Jaceidin Flavonoid Isolated from Chiliadenus montanus Attenuates Tumor Progression in Mice via VEGF Inhibition: In Vivo and In Silico Studies
Abstract
1. Introduction
2. Results and Discussion
2.1. Identification of the Isolated Compounds
2.2. Screening for Antitumor Activity
2.2.1. In Vitro Assay
2.2.2. In Vivo Assay
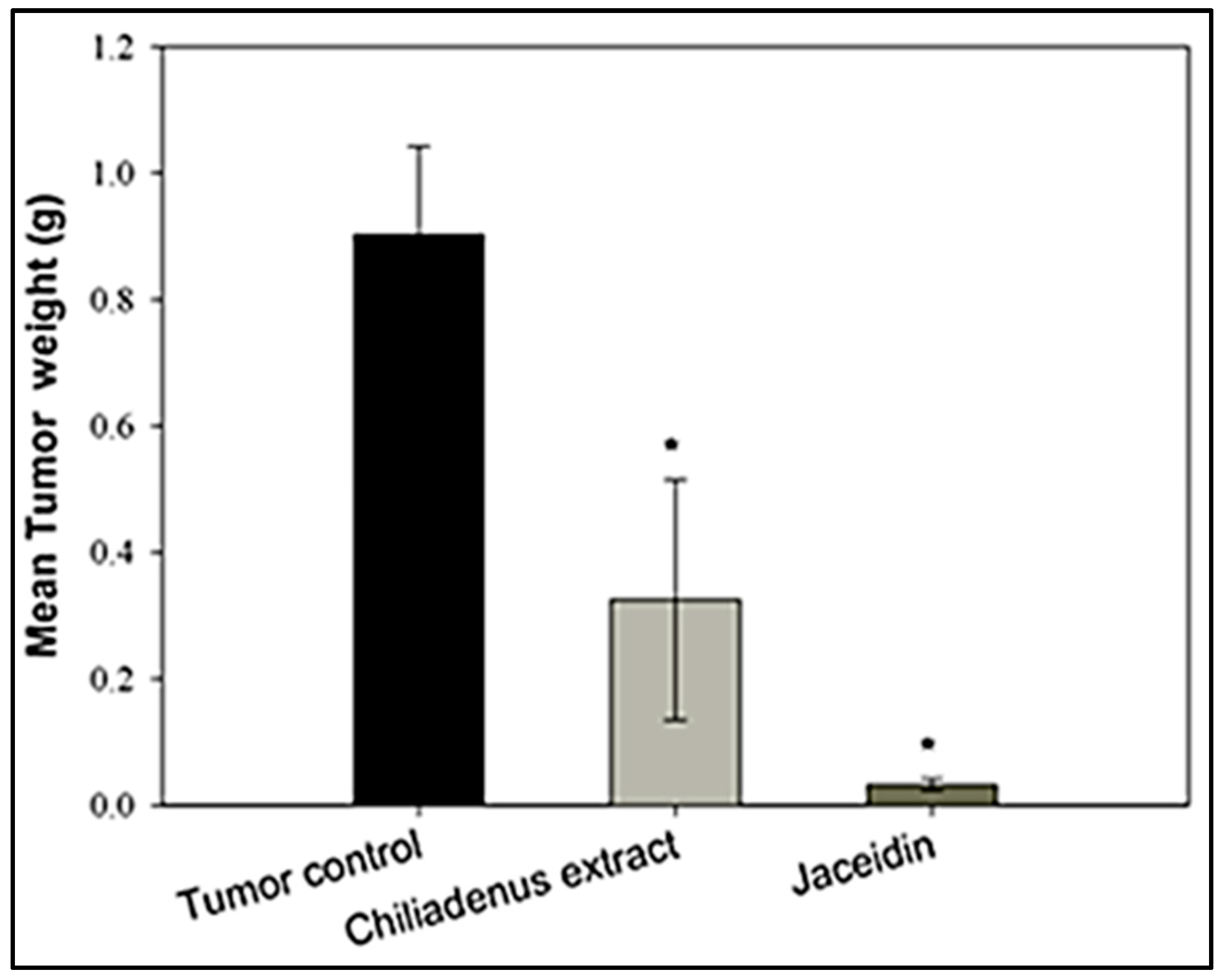
Tumor Weight
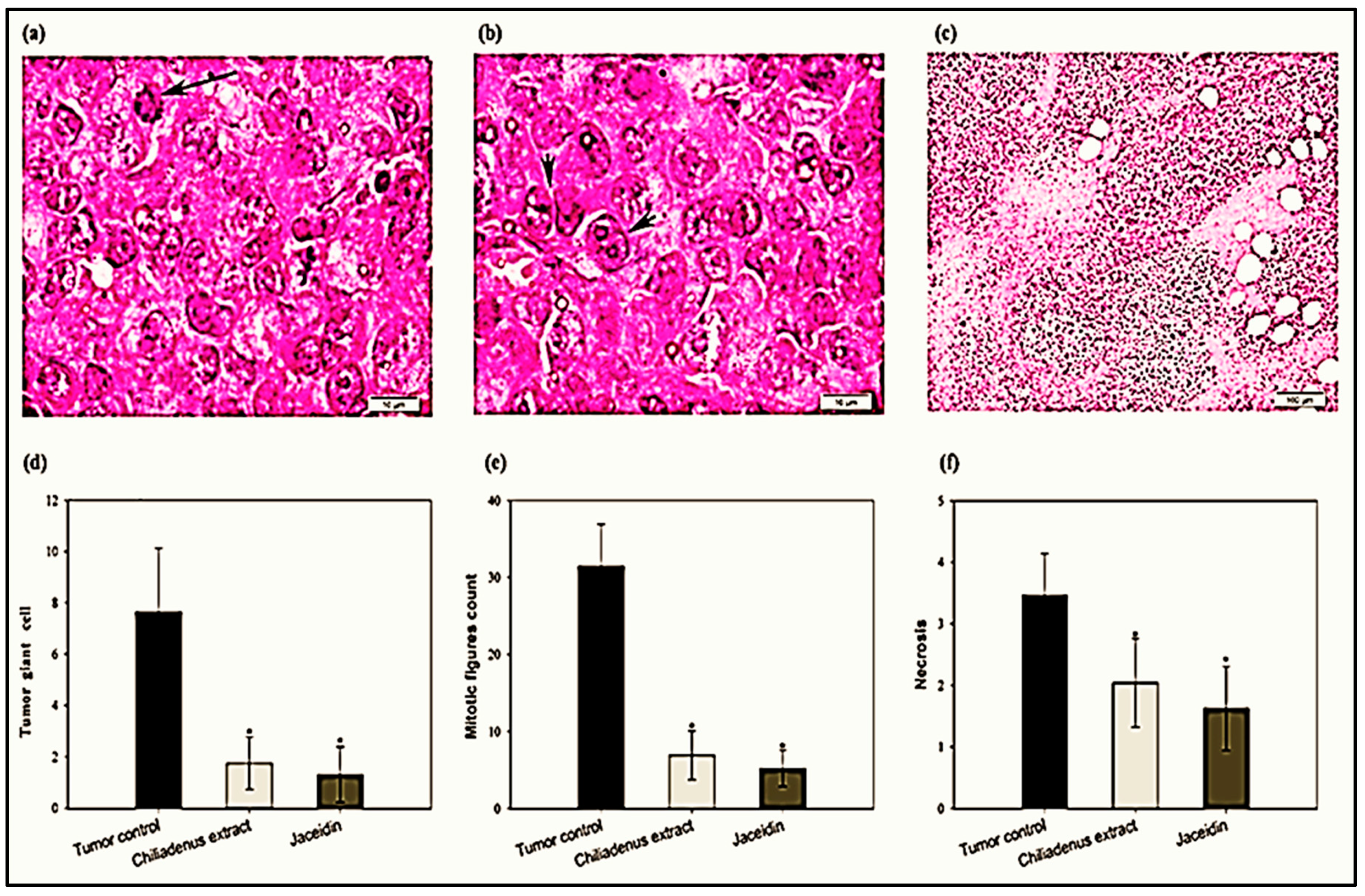
Histopathological Examination of Solid Tumors
2.2.3. Enzyme-Linked Immunosorbent Assays
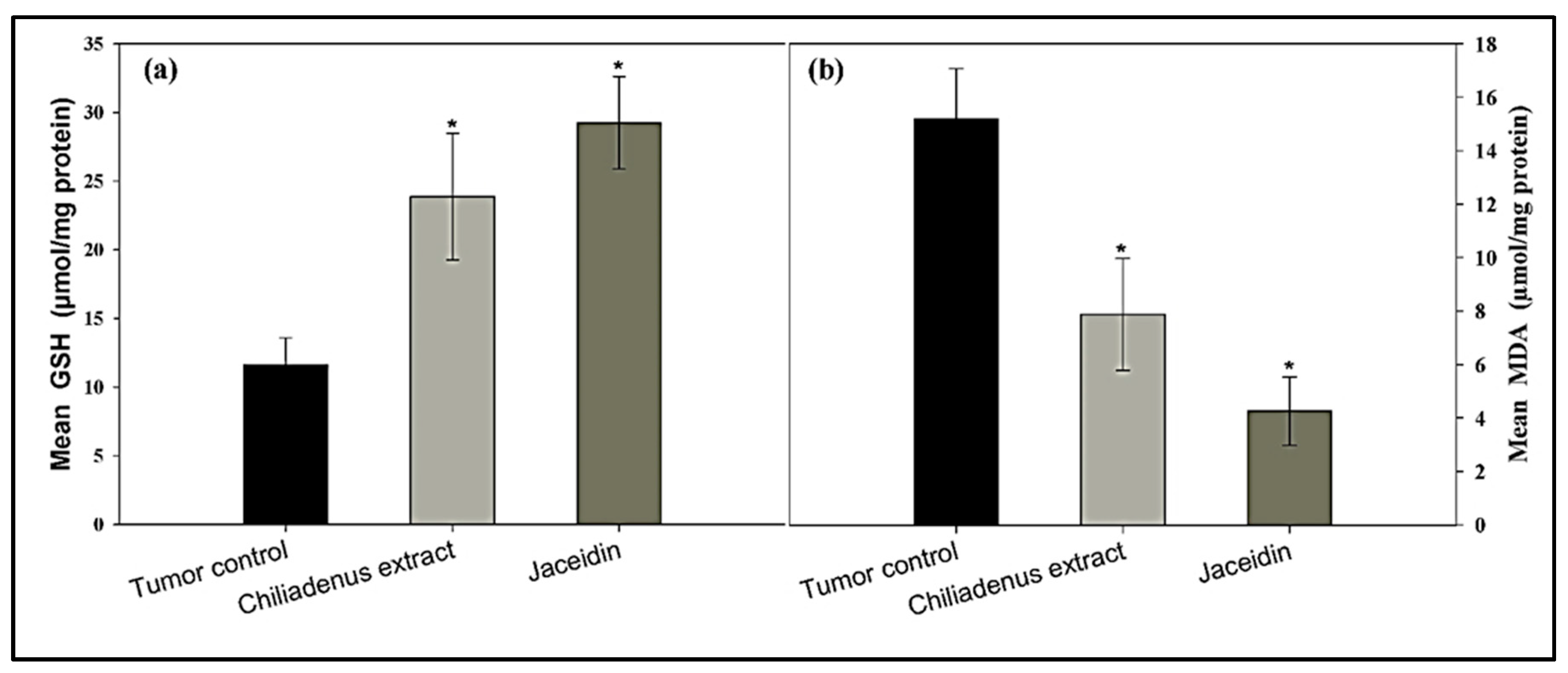
Determination of GSH and MDA Levels
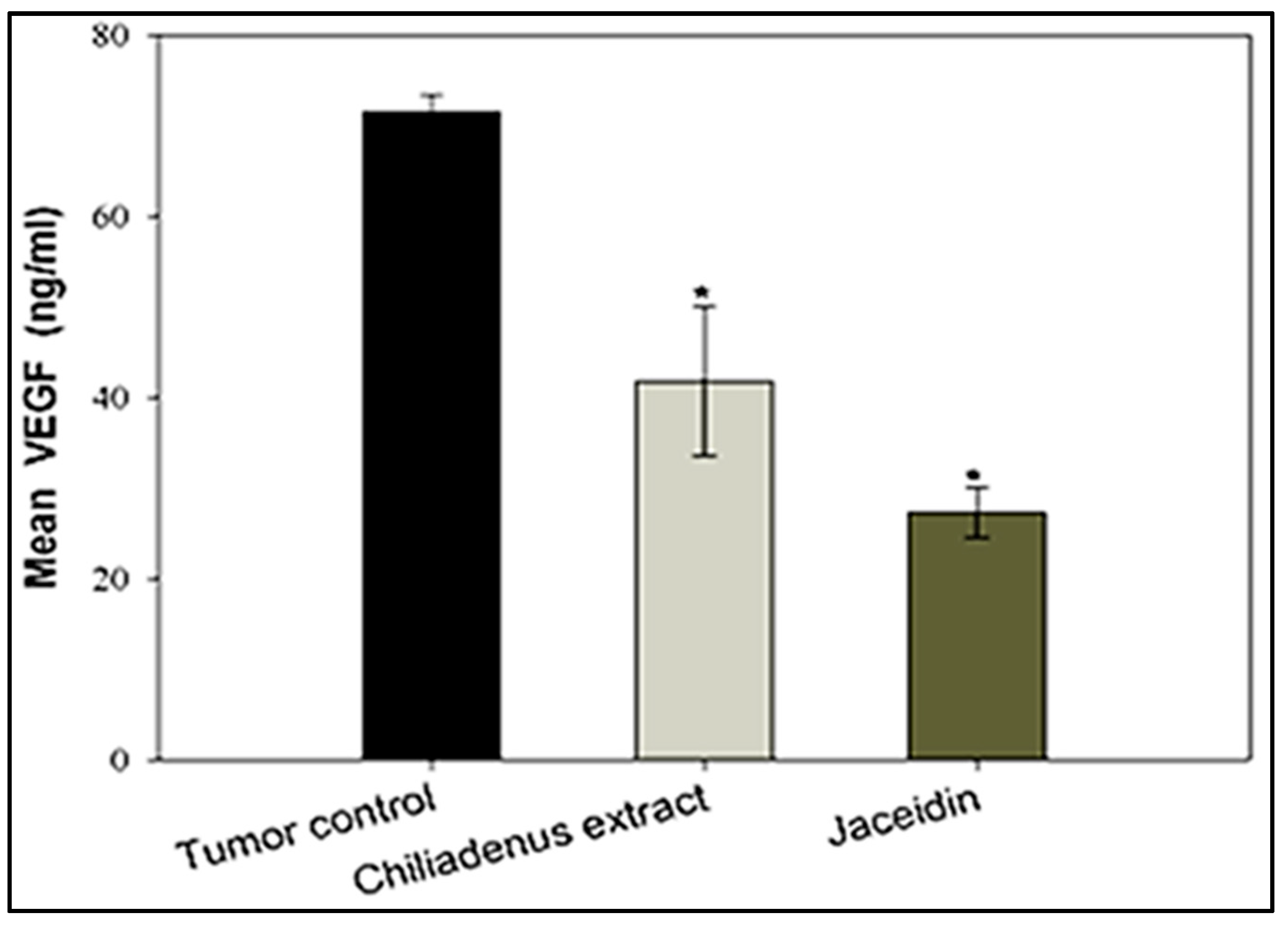
Estimation of VEGF-B Levels
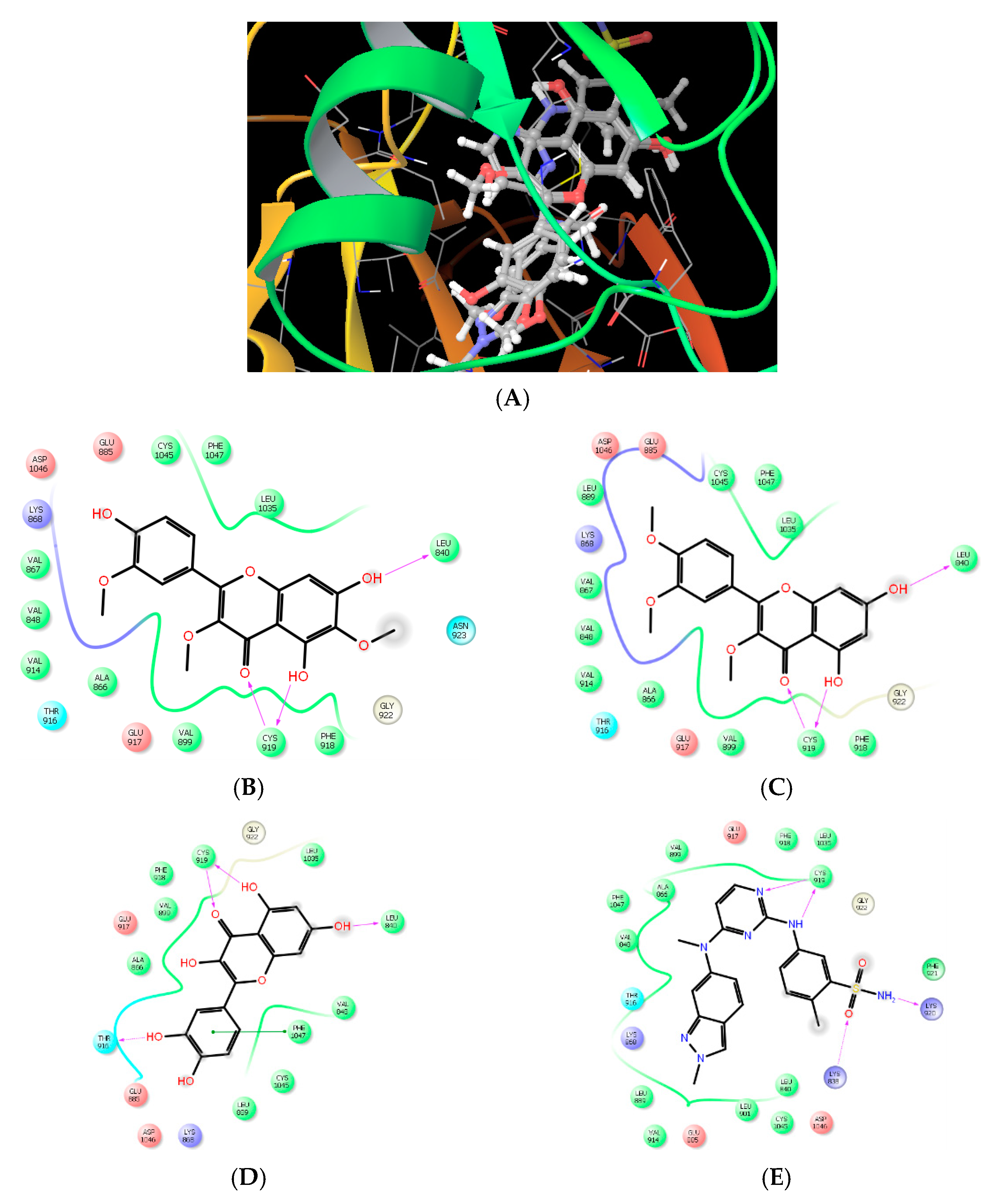
Molecular Modeling and Drug Design
In Silico ADME Predictive Study
3. Material and Methods
3.1. Instruments
3.2. Collection of Plant Materials
3.3. Extraction and Isolation
3.4. Screening for Antitumor Activity
3.4.1. In Vitro Assay
3.4.2. In Vivo Assay
Induction of EAC Solid Tumors in Mice
Experimental Design
Histopathological Examination of Solid Tumors
3.4.3. Enzyme-Linked Immunosorbent Assays
3.4.4. Molecular Modeling and Drug Design
3.4.5. In Silico ADME Predictive Study
3.4.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abotaleb, M.; Samuel, S.M.; Varghese, E.; Varghese, S.; Kubatka, P.; Liskova, A.; Büsselberg, D. Flavonoids in cancer and apoptosis. Cancers 2019, 11, 28. [Google Scholar] [CrossRef]
- Taiwo, B.J.; Fatokun, A.A.; Olubiyi, O.O.; Bamigboye-Taiwo, O.T.; Van Heerden, F.R.; Wright, C.W. Identification of compounds with cytotoxic activity from the leaf of the Nigerian medicinal plant, Anacardium occidentale L. (Anacardiaceae). Bioorg Med. Chem. 2017, 25, 2327–2335. [Google Scholar] [CrossRef] [PubMed]
- Naz, I.; Ramchandani, S.; Khan, M.R.; Yang, M.H.; Ahn, K.S. Anticancer Potential of Raddeanin A, a Natural Triterpenoid Isolated from Anemone raddeana Regel. Molecules 2020, 25, 1035. [Google Scholar] [CrossRef] [PubMed]
- Khan, T.; Ali, M.; Khan, A.; Nisar, P.; Jan, S.A.; Afridi, S.; Shinwari, Z.K. Anticancer Plants: A Review of the Active Phytochemicals, Applications in Animal Models, and Regulatory Aspects. Biomolecules 2020, 10, 47. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Zhou, J.; Lin, Q.; Gong, G.; Sun, H.; Liu, W.; Guo, Q.; Feng, F.; Qu, W. Anti-angiogenic and anticancer effects of baicalein derivatives based on transgenic zebrafish model. Bioorg Med. Chem. 2018, 26, 4481–4492. [Google Scholar] [CrossRef] [PubMed]
- Al Alawi, R.; Alhamdani, M.S.S.; Hoheisel, J.D.; Baqi, Y. Antifibrotic and tumor microenvironment modulating effect of date palm fruit (Phoenix dactylifera L.) extracts in pancreatic cancer. Biomed. Pharmacother. 2020, 121, 109522. [Google Scholar] [CrossRef]
- Klejborowska, G.; Urbaniak, A.; Preto, J.; Maj, E.; Moshari, M.; Wietrzyk, J.; Tuszynski, J.A.; Chambers, T.C.; Huczyński, A. Synthesis, biological evaluation and molecular docking studies of new amides of 4-bromothiocolchicine as anticancer agents. Bioorg Med. Chem. 2019, 27, 115144. [Google Scholar] [CrossRef]
- De Souza, P.O.; Bianchi, S.E.; Figueiró, F.; Heimfarth, L.; Moresco, K.S.; Gonçalves, R.M.; Hoppe, J.B.; Klein, C.P.; Salbego, C.G.; Gelain, D.P. Anticancer activity of flavonoids isolated from Achyrocline satureioides in gliomas cell lines. Toxicol. Vitr. 2018, 51, 23–33. [Google Scholar] [CrossRef]
- Boulos, L. Flora of Egypt: Verbenaceae-Compositae; Al Hadara Pubushing: Cairo, Eypt, 2002; pp. 1–373. [Google Scholar]
- Moustafa, A.A.; Fahmy, D.M.; Zaghloul, M.S.; El-Azeem, S.A.A. Autecology of Chiliadenus montanus as a threatened plant species growing in Saint Katherine protectorate, South Sinai, Egypt. J. Global Agric. Ecol. 2017, 7, 145–163. [Google Scholar]
- Hamed, A.R.; Mohamed, T.A.; Tawfik, W.A.; Hassan, E.M.; Higgins, M.; El-Toumy, S.A.; Dinkova-Kostova, A.T. Bioactive polymethoxylated flavonoids from Chiliadenus montanus. J. Chem. Pharm. Res. 2016, 8, 788–793. [Google Scholar]
- Soliman, F.M.; Moussa, M.Y.; Abdallah, H.M.; Othman, S.M. Cytotoxic activity of flavonoids of Jasonia montana Vahl. (Botsch)(Astraceae) growing in Egypt. Aust. J. Basic Appl. Sci. 2009, 3, 148–152. [Google Scholar]
- El-Sayed, M.; El-Sherif, F.; Elhassaneen, Y.; El-Rahman, A.A. Potential therapeutic effects of some Egyptian plant parts on hepatic toxicity induced by carbon tetrachloride in rats. Life Sci. J. 2012, 9, 3747–3755. [Google Scholar]
- Eissa, T.F.; González-Burgos, E.; Carretero, M.E.; Gómez-Serranillos, M.P. Phenolic Composition and Evaluation of Antioxidant and Cytoprotective Activity of Chiliadenus montanus. Rec. Nat. Prod. 2013, 7, 184. [Google Scholar]
- Hegazy, M.E.F.; Matsuda, H.; Nakamura, S.; Hussein, T.A.; Yoshikawa, M.; Paré, P.W. Chemical constituents and their antibacterial and antifungal activity from the Egyptian herbal medicine Chiliadenus montanus. Phytochemistry 2014, 103, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Helal, E.G.; Abou-Aouf, N.; Khattab, S.M. A possible hypoglycemic and antioxidant effect of herbal mixture extraction in diabetic rats. Egypt. J. Hosp. Med. 2015, 31, 109–119. [Google Scholar] [CrossRef]
- Hussein, M.A. Anti-obesity, antiatherogenic, anti-diabetic and antioxidant activities of J. montana ethanolic formulation in obese diabetic rats fed high-fat diet. Free Radic. Antioxid. 2011, 1, 49–60. [Google Scholar] [CrossRef]
- Ahmed, H.H.; Booles, H.F.; Khalil, W.K.; El Ashmaoui, H.M.; Othman, S.M. Possible Therapeutic Role of Jasonia Candicans and Jasonia Montana Extracts in the Regression of Alzehaimer’s Disease in Expermental Model. Am. J. Biochem. Biotechnol. 2013, 9, 144. [Google Scholar] [CrossRef][Green Version]
- Hegazy, M.E.F.; Ahmed, A.; Hamed, A.R.; El Aty, A.A.A.; Mohamed, N.S.; Paré, P.W. 3-Oxo-γ-costic acid fungal-transformation generates eudesmane sesquiterpenes with in vitro tumor-inhibitory activity. Bioorg Med. Chem. Lett. 2017, 27, 3825–3828. [Google Scholar] [CrossRef]
- El-Bassuony, A.; Kabbash, A. Montanone, a new sesquiterpene from Jasonia montana. Saudi Pharm. J. 2006, 14, 126. [Google Scholar] [CrossRef]
- Mahmoud, A.A. Jasomontanone, a novel bicyclic sesquiterpene from the leaves of Jasonia montana. Nat. Prod. Commun. 2006, 1, 15–19. [Google Scholar] [CrossRef]
- Mohamed, A.E.H.H. Jasonone, a nor-sesquiterepene from Jasonia montana. Z. Nat. B J. Chem Sci. 2007, 62, 125–128. [Google Scholar] [CrossRef]
- Ahmed, A.A.; Ali, A.A.; Mabry, T.J. Flavonoid aglycones from Jasonia montana. Phytochemistry 1989, 28, 665–667. [Google Scholar] [CrossRef]
- Daubney, J.; Bonner, P.L.; Hargreaves, A.J.; Dickenson, J.M. Cardioprotective and cardiotoxic effects of quercetin and two of its in vivo metabolites on differentiated H9c2 cardiomyocytes. Basic Clin. Pharmacol. Toxicol. 2015, 116, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, M.; Ruan, J. Metabolomics analysis reveals the metabolic and functional roles of flavonoids in light-sensitive tea leaves. BMC Plant. Biol. 2017, 17, 64. [Google Scholar] [CrossRef]
- Ramos, S. Effects of dietary flavonoids on apoptotic pathways related to cancer chemoprevention. J. Nutr. Biochem. 2007, 18, 427–442. [Google Scholar] [CrossRef]
- Prakash, S.; Elavarasan, N.; Subashini, K.; Kanaga, S.; Dhandapani, R.; Sivanandam, M.; Kumaradhas, P.; Thirunavukkarasu, C.; Sujatha, V. Isolation of hesperetin-A flavonoid from Cordia sebestena flower extract through antioxidant assay guided method and its antibacterial, anticancer effect on cervical cancer via in vitro and in silico molecular docking studies. J. Mol. Struct. 2020, 1207, 127751. [Google Scholar] [CrossRef]
- Mastinu, A.; Ribaudo, G.; Ongaro, A.; Bonini, S.A.; Memo, M.; Gianoncelli, A. Critical Review on the Chemical Aspects of Cannabidiol (CBD) and Harmonization of Computational Bioactivity Data. Curr. Med. Chem. 2020, 27, 1–24. [Google Scholar] [CrossRef]
- Abdelhameed, R.; Elgawish, M.S.; Mira, A.; Ibrahim, A.K.; Ahmed, S.A.; Shimizu, K.; Yamada, K. Anti-choline esterase activity of ceramides from the Red Sea marine sponge Mycale euplectellioides. RSC Adv. 2016, 6, 20422–20430. [Google Scholar] [CrossRef]
- Bai, M.; Chen, J.J.; Xu, W.; Dong, S.H.; Liu, Q.B.; Lin, B.; Huang, X.X.; Yao, G.D.; Song, S.J. Elephantopinolide AP, germacrane-type sesquiterpene lactones from Elephantopus scaber induce apoptosis, autophagy and G2/M phase arrest in hepatocellular carcinoma cells. Eur. J. Med. Chem. 2020, 112362. [Google Scholar] [CrossRef]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agric. Food Chem. 2001, 49, 2774–2779. [Google Scholar] [CrossRef] [PubMed]
- Croft, K.D. The Chemistry and Biological Effects of Flavonoids and Phenolic Acids a. Ann. N. Y. Acad. Sci. 1998, 854, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Awad, B.M.; Abd-Alhaseeb, M.M.; Habib, E.S.; Ibrahim, A.K.; Ahmed, S.A. Antitumor activity of methoxylated flavonoids separated from Achillea fragrantissima extract in Ehrlich’s ascites carcinoma model in mice. J. Herbmed Pharmacol. 2019, 9, 28–34. [Google Scholar] [CrossRef]
- Pomilio, A.B.; Solá, G.A.R.; Mayer, A.M.; Rumi, L.S. Antitumor and cytotoxic screen of 5, 6, 7-trisubstituted flavones from Gomphrena martiana. J. Ethnopharmacol. 1994, 44, 25–33. [Google Scholar] [CrossRef]
- Walle, T. Methylation of dietary flavones greatly improves their hepatic metabolic stability and intestinal absorption. Mol. Pharm. 2007, 4, 826–832. [Google Scholar] [CrossRef]
- Walle, T. Methylation of dietary flavones increases their metabolic stability and chemopreventive effects. Int. J. Mol. Sci. 2009, 10, 5002–5019. [Google Scholar] [CrossRef]
- Lemmens, K.J.; Vrolijk, M.F.; Bouwman, F.G.; Van Der Vijgh, W.J.; Bast, A.; Haenen, G.R. The minor structural difference between the antioxidants quercetin and 4’O-methylquercetin has a major impact on their selective thiol toxicity. Int. J. Mol. Sci. 2014, 15, 7475–7484. [Google Scholar] [CrossRef]
- Zajkowska, M.; Lubowicka, E.; Malinowski, P.; Szmitkowski, M.; Ławicki, S. Plasma levels of VEGF-A, VEGF B, and VEGFR-1 and applicability of these parameters as tumor markers in diagnosis of breast cancer. Acta Biochim. Pol. 2018, 65, 621–628. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, Y.; Hosaka, K.; Andersson, P.; Wang, J.; Tholander, F.; Cao, Z.; Morikawa, H.; Tegnér, J.; Yang, Y. VEGF-B promotes cancer metastasis through a VEGF-A–independent mechanism and serves as a marker of poor prognosis for cancer patients. Proc. Natl. Acad. Sci. USA 2015, 112, E2900–E2909. [Google Scholar] [CrossRef]
- Lautenschlaeger, T.; George, A.; Klimowicz, A.C.; Efstathiou, J.A.; Wu, C.L.; Sandler, H.; Shipley, W.U.; Tester, W.J.; Hagan, M.P.; Magliocco, A.M. Bladder preservation therapy for muscle-invading bladder cancers on Radiation Therapy Oncology Group trials 8802, 8903, 9506, and 9706: Vascular endothelial growth factor B overexpression predicts for increased distant metastasis and shorter survival. Oncologist 2013, 18, 685. [Google Scholar] [CrossRef]
- Kanda, M.; Nomoto, S.; Nishikawa, Y.; Sugimoto, H.; Kanazumi, N.; Takeda, S.; Nakao, A. Correlations of the expression of vascular endothelial growth factor B and its isoforms in hepatocellular carcinoma with clinico-pathological parameters. J. Surg. Oncol. 2008, 98, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Wu, S.; Liu, Z.; Zhang, W.; Xu, J.; Wang, Y.; Liu, J.; Zhang, D.; Tian, H.; Li, Y. Arenobufagin, a bufadienolide compound from toad venom, inhibits VEGF-mediated angiogenesis through suppression of VEGFR-2 signaling pathway. Biochem. Pharmacol. 2012, 83, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Tan, C.; Ding, H.; Xin, T.; Jiang, Y. Selective VEGFR inhibitors for anticancer therapeutics in clinical use and clinical trials. Curr. Pharm. Des. 2012, 18, 2921–2935. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, Y.; Imayoshi, Y.; Kato, M.; Maeda, K.; Iwabuchi, H.; Shimomura, K. New eudesmane--type sesquiterpenoids and other volatile constituents from the roots of Gynura bicolor DC. Flavour Frag. J. 2011, 26, 55–64. [Google Scholar] [CrossRef]
- El-Askary, H.; Meselhy, M.; Galal, A. Sesquiterpenes from Cymbopogon proximus. Molecules 2003, 8, 670–677. [Google Scholar] [CrossRef]
- Long, C.; Sauleau, P.; David, B.; Lavaud, C.; Cassabois, V.; Ausseil, F.; Massiot, G. Bioactive flavonoids of Tanacetum parthenium revisited. Phytochemistry 2003, 64, 567–569. [Google Scholar] [CrossRef]
- Wang, Y.F.; Wang, X.Y.; Lai, G.F.; Lu, C.H.; Luo, S.D. Three new sesquiterpenoids from the aerial parts of Homalomena occulta. Chem. Biodivers. 2007, 4, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Vichai, V.; Kirtikara, K. Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat. Protoc. 2006, 1, 1112–1116. [Google Scholar] [CrossRef]
- Ghareb, N.; El-Sayed, N.M.; Abdelhameed, R.; Yamada, K.; Elgawish, M.S. Toward a treatment of diabesity: Rational design, synthesis and biological evaluation of benzene-sulfonamide derivatives as a new class of PTP-1B inhibitors. Bioorg. Chem. 2019, 86, 322–338. [Google Scholar] [CrossRef]

| Compound No. | HepG2 (IC50) a,b in µM | MCF-7 (IC50) a,b in µM | HFB-4 (IC50) a,b in µM |
|---|---|---|---|
| 1 | 48.2 ± 0.30 | 45.6 ± 0.50 | >200 |
| 2 | 39.7 ± 0.65 | 41.5 ± 0.55 | >200 |
| 3 | 30.5 ± 0.61 | 10.7 ± 0.40 | >200 |
| 4 | 9.7 ± 0.26 | 9.3 ± 0.30 | >200 |
| 5 | 33.4 ± 0.49 | 38.5 ± 0.55 | >200 |
| 6 | 35.1 ± 0.60 | 33.2 ± 0.35 | >200 |
| Doxorubicin c | 4.58 ± 0.47 | 4 ± 0.25 | 14.7 ± 0.50 |
| ADME Prediction Parameters | Compound | Reference Flavonoid | ||
|---|---|---|---|---|
| 3 | 4 | Quercetin | Genistein | |
| Mol-MW a | 344.3 | 360.3 | 302.2 | 286.2 |
| DonorHB b | 1 | 2 | 3 | 4 |
| AcceptHB c | 5.2 | 6 | 5.2 | 4.5 |
| QPlogPo/w d | 2.7 | 2.2 | 0.517 | 1.05 |
| #rotor e | 5 | 6 | 5 | 4 |
| PSA f | 95.3 | 115.5 | 140.08 | 118.6 |
| QPlogS g | −4.1 | −3.8 | −3.09 | −3.05 |
| QPlogHERG h | −4.9 | −4.7 | −5.3 | −5.07 |
| QPPCaco i | 549.5 | 287.7 | 21.03 | 58.2 |
| QPP MDCK j | 259.01 | 128.7 | 7.62 | 22.89 |
| QPlogKhsa k | 0.163 | 0.013 | −0.318 | −0.198 |
| % Human Oral Absorption l | 92.2 | 83.92 | 53.65 | 64.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elhady, S.S.; Eltamany, E.E.; Shaaban, A.E.; Bagalagel, A.A.; Muhammad, Y.A.; El-Sayed, N.M.; Ayyad, S.N.; Ahmed, A.A.M.; Elgawish, M.S.; Ahmed, S.A. Jaceidin Flavonoid Isolated from Chiliadenus montanus Attenuates Tumor Progression in Mice via VEGF Inhibition: In Vivo and In Silico Studies. Plants 2020, 9, 1031. https://doi.org/10.3390/plants9081031
Elhady SS, Eltamany EE, Shaaban AE, Bagalagel AA, Muhammad YA, El-Sayed NM, Ayyad SN, Ahmed AAM, Elgawish MS, Ahmed SA. Jaceidin Flavonoid Isolated from Chiliadenus montanus Attenuates Tumor Progression in Mice via VEGF Inhibition: In Vivo and In Silico Studies. Plants. 2020; 9(8):1031. https://doi.org/10.3390/plants9081031
Chicago/Turabian StyleElhady, Sameh S., Enas E. Eltamany, Amera E. Shaaban, Alaa A. Bagalagel, Yosra A. Muhammad, Norhan M. El-Sayed, Seif N. Ayyad, Amal A. M. Ahmed, Mohamed S. Elgawish, and Safwat A. Ahmed. 2020. "Jaceidin Flavonoid Isolated from Chiliadenus montanus Attenuates Tumor Progression in Mice via VEGF Inhibition: In Vivo and In Silico Studies" Plants 9, no. 8: 1031. https://doi.org/10.3390/plants9081031
APA StyleElhady, S. S., Eltamany, E. E., Shaaban, A. E., Bagalagel, A. A., Muhammad, Y. A., El-Sayed, N. M., Ayyad, S. N., Ahmed, A. A. M., Elgawish, M. S., & Ahmed, S. A. (2020). Jaceidin Flavonoid Isolated from Chiliadenus montanus Attenuates Tumor Progression in Mice via VEGF Inhibition: In Vivo and In Silico Studies. Plants, 9(8), 1031. https://doi.org/10.3390/plants9081031








