Do Invasive Mammal Eradications from Islands Support Climate Change Adaptation and Mitigation?
Abstract
1. Introduction
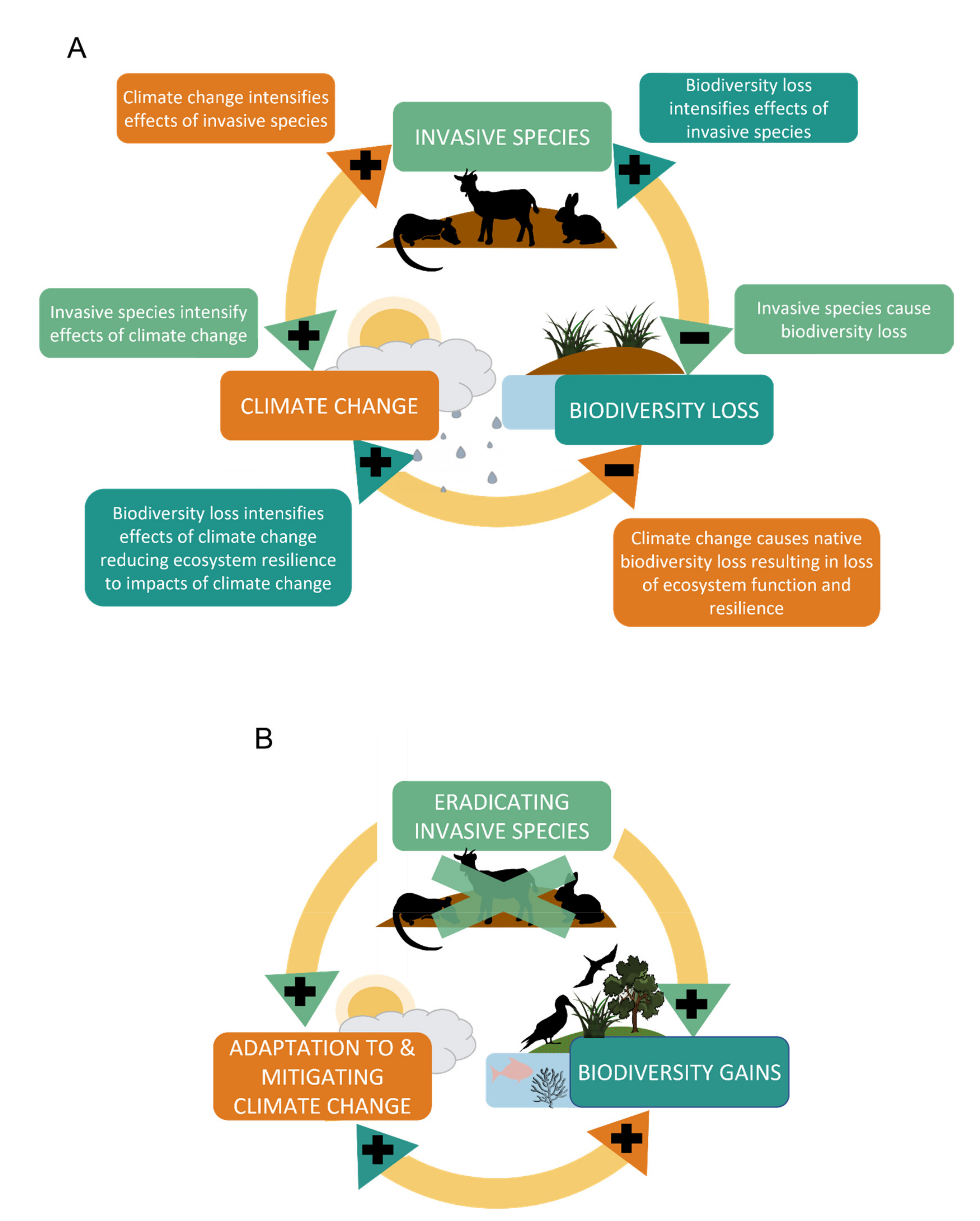
2. Climate Change, IAS, and Native Biodiversity Loss: The Positive Feedback Loop
| Synergistic Interaction | Focal IAS | Geography/Location | Impact | Determined | Reference |
|---|---|---|---|---|---|
| Review or summary papers documenting more than one type of synergistic interaction * | |||||
| Vertebrates, invertebrates, and plants | Global | Exacerbates and reduces IAS | Predicted and observed | [41,75,77,79,80,83] | |
| Australia’s World Heritage Sites | Exacerbates IAS | Predicted and observed | [81] | ||
| Antarctica and Southern Ocean | Exacerbates IAS | Predicted and observed | [84,104] | ||
| Australia | Exacerbates IAS | Predicted | [92] | ||
| Great Britain | Exacerbates and reduces IAS | Predicted | [105] | ||
| Plants, invertebrates, fishes, and birds | Global | Exacerbates and reduces IAS | Predicted | [78] | |
| Fish | Freshwater ecosystems | Exacerbates and reduces IAS | Predicted | [106] | |
| Top 100 most invasive species [107] | Global (including islands) | Exacerbates and reduces IAS | Predicted | [76] | |
| Climate change alters transport and introduction of IAS | |||||
| Tourism and new trade routes | Vertebrates, invertebrates, and plants | Global | Exacerbates IAS | Predicted and observed | [12] |
| Extreme weather events | African locusts (Schistocerca gregaria) | African windward islands | Exacerbates IAS | Observed | [108] |
| Plants | Coastal wetlands | Exacerbates IAS | Predicted | [109] | |
| Green iguana (Iguana iguana) | Anguilla | Exacerbates IAS | Observed | [86] | |
| Birds | Global | Exacerbates IAS | Observed | [88] | |
| Climate change alters distribution of existing IAS | |||||
| Vertebrates, invertebrates, and plants | Global | Exacerbates IAS | Predicted and observed | [110,111] | |
| Australia | Exacerbates IAS | Predicted | [92] | ||
| Norway rats (Rattus norvegicus) | South Georgia Island | Exacerbates IAS | Predicted | [91] | |
| Altered climatic constraints | |||||
| Creation of suitable habitat | Vertebrates, invertebrates, and plants | Global | Exacerbates IAS | Predicted and observed | [8,112] |
| Gastropods | Global | Exacerbates IAS | Predicted | [11] | |
| European rabbit | Tenerife, Canary Islands, Spain | Exacerbates IAS | Predicted | [93] | |
| Removal of barriers preventing IAS from establishing population | Norway rats | South Georgia Island | Exacerbates IAS | Predicted | [91] |
| Native species become invasive under altered climatic conditions | Vertebrates, invertebrates, and plants | Global | Exacerbates IAS | Observed | [113] |
| Mountain pine beetle (Dendroctonus ponderosae) | North American forests | Exacerbates IAS | Observed | [114] | |
| Extreme weather events | Rats (Rattus spp.) | Southeast Asia | Exacerbates IAS | Predicted and observed | [99] |
| Myanmar | Exacerbates IAS | Predicted and observed | [98] | ||
| Reduction in propagule pressure | Insects | Global | Exacerbates and reduces IAS | Predicted | [82] |
| Climate change alters impacts of existing IAS | |||||
| Vertebrates, invertebrates, and plants | Global | Exacerbates IAS | Predicted and observed | [111] | |
| House mouse | Sub-Antarctic Marion Island, South Africa | Exacerbates IAS | Observed | [90] | |
| European rabbit | Tenerife, Canary Islands, Spain | Exacerbates IAS | Predicted | [93] | |
| IAS outcompete native species under climate change | Maesopsis eminii | East Usambara mountain forests, Tanzania | Exacerbates IAS | Observed | [115] |
| Black rat (R. rattus) | Santiago Island, Galápagos Islands, Ecuador | Exacerbates IAS | Predicted | [97] | |
| Behavioral change | Feral cats (Felis catus) | San Clemente Island, California | Exacerbates IAS | Predicted and observed | [96] |
| Climate change alters effectiveness of IAS control and recovery of native biodiversity | |||||
| Seasonal limitations | European rabbit | Aotearoa/New Zealand | Exacerbates IAS | Observed and predicted | [116] |
| Kerguelen archipelago, France | Exacerbates IAS | Observed | [117] | ||
| IAS exacerbate climate change | |||||
| Reduction of carbon sequestration and release of carbon | Invertebrates and plants | Global | Exacerbates climate change | Predicted | [75] |
| Feral pigs | Global | Exacerbates climate change | Observed | [118] | |
| Mountain pine beetle | North American forests | Exacerbates climate change | Observed | [119] | |
| Rat spp. (Rattus spp.) | Aotearoa/New Zealand | Reduces climate change | Observed | [120] | |
| Destabilize coastal wetlands and anthropogenic flood-control structures | Nutria (Myocastor coypus) | Global | Exacerbates climate change | Predicted | [79] |
3. Evidence for Invasive Species Eradications as a Climate Adaptation Strategy
3.1. Invasive Mammal Eradications and Ecosystem Resilience
| Eradication | Focal Taxa | Location/Geography | Outcome | References |
|---|---|---|---|---|
| Enhance species/ecosystem resilience | ||||
| Improve native population (e.g., size, density, growth rate) or recruitment/rediscovery of extirpated native spp. (e.g., plants, seabirds) | Mammals | Global Islands | Positive: increases in population attributes and/or recruitment of new and/or extirpated species | [68] * |
| Positive: recovery of impacted populations | [54] * | |||
| Positive: increased population growth rate, nesting success, and enhanced adult survival | [69] * | |||
| Mammals | Aotearoa/New Zealand | Positive: recovery of numerous native plants, invertebrates, and >70 spp. of terrestrial vertebrates | [67,137] * | |
| Black rat (R. rattus) | Palmyra Atoll, Line Islands Central Pacific Ocean | Positive: increased recruitment of six native tree spp.; important for species dependent on these speciesNegative: increased recruitment of non-native tree spp. | [62] | |
| Create refugia | Invasive mammalian predators | Midway Atoll | Positive: creation of predator-free areas to recruit/translocate species at risk from sea-level rise | [138] |
| Lehua Island, Hawai’i, USA | Positive: creation of predator-free areas to recruit/translocate species at risk from sea-level rise | [139,140,141,142] | ||
| Restore food web and trophic dynamics, habitat quality | Rat (Rattus spp.) and feral cat (Felix catus) | Mercury Archipelago, Aotearoa/New Zealand | Positive: more diverse macroalgae communities at islands longer post eradication and more like never invaded islands | [128] |
| Mammals | Vanua Levu, Fiji | Positive: seabird-derived nutrient subsidies enhance growth of dominant reef-building spp. | [127] | |
| Rat spp. (Rattus spp.) | Aotearoa/New Zealand | Positive: seabird burrow density mediated soil and vegetation dynamics and were slightly higher on islands post eradication | [124] | |
| Norway rat (R. norvegicus) | Tromelin Island, Western Indian Ocean, France | Positive: increase in breeding pairs of two seabird species and recruitment of two seabird species; increase in vegetation coverNegative: increase in invasive House mouse (M. musculus) | [126] | |
| Hawadax Island, Aleutian Islands, Alaska | Positive: recovery of terrestrial birds, shorebirds, and recolonization by seabirds; community-level recovery: return of three-level trophic cascade in rocky intertidal, with decreases in invertebrate species and increases in fleshy algal cover | [143] | ||
| Black rat | Chagos Archipelago, Indian Ocean | Positive: seabird-derived subsidies enhance coral reef productivity and functioning and may increase resilience of reefs to climate change | [57,60,144,145] | |
| Reduce erosion, stabilize soils | Feral pig (Sus scrofa) | Isla del Coco, Costa Rica | Positive: vegetation recovery and reduction in erosion | [132] |
| O’ahu, Hawai’i, USA | Positive: Runoff volume was lower from plots excluding pigs | [135] | ||
| Feral sheep (Ovis aries) and cattle (Bos primigenius) | Santa Cruz Island, California | Positive: transition from grass-dominated systems to woody systems, increased woody vegetation and overstory | [133] | |
| Feral goat (Capra hircus) | Guadalupe Island, Mexico | Positive: recovery of native vegetation and rediscovery of species thought extinct or extirpated | [134] | |
| European rabbit (O. cuniculus) | Macquarie Island, Australia | Positive: recovery of native vegetation providing high-quality nesting habitat for three species of albatross, increased soil stabilization resulting in reduced erosion and increased reproductive success of albatrosses | [136] | |
| Kerguelen archipelago, France | Positive: combination of rabbits and climate change decimated plant cover and increased erosion; following eradication increased plant richness and reduction in erosionNegative: increased plant richness following eradication driven by invasive plant species adapted to warmer, drier climates due to climate change has resulted in an increase in soil erosion particularly where rabbits are still present | [117,146,147] | ||
| Mitigate climate change | ||||
| Increased carbon sequestration | Feral sheep and cattle | Santa Cruz Island, California | Positive: transition from grass-dominated systems to woody systems, increased woody vegetation and overstory result in 70% and 17% increase in stored carbon and nitrogen | [133] |
| Rat spp. | Aotearoa/New Zealand | Negative: islands with rats had higher rates of carbon sequestration | [120] | |
| Herbivores | Positive: removal resulted in increased carbon sequestration | [148] | ||
| European rabbits | Australia | Positive: removal of rabbits could be more effective way to sequester carbon than planting trees | [149] |
3.2. Invasive Mammal Eradication and the Creation of Refugia
3.3. Invasive Mammal Eradication and Climate Mitigation
4. Systematic Review of the Literature and Funding
4.1. Literature
4.2. Funding Sources: Awarded Projects
4.3. Literature and Funding Summary
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Doherty, T.S.; Glen, A.S.; Nimmo, D.G.; Ritchie, E.G.; Dickman, C.R. Invasive predators and global biodiversity loss. Proc. Natl. Acad. Sci. USA 2016, 113, 11261–11265. [Google Scholar] [CrossRef]
- Clavero, M.; Brotons, L.; Pons, P.; Sol, D. Prominent role of invasive species in avian biodiversity loss. Biol. Conserv. 2009, 142, 2043–2049. [Google Scholar] [CrossRef]
- Bellard, C.; Cassey, P.; Blackburn, T.M. Alien species as a driver of recent extinctions. Biol. Lett. 2016, 12, 20150623. [Google Scholar] [CrossRef] [PubMed]
- de Wit, L.A.; Croll, D.A.; Tershy, B.; Correa, D.; Luna-Pasten, H.; Quadri, P.; Kilpatrick, A.M. Potential public health benefits from cat eradications on islands. PLoS Neglect. Trop. Dis. 2019, 13, e0007040. [Google Scholar] [CrossRef] [PubMed]
- Reaser, J.K.; Meyerson, L.A.; Cronk, Q.; De Poorter, M.; Eldrege, L.G.; Green, E.; Kairo, M.; Latasi, P.; Mack, R.N.; Mauremootoo, J.; et al. Ecological and socioeconomic impacts of invasive alien species in island ecosystems. Environ. Conserv. 2007, 34, 98–111. [Google Scholar] [CrossRef]
- de Wit, L.A.; Croll, D.A.; Tershy, B.; Newton, K.M.; Spatz, D.R.; Holmes, N.D.; Kilpatrick, A.M. Estimating Burdens of Neglected Tropical Zoonotic Diseases on Islands with Introduced Mammals. Am. J. Trop. Med. Hyg. 2017, 96, 749–757. [Google Scholar] [CrossRef]
- Pyšek, P.; Hulme, P.E.; Simberloff, D.; Bacher, S.; Blackburn, T.M.; Carlton, J.T.; Dawson, W.; Essl, F.; Foxcroft, L.C.; Genovesi, P.; et al. Scientists’ warning on invasive alien species. Biol. Rev. 2020, 95, 1511–1534. [Google Scholar] [CrossRef] [PubMed]
- Meyerson, L.A.; Mooney, H.A. Invasive alien species in an era of globalization. Front. Ecol. Environ. 2007, 5, 199–208. [Google Scholar] [CrossRef]
- Westphal, M.I.; Browne, M.; MacKinnon, K.; Noble, I. The link between international trade and the global distribution of invasive alien species. Biol. Invasions 2008, 10, 391–398. [Google Scholar] [CrossRef]
- Chapman, D.; Purse, B.V.; Roy, H.E.; Bullock, J.M. Global trade networks determine the distribution of invasive non-native species. Glob. Ecol. Biogeogr. 2017, 26, 907–917. [Google Scholar] [CrossRef]
- Capinha, C.; Essl, F.; Seebens, H.; Moser, D.; Pereira, H.M. The dispersal of alien species redefines biogeography in the Anthropocene. Science 2015, 348, 1248–1251. [Google Scholar] [CrossRef]
- Seebens, H.; Blackburn, T.M.; Dyer, E.E.; Genovesi, P.; Hulme, P.E.; Jeschke, J.M.; Pagad, S.; Pysek, P.; Winter, M.; Arianoutsou, M.; et al. No saturation in the accumulation of alien species worldwide. Nat. Commun. 2017, 8, 14435. [Google Scholar] [CrossRef]
- Seebens, H.; Bacher, S.; Blackburn, T.M.; Capinha, C.; Dawson, W.; Dullinger, S.; Genovesi, P.; Hulme, P.E.; van Kleunen, M.; Kühn, I. Projecting the continental accumulation of alien species through to 2050. Glob. Chang. Biol. 2021, 27, 970–982. [Google Scholar] [CrossRef] [PubMed]
- McCann, K. Protecting biostructure. Nature 2007, 446, 29. [Google Scholar] [CrossRef] [PubMed]
- Russell, J.C.; Meyer, J.Y.; Holmes, N.D.; Pagad, S. Invasive alien species on islands: Impacts, distribution, interactions and management. Environ. Conserv. 2017, 44, 359–370. [Google Scholar] [CrossRef]
- SPREP. Pacific Invasive Species Battler Series: Protect Our Islands with Biosecurity; Secretariate of the Pacific Regional Environmental Programme (SPREP): Apia, Samoa, 2020. [Google Scholar]
- Stein, B.A.; Glick, P.; Edelson, N.; Staudt, A. Climate-Smart Conservation: Putting Adaption Principles into Practice; National Wildlife Federation: Washington, DC, USA, 2014. [Google Scholar]
- IPCC. Global warming of 1.5 °C. In An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty; IPCC: Geneva, Switzerland, 2018. [Google Scholar]
- Folke, C. Resilience: The emergence of a perspective for social-ecological systems analyses. Glob. Environ. Chang.-Hum. Policy Dimens. 2006, 16, 253–267. [Google Scholar] [CrossRef]
- Nelson, D.R.; Adger, W.N.; Brown, K. Adaptation to environmental change: Contributions of a resilience framework. Annu. Rev. Environ. Resour. 2007, 32, 395–419. [Google Scholar] [CrossRef]
- IUCN/SSC. Guidelines for Reintroductions and Other Conservation Translocations; Version 1.0; IUCN Species Survival Commission: Gland, Switzerland, 2013. [Google Scholar]
- Futuyma, D.J.; Kirkpatrick, M. Evolution, 4th ed.; Sinauer Associates, Inc., Publishers: Sunderland, MA, USA, 2017. [Google Scholar]
- Cromarty, P.; Broome, K.; Cox, A.; Empson, R.; Hutchinson, W. Eradication planning for invasive alien animal species on islands-the approach developed by the New Zealand Department of Conservation. In Turning the Tide: The Eradication of Invasive Species, Proceedings of the International Conference on Eradication of Island Invasives, Auckland, New Zealand, 19–23 February 2001; IUCN Species Specialist Group: Gland, Switzerland, 2002; pp. 85–91. [Google Scholar]
- IUCN. Invasive Alien Species and Climate Change; International Union for Conservation of Nature and Natural Resources: Gland, Switzerland, 2021; pp. 1–2. [Google Scholar]
- DeLong, D.C. Defining Biodiversity. Wildl. Soc. Bull. 1996, 24, 738–749. [Google Scholar]
- Blackburn, T.M.; Pyšek, P.; Bacher, S.; Carlton, J.T.; Duncan, R.P.; Jarošík, V.; Wilson, J.R.; Richardson, D.M. A proposed unified framework for biological invasions. Trends Ecol. Evol. 2011, 26, 333–339. [Google Scholar] [CrossRef]
- Essl, F.; Bacher, S.; Genovesi, P.; Hulme, P.E.; Jeschke, J.M.; Katsanevakis, S.; Kowarik, I.; Kuehn, I.; Pysek, P.; Rabitsch, W.; et al. Which Taxa Are Alien? Criteria, Applications, and Uncertainties. Bioscience 2018, 68, 496–509. [Google Scholar] [CrossRef]
- Williamson, M.H. Biological Invasions, 1st ed.; Chapman and Hall: New York, NY, USA, 1996; p. 244. [Google Scholar]
- Keppel, G.; Mokany, K.; Wardell-Johnson, G.W.; Phillips, B.L.; Welbergen, J.A.; Reside, A.E. The capacity of refugia for conservation planning under climate change. Front. Ecol. Environ. 2015, 13, 106–112. [Google Scholar] [CrossRef]
- United Nations Environment Programme. Making Peace with Nature: A Scientific Bluepring to Tackle the Climate, Biodiversity and Pollution Emergencies; UNEP: Nairobi, Kenya, 2021. [Google Scholar]
- Butchart, S.H.M.; Walpole, M.; Collen, B.; van Strien, A.; Scharlemann, J.P.W.; Almond, R.E.A.; Baillie, J.E.M.; Bomhard, B.; Brown, C.; Bruno, J.; et al. Global Biodiversity: Indicators of Recent Declines. Science 2010, 328, 1164–1168. [Google Scholar] [CrossRef] [PubMed]
- Novacek, M.J.; Cleland, E.E. The current biodiversity extinction event: Scenarios for mitigation and recovery. Proc. Natl. Acad. Sci. USA 2001, 98, 5466–5470. [Google Scholar] [CrossRef] [PubMed]
- Courchamp, F.; Hoffmann, B.D.; Russell, J.C.; Leclerc, C.; Bellard, C. Climate change, sea-level rise, and conservation: Keeping island biodiversity afloat. Trends Ecol. Evol. 2014, 29, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Dinerstein, E.; Joshi, A.R.; Vynne, C.; Lee, A.T.L.; Pharand-Deschenes, F.; Franca, M.; Fernando, S.; Birch, T.; Burkart, K.; Asner, G.P.; et al. A “Global Safety Net” to reverse biodiversity loss and stabilize Earth’s climate. Sci. Adv. 2020, 6, eabb2824. [Google Scholar] [CrossRef]
- Waller, N.L.; Gynther, I.C.; Freeman, A.B.; Lavery, T.H.; Leung, L.K.P. The Bramble Cay melomys Melomys rubicola (Rodentia: Muridae): A first mammalian extinction caused by human-induced climate change? Wildl. Res. 2017, 44, 9–21. [Google Scholar] [CrossRef]
- Fulton, G.R. The Bramble Cay melomys: The first mammalian extinction due to human-induced climate change. Pac. Conserv. Biol. 2017, 23, 1–3. [Google Scholar] [CrossRef]
- Bellard, C.; Bertelsmeier, C.; Leadley, P.; Thuiller, W.; Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol. Lett. 2012, 15, 365–377. [Google Scholar] [CrossRef]
- Urban, M.C. Climate change. Accelerating extinction risk from climate change. Science 2015, 348, 571–573. [Google Scholar] [CrossRef]
- Staude, I.R.; Navarro, L.M.; Pereira, H.M.; Storch, D. Range size predicts the risk of local extinction from habitat loss. Glob. Ecol. Biogeogr. 2020, 29, 16–25. [Google Scholar] [CrossRef]
- Newbold, T.; Hudson, L.N.; Contu, S.; Hill, S.L.L.; Beck, J.; Liu, Y.; Meyer, C.; Phillips, H.R.P.; Scharlemann, J.P.W.; Purvis, A. Widespread winners and narrow-ranged losers: Land use homogenizes biodiversity in local assemblages worldwide. PLoS Biol. 2018, 16, e2006841. [Google Scholar] [CrossRef]
- Manes, S.; Costello, M.J.; Beckett, H.; Debnath, A.; Devenish-Nelson, E.; Grey, K.A.; Jenkins, R.; Khan, T.M.; Kiessling, W.; Krause, C.; et al. Endemism increases species’ climate change risk in areas of global biodiversity importance. Biol. Conserv. 2021, 257, 109070. [Google Scholar] [CrossRef]
- Bellard, C.; Leclerc, C.; Leroy, B.; Bakkenes, M.; Veloz, S.; Thuiller, W.; Courchamp, F. Vulnerability of biodiversity hotspots to global change. Glob. Ecol. Biogeogr. 2014, 23, 1376–1386. [Google Scholar] [CrossRef]
- Bellard, C.; Leclerc, C.; Courchamp, F. Impact of sea level rise on the 10 insular biodiversity hotspots. Glob. Ecol. Biogeogr. 2014, 23, 203–212. [Google Scholar] [CrossRef]
- Caujape-Castells, J.; Tye, A.; Crawford, D.J.; Santos-Guerra, A.; Sakai, A.; Beaver, K.; Lobin, W.; Florens, F.B.V.; Moura, M.; Jardim, R.; et al. Conservation of oceanic island floras: Present and future global challenges. Perspect. Plant Ecol. Evol. Syst. 2010, 12, 107–129. [Google Scholar] [CrossRef]
- Kier, G.; Kreft, H.; Lee, T.M.; Jetz, W.; Ibisch, P.L.; Nowicki, C.; Mutke, J.; Barthlott, W. A global assessment of endemism and species richness across island and mainland regions. Proc. Natl. Acad. Sci. USA 2009, 106, 9322–9327. [Google Scholar] [CrossRef]
- Tershy, B.R.; Shen, K.-W.; Newton, K.M.; Holmes, N.D.; Croll, D.A. The importance of islands for the protection of biological and linguistic diversity. Bioscience 2015, 65, 592–597. [Google Scholar] [CrossRef]
- Russell, J.C.; Kueffer, C. Island Biodiversity in the Anthropocene. Annu. Rev. Environ. Resour. 2019, 44, 31–60. [Google Scholar] [CrossRef]
- Spatz, D.R.; Zilliacus, K.M.; Holmes, N.D.; Butchart, S.H.M.; Genovesi, P.; Ceballos, G.; Tershy, B.R.; Croll, D.A. Globally threatened vertebrates on islands with invasive species. Sci. Adv. 2017, 3, e1603080. [Google Scholar] [CrossRef]
- Dawson, W.; Moser, D.; van Kleunen, M.; Kreft, H.; Pergl, J.; Pyšek, P.; Weigelt, P.; Winter, M.; Lenzner, B.; Blackburn, T.M.; et al. Global hotspots and correlates of alien species richness across taxonomic groups. Nat. Ecol. Evol. 2017, 1, 0186. [Google Scholar] [CrossRef]
- Baillie, J.E.M.; Hilton-Taylor, C. 2004 IUCN Red List of Threatened Species: A Global Species Assessment; Stuart, S.N., Ed.; IUCN: Gland, Switzerland, 2004; p. 191. [Google Scholar]
- Vitousek, P.M. Diversity and biological invasions of oceanic islands. In Biodiversity; Wilson, E.O., Peter, F.M., Eds.; National Academy Press: Washington, DC, USA, 1988; pp. 181–189. [Google Scholar]
- Blackburn, T.M.; Cassey, P.; Duncan, R.P.; Evans, K.L.; Gaston, K.J. Avian extinction and mammalian introductions on oceanic islands. Science 2004, 305, 1955–1958. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.; Harper, G.; Algar, D.; Hanson, C.; Keitt, B.; Robinson, S. Review of feral cat eradications on islands. In Island Invasives: Eradication and Management; Veitch, C.R., Clout, M.N., Towns, D.R., Eds.; IUCN: Gland, Switzerland, 2011; pp. 37–46. [Google Scholar]
- Courchamp, F.; Chapuis, J.L.; Pascal, M. Mammal invaders on islands: Impact, control and control impact. Biol. Rev. 2003, 78, 347–383. [Google Scholar] [CrossRef]
- Hilton, G.M.; Cuthbert, R.J. The catastrophic impact of invasive mammalian predators on birds of the UK Overseas Territories: A review and synthesis. Ibis 2010, 152, 443–458. [Google Scholar] [CrossRef]
- Hamann, O. Regeneration of vegetation on Santa Fe and Pinta islands, Galápagos, after the eradication of goats. Biol. Conserv. 1979, 15, 215–236. [Google Scholar] [CrossRef]
- Graham, N.A.J.; Wilson, S.K.; Carr, P.; Hoey, A.S.; Jennings, S.; MacNeil, M.A. Seabirds enhance coral reef productivity and functioning in the absence of invasive rats. Nature 2018, 559, 250. [Google Scholar] [CrossRef]
- Towns, D.R.; Atkinson, I.A.E.; Daugherty, C.H. Have the harmful effects of introduced rats on islands been exaggerated? Biol. Invasions 2006, 8, 863–891. [Google Scholar] [CrossRef]
- Dias, M.P.; Martin, R.; Pearmain, E.J.; Burfield, I.J.; Lascelles, B.; Croxall, J.P.; Small, C.; Phillips, R.A.; Yates, O.; Borboroglu, P.G. Threats to seabirds: A global assessment. Biol. Conserv. 2019, 237, 525–537. [Google Scholar] [CrossRef]
- Benkwitt, C.E.; Wilson, S.K.; Graham, N.A.J. Biodiversity increases ecosystem functions despite multiple stressors on coral reefs. Nat. Ecol. Evol. 2020, 4, 919–926. [Google Scholar] [CrossRef]
- Kurle, C.M.; Croll, D.A.; Tershy, B.R. Introduced rats indirectly change marine rocky intertidal communities from algae-to invertebrate-dominated. Proc. Natl. Acad. Sci. USA 2008, 105, 3800–3804. [Google Scholar] [CrossRef]
- Wolf, C.A.; Young, H.S.; Zilliacus, K.M.; Wegmann, A.S.; McKown, M.; Holmes, N.D.; Tershy, B.R.; Dirzo, R.; Kropidlowski, S.; Croll, D.A. Invasive rat eradication strongly impacts plant recruitment on a tropical atoll. PLoS ONE 2018, 13, e0200743. [Google Scholar] [CrossRef]
- MacArthur, R.H.; Wilson, E.O. The Theory of Island Biogeography; Princeton University Press: Princeton, NJ, USA, 1967; p. 203. [Google Scholar]
- Borregaard, M.K.; Amorim, I.R.; Borges, P.A.V.; Cabral, J.S.; Fernadez-Palacios, J.M.; Field, R.; Heaney, L.R.; Kreft, H.; Matthews, T.J.; Olesen, J.M.; et al. Oceanic island biogeography through the lens of the general dynamic model: Assessment and prospect. Biol. Rev. 2017, 92, 830–853. [Google Scholar] [CrossRef] [PubMed]
- Moser, D.; Lenzner, B.; Weigelt, P.; Dawson, W.; Kreft, H.; Pergl, J.; Pysek, P.; van Kleunen, M.; Winter, M.; Capinha, C.; et al. Remoteness promotes biological invasions on islands worldwide. Proc. Natl. Acad. Sci. USA 2018, 115, 9270–9275. [Google Scholar] [CrossRef]
- Lenzner, B.; Latombe, G.; Capinha, C.; Bellard, C.; Courchamp, F.; Diagne, C.; Dullinger, S.; Golivets, M.; Irl, S.D.H.; Kuhn, I.; et al. What Will the Future Bring for Biological Invasions on Islands? An Expert-Based Assessment. Front. Ecol. Evol. 2020, 8, 280. [Google Scholar] [CrossRef]
- Towns, D.R.; West, C.J.; Broome, K.G. Purposes, outcomes and challenges of eradicating invasive mammals from New Zealand islands: An historical perspective. Wildl. Res. 2013, 40, 94–107. [Google Scholar] [CrossRef]
- Jones, H.P.; Holmes, N.D.; Butchart, S.H.M.; Tershy, B.R.; Kappes, P.J.; Corkery, I.; Aguirre-Munoz, A.; Armstrong, D.P.; Bonnaud, E.; Burbidge, A.A.; et al. Invasive mammal eradication on islands results in substantial conservation gains. Proc. Natl. Acad. Sci. USA 2016, 113, 4033–4038. [Google Scholar] [CrossRef]
- Brooke, M.D.; Bonnaud, E.; Dilley, B.J.; Flint, E.N.; Holmes, N.D.; Jones, H.P.; Provost, P.; Rocamora, G.; Ryan, P.G.; Surman, C.; et al. Seabird population changes following mammal eradications on islands. Anim. Conserv. 2018, 21, 3–12. [Google Scholar] [CrossRef]
- de Wit, L.A.; Zilliacus, K.M.; Quadri, P.; Will, D.; Grima, N.; Spatz, D.; Holmes, N.; Tershy, B.; Howald, G.R.; Croll, D.A. Invasive vertebrate eradications on islands as a tool for implementing global Sustainable Development Goals. Environ. Conserv. 2020, 47, 139–148. [Google Scholar] [CrossRef]
- DIISE. The Database of Island Invasive Species Eradications, Developed by Island Conservation, Coastal Conservation Action Laboratory UCSC, IUCN SSC Invasive Species Specialist Group, University of Auckland and Landcare Research New Zealand. Available online: http://diise.islandconservation.org (accessed on 19 July 2021).
- Glen, A.S.; Atkinson, R.; Campbell, K.J.; Hagen, E.; Holmes, N.D.; Keitt, B.S.; Parkes, J.P.; Saunders, A.; Sawyer, J.; Torres, H. Eradicating multiple invasive species on inhabited islands: The next big step in island restoration? Biol. Invasions 2013, 15, 2589–2603. [Google Scholar] [CrossRef]
- Secretariat of the Convention of Biological Diversity. Global Biodiversity Outlook 4. Available online: https://www.cbd.int/gbo/gbo4/publication/gbo4-en-hr.pdf (accessed on 28 February 2021).
- USGCRP. Impacts, Risks, and Adaptation in the United States: Fourth National Climate Assessment; Reidmiller, D., Avery, C., Easterling, D., Kunkel, K., Lewis, K., Maycock, T.K., Stewart, B., Eds.; U.S. Global Change Research Program: Washington, DC, USA, 2018; Volume 2. [Google Scholar]
- Mainka, S.A.; Howard, G.W. Climate change and invasive species: Double jeopardy. Integr. Zool. 2010, 5, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Bellard, C.; Thuiller, W.; Leroy, B.; Genovesi, P.; Bakkenes, M.; Courchamp, F. Will climate change promote future invasions? Glob. Chang. Biol. 2013, 19, 3740–3748. [Google Scholar] [CrossRef] [PubMed]
- Hellmann, J.J.; Byers, J.E.; Bierwagen, B.G.; Dukes, J.S. Five potential consequences of climate change for invasive species. Conserv. Biol. 2008, 22, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Walther, G.R.; Roques, A.; Hulme, P.E.; Sykes, M.T.; Pysek, P.; Kühn, I.; Zobel, M.; Bacher, S.; Botta-Dukát, Z.; Bugmann, H.; et al. Alien species in a warmer world: Risks and opportunities. Trends Ecol. Evol. 2009, 24, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Pyke, C.R.; Thomas, R.; Porter, R.D.; Hellmann, J.J.; Dukes, J.S.; Lodge, D.M.; Chavarria, G. Current practices and future opportunities for policy on climate change and invasive species. Conserv. Biol. 2008, 22, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Essl, F.; Lenzner, B.; Bacher, S.; Bailey, S.; Capinha, C.; Daehler, C.; Dullinger, S.; Genovesi, P.; Hui, C.; Hulme, P.E.; et al. Drivers of future alien species impacts: An expert-based assessment. Glob. Chang. Biol. 2020, 26, 4880–4893. [Google Scholar] [CrossRef]
- Australian National University. Implications of Climate Change for Australia’s World Heritage Properties: A Preliminary Assessment; A report to the Department of the Environment, Water, Heritage and the Arts by the Fenner School of Environment and Society; The Australian National University: Canberra, Australia, 2009. [Google Scholar]
- Gray, D.R. Climate change can reduce the risk of biological invasion by reducing propagule size. Biol. Invasions 2017, 19, 913–923. [Google Scholar] [CrossRef]
- Ricciardi, A.; Blackburn, T.M.; Carlton, J.T.; Dick, J.T.A.; Hulme, P.E.; Iacarella, J.C.; Jeschke, J.M.; Liebhold, A.M.; Lockwood, J.L.; MacIsaac, H.J.; et al. Invasion Science: A Horizon Scan of Emerging Challenges and Opportunities. Trends Ecol. Evol. 2017, 32, 464–474. [Google Scholar] [CrossRef]
- Duffy, G.A.; Coetzee, B.W.T.; Latombe, G.; Akerman, A.H.; McGeoch, M.A.; Chown, S.L. Barriers to globally invasive species are weakening across the Antarctic. Divers. Distrib. 2017, 23, 982–996. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Censky, E.J.; Hodge, K.; Dudley, J. Over-water dispersal of lizards due to hurricanes. Nature 1998, 395, 556. [Google Scholar] [CrossRef]
- Richardson, C.H.; Nemeth, D.J. Hurricane-borne African locusts (Schistocerca gregaria) on the Windward Islands. GeoJournal 1991, 23, 349–357. [Google Scholar] [CrossRef]
- Green, A.J.; Figuerola, J. Recent advances in the study of long-distance dispersal of aquatic invertebrates via birds. Divers. Distrib. 2005, 11, 149–156. [Google Scholar] [CrossRef]
- Deutsch, C.A.; Tewksbury, J.J.; Huey, R.B.; Sheldon, K.S.; Ghalambor, C.K.; Haak, D.C.; Martin, P.R. Impacts of climate warming on terrestrial ectotherms across latitude. Proc. Natl. Acad. Sci. USA 2008, 105, 6668–6672. [Google Scholar] [CrossRef] [PubMed]
- McClelland, G.T.W.; Altwegg, R.; van Aarde, R.J.; Ferreira, S.; Burger, A.E.; Chown, S.L. Climate change leads to increasing population density and impacts of a key island invader. Ecol. Appl. 2018, 28, 212–224. [Google Scholar] [CrossRef]
- Cook, A.J.; Poncet, S.; Cooper, A.P.R.; Herbert, D.J.; Christie, D. Glacier retreat on South Georgia and implications for the spread of rats. Antarct. Sci. 2010, 22, 255–263. [Google Scholar] [CrossRef]
- Steffen, W. Australia’s Biodiversity and Climate Change; CSIRO Publishing: Clayton, Australia, 2009. [Google Scholar]
- Bello-Rodriguez, V.; Mateo, R.G.; Pellissier, L.; Cubas, J.; Cooke, B.; Gonzalez-Mancebo, J.M. Forecast increase in invasive rabbit spread into ecosystems of an oceanic island (Tenerife) under climate change. Ecol. Appl. 2021, 31, e02206. [Google Scholar] [CrossRef] [PubMed]
- Singleton, G.R.; Brown, P.R.; Pech, R.P.; Jacob, J.; Mutze, G.J.; Krebs, C.J. One hundred years of eruptions of house mice in Australia—A natural biological curio. Biol. J. Linn. Soc. 2005, 84, 617–627. [Google Scholar] [CrossRef]
- Bronson, F.H. Climate change and seasonal reproduction in mammals. Philos. Trans. R. Soc. B-Biol. Sci. 2009, 364, 3331–3340. [Google Scholar] [CrossRef]
- Parsons, M.A.; Bridges, A.S.; Biteman, D.S.; Garcelon, D.K. Precipitation and prey abundance influence food habits of an invasive carnivore. Anim. Conserv. 2020, 23, 60–71. [Google Scholar] [CrossRef]
- Harris, D.B.; Macdonald, D.W. Interference competition between introduced black rats and endemic Galapagos rice rats. Ecology 2007, 88, 2330–2344. [Google Scholar] [CrossRef]
- Htwe, N.M.; Singleton, G.R.; Nelson, A.D. Can rodent outbreaks be driven by major climatic events? Evidence from cyclone Nargis in the Ayeyawady Delta, Myanmar. Pest Manag. Sci. 2013, 69, 378–385. [Google Scholar] [CrossRef]
- Singleton, G.R.; Belmain, S.; Brown, P.R.; Aplin, K.; Htwe, N.M. Impacts of rodent outbreaks on food security in Asia. Wildl. Res. 2010, 37, 355–359. [Google Scholar] [CrossRef]
- Oliver, T.H.; Heard, M.S.; Isaac, N.J.B.; Roy, D.B.; Procter, D.; Eigenbrod, F.; Freckleton, R.; Hector, A.; Orme, D.L.; Petchey, O.L.; et al. Biodiversity and Resilience of Ecosystem Functions. Trends Ecol. Evol. 2015, 30, 673–684. [Google Scholar] [CrossRef]
- MacDougall, A.S.; McCann, K.S.; Gellner, G.; Turkington, R. Diversity loss with persistent human disturbance increases vulnerability to ecosystem collapse. Nature 2013, 494, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Oliver, T.H.; Isaac, N.J.B.; August, T.A.; Woodcock, B.A.; Roy, D.B.; Bullock, J.M. Declining resilience of ecosystem functions under biodiversity loss. Nat. Commun. 2015, 6, 10122. [Google Scholar] [CrossRef]
- Ricciardi, A.; Iacarella, J.C.; Aldridge, D.C.; Blackburn, T.M.; Carlton, J.T.; Catford, J.A.; Dick, J.T.A.; Hulme, P.E.; Jeschke, J.M.; Liebhold, A.M.; et al. Four priority areas to advance invasion science in the face of rapid environmental change. Environ. Rev. 2021, 29, 119–141. [Google Scholar] [CrossRef]
- Frenot, Y.; Chown, S.L.; Whinam, J.; Selkirk, P.M.; Convey, P.; Skotnicki, M.; Bergstrom, D.M. Biological invasions in the Antarctic: Extent, impacts and implications. Biol. Rev. Camb. Philos. Soc. 2005, 80, 45–72. [Google Scholar] [CrossRef] [PubMed]
- Hulme, P.E. Climate change and biological invasions: Evidence, expectations, and response options. Biol. Rev. 2017, 92, 1297–1313. [Google Scholar] [CrossRef]
- Rahel, F.J.; Olden, J.D. Assessing the effects of climate change on aquatic invasive species. Conserv. Biol. 2008, 22, 521–533. [Google Scholar] [CrossRef]
- Lowe, S.; Browne, M.; Boudjelas, S.; De Poorter, M. 100 of the World’s Worst Invasive Alien Species: A Selection from the Global Invasive Species Database; Invasive Species Specialist Group Auckland: Auckland, New Zealand, 2000; Volume 12. [Google Scholar]
- Richardson, D.M.; Bond, W.J. Determinants of plant distribution: Evidence from pine invasions. Am. Nat. 1991, 137, 639–668. [Google Scholar] [CrossRef]
- Michener, W.K.; Blood, E.R.; Bildstein, K.L.; Brinson, M.M.; Gardner, L.R. Climate change, hurricanes and tropical storms, and rising sea level in coastal wetlands. Ecol. Appl. 1997, 7, 770–801. [Google Scholar] [CrossRef]
- Seebens, H.; Blackburn, T.M.; Hulme, P.E.; van Kleunen, M.; Liebhold, A.M.; Orlova-Bienkowskaja, M.; Pyšek, P.; Schindler, S.; Essl, F. Around the world in 500 years: Inter-regional spread of alien species over recent centuries. Glob. Ecol. Biogeogr. 2021, 30, 1621–1632. [Google Scholar] [CrossRef]
- Byers, J.E. Impact of non-indigenous species on natives enhanced by anthropogenic alteration of selection regimes. Oikos 2002, 97, 449–458. [Google Scholar] [CrossRef]
- Walther, G.R.; Post, E.; Convey, P.; Menzel, A.; Parmesan, C.; Beebee, T.J.C.; Fromentin, J.M.; Hoegh-Guldberg, O.; Bairlein, F. Ecological responses to recent climate change. Nature 2002, 416, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Seebens, H.; Blackburn, T.M.; Dyer, E.E.; Genovesi, P.; Hulme, P.E.; Jeschke, J.M.; Pagad, S.; Pyšek, P.; van Kleunen, M.; Winter, M. Global rise in emerging alien species results from increased accessibility of new source pools. Proc. Natl. Acad. Sci. USA 2018, 115, E2264–E2273. [Google Scholar] [CrossRef]
- Logan, J.A.; Powell, J.A. Ghost forest, global warming, and the mountain pine beetle (Coleoptera: Scolytidae). Am. Entomol. 2001, 47, 160–173. [Google Scholar] [CrossRef]
- Binggeli, P.; Hamilton, A.C. Biological invasion by Maesopsis eminii in the East Usambara forests, Tanzania. Opera Bot. 1993, 121, 229–235. [Google Scholar]
- Latham, A.D.M.; Latham, M.C.; Cieraad, E.; Tompkins, D.M.; Warburton, B. Climate change turns up the heat on vertebrate pest control. Biol. Invasions 2015, 17, 2821–2829. [Google Scholar] [CrossRef]
- Chapuis, J.L.; Frenot, Y.; Lebouvier, M. Recovery of native plant communities after eradication of rabbits from the subantarctic Kerguelen Islands, and influence of climate change. Biol. Conserv. 2004, 117, 167–179. [Google Scholar] [CrossRef]
- O’Bryan, C.J.; Patton, N.R.; Hone, J.; Lewis, J.S.; Berdejo-Espinola, V.; Risch, D.R.; Holden, M.H.; McDonald-Madden, E. Unrecognized threat to global soil carbon by a widespread invasive species. Glob. Chang. Biol. 2021. [Google Scholar] [CrossRef]
- Kurz, W.A.; Dymond, C.C.; Stinson, G.; Rampley, G.J.; Neilson, E.T.; Carroll, A.L.; Ebata, T.; Safranyik, L. Mountain pine beetle and forest carbon feedback to climate change. Nature 2008, 452, 987–990. [Google Scholar] [CrossRef]
- Wardle, D.A.; Bellingham, P.J.; Fukami, T.; Mulder, C.P.H. Promotion of ecosystem carbon sequestration by invasive predators. Biol. Lett. 2007, 3, 479–482. [Google Scholar] [CrossRef][Green Version]
- CCSP. Preliminary Review of Adaptation Options for Climate-Sensitive Ecosystems and Resources. U.S.; Climate Change Science Program and the Subcommittee on Global Change; U.S. Environmental Protection Agency: Washington, DC, USA, 2008; p. 873. [Google Scholar]
- Scheffer, M.; Barrett, S.; Carpenter, S.R.; Folke, C.; Green, A.J.; Holmgren, M.; Hughes, T.P.; Kosten, S.; van de Leemput, I.A.; Nepstad, D.C.; et al. Climate and conservation. Creating a safe operating space for iconic ecosystems. Science 2015, 347, 1317–1319. [Google Scholar] [CrossRef]
- Timpane-Padgham, B.L.; Beechie, T.; Klinger, T. A systematic review of ecological attributes that confer resilience to climate change in environmental restoration. PLoS ONE 2017, 12, e0173812. [Google Scholar] [CrossRef]
- Mulder, C.; Grant-Hoffman, M.; Towns, D.; Bellingham, P.; Wardle, D.; Durrett, M.; Fukami, T.; Bonner, K. Direct and indirect effects of rats: Does rat eradication restore ecosystem functioning of New Zealand seabird islands? Biol. Invasions 2009, 11, 1671–1688. [Google Scholar] [CrossRef]
- Peltzer, D.A.; Allen, R.B.; Lovett, G.M.; Whitehead, D.; Wardle, D.A. Effects of biological invasions on forest carbon sequestration. Glob. Chang. Biol. 2010, 16, 732–746. [Google Scholar] [CrossRef]
- Le Corre, M.; Danckwerts, D.K.; Ringler, D.; Bastien, M.; Orlowski, S.; Rubio, C.M.; Pinaud, D.; Micol, T. Seabird recovery and vegetation dynamics after Norway rat eradication at Tromelin Island, western Indian Ocean. Biol. Conserv. 2015, 185, 85–94. [Google Scholar] [CrossRef]
- Savage, C. Seabird nutrients are assimilated by corals and enhance coral growth rates. Sci. Rep. 2019, 9, 4284. [Google Scholar] [CrossRef] [PubMed]
- Rankin, L.L.; Jones, H.P. Nearshore ecosystems on seabird islands are potentially influenced by invasive predator eradications and environmental conditions: A case study at the Mercury Islands, New Zealand. Mar. Ecol. Prog. Ser. 2021, 661, 83–96. [Google Scholar] [CrossRef]
- Kurle, C.M.; Zilliacus, K.M.; Sparks, J.; Curl, J.; Bock, M.; Buckelew, S.; Williams, J.C.; Wolf, C.A.; Holmes, N.D.; Plissner, J. Indirect effects of invasive rat removal result in recovery of island rocky intertidal community structure. Sci. Rep. 2021, 11, 5395. [Google Scholar] [CrossRef] [PubMed]
- Munang, R.T.; Thiaw, I.; Rivington, M. Ecosystem management: Tomorrow’s approach to enhancing food security under a changing climate. Sustainability 2011, 3, 937–954. [Google Scholar] [CrossRef]
- McKergow, L.A.; Prosser, I.P.; Hughes, A.O.; Brodie, J. Sources of sediment to the Great Barrier Reef World Heritage Area. Mar. Pollut. Bull. 2005, 51, 200–211. [Google Scholar] [CrossRef]
- Sierra, C. El cerdo cimarrón (Sus scrofa, Suidae) en la Isla del Coco, Costa Rica: Escarbaduras, alteraciones al suelo y erosión. Rev. Biol. Trop. 2001, 49, 1158–1170. [Google Scholar]
- Beltran, R.S.; Kreidler, N.; Van Vuren, D.H.; Morrison, S.A.; Zavaleta, E.S.; Newton, K.; Tershy, B.R.; Croll, D.A. Passive recovery of vegetation after herbivore eradication on Santa Cruz Island, California. Restor. Ecol. 2014, 22, 790–797. [Google Scholar] [CrossRef]
- Luna-Mendoza, L.; Aguirre-Muñoz, A.; Hernández-Montoya, J.; Torres-Aguilar, M.; García-Carreón, J.; Puebla-Hernández, O.; Luvianos-Colín, S.; Cárdenas-Tapia, A.; Méndez-Sánchez, F. Ten years after feral goat eradication: The active restoration of plant communities on Guadalupe Island, Mexico. In Proceedings of the Island Invasives: Scaling up to Meet the Challenge, Dundee, Scotland, 10–14 July 2017; IUCN Species Specialist Group: Gland, Switzerland, 2019; pp. 571–575. [Google Scholar]
- Dunkell, D.O.; Bruland, G.L.; Evensen, C.I.; Litton, C.M. Runoff, Sediment Transport, and Effects of Feral Pig (Sus scrofa) Exclusion in a Forested Hawaiian Watershed. Pac. Sci. 2011, 65, 175–194. [Google Scholar] [CrossRef]
- Cleeland, J.B.; Pardo, D.; Raymond, B.; Terauds, A.; Alderman, R.; McMahon, C.R.; Phillips, R.A.; Lea, M.A.; Hindell, M.A. Introduced species and extreme weather as key drivers of reproductive output in three sympatric albatrosses. Sci. Rep. 2020, 10, e8199. [Google Scholar] [CrossRef] [PubMed]
- Bellingham, P.J.; Towns, D.R.; Cameron, E.K.; Davis, J.J.; Wardle, D.A.; Wilmshurst, J.M.; Mulder, C.P.H. New Zealand island restoration: Seabirds, predators, and the importance of history. New Zealand J. Ecol. 2010, 34, 115–136. [Google Scholar]
- Duhr, M.; Flint, E.; Hunter, S.; Taylor, R.; Flanders, B.; Howald, G.; Norwood, D. Control of house mice preying on adult albatrosses at Midway Atoll National Wildlife Refuge. In Island Invasives: Scaling up to Meet the Challenge; Occasional Paper SSC no. 62; Veitch, C.R., Clout, M.N., Martin, A.R., Russel, J.C., West, C.J., Eds.; IUCN: Gland, Switzerland, 2019; pp. 21–25. [Google Scholar]
- Reynolds, M.H.; Courtot, K.N.; Berkowitz, P.; Storlazzi, C.D.; Moore, J.; Flint, E. Will the Effects of Sea-Level Rise Create Ecological Traps for Pacific Island Seabirds? PLoS ONE 2015, 10, e0136773. [Google Scholar] [CrossRef]
- VanderWerf, E.A.; Wood, K.R.; Swenson, C.; LeGrande, M.; Eijzenga, H.; Walker, R.L. Avifauna of Lehua Islet, Hawai’i: Conservation value and management needs. Pac. Sci. 2007, 61, 39–52. [Google Scholar] [CrossRef]
- VanderWerf, E.; Young, L.; Kohley, C.; Dalton, M. Translocations of Laysan and black-footed albatrosses in Hawaii, USA, to create new protected breeding colonies safe from climate change. In Case Study in: Global Reintroduction Perspectives: 2018; Case Studies From around the Globe; Soorae, P., Ed.; IUCN/SSC Re-Introduction Specialist Group & Environment Agency: Abu Dhabi, United Arab Emirates, 2018; pp. 100–105. [Google Scholar]
- VanderWerf, E.A.; Young, L.C.; Kohley, C.R.; Dalton, M.E.; Fisher, R.; Fowlke, L.; Donohue, S.; Dittmar, E. Establishing Laysan and black-footed albatross breeding colonies using translocation and social attraction. Glob. Ecol. Conserv. 2019, 19, e00667. [Google Scholar] [CrossRef]
- Croll, D.A.; Newton, K.M.; McKown, M.; Bock, M.F.; Tershy, B.R.; Holmes, N.; Wolf, C.A.; Howald, G.; Williams, J.C.; Young, H.S.; et al. Passive recovery of an island bird community after rodent eradication. Biol. Invasions 2016, 18, 703–715. [Google Scholar] [CrossRef]
- Benkwitt, C.E.; Wilson, S.K.; Graham, N.A.J. Seabird nutrient subsidies alter patterns of algal abundance and fish biomass on coral reefs following a bleaching event. Glob. Chang. Biol. 2019, 25, 2619–2632. [Google Scholar] [CrossRef]
- Benkwitt, C.E.; Gunn, R.L.; Le Corre, M.; Carr, P.; Graham, N.A. Rat eradication restores nutrient subsidies from seabirds across terrestrial and marine ecosystems. Curr. Biol. 2021, 31, 2704–2711. [Google Scholar] [CrossRef]
- Ficetola, G.F.; Poulenard, J.; Sabatier, P.; Messager, E.; Gielly, L.; Leloup, A.; Etienne, D.; Bakke, J.; Malet, E.; Fanget, B.; et al. DNA from lake sediments reveals long-term ecosystem changes after a biological invasion. Sci. Adv. 2018, 4, eaar4292. [Google Scholar] [CrossRef]
- Robin, M.; Chapuis, J.L.; Lebouvier, M. Remote sensing of vegetation cover change in islands of the Kerguelen archipelago. Polar Biol. 2011, 34, 1689–1700. [Google Scholar] [CrossRef]
- Holdaway, R.J.; Burrows, L.E.; Carswell, F.E.; Marburg, A.E. Potential for invasive mammalian herbivore control to result in measurable carbon gains. N. Z. J. Ecol. 2012, 36, 252–264. Available online: https://www.jstor.org/stable/24060854 (accessed on 1 October 2021).
- Bengsen, A.; Cox, T. The Role of Rabbit and Other Invasive Herbivore Control in Reducing Australia’s Greenhouse Gas Emissions; PestSmart Toolkit publication, Invasive Animals Cooperative Research Centre: Canberra, Australia, 2014; p. 37. [Google Scholar]
- Hata, K.; Osawa, T.; Hiradate, S.; Kachi, N. Soil erosion alters soil chemical properties and limits grassland plant establishment on an oceanic island even after goat eradication. Restor. Ecol. 2019, 27, 333–342. [Google Scholar] [CrossRef]
- Jones, H.P. Prognosis for ecosystem recovery following rodent eradication and seabird restoration in an island archipelago. Ecol. Appl. 2010, 20, 1204–1216. [Google Scholar] [CrossRef]
- Kappes, P.J.; Jones, H.P. Integrating seabird restoration and mammal eradication programs on islands to maximize conservation gains. Biodivers Conserv. 2014, 23, 503–509. [Google Scholar] [CrossRef]
- Wooldridge, S.A.; Done, T.J. Improved water quality can ameliorate effects of climate change on corals. Ecol. Appl. 2009, 19, 1492–1499. [Google Scholar] [CrossRef] [PubMed]
- MacNeil, M.A.; Mellin, C.; Matthews, S.; Wolff, N.H.; McClanahan, T.R.; Devlin, M.; Drovandi, C.; Mengersen, K.; Graham, N.A.J. Water quality mediates resilience on the Great Barrier Reef. Nat. Ecol. Evol. 2019, 3, 620–627. [Google Scholar] [CrossRef]
- Sekercioglu, C.H. Increasing awareness of avian ecological function. Trends Ecol. Evol. 2006, 21, 464–471. [Google Scholar] [CrossRef] [PubMed]
- De La Peña-Lastra, S. Seabird droppings: Effects on a global and local level. Sci. Total Environ. 2021, 754, 142148. [Google Scholar] [CrossRef]
- McCauley, D.J.; DeSalles, P.A.; Young, H.S.; Dunbar, R.B.; Dirzo, R.; Mills, M.M.; Micheli, F. From wing to wing: The persistence of long ecological interaction chains in less-disturbed ecosystems. Sci. Rep. 2012, 2, 409. [Google Scholar] [CrossRef] [PubMed]
- Lorrain, A.; Houlbreque, F.; Benzoni, F.; Barjon, L.; Tremblay-Boyer, L.; Menkes, C.; Gillikin, D.P.; Payri, C.; Jourdan, H.; Boussarie, G.; et al. Seabirds supply nitrogen to reef-building corals on remote Pacific islets. Sci. Rep. 2017, 7, e3721. [Google Scholar] [CrossRef]
- Wiedenmann, J.; D’Angelo, C.; Smith, E.G.; Hunt, A.N.; Legiret, F.E.; Postle, A.D.; Achterberg, E.P. Nutrient enrichment can increase the susceptibility of reef corals to bleaching. Nat. Clim. Chang. 2013, 3, 160–164. [Google Scholar] [CrossRef]
- Ezzat, L.; Maguer, J.F.; Grover, R.; Ferrier-Pages, C. Limited phosphorus availability is the Achilles heel of tropical reef corals in a warming ocean. Sci. Rep. 2016, 6, 31768. [Google Scholar] [CrossRef] [PubMed]
- Cuttler, M.V.W.; Hansen, J.E.; Lowe, R.J.; Drost, E.J.F. Response of a fringing reef coastline to the direct impact of a tropical cyclone. Limnol. Oceanogr. Lett. 2018, 3, 31–38. [Google Scholar] [CrossRef]
- Ondiviela, B.; Losada, I.J.; Lara, J.L.; Maza, M.; Galvan, C.; Bouma, T.J.; van Belzen, J. The role of seagrasses in coastal protection in a changing climate. Coast. Eng. 2014, 87, 158–168. [Google Scholar] [CrossRef]
- Priddel, D.; Carlile, N.; Wheeler, R. Establishment of a new breeding colony of Gould’s petrel (Pterodroma leucoptera leucoptera) through the creation of artificial nesting habitat and the translocation of nestlings. Biol. Conserv. 2006, 128, 553–563. [Google Scholar] [CrossRef]
- Bellard, C.; Leclerc, C.; Courchamp, F. Potential impact of sea level rise on French islands worldwide. Nat. Conserv. 2013, 5, 75–86. [Google Scholar] [CrossRef]
- Church, J.A.; White, N.J.; Hunter, J.R. Sea-level rise at tropical Pacific and Indian Ocean islands. Glob. Planet. Chang. 2006, 53, 155–168. [Google Scholar] [CrossRef]
- Wetzel, F.T.; Beissmann, H.; Penn, D.J.; Jetz, A. Vulnerability of terrestrial island vertebrates to projected sea-level rise. Glob. Chang. Biol. 2013, 19, 2058–2070. [Google Scholar] [CrossRef]
- Spatz, D.R.; Holmes, N.D.; Reguero, B.G.; Butchart, S.H.M.; Tershy, B.R.; Croll, D.A. Managing Invasive Mammals to Conserve Globally Threatened Seabirds in a Changing Climate. Conserv. Lett. 2017, 10, 736–747. [Google Scholar] [CrossRef]
- Wetzel, F.T.; Kissling, W.D.; Beissmann, H.; Penn, D.J. Future climate change driven sea-level rise: Secondary consequences from human displacement for island biodiversity. Glob. Chang. Biol. 2012, 18, 2707–2719. [Google Scholar] [CrossRef] [PubMed]
- Raine, A.F.; Driskill, S.; Vynne, M.; Harvey, D.; Pias, K. Managing the effects of introduced predators on Hawaiian endangered seabirds. J. Wildl. Manag. 2020, 84, 425–435. [Google Scholar] [CrossRef]
- Reynolds, M.H.; Berkowitz, P.; Courtot, K.N.; Krause, C.M. Predicting Sea-Level Rise Vulnerability of Terrestrial Habitat and Wildlife of the Northwestern Hawaiian Islands; US Geological Survey Report No. 2012–1182; U.S. Geological Survey: Reston, VA, USA, 2012; p. 139. [Google Scholar]
- Lindsey, G.; Atkinson, C.; Banko, P.; Brenner, G.J.; Campbell, E.; David, R.; Foote, D.; Forbes, M.; Pratt, T.; Reynolds, M.H.; et al. Technical Options and Recommendations for Faunal Restoration of Kaho’olawe; Unpublished Technical Report to Kaho ‘Olawe Island Reserve Commission; USGS-Pacific Island Ecosystems Research Center, Kilauea Field Station: Hawai, Honolulu, 1997; p. 68. [Google Scholar]
- Pacific Rim Conservation. No Net Loss. Available online: https://www.islandarks.org/techniques.html (accessed on 29 September 2021).
- Minnich, R.A. Vegetation of Santa Cruz and Santa Catalina Islands. In The California Islands, Proceedings of the Multidisciplinary Symposium, Santa Barbara, CA, USA, 27 February–1 March 1978; Santa Barbara Museum of Natural History: Santa Barbara, CA, USA, 1980; pp. 123–138. [Google Scholar]
- Van Vuren, D.; Coblentz, B.E. Some ecological effects of feral sheep on Santa Cruz Island, California, USA. Biol. Conserv. 1987, 41, 253–268. [Google Scholar] [CrossRef]
- Nellemann, C.; Corcoran, E. Blue Carbon: The Role of Healthy Oceans in Binding Carbon: A Rapid Response Assessment; UNEP/Earthprint: Nairobi, Kenya, 2009. [Google Scholar]
- Laffoley, D. The Management of Natural Coastal Carbon Sinks; Grimsditch, G.D., Ed.; IUCN: Gland, Switzerland, 2009. [Google Scholar]
- Croxall, J.P.; Butchart, S.H.M.; Lascelles, B.; Stattersfield, A.J.; Sullivan, B.; Symes, A.; Taylor, P. Seabird conservation status, threats and priority actions: A global assessment. Bird Conserv. Int. 2012, 22, 1–34. [Google Scholar] [CrossRef]
- Russell, J.C.; Cole, N.C.; Zuel, N.; Rocamora, G. Introduced mammals on Western Indian Ocean islands. Glob. Ecol. Conserv. 2016, 6, 132–144. [Google Scholar] [CrossRef]
- McEachern, A.K.; Thomson, D.M.; Chess, K.A. Climate alters response of an endemic island plant to removal of invasive herbivores. Ecol. Appl. 2009, 19, 1574–1584. [Google Scholar] [CrossRef]
- Pettorelli, N.; Graham, N.A.; Seddon, N.; Bustamante, M.M.d.C.; Lowton, M.J.; Sutherland, W.; Koldewey, H.J.; Prentice, H.C.; Barlow, J. Time to integrate global climate change and biodiversity science-policy agendas. J. Appl. Ecol. 2021, 58, 2384–2393. [Google Scholar] [CrossRef]
| Term | Definition | Reference |
|---|---|---|
| Biodiversity loss | Loss of the variability among living organisms, including genetic (individual, subpopulation, and total population), species (uniqueness, abundances, and richness), functional (interactions and traits), and habitat diversity (different types and abiotic heterogeneity within them) from all sources, including terrestrial, marine, and aquatic ecosystems and ecological complexes of which they are part; for the purposes of our paper, this refers to loss of native biodiversity (see below) and not loss due to greater species richness resulting from the presence of IAS | [14,15] |
| Biosecurity | The actions needed to prevent, detect, and rapidly respond to the arrival of unwanted species, in a country (or island/archipelago) or between places within a country (or island/archipelago; with the common goal of protecting a country’s (island’s/archipelago’s) economy, environment, and people’s health from biological threats, such and plant and animal pests and diseases | [16] |
| Climate adaptation | Preparing for, coping with, or adjusting to climatic changes and their associated impacts | [17] |
| Climate change | Major shifts to the state of the climate (e.g., temperature, precipitation, and/or wind pattern) that occur over several decades or longer. May be due to natural processes or external anthropogenic changes to the composition of the atmosphere; in context of this paper change in climate is attributed directly or indirectly to human activity | [18] |
| Climate mitigation | Efforts to reduce or prevent emission of greenhouse gases and other anthropogenic climate forcing | |
| Climate resilience | The adaptive capacity for a socio-ecological system to: (1) absorb stresses and maintain function in the face of external stresses imposed upon it by climate change and (2) adapt, reorganize, and evolve into more desirable configurations that improve the sustainability of the system, leaving it better prepared for future climate change impacts | [19,20] |
| Conservation translocation | The deliberate movement of organisms from one site for release in another, with the intention to yield a measurable conservation benefit at the levels of a population, species, or ecosystem | [21] |
| Endemic species | Species that naturally occurs only in a single geographic area; in the context of this paper these species are located on single island/archipelago | [22] |
| Eradication | The complete and permanent removal of IAS | [23] |
| Invasive Alien Species (IAS) | Species that are either accidentally or intentionally introduced outside of their native range and have significant negative impacts on the native biodiversity, ecosystem services, and/or human well-being where they become established | [24] |
| Native biodiversity | Species that occur naturally in a given geographic area, as opposed to having been transported, inadvertently or purposefully, by humans | [25,26,27] |
| Propagule pressure | A composite measure of the introduction effort consisting of: the propagule size (i.e., the number of individuals introduced per introduction and the number/frequency of introduction events | [28] |
| Refugia | Areas that may facilitate the persistence of species during large-scale, long-term disruptive climatic change | [29] |
| Funding Entity | GEF | DI | GCF | WCS | Total |
|---|---|---|---|---|---|
| Projects listed as funded on website | 5288 | 1164 | 173 | 104 | 6729 |
| Projects remaining after initial filter | 2019 * | 633 ** | 173 | 104 | 2929 |
| Projects receiving secondary screening | 53 | 11 | 10 | 5 | 79 |
| Projects meeting our search criteria | 8 ** | 1 | 1 | 0 | 10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kappes, P.J.; Benkwitt, C.E.; Spatz, D.R.; Wolf, C.A.; Will, D.J.; Holmes, N.D. Do Invasive Mammal Eradications from Islands Support Climate Change Adaptation and Mitigation? Climate 2021, 9, 172. https://doi.org/10.3390/cli9120172
Kappes PJ, Benkwitt CE, Spatz DR, Wolf CA, Will DJ, Holmes ND. Do Invasive Mammal Eradications from Islands Support Climate Change Adaptation and Mitigation? Climate. 2021; 9(12):172. https://doi.org/10.3390/cli9120172
Chicago/Turabian StyleKappes, Peter J., Cassandra E. Benkwitt, Dena R. Spatz, Coral A. Wolf, David J. Will, and Nick D. Holmes. 2021. "Do Invasive Mammal Eradications from Islands Support Climate Change Adaptation and Mitigation?" Climate 9, no. 12: 172. https://doi.org/10.3390/cli9120172
APA StyleKappes, P. J., Benkwitt, C. E., Spatz, D. R., Wolf, C. A., Will, D. J., & Holmes, N. D. (2021). Do Invasive Mammal Eradications from Islands Support Climate Change Adaptation and Mitigation? Climate, 9(12), 172. https://doi.org/10.3390/cli9120172






