Irrational Use of Medicines—A Summary of Key Concepts
Abstract
:1. Introduction
2. What is Rational Use of Medicines?
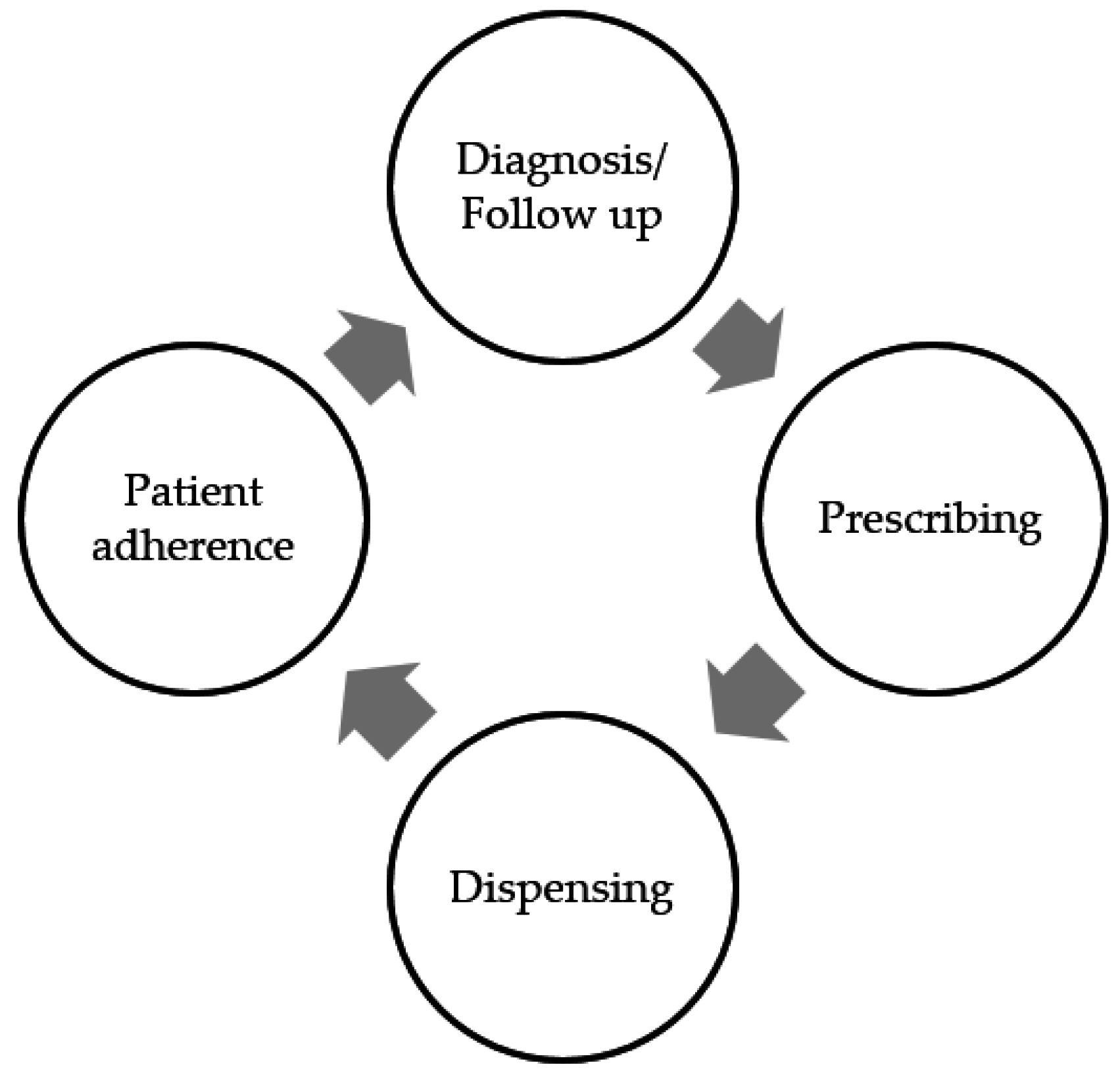
3. The Prescribing Process
4. What Constitutes a Good Prescribing?
“The NHS as a whole might define it as the lowest-cost prescribing that meets public health needs. The Department of Health and commissioners are keen to monitor prescribing and may measure good prescribing according to the available information and, as this largely relates to drug costs, their definitions of good prescribing tend to use cost as the focus. The pharmaceutical industry may look on good prescribing as prescribing of the latest drug to all patients who have need of treatment on the basis that new equals better. Evidence-based practitioners tend to define it as the use of therapies proven to be most effective in randomised controlled trials (RCTs), or according to evidence-based guidelines”.[27]
5. What is Irrational Prescribing?
6. The Art of Dispensing
7. Factors Contributing to the Irrational Use of Medicines
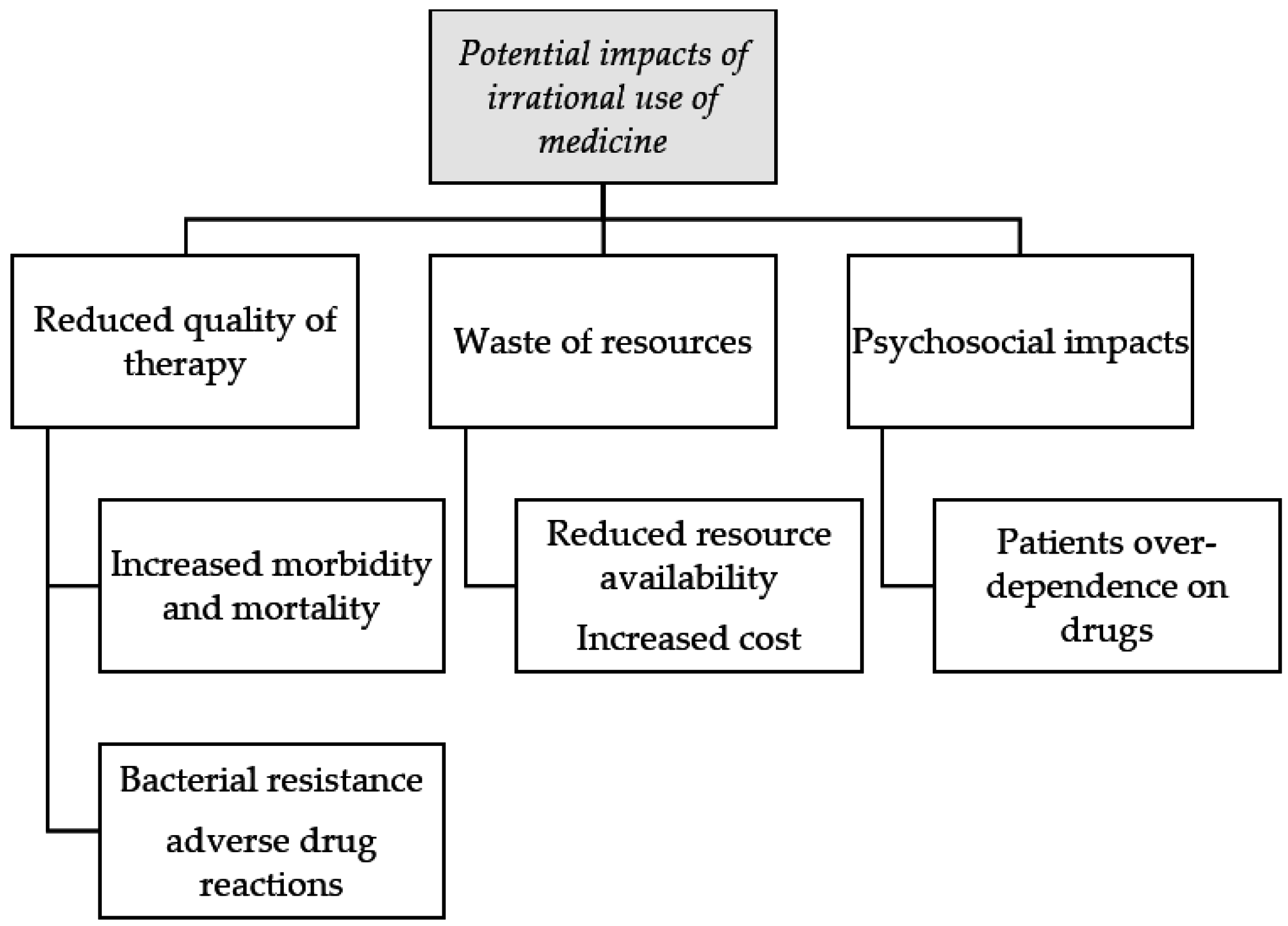
8. Impact of the Irrational Use of Medicines
9. Strategies to Tackle Irrational Prescribing
10. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. World Medicines Situation Report 2011; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- World Health Organization. The World Medicines Situation; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Ofori-Asenso, R.; Brhlikova, P.; Pollock, A.M. Prescribing indicators at primary health care centers within the WHO African region: A systematic analysis (1995–2015). BMC Public Health 2016, 16, 724. [Google Scholar] [CrossRef] [PubMed]
- Almarsdottir, A.B.; Traulsen, J.M. Rational use of medicines-an important issue in pharmaceutical policy. Pharm. World Sci. PWS 2005, 27, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Seiter, A. A Practical Approach to Pharmaceutical Policy; The World Bank: Washington, DC, USA, 2010. [Google Scholar]
- World Health Organization. Health Action International: Measuring Medicine Prices, Availability, Affordability and Price Components, 2nd ed.; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Shivhare, S.; Kunjwani, H.; Manikrao, A.; Bondre, A. Drug hazards and rational use of drugs: A review. J. Chem. Pharm. Res. 2010, 2, 106–112. [Google Scholar]
- Gopalakrishnan, S.; Udayshankar, P.M.; Rama, R. Standard treatment guidelines in primary healthcare practice. J. Fam. Med. Prim. Care 2014, 3, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Management Sciences for Health. MDS-3-Managing Access to Medicines and Health Technologies; Management Sciences for Health (MSH): Arlington, VA, USA, 2012. [Google Scholar]
- World Health Organization. Rational use of drugs: A review of major issues. In Proceedings of the Conference of Experts, Nairobi, Kenya, 22–29 November 1985.
- May, F. Whither ‘rational’ use of drugs? J. Pharm. Pract. Res. 2008, 38, 89–91. [Google Scholar] [CrossRef]
- Brahma, D.; Marak, M.; Wahlang, J. Rational use of drugs and irrational drug combinations. Internet J. Pharmacol. 2012, 10, 1. [Google Scholar]
- Laing, R.; Waning, B.; Gray, A.; Ford, N.; Hoen, E. 25 years of the WHO essential medicines lists: Progress and challenges. Lancet 2003, 361, 1723–1729. [Google Scholar] [CrossRef]
- Ofori-Asenso, R. A closer look at the World Health Organization’s prescribing indicators. J. Pharmacol. Pharmacother. 2016, 7, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Hogerzeil, H.V. The concept of essential medicines: Lessons for rich countries. BMJ 2004, 329, 1169–1172. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, S.R. Rational prescribing: The principles of drug selection. Clin. Med. 2016, 16, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Pollock, M.; Bazaldua, O.V.; Dobbie, A.E. Appropriate prescribing of medications: An eight-step approach. Am. Fam. Phys. 2007, 75, 231–236. [Google Scholar]
- Maxwell, S. Rational prescribing: The principles of drug selection. Clin. Med. 2009, 9, 481–485. [Google Scholar] [CrossRef]
- Claxton, A.J.; Cramer, J.; Pierce, C. A systematic review of the associations between dose regimens and medication compliance. Clin. Ther. 2001, 23, 1296–1310. [Google Scholar] [CrossRef]
- Elwyn, G.; Edwards, A.; Britten, N. What information do patients need about medicines? “Doing prescribing”: How doctors can be more effective. BMJ 2003, 327, 864–867. [Google Scholar] [CrossRef] [PubMed]
- Britten, N.; Stevenson, F.A.; Barry, C.A.; Barber, N.; Bradley, C.P. Misunderstandings in prescribing decisions in general practice: Qualitative study. BMJ 2000, 320, 484–488. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Roter, D.L.; Rand, C.S. Communication of affect between patient and physician. J. Health Soc. Behav. 1981, 22, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Ha, J.F.; Longnecker, N. Doctor-patient communication: A review. Ochsner J. 2010, 10, 38–43. [Google Scholar] [PubMed]
- Dong, R. Paternalism In Medical Decision Making; Duke University: Durham, NC, USA, 2011. [Google Scholar]
- Entwistle, V.A.; Carter, S.M.; Cribb, A.; McCaffery, K. Supporting patient autonomy: The importance of clinician-patient relationships. J. Gen. Intern. Med. 2010, 25, 741–745. [Google Scholar] [CrossRef] [PubMed]
- King, A.; Hoppe, R.B. “Best practice” for patient-centered communication: A narrative review. J. Grad. Med. Educ. 2013, 5, 385–393. [Google Scholar] [CrossRef] [PubMed]
- The King’s Fund. Improving the Quality of Care in General Practice; Report of an Independent Inquiry Commissioned by The King’s Fund; King’s Fund: London, UK, 2011. [Google Scholar]
- Aronson, J.K. Balanced prescribing. Br. J. Clin. Pharmacol. 2006, 62, 629–632. [Google Scholar] [CrossRef] [PubMed]
- Barber, N. What constitutes good prescribing? BMJ 1995, 310, 923–925. [Google Scholar] [CrossRef] [PubMed]
- Halczli, A.; Woolley, A. Medication underdosing and underprescribing: Issues that may contribute to polypharmacy, poor outcomes. Formulary 2013, 48, 194–196. [Google Scholar]
- Van den Heuvel, P.M.; Los, M.; van Marum, R.J.; Jansen, P.A. Polypharmacy and underprescribing in older adults: Rational underprescribing by general practitioners. J. Am. Geriatr. Soc. 2011, 59, 1750–1752. [Google Scholar] [CrossRef] [PubMed]
- Wauters, M.; Elseviers, M.; Vaes, B.; Degryse, J.; Dalleur, O.; Vander Stichele, R.; Christiaens, T.; Azermai, M. Too many, too few, or too unsafe? Impact of inappropriate prescribing on mortality, and hospitalization in a cohort of community-dwelling oldest old. Br. J. Clin. Pharmacol. 2016, 82, 1382–1392. [Google Scholar] [CrossRef] [PubMed]
- Llor, C.; Bjerrum, L. Antimicrobial resistance: Risk associated with antibiotic overuse and initiatives to reduce the problem. Ther. Adv. Drug Saf. 2014, 5, 229–241. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, M.N.; Gallagher, P.; O’Mahony, D. Inappropriate prescribing: Criteria, detection and prevention. Drugs Aging 2012, 29, 437–452. [Google Scholar] [CrossRef] [PubMed]
- Fiedorowicz, J.G.; Swartz, K.L. The role of monoamine oxidase inhibitors in current psychiatric practice. J. Psychiatr. Pract. 2004, 10, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Godman, B.; Shrank, W.; Andersen, M.; Berg, C.; Bishop, I.; Burkhardt, T.; Garuoliene, K.; Herholz, H.; Joppi, R.; Kalaba, M.; et al. Comparing policies to enhance prescribing efficiency in Europe through increasing generic utilization: Changes seen and global implications. Expert Rev. Pharmacoecon. Outcomes Res. 2010, 10, 707–722. [Google Scholar] [CrossRef] [PubMed]
- Godman, B.; Wettermark, B.; van Woerkom, M.; Fraeyman, J.; Alvarez-Madrazo, S.; Berg, C.; Bishop, I.; Bucsics, A.; Campbell, S.; Finlayson, A.E.; et al. Multiple policies to enhance prescribing efficiency for established medicines in Europe with a particular focus on demand-side measures: Findings and future implications. Front. Pharmacol. 2014, 5, 106. [Google Scholar] [CrossRef] [PubMed]
- Gurbani, N. Problems and impact of irrational medicines: Use and tools & interventions to improve medicines use. Pharm. Times 2011, 43, 7. [Google Scholar]
- Chou, Y.J.; Yip, W.C.; Lee, C.H.; Huang, N.; Sun, Y.P.; Chang, H.J. Impact of separating drug prescribing and dispensing on provider behaviour: Taiwan’s experience. Health Policy Plan. 2003, 18, 316–329. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.; Emery, J.D.; Lewis, J.; Sunderland, V.B. Australian dispensing doctors’ prescribing: Quantitative and qualitative analysis. Med. J. Aust. 2011, 195, 172–175. [Google Scholar] [PubMed]
- Wilcock, M. Dispensing doctors and non-dispensing doctors—A comparison of their prescribing costs. Int. J. Pharm. Pract. 2001, 9, 197–202. [Google Scholar] [CrossRef]
- Trap, B.; Hansen, E.H.; Hogerzeil, H.V. Prescription habits of dispensing and non-dispensing doctors in Zimbabwe. Health Policy Plan. 2002, 17, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Session Guide. Role of Dispensers in Promoting Rational Drug Use. Available online: http://archives.who.int/PRDUC2004/RDUCD/Session_Guides/role_of_dispensers_in_rational_d.htm (accessed on 24 October 2016).
- Dispensing Your Prescription Medicine: More Than Sticking a Label on a Bottle. Available online: https://www.guild.org.au/docs/default-source/public-documents/issues-and-resources/Fact-Sheets/the-dispensing-process.pdf?sfvrsn=4 (accessed on 24 October 2016).
- Md Rezal, R.S.; Hassali, M.A.; Alrasheedy, A.A.; Saleem, F.; Md Yusof, F.A.; Godman, B. Physicians’ knowledge, perceptions and behaviour towards antibiotic prescribing: A systematic review of the literature. Expert Rev. Anti-Infect. Ther. 2015, 13, 665–680. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, J.; Holmes, W.; Macfarlane, R.; Britten, N. Influence of patients’ expectations on antibiotic management of acute lower respiratory tract illness in general practice: Questionnaire study. BMJ 1997, 315, 1211–1214. [Google Scholar] [CrossRef] [PubMed]
- Denig, P.; Bradley, C. Prescribing in primary care. In How Doctors Choose Drugs; Hobbs, F., Bradley, C., Eds.; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Naicker, S.; Plange-Rhule, J.; Tutt, R.C.; Eastwood, J.B. Shortage of healthcare workers in developing countries—Africa. Ethn. Dis. 2009, 19, 60–64. [Google Scholar]
- Polage, C.R.; Bedu-Addo, G.; Owusu-Ofori, A.; Frimpong, E.; Lloyd, W.; Zurcher, E.; Hale, D.; Petti, C.A. Laboratory use in Ghana: Physician perception and practice. Am. J. Trop. Med. Hyg. 2006, 75, 526–531. [Google Scholar] [PubMed]
- Spurling, G.K.; Mansfield, P.R.; Montgomery, B.D.; Lexchin, J.; Doust, J.; Othman, N.; Vitry, A.I. Information from pharmaceutical companies and the quality, quantity, and cost of physicians’ prescribing: A systematic review. PLoS Med. 2010, 7, e1000352. [Google Scholar] [CrossRef] [PubMed]
- Ching, A.; Ishihara, M. The effects of detailing on prescribing decisions under quality uncertainty. Quant. Mark. Econ. 2010, 8, 123–165. [Google Scholar] [CrossRef]
- Davis, C.; Abraham, J. Is there a cure for corporate crime in the drug industry? BMJ 2013, 346, f755. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, H.J.; Gallagher, P.F.; O’Mahony, D. Inappropriate prescribing and adverse drug events in older people. BMC Geriatr. 2009, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Holman, C.D.; Price, S.D.; Sanfilippo, F.M.; Preen, D.B.; Bulsara, M.K. Comorbidity and repeat admission to hospital for adverse drug reactions in older adults: Retrospective cohort study. BMJ 2009, 338, a2752. [Google Scholar] [CrossRef] [PubMed]
- Godman, B.; Finlayson, A.E.; Cheema, P.K.; Zebedin-Brandl, E.; Gutierrez-Ibarluzea, I.; Jones, J.; Malmstrom, R.E.; Asola, E.; Baumgartel, C.; Bennie, M.; et al. Personalizing health care: Feasibility and future implications. BMC Med. 2013, 11, 179. [Google Scholar] [CrossRef] [PubMed]
- Rottenkolber, D.; Schmiedl, S.; Rottenkolber, M.; Farker, K.; Salje, K.; Mueller, S.; Hippius, M.; Thuermann, P.A.; Hasford, J. Adverse drug reactions in Germany: Direct costs of internal medicine hospitalizations. Pharmacoepidemiol. Drug Saf. 2011, 20, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Pirmohamed, M.; James, S.; Meakin, S.; Green, C.; Scott, A.K.; Walley, T.J.; Farrar, K.; Park, B.K.; Breckenridge, A.M. Adverse drug reactions as cause of admission to hospital: Prospective analysis of 18 820 patients. BMJ 2004, 329, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Lederberg, J. Infectious history. Science 2000, 288, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. Peer Rev. J. Formul. Manag. 2015, 40, 277–283. [Google Scholar]
- World Health Organization. Antimicrobial Resistance: Global Report on Surveillance; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Aina, B.A.; Tayo, F.; Taylor, O. Cost implication of irrational prescribing of chloroquine in Lagos State general hospitals. J. Infect. Dev. Ctries. 2008, 2, 68–72. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. World Medicines Strategy 2008–2013; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- World Health Organization. Teacher’s Guide to Good Prescribing; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- Hogerzeil, H. Promoting rational prescribing: An international perspective. Br. J. Clin. Pharmacol. 1995, 39, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kamarudin, G.; Penm, J.; Chaar, B.; Moles, R. Educational interventions to improve prescribing competency: A systematic review. BMJ Open 2013, 3, e003291. [Google Scholar] [CrossRef] [PubMed]
- Wettermark, B.; Godman, B.; Jacobsson, B.; Haaijer-Ruskamp, F.M. Soft regulations in pharmaceutical policy making: An overview of current approaches and their consequences. Appl. Health Econ. Health Policy 2009, 7, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, L.L.; Wettermark, B.; Godman, B.; Andersen-Karlsson, E.; Bergman, U.; Hasselstrom, J.; Hensjo, L.O.; Hjemdahl, P.; Jagre, I.; Julander, M.; et al. The ‘wise list’—A comprehensive concept to select, communicate and achieve adherence to recommendations of essential drugs in ambulatory care in Stockholm. Basic Clin. Pharmacol. Toxicol. 2011, 108, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Posner, J.; Griffin, J.P. Generic substitution. Br. J. Clin. Pharmacol. 2011, 72, 731–732. [Google Scholar] [CrossRef] [PubMed]
- The Pharmaceutical Benefits Scheme. Available online: http://www.pbs.gov.au/info/general/faq (accessed on 24 October 2016).
- Sabuncu, E.; David, J.; Bernede-Bauduin, C.; Pepin, S.; Leroy, M.; Boelle, P.Y.; Watier, L.; Guillemot, D. Significant reduction of antibiotic use in the community after a nationwide campaign in France, 2002–2007. PLoS Med. 2009, 6, e1000084. [Google Scholar] [CrossRef] [PubMed]
- Furst, J.; Cizman, M.; Mrak, J.; Kos, D.; Campbell, S.; Coenen, S.; Gustafsson, L.L.; Furst, L.; Godman, B. The influence of a sustained multifaceted approach to improve antibiotic prescribing in Slovenia during the past decade: Findings and implications. Expert Rev. Anti-Infect. Ther. 2015, 13, 279–289. [Google Scholar] [CrossRef] [PubMed]

| STEPS | ACTION |
|---|---|
| Accept and cross-check | Crosscheck prescription details
|
| Review | Script validity/Legality
|
Safety and appropriateness
| |
Check patients’ dispensing history
| |
| Prepare and check products | Product selection
|
Label and assemble dispensed products
| |
| Supply and counsel | Supply prescription to patient/carer: re-check
|
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ofori-Asenso, R.; Agyeman, A.A. Irrational Use of Medicines—A Summary of Key Concepts. Pharmacy 2016, 4, 35. https://doi.org/10.3390/pharmacy4040035
Ofori-Asenso R, Agyeman AA. Irrational Use of Medicines—A Summary of Key Concepts. Pharmacy. 2016; 4(4):35. https://doi.org/10.3390/pharmacy4040035
Chicago/Turabian StyleOfori-Asenso, Richard, and Akosua Adom Agyeman. 2016. "Irrational Use of Medicines—A Summary of Key Concepts" Pharmacy 4, no. 4: 35. https://doi.org/10.3390/pharmacy4040035
APA StyleOfori-Asenso, R., & Agyeman, A. A. (2016). Irrational Use of Medicines—A Summary of Key Concepts. Pharmacy, 4(4), 35. https://doi.org/10.3390/pharmacy4040035






