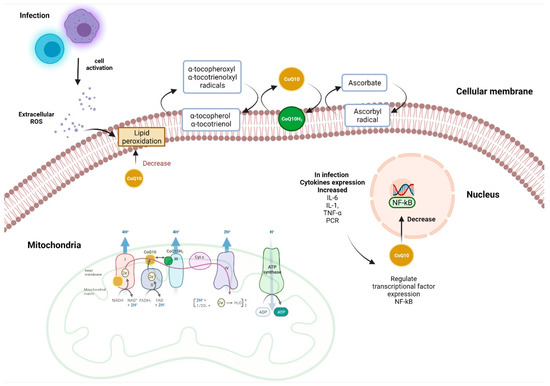
Figure 1.
Antioxidant and anti-inflammatory functions of CoQ10. During an infectious process, the cells of the immune response produce cytokines and reactive oxygen species (ROS) as a mechanism to combat a pathogen, these ROS can produce lipoperoxidation, which can be inhibited by the direct antioxidant effect of CoQ10. The main function of CoQ10 in the mitochondria is to transfer electrons to complex III (CIII). By transferring two electrons to Complex III, the reduced form of CoQ10 (ubiquinol) is oxidized to ubiquinone. The ubiquinol pool can be restored by accepting electrons from members of the electron transport chain (CI and CII). The anti-inflammatory effects of CoQ10 may be linked to the regulation of IL-1, IL-6, CRP and TNF-α gene expression through the NF-kB pathway. Figure was created with BioRender software, (Toronto, ON, Canada) ©biorender.com.
5. CoQ10 as a Possible Adjuvant in Infectious Diseases
As in cancer, migraine and aging, the role of inflammation in infections, both viral and bacterial, is well known. Likewise, it is known that oxidative stress plays a fundamental role in the immune response against pathogens [39]. For this reason, as a result, it is reasonable to believe that the use of substances with antioxidant and immunomodulatory effects can serve as an adjuvant in the management of infectious diseases. There is evidence that, in patients with infections such as influenza [40] or COVID-19 [41], endogenous CoQ10 levels are significantly lower than in healthy controls, as well as showing significant correlation with various inflammatory biomarkers. A computational study showed that CoQ10, like other quinones, exhibits a possible mechanism of action consistent with the anti-inflammatory drugs methylprednisolone and embelin [42], compounds that have been shown to inhibit viral infection through a variety of mechanisms and is active against influenza virus and hepatitis B [43].
A number of studies that have evaluated the effect of CoQ10 supplementation in infectious diseases (Table 1). In a study by Soltani et al., patients received 200 mg of CoQ10 daily for 7 days, resulting in a decrease in the levels of IL-6, TNF-α, glutathione peroxidase and malondialdehyde (MDA) [44].

Table 1.
Antioxidants and anti-inflammatory effects of CoQ10 in infectious diseases: human clinical trials.
Another similar study evaluated the effect of the administration of ubiquinol in patients with severe sepsis and septic shock; the study yielded inconclusive results on inflammatory markers and endothelial dysfunction, as there were no significant changes after treatment in levels of TNF-α, cytochrome C, nuclear and mitochondrial DNA [45]. The limitations of this study include the short follow-up time, which could have led to no significant changes being observable, as well as the severity of the patients’ conditions, which could hinder their recovery. Another clinical trial conducted in patients with HIV infection evaluated the effect of CoQ10 administration on CD4+ cell count and markers of liver and kidney function [46]. The study shows that in both groups, control and CoQ10, CD4+ count was significantly increased, without changes in markers of liver and kidney function, although the study does not demonstrate the efficacy of CoQ10 on T cell count, it is worth mentioning that conventional treatment could be responsible for the results obtained, however, in these patients with HIV infection it has been described that their CoQ10 levels endogenous are significantly decreased, so supplementation with CoQ10 helps to restore this deficiency.
In 2012, De Luca et al., examined the effects of antioxidant use, including CoQ10, on clinical benefits, inflammatory response, and antioxidant supplementation in patients with mucocutaneous viral lesions; the study shows that nutraceutical supplementation in patients suffering from viral infections, added to conventional antiviral drug therapy, can substantially improve clinical efficacy and prevent relapses, this same study shows that the use of antioxidants used in the experimental group improves viral load, decreasing it, compared to the control group can decrease viral load [47]. Another study conducted in 2016 evaluated the effect of oral administration of CoQ10 in patients with acute viral myocarditis. A clinical trial was carried out in which the patients were blinded and randomised into three groups based on the treatment: CoQ10; trimetazidine, a cytoprotective anti-ischaemic agent; and combination CQ10 and trimetazidine. Inflammation and oxidative markers and myocardial enzymes were evaluated. These markers decreased in all group, but the combination group showed the most powerful effect. This study demonstrated that the use of CoQ10 in combination with other agents with anti-inflammatory potential could be a therapeutic strategy in infectious diseases [48]. Moreover, a case report evidenced the possible beneficial effect of CoQ10 in anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis, observing improvement in the patient after the use of CoQ10 without response to standard immunotherapy. Although there have not been other studies evaluating the efficacy of CoQ10 supplementation in treating this condition, this report shows a possible complementary therapeutic option [49].
6. Conclusions
Although there are few clinical studies that demonstrate the effectiveness of CoQ10 supplementation in infectious diseases, its immunomodulatory and antioxidant effects in chronic diseases have been widely demonstrated. For this reason, its application in infectious processes is feasible and it can present benefits that contribute to the development of a potential new therapy for ID.
Author Contributions
Conceptualization, S.S.-F., D.C.S.-M.; Literature search, S.C.-I., V.A.S.-L., J.J.R.-V. and L.Y.Z.; Writing—original draft preparation, S.S.-F., J.J.R.-V., S.C.-I. and V.A.S.-L.; Writing—review and editing, S.S.-F. and D.C.S.-M. All authors have read and agreed to the published version of the manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Further Research
Further research is needed to support the confirmation of whether the administration of CoQ10 could improve the prognosis of patients with infectious diseases, as well as to evaluate its possible mechanism of action through biomarkers.
References
- Quinzii, C.M.; Lopez, L.C.; Von-Moltke, J.; Naini, A.; Krishna, S.; Schuelke, M.; Salviati, L.; Navas, P.; DiMauro, S.; Hirano, M. Respiratory chain dysfunction and oxidative stress correlate with severity of primary COQ10 deficiency. FASEB J. 2008, 22, 1874–1885. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, A.; Fonarow, G.C.; Butler, J.; Ezekowitz, J.A.; Felker, G.M. Coenzyme Q10 and heart failure: A state-of-the-art review. Circ. Heart Fail. 2016, 9, e002639. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Mariscal, F.M.; Arenas-de Larriva, A.P.; Limia-Perez, L.; Romero-Cabrera, J.L.; Yubero-Serrano, E.M.; López-Miranda, J. Coenzyme Q10 Supplementation for the Reduction of Oxidative Stress: Clinical Implications in the Treatment of Chronic Diseases. Int. J. Mol. Sci. 2020, 21, 7870. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Hawken, S.; Ôunpuu, S.; Dans, T.; Avezum, A.; Lanas, F.; McQueen, M.; Budaj, A.; Pais, P.; Varigos, J.; et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): Case-control study. Lancet 2004, 364, 937–952. [Google Scholar] [CrossRef]
- Ellwanger, J.H.; Kaminski, V.L.; Chies, J. Emerging infectious disease prevention: Where should we invest our resources and efforts? J. Infect. Public Health 2019, 12, 313–316. [Google Scholar] [CrossRef]
- Nii-Trebi, N.I. Emerging and Neglected Infectious Diseases: Insights, Advances, and Challenges. BioMed. Res. Int. 2017, 2017, 5245021. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization (WHO). Antibacterial Agents in Clinical Development: An Analysis of the Antibacterial Clinical Development Pipeline, Including Tuberculosis; WHO: Geneva, Switzerland, 2017; Available online: https://apps.who.int/iris/handle/10665/258965 (accessed on 20 February 2022).
- Alexander, J.; Tinkov, A.; Strand, T.A.; Alehagen, U.; Skalny, A.; Aaseth, J. Early Nutritional Interventions with Zinc, Selenium and Vitamin D for Raising Anti-Viral Resistance Against Progressive COVID-19. Nutrients 2020, 12, 2358. [Google Scholar] [CrossRef]
- Szkopińska, A. Ubiquinone. Biosynthesis of quinone ring and its isoprenoid side chain. Intracellular localization. Acta Biochim. Pol. 2000, 47, 469–480. [Google Scholar] [CrossRef] [Green Version]
- MBentinger, M.; Tekle, G. Dallner Coenzyme Q–biosynthesis and functions. Biochem. Biophys. Res. Commun. 2010, 396, 74–79. [Google Scholar] [CrossRef]
- Pallotti, F.; Bergamini, C.; Lamperti, C.; Fato, R. The Roles of Coenzyme Q in Disease: Direct and Indirect Involvement in Cellular Functions. Int. J. Mol. Sci. 2021, 23, 128. [Google Scholar] [CrossRef]
- Pravst, I.; Zmitek, K.; Zmitek, J. Coenzyme Q10 contents in foods and fortification strategies. Crit. Rev. Food Sci. Nutr. 2020, 50, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Mantle, D.; Dybring, A. Bioavailability of Coenzyme Q10: An Overview of the Absorption Process and Subsequent Metabolism. Antioxidants 2020, 9, 386. [Google Scholar] [CrossRef] [PubMed]
- Raizner, A.E. Coenzyme Q10. Methodist DeBakey Cardiovasc. J. 2019, 15, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Mantle, D.; Heaton, R.A.; Hargreaves, I.P. Coenzyme Q10 and Immune Function: An Overview. Antioxidants 2021, 10, 759. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, Y.; Xu, H.; Luo, X.; Yu, J.; Liu, J.; Chang, R.C. Neuroprotection of Coenzyme Q10 in Neurodegenerative Diseases. Curr. Top. Med. Chem. 2016, 16, 858–866. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Yang, K.L.; Zeng, L.T.; Wu, X.H.; Huang, H.Y. Effectiveness of Coenzyme Q10 Supplementation for Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis. Int. J. Endocrinol. 2018, 2018, 6484839. [Google Scholar] [CrossRef]
- Hargreaves, I.P.; Mantle, D. Coenzyme Q10 Supplementation in Fibrosis and Aging. Adv. Exp. Med. Biol. 2019, 1178, 103–112. [Google Scholar]
- Manzar, H.; Abdulhussein, D.; Yap, T.E.; Cordeiro, M.F. Cellular Consequences of Coenzyme Q10 Deficiency in Neurodegeneration of the Retina and Brain. Int. J. Mol. Sci. 2020, 21, 9299. [Google Scholar] [CrossRef]
- Farough, S.; Karaa, A.; Walker, M.; Slate, N.; Dasu, T.; Verbsky, J.; Fusunyan, R.; Canapari, C.; Kinane, T.; Van Cleave, J.; et al. Coenzyme Q10 and immunity: A case report and new implications for treatment of recurrent infections in metabolic diseases. Clin. Immunol. 2014, 155, 209–212. [Google Scholar] [CrossRef]
- Hidaka, T.; Fujii, K.; Funahashi, I.; Fukutomi, N.; Hosoe, K. Safety assessment of coenzyme Q10 (CoQ10). BioFactors 2008, 32, 199–208. [Google Scholar] [CrossRef]
- Deramaudt, T.B.; Dill, C.; Bonay, M. Regulation of oxidative stress by Nrf2 in the pathophysiology of infectious diseases. Med. Mal. Infect. 2013, 43, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hekimi, S. Understanding Ubiquinone. Trends Cell Biol. 2016, 26, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Alcazar-Fabra, M.; Navas, P.; Brea-Calvo, G. Coenzyme Q biosynthesis and its role in the respiratory chain structure. Biochim. Biophys. Acta 2016, 1857, 1073–1078. [Google Scholar] [CrossRef] [PubMed]
- Letts, J.A.; Fiedorczuk, K.; Degliesposti, G.; Skehel, M.; Sazanov, L.A. Structures of respiratory supercomplex I+ III2 reveal functional and conformational crosstalk. Mol. Cell 2019, 75, 1131–1146.e6. [Google Scholar] [CrossRef] [Green Version]
- Sohal, R.S. Coenzyme Q and vitamin E interactions. Methods Enzymol. 2004, 378, 146–151. [Google Scholar]
- Castaneda, O.A.; Lee, S.C.; Ho, C.T.; Huang, T.C. Macrophages in oxidative stress and models to evaluate the antioxidant function of dietary natural compounds. J. Food Drug Anal. 2017, 25, 111–118. [Google Scholar] [CrossRef] [Green Version]
- Forman, H.J.; Torres, M. Reactive oxygen species and cell signaling: Respiratory burst in macrophage signaling. Am. J. Respir. Crit. Care Med. 2002, 166, S4–S8. [Google Scholar] [CrossRef]
- Mazidi, M.; Kengne, A.P.; Banach, M.; Lipid and Blood Pressure Meta-analysis Collaboration Group. Effects of coenzyme Q10 supplementation on plasma C-reactive protein concentrations: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2018, 128, 130–136. [Google Scholar] [CrossRef]
- Heidari, A.; Hamidi, G.; Soleimani, A.; Aghadavod, E.; Asemi, Z. Effects of Coenzyme Q10 Supplementation on Gene Expressions Related to Insulin, Lipid, and Inflammation Pathways in Patients with Diabetic Nephropathy. Iran. J. Kidney Dis. 2018, 12, 14–21. [Google Scholar]
- Schmelzer, C.; Lindner, I.; Rimbach, G.; Niklowitz, P.; Menke, T.; Döring, F. Functions of coenzyme Q10 in inflammation and gene expression. Biofactors 2008, 32, 179–183. [Google Scholar] [CrossRef]
- Farsi, F.; Mohammadshahi, M.; Alavinejad, P.; Rezazadeh, A.; Zarei, M.; Engali, K.A. Functions of Coenzyme Q10 Supplementation on Liver Enzymes, Markers of Systemic Inflammation, and Adipokines in Patients Affected by Nonalcoholic Fatty Liver Disease: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial. J. Am. Coll. Nutr. 2016, 35, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Alehagen, U.; Lindahl, T.L.; Aaseth, J.; Svensson, E.; Johansson, P. Levels of sP-selectin and hs-CRP Decrease with Dietary Intervention with Selenium and Coenzyme Q10 Combined: A Secondary Analysis of a Randomized Clinical Trial. PLoS ONE 2015, 10, e0137680. [Google Scholar]
- Alehagen, U.; Alexander, J.; Aaseth, J.; Larsson, A. Decrease in inflammatory biomarker concentration by intervention with selenium and coenzyme Q10: A subanalysis of osteopontin, osteoprotergerin, TNFr1, TNFr2 and TWEAK. J. Inflamm. 2019, 16, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Folkers, K.; Brown, R.; Judy, W.V.; Morita, M. Survival of cancer patients on therapy with coenzyme Q10. Biochem. Biophys. Res. Commun. 1993, 192, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Lockwood, K.; Moesgaard, S.; Folkers, K. Partial and complete regression of breast cancer in patients in relation to dosage of coenzyme Q10. Biochem. Biophys. Res. Commun. 1994, 199, 1504–1508. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-T.; Huang, Y.-C.; Cheng, S.-B.; Huang, Y.-T.; Lin, P.-T. Effects of coenzyme Q10 supplementation on antioxidant capacity and inflammation in hepatocellular carcinoma patients after surgery: A randomized, placebo-controlled trial. Nutr. J. 2015, 15, 85. [Google Scholar] [CrossRef] [Green Version]
- Dahri, M.; Tarighat-Esfanjani, A.; Asghari-Jafarabadi, M.; Hashemilar, M. Oral coenzyme Q10 supplementation in patients with migraine: Effects on clinical features and inflammatory markers. Nutr. Neurosci. 2019, 22, 607–615. [Google Scholar] [CrossRef]
- Shastri, M.D.; Shukla, S.D.; Chong, W.C.; Dua, K.; Peterson, G.M.; Patel, R.P.; Hansbro, P.M.; Eri, R.; O’Toole, R.F. Role of Oxidative Stress in the Pathology and Management of Human Tuberculosis. Oxid. Med. Cell Longev. 2018, 2018, 7695364. [Google Scholar] [CrossRef] [Green Version]
- Chase, M.; Cocchi, M.N.; Liu, X.; Andersen, L.W.; Holmberg, M.J.; Donnino, M.W. Coenzyme Q10 in acute influenza. Influenza Other Respir. Viruses 2019, 13, 64–70. [Google Scholar] [CrossRef]
- Sumbalova, Z.; Kucharska, J.; Palacka, P.; Rausova, Z.; Langsjoen, P.H.; Langsjoen, A.M.; Gvozdjakova, A. Platelet mitochondrial function and endogenous coenzyme Q10 levels are reduced in patients after COVID-19. Bratisl. Lek. Listy 2022, 123, 9–15. [Google Scholar] [CrossRef]
- Caruso, F.; Rossi, M.; Pedersen, J.Z.; Incerpi, S. Computational studies reveal mechanism by which quinone derivatives can inhibit SARS-CoV-2. Study of embelin and two therapeutic compounds of interest, methyl prednisolone and dexamethasone. J. Infect. Public Health 2020, 13, 1868–1877. [Google Scholar] [CrossRef] [PubMed]
- Parvez, M.K.; Rehman, T.; Alam, P.; Al-Dosari, M.S.; Alqasoumi, S.I.; Mohammed, F.; Alajmi, M.F. Plant-derived antiviral drugs as novel hepatitis B virus inhibitors: Cell culture and molecular docking study. Saudi Pharm. J. 2019, 27, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Soltani, R.; Alikiaie, B.; Shafiee, F.; Amiri, H.; Mousavi, S. Coenzyme Q10 improves the survival and reduces inflammatory markers in septic patients. Bratisl. Lek. Listy 2020, 121, 154–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donnino, M.W.; Mortensen, S.J.; Andersen, L.W.; Chase, M.; Berg, K.M.; Balkema, J.; Radhakrishnan, J.; Gazmuri, R.J.; Liu, X.; Cocchi, M.N. Ubiquinol (reduced Coenzyme Q10) in patients with severe sepsis or septic shock: A randomized, double-blind, placebo-controlled, pilot trial. Crit. Care 2015, 19, 275. [Google Scholar] [CrossRef] [Green Version]
- Yousefi, F.; Roozbeh, F. The effect of coenzyme Q10 in comparison with placebo on CD4 in HIV-infected patients. Clin. Epidemiol. Glob. Health 2019, 7, 306–308. [Google Scholar] [CrossRef] [Green Version]
- De Luca, C.; Kharaeva, Z.; Raskovic, D.; Pastore, P.; Luci, A.; Korkina, L. Coenzyme Q(10), vitamin E, selenium, and methionine in the treatment of chronic recurrent viral mucocutaneous infections. Nutrition 2012, 28, 509–514. [Google Scholar] [CrossRef]
- Shao, L.; Ma, A.; Figtree, G.; Zhang, P. Combination Therapy with Coenzyme Q10 and Trimetazidine in Patients With Acute Viral Myocarditis. J. Cardiovasc. Pharmacol. 2016, 68, 150–154. [Google Scholar] [CrossRef]
- Rangel, G.R.; Camara, C.R.; Garcia, A.G.; Rodriguez, A.C.; Galarza, D.A. Could coenzyme Q10 supplementation have a role in the treatment of anti-NMDA receptor encephalitis? Acta Neurol. Belg. 2015, 115, 85–86. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).