Integration of Phenotype and Hormone Data during Adventitious Rooting in Carnation (Dianthus caryophyllus L.) Stem Cuttings
Abstract
:1. Introduction
2. Results
2.1. Phenotype Characterization of Parental Lines and the F1 Population Under Study
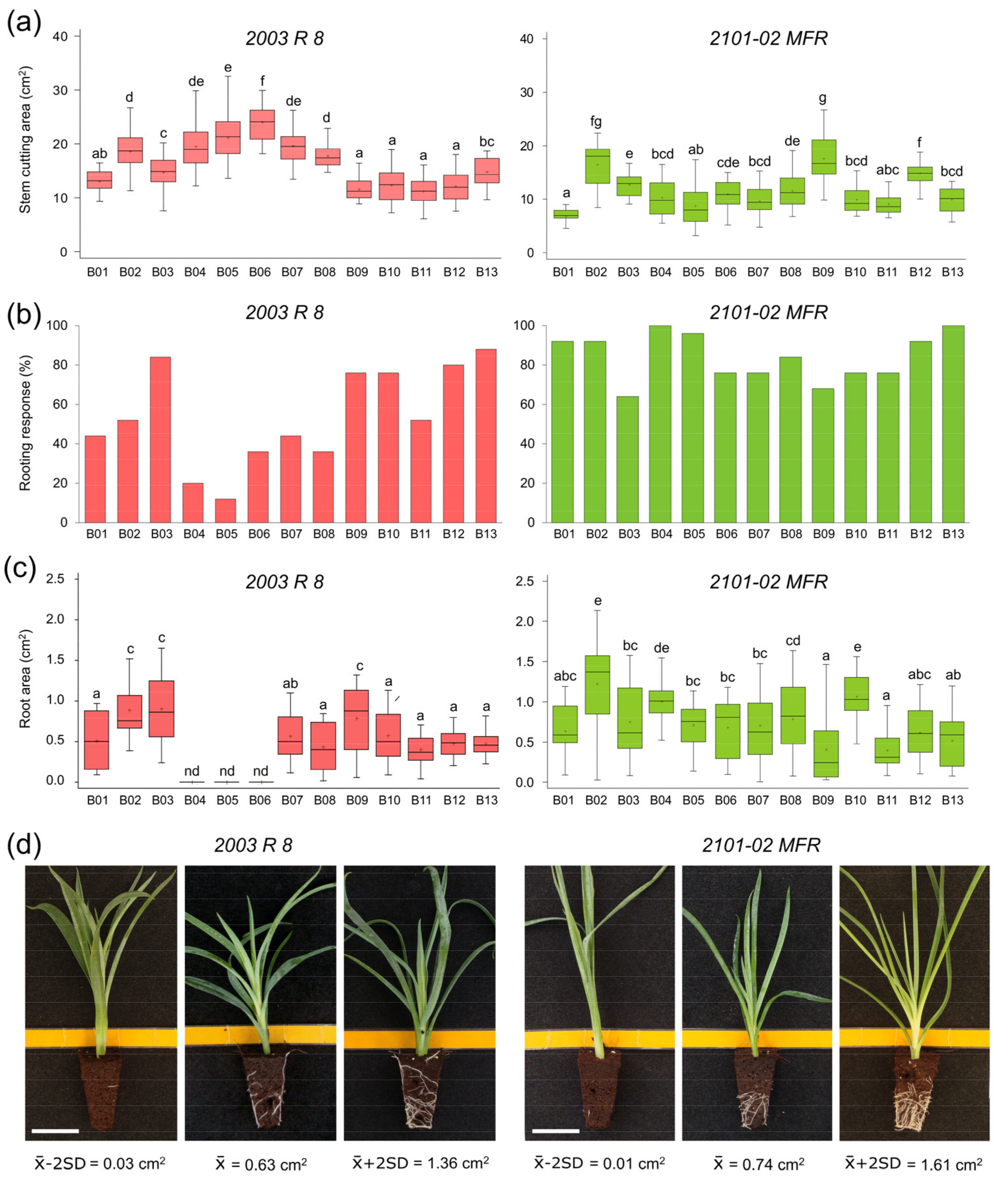
2.1.1. Morphological Variation in 2003 R 8 and 2101-02 MFR During Rooting
2.1.2. Morphological Variation of the Studied F1 Population
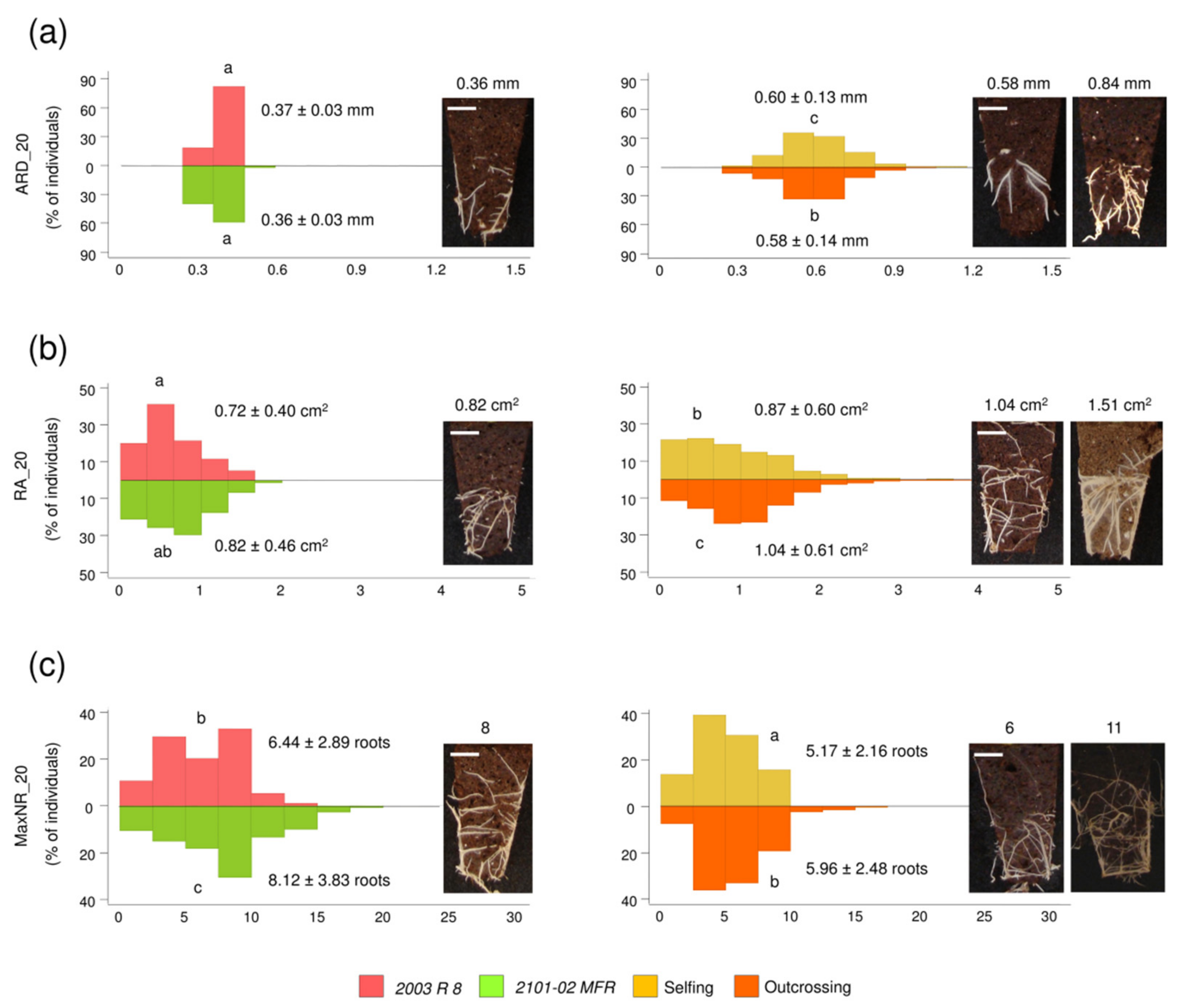
2.1.3. Transgressive Phenotypes in the Studied F1 Population
2.2. Study of a Reduced Panel of Double-Cross F1 Hybrids
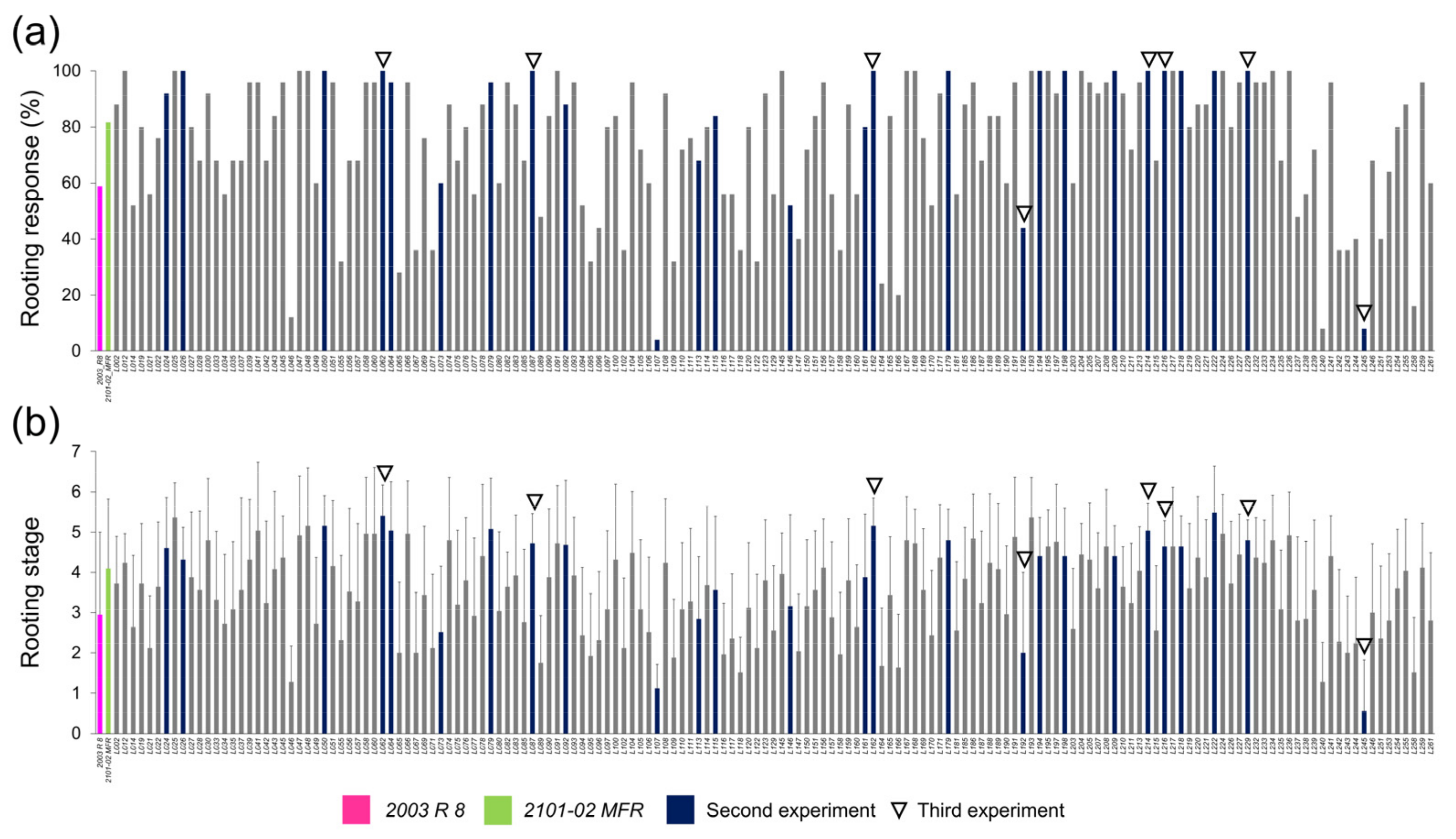
2.2.1. Selection of Double-Cross F1 Hybrids and Heritability Estimation of the Studied Traits
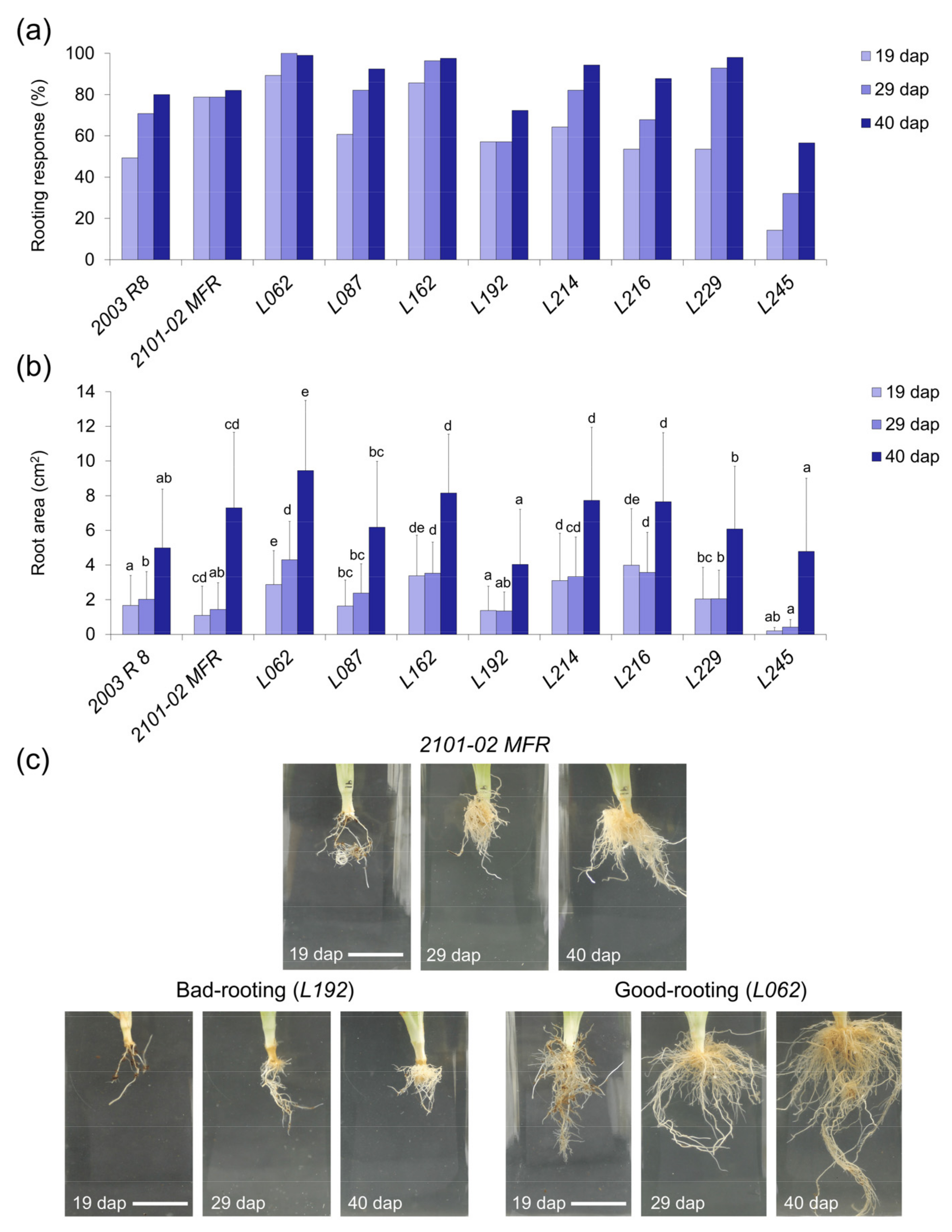
2.2.2. Rooting Performance of Double-Cross F1 Hybrids
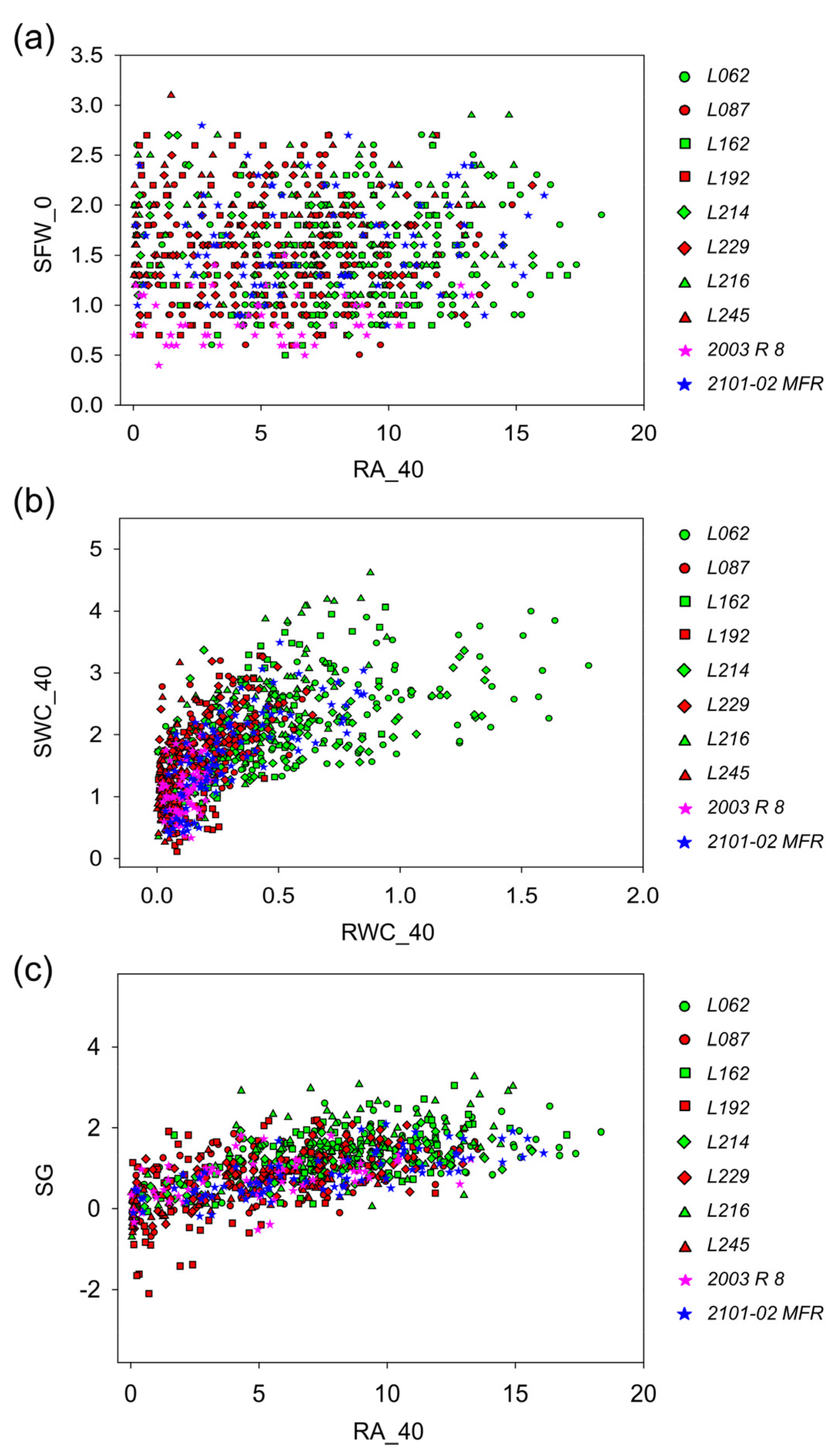
2.2.3. Stem Cutting Ecophysiology of Double-Cross F1 Hybrids
2.2.4. Capturing Variation in Morphometric, Ecophysiological, and Adventitious Rooting Traits in Carnation Stem Cuttings
2.2.5. Hormonal Profiling in the Stem Cutting Base During the Rooting of Double-Cross F1 Hybrids
3. Discussion
4. Materials and Methods
4.1. Plant Material, Growing Conditions, and Sample Collection
4.1.1. First and Second Experiments
4.1.2. Third Experiment
4.2. Picture and Image Processing
4.2.1. Image Collection
4.2.2. Image Processing
4.3. Statistical Analyses
4.3.1. Estimation of Heterosis in Selfing and Outcrossing F1 Populations
4.3.2. Estimation of Broad-Sense Heritability in Selected Lines
4.4. Phytohormone Extraction and Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bellini, C.; Pacurar, D.I.; Perrone, I. Adventitious roots and lateral roots: Similarities and differences. Annu. Rev. Plant Biol. 2014, 65, 639–666. [Google Scholar] [CrossRef] [PubMed]
- Steffens, B.; Rasmussen, A. The physiology of adventitious roots. Plant Physiol. 2016, 170, 603–617. [Google Scholar] [CrossRef] [PubMed]
- Druege, U.; Hilo, A.; Pérez-Pérez, J.M.; Koplotek, Y.; Acosta, M.; Shahinnia, F.; Zerche, S.; Franken, P.; Hajirezaei, M.R. Molecular and physiological control of adventitious rooting in cuttings: Phytohormone action meets resource allocation. Ann. Bot. 2019, 123, 929–949. [Google Scholar] [CrossRef] [PubMed]
- Garrido, G.; Arnao, M.B.; Acosta, M.; Sánchez-Bravo, J. Polar transport of indole-3-acetic acid in relation to rooting in carnation cuttings: Influence of cold storage duration and cultivar. Biol. Plant. 2003, 47, 481–485. [Google Scholar] [CrossRef]
- Birlanga, V.; Villanova, J.; Cano, A.; Cano, E.A.; Acosta, M.; Pérez-Pérez, J.M. Quantitative analysis of adventitious root growth phenotypes in carnation stem cuttings. PLoS ONE 2015, 10, e0133123. [Google Scholar] [CrossRef] [PubMed]
- Villacorta-Martín, C.; Sánchez-García, A.B.; Villanova, J.; Cano, A.; van de Rhee, M.; de Haan, J.; Acosta, M.; Passarinho, P.; Pérez-Pérez, J.M. Gene expression profiling during adventitious root formation in carnation stem cuttings. BMC Genom. 2015, 16, 789–806. [Google Scholar] [CrossRef] [PubMed]
- Cano, A.; Sánchez-García, A.B.; Albacete, A.; González-Bayón, R.; Justamante, M.S.; Ibáñez, S.; Acosta, M.; Pérez-Pérez, J.M. Enhanced conjugation of auxin by GH3 enzymes leads to poor adventitious rooting in carnation stem cuttings. Front. Plant Sci. 2018, 9, 566. [Google Scholar] [CrossRef]
- Villanova, J.; Cano, A.; Albacete, A.; López, A.; Cano, E.Á.; Acosta, M.; Pérez-Pérez, J.M. Multiple factors influence adventitious rooting in carnation (Dianthus caryophyllus L.) stem cuttings. Plant Growth Regul. 2017, 81, 511–521. [Google Scholar] [CrossRef]
- Galkovskyi, T.; Mileyko, Y.; Bucksch, A.; Moore, B.; Symonova, O.; Price, C.A.; Topp, C.N.; Iyer-Pascuzzi, A.S.; Zurek, P.R.; Fang, S.; et al. GiA Roots: Software for the high throughput analysis of plant root system architecture. BMC Plant Biol. 2012, 12, 116. [Google Scholar] [CrossRef]
- Agulló-Antón, M.Á.; Ferrández-Ayela, A.; Fernández-García, N.; Nicolás, C.; Albacete, A.; Pérez-Alfocea, F.; Sánchez-Bravo, J.; Pérez-Pérez, J.M.; Acosta, M. Early steps of adventitious rooting: Morphology, hormonal profiling and carbohydrate turnover in carnation stem cuttings. Physiol. Plant. 2014, 150, 446–462. [Google Scholar] [CrossRef]
- Lischweski, S.; Muchow, A.; Guthörl, D.; Hause, B. Jasmonates act positively in adventitious root formation in petunia cuttings. BMC Plant Biol. 2015, 15, 229. [Google Scholar] [CrossRef] [PubMed]
- Fattorini, L.; Hause, B.; Gutierrez, L.; Veloccia, A.; Della Rovere, F.; Piacentini, D.; Falasca, G.; Altamura, M.M. Jasmonate promotes auxin-induced adventitious rooting in dark-grown Arabidopsis thaliana seedlings and stem thin cell layers by a cross-talk with ethylene signalling and a modulation of xylogenesis. BMC Plant Biol. 2018, 18, 182. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zhao, F.; Chen, L.; Pan, Y.; Sun, L.; Bao, N.; Zhang, T.; Cui, C.X.; Qiu, Z.; Zhang, Y.; et al. Jasmonate-mediated wound signalling promotes plant regeneration. Nat. Plants 2019, 5, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Yang, L.; Mao, C.; Huang, Y.; Wu, P. Comparison of QTLs for rice seedling morphology under different water supply conditions. J. Genet. Genom. 2008, 35, 473–484. [Google Scholar] [CrossRef]
- Sato, S.; Katoh, N.; Yoshida, H.; Iwai, S.; Hagimori, M. Production of doubled haploid plants of carnation (Dianthus caryophyllus L.) by pseudofertilized ovule culture. Sci. Hortic. 2000, 83, 301–310. [Google Scholar] [CrossRef]
- Onozaki, T. Breeding of carnations (Dianthus caryophyllus L.) for long vase life. Breed. Sci. 2018, 68, 3–13. [Google Scholar] [CrossRef]
- Xu, X.; Ji, J.; Xu, Q.; Qi, X.; Weng, Y.; Chen, X. The major-effect quantitative trait locus CsARN6.1 encodes an AAA ATPase domain-containing protein that is associated with waterlogging stress tolerance by promoting adventitious root formation. Plant J. 2018, 93, 917–930. [Google Scholar] [CrossRef]
- Hoecker, N.; Keller, B.; Piepho, H.P.; Hochholdinger, F. Manifestation of heterosis during early maize (Zea mays L.) root development. Theor. Appl. Genet. 2006, 112, 421–429. [Google Scholar] [CrossRef]
- Mano, Y.; Muraki, M.; Fujimori, M.; Takamizo, T.; Kindiger, B. Identification of QTL controlling adventitious root formation during flooding conditions in teosinte (Zea mays ssp. huehuetenangensis) seedlings. Euphytica 2005, 142, 33–42. [Google Scholar] [CrossRef]
- Ochoa, I.E.; Blair, M.W.; Lynch, J.P. QTL analysis of adventitious root formation in common bean under contrasting phosphorus availability. Crop Sci. 2006, 46, 1609–1621. [Google Scholar] [CrossRef]
- Horii, H.; Nemoto, K.; Miyamoto, N.; Harada, J. Quantitative trait loci for adventitious and lateral roots in rice. Plant Breed. 2006, 125, 198–200. [Google Scholar] [CrossRef]
- Ribeiro, C.L.; Silva, C.M.; Drost, D.R.; Novaes, E.; Novaes, C.R.; Dervinis, C.; Kirst, M. Integration of genetic, genomic and transcriptomic information identifies putative regulators of adventitious root formation in Populus. BMC Plant Biol. 2016, 16, 66. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Chen, G.; Zhang, X.; Li, T.; Yu, H.; Liu, C. Quantitative trait locus analysis of adventitious and lateral root morphology of barley grown at low and high P. Funct. Plant Biol. 2018, 45, 957–967. [Google Scholar] [CrossRef]
- Dodd, I.C.; Puértolas, J.; Huber, K.; Pérez-Pérez, J.G.; Wright, H.R.; Blackwell, M.S. The importance of soil drying and re-wetting in crop phytohormonal and nutritional responses to deficit irrigation. J. Exp. Bot. 2015, 66, 2239–2252. [Google Scholar] [CrossRef] [PubMed]
- Faget, M.; Nagel, K.A.; Walter, A.; Herrera, J.M.; Jahnke, S.; Schurr, U.; Temperton, V.M. Root–root interactions: Extending our perspective to be more inclusive of the range of theories in ecology and agriculture using in-vivo analyses. Ann. Bot. 2013, 112, 253–266. [Google Scholar] [CrossRef]
- Franco, J.A.; Bañón, S.; Vicente, M.J.; Miralles, J.; Martínez-Sánchez, J.J. Root development in horticultural plants grown under abiotic stress conditions—A review. J. Hortic. Sci. Biotechnol. 2011, 86, 543–556. [Google Scholar] [CrossRef]
- Wu, F.; Yang, X.; Wang, Z.; Deng, M.; Ma, J.; Chen, G.; Wei, Y.; Liu, Y. Identification of major quantitative trait loci for root diameter in synthetic hexaploid wheat under phosphorus-deficient conditions. J. Appl. Genet. 2017, 58, 437–447. [Google Scholar] [CrossRef]
- Qiu, X.Q.; Gao, Y.; Huang, L.; Li, X.Q.; Sun, J.S.; Duan, A.W. Temporal and spatial distribution of root morphology of winter wheat. Sci. Agric. Sin. 2013, 46, 2211–2219. [Google Scholar]
- Zhao, C.; Craig, J.C.; Petzold, H.E.; Dickerman, A.W.; Beers, E.P. The xylem and phloem transcriptomes from secondary tissues of the Arabidopsis root-hypocotyl. Plant Physiol. 2005, 138, 803–818. [Google Scholar] [CrossRef]
- Yang, W.; Zhu, C.; Ma, X.; Li, G.; Gan, L.; Ng, D.; Xia, K. Hydrogen peroxide is a second messenger in the salicylic acid-triggered adventitious rooting process in mung bean seedlings. PLoS ONE 2013, 8, e84580. [Google Scholar] [CrossRef]
- Pacurar, D.I.; Perrone, I.; Bellini, C. Auxin is a central player in the hormone cross-talks that control adventitious rooting. Physiol. Plant. 2014, 151, 83–96. [Google Scholar] [CrossRef] [PubMed]
- De Klerk, G.J.; Hanecakova, J. Ethylene and rooting of mung bean cuttings. The role of auxin induced ethylene synthesis and phase-dependent effects. Plant Growth Regul. 2008, 56, 203. [Google Scholar] [CrossRef]
- Veloccia, A.; Fattorini, L.; Della Rovere, F.; Sofo, A.; D’angeli, S.; Betti, C.; Falasca, G.; Altamura, M.M. Ethylene and auxin interaction in the control of adventitious rooting in Arabidopsis thaliana. J. Exp. Bot. 2016, 67, 6445–6458. [Google Scholar] [CrossRef] [PubMed]
- Negi, S.; Sukumar, P.; Liu, X.; Cohen, J.D.; Muday, G.K. Genetic dissection of the role of ethylene in regulating auxin-dependent lateral and adventitious root formation in tomato. Plant J. 2010, 61, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Jawaharlal, M.; Ganga, M.; Padmadevi, K.; Jegadeeswari, V.; Karthikeyan, S. A Technical Guide on Carnation; Tamil Nadu Agricultural University: Coimbatore, India, 2009; pp. 1–56. [Google Scholar]
- Aguinis, H.; Gottfredson, R.K.; Joo, H. Best-practice recommendations for defining, identifying, and handling outliers. Organ. Res. Methods 2013, 16, 270–301. [Google Scholar] [CrossRef]
- Zeleke, H. Heterosis and combining ability for grain yield and yield component traits of maize in eastern Ethiopia. Sci. Technol. Arts Res. J. 2015, 4, 32–37. [Google Scholar] [CrossRef]
- Lynch, M.; Walsh, B. Genetics and Analysis of Quantitative Traits; Sinauer: Sunderland, MA, USA, 1998; Volume 1, pp. 535–557. [Google Scholar]
- Albacete, A.; Ghanem, M.E.; Martínez-Andújar, C.; Acosta, M.; Sánchez-Bravo, J.; Martínez, V.; Lutts, S.; Dodd, I.C.; Pérez-Alfocea, F. Hormonal changes in relation to biomass partitioning and shoot growth impairment in salinized tomato (Solanum lycopersicum L.) plants. J. Exp. Bot. 2008, 59, 4119–4131. [Google Scholar] [CrossRef]
- Gente, R.; Koch, M. Monitoring leaf water content with THz and sub-THz waves. Plant Methods 2015, 11, 15. [Google Scholar] [CrossRef]


| Studied Traits | Acronym | Measurement Mode a | Unit |
|---|---|---|---|
| Leaf number | LN_13 | Human operator | >1 |
| Average leaf section | ALS_13 | Average Width1 | mm |
| Cutting height | CH_13 | Network Depth1 | cm |
| Shoot area | SA_13, SA_20 | Network Area1 | cm2 |
| Shoot area increase | ΔSA | SA_20–SA_13 | cm2 |
| Rooting response | RR_20 | Human operator2 | % |
| Rooting stage | RS_20 | Human operator2 | 0 to 7 |
| Maximum number of roots | MaxNR_20 | Maximum number of roots1 | >1 |
| Average root diameter | ARD_20 | Average root width1 | mm |
| Root area | RA_20 | Network area1 | cm2 |
| Root convex area | RCA_20 | Network convex area1 | cm2 |
| Root perimeter | RP_20 | Network perimeter1 | cm |
| Shoot Traits | Acronym a | Measurement Mode b | Unit |
|---|---|---|---|
| Shoot fresh weight | SFW_0, SFW_40 | Precision scale | g |
| Cutting length | CL_0 | Digital caliper | cm |
| Shoot dry weight | SDW_40 | Heater, high precision scale | mg |
| Shoot water content | SWC | SFW_40 − SDW_40 | g |
| Shoot growth | SG | SFW_40 − SFW_0 | g |
| Root fresh weight/root growth | RFW_40 | Precision scale | g |
| Root dry weight | RDW_40 | Heater, high precision scale | mg |
| Root water content | RWC | RFW_40 − RDW_40 | g |
| Rooting response | RR | Human operator1 | % |
| Average root diameter | ARD | Average root width2 | mm |
| Root area | RA | Network area2 | cm2 |
| Root convex area | RCA | Network convex area2 | cm2 |
| Root depth | RD | Network depth2 | cm |
| Root length | RL | Network length2 | cm |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Justamante, M.S.; Acosta-Motos, J.R.; Cano, A.; Villanova, J.; Birlanga, V.; Albacete, A.; Cano, E.Á.; Acosta, M.; Pérez-Pérez, J.M. Integration of Phenotype and Hormone Data during Adventitious Rooting in Carnation (Dianthus caryophyllus L.) Stem Cuttings. Plants 2019, 8, 226. https://doi.org/10.3390/plants8070226
Justamante MS, Acosta-Motos JR, Cano A, Villanova J, Birlanga V, Albacete A, Cano EÁ, Acosta M, Pérez-Pérez JM. Integration of Phenotype and Hormone Data during Adventitious Rooting in Carnation (Dianthus caryophyllus L.) Stem Cuttings. Plants. 2019; 8(7):226. https://doi.org/10.3390/plants8070226
Chicago/Turabian StyleJustamante, María Salud, José Ramón Acosta-Motos, Antonio Cano, Joan Villanova, Virginia Birlanga, Alfonso Albacete, Emilio Á. Cano, Manuel Acosta, and José Manuel Pérez-Pérez. 2019. "Integration of Phenotype and Hormone Data during Adventitious Rooting in Carnation (Dianthus caryophyllus L.) Stem Cuttings" Plants 8, no. 7: 226. https://doi.org/10.3390/plants8070226
APA StyleJustamante, M. S., Acosta-Motos, J. R., Cano, A., Villanova, J., Birlanga, V., Albacete, A., Cano, E. Á., Acosta, M., & Pérez-Pérez, J. M. (2019). Integration of Phenotype and Hormone Data during Adventitious Rooting in Carnation (Dianthus caryophyllus L.) Stem Cuttings. Plants, 8(7), 226. https://doi.org/10.3390/plants8070226







