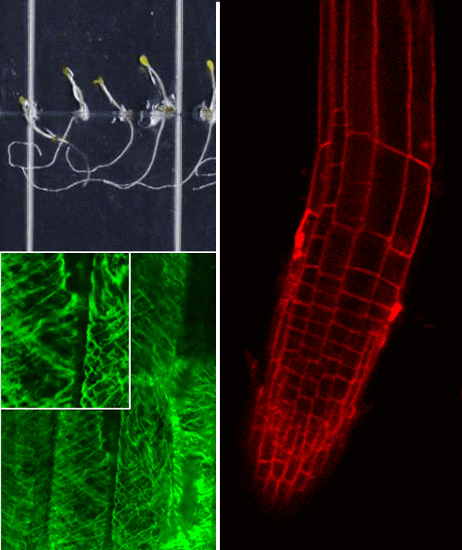
2.2. Growth Phenotypes of nhx1nhx2nhx3nhx4 and Responses to Na+, K+
We used the etiolation response of dark-grown seedlings to examine the contribution of
NHX1,
NHX2,
NHX3 and
NHX4 to cell expansion and seedling growth. Previously, we reported that etiolated
nhx1nhx2 double knockout seedlings had profound hypocotyl and root elongation phenotypes [
3]. Etiolated
nhx1nhx2nhx3nhx4 seedlings displayed significantly reduced growth of both roots and hypocotyls (
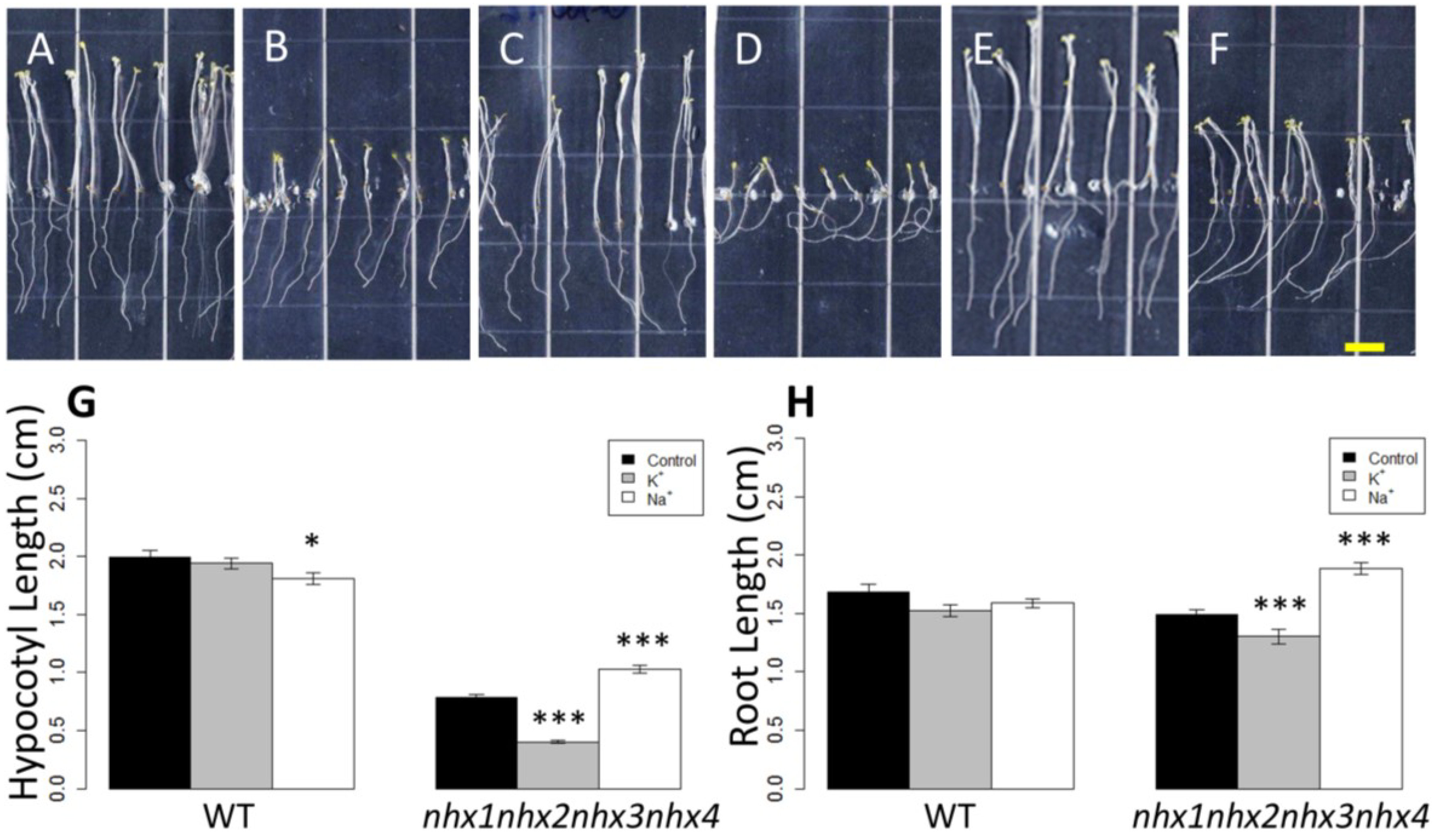
Figure 2). In control media, hypocotyl lengths of
nhx1nhx2nhx3nhx4 were less than 50% the length of WT after 11 days of etiolated growth (
Figure 2M). The hypocotyl elongation response of
nhx1nhx2nhx3nhx4 was highly responsive to both added Na
+ and K
+. For example, hypocotyls of
nhx1nhx2nhx3nhx4 grown in 30 mM NaCl were 31% longer than those of seedlings grown on control media (1 mM NaCl, 1 mM KCl), but hypocotyls of
nhx1nhx2nhx3nhx4 seedlings grown in 30 mM KCl were 49% shorter (
Figure 2D,H,K,L) than those grown in control media. By contrast, little change in hypocotyl length was observed in the WT seedlings grown on either 30 mM NaCl or KCl (
Figure 2B,F,J,L). Root growth was also markedly different between
nhx1nhx2nhx3nhx4 and WT vertically-grown seedlings, but only in response to either K
+ or Na
+. Similar to the K
+ and Na
+-dependent growth response of hypocotyls,
nhx1nhx2nhx3nhx4 roots were also significantly shorter (26%) under K
+ and longer (13%) under Na
+ as compared to
nhx1nhx2nhx3nhx4 grown in control media. In addition, the
nhx1nhx2nhx3nhx4 seedlings began to display prominent left-handed skewing (as viewed through the agar, following the convention assigned by Rutherford and Masson [
16]) after four days of growth on media supplemented with 30 mM K
+ (
Figure A2). Seedlings grown for another seven days on the same media displayed complete curling of roots (
Figure 2H). By contrast, WT root growth remained unchanged under 30 mM K
+ even after 11 days.
Figure 1.
Intracellular localization of NHX3 and NHX4. Transient co-expression, in Arabidopsis cotyledon epidermal cells, of 35S::NHX3-CFP (A) with 35S::NHX4-RFP (B) colocalized as shown in the merge fluorescent panel (C). Transient expression of 35S::NHX4-CFP (D) in stable 35S::NHX2-YFP background (E) also colocalized as shown in the merged image (F). Transient expression of 35S::NHX3-RFP (G) in stable expressing γTIP::γTIP-GFP (H) colocalized as shown in the merged panel (I). Transient expression of 35S::NHX4-CFP (J) in the stable expressing Ub10::VAMP711-RFP (K) colocalized as shown in the merged image (L). Arrowheads in (C,F,I,L) point to tonoplast invaginations and transvacuolar cytoplasmic strands.
Figure 1.
Intracellular localization of NHX3 and NHX4. Transient co-expression, in Arabidopsis cotyledon epidermal cells, of 35S::NHX3-CFP (A) with 35S::NHX4-RFP (B) colocalized as shown in the merge fluorescent panel (C). Transient expression of 35S::NHX4-CFP (D) in stable 35S::NHX2-YFP background (E) also colocalized as shown in the merged image (F). Transient expression of 35S::NHX3-RFP (G) in stable expressing γTIP::γTIP-GFP (H) colocalized as shown in the merged panel (I). Transient expression of 35S::NHX4-CFP (J) in the stable expressing Ub10::VAMP711-RFP (K) colocalized as shown in the merged image (L). Arrowheads in (C,F,I,L) point to tonoplast invaginations and transvacuolar cytoplasmic strands.
Figure 2.
Etiolation response of the knockout nhx1nhx2nhx3nhx4. Etiolated wild-type (A,C,E) and nhx1nhx2nhx3nhx4 (B,D,F) seedlings grown for 11 days in the dark on either 30 mM K+ (C,D) or 30 mM Na+ media (E,F). No significant difference in hypocotyl or root growth was noted in the wild-type; however, in nhx1nhx2nhx3nhx4, root and hypocotyl growth was significantly inhibited in seedlings grown in 30 mM K+ and increased in those grown in 30 mM Na+, as compared to control media. Quantification of hypocotyl (G) and root (H) growth normalized relative to growth on control media (1 mM Na+ and 1 mM K+). nhx1nhx2nhx3nhx4 displayed left-handed root skewing when grown on media containing 30 mM K+ (D), but not 1 mM K+ (B). Values represent the mean ± SE (n = 29). * p ≤ 0.05, *** p ≤ 0.001; p-values reflect significant differences between control and either K+ or Na+ treatments within each genotype. The yellow bar is 0.5 cm.
Figure 2.
Etiolation response of the knockout nhx1nhx2nhx3nhx4. Etiolated wild-type (A,C,E) and nhx1nhx2nhx3nhx4 (B,D,F) seedlings grown for 11 days in the dark on either 30 mM K+ (C,D) or 30 mM Na+ media (E,F). No significant difference in hypocotyl or root growth was noted in the wild-type; however, in nhx1nhx2nhx3nhx4, root and hypocotyl growth was significantly inhibited in seedlings grown in 30 mM K+ and increased in those grown in 30 mM Na+, as compared to control media. Quantification of hypocotyl (G) and root (H) growth normalized relative to growth on control media (1 mM Na+ and 1 mM K+). nhx1nhx2nhx3nhx4 displayed left-handed root skewing when grown on media containing 30 mM K+ (D), but not 1 mM K+ (B). Values represent the mean ± SE (n = 29). * p ≤ 0.05, *** p ≤ 0.001; p-values reflect significant differences between control and either K+ or Na+ treatments within each genotype. The yellow bar is 0.5 cm.
Previously, we reported that etiolated
nhx1nhx2 plants also displayed reduced hypocotyl growth, that was 70% the length of WT hypocotyls on media containing 1 mM K
+ and 50% of WT on 30 mM K
+ [
3], while in etiolated
nhx1nhx2nhx3nhx4 seedlings, the average hypocotyl length was 40% and 20% of WT on 1 and 30 mM K
+, respectively. A comparison of root growth responses between
nhx1nhx2nhx3nhx4 and
nhx1nhx2 to high K
+ [
3] indicates that root curling is also more pronounced and root growth more sensitive to high K
+, similar to that observed in hypocotyls. The dramatic differences in root and hypocotyl elongation between
nhx1nhx2 and
nhx1nhx2nhx3nhx4 under the higher K
+ suggest that NHX3 and NHX4 have additional roles in K
+ homeostasis and cell expansion.
To determine if the root curling observed in
nhx1nhx2nhx3nhx4 roots was specific to K
+, we compared the root growth of
nhx1nhx2nhx3nhx4 on media supplemented with KCl, KNO
3, NaCl or mannitol. Mannitol was included in order to determine whether the root curling response of
nhx1nhx2nhx3nhx4 under high K
+ was due to an osmotic effect; thus, 60 mM mannitol (60 mOsm, equivalent to the osmolarity of the added salt) was used. We found that
nhx1nhx2nhx3nhx4 root growth on mannitol was not different from the control (
Figure A3J,B), and therefore not likely due to an osmotic effect. As shown in
Figure A3, both 30 mM KCl and 30 mM KNO
3 supplemented media resulted in similar left-handed root skewing and curling of the root tip that was not seen when
nhx1nhx2nhx3nhx4 was grown on mannitol, suggesting that the response of
nhx1nhx2nhx3nhx4 was specific to K
+. Collectively, experiments comparing
nhx1nhx2nhx3nhx4 seedling growth (
i.e.,
Figure 2 and
Figure A3) indicate that the knockouts’ root curling response was caused by aberrant K
+ homeostasis.
Previous work has provided a strong link between ion stress and directional root growth. For example,
sos1(
nhx7) null mutants displayed a loss of normal gravitropic root growth when grown vertically on media supplemented with high (50–150 mM) Na
+, with young roots exhibiting skewing and eventual upward directed growth [
17]. Moreover, roots of both WT and
sos1(
nhx7) seedlings grown on high Na
+ showed delayed gravitropic responses, thereby suggesting that aberrant intracellular ion homeostasis can specifically affect directional root growth. The
sos1 skewing phenotype is remarkably similar to
nhx1nhx2nhx3nhx4 grown on K
+ supplemented media, and although effects of high cytosolic Na
+ have been suggested, the effects of K
+ on root skewing remain unclear. Furthermore,
sos1(
nhx7) mutants were more sensitive to microtubule reorganization, with whole mount immunostaining experiments in elongating root epidermal cells demonstrating that
sos1 (
nhx7) in the presence of 50 mM Na
+ had helically-orientated microtubules, while WT plants maintained the normal transverse array [
12]. Although microtubule remodeling has been shown to be induced by high external Na
+, reports of K
+-induced microtubule reorganization are sparse [
12,
13,
18]. In KCl (350 mM)-treated
Zea mays, a reorganization of the cortical microtubule array from the transverse to oblique orientation in root cells of the maturation zone occurred within 3 min after application. By contrast, treatment with sorbitol did not result in the reorganization of microtubules [
19]. These results suggested that cortical microtubule organization in roots was affected by high concentrations of Na
+ and K
+. Given the phenotypes of
nhx1nhx2nhx3nhx4, the knockout represents a useful tool to study the role of K
+ in cytoskeletal organization. The positive effect of Na
+ and the negative effect on K
+ on
nhx1nhx2nhx3nhx4 growth is well supported by similar results obtained in
nhx1nhx2 in which Na
+ was proposed to substitute for the lack of K
+ accumulation in vacuoles, providing the osmoticum needed to generate the turgor for cell expansion [
3,
10]. The sensitivity of both
nhx1nhx2 and
nhx1nhx2nhx3nhx4 to added K
+, as well as the aberrantly high cytosolic K
+ concentrations of
nhx1nhx2, together with previously reported effects of Na
+ on cytoskeleton disassembly and reorganization, suggests that aberrant K
+ homeostasis in the
nhx1nhx2nhx3nhx4 might affect the organization of the cytoskeleton.
2.4. Cells of the Root Transition Zone Lack Expansion in nhx1nhx2nhx3nhx4
To investigate the significance of NHX1 to NHX4-mediated Na
+ and K
+ transport on cell expansion and root skewing, we stained roots of
nhx1nhx2nhx3nhx4 seedlings with propidium iodide and visualized cell size. Compared with WT, cells in the
nhx1nhx2nhx3nhx4 root transition zone appeared to be much shorter when grown on 30 mM KCl-supplemented media (
Figure 3E,F). Cell size was similar between WT and
nhx1nhx2nhx3nhx4 in the apical meristem, but epidermal cells in the transition zone were markedly smaller in
nhx1nhx2nhx3nhx4. Whereas in the WT, a gradual increase in cell size from the meristem to the elongation zone (
Figure 3E, arrows) was apparent, the length of epidermal cells in the transition zone of
nhx1nhx2nhx3nhx4 was aberrantly small, and as a consequence, the transition zone appeared “compressed” (
Figure 3F, white arrows). This phenotype was not evident in
nhx1nhx2nhx3nhx4 grown on 30 mM NaCl-supplemented media, where no difference in cell size or shape was observed between WT and
nhx1nhx2nhx3nhx4 in any root zones (
Figure 3C,D).
Figure 3.
Root cell expansion in the nhx1nhx2nhx3nhx4 root transition zone is reduced. WT (A,C,E) or nhx1nhx2nhx3nhx4 (B,D,F) six-day-old dark-grown seedlings were stained with 10 µg·mL−1 propidium iodide. Seedlings were grown on control media (1 mM K+, 1 mM Na+) (A,B) or on media supplemented with 30 mM NaCl (C,D) or 30 mM KCl (E,F). The transition zone is bounded by the dashed lines. Arrowheads point to cells that show differences in cell size and are discussed in the Results. Bar is 50 μm.
Figure 3.
Root cell expansion in the nhx1nhx2nhx3nhx4 root transition zone is reduced. WT (A,C,E) or nhx1nhx2nhx3nhx4 (B,D,F) six-day-old dark-grown seedlings were stained with 10 µg·mL−1 propidium iodide. Seedlings were grown on control media (1 mM K+, 1 mM Na+) (A,B) or on media supplemented with 30 mM NaCl (C,D) or 30 mM KCl (E,F). The transition zone is bounded by the dashed lines. Arrowheads point to cells that show differences in cell size and are discussed in the Results. Bar is 50 μm.
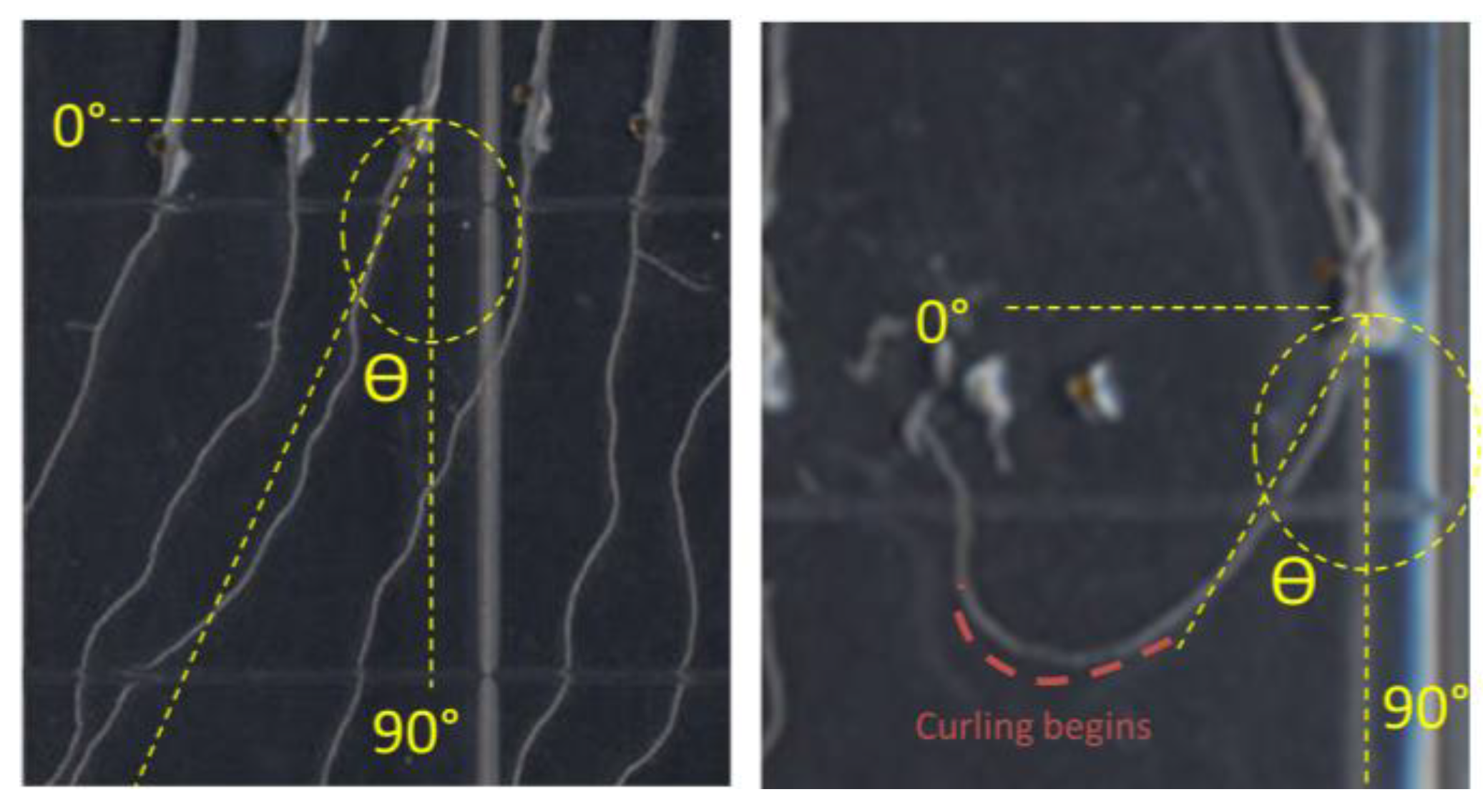
2.5. K+ Dependent Root Skewing Depends on the Dynamic Organization of the Cytoskeleton
In order to investigate whether the root growth response of
nhx1nhx2nhx3nhx4 to elevated K
+ concentrations was associated with dynamic changes in the organization of the cytoskeleton, we tested the effect of microtubule perturbing drugs on root morphology and directional root growth in etiolated
nhx1nhx2nhx3nhx4 seedlings as above. Propyzamide is a microtubule destabilizing drug that is known to induce right-handed root skewing (when viewed through the agar) without inhibiting overall growth and root elongation [
12,
20,
21]. When applied at 3 μM, anisotropic root growth of wild-type
Arabidopsis is moderately impaired without causing swelling of epidermal roots cells [
22]. We measured root growth angles for both WT and
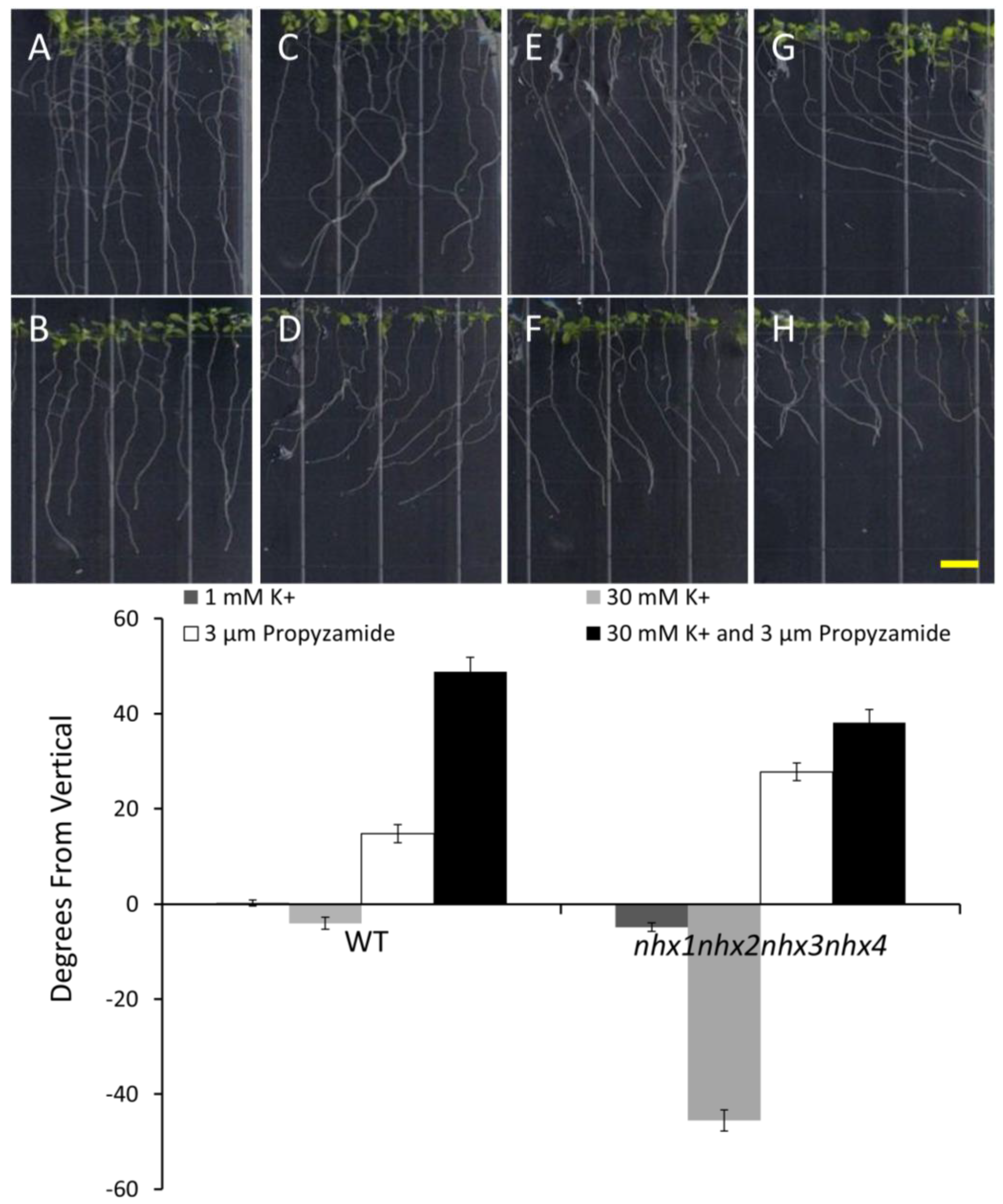
nhx1nhx2nhx3nhx4 vertically-grown seedlings on minimal media (containing 1 mM KCl) and an identical media supplemented with an additional 30 mM KCl. As shown in
Figure 4, both the WT and
nhx1nhx2nhx3nhx4 displayed the characteristic right-handed skewing (
Figure 4E,F) when grown in the presence of 3 µM propyzamide, but only
nhx1nhx2nhx3nhx4 displayed left-handed skewing in response to 30 mM K
+ (
Figure 4D). When both elevated K
+ and 3 µM propyzamide were included in the media, roots from WT seedlings continued to skew towards the right, unlike the roots from
nhx1nhx2nhx3nhx4, which displayed less pronounced skewing (
Figure 4G,H).
Figure 4.
Propyzamide alters potassium-induced root skewing in the nhx1nhx2nhx3nhx4 knockout. Wild-type (A,C,E,G) and nhx1nhx2nhx3nhx4 (B,D,F,H) seedlings were germinated on minimal media and transferred to plates with either 30 mM K+ (C,D), 3 µM propyzamide (E,F) or both 30 mM K+ and propyzamide (G,H) when four day-old and grown for an additional seven days. Root skewing is quantified as the angle of root growth in degrees from vertical (I). Values represent the mean ± SE (n = 28). The bar is 0.5 cm.
Figure 4.
Propyzamide alters potassium-induced root skewing in the nhx1nhx2nhx3nhx4 knockout. Wild-type (A,C,E,G) and nhx1nhx2nhx3nhx4 (B,D,F,H) seedlings were germinated on minimal media and transferred to plates with either 30 mM K+ (C,D), 3 µM propyzamide (E,F) or both 30 mM K+ and propyzamide (G,H) when four day-old and grown for an additional seven days. Root skewing is quantified as the angle of root growth in degrees from vertical (I). Values represent the mean ± SE (n = 28). The bar is 0.5 cm.
Wang and colleagues demonstrated that WT and
sos1(
nhx7) seedlings exhibited left-handed root skewing when grown vertically on NaCl-enriched MS media, similar to the root skewing of
nhx1nhx2nhx3nhx4 in the presence of high KCl. Their results indicated that 50 mM Na
+ was sufficient to initiate root skewing that eventually resulting in strong root curling and the formation of root “loops”. Interestingly, Na
+-dependent root skewing was exacerbated in
sos1(
nhx7) mutants, which displayed more prominent root skewing than the WT at lower Na
+ treatments, suggesting that cytosolic ion accumulation played an important role in directional root growth. NaCl-induced salt stress resulted in seedling death, which occurred more frequently in
sos1(
nhx7) than WT seedlings, but was partially prevented by propyzamide treatment, suggesting that microtubule reorganization was required for salt tolerance [
18].
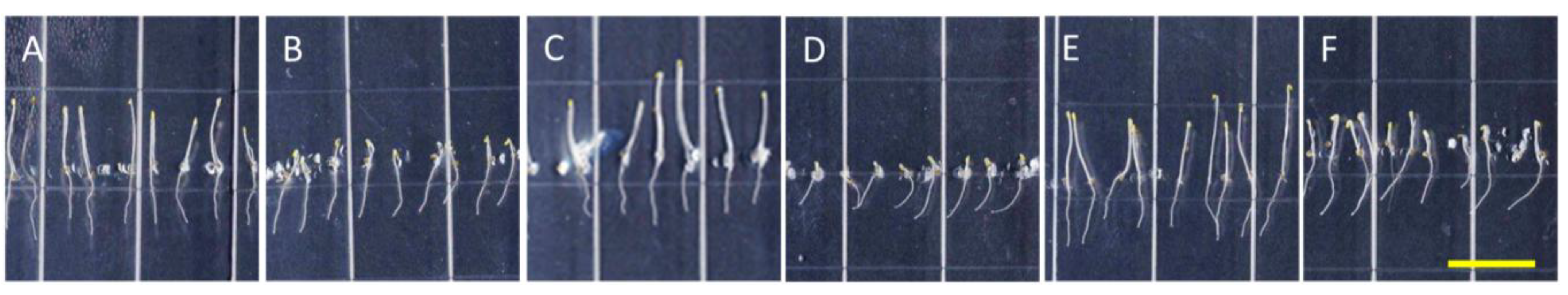
We also tested the effect of Taxol, a microtubule stabilizing drug, on K
+-induced root skewing in
nhx1nhx2nhx3nhx4 plants
. A characteristic effect of Taxol on roots is a right-handed skewing response that is dose dependent [
23]. We observed that 0.5 µM Taxol was sufficient to induce right-handed root skewing in the WT (
Figure 5E), but the same concentration did not significantly change the root growth direction of
nhx1nhx2nhx3nhx4 plants (
Figure 5F). It is unlikely that the lack of
nhx1nhx2nhx3nhx4 root skewing observed in the 0.5 µM Taxol treatment was caused by a lack of overall root growth, because
nhx1nhx2nhx3nhx4 roots did not vary significantly in length between 0.5 µM or 1.0 µM Taxol treatments. Indeed, the addition of 30 mM KCl was sufficient to induce left-handed skewing in
nhx1nhx2nhx3nhx4, despite the presence of 0.5 µM Taxol (
Figure 5J). In contrast, the added KCl had no effect on WT root growth, with seedlings continuing to skew to the right (
Figure 5I). We observed that a threshold of 1.0 µM Taxol was sufficient to overcome the K
+-induced left-handed skewing in
nhx1nhx2nhx3nhx4 plants and to cause right-handed skewing, as shown (
Figure 5L). Previously, it was shown that 0.5 μM Taxol counters the effect of Na
+-induced root skewing, which is dose dependent [
18]; a result that is strikingly similar to what we observed in Taxol- and K
+-treated
nhx1nhx2nhx3nhx4 (
Figure 5).
Figure 5.
Taxol reverses directional root growth in nhx1nhx2nhx3nhx4 grown on potassium-supplemented media. Wild-type (top) and nhx1nhx2nhx3nhx4 (bottom) seedlings were germinated on minimal media containing 1 mM K+ for three days and transferred to plates containing two concentrations of Taxol and K+ (indicated below) and grown for an additional four days. (A,B) 1 mM K+; (C,D) 30 mM K+; (E,F) 0.5 µM Taxol and 1 mM K+; (G,H) 1 µM Taxol and 1 mM K+; (I,J) 0.5 µM Taxol and 30 mM K+; (K,L) 1 µM Taxol and 30 mM K+. (M) The angle of root growth was quantified as the degrees from vertical; values represent the mean ± SE (n = 24). The bar is 0.5 cm.
Figure 5.
Taxol reverses directional root growth in nhx1nhx2nhx3nhx4 grown on potassium-supplemented media. Wild-type (top) and nhx1nhx2nhx3nhx4 (bottom) seedlings were germinated on minimal media containing 1 mM K+ for three days and transferred to plates containing two concentrations of Taxol and K+ (indicated below) and grown for an additional four days. (A,B) 1 mM K+; (C,D) 30 mM K+; (E,F) 0.5 µM Taxol and 1 mM K+; (G,H) 1 µM Taxol and 1 mM K+; (I,J) 0.5 µM Taxol and 30 mM K+; (K,L) 1 µM Taxol and 30 mM K+. (M) The angle of root growth was quantified as the degrees from vertical; values represent the mean ± SE (n = 24). The bar is 0.5 cm.
To better understand the possible effect of high K+ on cytoskeletal organization, we used a whole mount immunostaining approach to visualize the organization of cortical microtubules in cells of the root elongation zone, where pronounced skewing and curling occurred.
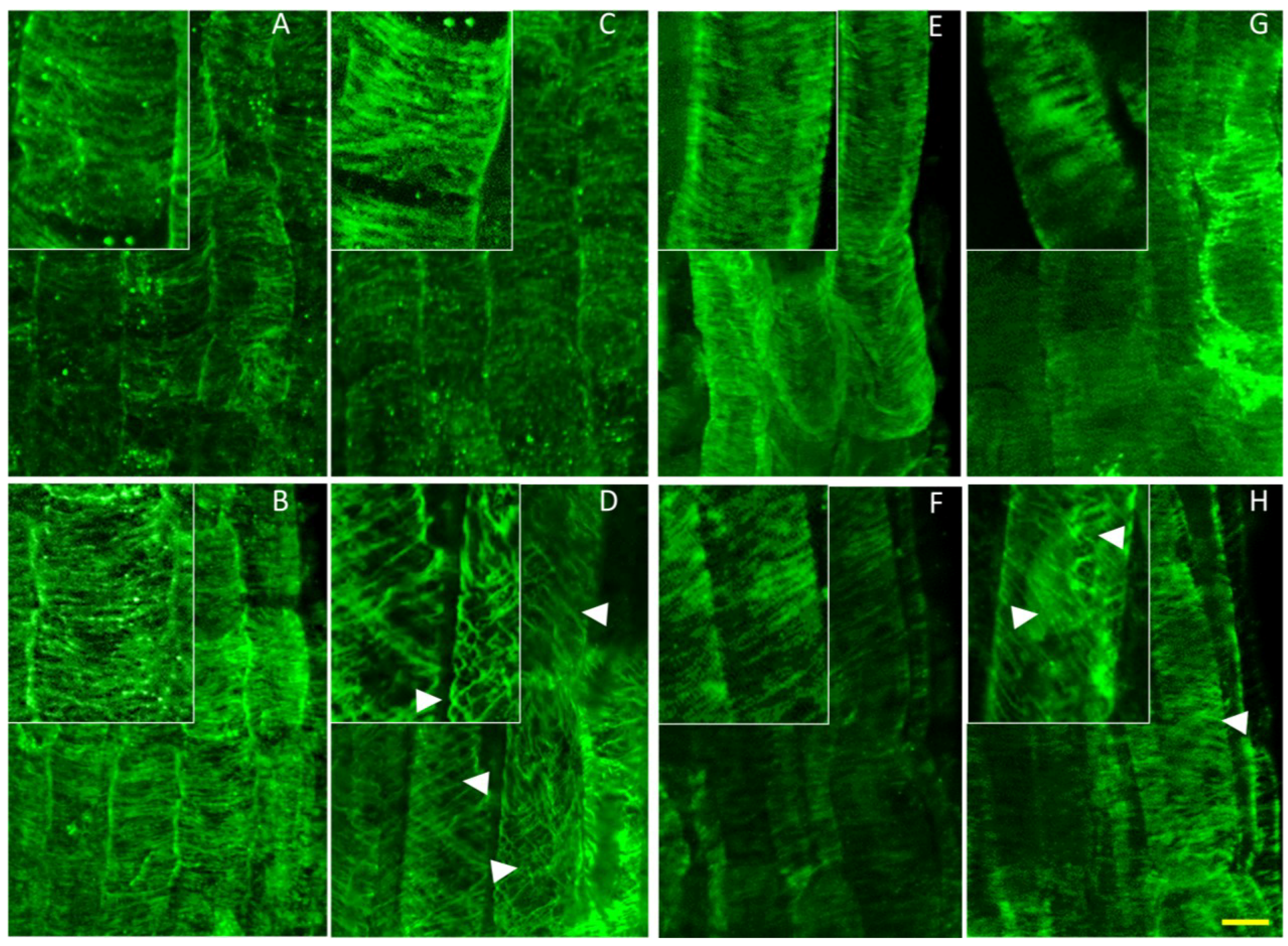
As depicted in
Figure 6, no differences were observed between the microtubule organization of roots from
nhx1nhx2nhx3nhx4 and the WT when grown on media containing 1 mM K
+ (
Figure 6A,B). By contrast,
nhx1nhx2nhx3nhx4 seedlings grown on media containing 30 mM K
+ displayed a helical orientation of the cortical microtubule array in elongating
nhx1nhx2nhx3nhx4 root cells (
Figure 6D) that was observed only when roots exhibited left-handed skewing. Both WT and
nhx1nhx2nhx3nhx4 displayed the characteristic Taxol-induced “bundling” of microtubules [
24,
25] with a highly parallel microtubule organization (
Figure 6E,F). However, when seedlings were grown on media with both 0.5 µM Taxol and 30 mM KCl, the
nhx1nhx2nhx3nhx4 microtubules displayed a similar spiraling pattern that was seen when grown on 30 mM KCl plates (
Figure 6H,D). Higher K
+ had no observable effect on microtubule organization in the WT, even in the presence of Taxol (
Figure 6C,G).
Figure 6.
Potassium induces helical reorganization of cortical microtubules of nhx1nhx2nhx3nhx4 epidermal cells in the root elongation zone. Immunolabeled microtubules in WT (top) and nhx1nhx2nhx3nhx4 (bottom). Seedlings were germinated on minimal media containing 1 mM K+ for three days, then transferred to plates with two concentrations of Taxol and grown for an additional five days before fixation. Normal microtubule orientation is observed in WT (A) and nhx1nhx2nhx3nhx4 (B) when grown on media containing 1 mM K+. WT seedlings grown on 30 mM K+ displayed normally-orientated microtubules (C), whereas 30 mM K+ induced a helical orientation of the cortical microtubules in nhx1nhx2nhx3nhx4 (D). In the presence of 1 mM K+ and 0.5 µM Taxol, both WT (E) and nhx1nhx2nhx3nhx4 (F) display characteristic bundling of cortical microtubules. Whereas 30 mM K+ had no effect on microtubule orientation in WT grown with 0.5 µM Taxol (G), nhx1nhx2nhx3nhx4 seedlings grown with 30 mM K+ and 0.5 µM Taxol (H) exhibited a helical microtubule organization similar to that seen when grown with just 30 mM K+. Arrows point to helically-oriented microtubules. The bar is 10 μM. The upper left insets are enlarged 50% to highlight microtubule orientation.
Figure 6.
Potassium induces helical reorganization of cortical microtubules of nhx1nhx2nhx3nhx4 epidermal cells in the root elongation zone. Immunolabeled microtubules in WT (top) and nhx1nhx2nhx3nhx4 (bottom). Seedlings were germinated on minimal media containing 1 mM K+ for three days, then transferred to plates with two concentrations of Taxol and grown for an additional five days before fixation. Normal microtubule orientation is observed in WT (A) and nhx1nhx2nhx3nhx4 (B) when grown on media containing 1 mM K+. WT seedlings grown on 30 mM K+ displayed normally-orientated microtubules (C), whereas 30 mM K+ induced a helical orientation of the cortical microtubules in nhx1nhx2nhx3nhx4 (D). In the presence of 1 mM K+ and 0.5 µM Taxol, both WT (E) and nhx1nhx2nhx3nhx4 (F) display characteristic bundling of cortical microtubules. Whereas 30 mM K+ had no effect on microtubule orientation in WT grown with 0.5 µM Taxol (G), nhx1nhx2nhx3nhx4 seedlings grown with 30 mM K+ and 0.5 µM Taxol (H) exhibited a helical microtubule organization similar to that seen when grown with just 30 mM K+. Arrows point to helically-oriented microtubules. The bar is 10 μM. The upper left insets are enlarged 50% to highlight microtubule orientation.
![]()
In particular, cortical microtubules depolymerize and reorganize in a Ca
2+-dependent manner under salt stress; preventing this response with microtubule stabilizing drugs, such as Taxol, results in seedling death [
12,
13], suggesting that cytoskeletal reorganization of root cells is important during salt stress. In addition, mutations of the Na
+/H
+ antiporter SOS1 and associated SOS2 kinase were shown to suppress the cortical microtubule disruptions and subsequent helical growth associated with the
Arabidopsis spiral1 mutants. This study also demonstrated that even in the absence of salt stress, loss of function SOS mutants resulted in altered cortical microtubule arrays when seedlings were treated with the same dose of propyzamide as used in the present study [
12].