Within-Host Phenotypic Evolution and the Population-Level Control of Chronic Viral Infections by Treatment and Prophylaxis
Abstract
1. Introduction
2. Description of the Models and Their Structural Properties
2.1. A Baseline Model of a Chronic Multi-Strain Virus Infection
2.2. A Generalized Model with Differentially Effective Control, Variable Transmissibility and Prophylaxis
- The efficacy of the treatment program depends on the viral strain. That is, the treatment program fails with certain probability, which varies depending on the virus strain, causing the treated individuals to thus revert back to active chronic infection.
- Virus strains have different levels of contagiousness.
- The efficacy of prophylactic measures depends on the viral strain. While on prophylaxis, an individual acquires protection against the virus depending on the specific viral strain.
2.3. Structural Analysis
3. Local Analysis at a Disease-Free Equilibrium
3.1. Basic Reproduction Number for the Baseline Model
- 1.
- Locally efficient if the respective sensitivity parameter is negative, i.e., ;
- 2.
- (Globally) efficient if there exists a non-negative value such that .
3.2. Basic Reproduction Number for the Extended Model
4. Endemic Equilibrium
Structure of the Matrix : Uniform within Host Mutations
5. Numerical Simulation for Different Scenarios and Illustration of Results
- Case 1.
- , . Assuming that one can compute the remaining probabilities using Equation (22): , , and . Finally, the endemic frequencies are .
- Case 2.
- , . Similarly to the previous case, we fix and compute the remaining probabilities , , and . The respective endemic frequencies are .
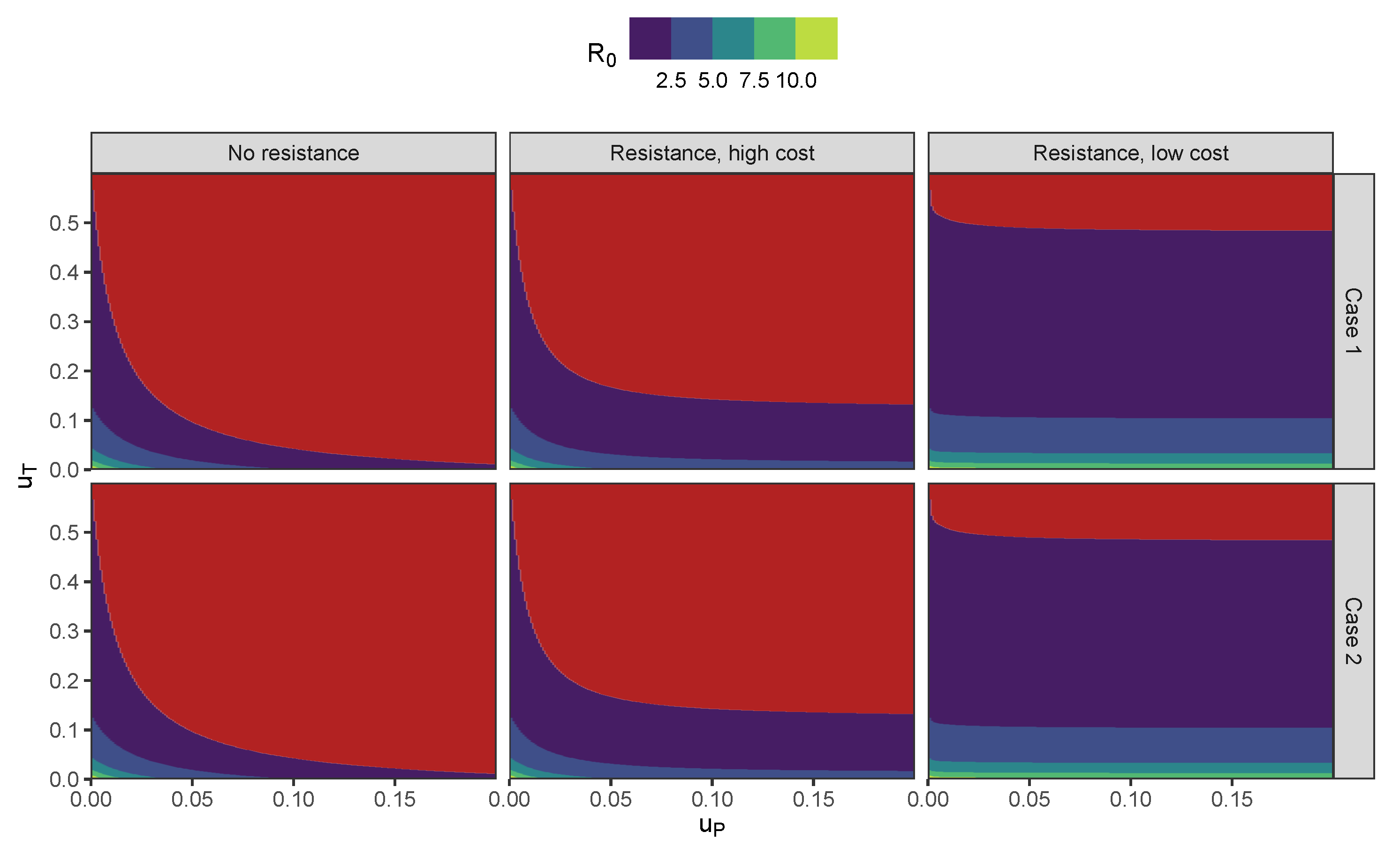
5.1. Controlled Basic Reproduction Number
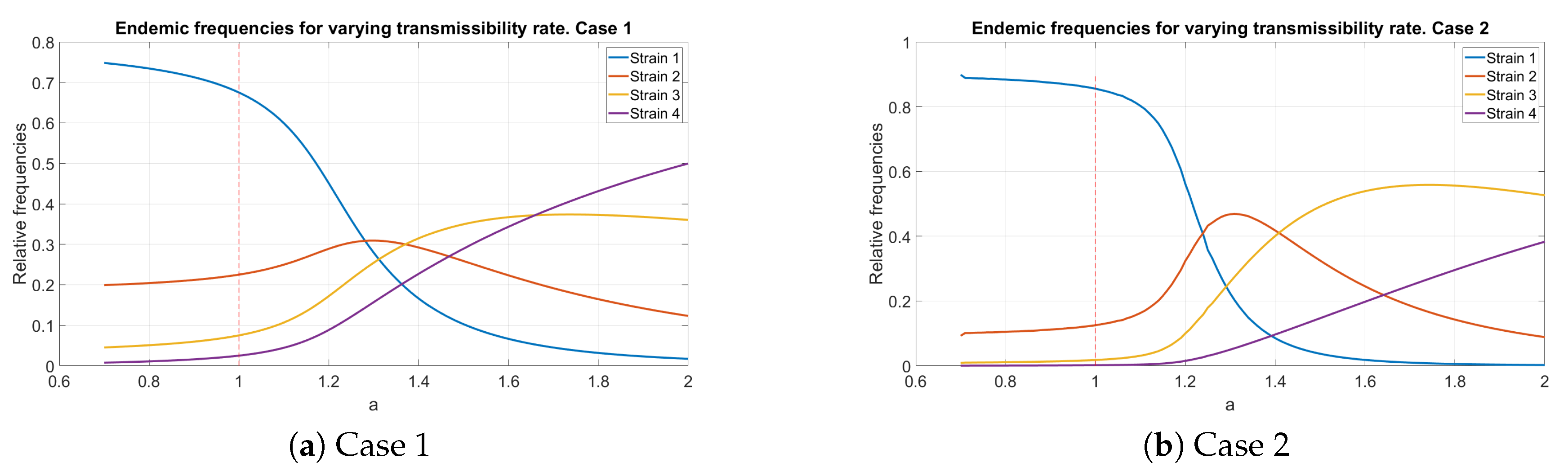
5.2. Endemic Distribution with Variable Transmissibility
5.3. Endemic Distribution with Variable Prophylaxis Effects
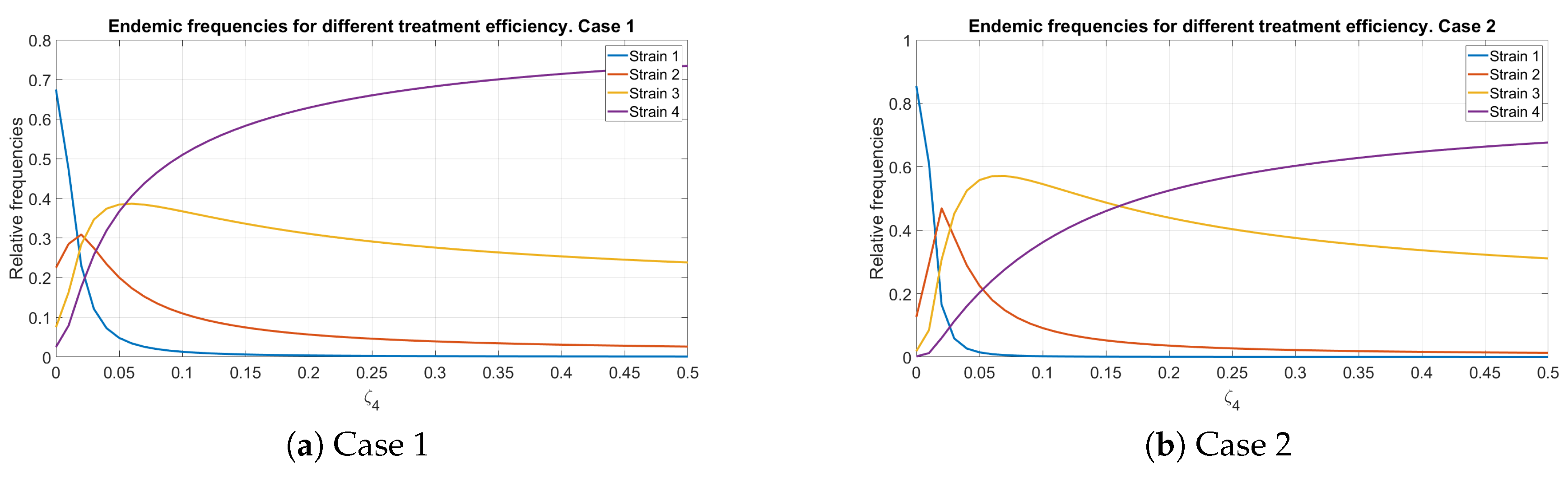
5.4. Endemic Distribution with Imperfect Treatment
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A. Necessary Ingredients from Matrix Algebra
Appendix A.1. Non-Negative Matrices
Appendix A.2. Stochastic Matrices
Appendix B. Proofs
References
- Melchjorsen, J.; Matikainen, S.; Paludan, S.R. Activation and Evasion of Innate Antiviral Immunity by Herpes Simplex Virus. Viruses 2009, 1, 737–759. [Google Scholar] [CrossRef] [PubMed]
- Perelson, A.S.; Neumann, A.U.; Markowitz, M.; Leonard, J.M.; Ho, D.D. HIV-1 Dynamics in Vivo: Virion Clearance Rate, Infected Cell Life-Span, and Viral Generation Time. Science 1996, 271, 1582–1586. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. HIV Drug Resistance Report 2019; Technical Report WHO/CDS/HIV/19.21; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Lythgoe, K.A.; Pellis, L.; Fraser, C. Is Hiv short-sighted? Insights from a multistrain nested model. Evolution 2013, 67, 2769–2782. [Google Scholar] [CrossRef] [PubMed]
- Kryazhimskiy, S.; Dieckmann, U.; Levin, S.A.; Dushoff, J. On State-Space Reduction in Multi-Strain Pathogen Models, with an Application to Antigenic Drift in Influenza A. PLoS Comput. Biol. 2007, 3, e159. [Google Scholar] [CrossRef] [PubMed]
- Minayev, P.; Ferguson, N. Improving the realism of deterministic multi-strain models: Implications for modelling influenza A. J. R. Soc. Interface 2008, 6, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Bianco, S.; Shaw, L.B.; Schwartz, I.B. Epidemics with multistrain interactions: The interplay between cross immunity and antibody-dependent enhancement. Chaos Interdiscip. J. Nonlinear Sci. 2009, 19, 043123. [Google Scholar] [CrossRef] [PubMed]
- Van der Ventel, B.I. Analysis of the multistrain asymmetric SI model for arbitrary strain diversity. Math. Comput. Model. 2011, 53, 1007–1025. [Google Scholar] [CrossRef]
- De Leenheer, P.; Pilyugin, S.S. Multistrain virus dynamics with mutations: A global analysis. Math. Med. Biol. J. IMA 2008, 25, 285–322. [Google Scholar] [CrossRef] [PubMed]
- Breban, R.; Drake, J.M.; Rohani, P. A general multi-strain model with environmental transmission: Invasion conditions for the disease-free and endemic states. J. Theor. Biol. 2010, 264, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Kooi, B.W.; Aguiar, M.; Stollenwerk, N. Bifurcation analysis of a family of multi-strain epidemiology models. J. Comput. Appl. Math. 2013, 252, 148–158. [Google Scholar] [CrossRef]
- Kucharski, A.J.; Andreasen, V.; Gog, J.R. Capturing the dynamics of pathogens with many strains. J. Math. Biol. 2016, 72, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Wikramaratna, P.S.; Kucharski, A.; Gupta, S.; Andreasen, V.; McLean, A.R.; Gog, J.R. Five challenges in modelling interacting strain dynamics. Epidemics 2015, 10, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Gromov, D.; Bulla, I.; Serea, O.S.; Romero-Severson, E.O. Numerical optimal control for HIV prevention with dynamic budget allocation. Math. Med. Biol. J. IMA 2017, 35, 469–491. [Google Scholar] [CrossRef] [PubMed]
- Van den Driessche, P.; Watmough, J. Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 2002, 180, 29–48. [Google Scholar] [CrossRef]
- Gromov, D.; Bulla, I.; Romero-Severson, E.O. Systematic evaluation of the population-level effects of alternative treatment strategies on the basic reproduction number. J. Theor. Biol. 2019, 462, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Wagh, K.; Seaman, M.S.; Zingg, M.; Fitzsimons, T.; Barouch, D.H.; Burton, D.R.; Connors, M.; Ho, D.D.; Mascola, J.R.; Nussenzweig, M.C.; et al. Potential of conventional & bispecific broadly neutralizing antibodies for prevention of HIV-1 subtype A, C & D infections. PLoS Pathog. 2018, 14, 1–24. [Google Scholar] [CrossRef]
- Shen, Z.; Xiao, Y.; Kang, L.; Ma, W.; Shi, L.; Zhang, L.; Zhou, Z.; Yang, J.; Zhong, J.; Yang, D.; et al. Genomic Diversity of Severe Acute Respiratory Syndrome–Coronavirus 2 in Patients with Coronavirus Disease 2019. Clin. Infect. Dis. 2020, 71, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Korber, B.; Fischer, W.M.; Gnanakaran, S.; Yoon, H.; Theiler, J.; Abfalterer, W.; Hengartner, N.; Giorgi, E.E.; Bhattacharya, T.; Foley, B.; et al. Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus. Cell 2020, 182, 812–827.e19. [Google Scholar] [CrossRef] [PubMed]
- Meyer, C.D. Matrix Analysis and Applied Linear Algebra; SIAM: Philadelphia, PA, USA, 2000. [Google Scholar]
- Kemeny, J.G.; Snell, J.L. Finite Markov Chains, 2nd ed.; Undergraduate Texts in Mathematics; Springer: Berlin/Heidelberg, Germany, 1976. [Google Scholar]
- Gursoy, B.B.; Kirkland, S.; Mason, O.; Sergeev, S. On the Markov chain tree theorem in the Max algebra. Electron. J. Linear Algebra 2013, 26, 2. [Google Scholar] [CrossRef][Green Version]
- Wicks, J.R. An Algorithm to Compute the Stochastically Stable Distribution of a Perturbed Markov Matrix. Ph.D. Thesis, Brown University, Providence, RI, USA, 2009. [Google Scholar]


| State Variable | Range | Description |
|---|---|---|
| Fraction of acutely infected individuals infected by the virus of type i. | ||
| Fraction of chronically infected individuals infected by the virus of type i. | ||
| S | Fraction of susceptible individuals | |
| T | Fraction of patients involved in treatment | |
| Fraction of patients infected by the virus of type i that are involved in treatment | ||
| Fraction of patients involved in prophylaxis | ||
| Parameter | Range | Description |
| Rate at which chronically infected are enrolled into treatment (controlled parameter) | ||
| Rate at which susceptible individuals are enrolled into prophylaxis (controlled parameter) | ||
| Inverse duration of the acute phase | ||
| Mortality rate | ||
| [0, 1] | Fraction of type i viruses in the viral population of an individual initially infected by the type j virus. | |
| , | Transmissibility rates of acute and chronically infected individuals. | |
| Proportionality coefficient of the transmissibility in acute and chronic stages | ||
| Failure rate of treatment for individuals infected by the virus of type i | ||
| Failure rate of prophylaxis | ||
| [0, 1] | The level of protection against the virus strain i, which is conferred by prophylaxis; corresponds to full protection |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gromov, D.; Romero-Severson, E.O. Within-Host Phenotypic Evolution and the Population-Level Control of Chronic Viral Infections by Treatment and Prophylaxis. Mathematics 2020, 8, 1500. https://doi.org/10.3390/math8091500
Gromov D, Romero-Severson EO. Within-Host Phenotypic Evolution and the Population-Level Control of Chronic Viral Infections by Treatment and Prophylaxis. Mathematics. 2020; 8(9):1500. https://doi.org/10.3390/math8091500
Chicago/Turabian StyleGromov, Dmitry, and Ethan O. Romero-Severson. 2020. "Within-Host Phenotypic Evolution and the Population-Level Control of Chronic Viral Infections by Treatment and Prophylaxis" Mathematics 8, no. 9: 1500. https://doi.org/10.3390/math8091500
APA StyleGromov, D., & Romero-Severson, E. O. (2020). Within-Host Phenotypic Evolution and the Population-Level Control of Chronic Viral Infections by Treatment and Prophylaxis. Mathematics, 8(9), 1500. https://doi.org/10.3390/math8091500






