1. Introduction
The task of segmenting anatomical structures in medical images is known to be challenged by significant inter-reader variability, which can impact the performance of subsequent supervised machine learning models. This issue is particularly pressing in the medical and biological domains, where labeled data are often limited due to the high costs associated with expert annotations. Across diverse fields and applications, differing biases and levels of expertise among annotators lead to considerable variations in segmentation annotations of structures in medical images [
1]. For instance, in the realm of assisted reproduction, expert annotators are primarily responsible for the segmentation of relevant structures, both in human and other mammalian data. For this kind of tasks, the UNet architecture [
2] has been used extensively in several medical and biological fields [
3]. Despite their expertise, disagreements and inconsistencies in annotations are not uncommon, adding another layer of complexity to the already challenging task. As a result, despite the proliferation of digital medical imaging data over the past couple of decades, access to data with curated labels that can be readily used for machine learning remains relatively scarce [
4]. This underlines the pressing need for advanced methodologies capable of robust learning from such noisy annotations.
Various pre-processing techniques are routinely employed to reconcile inter-reader variations and curate segmentation annotations by merging labels from multiple experts. A particularly common and straightforward approach utilizes the majority-vote approach [
5], whereby the ground truth is derived from the most frequently occurring opinion among the experts. This method has been effectively applied in various contexts, including Assisted Reproductive Technology (ART) for COC, as demonstrated by Athanasiou et al. [
6]. Furthermore, an advanced variant of this method, which considers class similarities, has demonstrated effectiveness in the aggregation of brain tumor segmentation labels [
1]. However, a primary limitation of these approaches is the assumption that all experts hold equal reliability. To circumvent this, Warfield et al. [
7] proposed a label fusion technique, known as STAPLE, which explicitly models the individual reliability of each expert and leverages this information to ‘weight’ their opinions during the label aggregation process. Due to its consistent superiority over traditional majority-vote pre-processing across various applications, STAPLE has emerged as the preferred label fusion method.
Recently, STAPLE has seen numerous enhancements, particularly in the context of multi-atlas segmentation problems [
8]. These enhancements particularly shine when image registration transfers segments from labeled images (“atlases”) to a new scan. A notable improvement, known as STEPS, was introduced by Cardoso et al. [
9]. This approach took things a step further by incorporating the local morphological similarity between atlases and target images. Numerous extensions of this approach, such as in the work of Asman et al. [
10], have since been explored. However, a shared limitation across these label fusion methods is the absence of a mechanism allowing information integration across different training images. This restriction essentially limits the applicability of these methods to cases where each image comes with a substantial number of annotations from multiple experts. In practice, obtaining such many annotations can be prohibitively expensive. Moreover, these methods often utilize relatively simplistic functions to model the relationship between observed noisy annotations, true labels, and expert reliability. As a result, they may fall short of capturing the complex characteristics of human annotators.
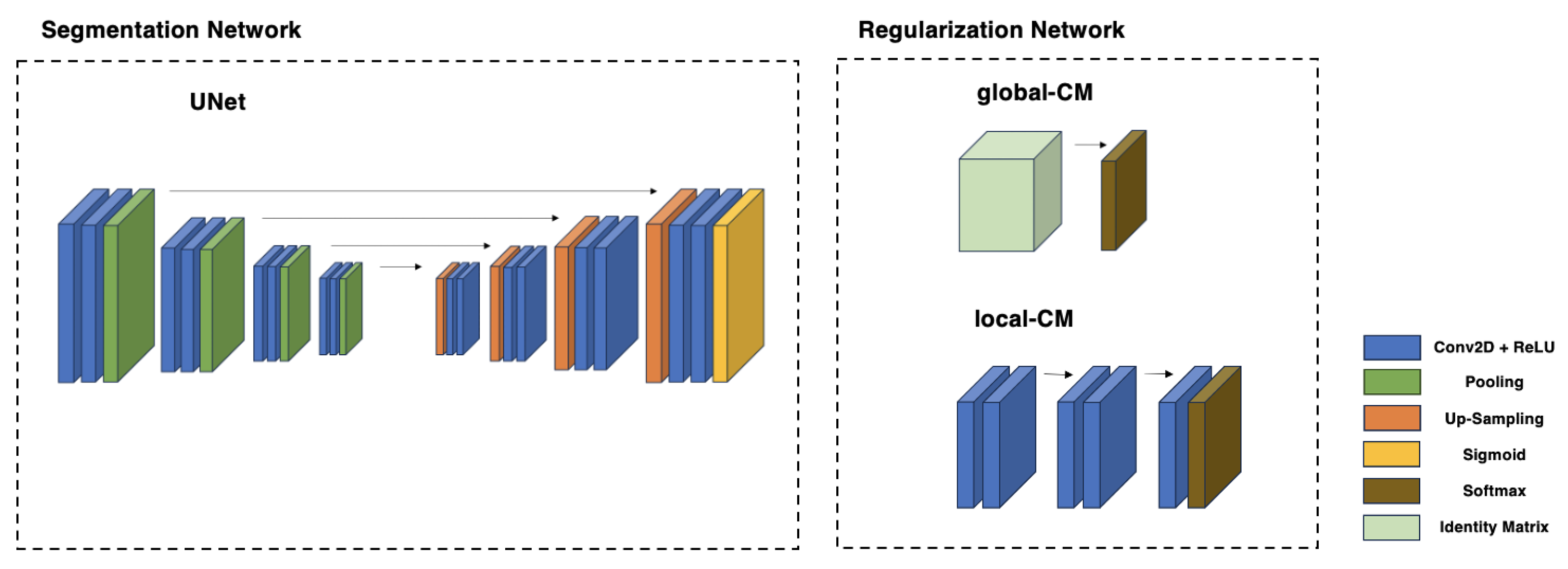
Several studies suggest solutions to these challenges by devising models that execute an end-to-end supervised segmentation process. This process aims to simultaneously determine the reliability of multiple human annotators and the true segmentation labels using only noisy labels. The proposed architectures [
11,
12,
13,
14] involve two integrated CNNs. The first one calculates the true segmentation probabilities. In contrast, the second characterizes individual annotators (e.g., tendencies to over-segment, mix-ups between different classes, etc.) by computing the pixel-wise confusion matrices (CMs) on a per-image basis. These models purport to outperform methods such as STAPLE and its variants by employing deep neural networks to unravel the complex mappings from the input images to the behaviors of the annotators and to the actual segmentation labels. Moreover, the CNNs’ parameters are “global variables” optimized over multiple image samples. This feature empowers the model to disentangle the true labels from the annotators’ errors robustly, drawing on correlations between analogous image samples. This robustness holds even when the number of available annotations per image is minimal (e.g., a single annotation per image). However, these techniques have not been tested on actual medical data yet, where challenges such as the absence of clear-cut differences among experts may occur, unlike in the artificially created data used in their previous studies. Furthermore, the feasibility of these architectures on small-sized datasets, with their unique challenges, has not been investigated yet. In this study, we implement the methods suggested in the literature [
12] on a small-sized dataset of COC, annotated by three experts. This marks the first instance where such techniques are employed with real-world medical data and masks from multiple experts, and not synthetic masks as in the previously mentioned work. Our findings reveal that while these techniques have potential, they may not be entirely effective in this context, and several challenges emerge, suggesting room for refinement in these proposals. Venturing a step further, we turn our attention towards areas of high uncertainty, as the majority of the areas exhibit considerable agreement among experts. By concentrating on these areas with high predictive uncertainty, we succeed in crafting a unique confusion matrix for each expert. This matrix serves as a signature of each expert’s annotation style within the field of Assisted Reproductive Technology.
We also establish a similar ‘identity’ for the final Deep Learning model and compare the behaviors of the model and human experts. Ultimately, we use these ‘identities’ within the uncertain areas to derive a new ground truth. This novel ground truth proves to be more reliable than the commonly employed majority-vote or STAPLE methods in the literature, thereby providing a promising direction for future work in this domain.
The structure of this paper is as follows: Following the introduction, we delve into related work in the field in
Section 2. This is followed by a detailed description of the methodology we adopted in
Section 3. Subsequently, we present our findings and engage in a thoughtful discussion about the implications and significance of our results in
Section 4. Finally, in
Section 5, we draw conclusions from our study and suggest possible directions for future work.
2. Related Work
Image segmentation in medical data has witnessed numerous advancements due to the introduction and utilization of the UNet architecture [
2]. There are numerous cases in the literature demonstrating the effectiveness of utilizing the UNet [
3]. Assessing the efficacy of image segmentation techniques is a longstanding challenge. Interactive segmentation by expert annotators is a commonly accepted practice, yet it introduces variability between different experts. A variety of methods has been put forth to estimate annotator skills and establish ground truth labels. These approaches can be broadly categorized into two groups: (i) two-stage approaches [
7,
15,
16,
17] and (ii) simultaneous approaches [
12,
13,
18,
19,
20].
In two-stage approaches, label aggregation and supervised model training occur independently. Initially, noisy labels are consolidated using a probabilistic model that incorporates unknown variables—annotator skills and ground truth labels. A machine learning model is then trained on ground truth labels to perform the task. Unfortunately, these methods often neglect raw input information during label aggregation, impacting estimated true labels.
The STAPLE algorithm was introduced by Warfield et al. [
15] in 2002. It characterizes each annotator
w through a confusion matrix
, where
L is the number of classes, and
denotes the probability that expert
w labels a pixel as
given
c in consensus. STAPLE employs Expectation Maximization [
7] for maximum likelihood consensus segmentation. Enhancements followed, including Spatial STAPLE [
16] and Local MAP STAPLE [
17]. Non-Local STAPLE (NLS) [
10] proposed by Asman and Landman addresses intensity inclusion in consensus computation. MOJITO [
21] minimizes Fréchet variance to counter STAPLE sensitivity. Carass et al. [
22] used STAPLE for a challenge, revealing experts’ superiority over algorithms.
Simultaneous approaches combine supervised model prediction with noisy label handling for enhanced accuracy. Employing expectation maximization, these methods require ample labels. However, real-life label collection constraints limit their practicality.
This category simultaneously curates labels and train models, achieving synergy. Despite favoring basic classification, these approaches improve predictive power and sample efficiency over the first category. Yet, structured prediction tasks with high-dimensional outputs lack attention. Zhang et al. [
12] introduced a supervised segmentation method that estimated annotator reliability and true labels using noisy data. While they evaluate their methodology in several synthetic scenarios, it has not been tested on real medical data with masks from multiple annotators. Zhang et al. [
13] refined this approach with crowdsourcing. Liu et al. [
18] shifted their focus to learning dynamics—a concept previously studied in classification problems but scarcely explored in the realm of image segmentation. They propose the ADELE model, but the results provided are not grounded in the core issue of multiple annotators with varying annotation methods. Other methods were proposed [
19,
20,
23,
24], but none handle real medical data from multiple experts.
Our work represents the first attempt at implementing a simultaneous approach to this tangible problem, using data directly provided by three distinct experts in the field.
4. Results and Discussion
In the following section, we present and discuss the outcomes derived from the application of the methodology outlined in the previous sections. These results correspond to (i) the performance of the dual-CNN framework as depicted in existing literature, along with the modifications that we introduced, (ii) the extraction and understanding of the unique annotating behavior profile exhibited by each expert, and (iii) the advancements made in developing a more sophisticated ground truth, focused on the uncertain areas. This analysis aims to shed light on the effectiveness of our multi-faceted approach for addressing the challenges inherent in the segmentation task.
4.1. Performance Coupled CNNs
The performance of the dual-CNN structure did not meet the expectations formed by its previous applications on artificial data. This observation held true across all different iterations, irrespective of the use of global or local CMs and regardless of the use of transfer learning. Multiple factors contributed to this discordance between the expected potential of the methodology and the ultimate results achieved.
Two key challenges became evident during our study. Firstly, the nature of real-world medical problems, such as the present COC investigation, inherently restricts the size of available data. In our scenario, the dataset was composed of a maximum of 200 COC images. Secondly, experts in the medical fields typically exhibit high accuracy in identifying the regions of COCs, thereby diminishing the degree of disagreement amongst them. This relatively high consensus among experts further complicates the task of segmenting and distinguishing various expert behaviors.
Eventually, neither the local CM’s nor the global CM’s strategy allowed the dual-CNN model to learn the segmentation of COC areas successfully. In both cases, the model began to diverge after a few epochs. Various attempts were made to tackle this issue, including adjusting multiple hyperparameters and the exploration of different values to manage the trace loss better. Despite these efforts, the performance remained poor, underscoring the challenge of adapting the model to this specific task.
In a preceding study by Athanasiou et al. [
6], transfer learning emerged as an indispensable tool to achieve high performance in COC area segmentation. The first approach, despite starting from an advantageous position in training, could not sustain high performance for both the local CM and global CM implementations. The second approach involved freezing the segmenting CNN to ensure its high performance, shifting the focus towards learning the CMs on the annotating CNN.
While this model could learn three different CMs, the values were strikingly similar across the experts. The True Positive (TP) and True Negative (TN) parts were extremely close to 1.0, and the False Positive (FP) and False Negative (FN) parts were extremely close to 0.0. Upon further examination, we discovered that this was to be expected, as there was substantial agreement among the experts on the majority of the pixels forming either part of the image background or the COC itself.
The same poor results were achieved even when a pre-trained network (by Athanasiou et al. [
6]) was used as the starting point for this approach. Despite its advantageous initial standing, the network could not maintain high performance when applied to either the local CM or global CM variants. In the latter approach, the segmentation CNN was ‘frozen’ to maintain its high performance, thus shifting the emphasis towards learning the CMs on the annotation CNN. Although this model was able to discern three different CMs, their values were strikingly alike across all the experts, with True Positive (TP) and True Negative (TN) values approximating 1.0 and False Positive (FP) and False Negative (FN) values near 0.0.
As it is already assumed, the high degree of consensus among experts on most of the pixels led to this poor outcome. Finally, we accepted that the proposed approach still needs improvements to fulfill the idea of their creators and alter our focus on the CMs.
4.2. Performance on CMs—Learning
In this phase of our work, our primary aim is to learn the unique CMs of each expert in areas of high uncertainty (
Figure 5). Given that the experts largely agree in their assessments, the only viable approach to characterize their distinct annotating “behaviors” lies in these contentious areas. Initially, the areas of high uncertainty are pinpointed. This is achieved through the use of our deep learning model, which discerns the zones where there is an elevated level of indecision regarding pixel classification, as described in
Section 3.4. Subsequently, we compare each pixel of every annotator’s mask to the majority-vote ground truth. This process is repeated five times, and ultimately, we synthesize a mean confusion matrix for each annotator drawn from the complete dataset. The resulting matrices, which shed light on the unique ‘identity’ of each expert, are illustrated in
Figure 6.
At this point, the approach of each expert towards the ambiguous areas—those with the highest likelihood of disagreement—has become clear. The first expert scores significantly high on the True Negative metric but averages on the True Positive one, indicating a greater tendency to categorize a pixel as part of the background and stricter criteria for identifying a pixel as part of the COC. Conversely, the third expert shows a somewhat opposing approach. He appears more lenient in categorizing pixels as part of the COC, even at the risk of falsely including background pixels in the COC region, as evidenced by his high True Positive score and average True Negative score. Lastly, the second expert sits in between these two extremes, exhibiting a more balanced approach. He demonstrates relatively high accuracy in correctly identifying pixels as part of the COC when they indeed are, and as parts of the background when they are not.
4.3. Ground Truth
Using the maximum likelihood approach as described in
Section 3.4.3, a new and more sophisticated method for computing the ground truth is proposed, incorporating the personal annotations of experts. An illustrative example of this novel ground truth computation is depicted in
Figure 7.
The first approach utilizes a majority vote strategy (
Figure 7a). In cases where three experts are involved, an uncertainty level of only 0.67 is achieved when two out of the three experts agree while the third expert disagrees. The second approach (
Figure 7b), however, leverages the previously derived confusion matrices from the annotation processes to gain insight into the annotators’ behaviors. This knowledge is then utilized to enhance the certainty level for each pixel beyond 0.67, encompassing a range of 0.67–1. Consequently, the final ground truth determination is fortified, as it has the capacity to identify erroneous pixels that may have been produced by the majority vote method.
Figure 7c exhibits the disparities in the areas of the image where the majority vote and maximum likelihood methods diverge.
The maximum likelihood method’s qualitative superiority over previous approaches is readily apparent. What makes our method stand out is how it tackles existing limitations. We have found that current methods fall short when applied to real-world annotation medical data from assisted reproduction. They were efficient with synthetic data or annotations by non-experts, but in real cases, they need improvement.
Our method is designed to address limitations that have been identified in current practices. It has come to light that these methods fall short when dealing with actual medical data from assisted reproduction, a significant departure from their performance with synthetic data or annotations by non-experts (crowdsourcing). A distinctive feature of our approach is its focus on creating customized profiles for each expert based on their annotations. These profiles, derived using confusion matrices, target areas where experts exhibit discrepancies or uncertainty. This capability is achieved by working with real-world annotation data that accurately capture genuine expert disagreement.
Furthermore, our approach allows us to discern the distinct annotation behaviors of each expert. This step holds importance as it enables the reuse of these behavioral patterns for future annotations, thereby sidestepping the need for repetitive experimentation. This not only conserves time and resources, but also bolsters model training accuracy. Notably, our approach does away with the prerequisite of an odd number of experts, which is a constraint imposed by prevailing methods.
A notable practical advantage of our methodology is its seamless integration into clinical and laboratory environments. This integration permits the preservation and application of personalized annotation profiles, streamlining the process of generating augmented results without the intricacies of sourcing and amalgamating numerous annotations.
In conclusion, our innovative approach revolutionizes the computation of ground truth. It actively addresses shortcomings in current methodologies while delivering tangible benefits in terms of precision and operational efficiency in medical image analysis.
5. Conclusions
The focus of the study was an exploration of multi-annotator segmentation using a COC dataset. This research consisted of three main steps: the application of a coupled CNN architecture to real medical data as proposed in the literature, a focus on uncertain areas to extract individual expert annotating profiles, and an attempt to optimize the ground truth by emphasizing areas of high uncertainty.
A primary finding of this investigation was the distinct disparity between results obtained from artificial data that have been used in previous works, and those from real-life medical datasets. Significantly, strategies that showed considerable success with artificial data proved unsatisfactory when applied to real-life medical data. This underscored the inherent complexities and unique characteristics of real-world annotation medical data, serving as a reminder that solutions developed in idealized environments may not necessarily translate directly to practical applications. Efforts to enhance the model’s performance using different strategies, such as transfer learning and variations in CMs, did not yield any notable improvements.
Another obstacle encountered was the difficulty in achieving a consensus among the experts. Although high agreement among experts is generally desirable, it presented a hurdle in the quest to improve the coupled CNN’s segmentation model using the individuals’ CMs, since minimal knowledge could be obtained in a highly agreeable environment. This suggests that models designed for scenarios with significant annotation disagreement may struggle when the discrepancies are minimal.
In response to these challenges, the focus shifted toward the regions of uncertainty within the data. An opportunity was found to delve deeper into understanding the individual behaviors of each expert, specifically in the context of these ambiguous areas. This introspection proved to be a fruitful endeavor, unveiling unique tendencies and preferences for each expert.
With this newfound understanding of annotator behaviors, a transition was made to redefine the ground truth (GT) from a more informed perspective. This was not an attempt to divorce the GT from expert annotations, but rather, it was a sophisticated endeavor to better integrate expert preferences into the construction of the GT. A novel GT was proposed and formulated based on maximum likelihood and with a specific focus on areas of uncertainty identified through the utilization of the deep learning model. This new approach surpasses the simplistic majority-vote strategy, providing a more sophisticated reflection of expert knowledge in the resulting GT.
The proposed method offers distinct advantages, primarily centered on addressing multi-labeling challenges by focusing on uncertain areas. Additionally, they create personalized annotating profiles for experts, enhancing and assessing future tasks. Moreover, these methods hold practical value in real-world settings, saving time and resources while overcoming odd-expert-number limitations for accurate ground truth determination.
In conclusion, this research illuminated the shortcomings of transferring methodologies developed from artificial data to real-world applications. However, through this exploration, the value of focusing on uncertain areas and individual expert behaviors was revealed. These insights fostered a more sophisticated construction of the ground truth, highlighting the potential for future research in multi-annotator segmentation.