Functional and Transcriptome Analysis Reveal Specific Roles of Dimocarpus longan DlRan3A and DlRan3B in Root Hair Development, Reproductive Growth, and Stress Tolerance
Abstract
1. Introduction
2. Results
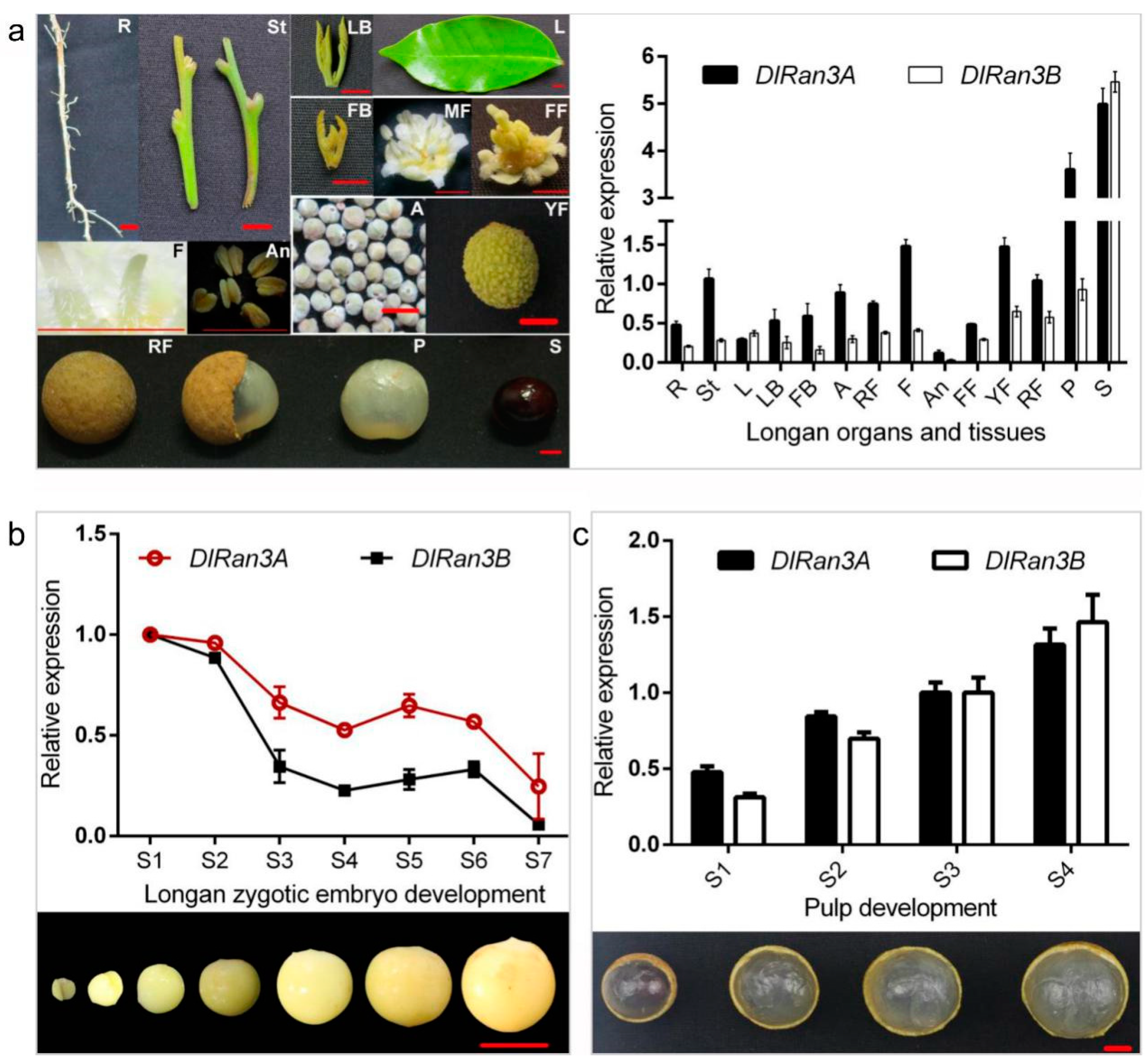
2.1. Longan DlRan3A and DRan3B Are Highly Expressed in Early Embryos and Pulp (Aril)
2.2. Characterization of DlRan3A and DlRan3B Genes and the Use of Their Own Promoters by Heterologous Expression in Tobacco Plants
2.3. Heterologous Expression in Tobaccos Reveals Shared and Distinct Roles of DlRan3A and DlRan3B in Various Stresses
2.4. Transcriptome Profiling of Transgenic Tobaccos Revealed Alteration in Genes Involved in Biosynthetic Processes Related to the Cell Wall, Hormone Signaling, and Stress Responses
2.5. Functional Classification, and Kyoto Encyclopedia of Genes and Genomes (KEGG) Analyses of DEGs
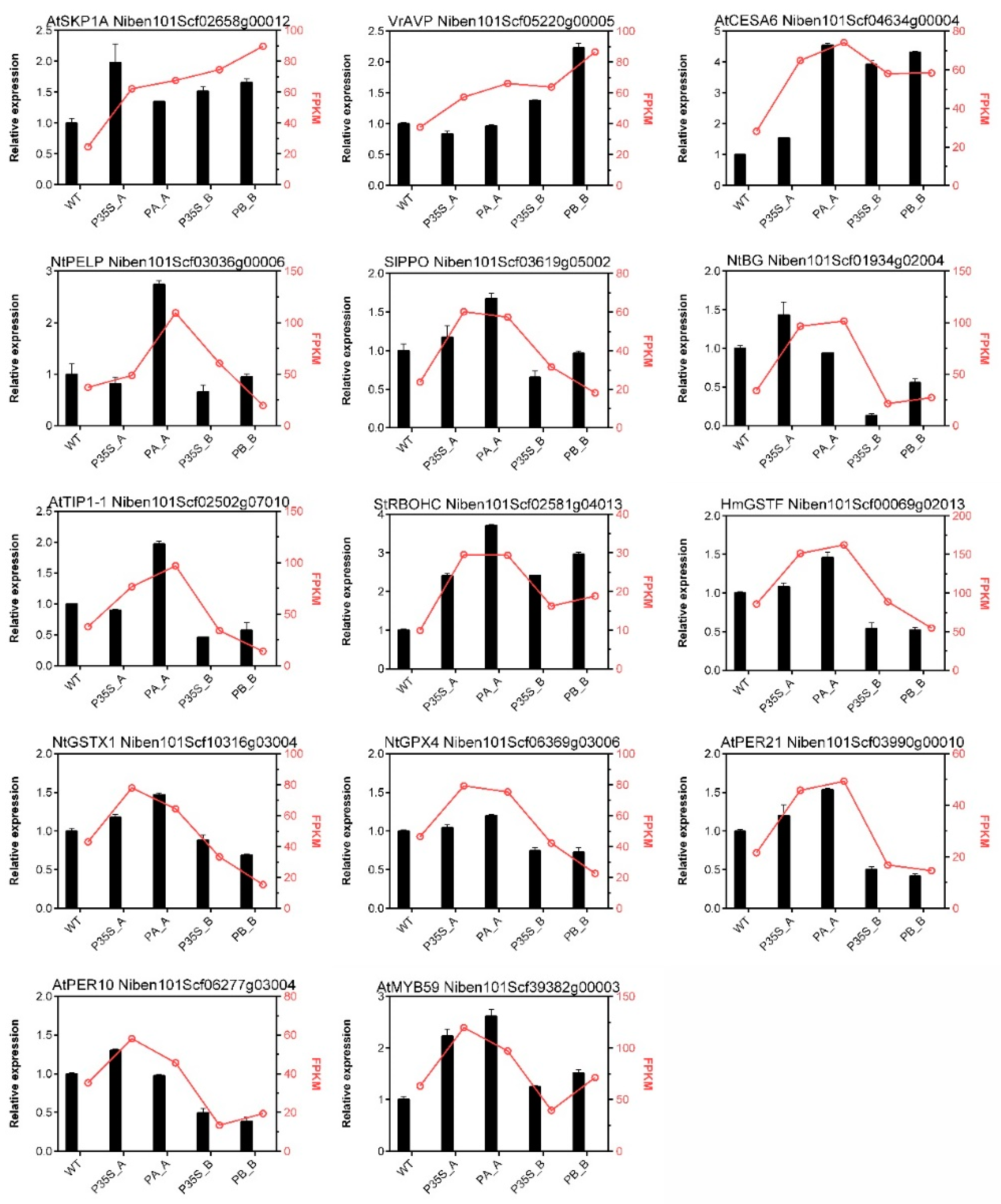
2.6. Verification qPCR Validation of Candidate DEGs
3. Discussion
3.1. DlRan3A and DlRan3B Up-Regulate Cell Wall Biosynthesis and Expansion Genes to Regulate Early Embryo and Pulp Development in Longan
3.2. pDlRan3A Driven Expression of DlRan3A Led to Stunted Plant Growth, Higher Root Hair Density, Abnormal Fruits, and Bigger Seeds, Potentially via Partial Regulation of Expansin- and Extensin-like Genes
3.3. DlRan3A and DlRan3B Function in Stress Tolerance by Regulating Different Stress-Responsive Genes
4. Materials and Methods
4.1. Plant Materials
4.2. Gene Expression Analysis
4.3. Vector Construction
4.4. Generation of T2 Transgenic Tobacco Plants
4.5. Phenotypic Analysis
4.6. Analysis of Environmental Stress Response
4.7. RNA-Sequencing
4.8. qPCR Validation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Lin, Y.; Min, J.; Lai, R.; Wu, Z.; Chen, Y.; Yu, L.; Cheng, C.; Jin, Y.; Tian, Q.; Liu, Q.; et al. Genome-wide sequencing of longan (Dimocarpus longan Lour.) provides insights into molecular basis of its polyphenol-rich characteristics. Gigascience 2017, 6, gix023. [Google Scholar] [CrossRef] [PubMed]
- Jue, D.; Liu, L.; Sang, X.; Shu, B.; Wang, J.; Wang, Y.; Zhang, C.; Shi, S. SNP-based high-density genetic map construction and candidate gene identification for fruit quality traits of Dimocarpus longan Lour. Sci. Hortic. 2021, 284, 110086. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, C.; Xu, X.; Cai, R.; Guan, Q.; Chen, X.; Chen, Y.; Zhang, Z.; XuHan, X.; Lin, Y.; et al. Riboflavin mediates m6A modification targeted by miR408, promoting early somatic embryogenesis in longan. Plant Physiol. 2023, 192, 1799–1820. [Google Scholar] [CrossRef] [PubMed]
- Salus, S.S.; Demeter, J.; Sazer, S. The Ran GTPase system in fission yeast affects microtubules and cytokinesis in cells that are competent for nucleocytoplasmic protein transport. Mol. Cell. Biol. 2002, 22, 8491–8505. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Scrofani, J.; Sardon, T.; Meunier, S.; Vernos, I. Microtubule Nucleation in Mitosis by a RanGTP-Dependent Protein Complex. Curr. Biol. 2015, 25, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xu, Y.; Han, Y.; Bao, S.; Du, J.; Yuan, M.; Xu, Z.; Chong, K. Overexpression of RAN1 in rice and Arabidopsis alters primordial meristem, mitotic progress, and sensitivity to auxin. Plant Physiol. 2006, 140, 91–101. [Google Scholar] [CrossRef]
- Malambane, G.; Madumane, K.; Sewelo, L.T.; Batlang, U. Drought stress tolerance mechanisms and their potential common indicators to salinity, insights from the wild watermelon (Citrullus lanatus): A review. Front. Plant Sci. 2022, 13, 1074395. [Google Scholar] [CrossRef]
- Zang, A.; Xu, X.; Neill, S.; Cai, W. Overexpression of OsRAN2 in rice and Arabidopsis renders transgenic plants hypersensitive to salinity and osmotic stress. J. Exp. Bot. 2010, 61, 777–789. [Google Scholar] [CrossRef]
- Chen, N.; Xu, Y.; Wang, X.; Du, C.; Du, J.; Yuan, M.; Xu, Z.; Chong, K. OsRAN2, essential for mitosis, enhances cold tolerance in rice by promoting export of intranuclear tubulin and maintaining cell division under cold stress. Plant Cell Environ. 2011, 34, 52–64. [Google Scholar] [CrossRef]
- Xu, P.; Ma, W.; Liu, J.; Hu, J.; Cai, W. Overexpression of a small GTP-binding protein Ran1 in Arabidopsis leads to promoted elongation growth and enhanced disease resistance against P. syringae DC3000. Plant J. 2021, 108, 977–991. [Google Scholar] [CrossRef]
- Yan, J.J.; Xie, B.; Zhang, L.; Li, S.J.; van Peer, A.F.; Wu, T.J.; Chen, B.Z.; Xie, B.G. Small GTPases and Stress Responses of vvran1 in the Straw Mushroom Volvariella volvacea. Int. J. Mol. Sci. 2016, 17, 1527. [Google Scholar] [CrossRef]
- Fang, Z.-Z.; Zhang, Y.-L.; Lai, C.C.; Lai, Z. Developmental regulation of Ran 3untranslated region during somatic embryogenesis in Dimocarpus longan Lour. Sci. Hortic. 2014, 176, 297–302. [Google Scholar] [CrossRef]
- Tian, Q.; Lin, Y.; Lai, Z.; Wang, T. Cloning and Bioinformatic Analysis of the Promoters of DlRan3A and DlRan3B from Embryogenic Callus in Dimocarpus longan. Chin. J. Trop. Crops 2014, 35, 82–89. [Google Scholar]
- Tian, Q.; Lin, Y.; Yang, M.; Zhang, D.; Lai, R.; Lai, Z. DlRan3A is involved in hormone, light, and abiotic stress responses in embryogenic callus of Dimocarpus longan Lour. Gene 2015, 569, 267–275. [Google Scholar] [CrossRef]
- Tian, Q.; Lin, Y.; Zhang, D.; Lai, R.; Lai, Z. Ras-Related Nuclear Protein Ran3B Gene Is Involved in Hormone Responses in the Embryogenic Callus of Dimocarpus longan Lour. Int. J. Mol. Sci. 2016, 17, 873. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, X.; Chen, X.; Chen, Y.; Zhang, Z.; Xuhan, X.; Lin, Y.; Lai, Z. Seed-Specific Gene MOTHER of FT and TFL1(MFT) Involved in Embryogenesis, Hormones and Stress Responses in Dimocarpus longan Lour. Int. J. Mol. Sci. 2018, 19, 2403. [Google Scholar] [CrossRef] [PubMed]
- Ach, R.A.; Gruissem, W. A small nuclear GTP-binding protein from tomato suppresses a Schizosaccharomyces pombe cell-cycle mutant. Proc. Natl. Acad. Sci. USA 1994, 91, 5863–5867. [Google Scholar] [CrossRef] [PubMed]
- Haizel, T.; Merkle, T.; Pay, A.; Fejes, E.; Nagy, F. Characterization of proteins that interact with the GTP-bound form of the regulatory GTPase Ran in Arabidopsis. Plant J. 1997, 11, 93–103. [Google Scholar] [CrossRef]
- Lü, S.H.; Fan, Y.L.; Jin, C.X. Overexpression of a Ran GTPase homologous gene, FaRan from tall fescue, in transgenic Arabidopsis. Biol. Plant. 2011, 55, 331–334. [Google Scholar] [CrossRef]
- Huang, N.; Ling, H.; Zhang, X.; Mao, H.; Su, Y.; Su, W.; Liu, F.; Xu, L.; Chen, R.; Que, Y. A Small GTP-Binding Gene ScRan from Sugarcane is Involved in Responses to Various Hormone Stresses and Sporisirium scitamineum Challenge. Sugar Technol. 2018, 20, 669–680. [Google Scholar] [CrossRef]
- Reynolds, J.P. Characterisation of the Defective Embryo and Meristems (DEM) Genes of Plants. Ph.D. Thesis, The University of Queensland, Townsville, QLD, Australia, 2013. [Google Scholar]
- Liu, P.; Qi, M.; Wang, Y.; Chang, M.; Liu, C.; Sun, M.; Yang, W.; Ren, H. Arabidopsis RAN1 mediates seed development through its parental ratio by affecting the onset of endosperm cellularization. Mol. Plant 2014, 7, 1316–1328. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wolf, S.; Hématy, K.; Höfte, H. Growth control and cell wall signaling in plants. Annu. Rev. Plant Biol. 2012, 63, 381–407. [Google Scholar] [CrossRef] [PubMed]
- Malinowski, R.; Filipecki, M.; Tagashira, N.; Wiśniewska, A.; Gaj, P.; Plader, W.; Malepszy, S. Xyloglucan endotransglucosylase/hydrolase genes in cucumber (Cucumis sativus)-differential expression during somatic embryogenesis. Physiol. Plant. 2004, 120, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Hall, Q.; Cannon, M.C. The cell wall hydroxyproline-rich glycoprotein RSH is essential for normal embryo development in Arabidopsis. Plant Cell 2002, 14, 1161–1172. [Google Scholar] [CrossRef] [PubMed]
- Cannon, M.C.; Terneus, K.; Hall, Q.; Tan, L.; Wang, Y.; Wegenhart, B.L.; Chen, L.; Lamport, D.T.; Chen, Y.; Kieliszewski, M.J. Self-assembly of the plant cell wall requires an extensin scaffold. Proc. Natl. Acad. Sci. USA 2008, 105, 2226–2231. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Wu, K.; Harberd, N.P.; Fu, X. Green Revolution DELLAs: From translational reinitiation to future sustainable agriculture. Mol. Plant 2021, 14, 547–549. [Google Scholar] [CrossRef] [PubMed]
- Budot, B.O.; Encabo, J.R.; Ambita, I.D.; Atienza-Grande, G.A.; Satoh, K.; Kondoh, H.; Ulat, V.J.; Mauleon, R.; Kikuchi, S.; Choi, I.R. Suppression of cell wall-related genes associated with stunting of Oryza glaberrima infected with Rice tungro spherical virus. Front. Microbiol. 2014, 5, 26. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.; Lee, Y.; Cho, H.T.; Kende, H. Regulation of expansin gene expression affects growth and development in transgenic rice plants. Plant Cell 2003, 15, 1386–1398. [Google Scholar] [CrossRef]
- Ilias, I.A.; Negishi, K.; Yasue, K.; Jomura, N.; Morohashi, K.; Baharum, S.N.; Goh, H.H. Transcriptome-wide effects of expansin gene manipulation in etiolated Arabidopsis seedling. J. Plant Res. 2019, 132, 159–172. [Google Scholar] [CrossRef]
- Koevoets, I.T.; Venema, J.H.; Elzenga, J.T.; Testerink, C. Roots Withstanding their Environment: Exploiting Root System Architecture Responses to Abiotic Stress to Improve Crop Tolerance. Front. Plant Sci. 2016, 7, 1335. [Google Scholar] [CrossRef]
- Lei, M.J.; Wang, Q.; Li, X.; Chen, A.; Luo, L.; Xie, Y.; Li, G.; Luo, D.; Mysore, K.S.; Wen, J.; et al. The small GTPase ROP10 of Medicago truncatula is required for both tip growth of root hairs and nod factor-induced root hair deformation. Plant Cell 2015, 27, 806–822. [Google Scholar] [CrossRef]
- Kang, E.; Zheng, M.; Zhang, Y.; Yuan, M.; Yalovsky, S.; Zhu, L.; Fu, Y. The Microtubule-Associated Protein MAP18 Affects ROP2 GTPase Activity during Root Hair Growth. Plant Physiol. 2017, 174, 202–222. [Google Scholar] [CrossRef]
- Bucher, M.; Brunner, S.; Zimmermann, P.; Zardi, G.I.; Amrhein, N.; Willmitzer, L.; Riesmeier, J.W. The expression of an extensin-like protein correlates with cellular tip growth in tomato. Plant Physiol. 2002, 128, 911–923. [Google Scholar] [CrossRef] [PubMed]
- Kwasniewski, M.; Janiak, A.; Mueller-Roeber, B.; Szarejko, I. Global analysis of the root hair morphogenesis transcriptome reveals new candidate genes involved in root hair formation in barley. J. Plant Physiol. 2010, 167, 1076–1083. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, S.M.; Ricardi, M.M.; Dorosz, J.G.; Fernandez, P.V.; Nadra, A.D.; Pol-Fachin, L.; Egelund, J.; Gille, S.; Harholt, J.; Ciancia, M.; et al. O-glycosylated cell wall proteins are essential in root hair growth. Science 2011, 332, 1401–1403. [Google Scholar] [CrossRef] [PubMed]
- Eberle, C.A.; Anderson, N.O.; Clasen, B.M.; Hegeman, A.D.; Smith, A.G. PELPIII: The class III pistil-specific extensin-like Nicotiana tabacum proteins are essential for interspecific incompatibility. Plant J. 2013, 74, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.M.L.; Noyszewski, A.K.; Smith, A.G. Structure and function of class III pistil-specific extensin-like protein in interspecific reproductive barriers. BMC Plant Biol. 2019, 19, 118. [Google Scholar] [CrossRef] [PubMed]
- Ralevski, A. The Interaction of Early Salt Stress-Induced 2 (ESI2) and the Ran G Protein in Arabidopsis. Master’s Thesis, Concordia University, Montreal, QC, Canada, 2013. [Google Scholar]
- Xu, P.; Cai, W. RAN1 is involved in plant cold resistance and development in rice (Oryza sativa). J. Exp. Bot. 2014, 65, 3277–3287. [Google Scholar] [CrossRef] [PubMed]
- Karuppanapandian, T.; Rhee, S.-J.; Kim, E.-J.; Han, B.K.; Hoekenga, O.A.; Lee, G.P. Proteomic analysis of differentially expressed proteins in the roots of Columbia-0 and Landsberg erecta ecotypes of Arabidopsis thaliana in response to aluminum toxicity. Can. J. Plant Sci. 2012, 92, 1267–1282. [Google Scholar] [CrossRef]
- Kagale, S.; Divi, U.K.; Krochko, J.E.; Keller, W.A.; Krishna, P. Brassinosteroid confers tolerance in Arabidopsis thaliana and Brassica napus to a range of abiotic stresses. Planta 2007, 225, 353–364. [Google Scholar] [CrossRef]
- Xia, X.-J.; Wang, Y.-J.; Zhou, Y.-H.; Tao, Y.; Mao, W.-H.; Shi, K.; Asami, T.; Chen, Z.; Yu, J.-Q. Reactive Oxygen Species Are Involved in Brassinosteroid-Induced Stress Tolerance in Cucumber. Plant Physiol. 2009, 150, 801–814. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, H.; Zhao, Z.; An, L. Abscisic acid is involved in brassinosteroids-induced chilling tolerance in the suspension cultured cells from Chorispora bungeana. J. Plant Physiol. 2011, 168, 853–862. [Google Scholar] [CrossRef]
- Fu, J.; Sun, P.; Luo, Y.; Zhou, H.; Gao, J.; Zhao, D.; Pubu, Z.; Liu, J.; Hu, T. Brassinosteroids enhance cold tolerance in Elymus nutans via mediating redox homeostasis and proline biosynthesis. Environ. Exp. Bot. 2019, 167, 103831. [Google Scholar] [CrossRef]
- Arfan, M.; Zhang, D.W.; Zou, L.J.; Luo, S.S.; Tan, W.R.; Zhu, T.; Lin, H.H. Hydrogen Peroxide and Nitric Oxide Crosstalk Mediates Brassinosteroids Induced Cold Stress Tolerance in Medicago truncatula. Int. J. Mol. Sci. 2019, 20, 144. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, M.A.; Mateos, I.; Labrador, E.; Dopico, B. Brassinolides and IAA induce the transcription of four alpha-expansin genes related to development in Cicer arietinum. Plant Physiol. Biochem. 2004, 42, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Lisso, J.; Steinhauser, D.; Altmann, T.; Kopka, J.; Müssig, C. Identification of brassinosteroid-related genes by means of transcript co-response analyses. Nucleic Acids Res. 2005, 33, 2685–2696. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Divi, U.K.; Rahman, T.; Krishna, P. Gene expression and functional analyses in brassinosteroid-mediated stress tolerance. Plant Biotechnol. J. 2016, 14, 419–432. [Google Scholar] [CrossRef] [PubMed]
- Orman-Ligeza, B.; Parizot, B.; de Rycke, R.; Fernandez, A.; Himschoot, E.; Van Breusegem, F.; Bennett, M.J.; Perilleux, C.; Beeckman, T.; Draye, X. RBOH-mediated ROS production facilitates lateral root emergence in Arabidopsis. Development 2016, 143, 3328–3339. [Google Scholar] [CrossRef] [PubMed]
- Verma, P.; Sanyal, S.K.; Pandey, G.K. Ca2+–CBL–CIPK: A modulator system for efficient nutrient acquisition. Plant Cell Rep. 2021, 40, 2111–2122. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.; Yan, M.; Zhang, Q. Functional regulation of plant NADPH oxidase and its role in signaling. Plant Signal. Behav. 2017, 12, 1356970. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.L.; Lai, Z.X. Reference gene selection for qPCR analysis during somatic embryogenesis in longan tree. Plant Sci. 2010, 178, 359–365. [Google Scholar] [CrossRef]
- Feng, X. Genome-Wide Identification and Function Analysis of the Superoxide Dismutase Gene Family in Musa spp. Ph.D. Thesis, Fujian Agriculture and Forestry University, Fuzhou, China, 2016. [Google Scholar]
- Cui, Y.; Zhu, Y.; Lin, Y.; Chen, L.; Feng, Q.; Wang, W.; Xiang, H. New insight into the mechanism underlying the silk gland biological process by knocking out fibroin heavy chain in the silkworm. BMC Genome 2018, 19, 215. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chen, H.; Zhang, Y.; Thomas, H.R.; Frank, M.H.; He, Y.; Xia, R. TBtools: An Integrative Toolkit Developed for Interactive Analyses of Big Biological Data. Mol. Plant 2020, 13, 1194–1202. [Google Scholar] [CrossRef]
- Pérez-Rodríguez, P.; Riaño-Pachón, D.M.; Corrêa, L.G.; Rensing, S.A.; Kersten, B.; Mueller-Roeber, B. PlnTFDB: Updated content and new features of the plant transcription factor database. Nucleic Acids Res. 2010, 38, D822–D827. [Google Scholar] [CrossRef]
- Jin, J.; Zhang, H.; Kong, L.; Gao, G.; Luo, J. PlantTFDB 3.0: A portal for the functional and evolutionary study of plant transcription factors. Nucleic Acids Res. 2014, 42, D1182–D1187. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, X.; Yu, H.; Jiang, W.; Sun, N.; Liu, X.; Liu, X.; Zhang, X.; Wang, Y.; Gu, X. RNA-Seq-based transcriptome profiling of early nitrogen deficiency response in cucumber seedlings provides new insight into the putative nitrogen regulatory network. Plant Cell Physiol. 2015, 56, 455–467. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tian, Q.; Xie, X.; Lai, R.; Cheng, C.; Zhang, Z.; Chen, Y.; XuHan, X.; Lin, Y.; Lai, Z. Functional and Transcriptome Analysis Reveal Specific Roles of Dimocarpus longan DlRan3A and DlRan3B in Root Hair Development, Reproductive Growth, and Stress Tolerance. Plants 2024, 13, 480. https://doi.org/10.3390/plants13040480
Tian Q, Xie X, Lai R, Cheng C, Zhang Z, Chen Y, XuHan X, Lin Y, Lai Z. Functional and Transcriptome Analysis Reveal Specific Roles of Dimocarpus longan DlRan3A and DlRan3B in Root Hair Development, Reproductive Growth, and Stress Tolerance. Plants. 2024; 13(4):480. https://doi.org/10.3390/plants13040480
Chicago/Turabian StyleTian, Qilin, Xiying Xie, Ruilian Lai, Chunzhen Cheng, Zihao Zhang, Yukun Chen, Xu XuHan, Yuling Lin, and Zhongxiong Lai. 2024. "Functional and Transcriptome Analysis Reveal Specific Roles of Dimocarpus longan DlRan3A and DlRan3B in Root Hair Development, Reproductive Growth, and Stress Tolerance" Plants 13, no. 4: 480. https://doi.org/10.3390/plants13040480
APA StyleTian, Q., Xie, X., Lai, R., Cheng, C., Zhang, Z., Chen, Y., XuHan, X., Lin, Y., & Lai, Z. (2024). Functional and Transcriptome Analysis Reveal Specific Roles of Dimocarpus longan DlRan3A and DlRan3B in Root Hair Development, Reproductive Growth, and Stress Tolerance. Plants, 13(4), 480. https://doi.org/10.3390/plants13040480







