Temporal Changes in Biochemical Responses to Salt Stress in Three Salicornia Species
Abstract
1. Introduction
2. Results
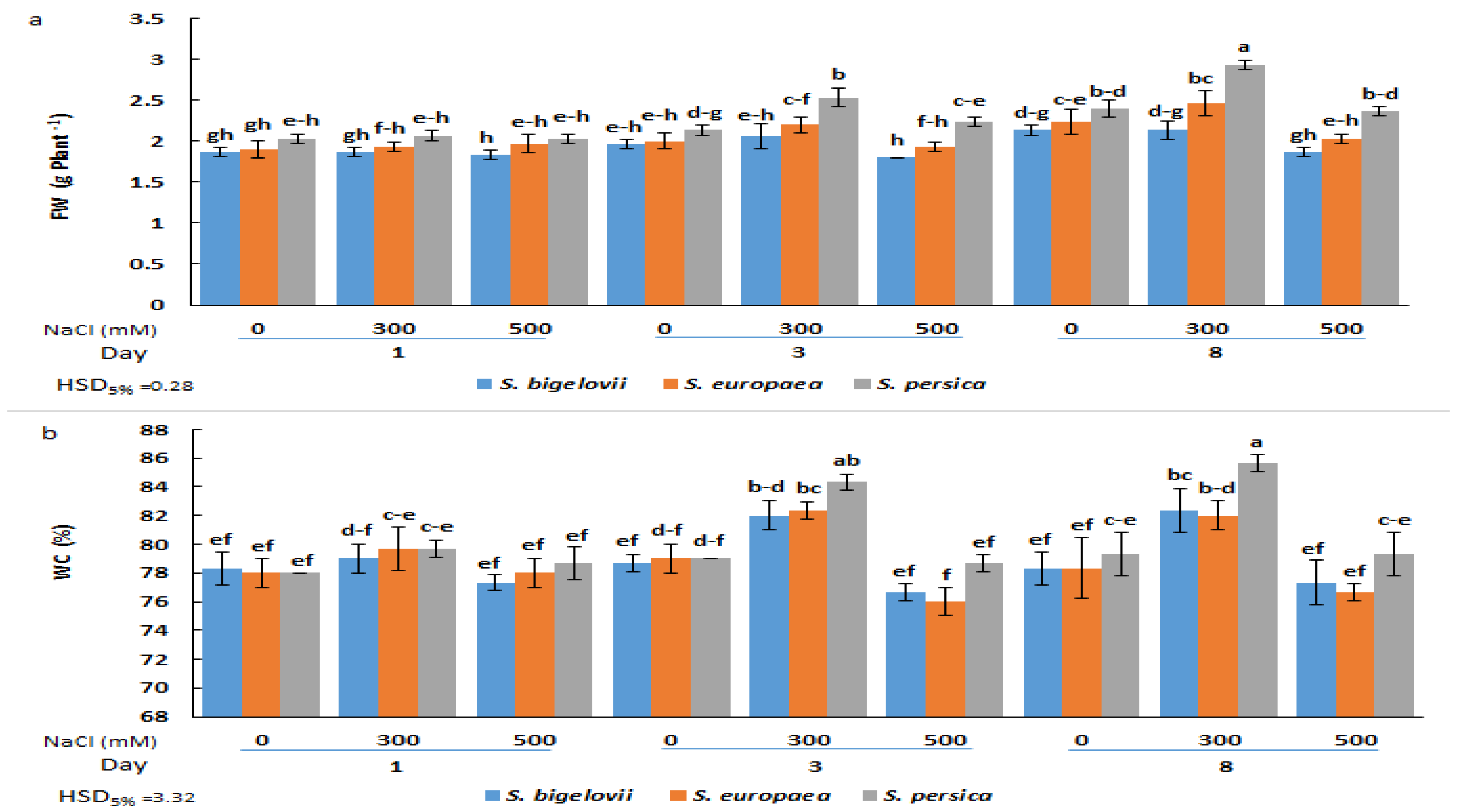
2.1. Growth Parameters
2.2. Photosynthetic Pigments Contents
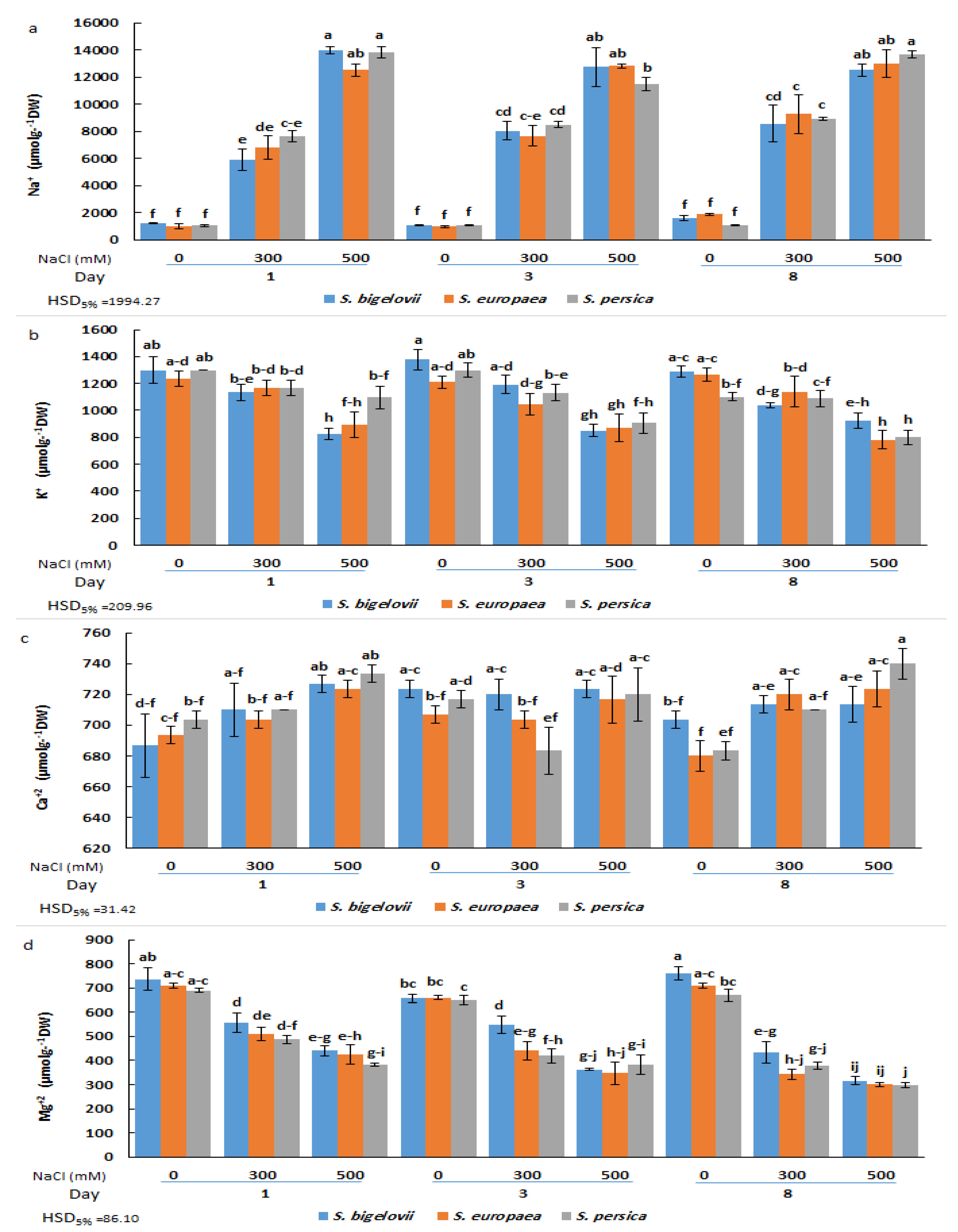
2.3. Cation Accumulation
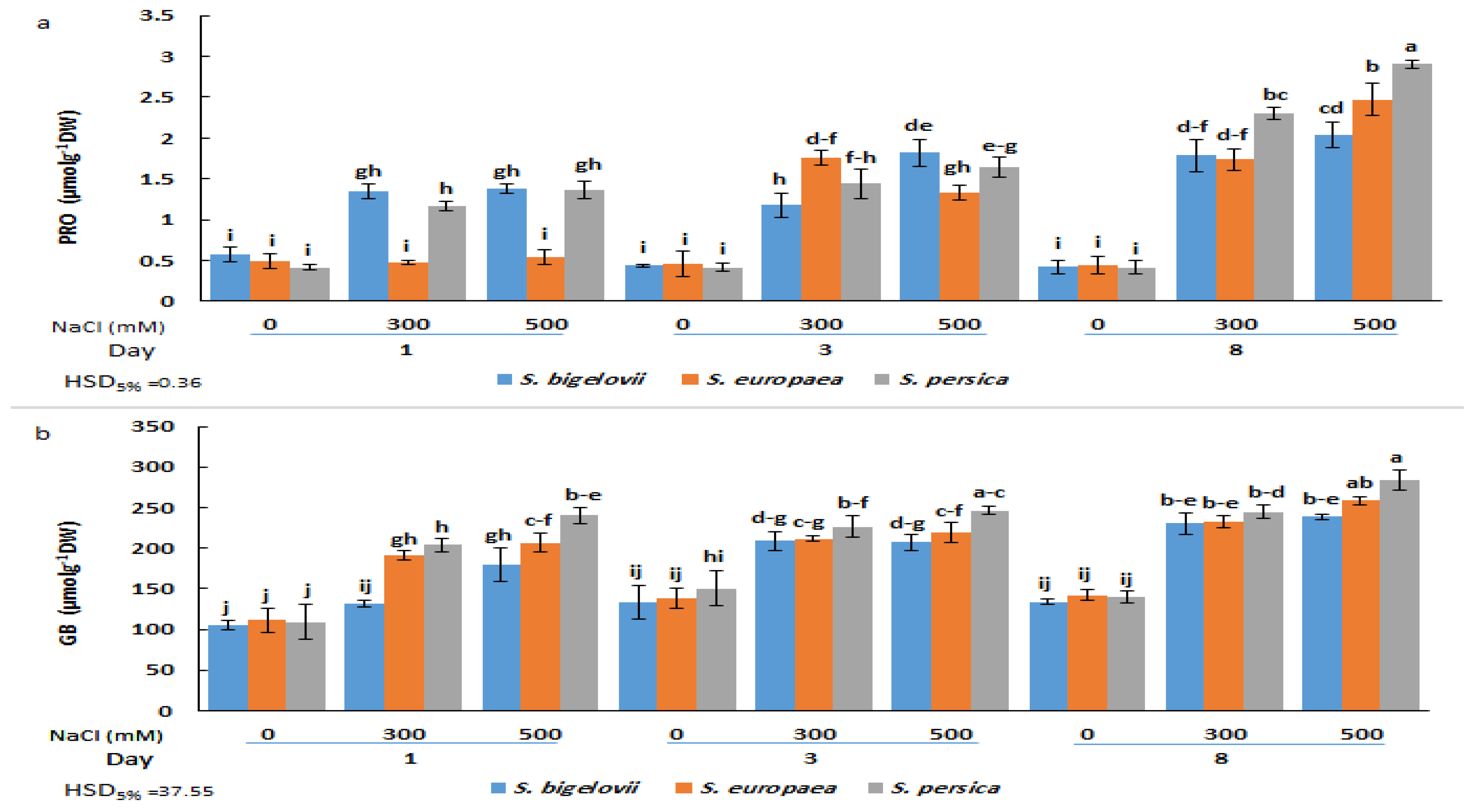
2.4. Shoot Osmolytes Contents
2.5. Oxidative Stress Markers
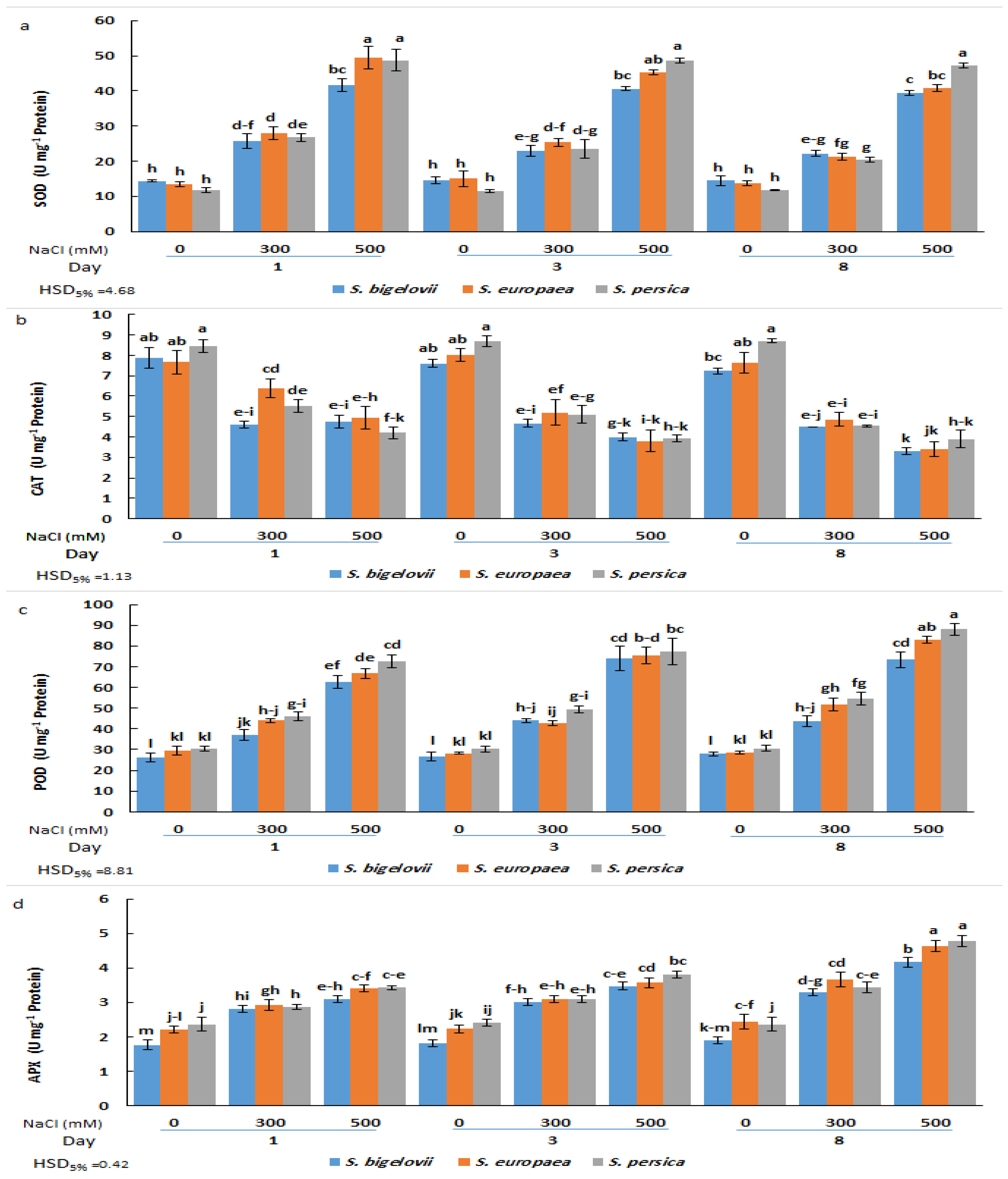
2.6. Antioxidant Enzymes Activities
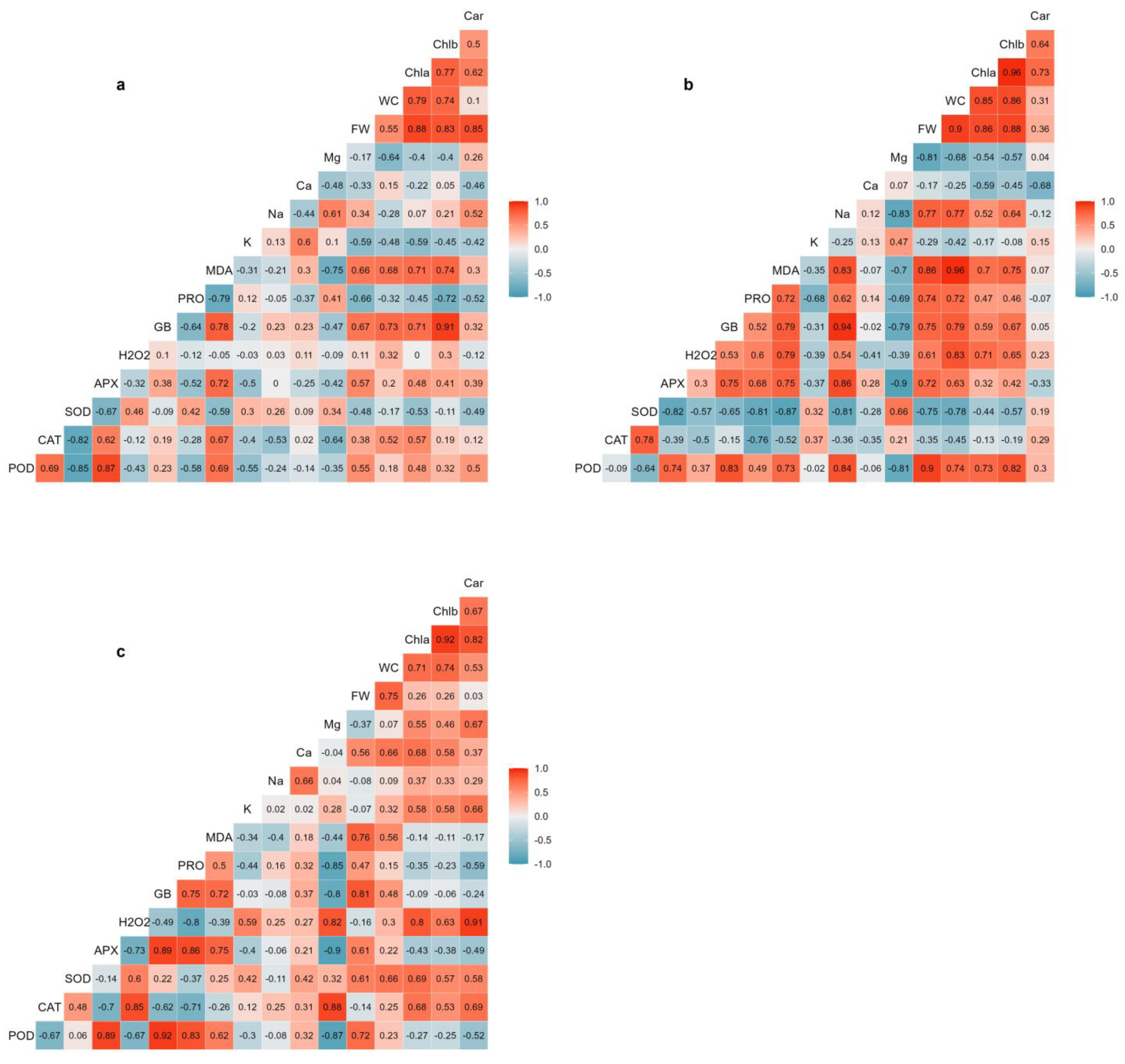
2.7. Correlation Analysis
2.8. Principal Component Analysis
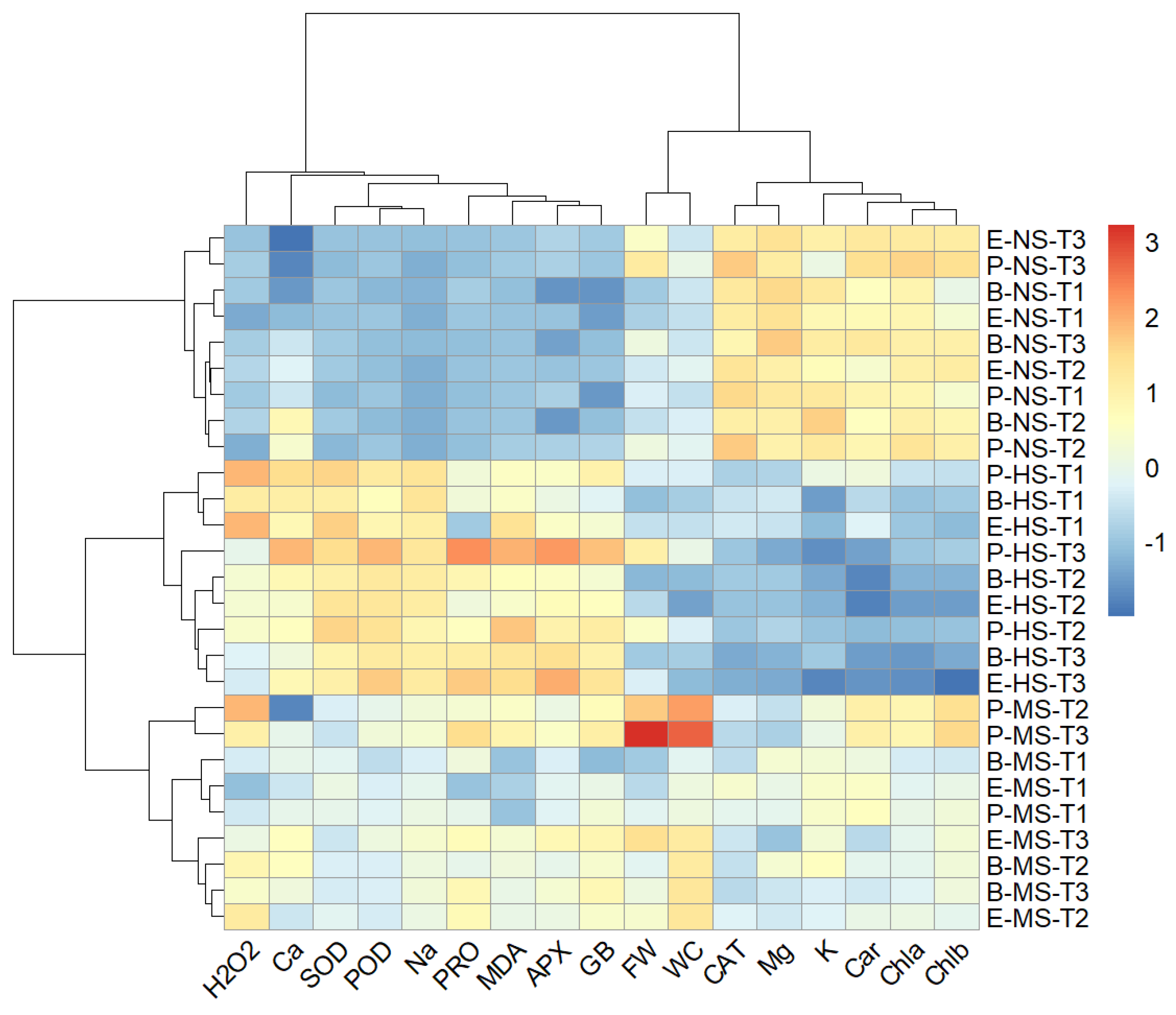
2.9. Hierarchical Clustering
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Growth Parameters
4.3. Photosynthetic Pigments
4.4. Quantification of Cations
4.5. Proline and Glycine Betaine
4.6. MDA and H2O2
4.7. Antioxidant Enzyme Assays
4.8. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hopmans, J.W.; Qureshi, A.; Kisekka, I.; Munns, R.; Grattan, S.; Rengasamy, P.; Ben-Gal, A.; Assouline, S.; Javaux, M.; Minhas, P. Critical knowledge gaps and research priorities in global soil salinity. Adv. Agron. 2021, 169, 1–191. [Google Scholar] [CrossRef]
- Morton, M.J.; Awlia, M.; Al-Tamimi, N.; Saade, S.; Pailles, Y.; Negrão, S.; Tester, M. Salt stress under the scalpel–dissecting the genetics of salt tolerance. Plant J. 2019, 97, 148–163. [Google Scholar] [CrossRef] [PubMed]
- Accogli, R.; Tomaselli, V.; Direnzo, P.; Perrino, E.V.; Albanese, G.; Urbano, M.; Laghetti, G. Edible halophytes and halo-tolerant species in Apulia region (Southeastern Italy): Biogeography, traditional food use and potential sustainable crops. Plants 2023, 12, 549. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas-Pérez, S.; Piernik, A.; Chanona-Pérez, J.; Grigore, M.; Perea-Flores, M. An overview of the emerging trends of the Salicornia L. genus as a sustainable crop. Environ. Exp. Bot. 2021, 191, 104606. [Google Scholar] [CrossRef]
- Arya, S.S.; Devi, S.; Ram, K.; Kumar, S.; Kumar, N.; Mann, A.; Kumar, A.; Chand, G. Halophytes: The plants of therapeutic medicine. In Ecophysiology, Abiotic Stress Responses and Utilization of Halophytes; Springer: Singapore, 2019; pp. 271–287. [Google Scholar] [CrossRef]
- Pérez Cuadra, V.; Verolo, M.; Cambi, V. Morphological and anatomical traits of halophytes: Adaptive versus phylogenetic value. In Handbook of Halophytes: From Molecules to Ecosystems towards Biosaline Agriculture; Springer: Cham, Switzerland, 2020; pp. 1–20. [Google Scholar]
- Singh, D.; Buhmann, A.K.; Flowers, T.J.; Seal, C.E.; Papenbrock, J. Salicornia as a crop plant in temperate regions: Selection of genetically characterized ecotypes and optimization of their cultivation conditions. AoB Plants 2014, 6, plu071. [Google Scholar] [CrossRef]
- Patel, K.K. The new deal. In The New Deal; Princeton University Press: Princeton, NJ, USA, 2016. [Google Scholar] [CrossRef]
- Nakahara, Y.; Sawabe, S.; Kainuma, K.; Katsuhara, M.; Shibasaka, M.; Suzuki, M.; Yamamoto, K.; Oguri, S.; Sakamoto, H. Yeast functional screen to identify genes conferring salt stress tolerance in Salicornia europaea. Front. Plant Sci. 2015, 6, 920. [Google Scholar] [CrossRef]
- Aliakbari, M.; Razi, H.; Alemzadeh, A.; Tavakol, E. RNA-seq transcriptome profiling of the halophyte Salicornia persica in response to salinity. J. Plant Growth Regul. 2021, 40, 707–721. [Google Scholar] [CrossRef]
- Yadav, N.S.; Rashmi, D.; Singh, D.; Agarwal, P.K.; Jha, B. A novel salt-inducible gene SbSI-1 from Salicornia brachiata confers salt and desiccation tolerance in E. coli. Mol. Biol. Rep. 2012, 39, 1943–1948. [Google Scholar] [CrossRef]
- Duan, H.; Tiika, R.J.; Tian, F.; Lu, Y.; Zhang, Q.; Hu, Y.; Cui, G.; Yang, H. Metabolomics analysis unveils important changes involved in the salt tolerance of Salicornia europaea. Front. Plant Sci. 2023, 13, 1097076. [Google Scholar] [CrossRef]
- Grigore, M.-N.; Vicente, O. Wild Halophytes: Tools for understanding salt tolerance mechanisms of plants and for adapting agriculture to climate change. Plants 2023, 12, 221. [Google Scholar] [CrossRef]
- Asadullah; Bano, A. Climate change modulates halophyte secondary metabolites to reshape rhizosphere halobacteria for biosaline agriculture. Appl. Sci. 2023, 13, 1299. [Google Scholar] [CrossRef]
- Koca, H.; Bor, M.; Özdemir, F.; Türkan, İ. The effect of salt stress on lipid peroxidation, antioxidative enzymes and proline content of sesame cultivars. Environ. Exp. Bot. 2007, 60, 344–351. [Google Scholar] [CrossRef]
- Aghaleh, M.; Niknam, V.; Ebrahimzadeh, H.; Razavi, K. Effect of salt stress on physiological and antioxidative responses in two species of Salicornia (S. persica and S. europaea). Acta Physiol. Plant 2011, 33, 1261–1270. [Google Scholar] [CrossRef]
- Aghaleh, M.; Niknam, V.; Ebrahimzadeh, H.; Razavi, K. Antioxidative enzymes in two in vitro cultured Salicornia species in response to increasing salinity. Biol. Plant 2014, 58, 391–394. [Google Scholar] [CrossRef]
- Parida, A.K.; Jha, B. Salt tolerance mechanisms in mangroves: A review. Trees 2010, 24, 199–217. [Google Scholar] [CrossRef]
- Cárdenas-Pérez, S.; Rajabi Dehnavi, A.; Leszczyński, K.; Lubińska-Mielińska, S.; Ludwiczak, A.; Piernik, A. Salicornia europaea L. functional traits indicate its optimum growth. Plants 2022, 11, 1051. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, A.-M.F.; Mohamed, E.; Kasem, A.M.; El-Ghamery, A.A. Differential salt tolerance strategies in three halophytes from the same ecological habitat: Augmentation of antioxidant enzymes and compounds. Plants 2021, 10, 1100. [Google Scholar] [CrossRef] [PubMed]
- Calone, R.; Mircea, D.-M.; González-Orenga, S.; Boscaiu, M.; Lambertini, C.; Barbanti, L.; Vicente, O. Recovery from salinity and drought stress in the perennial Sarcocornia fruticosa vs. the annual Salicornia europaea and S. veneta. Plants 2022, 11, 1058. [Google Scholar] [CrossRef] [PubMed]
- Aghaleh, M.; Niknam, V.; Ebrahimzadeh, H.; Razavi, K. Salt stress effects on growth, pigments, proteins and lipid peroxidation in Salicornia persica and S. europaea. Biol. Plant 2009, 53, 243–248. [Google Scholar] [CrossRef]
- Mishra, A.; Tanna, B. Halophytes: Potential resources for salt stress tolerance genes and promoters. Front. Plant Sci. 2017, 8, 829. [Google Scholar] [CrossRef]
- Suriyan, C.-u.; Chalermpol, K. Proline accumulation, photosynthetic abilities and growth characters of sugarcane (Saccharum officinarum L.) plantlets in response to iso-osmotic salt and water-deficit stress. Agric. Sci. China 2009, 8, 51–58. [Google Scholar] [CrossRef]
- Mansour, M.M.F.; Ali, E.F. Glycinebetaine in saline conditions: An assessment of the current state of knowledge. Acta Physiol. Plant 2017, 39, 1–17. [Google Scholar] [CrossRef]
- Siddiqui, S.A.; Kumari, A.; Rathore, M.S. Glycine betaine as a major osmolyte under abiotic stress in halophytes. In Handbook of Halophytes: From Molecules to Ecosystems towards Biosaline Agriculture; Springer: Cham, Switzerland, 2021; pp. 2069–2087. [Google Scholar] [CrossRef]
- Moghaieb, R.E.; Saneoka, H.; Fujita, K. Effect of salinity on osmotic adjustment, glycinebetaine accumulation and the betaine aldehyde dehydrogenase gene expression in two halophytic plants, Salicornia europaea and Suaeda maritima. Plant Sci. 2004, 166, 1345–1349. [Google Scholar] [CrossRef]
- Ahmad, S.T.; Sima, N.A.K.K.; Mirzaei, H.H. Effects of sodium chloride on physiological aspects of Salicornia persica growth. J. Plant Nutr. 2013, 36, 401–414. [Google Scholar] [CrossRef]
- Shabala, S.; Mackay, A. Ion transport in halophytes. In Advances in Botanical Research; Elsevier: Amsterdam, The Netherlands, 2011; Volume 57, pp. 151–199. [Google Scholar] [CrossRef]
- Amiri, B.; Assareh, M.; Rasouli, B.; Jafari, M.; Arzani, H.; Jafari, A. Effect of salinity on growth, ion content and water status of glasswort (Salicornia herbacea L.). Casp. J. Environ. Sci. 2010, 8, 79–87. [Google Scholar]
- Lee, S.J.; Jeong, E.-M.; Ki, A.Y.; Oh, K.-S.; Kwon, J.; Jeong, J.-H.; Chung, N.-J. Oxidative defense metabolites induced by salinity stress in roots of Salicornia herbacea. J. Plant Physiol. 2016, 206, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Akhani, H. Salicornia persica Akhani (Chenopodiaceae), a remarkable new species from Central Iran. Linz. Biol. Beitr. 2003, 35, 607–612. [Google Scholar]
- Szabados, L.; Savouré, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2010, 15, 89–97. [Google Scholar] [CrossRef]
- Arif, Y.; Singh, P.; Siddiqui, H.; Bajguz, A.; Hayat, S. Salinity induced physiological and biochemical changes in plants: An omic approach towards salt stress tolerance. Plant Physiol. Biochem. 2020, 156, 64–77. [Google Scholar] [CrossRef]
- Flowers, T.J.; Munns, R.; Colmer, T.D. Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Ann. Bot. 2015, 115, 419–431. [Google Scholar] [CrossRef]
- Hernandez, M.; Fernandez-Garcia, N.; Diaz-Vivancos, P.; Olmos, E. A different role for hydrogen peroxide and the antioxidative system under short and long salt stress in Brassica oleracea roots. J. Exp. Bot 2010, 61, 521–535. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; He, S.; Sun, J.; Pan, Z.; Gong, W.; Lu, Y.; Du, X. Na+ compartmentalization related to salinity stress tolerance in upland cotton (Gossypium hirsutum) seedlings. Sci. Rep. 2016, 6, 34548. [Google Scholar] [CrossRef] [PubMed]
- Hsouna, A.B.; Ghneim-Herrera, T.; Romdhane, W.B.; Dabbous, A.; Saad, R.B.; Brini, F.; Abdelly, C.; Hamed, K.B. Early effects of salt stress on the physiological and oxidative status of the halophyte Lobularia maritima. Funct. Plant Biol. 2020, 47, 912–924. [Google Scholar] [CrossRef] [PubMed]
- Percey, W.J.; Shabala, L.; Wu, Q.; Su, N.; Breadmore, M.C.; Guijt, R.M.; Bose, J.; Shabala, S. Potassium retention in leaf mesophyll as an element of salinity tissue tolerance in halophytes. Plant Physiol. Biochem. 2016, 109, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Kefu, Z.; Hai, F.; San, Z.; Jie, S. Study on the salt and drought tolerance of Suaeda salsa and Kalanchoe claigremontiana under iso-osmotic salt and water stress. Plant Sci. 2003, 165, 837–844. [Google Scholar] [CrossRef]
- Parida, A.K.; Das, A.; Mittra, B. Effects of salt on growth, ion accumulation, photosynthesis and leaf anatomy of the mangrove, Bruguiera parviflora. Trees 2004, 18, 167–174. [Google Scholar] [CrossRef]
- Greenway, H.; Munns, R. Mechanisms of salt tolerance in nonhalophytes. Annu. Rev. Plant Physiol. 1980, 31, 149–190. [Google Scholar] [CrossRef]
- Flowers, T.; Hajibagheri, M.; Clipson, N. Halophytes. Q. Rev. Biol. 1986, 61, 313–337. [Google Scholar] [CrossRef]
- Julkowska, M.M.; Testerink, C. Tuning plant signaling and growth to survive salt. Trends Plant Sci. 2015, 20, 586–594. [Google Scholar] [CrossRef]
- Khan, M.A.; Gul, B.; Weber, D.J. Effect of salinity on the growth and ion content of Salicornia rubra. Commun. Soil Sci. Plant Anal. 2001, 32, 2965–2977. [Google Scholar] [CrossRef]
- Khan, M.A.; Ungar, I.A.; Showalter, A.M. The effect of salinity on the growth, water status, and ion content of a leaf succulent perennial halophyte, Suaeda fruticosa (L.) Forssk. J. Arid Environ. 2000, 45, 73–84. [Google Scholar] [CrossRef]
- Rhodes, D.; Hanson, A. Quaternary ammonium and tertiary sulfonium compounds in higher plants. Annu. Rev. Plant Biol. 1993, 44, 357–384. [Google Scholar] [CrossRef]
- Akcin, A.; Yalcin, E. Effect of salinity stress on chlorophyll, carotenoid content, and proline in Salicornia prostrata Pall. and Suaeda prostrata Pall. subsp. prostrata (Amaranthaceae). Rev. Bras. Bot. 2016, 39, 101–106. [Google Scholar] [CrossRef]
- Ashraf, M.; Foolad, M.R. Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Gil, R.; Bautista, I.; Boscaiu, M.; Lidón, A.; Wankhade, S.; Sánchez, H.; Llinares, J.; Vicente, O. Responses of five Mediterranean halophytes to seasonal changes in environmental conditions. AoB Plants 2014, 6, plu049. [Google Scholar] [CrossRef] [PubMed]
- Annunziata, M.G.; Ciarmiello, L.F.; Woodrow, P.; Dell’Aversana, E.; Carillo, P. Spatial and temporal profile of glycine betaine accumulation in plants under abiotic stresses. Front. Plant Sci. 2019, 10, 230. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, J.M.C. Free Radicals in Biology and Medicine, 5th ed.; Oxforf University Press: Oxford, UK, 2015. [Google Scholar]
- Moghaddam, A.; Larijani, H.R.; Oveysi, M.; Moghaddam, H.R.T.; Nasri, M. Alleviating the adverse effects of salinity stress on Salicornia persica using sodium nitroprusside and potassium nitrate. BMC Plant Biol. 2023, 23, 166. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Redox sensing and signalling associated with reactive oxygen in chloroplasts, peroxisomes and mitochondria. Physiol. Plant. 2003, 119, 355–364. [Google Scholar] [CrossRef]
- Rahman, M.M.; Mostofa, M.G.; Keya, S.S.; Siddiqui, M.N.; Ansary, M.M.U.; Das, A.K.; Rahman, M.A.; Tran, L.S.-P. Adaptive mechanisms of halophytes and their potential in improving salinity tolerance in plants. Int. J. Mol. Sci. 2021, 22, 10733. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Mohamed, E.; Matsuda, R.; El-Khatib, A.A.; Takechi, K.; Takano, H.; Takio, S. Characterization of the superoxide dismutase genes of the halophyte Suaeda maritima in Japan and Egypt. Plant Cell Rep. 2015, 34, 2099–2110. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, E.; Kasem, A.M.; Gobouri, A.A.; Elkelish, A.; Azab, E. Influence of maternal habitat on Salinity tolerance of Zygophyllum coccineum with regard to seed germination and growth parameters. Plants 2020, 9, 1504. [Google Scholar] [CrossRef] [PubMed]
- Shang, W.; Feierabend, J. Dependence of catalase photoinactivation in rye leaves on light intensity and quality and characterization of a chloroplast-mediated inactivation in red light. Photosynth. Res. 1999, 59, 201–213. [Google Scholar] [CrossRef]
- Mohammadi, F.; Kavousi, H.R.; Mansouri, M. Effects of salt stress on physio-biochemical characters and gene expressions in halophyte grass Leptochloa fusca (L.) Kunth. Acta Physiol. Plant 2019, 41, 143. [Google Scholar] [CrossRef]
- Muchate, N.S.; Nikalje, G.C.; Rajurkar, N.S.; Suprasanna, P.; Nikam, T.D. Physiological responses of the halophyte Sesuvium portulacastrum to salt stress and their relevance for saline soil bio-reclamation. Flora 2016, 224, 96–105. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Wellburn, A.R. Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem. Soc. Trans. 1983, 603, 591–593. [Google Scholar] [CrossRef]
- Overman, R.R.; Davis, A. The application of flame photometry to sodium and potassium determinations in biological fluids. J. Biol. Chem. 1947, 168, 641–649. [Google Scholar] [CrossRef]
- Hanlon, E. Determination of potassium, calcium, and magnesium in plants by atomic absorption techniques. Plant Anal. Ref. Proced. South. Reg. United States South. Coop. Ser. Bull. 1992, 368, 30–33. [Google Scholar] [CrossRef]
- Bates, L.; Waldren, R.A.; Teare, I. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Grieve, C.; Grattan, S. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 1983, 70, 303–307. [Google Scholar] [CrossRef]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Sergiev, I.; Alexieva, V.; Karanov, E. Effect of spermine, atrazine and combination between them on some endogenous protective systems and stress markers in plants. Compt. Rend. Acad. Bulg. Sci. 1997, 51, 121–124. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef] [PubMed]
- Dhindsa, R.S.; Plumb-Dhindsa, P.; Thorpe, T.A. Leaf senescence: Correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J. Exp. Bot. 1981, 32, 93–101. [Google Scholar] [CrossRef]
- Chance, B.; Maehly, A. Assay of catalases and peroxidases. Methods Enzymol. 1955, 2, 764–775. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]



| Mean Squares | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Df | FW | WC | Chl. a | Chl. b | Car | Na+ | K+ | Ca2+ | Mg2+ | |
| Species | 2 | 0.88 ** | 17.83 ** | 1.26 ** | 0.87 ** | 0.24 ** | 218,549 ns | 10,357 ns | 211 ns | 19,505 ** |
| Salinity | 2 | 0.40 ** | 135.49 ** | 20.89 ** | 8.13 ** | 2.28 ** | 935,837,253 ** | 996,868 ** | 4293 ** | 787,916 ** |
| Time | 2 | 0.79 ** | 14.86 ** | 0.02 ns | 0.31 ** | 0.16 ** | 4,431,883 ** | 40,298 ** | 70 ns | 46,001 ** |
| Species × Salinity | 4 | 0.05 * | 2.72 * | 0.10 ns | 0.19 ** | 0.03 ns | 857,253 ns | 15,407 * | 393 ** | 3842 ** |
| Species × Time | 4 | 0.07 ** | 2.98 * | 0.10 ns | 0.06 ns | 0.01 ns | 882,716 ns | 29,642 ** | 331 * | 777 ns |
| Salinity × Time | 4 | 0.10 ** | 14.64 ** | 0.49 ** | 0.42 ** | 0.19 ** | 4,675,309 ** | 4098 ns | 1096 ** | 16,638 ** |
| Species × Salinity × Time | 8 | 0.02 * | 0.62 ns | 0.03 ns | 0.04 ns | 0.02 * | 1,188,642 ** | 15,534 ** | 257 * | 1948 ** |
| Residuals | 54 | 0.008 | 1.11 | 0.07 | 0.05 | 0.01 | 392,747 | 4352 | 98 | 732 |
| CV% | 4.11 | 1.33 | 10.48 | 10.50 | 9.87 | 8.51 | 6.05 | 1.38 | 5.36 | |
| Mean Squares | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| df | PRO | GB | MDA | H2O2 | SOD | CAT | POD | APX | |
| Species | 2 | 0.46 ** | 6304 ** | 0.65 ** | 0.06 * | 25.94 ** | 1.88 ** | 338.41 ** | 1.04 ** |
| Salinity | 2 | 12.16 ** | 77,777 ** | 21.30 ** | 1.64 ** | 6828.96 ** | 114.59 ** | 14,595 ** | 18.60 ** |
| Time | 2 | 3.88 ** | 1527 ** | 2.70 ** | 0.11 ** | 65.91 ** | 3.31 ** | 366.75 ** | 2.99 ** |
| Species × Salinity | 4 | 0.19 ** | 935 ** | 0.10 ** | 0.04 * | 65.55 ** | 1.36 ** | 22.73 * | 0.13 ** |
| Species × Time | 4 | 0.47 ** | 524 ** | 0.40 ** | 0.009 ns | 6.81 * | 0.28 ns | 23.84 * | 0.03 ns |
| Salinity × Time | 4 | 1.27 ** | 1133 ** | 0.99 ** | 0.63 ** | 19.79 ** | 0.65 ** | 110.32 ** | 0.73 ** |
| Species × Salinity × Time | 8 | 0.21 ** | 361 * | 0.18 ** | 0.02 * | 4.40 ns | 0.37 ** | 15.49 * | 0.02 * |
| Residuals | 54 | 0.01 | 139 | 0.02 | 0.01 | 2.16 | 0.13 | 7.37 | 0.01 |
| CV% | 9.33 | 6.22 | 9.39 | 6.89 | 5.37 | 6.24 | 5.56 | 4.43 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Homayouni, H.; Razi, H.; Izadi, M.; Alemzadeh, A.; Kazemeini, S.A.; Niazi, A.; Vicente, O. Temporal Changes in Biochemical Responses to Salt Stress in Three Salicornia Species. Plants 2024, 13, 979. https://doi.org/10.3390/plants13070979
Homayouni H, Razi H, Izadi M, Alemzadeh A, Kazemeini SA, Niazi A, Vicente O. Temporal Changes in Biochemical Responses to Salt Stress in Three Salicornia Species. Plants. 2024; 13(7):979. https://doi.org/10.3390/plants13070979
Chicago/Turabian StyleHomayouni, Hengameh, Hooman Razi, Mahmoud Izadi, Abbas Alemzadeh, Seyed Abdolreza Kazemeini, Ali Niazi, and Oscar Vicente. 2024. "Temporal Changes in Biochemical Responses to Salt Stress in Three Salicornia Species" Plants 13, no. 7: 979. https://doi.org/10.3390/plants13070979
APA StyleHomayouni, H., Razi, H., Izadi, M., Alemzadeh, A., Kazemeini, S. A., Niazi, A., & Vicente, O. (2024). Temporal Changes in Biochemical Responses to Salt Stress in Three Salicornia Species. Plants, 13(7), 979. https://doi.org/10.3390/plants13070979








