Epidemiological Algorithm for Early Detection of COVID-19 Cases in a Mexican Oncologic Center
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. O.S.M. Training and Personal Protection Equipment (P.P.E.)
2.3. Application of Electronic Surveys to O.S.M.
2.3.1. R-Track Predictive Model
2.3.2. S-Facts Predictive Model
2.4. Detection of SARS-CoV-2 IgM and IgG Antibodies in O.S.M.
2.5. Paper Surveys for Radiotherapy Area Patients
2.6. Sample Collection for SARS-CoV-2 qPCR Assay
2.7. RNA Extraction
2.8. qRT-PCR Tests
3. Results
3.1. Electronic Surveys and Antibodies Detection in O.S.M.
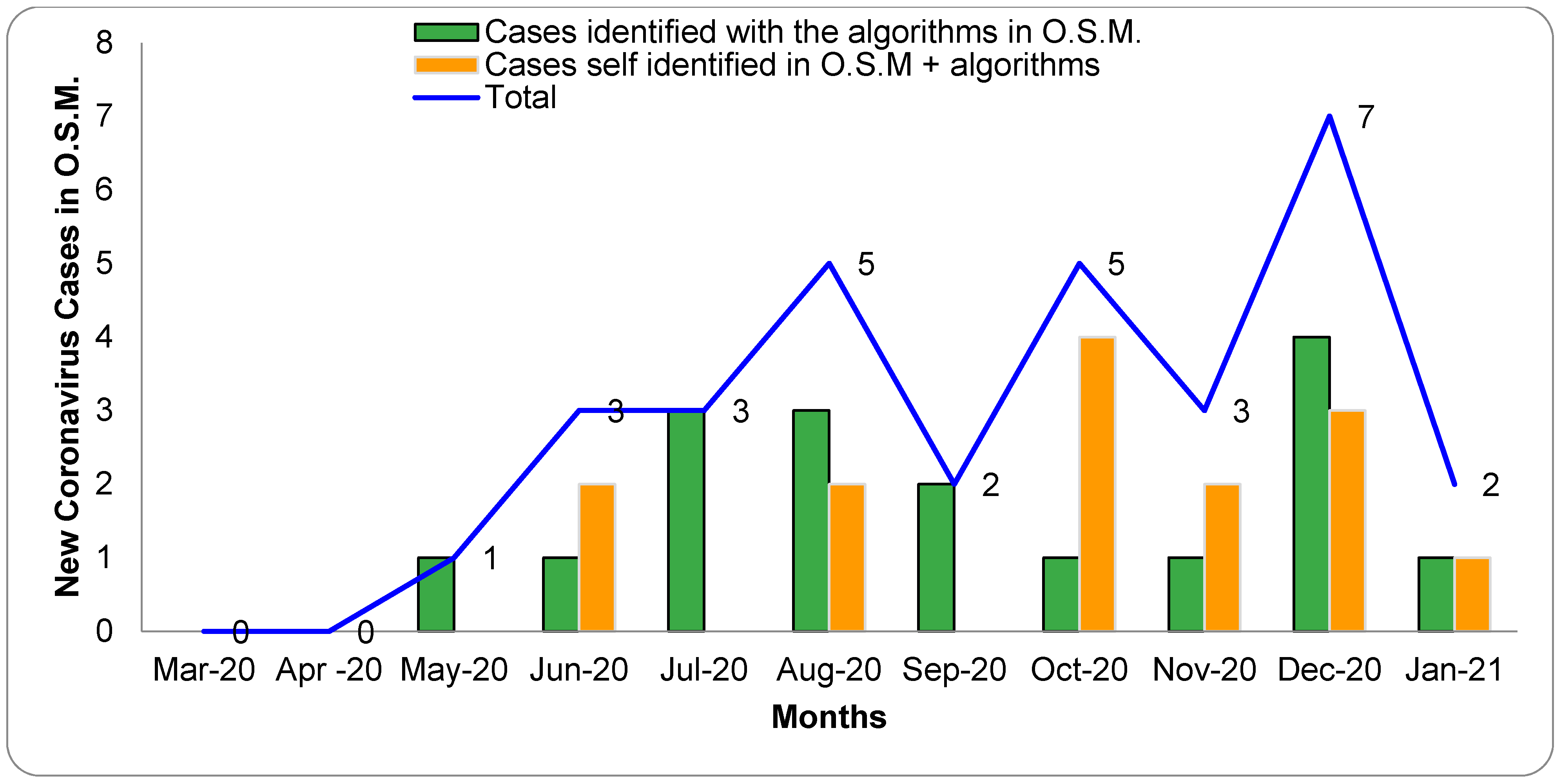
3.1.1. R-Track and S-Facts Algorithm for O.S.M Screening
3.1.2. Identification of Antibodies in O.S.M.
3.2. Application of Paper Surveys for Patients in the Radiotherapy Area
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. WHO Timeline—COVID-19. Available online: https://www.who.int/news/item/27-04-2020-who-timeline---covid-19 (accessed on 21 April 2021).
- Liang, W.; Guan, W.; Chen, R.; Wang, W.; Li, J.; Xu, K.; Li, C.; Ai, Q.; Lu, W.; Liang, H.; et al. Cancer patients in SARS-CoV-2 infection: A nationwide analysis in China. Lancet Oncol. 2020, 21, 335–337. [Google Scholar] [CrossRef]
- Zhang, L.; Zhu, F.; Xie, L.; Wang, C.; Wang, J.; Chen, R.; Jia, P.; Guan, H.Q.; Peng, L.; Chen, Y.; et al. Clinical characteristics of COVID-19-infected cancer patients: A retrospective case study in three hospitals within Wuhan, China. Ann. Oncol. 2020, 31, 894–901. [Google Scholar] [CrossRef]
- Han, H.J.; Nwagwu, C.; Anyim, O.; Ekweremadu, C.; Kim, S. COVID-19 and cancer: From basic mechanisms to vaccine development using nanotechnology. Int. Immunopharmacol. 2020, 90, 107247. [Google Scholar] [CrossRef]
- Li, X.; Geng, M.; Peng, Y.; Meng, L.; Lu, S. Molecular immune pathogenesis and diagnosis of COVID-19. J. Pharm. Anal. 2020, 10, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Campos Nonato, I.; Cuevas Nasu, L.; González Castell, L.D.; Hernández Barrera, L.; Shamah Levy, T.; González de Cosío, T. Epidemiología de la obesidad y sus principales comorbilidades en México. In La Obesidad en México, Primera edición ed.; Dommarco, J.Á.R., Ed.; Instituto Nacional de Salud Pública: Cuernavaca, Mexico, 2018; p. 9. [Google Scholar]
- Cui, M.; Wu, X.; Mao, J.; Wang, X.; Nie, M. T2DM Self-Management via Smartphone Applications: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0166718. [Google Scholar] [CrossRef]
- Formagini, T.D.B.; Ervilha, R.R.; Machado, N.M.; De Andrade, B.A.B.B.; Gomide, H.P.; Ronzani, T.M. A review of smartphone apps for smoking cessation available in Portuguese. Cad. Saude Publica 2017, 33, e00178215. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Garg, S.; Williams, N.L.; Ip, A.; Dicker, A.P. Clinical Integration of Digital Solutions in Health Care: An Overview of the Current Landscape of Digital Technologies in Cancer Care. JCO Clin. Cancer Inform. 2018, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Logishetty, K. Adopting and sustaining a Virtual Fracture Clinic model in the District Hospital setting—A quality improvement approach. BMJ Qual. Improv. Rep. 2017, 6, u220211–w7861. [Google Scholar] [CrossRef]
- Menni, C.; Valdes, A.M.; Freidin, M.B.; Sudre, C.H.; Nguyen, L.H.; Drew, D.A.; Ganesh, S.; Varsavsky, T.; Cardoso, M.J.; El-Sayed Moustafa, J.S.; et al. Real-time tracking of self-reported symptoms to predict potential COVID-19. Nat. Med. 2020, 26, 1037–1040. [Google Scholar] [CrossRef]
- Alimohamadi, Y.; Sepandi, M.; Taghdir, M.; Hosamirudsari, H. Determine the most common clinical symptoms in COVID-19 patients: A systematic review and meta-analysis. J. Prev. Med. Hyg. 2020, 61, E304–E312. [Google Scholar] [PubMed]
- CDC. Centers for Disease Control and Prevention (CDC). Real-TIme RT-PCR Diagnostic Panel. Available online: https://www.fda.gov/media/134922/download (accessed on 21 April 2021).
- ESMO. European Society of Medical Oncology. Cancer Patiet Manangement during the COVID-19 Pandemic. Available online: https://www.esmo.org/guidelines/cancer-patient-management-during-the-covid-19-pandemic (accessed on 16 April 2021).
- ASCO. General Information about COVID-19 & Cancer. Available online: https://www.asco.org/asco-coronavirus-resources/care-individuals-cancer-during-covid-19/general-information-about-covid-19 (accessed on 16 April 2021).
- Memorial Sloan Kettering Cancer Center. Available online: https://www.mskcc.org/search?msk_promote[query]=covid (accessed on 1 March 2021).
- Graham, M.S.; Sudre, C.H.; May, A.; Antonelli, M.; Murray, B.; Varsavsky, T.; Kläser, K.; Canas, L.S.; Molteni, E.; Modat, M.; et al. Changes in symptomatology, reinfection, and transmissibility associated with the SARS-CoV-2 variant B.1.1.7: An ecological study. Lancet Public Health 2021, 6, e335–e345. [Google Scholar] [CrossRef]
- Montes, J. Covid-19 Takes Outsize Toll on Mexican Health Workers. Available online: https://www.wsj.com/livecoverage/covid-2021-01-08/card/vzTb2JoN4tpreIdBu5Kh (accessed on 16 April 2021).
- MD Anderson Center/COVID Prevention. Available online: https://www.mdanderson.org/patients-family/search-results.html?q=covid%20prevention# (accessed on 1 March 2021).
- Dana-Farber Cancer Institute. Available online: https://www.dana-farber.org (accessed on 21 April 2021).
- Motlagh, A.; Yamrali, M.; Azghandi, S.; Azadeh, P.; Vaezi, M.; Ashrafi, F.; Zendehdel, K.; Mirzaei, H.; Basi, A.; Rakhsha, A.; et al. COVID19 Prevention & Care; A Cancer Specific Guideline. Arch. Iran. Med. 2020, 23, 255–264. [Google Scholar] [CrossRef]
- Bastiani, L.; Fortunato, L.; Pieroni, S.; Bianchi, F.; Adorni, F.; Prinelli, F.; Giacomelli, A.; Pagani, G.; Maggi, S.; Trevisan, C.; et al. Rapid COVID-19 Screening Based on Self-Reported Symptoms: Psychometric Assessment and Validation of the EPICOVID19 Short Diagnostic Scale. J. Med. Internet Res. 2021, 23, e23897. [Google Scholar] [CrossRef] [PubMed]
- Cherry, G.; Rocke, J.; Chu, M.; Liu, J.; Lechner, M.; Lund, V.J.; Kumar, B.N. Loss of smell and taste: A new marker of COVID-19? Tracking reduced sense of smell during the coronavirus pandemic using search trends. Expert Rev. Anti-Infect. Ther. 2020, 18, 1165–1170. [Google Scholar] [CrossRef]
- Lee, L.Y.W.; Cazier, J.-B.; Starkey, T.; Briggs, S.E.W.; Arnold, R.; Bisht, V.; Booth, S.; Campton, N.A.; Cheng, V.W.T.; Collins, G.; et al. COVID-19 prevalence and mortality in patients with cancer and the effect of primary tumour subtype and patient demographics: A prospective cohort study. Lancet Oncol. 2020, 21, 1309–1316. [Google Scholar] [CrossRef]
- Henry, B.M.; Oliveira, M.H.; Benoit, J.; Lippi, G. Gastrointestinal symptoms associated with severity of coronavirus disease 2019 (COVID-19): A pooled analysis. Intern. Emerg. Med. 2020, 15, 857–859. [Google Scholar] [CrossRef]
- Li, J.; Chen, Z.; Nie, Y.; Ma, Y.; Guo, Q.; Dai, X. Identification of Symptoms Prognostic of COVID-19 Severity: Multivariate Data Analysis of a Case Series in Henan Province. J. Med. Internet Res. 2020, 22, e19636. [Google Scholar] [CrossRef]
- Dubois, R.N. COVID-19, Cancer Care and Prevention. Cancer Prev. Res. 2020, 13, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Fuereder, T.; Berghoff, A.S.; Heller, G.; Haslacher, H.; Perkmann, T.; Strassl, R.; Berger, J.M.; Puhr, H.C.; Kreminger, J.; Moik, F.; et al. SARS-CoV-2 seroprevalence in oncology healthcare professionals and patients with cancer at a tertiary care centre during the COVID-19 pandemic. ESMO Open 2020, 5, e000889. [Google Scholar] [CrossRef]
- Mahto, M.; Banerjee, A.; Biswas, B.; Kumar, S.; Agarwal, N.; Singh, P.K. Seroprevalence of IgG against SARS-CoV-2 and its determinants among healthcare workers of a COVID-19 dedicated hospital of India. Am. J. Blood Res. 2021, 11, 44–52. [Google Scholar] [PubMed]
- Ladoire, S.; Goussot, V.; Redersdorff, E.; Cueff, A.; Ballot, E.; Truntzer, C.; Ayati, S.; Bengrine-Lefevre, L.; Bremaud, N.; Coudert, B.; et al. Seroprevalence of SARS-CoV-2 among the staff and patients of a French cancer centre after first lockdown: The canSEROcov study. Eur. J. Cancer 2021, 148, 359–370. [Google Scholar] [CrossRef]
- Garcia-Basteiro, A.L.; Moncunill, G.; Tortajada, M.; Vidal, M.; Guinovart, C.; Jiménez, A.; Santano, R.; Sanz, S.; Méndez, S.; Llupià, A.; et al. Seroprevalence of antibodies against SARS-CoV-2 among health care workers in a large Spanish reference hospital. Nat. Commun. 2020, 11, 3500. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Xu, Y.; Sun, C.; Wang, X.; Guo, Y.; Qiu, S.; Ma, K. A systematic review of asymptomatic infections with COVID-19. J. Microbiol. Immunol. Infect. 2020, 54, 12–16. [Google Scholar] [CrossRef]
- Fuereder, T.; Memo, E.O.; Gunsilius, E.; Bartsch, R.; Hilbe, W. Circumnavigating the challenges of COVID-19 in oncology. memo Mag. Eur. Med. Oncol. 2020, 13, 135–138. [Google Scholar] [CrossRef]
- Long, Q.-X.; Tang, X.-J.; Shi, Q.-L.; Li, Q.; Deng, H.-J.; Yuan, J.; Hu, J.-L.; Xu, W.; Zhang, Y.; Lv, F.-J.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef] [PubMed]
- Burrows, C.S.; Weigel, F.K. Mobile Phone Health Applications for the Federal Sector. U.S. Army Med. Dep. J. 2016, Jan-Mar, 71–75. [Google Scholar]
| Characteristics | n (%) |
|---|---|
| Gender | |
| Male | 7 (41.18) |
| Female | 10 (58.82) |
| Co- morbidities | |
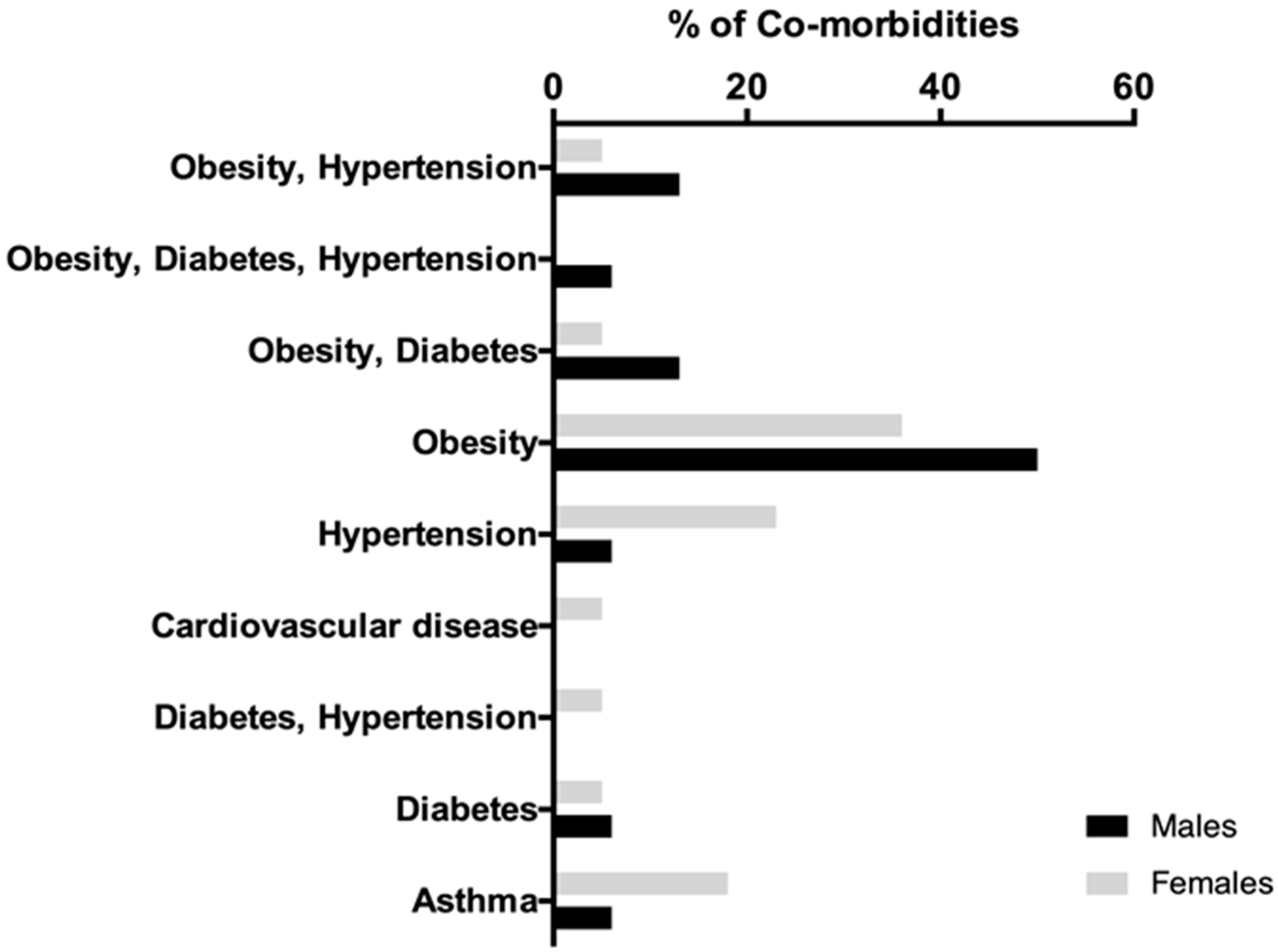
| Obesity | 4 (23.53) |
| Diabetes, Hypertension | 1 (5.88) |
| Any | 12 (70.59) |
| CUCC Work area | |
| Nursery | 6 (35.29) |
| Interns/Residents | 1 (5.88) |
| Administration | 6 (35.29) |
| Radiation therapist | 4 (23.53) |
| Symptoms | |
| Fatigue * | 6 (35.29) |
| Skipped meals * | 13 (76.47) |
| Loss of taste/smell * | 14 (82.35) |
| Cough * | 9 (52.94) |
| Fever * | 8 (47.06) |
| Shortness of breath * | 4 (23.53) |
| Chest pain | 6 (35.29) |
| Diarrhea or vomiting | 5 (29.41) |
| Sore throat | 10 (58.82) |
| Pink eyes | 3 (17.65) |
| Runny nose | 10 (58.82) |
| Muscle or body aches | 12 (70.59) |
| Headaches | 12 (70.59) |
| Risk exposure | |
| Exposure with COVID-19 positive patients | 11 (64.71) |
| Exposure with family/friends with symptoms of COVID-19 | 9 (52.94) |
| Exposure with positive family/friends COVID-19 | 10 (58.82) |
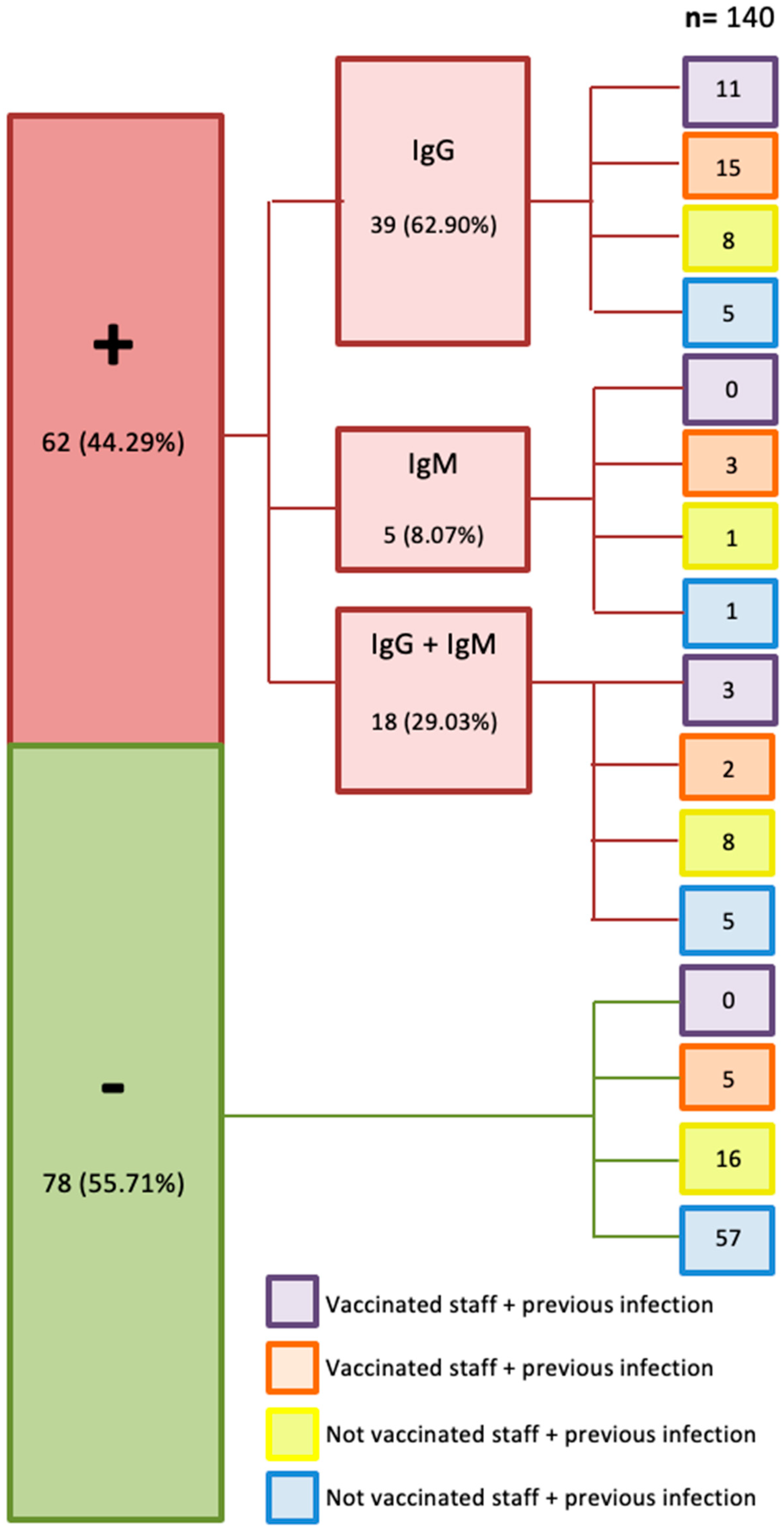
| Antibody Test | Oncology Staff n = 140 (%) | Vaccinated | With Previous Infection | Vaccinated + Previous Infection | Non Vaccinated without Previous Infection |
|---|---|---|---|---|---|
| Positive | 62 (45%) | 20 | 17 | 14 | 11 |
| Negative | 78 (57%) | 5 | 16 | 0 | 57 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Escamilla, M.; Pérez-Ibave, D.C.; Burciaga-Flores, C.H.; Ortiz-Murillo, V.N.; Ramírez-Correa, G.A.; Rodríguez-Niño, P.; Piñeiro-Retif, R.; Rodríguez-Gutiérrez, H.F.; Alcorta-Nuñez, F.; González-Guerrero, J.F.; et al. Epidemiological Algorithm for Early Detection of COVID-19 Cases in a Mexican Oncologic Center. Healthcare 2022, 10, 462. https://doi.org/10.3390/healthcare10030462
González-Escamilla M, Pérez-Ibave DC, Burciaga-Flores CH, Ortiz-Murillo VN, Ramírez-Correa GA, Rodríguez-Niño P, Piñeiro-Retif R, Rodríguez-Gutiérrez HF, Alcorta-Nuñez F, González-Guerrero JF, et al. Epidemiological Algorithm for Early Detection of COVID-19 Cases in a Mexican Oncologic Center. Healthcare. 2022; 10(3):462. https://doi.org/10.3390/healthcare10030462
Chicago/Turabian StyleGonzález-Escamilla, Moisés, Diana Cristina Pérez-Ibave, Carlos Horacio Burciaga-Flores, Vanessa Natali Ortiz-Murillo, Genaro A. Ramírez-Correa, Patricia Rodríguez-Niño, Rafael Piñeiro-Retif, Hazyadee Frecia Rodríguez-Gutiérrez, Fernando Alcorta-Nuñez, Juan Francisco González-Guerrero, and et al. 2022. "Epidemiological Algorithm for Early Detection of COVID-19 Cases in a Mexican Oncologic Center" Healthcare 10, no. 3: 462. https://doi.org/10.3390/healthcare10030462
APA StyleGonzález-Escamilla, M., Pérez-Ibave, D. C., Burciaga-Flores, C. H., Ortiz-Murillo, V. N., Ramírez-Correa, G. A., Rodríguez-Niño, P., Piñeiro-Retif, R., Rodríguez-Gutiérrez, H. F., Alcorta-Nuñez, F., González-Guerrero, J. F., Vidal-Gutiérrez, O., & Garza-Rodríguez, M. L. (2022). Epidemiological Algorithm for Early Detection of COVID-19 Cases in a Mexican Oncologic Center. Healthcare, 10(3), 462. https://doi.org/10.3390/healthcare10030462









