Influence of Boiling, Steaming and Frying of Selected Leafy Vegetables on the In Vitro Anti-inflammation Associated Biological Activities
Abstract
:1. Introduction
2. Results and Discussion
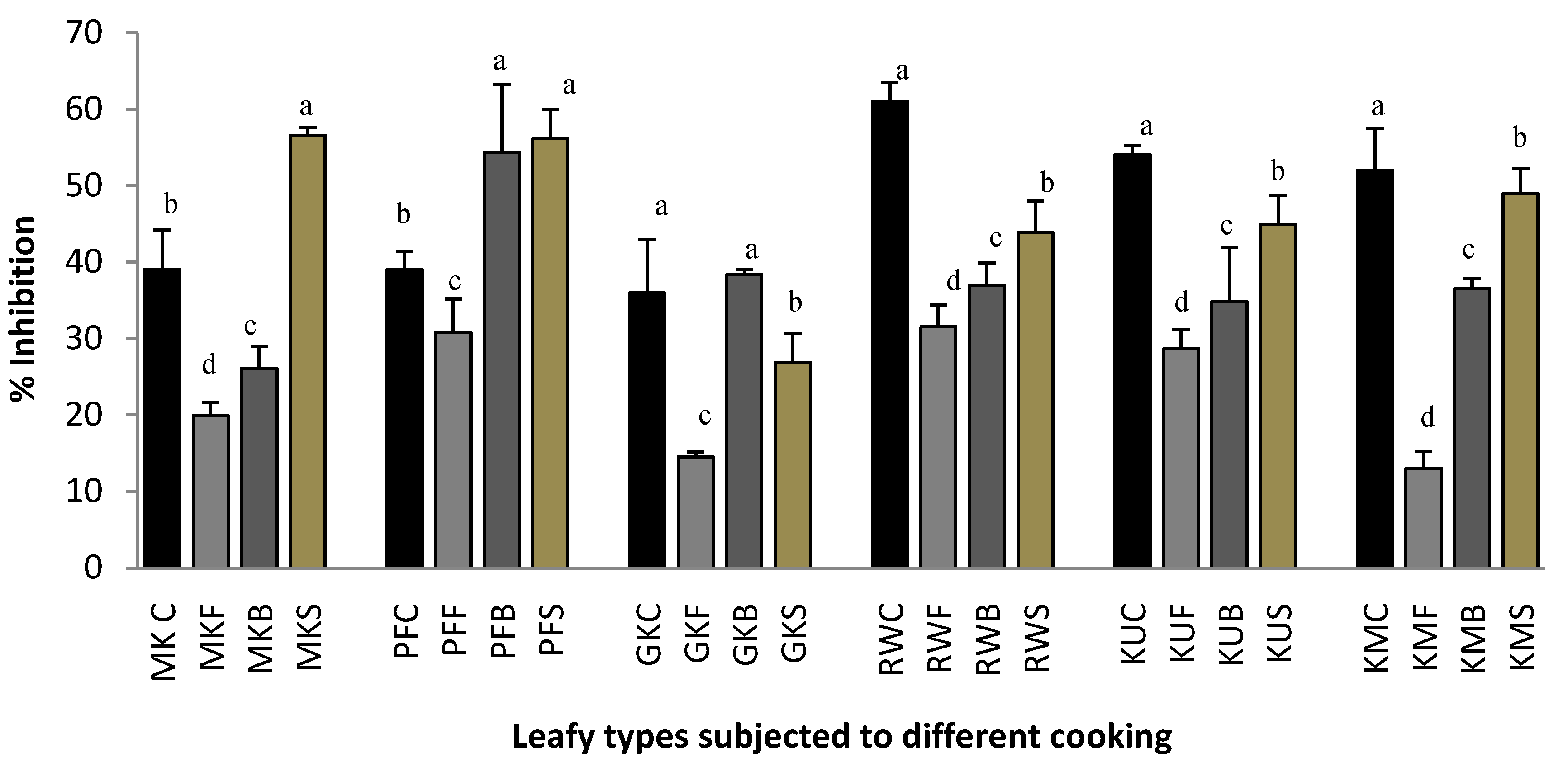
2.1. Protein Denaturation
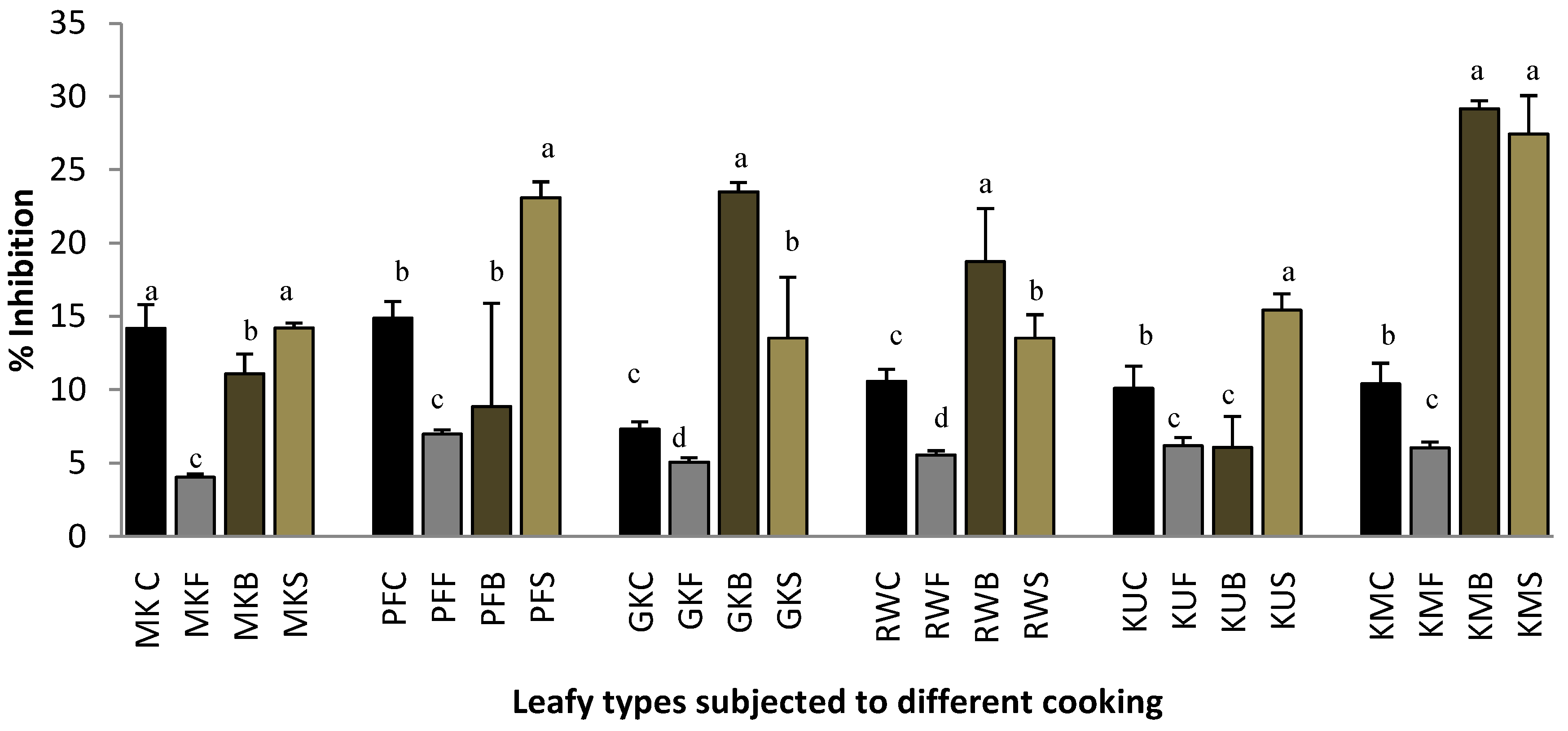
2.2. Heat-Induced Hemolysis
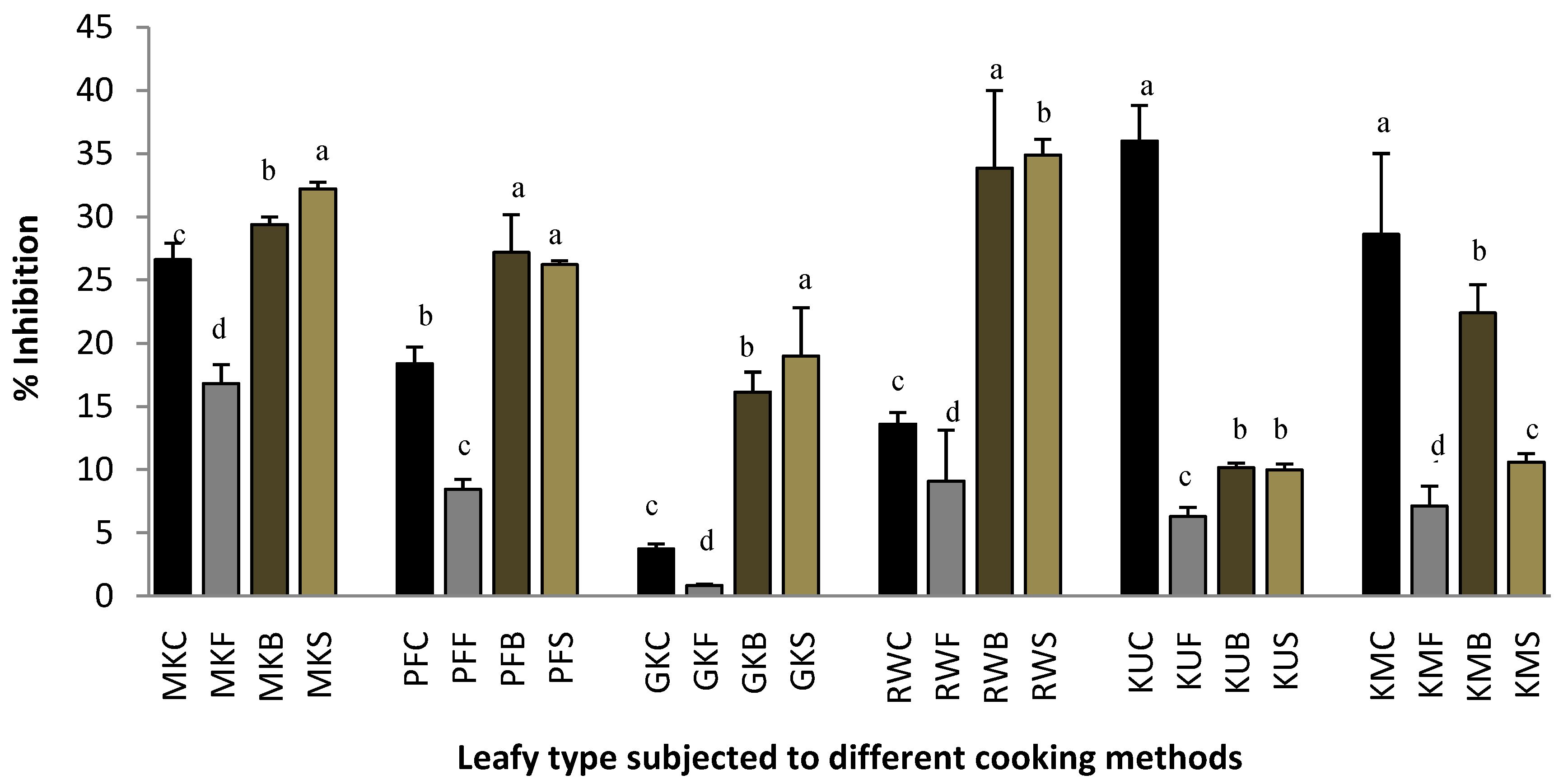
2.3. Lipoxygenase Inhibitory Activity.
2.4. Proteinase Inhibitory Activity
3. Materials and Methods
3.1. Materials
3.2. Preparation of Cooked Samples
3.3. Preparation of Methanolic Extracts.
3.4. Anti-Inflammatory Properties
3.4.1. Heat-Induced Hemolysis
3.4.2. Effect on Protein Denaturation
3.4.3. Proteinase Inhibitory Activity
3.4.4. Lipoxygenase Inhibition Assay
3.5. Analysis of Phenolics, Flavonoids and Carotenoids
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kaliora, A.C.; Dedoussis, G.V.Z.; Schmidt, H. Dietary antioxidants in preventing atherogenesis—A review. J. Atheroscler. 2006, 187, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Ferrero-Millani, L.; Nelsen, O.H.; Anderson, P.S.; Girardin, S.E. Chronic inflammation: Importance of NOD2 and NALP3 in interleukin-1β generation. Clin. Exp. Immunol. 2007, 147, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Albers, R.; Antoine, J.M.; Blum, S.; Bourdet-Sicard, R.; Ferns, G.A.; Folkerts, G.; Friedmann, P.S.; Frost, G.S.; Guarner, F.; et al. Inflammatory disease processes and interactions with nutrition. Br. J. Nutr. 2009, 101, 1–45. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Hettiarachchy, N.S.; Horax, R.; Kannan, A.; Praisoody, A.; Muhundan, A. Phytochemicals, the antioxidant and antimicrobial activity of Hibiscus sabdariffa, Centella asiatica, Moringa oleifera and Murraya koenigii leaves. J. Med. Plants Res. 2011, 5, 6672–6680. [Google Scholar]
- Oboh, G. Effect of blanching on the antioxidant properties of some tropical green leafy vegetables. LWT Food Sci. Technol. 2005, 38, 513–517. [Google Scholar] [CrossRef]
- Gunathilake, K.D.P.P.; Ranaweera, K.K.D.S.; Rupasinghe, H.P.V. Change of phenolics, carotenoids, and antioxidant capacity following simulated gastrointestinal digestion and dialysis of selected edible green leaves. Food Chem. 2018, 245, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Gunathilake, K.D.P.P.; Ranaweera, K.K.D.S. Antioxidative properties of 34 green leafy vegetables. J. Funct. Foods 2016, 26, 176–186. [Google Scholar] [CrossRef]
- Andarwulan, N.; Kurniasih, D.; Apriady, R.A.; Rahmat, H.; Roto, A.V.; Bolling, B.W. Polyphenols, carotenoids, and ascorbic acid in underutilized medicinal vegetables. J. Funct. Foods 2012, 4, 339–347. [Google Scholar] [CrossRef]
- Sakat, S.; Juvekar, A.R.; Gambhire, M.N. In vitro antioxidant and anti-inflammatory activity of methanol extract of Oxalis corniculata Linn. Int. J. Pharm. Pharm. Sci. 2010, 2, 146–155. [Google Scholar]
- Akinwunmi, K.F.; Oyedapo, O.O. In vitro Anti-inflammatory Evaluation of African Nutmeg (Monodora myristica) Seeds. Eur. J. Med. Plants 2015, 8, 167–174. [Google Scholar] [CrossRef]
- Govindappaaga, S.S.; Poojashri, M.N.; Sadananda, T.S.; Chandrappa, C.P. Antimicrobial, antioxidant and in vitroantiinflammatory activity of ethanol extract and active phytochemical screening of Wedelia trilobata (L) Hitchc. J. Pharm. Phytother. 2011, 3, 43–51. [Google Scholar]
- Chou, C.T. The anti inflammatory effect of Tripterygium wilfordii Hook F on adjuvant induced paw edema in rats and inflammatory mediator release. Phytother. Res. 1997, 11, 152–154. [Google Scholar] [CrossRef]
- Chippada, S.C.; Vangalapati, M. Antioxidant, an anti-inflammatory and anti-arthritic activity of Centella asiatica extracts. J. Chem. Biol. Phys. Sci. 2011, 1, 260. [Google Scholar]
- Alitonou, G.A.; Avlessi, F.; Sohounhloue, D.K.; Agnaniet, H.; Bessiere, J.M.; Menut, C. Investigations on the essential oil of Cymbopogon giganteus from Benin for its potential use as an anti-inflammatory agent. Int. J. Aromather. 2006, 16, 37–41. [Google Scholar] [CrossRef]
- Wachtel-Galor, S.; Wong, K.W.; Benzie, I.F. The effect of cooking on Brassica vegetables. Food Chem. 2008, 110, 706–710. [Google Scholar] [CrossRef]
- Palermo, M.; Pellegrini, N.; Fogliano, V. The effect of cooking on the phytochemical content of vegetables. J. Sci. Food Agric. 2014, 94, 1057–1070. [Google Scholar] [CrossRef] [PubMed]
- Mayeaux, M.; Xu, Z.; King, J.M.; Prinyawiwatkul, W. Effects of cooking conditions on the lycopene content in tomatoes. J. Food Sci. 2006, 71, 461–464. [Google Scholar] [CrossRef]
- Turkmen, N.; Sari, F.; Velioglu, Y.S. The effect of cooking methods on total phenolics and antioxidant activity of selected green vegetables. Food Chem. 2005, 93, 713–718. [Google Scholar] [CrossRef]
- Shinde, U.A.; Phadke, A.S.; Nari, A.M.; Mungantiwar, A.A.; Dikshit, V.J.; Saraf, M.N. Membrane stabilization activity—A possible mechanism of action for the anti-inflammatory activity of Cedrus deodora wood oil. Fitoterapia 1999, 70, 251–257. [Google Scholar] [CrossRef]
- Gunathilake, K.D.P.P. Antioxidant and Anti-Inflammatory of Selected Green Leafy Vegetables in Sri Lanka. Ph.D. Thesis, University of Sri Jayewardenepura, Nugegoda, Sri Lanka, 2017. [Google Scholar]
- Okoli, C.O.; Akah, P.A.; Onuoha, N.J.; Okoye, T.C.; Nwoye, A.C.; Nworu, C.S. Acanthus montanus: An experimental evaluation of the antimicrobial, anti-inflammatory and immunological properties of a traditional remedy for furuncles. BMC Complement. Altern. Med. 2008, 8, 27. [Google Scholar] [CrossRef] [PubMed]
- Gambhire, M.; Juvekar, A.; Wankhede, S. Evaluation of the anti-inflammatory activity of methanol extract of Barleria cristata leaves by in vivo and in vitro methods. Int. J. Pharm. 2009, 7, 1–6. [Google Scholar]
- Wu, H. Affecting the activity of soybean lipoxygenase-1. J. Mol. Graph. 1996, 14, 331–337. [Google Scholar] [CrossRef]
- Gunathilake, K.D.P.P.; Rupasinghe, H.P.V. Optimization of water based-extraction methods for the preparation of bioactive-rich ginger extract using response surface methodology. Eur. J. Med. Plants 2014, 4, 893. [Google Scholar] [CrossRef]

| Parameters | r | P |
|---|---|---|
| Total phenolics versus protein denaturation | 0.646 | 0.001 |
| Total phenolics versus hemolysis | 0.294 | 0.024 |
| Total phenolics versus lipoxygenase activity | 0.558 | 0.000 |
| Total phenolics versus proteinase activity | 0.594 | 0.001 |
| Total flavonoids versus protein denaturation | 0.519 | 0.000 |
| Total flavonoids versus hemolysis | 0.444 | 0.000 |
| Total flavonoids versus lipoxygenase activity | 0.592 | 0.001 |
| Total flavonoids versus proteinase activity | 0.666 | 0.000 |
| Total carotenoids versus protein denaturation | 0.106 | 0.420 |
| Total carotenoids versus hemolysis | 0.203 | 0.120 |
| Total carotenoids versus lipoxygenase activity | 0.564 | 0.000 |
| Total carotenoids versus proteinase activity | 0.634 | 0.000 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gunathilake, K.D.P.P.; Ranaweera, K.K.D.S.; Rupasinghe, H.P.V. Influence of Boiling, Steaming and Frying of Selected Leafy Vegetables on the In Vitro Anti-inflammation Associated Biological Activities. Plants 2018, 7, 22. https://doi.org/10.3390/plants7010022
Gunathilake KDPP, Ranaweera KKDS, Rupasinghe HPV. Influence of Boiling, Steaming and Frying of Selected Leafy Vegetables on the In Vitro Anti-inflammation Associated Biological Activities. Plants. 2018; 7(1):22. https://doi.org/10.3390/plants7010022
Chicago/Turabian StyleGunathilake, K. D. P. P., K. K. D. S. Ranaweera, and H. P. V. Rupasinghe. 2018. "Influence of Boiling, Steaming and Frying of Selected Leafy Vegetables on the In Vitro Anti-inflammation Associated Biological Activities" Plants 7, no. 1: 22. https://doi.org/10.3390/plants7010022
APA StyleGunathilake, K. D. P. P., Ranaweera, K. K. D. S., & Rupasinghe, H. P. V. (2018). Influence of Boiling, Steaming and Frying of Selected Leafy Vegetables on the In Vitro Anti-inflammation Associated Biological Activities. Plants, 7(1), 22. https://doi.org/10.3390/plants7010022






