Hereditary Breast and Ovarian Cancer Service in Sparsely Populated Western Pomerania
Abstract
:1. Introduction
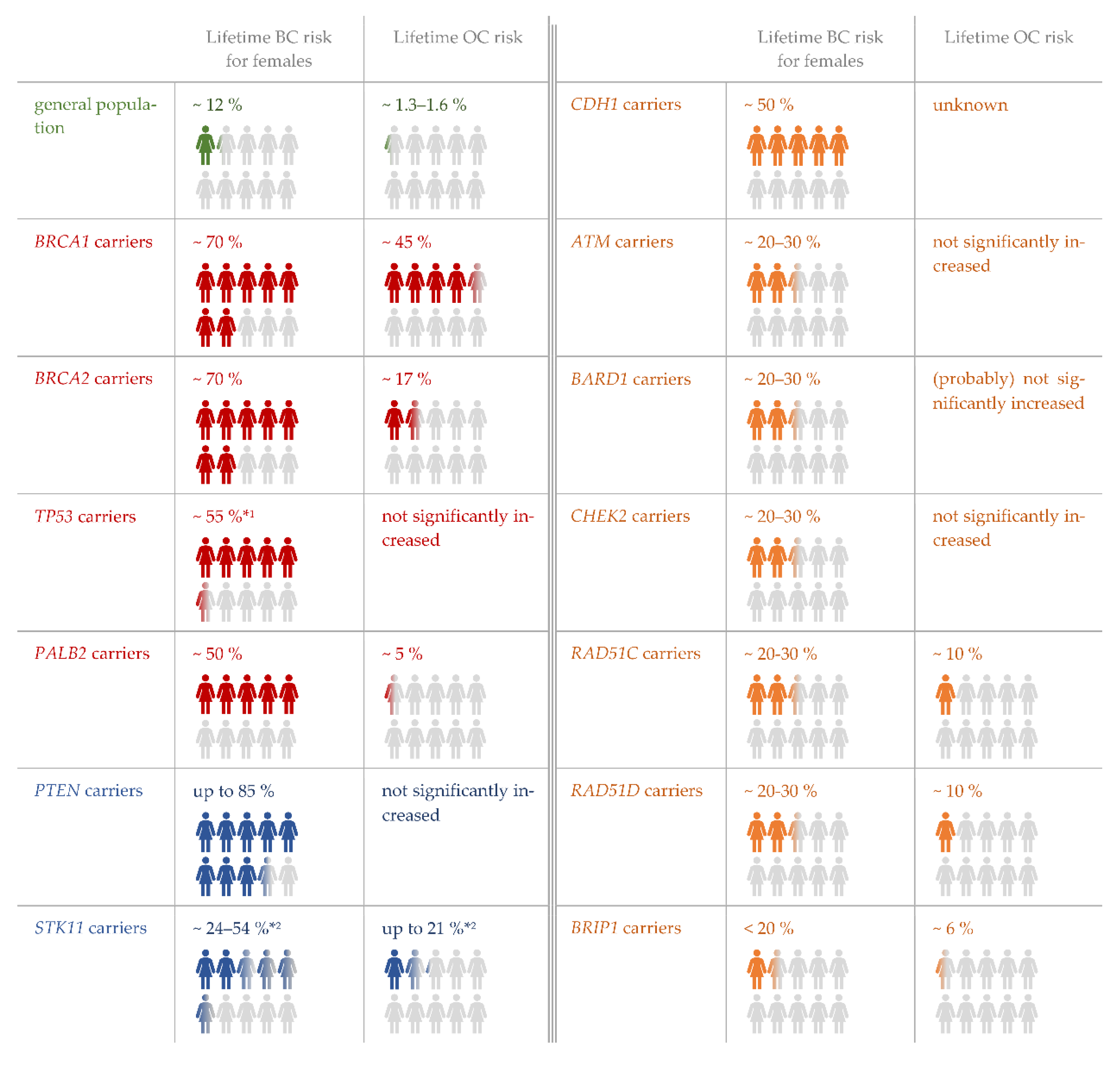

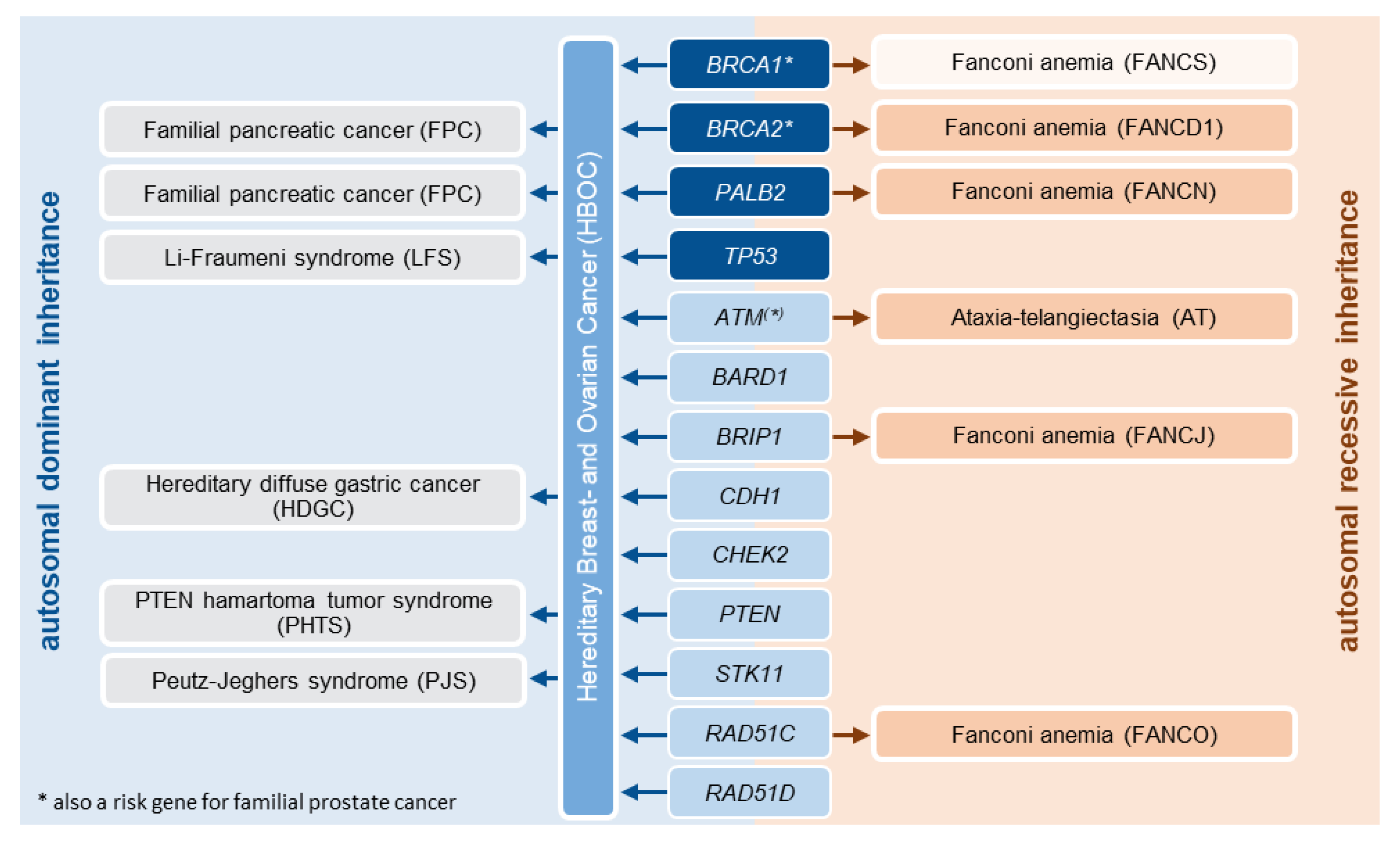
2. Materials and Methods
2.1. Implementation of the Clinical Framework
2.2. Molecular Genetic Analyses
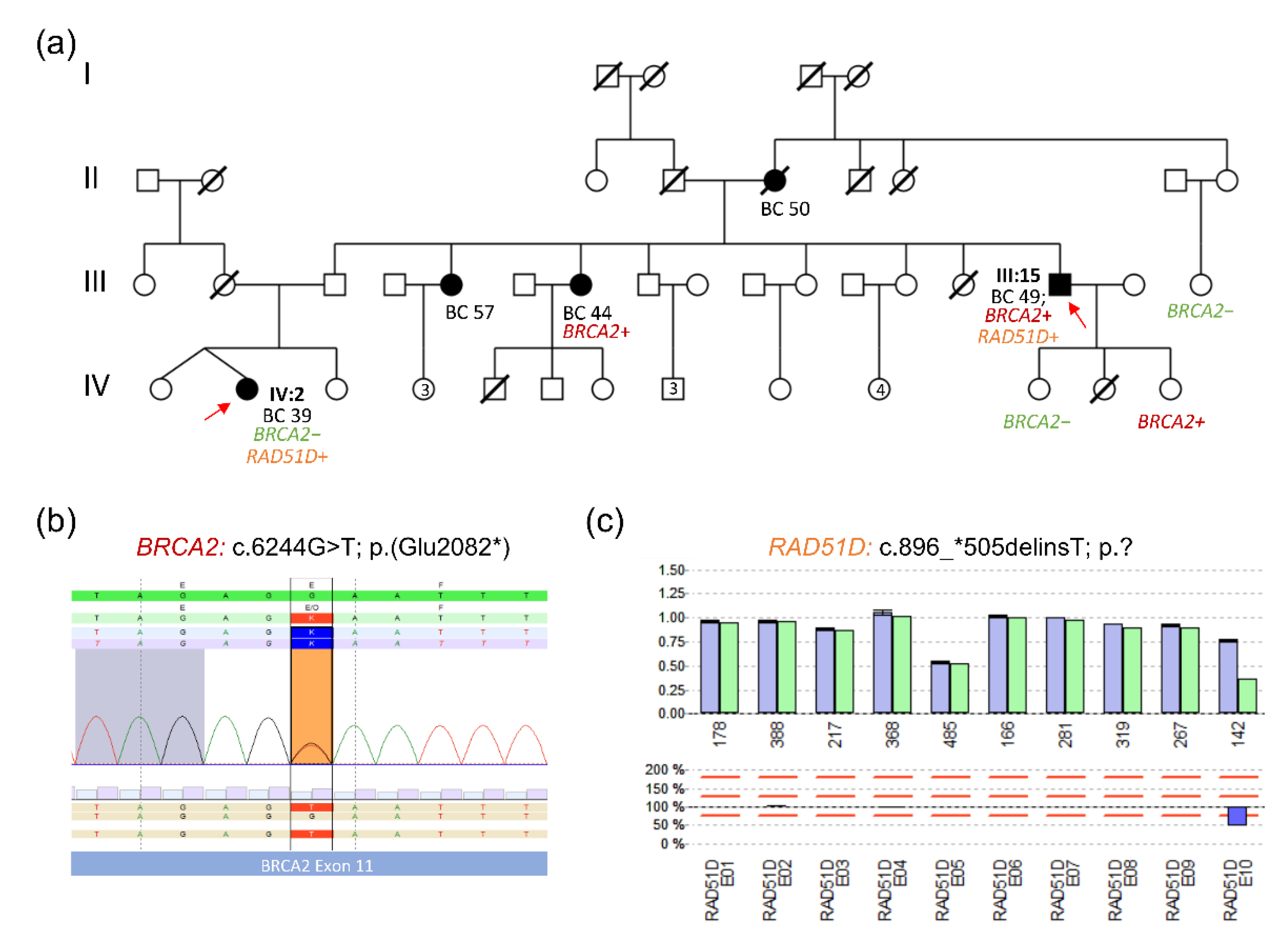
2.3. Establishment of a PCR Validation Assay for a RAD51D Deletion
2.4. Data Analysis and Visualization
3. Results
3.1. Familial Breast and Ovarian Cancer Center Greifswald
3.2. Clinical Genetic Testing
3.2.1. Individuals with Fulfilled Inclusion Criteria
3.2.2. Patients with TNBC in the 50-to-59 Age Group
3.2.3. Variants of Unknown Significance (VUS)
3.2.4. Predictive and Diagnostic Genetic Analyses for At-Risk Family Members
3.3. Selected Family Report: Pathogenic BRCA2 and RAD51D Variants in an HBOC Family
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rhiem, K.; Auber, B.; Briest, S.; Dikow, N.; Ditsch, N.; Dragicevic, N.; Grill, S.; Hahnen, E.; Horvath, J.; Jaeger, B.; et al. Consensus Recommendations of the German Consortium for Hereditary Breast and Ovarian Cancer. Breast Care 2022, 17, 199–207. [Google Scholar] [CrossRef] [PubMed]
- OncoMap—Certified Breast Cancer Centers in Mecklenburg-Western Pomerania. Available online: https://www.oncomap.de/centers?selectedOrgan=Brust&selectedCounty=Deutschland&selectedState=Mecklenburg-Vorpommern (accessed on 11 August 2022).
- Consensus Recommendations of the German Consortium for Hereditary Breast and Ovarian Cancer; German Consortium for Hereditary Breast and Ovarian Cancer; Germany. Available online: https://www.konsortium-familiaerer-brustkrebs.de/konsensusempfehlung/ (accessed on 8 August 2022).
- Klimkowski, S.; Ibrahim, M.; Ibarra Rovira, J.J.; Elshikh, M.; Javadi, S.; Klekers, A.R.; Abusaif, A.A.; Moawad, A.W.; Ali, K.; Elsayes, K.M. Peutz-Jeghers Syndrome and the Role of Imaging: Pathophysiology, Diagnosis, and Associated Cancers. Cancers 2021, 13, 5121. [Google Scholar] [CrossRef]
- McGarrity, T.J.; Amos, C.I.; Baker, M.J. Peutz-Jeghers Syndrome. 2001 Feb 23 [Updated 2021 Sep 2]. In GeneReviews® [Internet]; Adam, M.P., Everman, D.B., Mirzaa, G.M., Eds.; University of Washington: Seattle, WA, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK1266/ (accessed on 8 August 2022).
- Giardiello, F.M.; Brensinger, J.D.; Tersmette, A.C.; Goodman, S.N.; Petersen, G.M.; Booker, S.V.; Cruz-Correa, M.; Offerhaus, J.A. Very high risk of cancer in familial Peutz-Jeghers syndrome. Gastroenterology 2000, 119, 1447–1453. [Google Scholar] [CrossRef] [Green Version]
- Resta, N.; Pierannunzio, D.; Lenato, G.M.; Stella, A.; Capocaccia, R.; Bagnulo, R.; Lastella, P.; Susca, F.C.; Bozzao, C.; Loconte, D.C.; et al. Cancer risk associated with STK11/LKB1 germline mutations in Peutz-Jeghers syndrome patients: Results of an Italian multicenter study. Dig. Liver Dis. 2013, 45, 606–611. [Google Scholar] [CrossRef]
- Hosten, N.; Bulow, R.; Volzke, H.; Domin, M.; Schmidt, C.O.; Teumer, A.; Ittermann, T.; Nauck, M.; Felix, S.; Dörr, M.; et al. SHIP-MR and Radiology: 12 Years of Whole-Body Magnetic Resonance Imaging in a Single Center. Healthcare 2021, 10, 33. [Google Scholar] [CrossRef] [PubMed]
- Hosten, N.; Rosenberg, B.; Kram, A. Project Report on Telemedicine: What We Learned about the Administration and Development of a Binational Digital Infrastructure Project. Healthcare 2021, 9, 400. [Google Scholar] [CrossRef] [PubMed]
- Krebs in Mecklenburg-Vorpommern 2014–2015 (Landesbericht); Gemeinsames Krebsregister der Länder Berlin, Brandenburg, Mecklenburg-Vorpommern, Sachsen—Anhalt und der Freistaaten Sachsen und Thüringen (GKR); Berlin (Germany). Available online: https://www.berlin.de/gkr/dienstleistungen/publikationen/landesberichte/ (accessed on 8 August 2022).
- Rhiem, K.; Bucker-Nott, H.J.; Hellmich, M.; Fischer, H.; Ataseven, B.; Dittmer-Grabowski, C.; Latos, K.; Pelzer, V.; Seifert, M.; Schmidt, A.; et al. Benchmarking of a checklist for the identification of familial risk for breast and ovarian cancers in a prospective cohort. Breast J. 2019, 25, 455–460. [Google Scholar] [CrossRef]
- Datenreport 2021—Ein Sozialbericht für die Bundesrepublik Deutschland; Statistisches Bundesamt (Destatis), Wissenschaftszentrum Berlin für Sozialforschung (WZB), Bundesinstitut für Bevölkerungsforschung (BiB), Das Sozio-oekonomische Panel (SOEP) am Deutschen Institut für Wirtschaftsforschung (DIW Berlin); Bonn (Germany). Available online: https://www.destatis.de/DE/Service/Statistik-Campus/Datenreport/Downloads/datenreport-2021.html (accessed on 8 August 2022).
- Mecklenburg-Vorpommern Location Map Simplified.svg; TUBS Derivative Work: MichaelBueker, CC0, via Wikimedia Commons. Available online: https://commons.wikimedia.org/wiki/File:Mecklenburg-Vorpommern_location_map_simplified.svg (accessed on 4 August 2022).
- Guidelines Breast (Version 2021.1E) of the Working Group Gynecological Oncology (Arbeitsgemeinschaft Gynäkologische Onkologie/AGO): Breast Cancer Risk and Prevention; Expert Panel ’breast‘; AGO e. V. Available online: https://www.ago-online.de/leitlinien-empfehlungen/leitlinien-empfehlungen/kommission-mamma (accessed on 7 August 2022).
- Vaughn, C.P.; Robles, J.; Swensen, J.J.; Miller, C.E.; Lyon, E.; Mao, R.; Bayrak-Toydemir, P.; Samowitz, W.S. Clinical analysis of PMS2: Mutation detection and avoidance of pseudogenes. Hum. Mutat. 2010, 31, 588–593. [Google Scholar] [CrossRef]
- Bick, U. High-risk screening using magnetic resonance imaging. Radiologe 2021, 61, 150–158. [Google Scholar] [CrossRef]
- Meindl, A.; Ditsch, N.; Kast, K.; Rhiem, K.; Schmutzler, R.K. Hereditary breast and ovarian cancer: New genes, new treatments, new concepts. Dtsch. Arztebl. Int. 2011, 108, 323–330. [Google Scholar] [CrossRef]
- Quante, A.S.; Engel, C.; Kiechle, M.; Schmutzler, R.K.; Fischer, C. Umstrukturierung der Risikoberechnung für die intensivierte Früherkennung im Deutschen Konsortium für Brust- und Eierstockkrebs. Der Gynäkologe 2020, 53, 259–264. [Google Scholar] [CrossRef] [Green Version]
- Schmutzler, R.K. Quality and Quantity: How to Organize a Countrywide Genetic Counseling and Testing. Breast Care 2021, 16, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Ataseven, B.; Tripon, D.; Rhiem, K.; Harter, P.; Schneider, S.; Heitz, F.; Baert, T.; Traut, A.; Pauly, N.; Ehmann, S.; et al. Prevalence of BRCA1 and BRCA2 Mutations in Patients with Primary Ovarian Cancer—Does the German Checklist for Detecting the Risk of Hereditary Breast and Ovarian Cancer Adequately Depict the Need for Consultation? Geburtshilfe Frauenheilkd 2020, 80, 932–940. [Google Scholar] [CrossRef]
- Hauke, J.; Wappenschmidt, B.; Faust, U.; Niederacher, D.; Wiesmüller, L.; Schmidt, G.; Groß, E.; Meindl, A.; Gehrig, A.; Sutter, C.; et al. Aktualisierte Kriterien des Deutschen Konsortiums Familiärer Brust- und Eierstockkrebs zur Klassifizierung von Keimbahn-Sequenzvarianten in Risikogenen für familiären Brust- und Eierstockkrebs. Senol. Z. Mammadiagnostik Ther. 2021, 18, 136–162. [Google Scholar] [CrossRef]
- Wappenschmidt, B.; Hauke, J.; Faust, U.; Niederacher, D.; Wiesmuller, L.; Schmidt, G.; Gross, E.; Gehrig, A.; Sutter, C.; Ramser, J.; et al. Criteria of the German Consortium for Hereditary Breast and Ovarian Cancer for the Classification of Germline Sequence Variants in Risk Genes for Hereditary Breast and Ovarian Cancer. Geburtshilfe Frauenheilkd 2020, 80, 410–429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef] [Green Version]
- Lee, A.; Mavaddat, N.; Wilcox, A.N.; Cunningham, A.P.; Carver, T.; Hartley, S.; Babb de Villiers, C.; Izquierdo, A.; Simard, J.; Schmidt, M.K.; et al. BOADICEA: A comprehensive breast cancer risk prediction model incorporating genetic and nongenetic risk factors. Genet. Med. 2019, 21, 1708–1718. [Google Scholar] [CrossRef] [Green Version]
- Lee, A.; Yang, X.; Tyrer, J.; Gentry-Maharaj, A.; Ryan, A.; Mavaddat, N.; Cunningham, A.P.; Carver, T.; Archer, S.; Leslie, G.; et al. Comprehensive epithelial tubo-ovarian cancer risk prediction model incorporating genetic and epidemiological risk factors. J. Med. Genet. 2022, 59, 632–643. [Google Scholar] [CrossRef]
- Carver, T.; Hartley, S.; Lee, A.; Cunningham, A.P.; Archer, S.; Babb de Villiers, C.; Roberts, J.; Ruston, R.; Walter, F.M.; Tischkowitz, M.; et al. CanRisk Tool-A Web Interface for the Prediction of Breast and Ovarian Cancer Risk and the Likelihood of Carrying Genetic Pathogenic Variants. Cancer Epidemiol. Biomark. Prev. 2021, 30, 469–473. [Google Scholar] [CrossRef]
- Bick, U.; Engel, C.; Krug, B.; Heindel, W.; Fallenberg, E.M.; Rhiem, K.; Maintz, D.; Golatta, M.; Speiser, D.; Rjosk-Dendorfer, D.; et al. High-risk breast cancer surveillance with MRI: 10-year experience from the German consortium for hereditary breast and ovarian cancer. Breast Cancer Res. Treat. 2019, 175, 217–228. [Google Scholar] [CrossRef]
- Wang, J.; Singh, P.; Yin, K.; Zhou, J.; Bao, Y.; Wu, M.; Pathak, K.; McKinley, S.K.; Braun, D.; Hughes, K.S. Disease Spectrum of Breast Cancer Susceptibility Genes. Front. Oncol. 2021, 11, 663419. [Google Scholar] [CrossRef] [PubMed]
- Weber-Lassalle, N.; Borde, J.; Weber-Lassalle, K.; Horvath, J.; Niederacher, D.; Arnold, N.; Kaulfuss, S.; Ernst, C.; Paul, V.G.; Honisch, E.; et al. Germline loss-of-function variants in the BARD1 gene are associated with early-onset familial breast cancer but not ovarian cancer. Breast Cancer Res. 2019, 21, 55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weber-Lassalle, N.; Hauke, J.; Ramser, J.; Richters, L.; Gross, E.; Blümcke, B.; Gehrig, A.; Kahlert, A.K.; Muller, C.R.; Hackmann, K.; et al. BRIP1 loss-of-function mutations confer high risk for familial ovarian cancer, but not familial breast cancer. Breast Cancer Res. 2018, 20, 7. [Google Scholar] [CrossRef]
- Hauke, J.; Horvath, J.; Gross, E.; Gehrig, A.; Honisch, E.; Hackmann, K.; Schmidt, G.; Arnold, N.; Faust, U.; Sutter, C.; et al. Gene panel testing of 5589 BRCA1/2-negative index patients with breast cancer in a routine diagnostic setting: Results of the German Consortium for Hereditary Breast and Ovarian Cancer. Cancer Med. 2018, 7, 1349–1358. [Google Scholar] [CrossRef] [PubMed]
- Kast, K.; Rhiem, K.; Wappenschmidt, B.; Hahnen, E.; Hauke, J.; Bluemcke, B.; Zarghooni, V.; Herold, N.; Ditsch, N.; Kiechle, M.; et al. Prevalence of BRCA1/2 germline mutations in 21 401 families with breast and ovarian cancer. J. Med. Genet. 2016, 53, 465–471. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harter, P.; Hauke, J.; Heitz, F.; Reuss, A.; Kommoss, S.; Marme, F.; Heimbach, A.; Prieske, K.; Richters, L.; Burges, A.; et al. Prevalence of deleterious germline variants in risk genes including BRCA1/2 in consecutive ovarian cancer patients (AGO-TR-1). PLoS ONE 2017, 12, e0186043. [Google Scholar] [CrossRef] [PubMed]
- Engel, C.; Rhiem, K.; Hahnen, E.; Loibl, S.; Weber, K.E.; Seiler, S.; Zachariae, S.; Hauke, J.; Wappenschmidt, B.; Waha, A.; et al. Prevalence of pathogenic BRCA1/2 germline mutations among 802 women with unilateral triple-negative breast cancer without family cancer history. BMC Cancer 2018, 18, 265. [Google Scholar] [CrossRef] [Green Version]
- Rolfes, M.; Borde, J.; Mollenhoff, K.; Kayali, M.; Ernst, C.; Gehrig, A.; Sutter, C.; Ramser, J.; Niederacher, D.; Horvath, J.; et al. Prevalence of Cancer Predisposition Germline Variants in Male Breast Cancer Patients: Results of the German Consortium for Hereditary Breast and Ovarian Cancer. Cancers 2022, 14, 3292. [Google Scholar] [CrossRef]
- Engel, C.; Wieland, K.; Zachariae, S.; Bucksch, K.; Enders, U.; Schoenwiese, U.; Yahiaoui-Doktor, M.; Keupp, K.; Waha, A.; Hahnen, E.; et al. HerediCaRe: Documentation and IT Solution of a Specialized Registry for Hereditary Breast and Ovarian Cancer. Gesundheitswesen 2021, 83, S12–S17. [Google Scholar] [CrossRef]
- Breast Cancer Association, C.; Dorling, L.; Carvalho, S.; Allen, J.; Gonzalez-Neira, A.; Luccarini, C.; Wahlstrom, C.; Pooley, K.A.; Parsons, M.T.; Fortuno, C.; et al. Breast Cancer Risk Genes—Association Analysis in More than 113,000 Women. N. Engl. J. Med. 2021, 384, 428–439. [Google Scholar] [CrossRef]
- Binion, S.; Sorgen, L.J.; Peshkin, B.N.; Valdimarsdottir, H.; Isaacs, C.; Nusbaum, R.; Graves, K.D.; DeMarco, T.; Wood, M.; McKinnon, W.; et al. Telephone versus in-person genetic counseling for hereditary cancer risk: Patient predictors of differential outcomes. J. Telemed. Telecare 2021. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Danylchuk, N.R.; Cook, L.; Shane-Carson, K.P.; Cacioppo, C.N.; Hardy, M.W.; Nusbaum, R.; Steelman, S.C.; Malinowski, J. Telehealth for genetic counseling: A systematic evidence review. J. Genet. Couns. 2021, 30, 1361–1378. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Felbor, U.; Bülow, R.; Schmutzler, R.K.; Rath, M. Hereditary Breast and Ovarian Cancer Service in Sparsely Populated Western Pomerania. Healthcare 2022, 10, 2021. https://doi.org/10.3390/healthcare10102021
Felbor U, Bülow R, Schmutzler RK, Rath M. Hereditary Breast and Ovarian Cancer Service in Sparsely Populated Western Pomerania. Healthcare. 2022; 10(10):2021. https://doi.org/10.3390/healthcare10102021
Chicago/Turabian StyleFelbor, Ute, Robin Bülow, Rita K. Schmutzler, and Matthias Rath. 2022. "Hereditary Breast and Ovarian Cancer Service in Sparsely Populated Western Pomerania" Healthcare 10, no. 10: 2021. https://doi.org/10.3390/healthcare10102021
APA StyleFelbor, U., Bülow, R., Schmutzler, R. K., & Rath, M. (2022). Hereditary Breast and Ovarian Cancer Service in Sparsely Populated Western Pomerania. Healthcare, 10(10), 2021. https://doi.org/10.3390/healthcare10102021





