2. Natural Macrocyclic Meroterpenoids
Among natural macrocyclic meroterpenoids, isoprenylated flavonoids and phenols [
6,
13], isoprenoid lipids [
14], prenylated amino acids [
15], and isoprenylpolyketide alkaloids [
5] have been most extensively studied. All of these macrocyclic meroterpenoids are derived directly from natural raw materials. Today, there are a number of methods that allow the isolation and characterization of natural meroterpenoids, among them: chromatographic technique, solvent extraction, membrane separation, distillation techniques, supercritical fluid fractionation, and others. All of these methods have both advantages and disadvantages. Thus, chromatographic techniques can provide high purity of key substances; at the same time, this is a laborious method that requires sophisticated equipment and entails low yields of target products. Extraction techniques are the easiest to implement but are associated with environmental pollution with organic solvents and also involve expensive recovery operations. Membrane separation is a highly selective method for meroterpenoids disengagement and is less labor-intensive consuming than others. However, the liquid–liquid interface is unstable, which causes a short membrane life. Distillation techniques are suitable for preparation of high purification and enriched fractions; however, these methods involve risk of implosion. The supercritical fluid fractionation is green chemistry, highly selective, and provides a high yield of target products. However, this is the most expensive method, requiring special operating system security, a large number of configurable factors, and the addition of organic modifiers [
16]. The formation of macrocyclic secondary metabolites during biosynthesis is a thermodynamically beneficial process since a whole series of final biochemical metabolic products can be obtained from one macrocyclic metabolite by transannular cyclization of the macrocyclic structure [
17]. Thus, Zhang et al. [
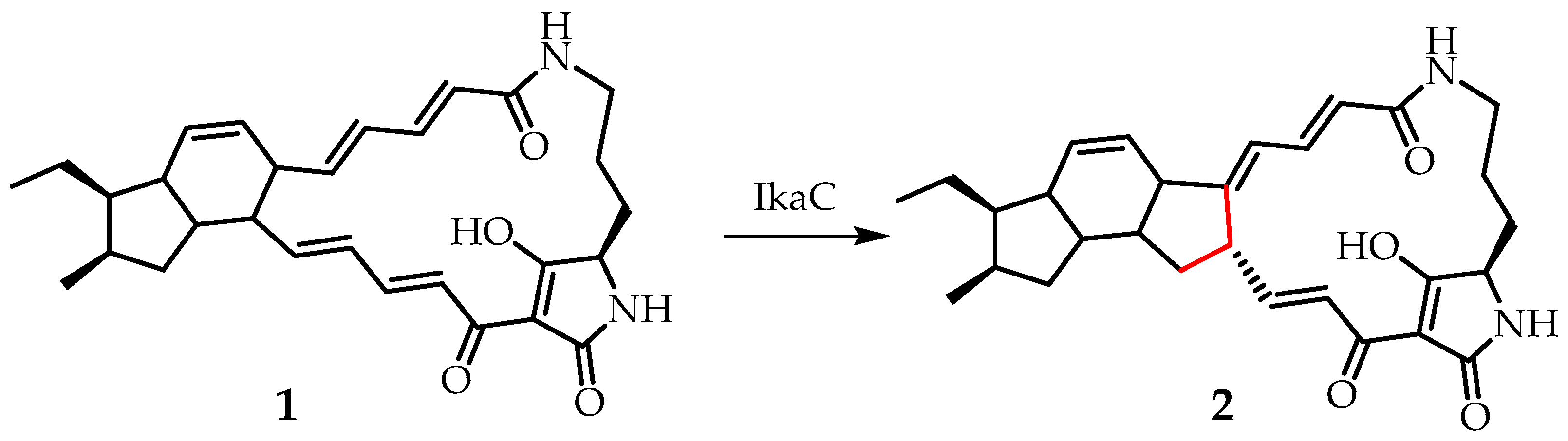
18] showed that alcohol dehydrogenase (IkaC) catalyzes an unusual reaction (
Figure 1) of reductive cyclization of compound
1 to ikarugamycin (
2) with the formation of an inner five-membered ring [
18]. Ikarugamycin (
2) is isolated from strains of
Streptomyes bacteria. In addition to strong antiprotozoal, antiulcer, antibacterial, antiviral, cytotoxic and apoptosis-inducing activities, it has also been reported that
2 inhibits the penetration of oxidized low-density lipoproteins into macrophage cells, thereby preventing the formation of atherosclerotic plaques, and clathrin-dependent endocytocytosis, which opens up the possibility of using
2 as a therapeutic agent [
18].
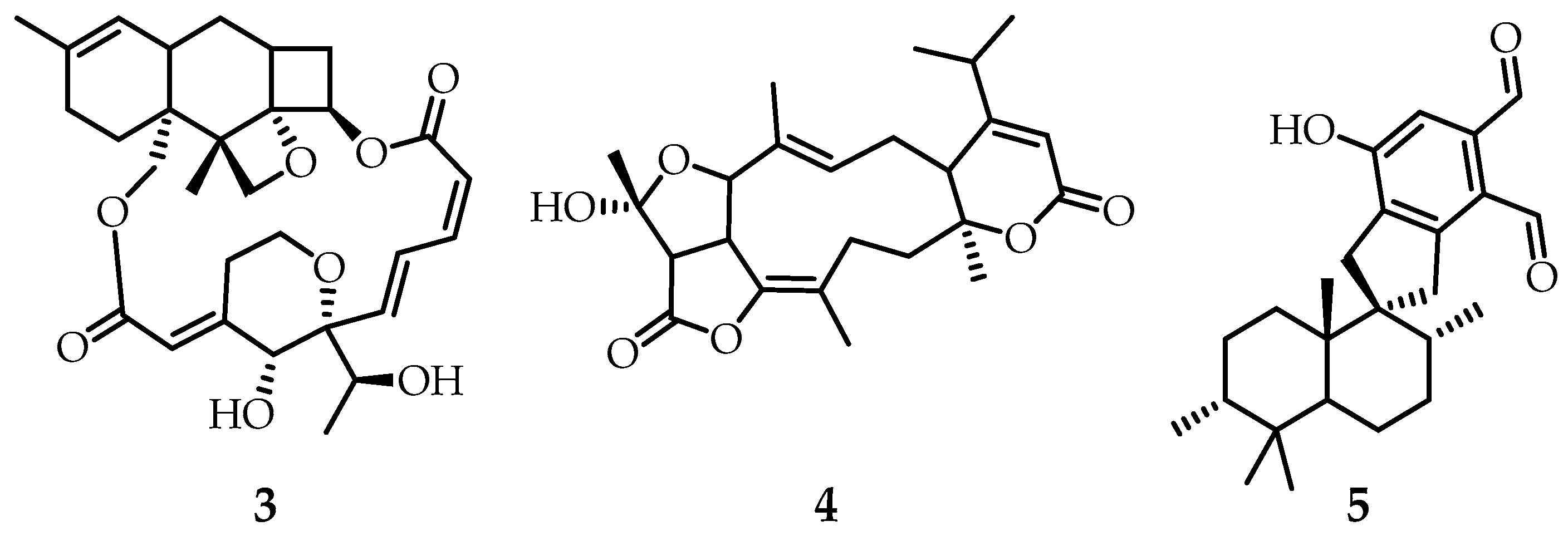
The study of the formation of secondary metabolites in fungi of the genus
Stachybotry, namely mycelial culture DSM 12880 (chemotype S), led to the discovery of three new meroterpenoids
3-
5 [
13], which contain a fragment of the chromene ring with side isoprenoid fragments. Taking into account the biosynthetic pathway, it can be assumed that stachibotrichromen C (
5) (
Figure 2) is the final product of the process and is formed through the intermediate stachibotrichromen A (
3) and stachibotrichromen B (
4) (
Figure 2). From the point of view of cytotoxicity, substances
3 and
4 exhibit a moderate cytotoxic effect on
HepG2 cells after 24 h of exposure, while
5 did not exhibit cytotoxicity over the concentration range studied (0.1–100 μM) [
13].
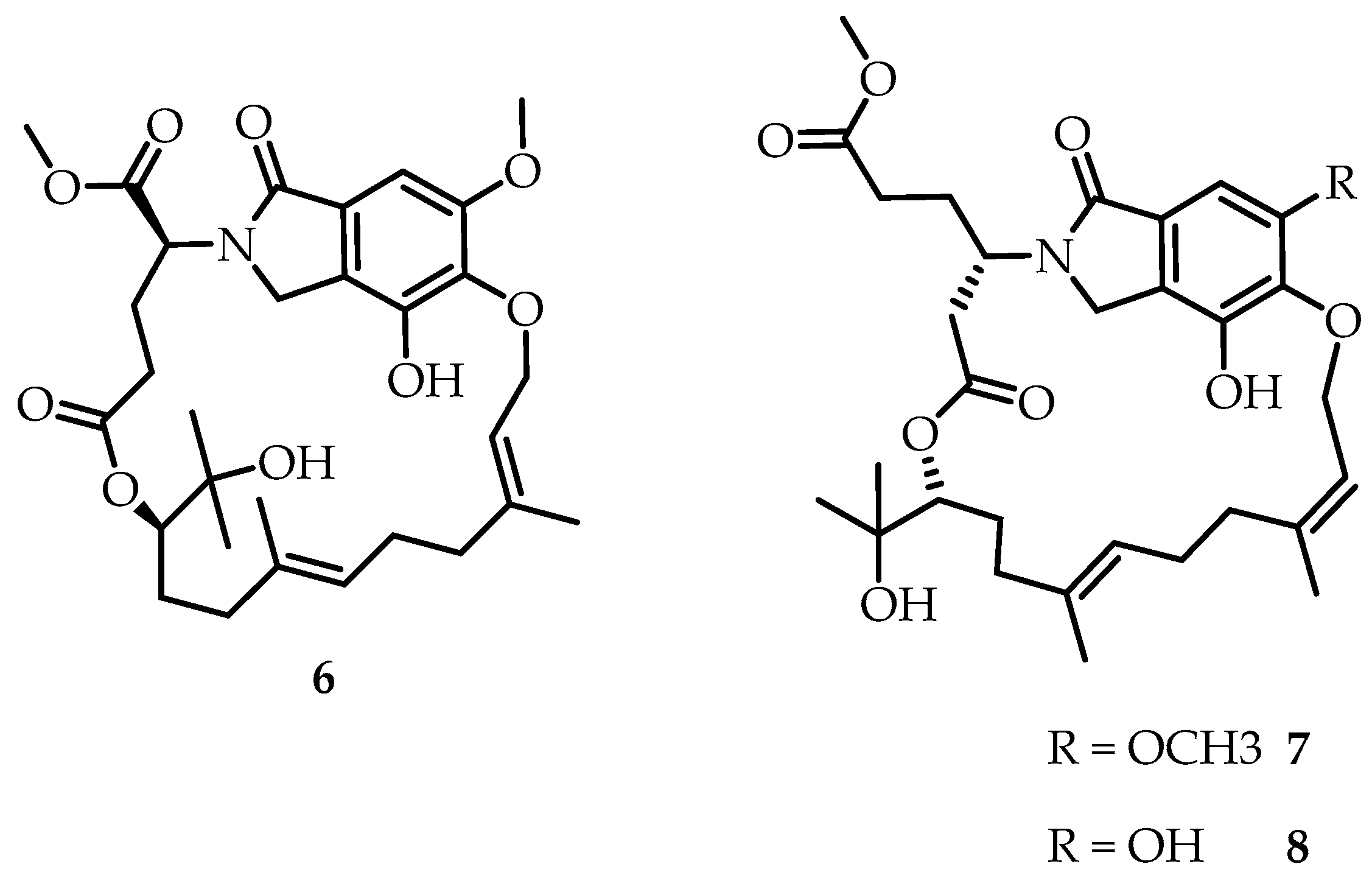
Li’s group [
19] discovered two new macrocyclic meraterpenoids derived from isoindolone from the culture of the endophytic fungus
Emericella nidulans HDN12-249. Emericellolides A-C (
6–
8) (
Figure 3) possess an unprecedented macrolide skeleton. The resulting compounds have potential antitumor activity.
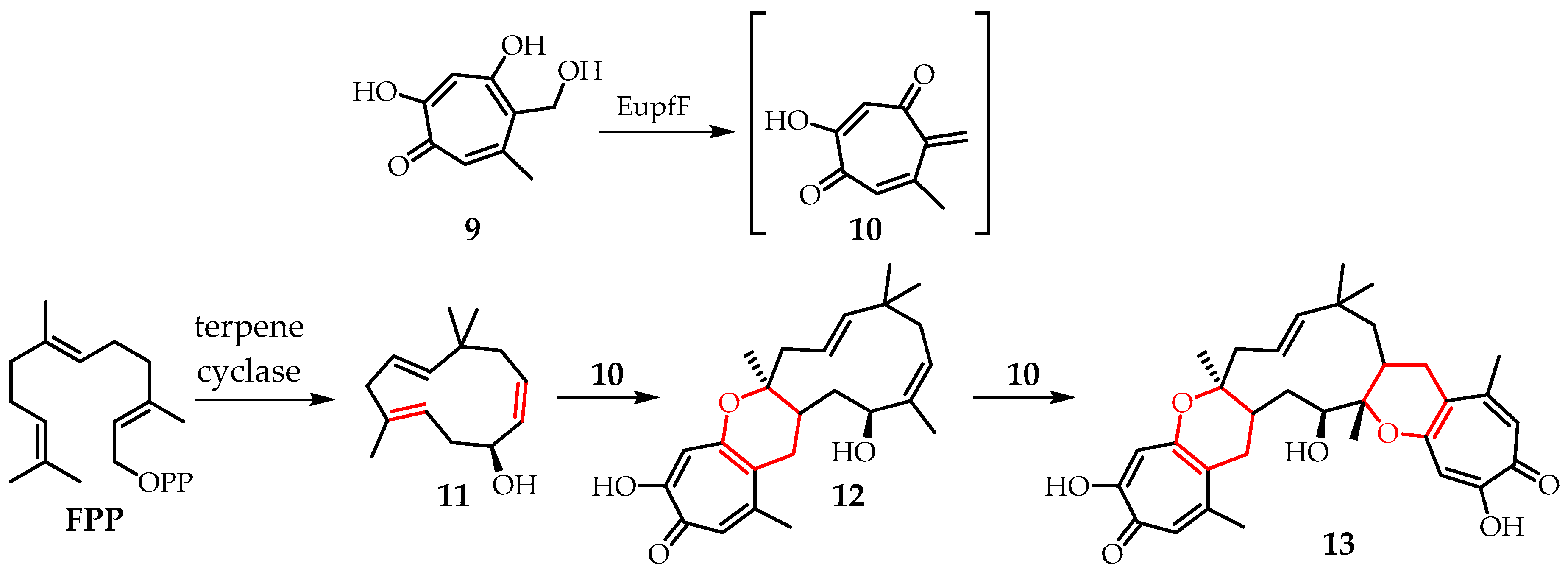
The Diels–Alder reaction is one of the most demanded synthetic transformations for the creation of complex natural compounds [
20]. However, only a few examples of its use in enzyme-mediated cascade reactions of macrocyclic meroterpenoids have been described [
21,
22]. Thus, in 2019, Houk et al. [
23] reported on the intermolecular hetero-Diels–Alder reaction in the biosynthesis of macrocyclic sesquiterpenes. It is noteworthy that neosetofomon B (
12), isolated as an optically pure compound from various mushrooms, can be obtained via one or two tandem hetero-Diels–Alder reactions. Neosetofomon B (
12) has a unique central 11-membered macrocycle consisting of a dihydropyran ring linked to tropolone
10 and has very strong antitumor activity at the nanomolar level. It has been shown (
Figure 4) that the enzyme
EupfF catalyzes the dehydration of hydroxymethyl-containing tropolone
9 to form the reactive o-quinone tropolone
10. The
EupfF enzyme is also able to stereoselectively control the subsequent intermolecular hetero-Diels–Alder reaction with (1E, 4E, 8Z)-humulenol (
11), obtained from farnesyl pyrophosphate (
FPP) by the action of the
terpene cyclase enzyme. The result is an enantiomerically pure neosetofomon B (
12) (
Figure 4), to which the addition of an excess of
10 leads to upenifeldine (
13), a neosetofomon containing two dihydropyran fragments.
A series of new isoprenylated phenols was isolated by Liu, An et al. [
24] from
Psidium guajava. The spatial structures of the meroterpenoids psiguajavadial A (
14) and B (
15) (
Figure 5) have been fully established using extensive spectroscopic methods and theoretical calculations. Flow cytometric analysis showed that the novel meroterpenoids
14 and
15 can induce apoptosis in
HCT116 cells. These data suggest that meroterpenoids from guava fruits can be used for the development of anticancer agents.
A unique macrocyclic isoprenylated phenol, napyradiomycin D1 (
16), containing a 14-membered cyclic ether ring, was isolated from
Streptomyces sp. strain CA-271078 [
25]. The authors report that meroterpenoid
16 (
Figure 5) exhibited significant inhibitory activity against methicillin-resistant
Staphylococcus aureus,
Mycobacterium tuberculosis and
HepG2.
Another unique natural source of macrocyclic meroterpenoids is archaeal lipids consisting of methylated isoprenoid chains, which are ether-linked to the glycerol-1-phosphate backbone [
26,
27]. The chemical structure and variety of archaeal lipids provide the necessary stability under extreme environmental conditions, as many archaea thrive in such conditions, including high or low temperatures, high salinity, and extreme acidic or alkaline pH values. Recently, higher homologues of the widely known macrocyclic isoprenoid lipid nuclei C
86, containing 0–6 cyclopentyl rings in (hyper) thermophilic archaea, have been identified [
28], which accounts for up to 21% of the total amount of tetraether lipids in cells. Tandem liquid chromatography-mass-spectrometry confirms that the additional carbon atoms in the C
87–88 homologues are located in the esterified chains. The structures identified include dialkyl and monoalkyl ("H-shaped") tetraesters containing C
40–42 or C
81–82 hydrocarbons, respectively, many of which are new compounds.
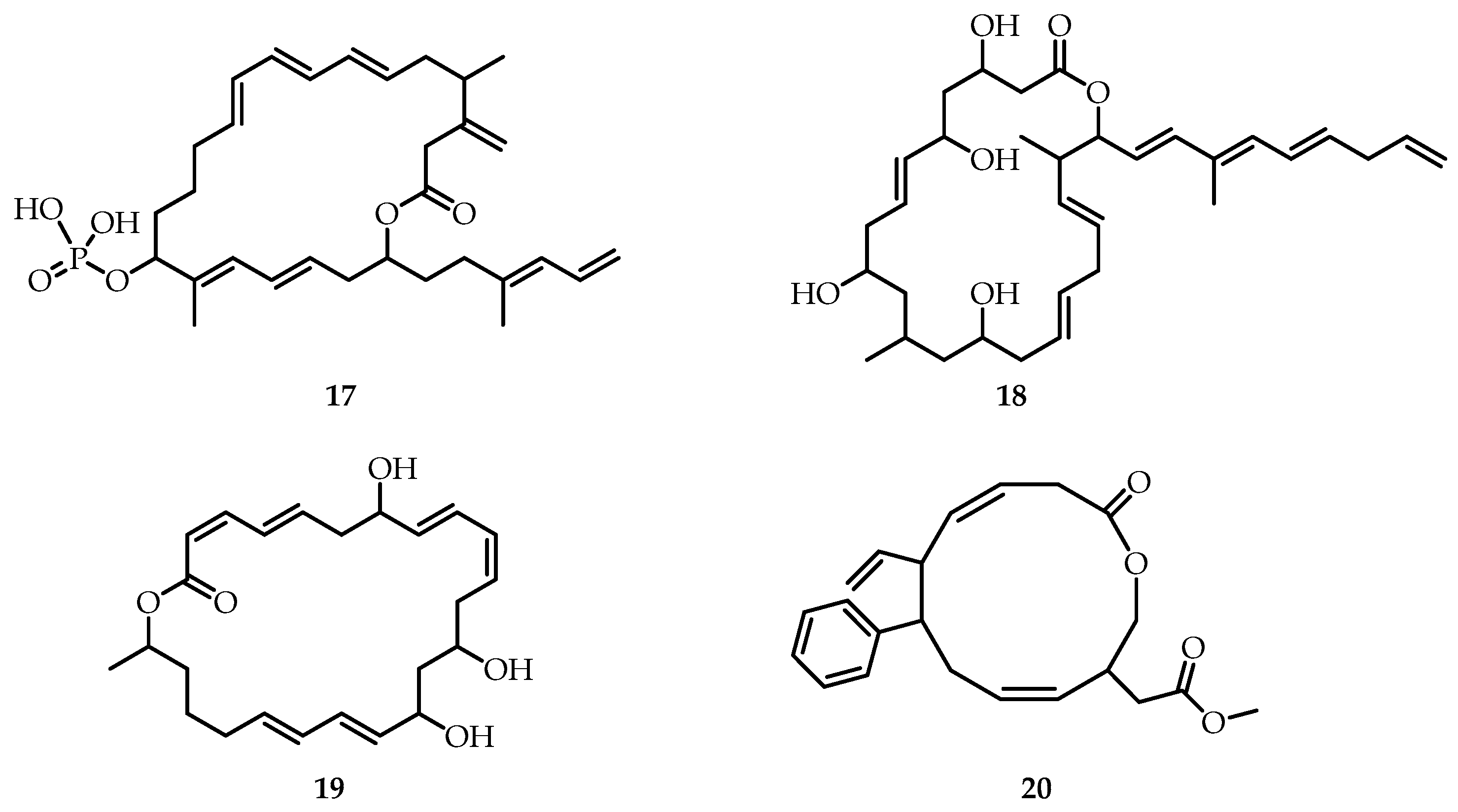
Recent studies have shown [
29] that macrocyclic meroterpenoid polyketides produced by
Bacillus and
Paenibacillus species can be used as antimicrobial agents to combat outbreaks of phytopathogenic diseases, as well as multidrug-resistant human pathogens. Among them are difficidin (
17), paenimacrolidin (
18), and macrolactin (
19) (
Figure 6). Difficidin (
17) is a highly unsaturated macrocyclic polyene, consisting of a 22-membered carbon skeleton with a phosphate group. Although difficidin is primarily known for its activity against human pathogens, it is also effective against plant diseases caused by
Erwinia amylovora and
Pectobacterium carotovorum, as well as
Xanthomonas oryzae. Macrolactins are a class of over 30 different 24-membered macrolactones with three diene moieties in the carbon backbone, isolated from various bacterial sources, including
Bacillus species such as
B. subtilis and
B. amyloliquefaciens. Recently, a new derivative of macrolactins, macrolactin A (
19), was isolated from the
Bacillus subtilis B5 bacterial strain. Bacillus macrolactins exhibit antimicrobial activity against gram-negative and gram-positive bacteria, including MRSA and VRE, as well as antiviral activity against Herpes simplex and HIV in lymphoblastic cells. Paenimacrolidin (
18) is a secondary metabolite of
Paenibacillus sp.
F6-B70. It exhibited mainly bacteriostatic activity and exhibited antimicrobial activity against the pair of MRSA strains tested and ampicillin resistant
Staphylococcus epidermidis.
Interestingly, macrocyclic polyketide (
20) (
Figure 6) showed [
30] significantly stronger antioxidant and anti-inflammatory properties compared to some commercially available antioxidant agents and is a candidate for preventing food spoilage and inflammatory diseases caused by oxidative stress.
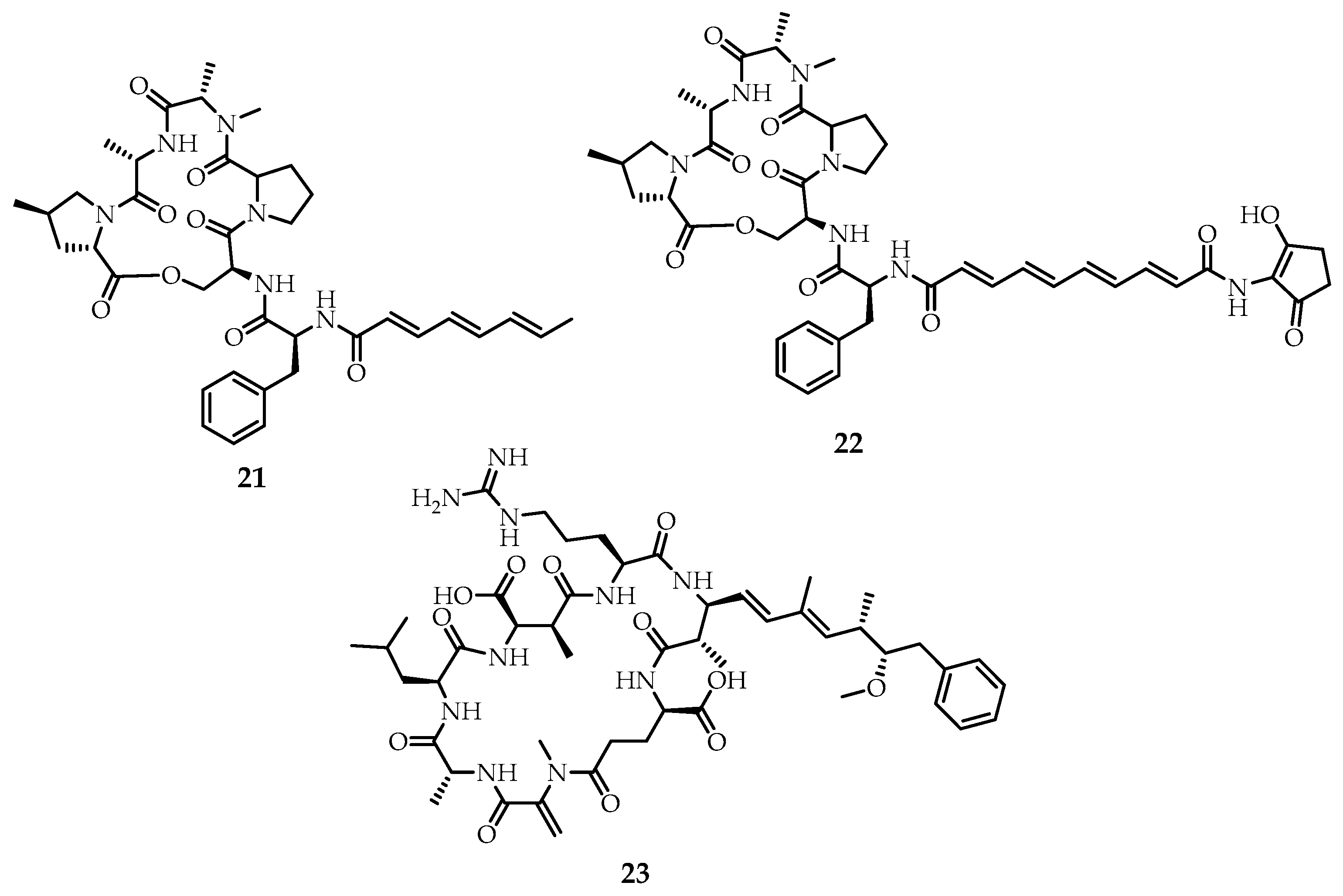
It should be noted that the majority of natural meroterpenoids obtained during enzyme-mediated cascade reactions of coupled biosynthesis, although they are macrocyclic compounds, do not exhibit "classical" supramolecular properties. They are important secondary metabolites in various biochemical processes and many of them have a sufficiently high biological activity, which makes them promising drug candidates [
1]. However, among the natural macrocyclic meroterpenoids, a class of compounds with the properties of supramolecular systems can be distinguished. These are the prenylated bacterial nonribosomal lipopeptides [
31], which represent the smallest group of isoprenoids of conjugated biosynthesis but have been actively studied in recent decades. The study of prenylated macrocyclic peptides as allosteric activators of chamber bacterial proteases made it possible to place them among potential antibiotics. The first representatives of this class of compounds are acyldepsipeptides
21–
23 (
Figure 7) [
15,
29,
32].
Since the structure and composition of nonribosomal lipopeptides are not limited or determined by the genetic code, they may contain nonproteinogenic (ornithine, homoserine, β-amino acids) and modified amino acids (hydroxylated, chlorinated, N-methylated or glycosylated) in the L- or D-configurations. The fatty acid moiety can be composed of a linear, cyclic, or branched terpene tail, the length of which can vary (typically C
6-C
18) and contain functional groups (e.g., hydroxyl or amino). A macrocyclic ring of nonribosomal prenylated lipopeptides is formed between the C-terminus of the peptide and the hydroxyl, phenolic, or amine functional groups that belong to either the amino acid side chain or the terpene moiety. The resulting macrolactones or macrolactams differ in ring size, which ranges from 4 to 16 amino acids [
15].
Many nonribosomal prenylated lipopeptides have amphiphilic properties, which make them useful as effective biosurfactants [
33]. They influence biofilm formation or degradation by changing the surface tension of the environment. Nonribosomal prenylated lipopeptides also have membranotropic properties. In addition, they have the ability to form supramolecular associates containing water-insoluble nutrients to facilitate the transport of the latter into the cells [
34].
Thus, the number of detected representatives of natural macrocyclic meroterpenoids is increasing each year. This is primarily due to the establishment of biochemical mechanisms of the formation of meroterpenoids in the course of metabolism. Such identified biosynthetic pathways open up new perspectives in the discovery of previously unknown representatives of macrocyclic meroterpenoids. Extension of a number of macrocyclic meroterpenoids will make it possible to finally consolidate this class of compounds in a number of macromolecules. Initially nontoxic, biodegradable, available meroterpenoids with a macrocyclic cavity can replace or be an alternative to classical macrocyclic platforms, which usually exhibit high toxicity. The disadvantages of using natural macrocyclic meroterpenoids include relatively low yields of target products during their disengagement from biomass. This imposes serious restrictions on the use of natural meroterpenoids. However, the development of semisynthetic production methods resolves these restrictions.
3. Synthetic Macrocyclic Meroterpenoids
Synthetic macrocyclic meroterpenoids are obtained by combining “classic” macrocyclic platforms, such as cyclodextins or meta- and paracyclophanes, with various kinds of terpenoid fragments or through macrocyclization of several terpene derivatives [
4].
In this regard the most studied synthon for macrocyclization is the triterpene steroid framework. The steroid core is one of the largest and most conformationally rigid readily available terpene molecules with axial or equatorial substitution patterns available, occurring in a homochiral form. Moreover, because of the importance of steroids in biochemistry and medicine, steroids have been studied in great detail [
35]. Consideration of macrocyclic meroterpenoids should start with cholaphanes and cyclocholates. Cholaphanes are bile acid macrocycles composed of two to four steroid subunits linked together by different groups of atoms as spacers. Macrocyclic cholaphanes, consisting of two steroid fragments, in contrast to their acyclic analogs, tend to form host–guest complexes [
35]. Cyclocholates are macrocyclic polyesters formed by macrocyclization of cholic acid derivatives [
36].
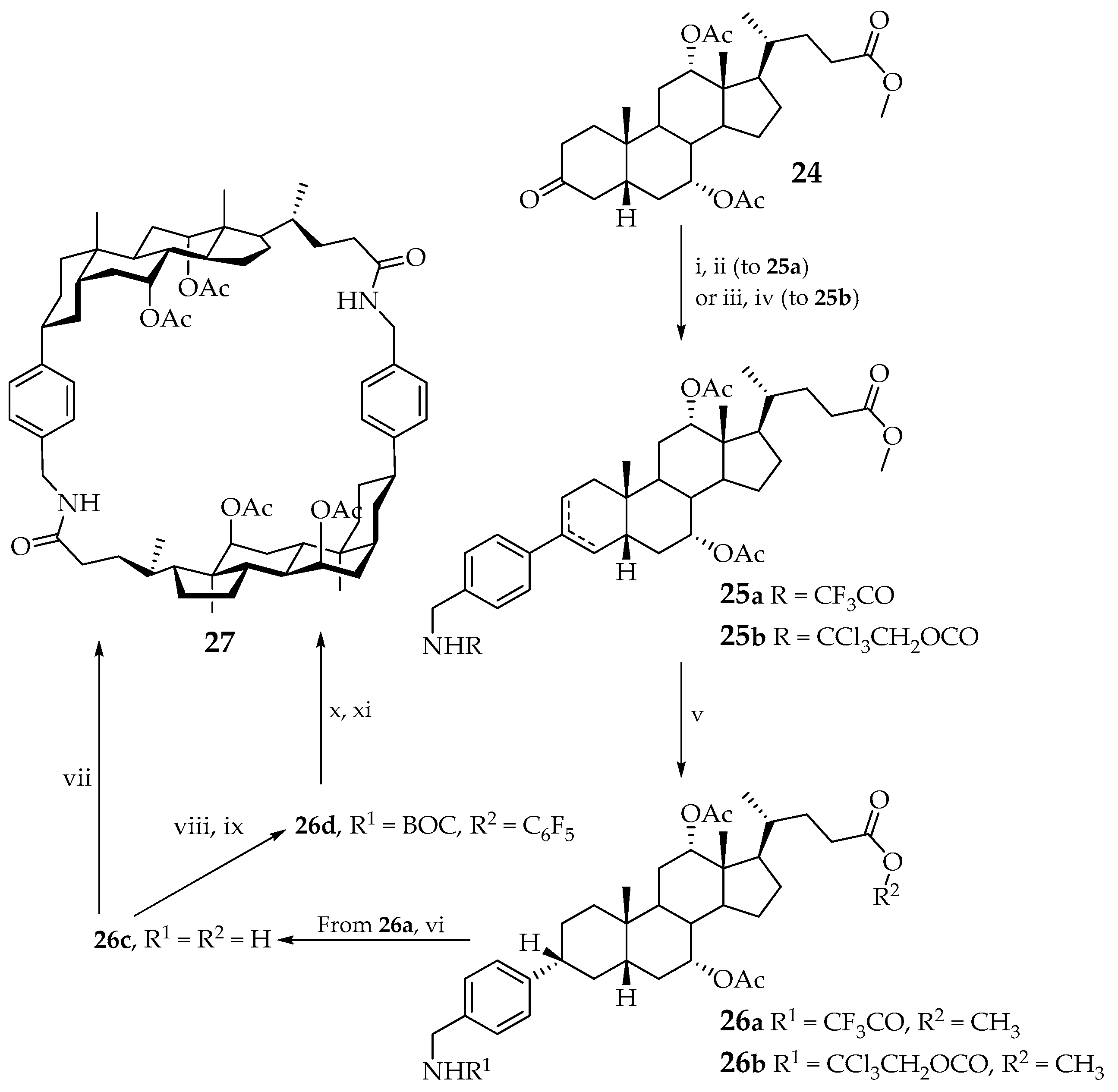
Davis was the first to report the synthesis of a cyclic dimer of cholic acid in 1989 [
37]. As part of this work, macrocycle
27, containing two cholic acid fragments linked by a rigid para-substituted benzyl bridge, was synthesized. The introduction of a conformationally rigid bridge made it possible to avoid excessive flexibility of the skeleton (
Scheme 1) [
37].
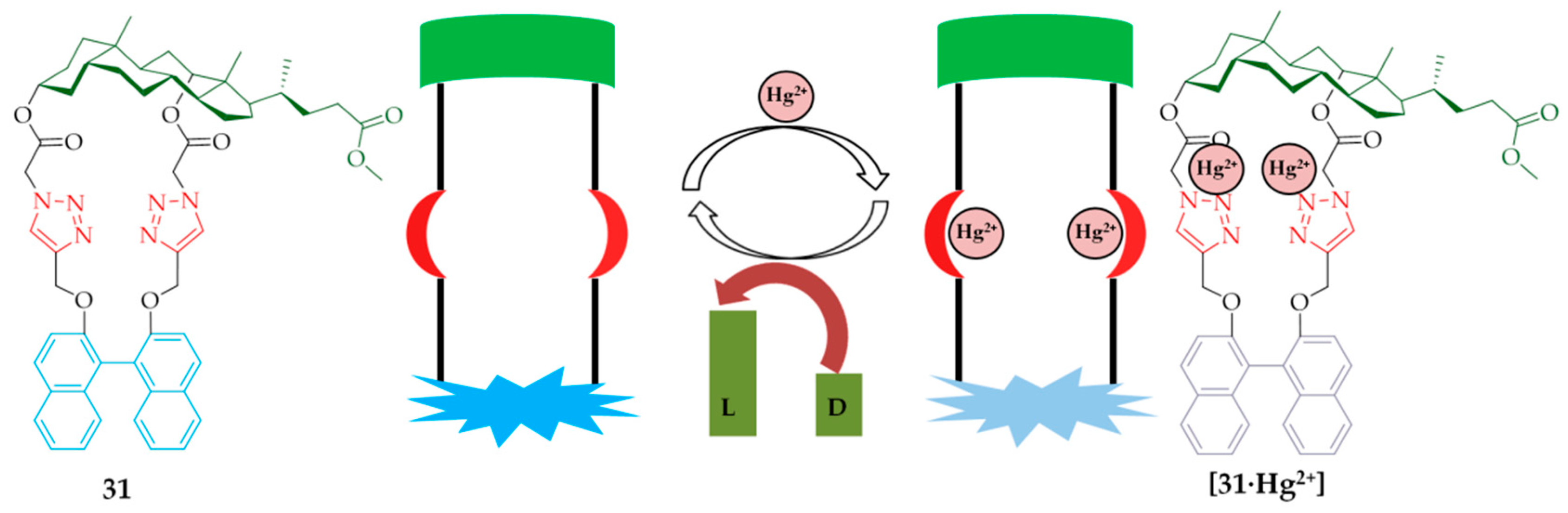
Currently, cholaphanes are an actively studied class of macrocycles. Recently a group led by Ju developed [
38] and synthesized a new macrocyclic sensor,
31, (
Scheme 2) consisting of cholesterol, binaphthalene and 1,2,3-triazole fragments. Macrocyclization was carried out by azizide-alkyne cycloaddition between azide
28 and alkyne derivative
30, obtained by alkylation of
29 with propargyl bromide. Fragments of the triterpenoid play the role of a chiral framework, the binaphthyl fragment acts as a fluorophore fragment, and the 1,2,3-triazole fragment plays the role of a coordination center [
38].
The authors have shown [
38] that macrocycle
31 can serve as an “on-off” fluorescent molecular sensor for the Hg
2+ ion due to the 1,2,3-triazole fragment introduced as a result of the CuAAC click reaction. This complex [
31∙Hg
2+] can be used in the cascade recognition of amino acids with high enantioselectivity (
Figure 8).
The group led by Pandey reported [
39] the efficient synthesis of a bis-1,2,3-triazolium cholaphane macrocycle based on bile acid via the click reaction (
Scheme 3) and the use of the product,
38, to recognize the chloride ion. The propargyl derivative of methyldeoxycholate,
33, was synthesized by the reaction of deoxycholic acid
32 with propargyl bromide in the presence of sodium hydride in THF followed by the addition of MeOH-H
2SO
4. Monobromoacetyl derivative
34 was prepared by reacting propargyl ether
33 with bromoacetyl bromide in the presence of K
2CO
3 in trichloromethane. Compound
34 upon treatment with sodium azide in DMF gives precursor
35, which in the presence of CuSO
4 (10 mol %) and sodium ascorbate (20 mol %) gives cholaphane
36 based on triterpene bile acid in 50% yield. Cholaphane
36 was methylated at the nitrogen of the triazole moieties with methyl iodide to give compound
37. The PF
6-salt
38 was obtained by ion exchange of the iodide salt of
37 with NH
4PF
6 in MeOH [
39].
The receptor properties of compound
38 were studied by
1H NMR spectroscopy by adding tetrabutylammonium salts to a solution of receptor
38 in CDCl
3. Upon addition of aliquots of the solution of Bu
4NX (X = F
−, Cl
−, Br
−, I
−, CH
3COO
−, H
2PO
4−) to a solution of
38, significant downfield shifts were observed for the triazolium groups suggesting the interaction of the anion with triazolium by forming C
+-H-X−hydrogen bonds. In addition, noticeable downfield shifts were also observed for one of the methylene protons of both acetyl groups, which indicates their participation in forming hydrogen bonds with anions [
39]. The stoichiometry in all studied cases was 1:1. Receptor
38 was found to be highly selective for chloride with a binding constant K
a = 3700 M
−1. The strength of the interaction decreased in the order: Cl
− > HSO
4− > H
2PO
4− > F
− > Br
− > CH
3COO
− > I
−. The high selectivity of this receptor with respect to the chloride ion was explained by the authors as being due to the corresponding size of the receptor cavity, which provides the most effective binding with the chloride anion. Consequently, this system may be an attractive carrier for chloride ions. Carriers of chloride ions have direct medical use in the treatment of cystic fibrosis and other diseases caused by insufficient transport of chloride ions across biological membranes [
40].
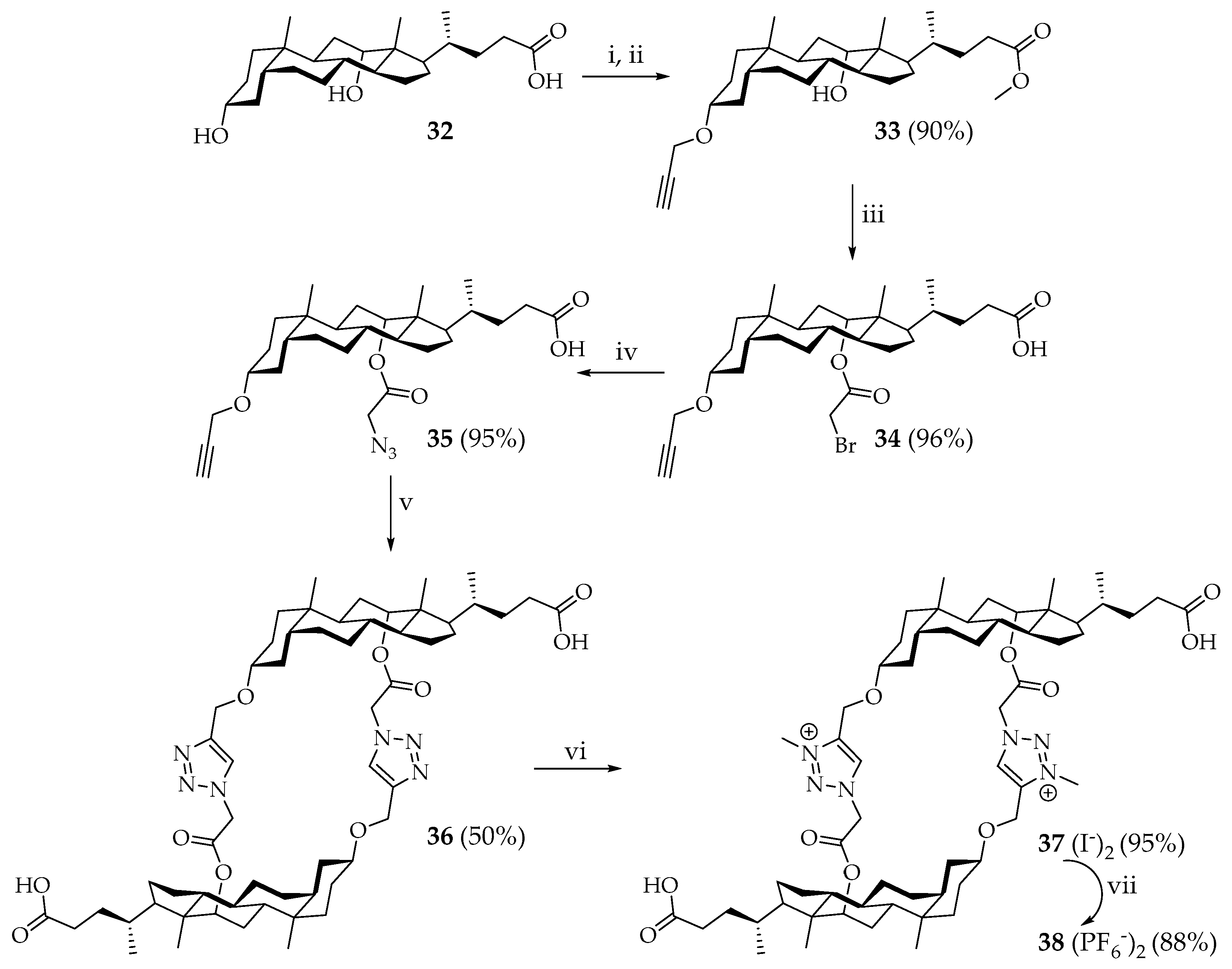
A group of Polish scientists [
41] reported the preparation of new dimeric cholaphanes
44 and
45 with disulfide spacers (
Scheme 4). The synthetic route consisted in a series of successive reactions of the conversion of cholic acid ester
39 into alcohol
40, followed by the substitution of two alcohol groups by the Appel reaction. Then the precursor
41 was converted to diisocyanate
42, which was reduced to the corresponding dithiol
43. Then, macrocycles
44 and
45 were obtained by oxidative macrocyclization to give
43. Disulfide spacers link two identical steroid subunits head-to-head, to give cis-dimer
45, or head-to-tail to give trans-dimer
44. Another cyclic dimer,
48, was also obtained (
Scheme 5), containing both disulfide and sulfide spacers in its structure. Cholaphanes
44 and
45 can be potentially useful for molecular or ionic recognition [
41].
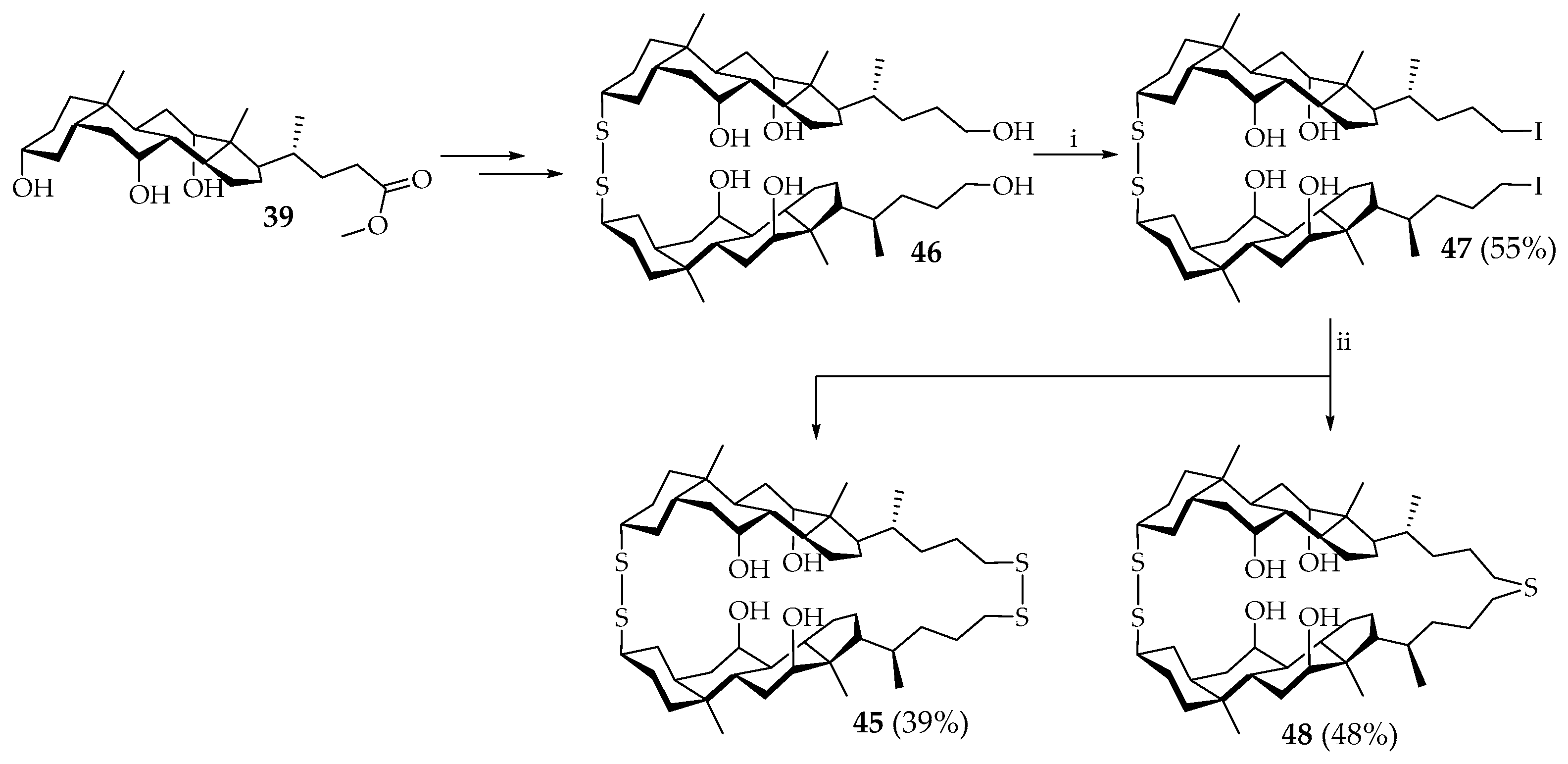
An attempt was also made to selectively obtain isomer
45 directly from the acyclic disulfide dimer. The “cis-dimer”
45 was finally obtained from acyclic disulfide
46 and, during this reaction, cholaphane
48 with a sulfide linker was also formed (
Scheme 5). The latter was also selectively formed by using a large excess of NaSH∙2H
2O in the reaction with diiodide
47 [
41].
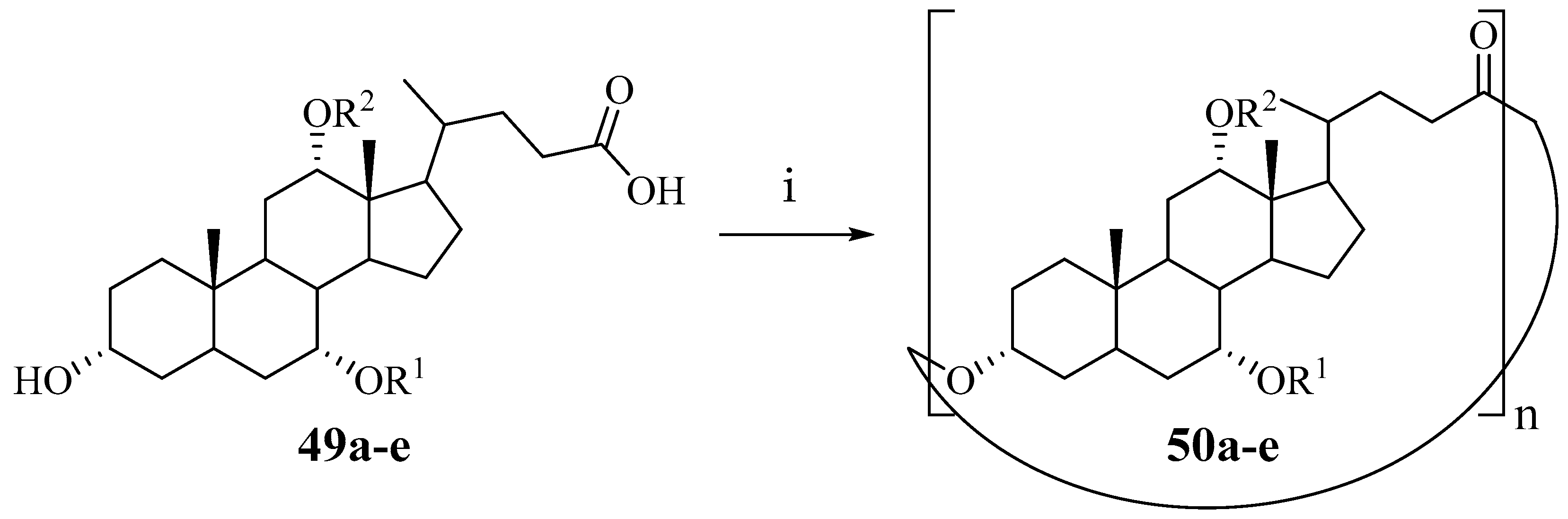
Cyclocholates-macrocyclic polyesters of cholic acid were first described by Bonar-Law [
36]. Cyclocholates
50a–e were obtained by a Yamaguchi macrolactonization from monomeric hydroxy acids
49a–e using 2,6-dichlorobenzoylchloride as coupling reagent in a one-pot synthesis (
Scheme 6). The resulting mixture of cyclic compounds was separated by flash chromatography. The individual yields of target cyclooligomers are shown in
Table 1 [
36].
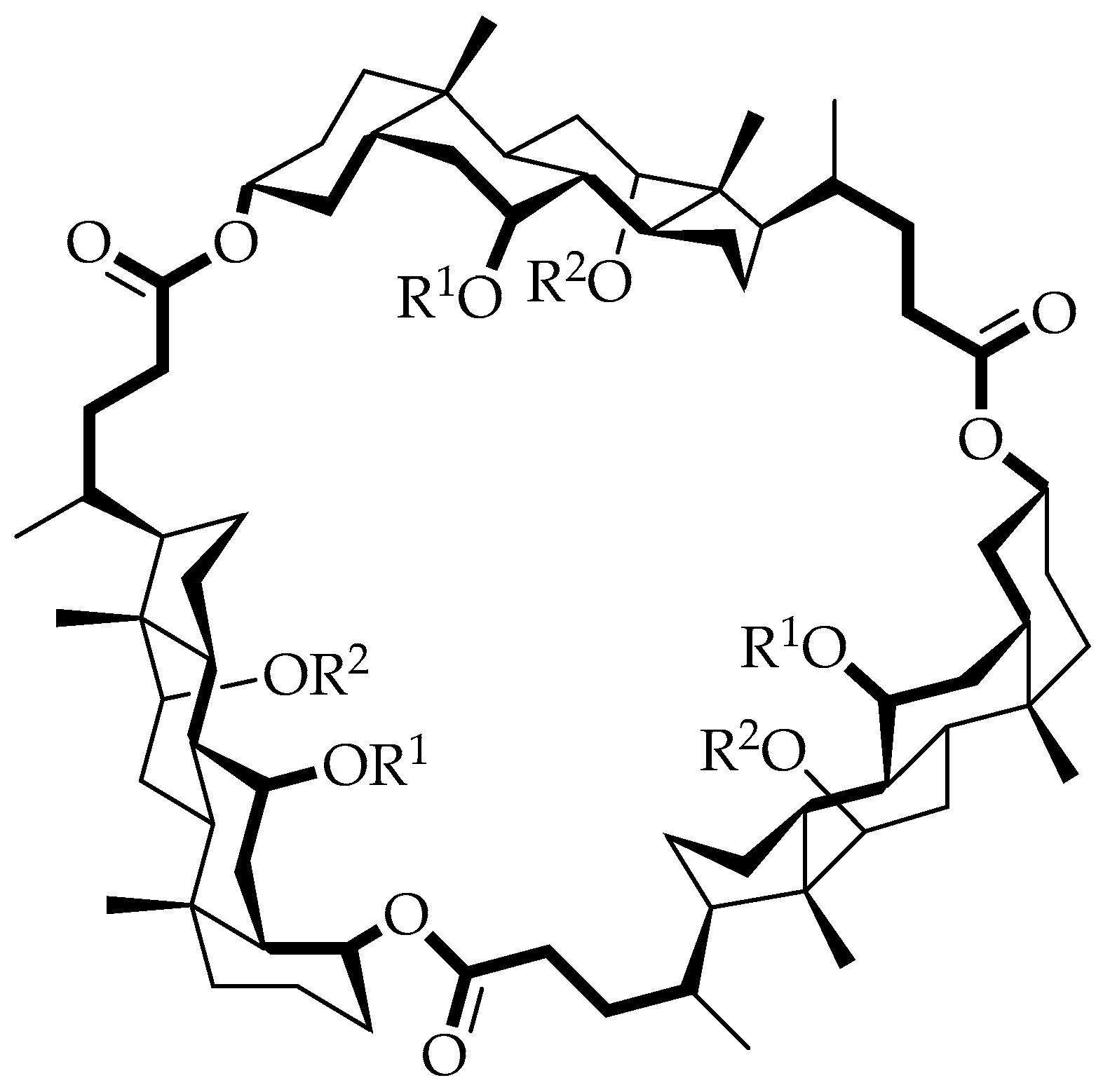
The dependence of the macrocycle size on the initial monomer concentration was established using
19F NMR spectroscopy for trifluoroacetate-protected cyclocholates, since the fluorine resonance turned out to be a sensitive indicator of the chemical environment. The relatively high yields of cyclotrimers (
Table 1) make this particular series of cyclooligomers (
Figure 9) available in gram quantities without the need for high dilution [
36].
It was also found that the size of the macrocycle formed depended on the nature of the substituents on the axial hydroxyl groups. It should be noted that polyester chains facilitate the formation of not only cyclotrimers but also larger macrocycles. The authors note that cyclocholates, in this work, are considered as semirigid macrocyclic lactones, built from large units carrying convergent functionality, and they represent a universal type of receptor architecture. However, quantum-chemical calculations [
42] using a nonfunctionalized cyclotrimer
50a show that the current generation of cyclocholates is too flexible to be used as macrocyclic receptors in molecular recognition [
37].
Gao and Dias [
43] reported the synthesis of seven macrocyclic bile acid oligomers (
Scheme 7). In this work, as in the previous one, the Yamaguchi method was used and the yields of
52a–f varied from 3 to 45% (
Table 2). Cyclomonomer
52g and macrocyclic dimer
52f were obtained only as a result of the cyclization reaction of 7-deoxycholic acid. Under Yamaguchi reaction conditions, cholic acid derivatives give mainly macrocyclic trimeric products
52a,b,e, while 24-norcholic acid derivatives give mainly macrocyclic tetrameric products
52c [
43].
It should be noted that there are far fewer papers devoted to cyclocholates than there are related to cholaphanes. This lack of interest in cyclocholates may be due to the absence of potential coordination centers in their structure, in contrast to the related cholaphanes, which contain functional groups of different nature that can act as coordination centers.
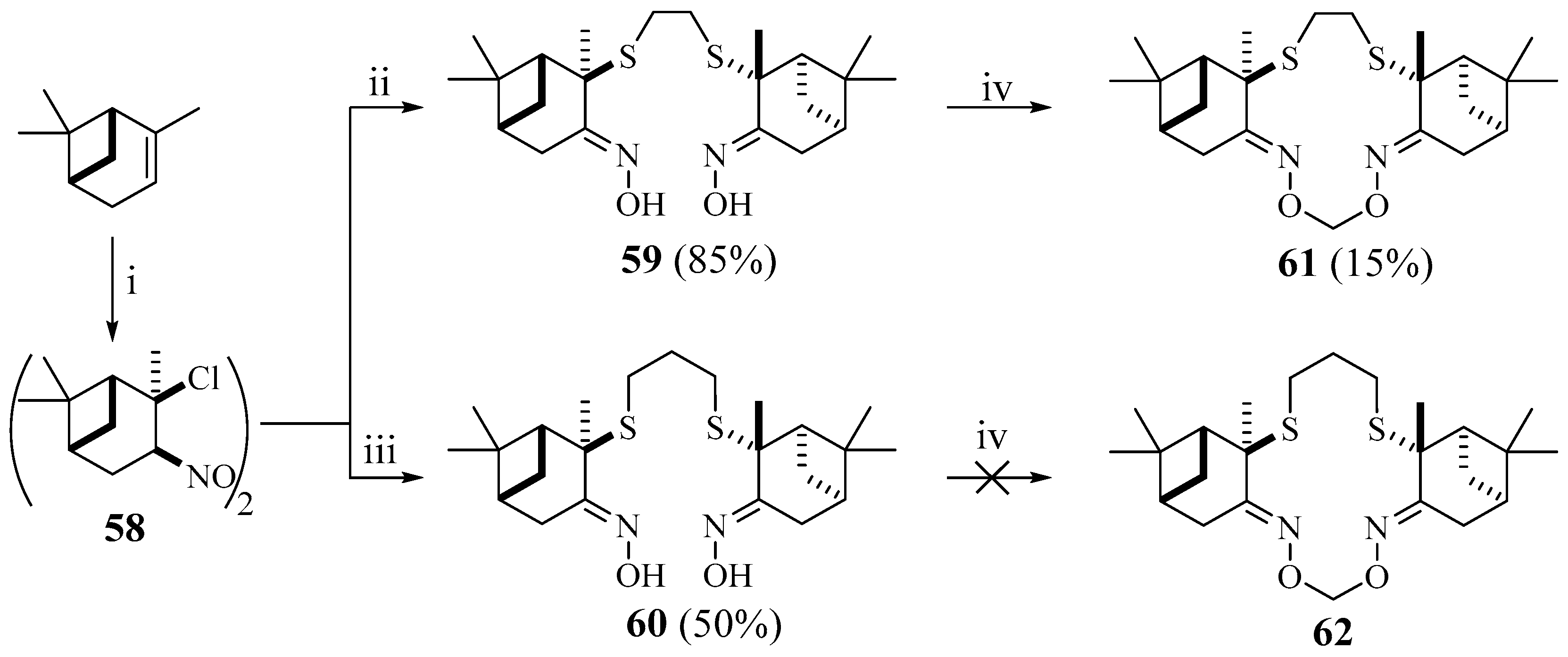
Syntheses of macrocyclic dimers containing terpene fragments other than cholesterol are also poorly presented in the literature. In one example, a group of scientists from Novosibirsk [
44] synthesized macrocyclic meroterpenoids
56,
57 and
61 by cyclization. The authors proposed a simple and effective method for the synthesis of bis-α-sulfanyloximes
54,
55 and
59,
60 from unsaturated monoterpene hydrocarbons, such as (+)-3-carene and (−)-α-pinene, previously converted into the corresponding nitrosochlorides
53,
58 (
Scheme 8 and
Scheme 9).
The reaction of dimeric nitrosochloride
53 or
58 with a solution of the dithiolate in anhydrous methanol under an inert gas atmosphere led to the corresponding bis-α sulfanyloximes
54,
55,
59 and
60 (
Scheme 8 and
Scheme 9). The maximum yields of 1,2-dithiol derivatives
54 and
59 were achieved using Na
+ as a counter ion, while for 1,3-dithiol derivatives
55 and
60 K
+ was more successful. The authors explain this by the template effect of the alkali metal cation, which is similar to that observed during the formation of crown ethers. The cyclization of bis-α-sulfanyloximes
54,
55,
59, and
60 had been carried out by bonding hydroxyl groups with dichloromethane under conditions of phase transfer catalysis. It is likely that macrocyclization follows the generally accepted mechanism of ether formation under conditions of phase transfer catalysis. In the case of compound
60, the target macrocycle
62 was not obtained; the reaction resulted in the formation of a very complex mixture of oligomerization products [
44].
The group of Kataev [
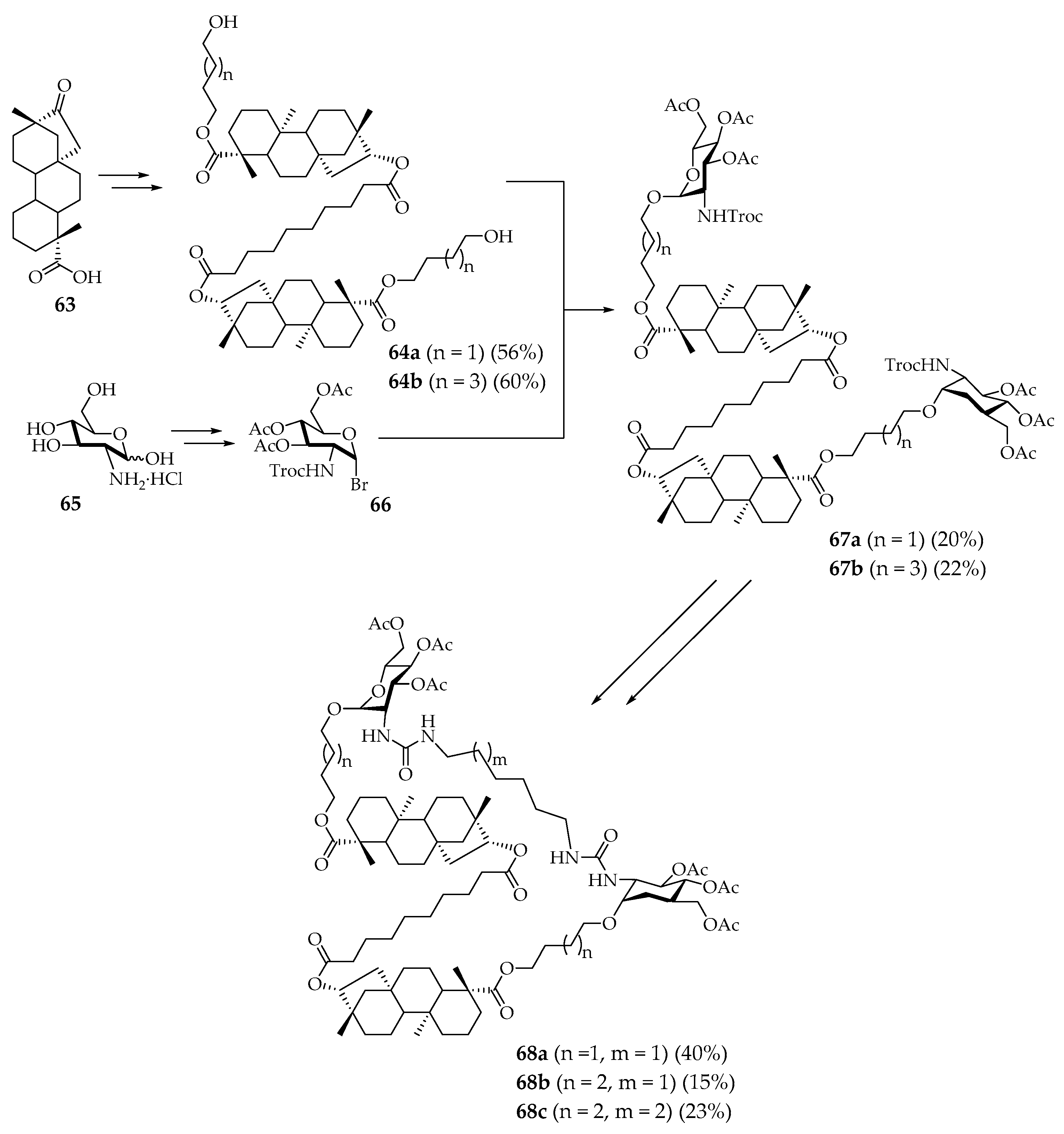
45] synthesized a number of macrocyclic glycoterpenoids. As a basis for creating macrocycles, diterpenoid isosteviol
63 and commercially available glucosamine hydrochloride
65 were used and the entire synthesis can be divided into four stages (
Scheme 10). In the first stage, two isosteviol molecules were combined using a linker, and the carboxyl groups of isosteviol fragments were functionalized with hydroxypolymethylene chains, giving precursors
64a–
b. Reactive carbohydrate
66 was obtained in a series of sequential reactions from glucosamine
65 by introducing three acetate fragments, one trichloroethyl chloroformate protection of the amino group, and replacing the glycoside hydroxyl with bromine. Products
64a–b and
66 were then combined to form diglycosides
67a–
b. Trichloroethylchloroformate protecting groups were removed from the amines before the target macrocyclic glycoterpenoids
68a–
c were obtained by macrocyclization of
67a–
b with alkydiisocyanates [
45].
Macrocycles
68a–c were tested for their ability to inhibit the growth of
M. tuberculosis H37Rv cells in vitro and all showed moderate tuberculostatic activity. Their minimum inhibitory concentrations (MIC) were in the range 6.4–17.4 mM, while the MIC for the antitubercular drug isoniazid (the control compound in the experiment) was 0.7 mM [
45].
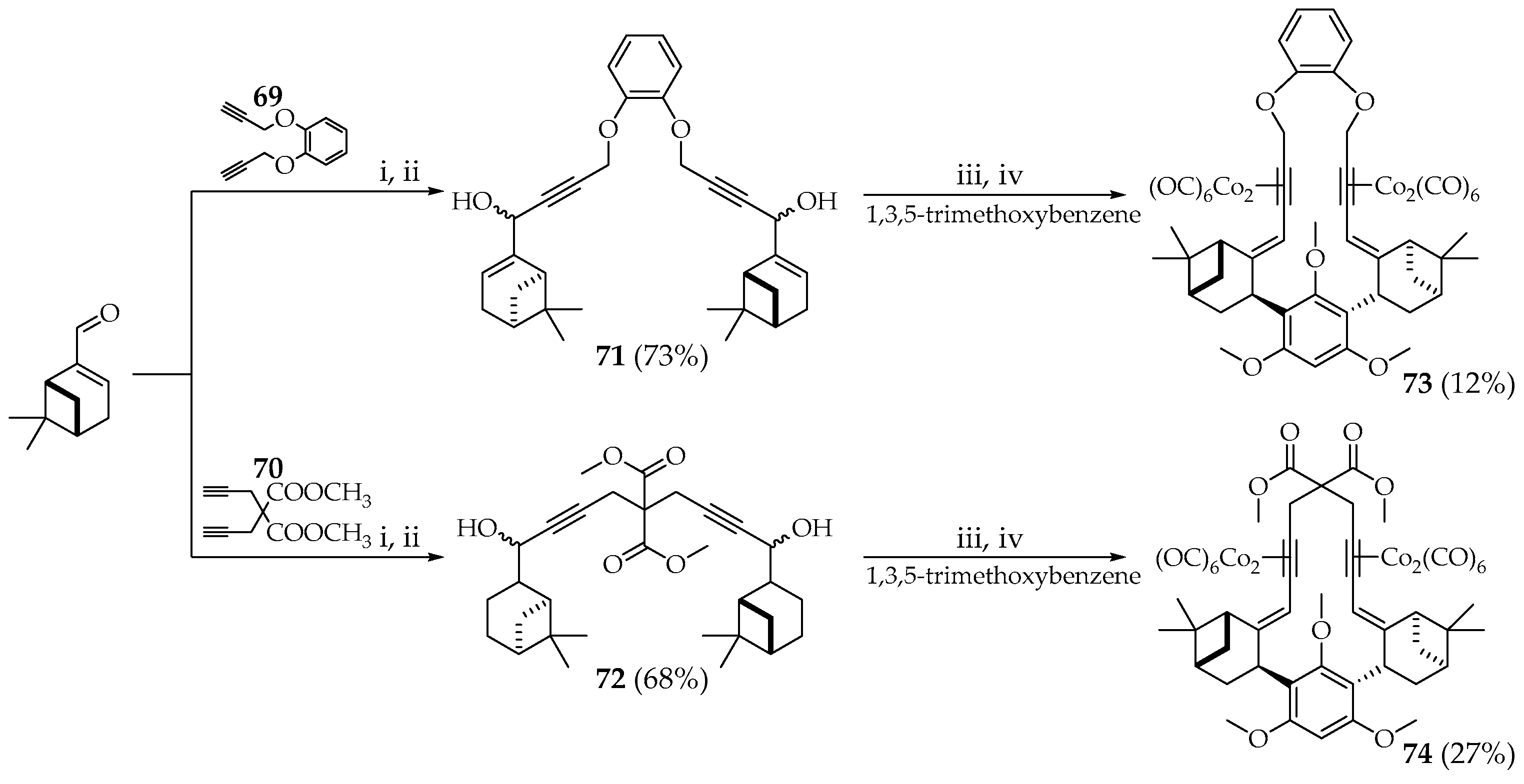
Spanish scientists synthesized [
46] polymetallic macrocyclic hybrids
73,
74, and
77 based on terpenoids. The two terpenoid fragments were linked together by a diyne linker, resulting in the formation of structures that involved a double Nicholas reaction to produce macrocyclic meroterpenoids
73 and
74. The role of the initial diyne linker was performed by 1,2-(bipropynyloxy)benzene
69 and 2,2-di(propynyl)malonate
70. Condensation of lithium salts of compounds
69 and
70 with (1R)-(−)-myrtenal gave diols
71 and
72 in the form of diastereomeric mixtures in high yields (
Scheme 11). The macrocycle was formed through a double Nicholas reaction with 1,3,5-trimethoxybenzene. Macrocyclization of one equivalent of compound
71 or
72 with one equivalent of 1,3,5-trimethoxybenzene was carried out in the presence of BF
3∙OEt
2. Macrocyclic compounds
73 and
74 were obtained as individual diastereoisomers (
Scheme 11) [
46].
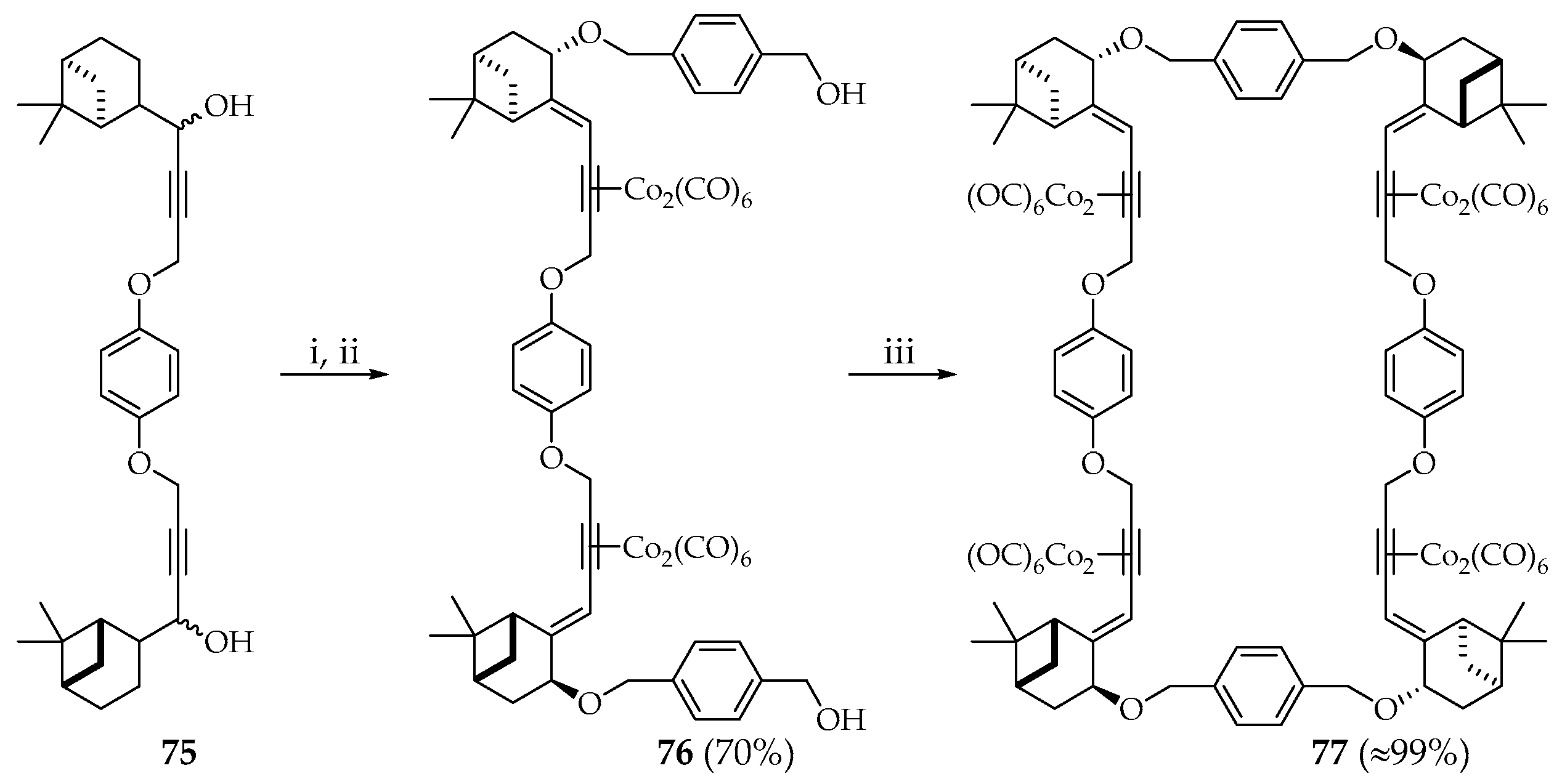
Macrocycle
77 was also obtained from precursor
75 in two stages. Compound
75 was stereoselectively converted to the Co
2(CO)
6-bis-alkyne complex
76 followed by cyclization with 1,4-benzenedimethanol in the presence of BF
3∙OEt
2 to obtain macrocycle
77 in almost quantitative yield (
Scheme 12).
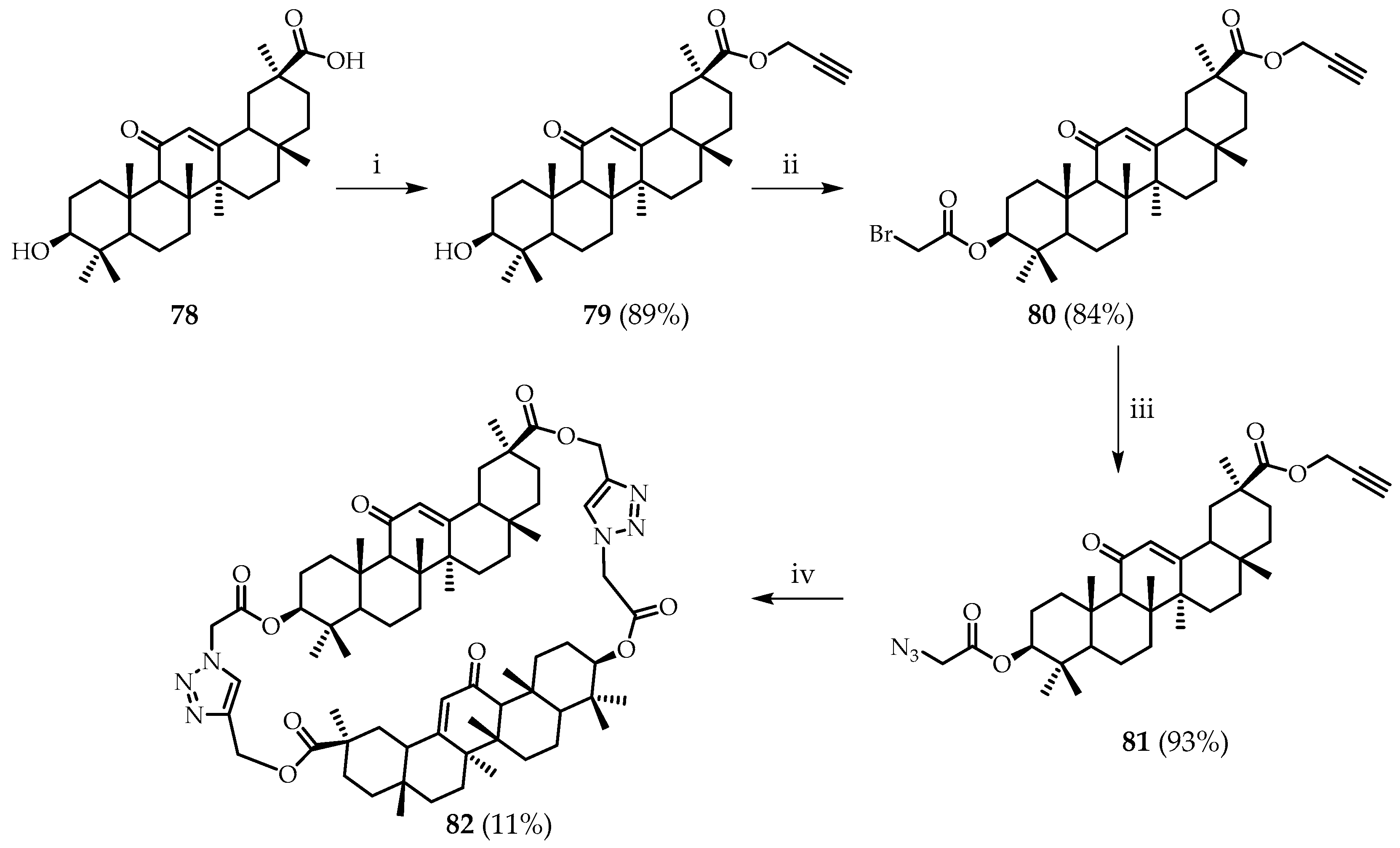
Ju et al. [
47] synthesized macrocycle
82 from 18b-glycyrrhetinic acid,
78, using click chemistry. Glycyrrhetinic acid,
78 (
Scheme 13), reacted with proparogyl bromide in the presence of cesium carbonate to give
79 in a yield of 89%. Subsequent acylation gave bromo derivative
80 in good yield. For the final stage of cyclization, precursor
80 was converted to azide
81 in preparation for a Huisgen reaction to form the target macrocycle
82 in 11% yield (
Scheme 13). Macrocycle
82 showed selectivity in recognizing fluoride and Hg
2+ ions through binding to the 1,2,3-triazole moiety and carbonyl group, respectively.
Metacyclophanes and paracyclophanes are among the most well-studied class of macrocyclic compounds and include the calix[n]arenes, important macrocyclic platforms exhibiting various receptor properties depending on their size and conformation [
48,
49,
50,
51,
52,
53], (thia)calix[n]arenes, and pillar[n]arenes.
To date, various macrocyclic systems and supramolecular assemblies based on (thia)calixarenes have been developed and synthesized [
54,
55,
56]. A group of Chinese scientists led by Lai synthesized calix[4]arenes
85a–
b and
86a–
b, containing triterpenoid (cholesterol) fragments which had liquid crystal properties [
53]. Cholesterol,
73, is often used as a good structural unit for the creation of various types of liquid crystal materials with interesting mesomorphic properties [
57,
58,
59,
60,
61]. The first liquid crystals were obtained from calix[4]arene-cholesterol derivatives
85a–
b and
86a–
b (Scheme XIV), which were synthesized in 50–80% yields [
57]. The synthetic pathway,
Scheme 14, shows the alkylation of
p-tert-butylthiacalix[4]arene with cholesterol derivatives
74a–
b and allowed the synthesis of di- and tetrasubstituted macrocycles
85a–
b and
86a–
b. The yield of disubstituted products is higher than that of the tetrasubstituted, which is most likely due to steric hindrance in the reaction with a cholesterol derivative. Differential scanning calorimetry (DSC) was used in the preliminary study of the mesomorphic behavior of compounds
85a–
b and
86a–
b. All new compounds have two thermal peaks on the second heating and cooling, respectively. Judging by the heats of fusion and hysteresis between the peaks of crystallization and melting, for compounds
85a–
b and
86a–
b there are two solid-state-mesophase and mesophase-isotropic phase transitions upon cooling and heating, which is also confirmed by the data of polarization optical microscopy (POM). Compounds
85b and
86b represent a broader mesophase spectrum than compounds
85a and
86a, suggesting that the spacers between the cholesterol moiety and calixarene strongly affect the mesomorphic properties. The long spacer proved to be more suitable so that each part of the structure could find its own counterpart in the resulting mesophase. Moreover, the mesomorphic temperature range of compounds
86a and
86b with four cholesterol units was greater than that of compounds
85a and
85b with two cholesterol units. These results showed that the number of cholesterol units also plays an important role in mesomorphic properties. The more cholesterol units were in the compound, the wider the mesophase temperature range. For compounds
85a–
b and
86a–
b, clear textures were observed upon cooling. These textures were similar to typical focal-conical columnar liquid crystals, which were further confirmed by X-ray diffraction analysis. In addition, X-ray diffraction showed that different lengths of spacers and different amounts of cholesterol units in the calixarene structure affected the crystal packing to a certain extent [
57].
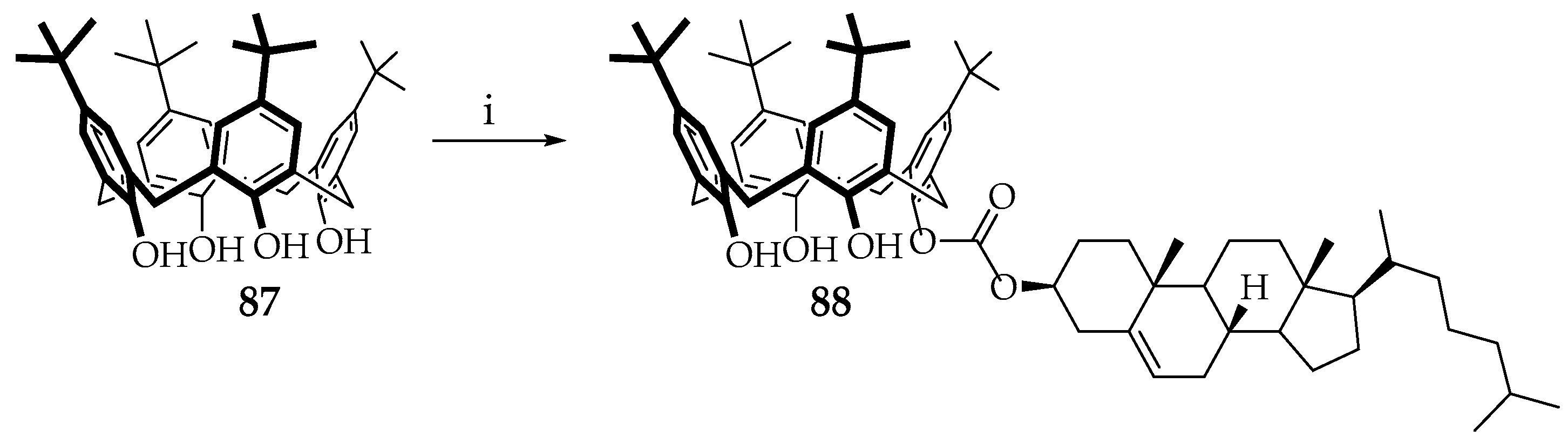
Cholesterol moieties in the calixarene structure are also used to obtain the properties of gels. Thus, a group of Indian scientists led by Rao reported [
62] the synthesis of monosubstituted
p-tert-butylthiacalix[4]arene,
88, functionalized with a triterpenoid cholesterol fragment. The target compound was synthesized by one-stage condensation of chloroformylcholesterol with
p-tert-butylcalix[4]arene (
Scheme 15). Product
88 was characterized by
1H and
13C NMR, ESI-MS, FTIR, and elemental analysis [
62].
The gel capability of
88 was tested in twenty organic solvents and demonstrated versatile gelling properties with various solvent combinations. The minimum concentration of
88 in the gel agent in THF/acetonitrile (1:1) was 0.6 wt%. SEM and AFM images of the gel showed a well-ordered nanostructured system, while the sol showed spherical nanoaggregates. The ability of the synthesized compounds to interact with rhodamine, fluorescein, doxorubicin, curcumin, and tocopheryl acetate was shown by fluorescence spectroscopy, while gel processes were also observed. Detailed fluorescence measurements clearly confirmed the uptake of drug compounds in the gel. Quantum-chemical modeling showed that there is an interaction between the guest π-systems with the C-H cholesteric fragment, indicating that the captured guest molecules are stabilized due to C-H ··· π-interactions. Encapsulated guests are easily released when the gel is heated above room temperature [
62].
A group led by Zhao synthesized [
63] calix[4]arene
95, functionalized on the upper rim with triterpenoid (cholesterol) fragments (
Figure 10 and
Scheme 16).
The compounds obtained are amphiphilic macrocycles with one (
96a–
b,
97 and
98) (
Figure 10) or two (
95) (
Scheme 16) cholesterol fragments on the upper rim of the tetrasubstituted macrocycle. The conformational rigidity of the spacer between the terpenoid and macrocyclic parts also varies with rigid 4-aminobenzoyl (
97) or flexible 4-aminobutyroyl (
98) linkers. The synthesized macrocycles with cholesterol fragments attached to the macrocyclic framework can form multiple hydrogen bonds due to the presence of secondary amide groups. Due to this, calix[4]arenes
95-
98 can easily form supramolecular assemblies with the formation of monomolecular and reversed micelles. Containers based on calix[4]arenes
95-
98 give stable micelles regardless of the nature of the rigidity of the spacer. The authors suggest that cholesterol fragments directly attached to macrocycle
95 in polar solvents have fewer degrees of freedom in comparison with terminal cholesterol fragments, which leads to the formation of more ordered supramolecular structures of calix[4]arene
95 compared to
96-
98 (
Figure 10) [
63].
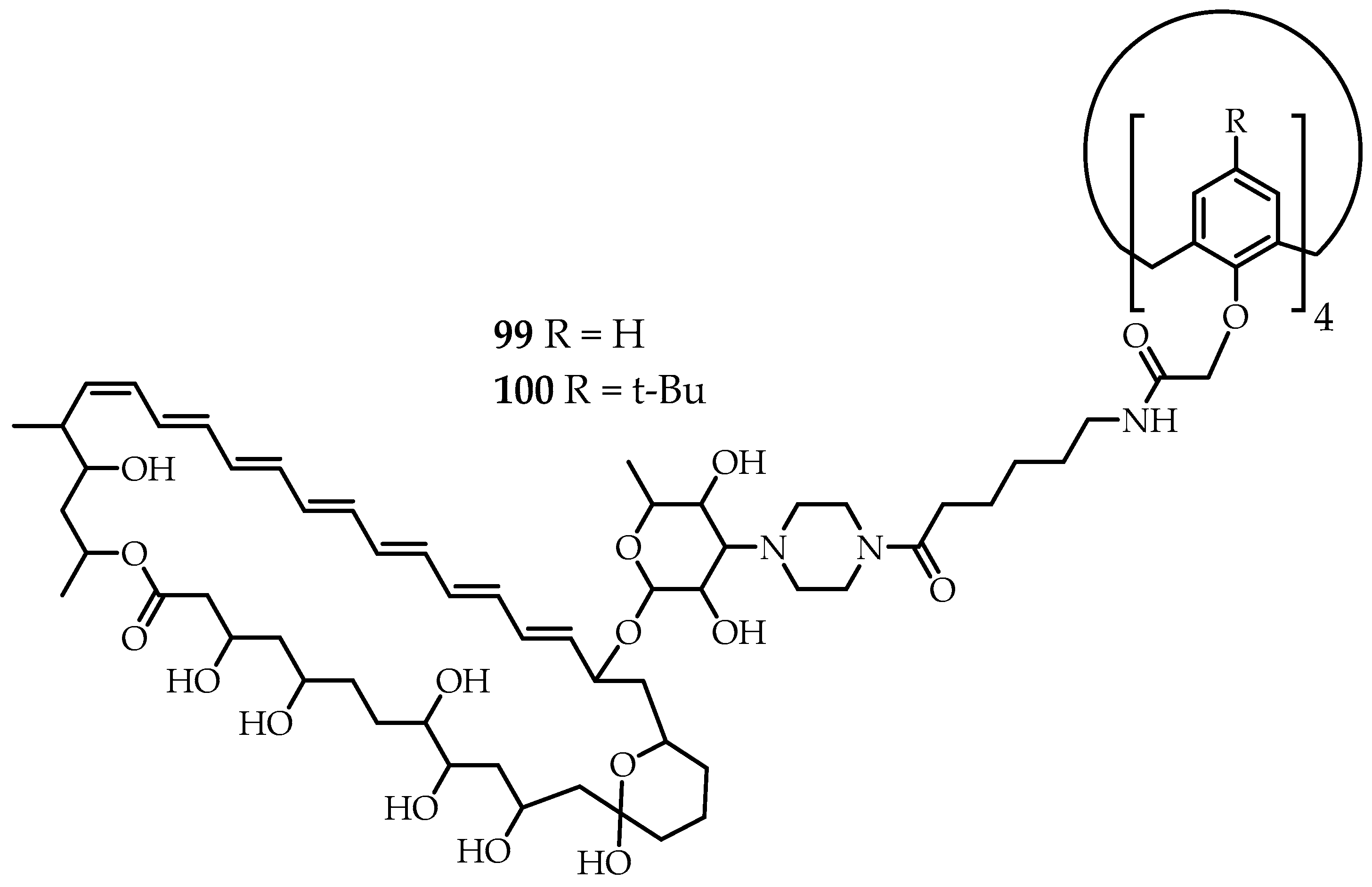
Carreira’s group [
64] synthesized new calix[4]arene derivatives
99 and
100 (
Figure 11), containing four meroterpenoid fragments (amphotericin B). The synthesized macromolecules assume a cone-shaped conformation that mimics the structure of a transmembrane pore. The antifungal activity of derivatives
90 and
100 was similar to amphotericin B with the minimum inhibitory concentrations of 0.10 and 0.25 μM, respectively. In addition, the hemotoxicity of the new derivatives was significantly lower (at least 10×) than the hemotoxicity of monomeric amphotericin B. Finally, the ability of compounds
99 and
100 was shown to form ion channels in the lipid bilayer [
64].
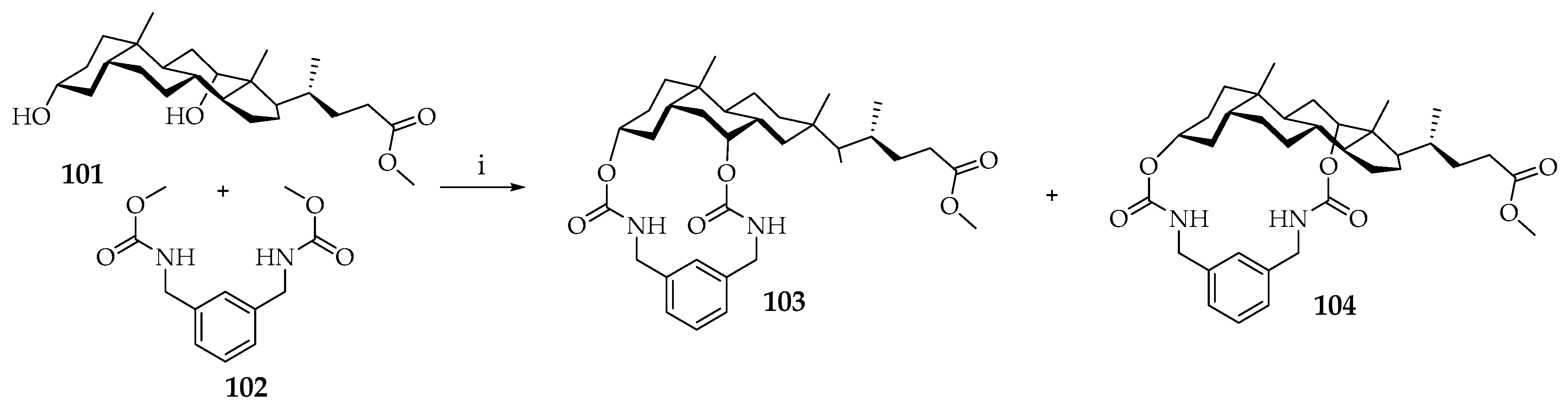
A group led by Yamada [
65] synthesized steroid metacyclophans
103 and
104 (
Scheme 17). The authors carried out regioselective macrocyclization of methylcholate
101 using dimethyl-α,α,α’,α’-tetramethyl-
m-xylylenedicarbamate
102. The reaction was carried out by transesterification in boiling toluene (0.004 M) using 3% SnCl
2 and 1% glycoluril as catalysts. After 2 h of reaction, product
103 has been obtained selectively in high yield (94%). Conversion of 16-membered macrocycle
103 to 18-membered cyclophane
104 was achieved by equilibration in 7 days to obtain thermodynamically stable
104 in yield 68%. New steroidal carbamoyloxy-bridging cyclophans
103 and
104 are examples of regioisomeric steroidal cyclophans [
65].
As a low molecular weight gelling agent, Leyong Wang et al. synthesized a pillar[6]arene functionalized with a derivative of cholesterol
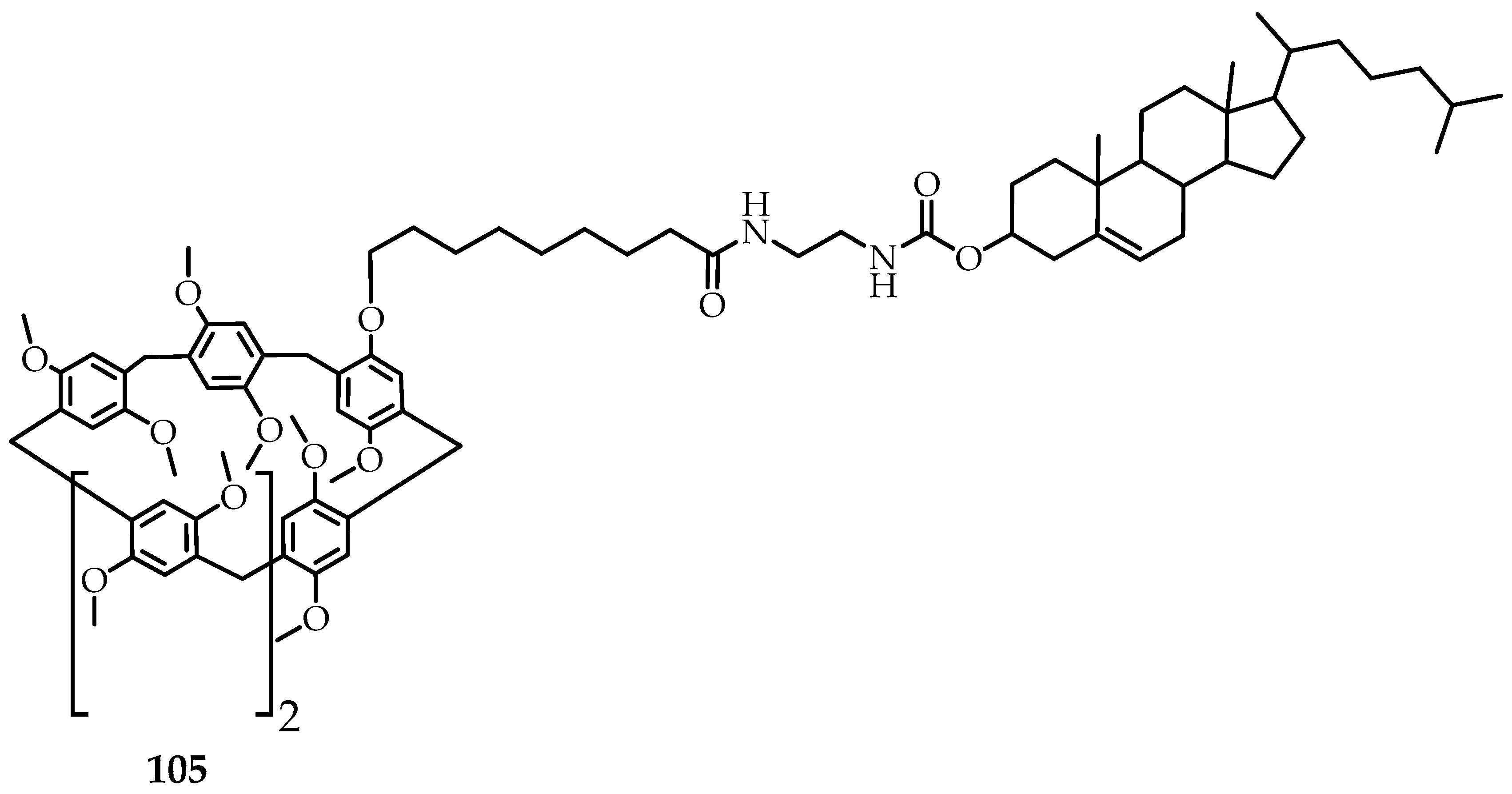
105 (
Figure 12), which forms an organogel in a cyclohexane/n-hexanol mixture (10:1 V/V) at 10 °C. The resulting organogel reacts reversibly to temperature changes and forms guest–host complexes [
66].
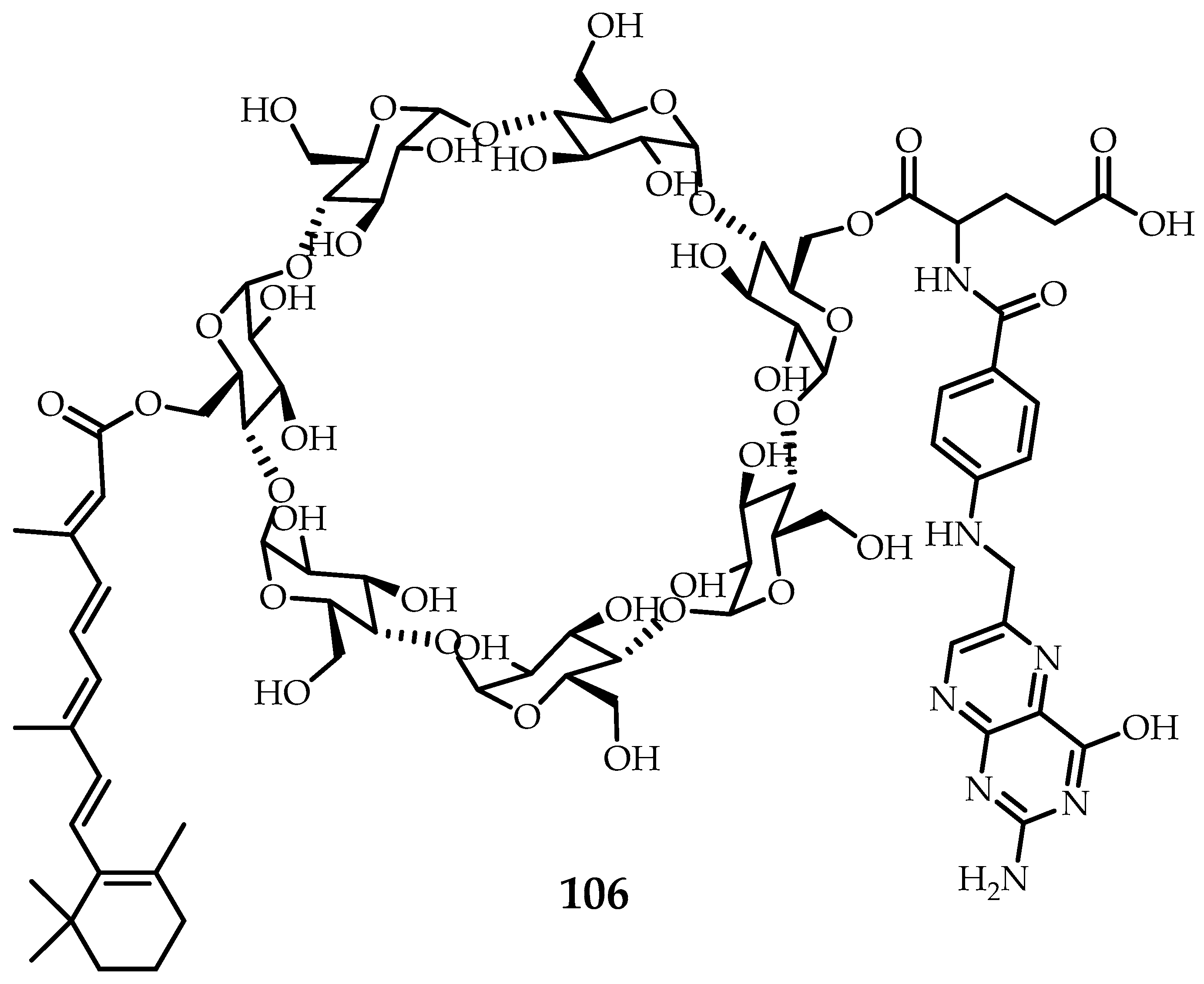
Varshosaz et al. [
67] successfully synthesized meroterpenoid
106 (
Figure 13) based on β-cyclodextrin, folic, and retinoic acid which formed stable submicron self-associates in water. The stability of the formed colloidal system was additionally confirmed by measuring the value of the ζ-potential. It was shown that macrocycle
106 (
Figure 13) is able to effectively encapsulate the anticancer drug, doxorubicin [
67].
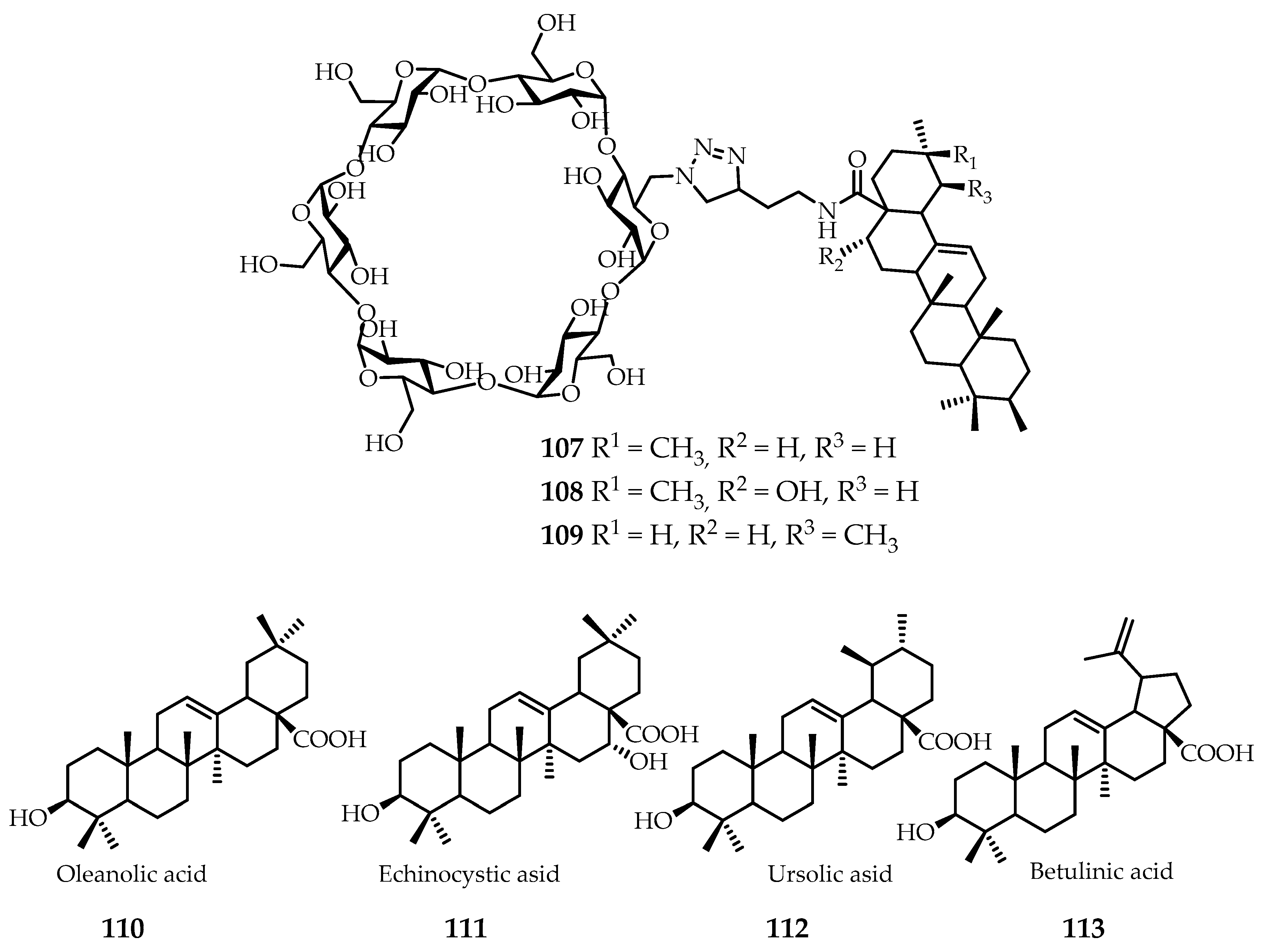
In 2016 a series of water-soluble meroterpenoids
107–
109 (
Figure 14) was synthesized based on α-cyclodextrin/pentacyclic hopanoids (
110–
113) (
Figure 14) and obtained in good yields [
68]. Inhibiting the entry of HCV (hepatitis C virus) is a key goal in the treatment of chronic hepatitis C virus infection. HCV entry inhibition activity was determined based on HCVpp/VSVGpp penetration assays. The best results were found for compounds
107 and
108, which showed the most promising activity inhibition of HCV penetration with mean IC
50 values of 1.18 mM and 0.25 mM. In addition, the in vitro cytotoxic activity of these two compounds against MDCK cells was absent even at 100 mM. Five different binding assays were identified to determine the mechanism of action; the results showed that the compounds exhibit their inhibitory activity at the stage after HCV binding and subsequently prevent the penetration of the virus [
68].
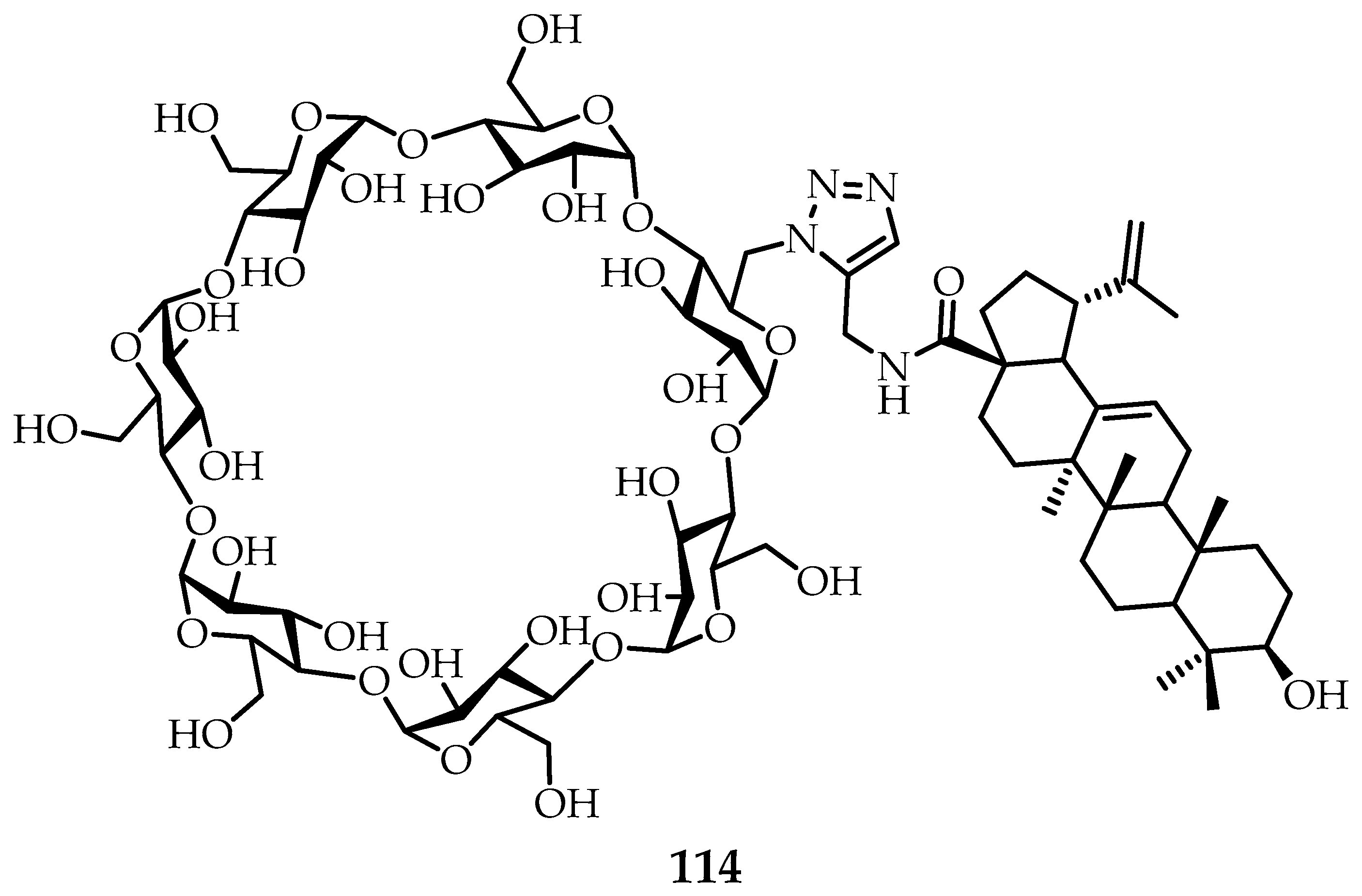
The results obtained are in good agreement with studies carried out on β-cyclodextrins [
69]. It was previously found that oleanolic acid
110 and echinocystic acid
111, isolated from
Dipsacusasperoides, have the properties of inhibiting HCV penetration. The main problem in using this type of triterpenes is their low aqueous solubility. Thus, the introduction of fragments of hopanoids (
110–
113) into the structure of α- and β-cyclodextrins increases the solubility of terpenes in water. In the case of β-cyclodextrin
114 meroterpenoid (
Figure 15), moderate activity inhibition of HCV penetration was found. All synthesized meroterpenoids showed no cytotoxicity based on the
alamarBlue assay performed on
HeLa,
HepG2,
MDCK, and
293T cells. It should also be noted that when the terpene and β-cyclodextrin structures were combined, the obtained triterpenes became hemolytically inactive. A simple synthetic approach to the preparation of meroterpenoids, combining the properties of β-cyclodextrin and pentacyclic triterpenoids, based on the concept of “click chemistry”, can provide a way to obtain a new class of inhibitors of HCV penetration [
68,
69].
Thus, the variety of synthetic, macrocyclic meroterpenoids leads to the conclusion that the development of new generation materials with biologically significant properties is impossible without the use of natural products. Terpenoids, such as natural products, are ideal for these purposes, as they have low toxicity and are completely biocompatible. The advantages of targeted synthesis of macrocyclic meroterpenoids include: (1) ease of production (a variety of synthetic methods makes it easy to vary the structure of the target meroterpenoids), (2) scalability (the developed synthetic methods allow obtaining a large amount of substance in a short time), (3) independence from external conditions (any specialist equipped with the appropriate equipment, can easily reproduce the synthetic method). However, the directed synthesis of macrocyclic meroterpenoids has one very important drawback, which is the loss of biological activity of the terpenoid fragment, which is part of the macrocyclic compound, caused by chemical modification. This disadvantage must always be taken into account when drawing up a synthetic strategy.