1. Introduction
Fruit trees present a widely genetic diversity reflected in their broad range of mopho-agronomic, multiple pharmacological activities, and biochemical composition, which are, accordingly, very diverse. Their fruits are fundamentally very rich in terms of bioactive molecules, including phytochemicals (phenolics, carotenoids, lignans, stilbenes, etc.), vitamins (mainly vitamins, A, C, E, and K), minerals (i.e., potassium, calcium, and magnesium), and dietary fibers, which have vital functions in human health by alleviating several chronic diseases [
1,
2,
3,
4].
Fruits (berries in particular) as a source of nutrients and bioactive molecules and health-promoting properties, remains, so far, a hot topic in the scientific community. Epidemiological reports have consistently shown sufficient evidence proving that the regular consumption of berries is directly linked to the prevention of coronary diseases. The antioxidant attributes of these compounds act as reducing agents, metal chelators, hydrogen donors, and singlet oxygen quenchers [
2,
3,
4].
The strawberry tree (
A. unedo) is generally considered a small tree, usually smaller than 4 m. During autumn, it bears orange colored fruit, naturally grown as a population or solitary tree in Mediterranean countries, such as Morocco, Tunisia, Algeria, Turkey, Syria, Greece, Croatia, France, Portugal, and Spain [
5]. The strawberry tree is recognized as a medicinal species, with high antioxidant potential, due mainly to polyphenols concentrated in its fruit, which play a major role in safeguarding health, because of their biological functions, such as antimutagenicity, anticarcinogenicity, and antiaging [
6]. Strawberry tree fruit, being a red spherical berry, is not only suitable for the production of alcoholic beverages, jams, jellies, and marmalades [
7], but also for medicinal purposes [
8].
In Morocco—a hotspot of fruit tree diversity—
A. unedo is known as “Sasnou”, and it is widely used by the local population in traditional medicine as antiseptics, diuretics, laxatives, and, more recently, used in therapy for diabetes and arterial hypertension [
8,
9]. The latter has been demonstrated in previous reports (the total tannin concentration of the leaf extract produces an in vitro inhibition of platelet aggregation) [
10,
11]. Both fruits and leaves have been used for medicinal purposes for centuries, as they possess good antimicrobial and antioxidant proprieties. Furthermore, strawberry tree fruits are well-known as a good dietary source of antioxidants, including phenolic compounds (e.g., anthocyanins and other flavonoids, gallic acid derivatives, and tannins), vitamins C and E, and carotenoids [
7,
8,
12,
13,
14,
15,
16,
17]. These bioactive plant secondary metabolites are systematically involved in the species’ biological systems, such as pigmentation, growth, reproduction mechanisms, protection against predators, etc. [
14]. Moreover, they have been used since ancient times as primary and supplemental treatments for various ailments, supporting normal physiological functions [
18]. Phenolic compounds can amplify the human defense system to eliminate cancer cells and block angiogenesis, which is the formation of new blood vessels, essential for tumor development [
19]. Recently, several studies have shown that strawberry antioxidants and bioactive compound amounts strongly depend on genetic background. Moreover, geographical origin has significant influence on the biosynthesis of these nutriments during ripening [
20,
21,
22].
More recently, there has been an increased interest in using naturally occurring phytochemicals from novel, raw material, for the prevention and treatment of different chronic human diseases [
23,
24,
25,
26,
27]. Among phytochemicals, phenolics from a large number of fruits and beverages have been shown to prevent cancer and cardiovascular diseases [
28]. Strawberry tree fruit is also a rich source of phytochemicals. Previous phytochemical studies on the plant showed the presence of three anthocyanins: delphinidin 3-
O-galactoside, cyanidin 3-
O-galactoglucoside, and cyanidin 3-
O-galactoside [
29].
Arbutus unedo L. fruits were reported very high when compared with 27 of the other fruits [
30].
Despite the high popularity of these wild fruits in Morocco, the diversity within the species is still largely unknown. Moreover, data regarding a complete ethnomedicinal and nutritional assessment of A. unedo fruits are missing. The high nutritional quality and bioactive compounds of these berries are likely to be lost if not documented. Therefore, this species remains, so far, underexploited, due to the lack of awareness of their potential, market demand, and value addition. However, to date, the genetic resources of A. unedo still face a serious threat of extinction, mainly because of climate change and extensive urbanization. To preserve the existing diversity within the species, scientific survey, core-collection building, and large-scale assessment are urgently required to ensure food and nutritional security of rural populations and to achieve sustainable development.
Biochemical markers have been widely used in breeding studies and in investigations into diversity of species, and the relationship between genotypes and their wild relatives. More recently, biochemical content, in particular, bioactive content of fruits, has been widely researched, in terms of their human health benefits. Scientists are now searching to find genotypes that can meet farmers’ and industrial requirements, regarding their agronomic and functional properties, in order to use them in breeding programs to develop new chemotypes that hold high nutritional proprieties, making them suitable ingredients for the food industry and for health applications [
31]. However, in Morocco, very few studies have been devoted toward strawberry tree fruits. Thus, the objective of this study was to investigate, for the first time, strawberry tree fruits in terms of their main physical and biochemical characteristics, in a comparative scheme of five prospected Moroccan clones. The main purposes of this work are: (i) to assess the quality of strawberry tree fruits (pH, titratable acidity, soluble solid); (ii) to evaluate the polyphenolic profiles and antioxidant activities of strawberry tree fruits using three methods (DPPH, ABTS, and beta-carotene bleaching assays); (iii) to identify the correlations among all studied parameters in order to determine the ones that are potentially important in assessing strawberry tree genotypes; and (iv) to evaluate the biochemical diversity among the strawberry tree genotypes belonging to several Moroccan geographical origins. Herein, we intend to present a complete database regarding biochemical composition and antioxidant properties of these cultivars in order to valorize them as an invaluable source of nutrients and nutraceuticals. This will facilitate the breeding and selection of new strawberry tress cultivars by integrating the scattered desired attributes.
2. Materials and Methods
2.1. Plant Material
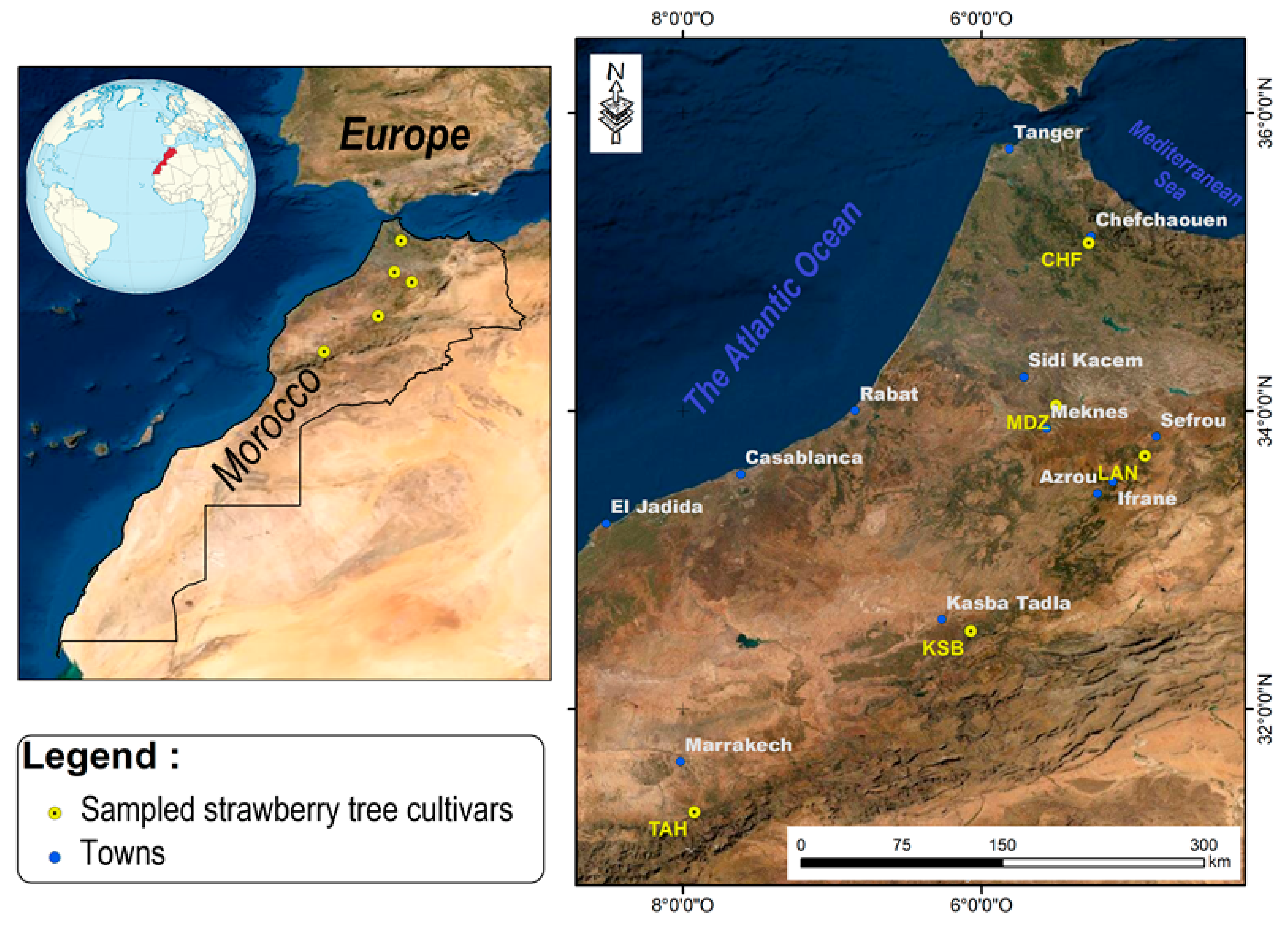
Five genotypes of the strawberry tree (
Arbutus unedo L.) were collected between October and November 2019 named (Chefchaouen (CHF), Moulay Driss Zerhoun (MDZ), Laanoucer (LAN), El Ksiba (KSB), and Thnaout (TAH)), where they grew spontaneously (
Table 1;
Figure 1). Morphological key characters of both tree and its flowers and leaves were used for in-situ trees selection (International Plant Genetic Resources Institutes (IPGRI)) and CIHEAM) [
32]. This selection was also performed through a survey with a local population, since surveyed geographical sites represent an endemic area for species growth in Morocco. Each area hosts typical, spontaneously growing strawberry tree clones.
The strawberry tree cultivars, herein examined, presented round-shaped fruits, with an index varying from 0.916 to 1.001, and weight ranging between 1.32 and 4.46 g. The cluster length ranged from 32.64 to 74.01, while the number of fruits per cluster varied from 2.71 to 5.34. Moreover, leaf weight was in the range of 0.246 to 0.486 g. Likewise, the flower cluster length varied from 7.15 to 13.41. The investigated cultivars displayed significant differences based on their fruits, flowers, and leaf morphology.
At each geographical location, random fruits, having uniform size and maturity, with no diseases and visual blemishes, were harvested at their fully ripened stage, and transferred to the laboratory for biochemical and phytochemical analysis. Fruits picked at different positions around the canopy were considered fully ripened when their color turned from yellow/green to red, and when they were easily separated from the twig. Fruits were frozen at −80 °C, freeze-dried, and ground, then kept in appropriate conditions for subsequent use.
2.2. UV-VIS Profile Determination
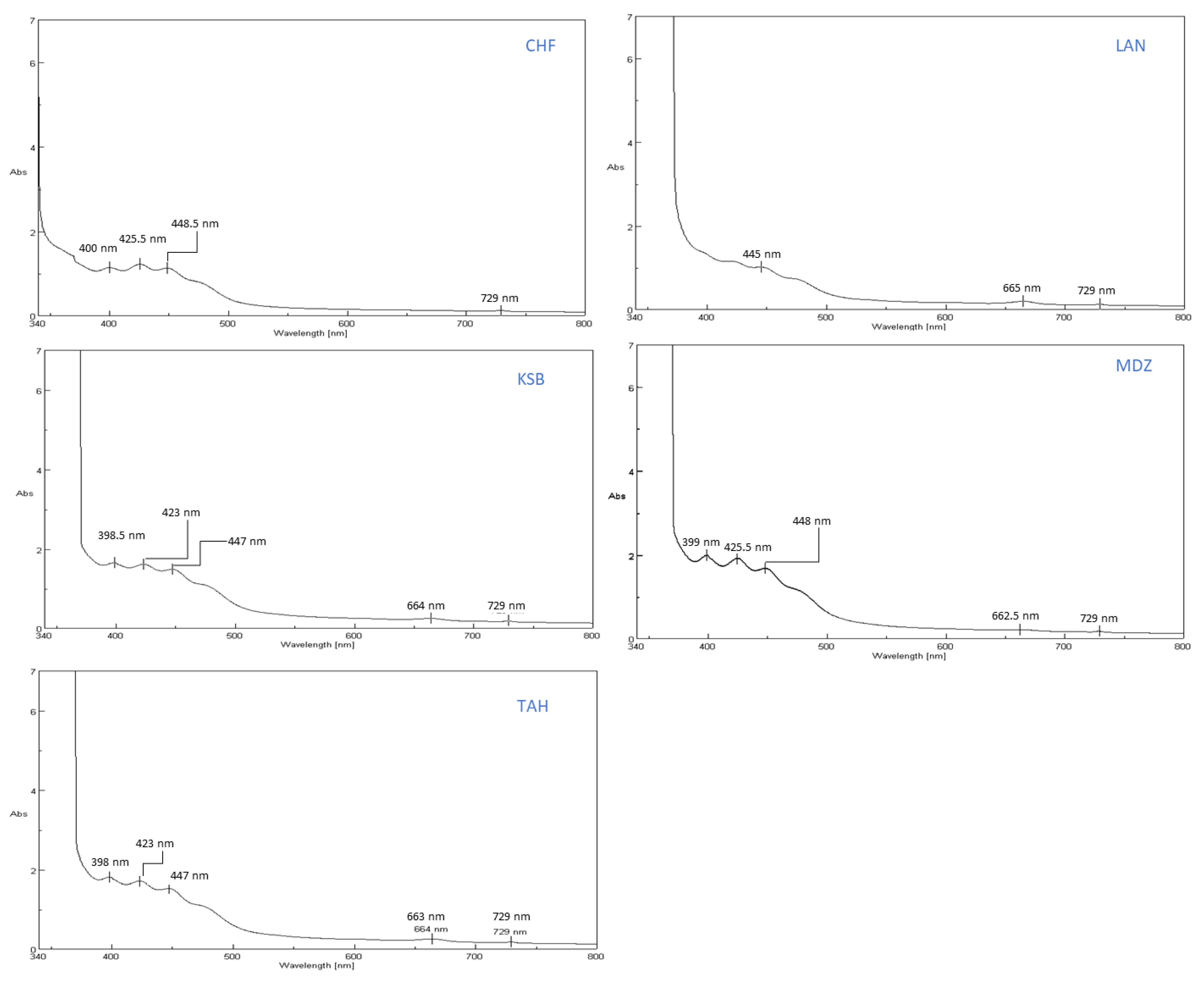
The bioactive constituents of the sample extracts were scanned in a wavelength ranging from 340–800 nm by using a UV-VIS spectrophotometer (spectrophotometer Spectra Physics JASCO V730, instrument, JASCO corporation 2967-5 Ishikawa-matchi Hachioji-shi, Tokyo 192-8537, Japan), and the main absorbance peaks were identified.
2.3. Biochemical Analyses
Total soluble solids (TSS) were assessed according to Association of Official Analytical Chemists (AOAC) [
33] with a digital refractometer (Atago N1; Atago Co. Ltd., Tokyo, Japan), at room temperature, and expressed as Brix. Total titratable acidity (TA) was assessed according to AOAC [
33] using an automatic titration device (877 Titrino plus, Metrohm ion analyses CH9101, Herisau, Switzerland), 0.1 N NaOH up to pH 8.1, using 1 mL diluted juice in 25 mL distilled H
2O. Results were expressed as g of malic acid per 100 g FW. The pH was measured using a pH meter, according to the method described by AOAC [
33].
2.4. Organic Acids and Ascorbic Acid Profiles
A total of 0.5 g of each sample was extracted with 5 mL of Milli-Q water by incubation for 30 min under ultra-sonication. The mixture was then centrifuged at 150,000× g for 20 min (Sigma 3–18 K; Sigma, Laborzentrifugen GmbH, Osterode am Harz, Germany). The supernatant was filtered using a 0.45 μm Millipore filter and immediately used for analysis. All extractions were carried out in triplicate.
The chromatographic analysis was performed as reported by Garcia-Salas et al. [
34]. Briefly, 10 μL of each extract were injected into a Hewlett-Packard HPLC Series 1100 (Wilmington, DE, USA) using an autosampler. The UV detector was set at 210 nm and coupled with a refractive index detector (HP 1100, G1362A). A column (Supelcogel TM C−610H column 30 cm × 7.8 mm) and apre-column (Supelguard 5 cm × 4.6 mm; Supelco, Bellefonte, PA, USA) were used for the analyses of both organic acids and ascorbic acid. The elution buffer consisted of 0.1% phosphoric (
v/v) at a flow rate of 0.5 mL min
−1. Organic acids were measured at a wavelength of 210 nm using a diode-array detector (DAD). Analysis were performed in triplicated and results were expressed as g 100 g
−1 of dry weight (DW).
2.5. Phytochemical Composition
2.5.1. Extraction Procedure
One g of powder from each sample was mixed with 25 mL of ethanol (1:25, w/v) at 25 °C for 15 min using an IKA T−18 digital Ultra-Turrax homogenizer. The homogenate was then centrifuged for 10 min at 6000 rpm and the supernatant was removed from the residue. The latter was homogenized and the supernatant removed as above. The supernatants were then combined and filtered.
2.5.2. Total Phenols (TP)
Phenolic contents (PT) of sampled strawberry tree fruits were determined, according to Ben Salem et al. [
35]. Moreover, 100 µL of diluted sample (1/100) with ethanol was added to 400 µL of 1/10 diluted Folin Ciocalteu reagent. After 5 min, 500 µL of 10% (
w/v) sodium carbonate solution was added. After 1 h of incubation at room temperature, absorbance at 765 nm (spectrophotometer Spectra Physics JASCO V730, corporation 2967-5 Ishikawa-matchi Hachioji-shi, Tokyo 192-8537, Japan) was measured in triplicate. Total polyphenols content (TP) was expressed as milligrams gallic acid equivalents (GAE) per g dry weight of strawberry tree fruit.
2.5.3. Total Flavonoids (TF)
Total flavonoids of samples were determined, as described by Lamaison and Carnat [
36]. Moreover, 1 mL of the sample was mixed with 1 mL of a 2% aluminum chloride solution. The mixture was incubated at room temperature for 15 min. The absorbance was measured at 430 nm (with a spectrophotometer Spectra Physics JASCO V730, Japan). The results were expressed as rutin equivalent per dry weight of strawberry tree fruit.
2.5.4. Total Anthocyanins
Total anthocyanins content (TAC) of samples were determined using the pH differential method with some modifications, according to Giusti and Wrolstad [
37]. A 1 mL aliquot of each sample extract was mixed with 980 μL of KCl buffer (pH1.0) and NaOAc buffer (pH 4.5). After the mixtures were incubated at a room temperature for 15 min, the absorbance was read at 510 nm and 700 nm for both sets of pH 1.0 and 4.5 solutions. Total anthocyanins were estimated using Equation (1), and their concentrations were expressed as milligrams of cyanidin-3-glucoside equivalents in 100 g of DW.
where,
A: absorbance = (A510 nm–A700 nm)
pH1.0—(A510 nm–A700 nm)
pH4.5;
MW: molecular weight (449.2 g/mol);
DF: dilution factor;
Ɛ: molar absorptivity coefficient of cyanidin-3-glucoside (260.900 L/mol cm).
2.6. Antioxidant Activities
The antioxidant activities were determined using three different assays: (i) DPPH assay, (ii) ABTS assay, and (iii) the β-Carotene bleaching test. Each essay was performed in triplicate, using Lambda EZ 150 (Spectra Physics JASCO V730, Japan) spectrophotometer. For all essays, a calibration curve within a range of 0.5−5.0 mg of ascorbic acid g−1 was used for the quantification of the three methods, showing good linearity (R2 ≥ 0.998). Results were expressed accordingly as mg ascorbic acid equivalent (AAE) per dry weight.
2.6.1. DPPH Free Radical-Scavenging Capacity
The DPPH (10.1-diphenyl−2-picrylhydrazyl) radical scavenging activity of the samples was determined according to Ben Salem et al. [
35]. Thus, DPPH solution was prepared by dissolving 0.1 g of DPPH in 1 L methanol (HPLC quality). Then, to each extract (125 µL), 1 mL of this solution was added. The mixture was stirred thoroughly and incubated in the dark at room temperature for 10 min. A control solution was prepared by adding equal volumes of DPPH and methanol. The absorbance of both sample and control was measured at 517 nm, and their scavenging activity of DPPH radicals was determined using the following equation: DPPH scavenging activity % inhibition).
where, Ac and AS refer to the control and sample absorbances, respectively.
IC50 value (mg extract/g DW) defines the inhibitory concentration at which tested radicals were scavenged by 50%. It was calculated by plotting inhibition percentage of each test against the sample extract dilutions.
2.6.2. ABTS Assay
The ABTS (20.2-azinobis- (3-ethylbenzothiazoline−6-sulphonic acid)) radical scavenging assay was performed according to Dorman and Hiltunen. [
38]. Thus, 990 µL of each sample extract was incubated in 10 µL ABTS (7 mM)-ETOH and 2.45 mM potassium persulfate solution after sonicated at 20 °C for 15 min during 16 h in the dark. The mixtures were the incubated for 18 h in the darkness at room temperature. The ethanol (HPLC quality) was used to dilute the stock solution of ABTS until absorbance of 0.70 ± 0.05 was reached at a wavelength of 734 nm.
2.6.3. β-Carotene Bleaching Assay
The Beta-carotene blanching essay was carried out according to Barros et al. [
39]. β-carotene (0.5 mg) in 1 mL of chloroform was taken in an amber bottle and mixed with 200 mg of linolenic acid and 600 mg of Tween 80 (polyoxyethylene sorbitan monopalmitate). The chloroform was removed under nitrogen, and the resulting solution was immediately diluted with 30 mL of triple distilled water; the emulsion was mixed well for 1 min. The emulsion was further diluted with 120 mL of oxygenated water and used for assay. To each sample extract (0.5 mL), 2.5 mL of the prepared emulsion mixture was added and then vigorously mixed. A control consisting 0.5 mL of ethanol and 2.5 mL of emulsion was also analyzed. The absorbance of reaction mixture was read immediately (t = 0) at 470 nm against blank, consisting of emulsion mixture, except β-carotene, and at the 60 min interval for 2 h (t = 120). The tubes were incubated in a water bath at a temperature of 50 °C between measurements. Color measurement was monitored until the β-carotene color disappeared. The linoleic acid peroxidation inhibition uses the following Equation (3):
where, A
o and A
oo refer to the absorbance measured at the beginning of samples and control incubation, respectively. A
t and A
ot are the final absorbance of samples and control, respectively.
2.7. Extraction and Determination of Polyphenolic Compounds
2.7.1. Extraction Method
Samples (1 g) were mixed with 10 mL of methanol:water (80:20, v/v); the mixtures were then sonicated for 30 min and macerated for one hour in refrigeration (4 °C). The samples were then centrifuged for 10 min, 8000× g at 4 °C. The supernatants were collected and the pellets were mixed with 10 mL of acetone:water (70:30, v/v), and the same steps were repeated (sonication, maceration, and centrifugation). Then, the supernatants were combined and evaporated to dryness using a rotary evaporator R−205 under reduced pressure, at 40 °C. Moreover, 5 mL of methanol were added to the residue, and the mixture was well-shaken in a Vortex for 2 min. Due to the high sugar content present in the samples, which could interfere with the HPLC column, the samples were loaded onto a C18 Sep-Pak cartridge, previously conditioned with 5 mL of methanol, 5 mL of pure water, and then, with 5 mL of 0.01 mol/L HCl. The cartridge was washed with 5 mL of pure water and then eluted with acidified methanol (0.1 g/L HCl). The collected fractions were stored at −20 °C until further use.
2.7.2. Determination of Polyphenolic Profiles
Polyphenolic profiles of all samples were determined according to Genskowsky [
40]. A volume of 20 µL of the samples were injected into a Hewlett-Packard HPLC series 1200 instrument equipped with a diode array detector (DAD) and a C18 column (Mediterranean sea 18, 25 × 0.4 cm, 5 cm particle size) from Teknokroma, (Barcelona, Spain). This spectrophotometer uses a Xenon lamp, illuminant D65, 10° observer, SCI mode, 11 mm aperture of the instrument for illumination and 8 mm for measurement. Polyphenolic compounds were analyzed in standard and sample solutions, using a gradient elution at 1 mL/min. The mobile phases were composed by formic acid in water (1:99,
v/v) as solvent A and acetonitrile as solvent B. The chromatograms were recorded at 280, 320, 360, and 520 nm. Polyphenolic compound identification was carried out by comparing UV absorption spectra and retention times of each compound with those of pure standards injected in the same conditions.
2.8. Statistical Analysis
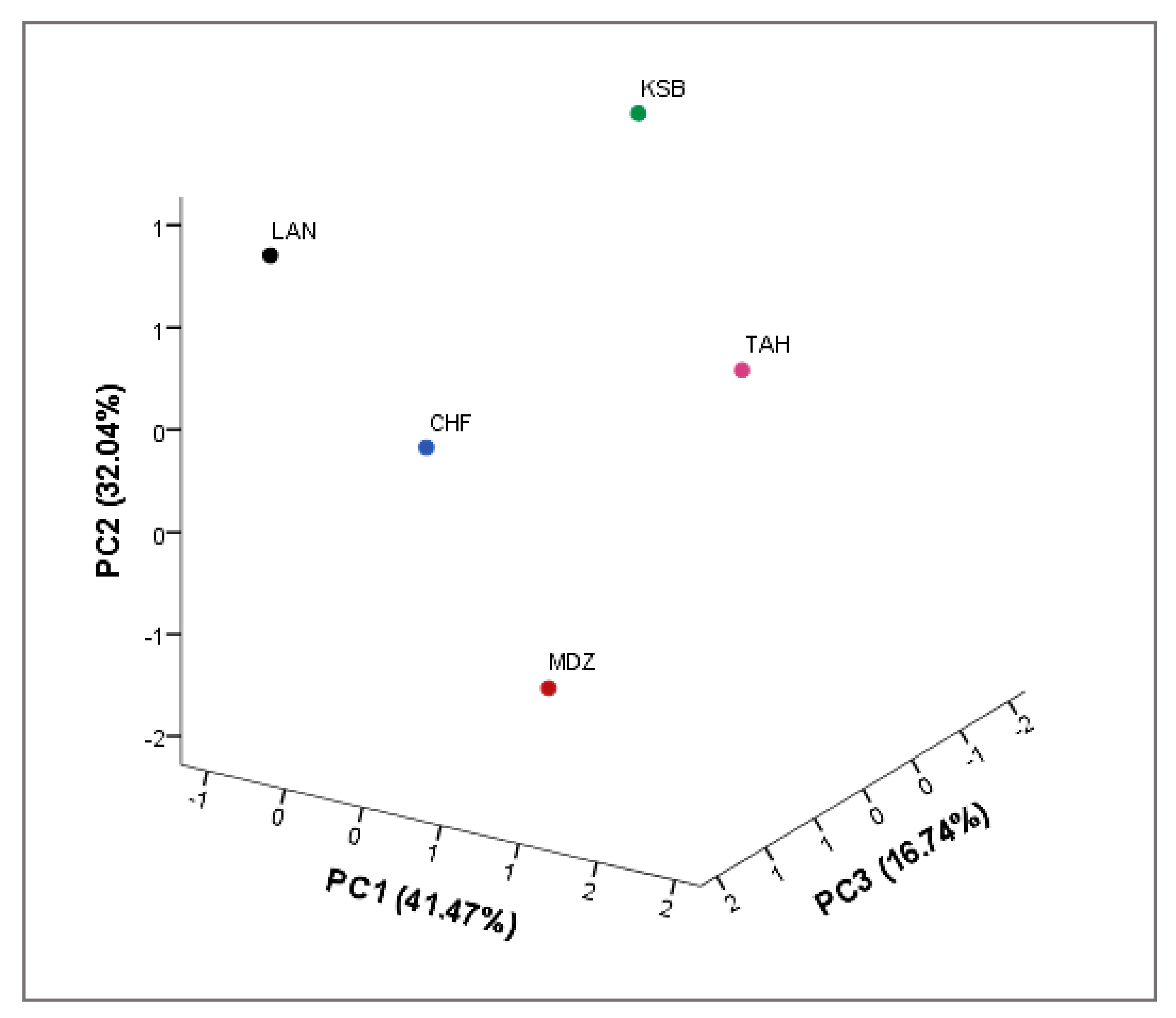
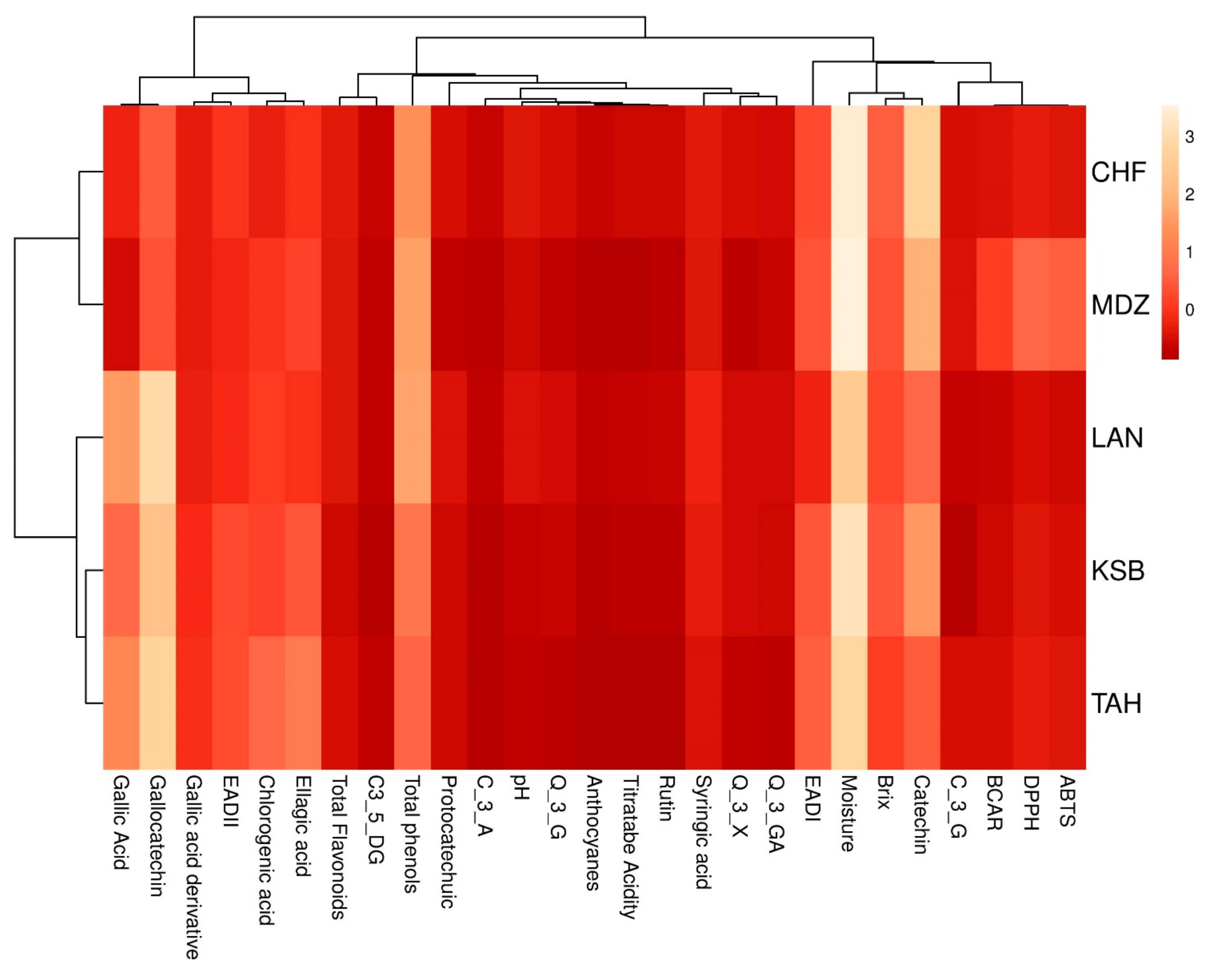
All analyses were performed in triplicate. The means were evaluated according to descriptive statistics represented as mean ± SE. Data analysis was performed using IBM SPSS v22. Analysis of variance (ANOVA) was performed to test significant differences among the samples. The differences in studied variables were estimated with Duncan new multiple range (DMRT) test. Correlation coefficients and their levels of significance were calculated using the Pearson correlation. Principal Component Analysis (PCA) was carried out using correlation matrix to achieve a better understanding of the trends and relationships among investigated biochemical variables. Moreover, it served to determine the main factors to reduce the number of effective parameters to use in decimating the sampled genotypes. In addition, a scatter plot was created according to the first three principal components (PC1, PC2, and PC3). A two-dimensional hierarchical clustered heatmap was applied to the dataset using R software 3.0.2. Prior to these analyses, data were standardized to a comparable scale (µ = 0 and σ = 1). In this presentation of data, the effect size measure is represented by the color intensity. The heatmap groups similar rows and columns together, with their similarity represented by a dendrogram.
With the advent of high-throughput experiments, biochemical attributes have often been coupled with chemometrics to extract features relevant for better understanding of multiple associations between these attributes. For this purpose, heatmap and PCA are the two popular methods for analyzing this type of data. In this study, both methods were used to investigate the change of investigated metabolites of sampled cultivars from different geographical origins. These methods are of importance to achieve better understanding of complex biological systems, where one-way direction is assumed [
41]. They also aim to explore the associations between these factors with regards to the genetic factor.
4. Conclusions
This study is likely to provide the first set of data on the physicobiochemical attributes of strawberry tree fruits (Arbutus unedo) prospected in an endemic area, where this species is spontaneously growing in Morocco. Results displayed significant differences among sampled trees based on the investigated physicobiochemical attributes. Thus, titratable acidity was in the range of 0.65 and 1.01 g malic acid/100g FW, whereas, total soluble solids varied from 14.83% to 18.53%. Citric acid was the major organic acid, followed by malic acid, where the average concentrations ranged from 1.74 to 5.32 g/100 g and from 1.53 to 2.86 g/100 g, respectively. Results also showed that the strawberry tree fruits could be considered as interesting, high-value nutraceuticals, being a novel source of bioactive compounds for dietary supplements or functional foods. Indeed, total phenols ranged from 25.37 to 39.06 mg GAE/g DW, while total flavonoid content varied between 3.30 and 7.07 mg GAE/g DW. Seventeen phenolic compounds were identified by HPLC, of which gallocatechol and catechin were the most abundant, of which the highest level was reported in “TAH” (65.31 mg/100 g DW). Given the current day, the biochemical composition of the strawberry tree fruits could be useful to improve future pharmacological and cosmetic usages. In addition, the results found in this study may be helpful for nutritionists, as well as berry growers and breeders, who can promote the cultivation of species and new cultivars with higher phenolic content and antioxidant activity. The high variability in biochemical composition observed among genotypes could be attributed to genetic factors. Therefore, it will be important to study and identify the genes responsible for the biochemical properties in order to understand the pattern of variation in the biochemical composition of strawberry tree genotypes. The present work provides important data for the food and pharmaceutical industries to consider this fruit as an exotic (or unusual) source of bioactive compounds, colors, and flavors.