Endophytic Trichoderma Species Isolated from Persea americana and Cinnamomum verum Roots Reduce Symptoms Caused by Phytophthora cinnamomi in Avocado
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling of Symptomatic Avocado and Cinnamon Plants
2.2. Fungal Isolates
2.3. DNA Extraction, PCR Amplification and Sequencing
2.4. Phylogenetic Analysis
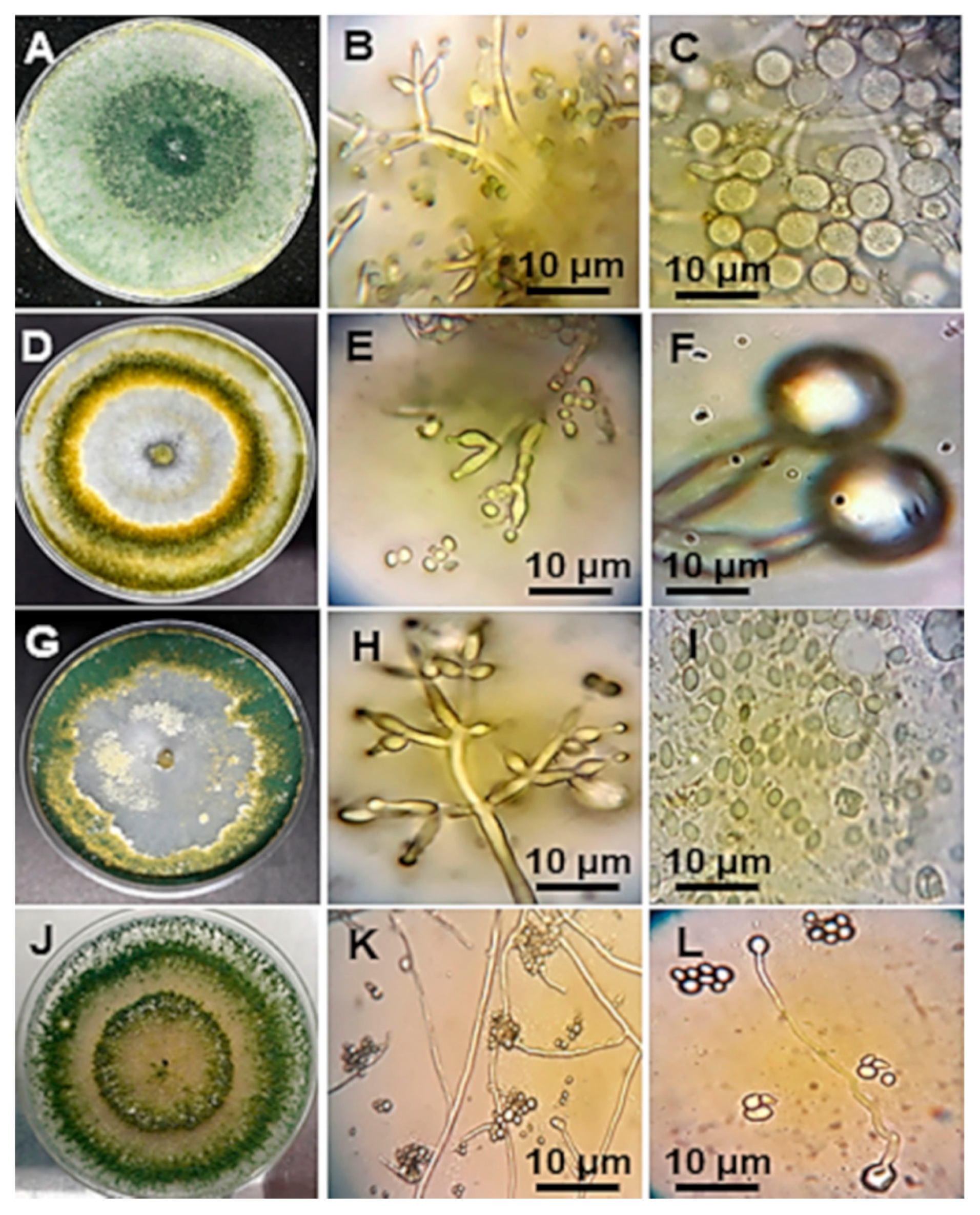
2.5. Morphological Characterization of Trichoderma spp.
2.6. Antagonistic In Vitro Assessment of Trichoderma spp.
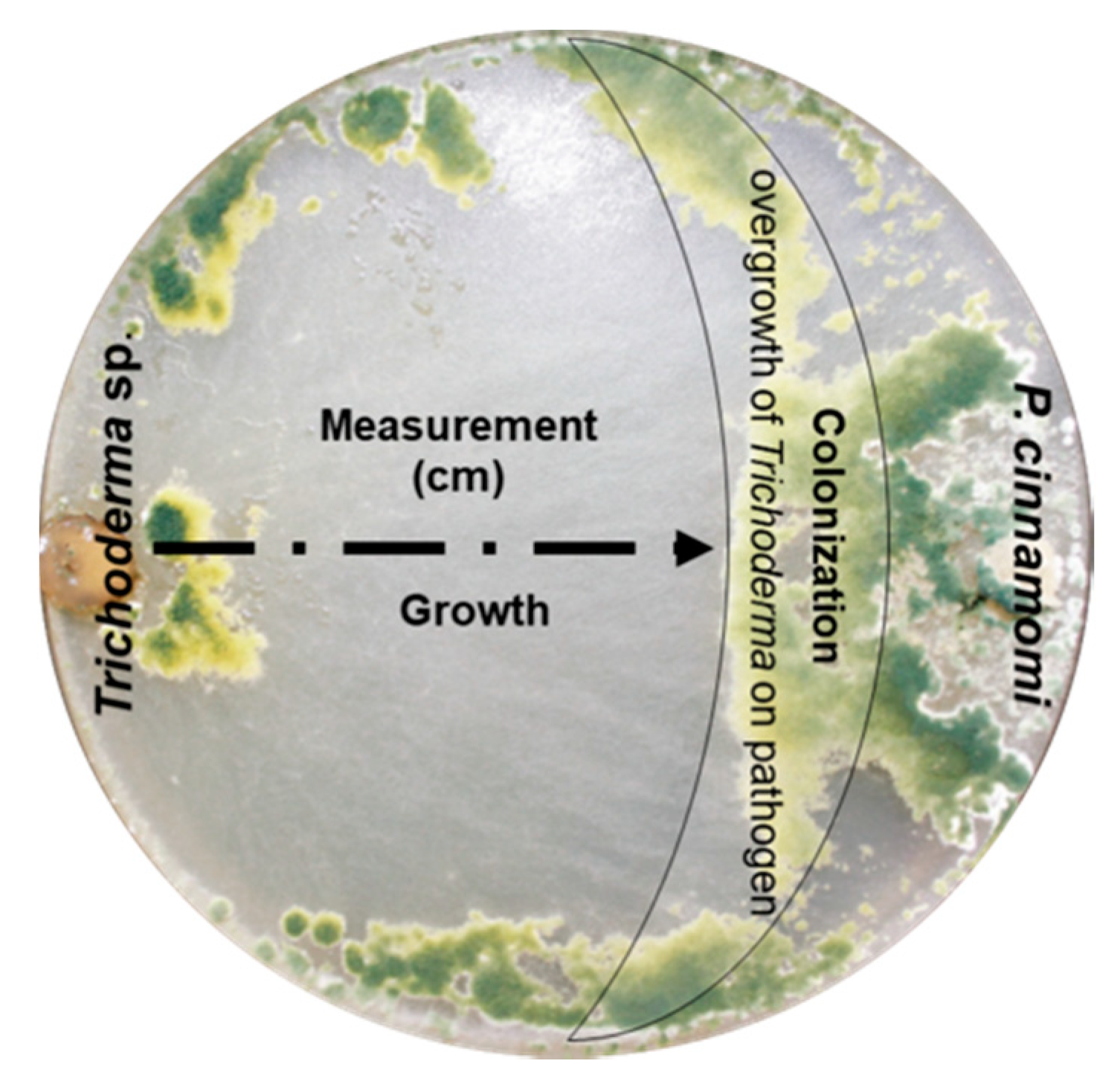
2.7. Mycoparasitism and Colonization by Trichoderma spp. over P. cinnamomi
2.8. Fungal Growth and Plant Inoculation
2.9. Avocado Seedling Inoculation
2.10. Statistical Analysis
3. Results
3.1. Isolates Recovered
3.2. Phylogenetic Analysis
3.3. Morphological Characterization of Trichoderma spp.
3.4. Percentage of Growth Inhibition In Vitro
3.5. Mycoparasitism, Percentage of Colonization, and Growth Rate
3.6. Biocontrol of P. cinnamomi by Trichoderma Strains in the Greenhouse Assay
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Newett, S.D.; Crandy, J.H.; Balerdi, C.F. Cultivars and rootstocks. In The Avocado: Botany, Production and Uses; Schaffer, B., Wolstenholme, B.N., Whiley, A.W., Eds.; CABI: London, UK, 2013; p. 560. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Data-Production. Crops-FAOSTAT. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 14 October 2019).
- Rodrigues, D.S.; Cantuarias-Avilés, T.; Bremer, N.H.; Mourão, F.A.; Bordignon, M.R. Management of root rot in avocado trees. Rev. Bras. Frutic. 2016, 38, 1–5. [Google Scholar]
- Dunstan, W.A.; Howard, K.; Hardy, G.E.; Burgess, T.I. An overview of Australia’s Phytophthora species assemblage in natural ecosystems recovered from a survey in Victoria. IMA Fungus 2016, 7, 47–58. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malarczyk, D.; Panek, J.; Frac, M. Alternative molecular based diagnostic methods of plant pathogenic fungi affecting berry crops, a review. Molecules 2019, 24, 1200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrade-Hoyos, P.; De León, C.; Espíndola-Barquera, M.C.; Alvarado-Rosales, D.; López-Jiménez, A.; García-Espinosa, R. Selection of avocado rootstocks for tolerance-resistance to Phytophthora cinnamomi Rands. Span. J. Rural Dev. 2012, 3, 23–30. [Google Scholar]
- Sánchez-González, E.I.; Gutiérrez-Soto, J.G.; Olivares-Sáenz, E.; Gutiérrez-Díez, A.; Barrientos-Priego, A.F.; Ochoa-Ascencio, S. Screening progenies of Mexican race avocado genotypes for resistance to Phytophthora cinnamomi Rands. HortScience 2019, 54, 809–813. [Google Scholar] [CrossRef] [Green Version]
- Hermosa, R.; Viterbo, A.; Chet, I.; Monte, E. Plant-beneficial effects of Trichoderma and of its genes. Microbiology 2012, 158, 17–25. [Google Scholar] [CrossRef] [Green Version]
- Reeksting, B.J.; Olivier, N.A.; Van-Den, B.N. Transcriptome responses of an ungrafted Phytophthora root rot tolerant avocado (Persea americana) rootstock to flooding and Phytophthora cinnamomi. BMC Plant Biol. 2016, 16, 205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Contreras-Cornejo, H.A.; Macías-Rodríguez, L.; del-Val, E.; Larsen, J. Interactions of Trichoderma with plants, insects, and plant pathogen microorganisms: Chemical and molecular bases. In Co-Evolution of Secondary Metabolites; Merillon, J.M., Ramawat, K., Eds.; Springer: Cham, Switzerland, 2019; pp. 1–28. [Google Scholar]
- Naher, L.; Yusuf, U.K.; Ismail, A.; Hossain, K. Trichoderma spp. a biocontrol agent for sustainable management of plant diseases. Pak. J. Bot. 2014, 46, 1489–1493. [Google Scholar]
- Mayo, S.; Gutiérrez, S.; Malmierca, M.G.; Lorenzana, A.; Campelo, M.P.; Hermosa, R.; Casquero, P.A. Influence of Rhizoctonia solani and Trichoderma spp. in growth of bean (Phaseolus vulgaris L.) and in the induction of plant defense-related genes. Front. Plant Sci. 2015, 6, 685. [Google Scholar] [CrossRef] [Green Version]
- Benocci, T.; Aguilar-Pontes, M.V.; Kun, R.S.; Lubbers, R.J.; Lail, K.; Wang, M.; Lipzen, A.; Ng, V.; Grigoriev, I.V.; Seiboth, B.; et al. Deletion of either the regulatory gene ara1 or metabolic gene xki1 in Trichoderma reesei leads to increased CAZyme gene expression on crude plant biomass. Biotechnol. Biofuels 2019, 12, 81. [Google Scholar] [CrossRef]
- Kashyap, P.L.; Rai, P.; Srivastava, A.K.; Kumar, S. Trichoderma for climate resilient agriculture. World J. Microbiol. Biotechnol. 2017, 33, 155. [Google Scholar] [PubMed]
- Nawaz, K.; Shahid, A.A.; Bengyell, L.; Subhani, M.N.; Alia, M.; Anwar, W.; Iftikhar, S.; Ali, S.W. Diversity of Trichoderma species in chili rhizosphere that promote vigor and antagonism against virulent Phytophthora capsici. Sci. Hortic. 2018, 239, 242–252. [Google Scholar]
- Singh, B.N.; Singh, A.; Singh, S.P.; Singh, H.B. Trichoderma harzianum mediated reprogramming of oxidative stress response in root apoplast of sunflower enhances defence against Rhizoctonia solani. Eur. J. Plant Pathol. 2011, 131, 121–134. [Google Scholar]
- Li, Y.; Sun, R.; Yu, J.; Saravanakumar, K.; Chen, J. Antagonistic and biocontrol potential of Trichoderma asperellum ZJSX5003 against the maize stalk rot pathogen Fusarium graminearum. Indian J. Microbiol. 2016, 56, 318–327. [Google Scholar]
- Macías-Rodríguez, L.; Guzmán-Gómez, A.; García-Juárez, P.; Contreras-Cornejo, H.A. Trichoderma atroviride promotes tomato development and alters the root exudation of carbohydrates, which stimulates fungal growth and the biocontrol of the phytopathogen Phytophthora cinnamomi in a tripartite interaction system. Microb. Ecol. 2018, 94, 1–11. [Google Scholar]
- Ros, M.; Raut, I.; Santisima-Trinidad, A.B.; Pascual, J.A. Relationship of microbial communities and suppressiveness of Trichoderma fortified composts for pepper seedlings infected by Phytophthora nicotianae. PLoS ONE 2017, 12, e0174069. [Google Scholar]
- Sabbagh, S.K.; Roudini, M.; Panjehkeh, N. Systemic resistance induced by Trichoderma harzianum and Glomus mossea on cucumber damping-off disease caused by Phytophthora melonis. Arch. Phytopathol. Plant Prot. 2017, 50, 375–388. [Google Scholar]
- Vinale, F.; Girona, I.A.; Nigro, M.; Mazzei, P.; Piccolo, A.; Ruocco, M.; Woo, S.; Rosa, D.R.; Herrera, C.L.; Lorito, M. Cerinolactone, a hydroxy-lactone derivative from Trichoderma cerinum. J. Nat. Prod. 2012, 75, 103–106. [Google Scholar]
- Erwin, D.C.; Ribeiro, O.K. Phytophthora Diseases Worldwide; American Phytopathology Society Press: St Paul, MN, USA, 1996; p. 562. [Google Scholar]
- Doyle, J.J.; Doyle, J.L. Isolation of plant DNA from fresh tissue. Focus 1990, 12, 13–15. [Google Scholar]
- Rivera-Jiménez, M.N.; Zavaleta-Mancera, H.A.; Rebollar-Alviter, A.; Aguilar-Rincón, V.H.; García de los Santos, G.; Vaquera-Huerta, H.; Silva-Rojas, H.V. Phylogenetics and histology provide insight into damping-off infections of “Poblano” pepper seedlings caused by Fusarium wilt in greenhouses. Mycol. Prog. 2018, 17, 1237–1249. [Google Scholar]
- White, T.J.; Bruns, T.D.; Lee, S.B.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Juárez-Vázquez, S.B.; Silva-Rojas, H.V.; Rebollar-Alviter, A.; Maidana-Ojeda, M.; Osnaya-González, M.; Fuentes-Aragón, D. Phylogenetic and morphological identification of Colletotrichum godetiae, a novel pathogen causing anthracnose on loquat fruit (Eriobotrya japonica). J. Plant Dis. Prot. 2019, 126, 593–598. [Google Scholar] [CrossRef]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Altschul, S.F.; Madden, T.L.; Schaffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSIBLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 35, 1547–1549. [Google Scholar] [CrossRef]
- Huelsenbeck, J.P.; Ronquist, F. MrBayes: Bayesian inference of phylogenetic trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef] [Green Version]
- Ronquist, F.; Teslenko, M.; Van Der, M.P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbecck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [Green Version]
- Barnett, H.L.; Hunter, B.B. Illustrated Genera of Imperfect Fungi; American Phytopathology Society Press: St Paul, MN, USA, 1998; p. 240. [Google Scholar]
- Harris, I.J. Modified method for fungal slide culture. J. Clin. Microbiol. 1986, 24, 460–461. [Google Scholar] [CrossRef] [Green Version]
- Chaverri, P.; Branco-Rocha, F.; Jaklitsch, W.; Gazis, R.; Degenkolb, T.; Samuels, G.J. Systematics of the Trichoderma harzianum species complex and the re-identification of commercial biocontrol strains. Mycologia 2015, 107, 558–590. [Google Scholar] [CrossRef] [Green Version]
- Du-Plessis, I.L.; Druzhinina, I.S.; Atanasova, L.; Yarden, O.; Jacobs, K. The diversity of Trichoderma species from soil in South Africa, with five new additions. Mycologia 2018, 110, 559–583. [Google Scholar] [CrossRef]
- Gams, W.; Bissett, J. Morphology and identification of Trichoderma. In Trichoderma and Gliocladium: Basic Biology, Taxonomy and Genetics; Kubicek, C.P., Harman, G.E., Eds.; Taylor and Francis: Oxfordshire, UK, 2002; pp. 1–34. [Google Scholar]
- Jang, S.; Kwon, S.L.; Lee, H.; Jang, Y.; Park, M.S.; Lim, Y.W.; Kim, C.; Kim, J.J. New report of three unrecorded species in Trichoderma harzianum species complex in Korea. Mycobiology 2018, 46, 177–184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bell, D.K.; Wells, H.D.; Markam, C.R. In vitro antagonism of Trichoderma spp. against six fungal pathogens. Phytopathology 1982, 72, 379–382. [Google Scholar] [CrossRef]
- Camporota, P. Antagonisme in vitro de Trichoderma spp. vis-à-vis de Rhizoctonia solani Kühn. Agronomie 1985, 5, 613–620. [Google Scholar] [CrossRef] [Green Version]
- Ezziyyani, M.; Requena, M.E.; Egea-Gilabert, C.; Candela, M.E. Biological control of Phytophthora root rot of pepper using Trichoderma harzianum and Streptomyces rochei in combination. J. Phytopathol. 2007, 155, 342–349. [Google Scholar] [CrossRef]
- Toghueo, R.M.; Eke, P.; González, I.Z.; De Aldana, B.R.; Nana, L.W.; Boyom, F.F. Biocontrol and growth enhancement potential of two endophytic Trichoderma spp. from Terminalia catappa against the causative agent of common bean root rot (Fusarium solani). Biol. Control 2016, 96, 8–20. [Google Scholar] [CrossRef]
- Statistical Analysis System (SAS) Institute. SAS/STAT User Guide, Release 9.1; SAS. Institute Inc.: Cary, NC, USA, 2002; p. 292. [Google Scholar]
- Adnan, M.; Islam, W.; Shabbir, A.; Khan, K.A.; Ghramh, H.A.; Huang, Z.; Chen, H.Y.; Lu, G.D. Plant defense against fungal pathogens by antagonistic fungi with Trichoderma in focus. Microb. Pathog. 2019, 129, 7–18. [Google Scholar] [CrossRef]
- Alfiky, A. Effects of ultraviolet irradiation on the in vitro antagonistic potential of Trichoderma spp. against soil-borne fungal pathogens. Heliyon 2019, 5, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Harman, G.E. Overview of mechanisms and uses of Trichoderma spp. Phytopathology 2006, 96, 190–194. [Google Scholar] [CrossRef] [Green Version]
- Guzmán-Guzmán, P.; Porras-Troncoso, M.D.; Olmedo-Monfil, V.; Herrera-Estrella, A. Trichoderma species: Versatile plant symbionts. Phytopathology 2019, 109, 6–16. [Google Scholar] [CrossRef] [Green Version]
- Mahmoud, H.K.; Amgad, A.S.; Anas, E.; Younes, Y.M. Characterization of novel Trichoderma asperellum isolates to select effective biocontrol agents against tomato Fusarium wilt. Plant Pathol. 2015, 3, 50–60. [Google Scholar]
- Błaszczyk, L.; Strakowska, J.; Chełkowski, J.; Gąbka-Buszek, A.; Kaczmarek, J. Trichoderma species occurring on wood with decay symptoms in mountain forests in Central Europe: Genetic and enzymatic characterization. J. Appl. Genet. 2016, 57, 397–407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, Y.; Wang, J.L.; Chen, J.; Mao, L.J.; Feng, X.; Zhang, C.L.; Lin, F.C. Trichoderma biodiversity of agricultural fields in east China reveals a gradient distribution of species. PLoS ONE 2016, 11, e0160613. [Google Scholar] [CrossRef] [PubMed]
- Fukasawa, Y.; Osono, T.; Takeda, H. Wood decomposing abilities of diverse lignicolous fungi on nondecayed and decayed beech wood. Mycologia 2011, 103, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Margullis, L. Symbiogenesis and symbionticism. In Symbiosis as a Source of Evolutionary Innovation. Speciation and Morphogenesis; Margulis, L., Fester, R., Eds.; The MIT Press: Cambridge, MA, USA; London, UK, 1991; pp. 1–21. [Google Scholar]
- Harman, G.E.; Uphoff, N. Symbiotic root-endophytic soil microbes improve crop productivity and provide environmental benefits. Scientifica 2019, 2019, 9106395. [Google Scholar] [CrossRef] [Green Version]
- Hewedy, O.A.; Abdel-Lateif, K.; Bakr, R.A. Genetic diversity and biocontrol efficacy of indigenous Trichoderma isolates against Fusarium wilt of pepper. J. Basic Microbiol. 2019, 60, 1–10. [Google Scholar] [CrossRef]
- Leandro, L.S.; Guzman, T.; Ferguson, L.M.; Fernandez, G.E.; Louws, F.J. Population dynamics of Trichoderma in fumigated and compost-amended soil and on strawberry roots. Appl. Soil Ecol. 2007, 35, 237–246. [Google Scholar] [CrossRef]
- Chet, I.; Harman, G.E.; Baker, R. Trichoderma hamatum: Its hyphal interactions with Rhizoctonia solani and Pythium spp. Microb. Ecol. 1981, 7, 29–38. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, F.; Zhang, Y.; Zhang, J. Involvement of Trichoderma asperellum strain T6 in regulating iron acquisition in plant. J. Basic Microbiol. 2014, 54, S115–S124. [Google Scholar] [CrossRef]
- Chen, S.; Li, H.; Chen, Y.; Li, S.; Xu, J.; Guo, H.; Liu, Z.; Zhu, S.; Liu, H.; Zhang, W. Three new diterpenes and two new sesquiterpenoids from the endophytic fungus Trichoderma koningiopsis A729. Bioorg. Chem. 2019, 86, 368–374. [Google Scholar] [CrossRef]




| Strain | Trichoderma Species a | Host | GenBank Accession Number | Location |
|---|---|---|---|---|
| T-AS1 | T. asperellum | Persea americana | MK778890 | Hidalgo |
| T-AS2 | T. asperellum | Persea americana | MK779008 | Hidalgo |
| T-H3 | T. harzianum | Persea americana | MK780094 | Tepeyanco, Tlaxcala |
| T-H4 | T. harzianum | Persea americana | MK779064 | Tetela de Ocampo, Puebla |
| T-H5 | T. harzianum | Persea americana | MK784067 | La Aurora, Tepeyanco, Tlaxcala |
| T-AS6 | T. asperellum | Persea americana | MK791646 | Uruapan, Michoacan |
| T-AS7 | T. asperellum | Persea americana | MK791647 | Uruapan, Michoacan |
| T-K11 | T. koningiopsis | Persea americana | MK791648 | Jalisco, Guadalajara |
| T-K8 | T. koningiopsis | Persea americana | MK791649 | Atlixco, Puebla |
| T-A12 | T. hamatum | Cinnamomun verum | MK791650 | Tlaltipa, Zozocolco de Hidalgo, Veracruz |
| CPO-PCU b | Phytophthora cinnamomi | Persea americana | JQ266267 | Uruapan, Michoacan |
| Scale | Antagonistic Ability |
|---|---|
| 1 | Trichoderma completely overgrew the P. cinnamomi and covered the entire medium surface. |
| 2 | Trichoderma overgrew at least two-thirds of the medium surface. |
| 3 | Trichoderma and the P. cinnamomi each colonized approximately one-half of the medium surface and neither organism appeared to dominate the other. |
| 4 | P. cinnamomi colonized at least two-thirds of the medium surface and appeared to withstand encroachment by Trichoderma. |
| 5 | P. cinnamomi completely overgrew the Trichoderma and occupied the entire medium surface. |
| Strain | Trichoderma Species | PIRG | Standard | Colonization | Standard | ||
|---|---|---|---|---|---|---|---|
| % | Error | % | Error | ||||
| T-AS2 | T. asperellum | 78.32 | a | 5.03 | 44.33 | c | 4.66 |
| T-H3 | T. harzianum | 73.33 | ab | 2.87 | 60.00 | ab | 1.63 |
| T-H4 | T. harzianum | 69.15 | b | 1.13 | 30.00 | d | 1.27 |
| T-AS1 | T. asperellum | 65.30 | b | 1.51 | 29.66 | d | 3.28 |
| T-AS6 | T. asperellum | 53.33 | c | 2.84 | 51.00 | bc | 2.84 |
| T-AS7 | T. asperellum | 51.50 | c | 6.06 | 46.83 | c | 1.10 |
| T-A12 | T. hamatum | 50.99 | c | 6.65 | 27.33 | d | 2.21 |
| T-K11 | T. koningiopsis | 47.33 | cd | 4.55 | 67.83 | a | 2.47 |
| T-K8 | T. koningiopsis | 46.99 | cd | 1.27 | 55.00 | bc | 2.20 |
| T-H5 | T. harzianum | 38.66 | d | 5.10 | 23.83 | d | 3.53 |
| Strain | Trichoderma Species | Growth Rate | |||
|---|---|---|---|---|---|
| Trichoderma spp. | P. cinnamomi | ||||
| T-K8 | T. koningiopsis | 0.1176 | a | 0.0526 | a |
| T-A12 | T. hamatum | 0.1132 | ab | 0.0462 | a |
| T-H3 | T. harzianum | 0.0985 | abc | 0.0098 | a |
| T-H4 | T. harzianum | 0.091 | abcd | 0.0384 | a |
| T-AS1 | T. asperellum | 0.0346 | bcde | 0.0379 | a |
| T-AS2 | T. asperellum | 0.0295 | cde | 0.0342 | a |
| T-AS7 | T. asperellum | 0.0212 | cde | 0.0169 | a |
| T-K11 | T. koningiopsis | 0.0285 | cde | 0.0512 | a |
| T-AS6 | T. asperellum | 0.0174 | de | 0.0240 | a |
| T-H5 | T. harzianum | 0.0077 | e | 0.0316 | a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andrade-Hoyos, P.; Silva-Rojas, H.V.; Romero-Arenas, O. Endophytic Trichoderma Species Isolated from Persea americana and Cinnamomum verum Roots Reduce Symptoms Caused by Phytophthora cinnamomi in Avocado. Plants 2020, 9, 1220. https://doi.org/10.3390/plants9091220
Andrade-Hoyos P, Silva-Rojas HV, Romero-Arenas O. Endophytic Trichoderma Species Isolated from Persea americana and Cinnamomum verum Roots Reduce Symptoms Caused by Phytophthora cinnamomi in Avocado. Plants. 2020; 9(9):1220. https://doi.org/10.3390/plants9091220
Chicago/Turabian StyleAndrade-Hoyos, Petra, Hilda Victoria Silva-Rojas, and Omar Romero-Arenas. 2020. "Endophytic Trichoderma Species Isolated from Persea americana and Cinnamomum verum Roots Reduce Symptoms Caused by Phytophthora cinnamomi in Avocado" Plants 9, no. 9: 1220. https://doi.org/10.3390/plants9091220





