A Comprehensive Review of Microalgae and Cyanobacteria-Based Biostimulants for Agriculture Uses
Abstract
1. Introduction
2. Microalgae-Based Biostimulants: From Phototrophic Microorganisms to Biostimulants
2.1. Selection of Microalgae Strains with High Biostimulant Potential
2.2. Microalgae Cultivation and Biomass Production
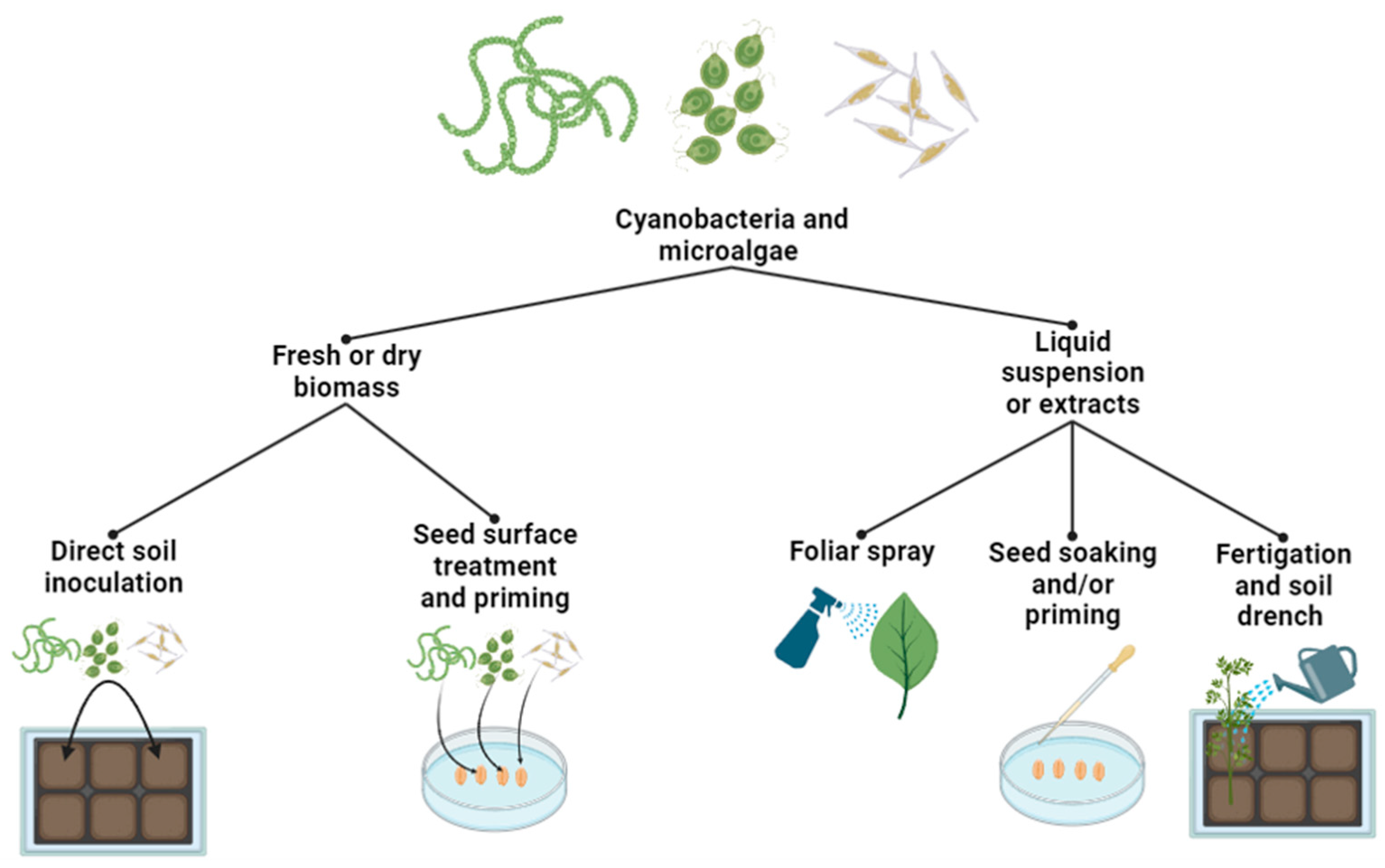
2.3. Extraction and Application Methods of Microalgae- and Cyanobacteria-Based Biostimulants
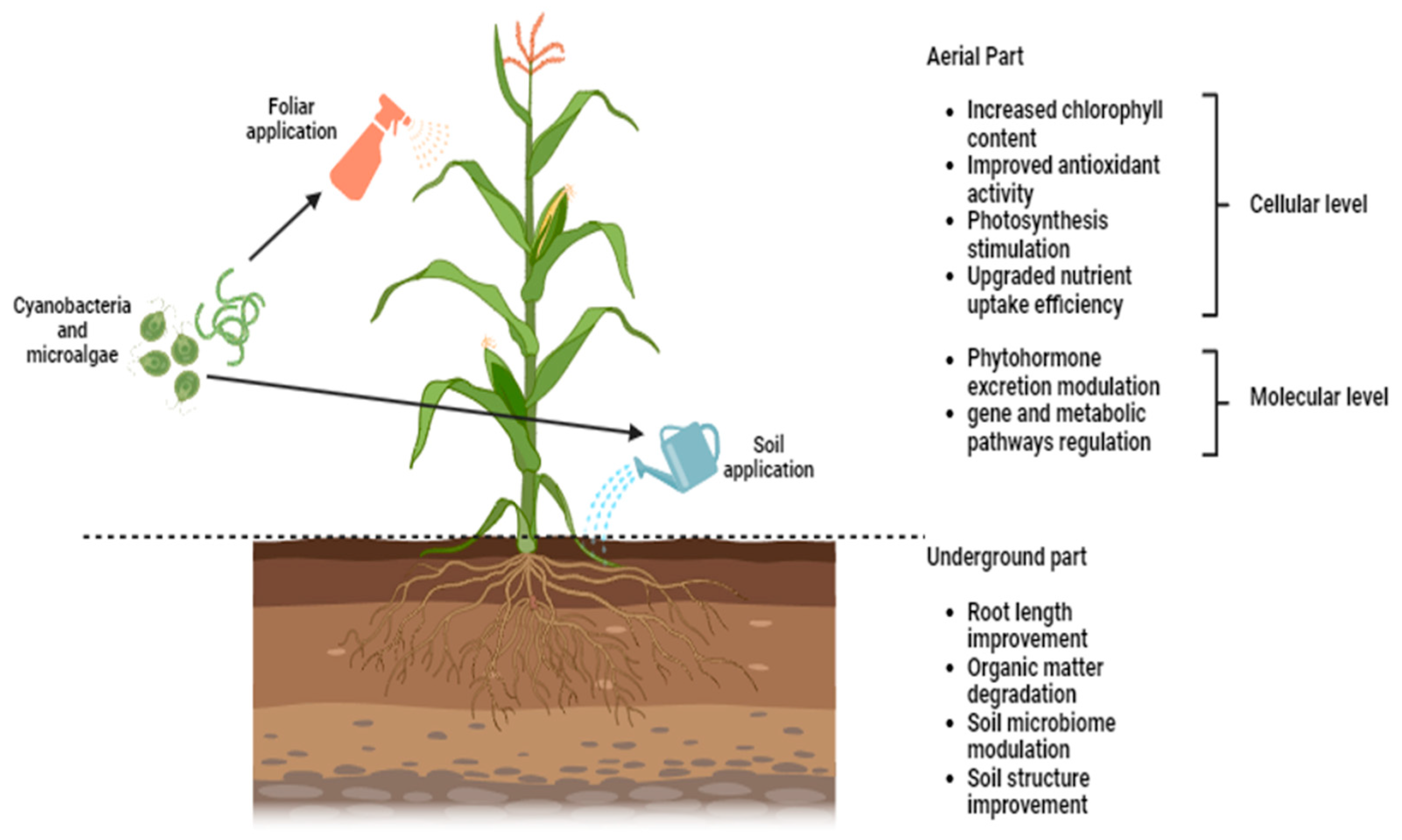
2.4. Mechanisms and Modes of Action of Microalgae- and Cyanobacteria-Based Biostimulants
3. Microalgae and Cyanobacteria Strains as Biostimulants in Agriculture
3.1. Cyanobacteria Use as Plant Biostimulants
3.2. Green Microalgae Use as Plant Biostimulants
3.3. Diatoms Use as Plant Biostimulants/Biofertilizers
3.4. The Use of Microalgae- and Cyanobacteria-Based Consortia/Associations as Plant Biostimulant
4. Economic and Research Trends in Microalgae- and Cyanobacteria-Based Biostimulants for Agriculture Uses
5. Commercialized Microalgae- and Cyanobacteria-Based Biostimulant Products and Consumer Acceptance
6. Regulations and Legal Framework of Biostimulants for Agriculture Uses
7. Bottlenecks and Use Limitations of Microalgae and Cyanobacteria Biostimulants
- Fully explore the biodiversity of particularly microalgae in aquatic and soil ecosystems, plus broadening tests to include all groups of microalgae in particular diatoms;
- To address the production challenge, it is necessary to focus on minimizing the costs of production and application through the optimization of production schemes mainly targeting the use of waste resources by adopting biorefinery approach, as well as scaling up application methods to be used on an agricultural scale;
- In order to fully use the biostimulant potential of microalgae, it is necessary to understand the physiological and molecular mechanisms by which these compounds affect the plant and soil ecosystem by adopting new approaches mainly molecular and omics;
- The need to fully explore the conditions under which the benefits of biostimulants formulations are optimal, encompassing pedology and agroclimatic conditions, crop type and growth stage, timing of use and frequency of use, as well as revealing the possible interactions with soil components such as bacteria, fungi, and microfauna;
- For a successful input in agriculture, it is necessary to raise the awareness of farmers regarding the benefits of using these tools.
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The future of food and agriculture—Alternative pathways to 2050|Global Perspectives Studies|Food and Agriculture Organization of the United Nations. In Food and Agriculture Organization; FAO: Rome, Italy, 2018; p. 224. ISBN 978-92-5-130158-6. [Google Scholar]
- Bulgari, R.; Cocetta, G.; Trivellini, A.; Vernieri, P.; Ferrante, A. Biostimulants and crop responses: A review. Biol. Agric. Hortic. 2014, 31, 1–17. [Google Scholar] [CrossRef]
- González-Pérez, B.K.; Rivas-Castillo, A.M.; Valdez-Calderón, A.; Gayosso-Morales, M.A. Microalgae as biostimulants: A new approach in agriculture. World J. Microbiol. Biotechnol. 2021, 38, 4. [Google Scholar] [CrossRef] [PubMed]
- Pahalvi, H.N.; Rafiya, L.; Rashid, S.; Nisar, B.; Kamili, A.N. Microbiota and Biofertilizers, Ecofriendly Tools for Reclamation of Degraded Soil Environs; Springer: Berlin/Heidelberg, Germany, 2022; Volume 2, ISBN 9783030610098. [Google Scholar]
- Calabi-Floody, M.; Medina, J.; Rumpel, C.; Condron, L.M.; Hernandez, M.; Dumont, M.; Mora, M.d.L. Smart Fertilizers as a Strategy for Sustainable Agriculture, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; Volume 147. [Google Scholar]
- Abinandan, S.; Subashchandrabose, S.R.; Venkateswarlu, K.; Megharaj, M. Soil microalgae and cyanobacteria: The biotechnological potential in the maintenance of soil fertility and health. Crit. Rev. Biotechnol. 2019, 39, 981–998. [Google Scholar] [CrossRef] [PubMed]
- Bomfim, C.A.; Coelho, L.G.F.; do Vale, H.M.M.; de Carvalho Mendes, I.; Megías, M.; Ollero, F.J.; dos Reis Junior, F.B. Brief history of biofertilizers in Brazil: From conventional approaches to new biotechnological solutions. Braz. J. Microbiol. 2021, 52, 2215–2232. [Google Scholar] [CrossRef]
- Yousfi, S.; Marín, J.; Parra, L.; Lloret, J.; Mauri, P.V. A Rhizogenic Biostimulant Effect on Soil Fertility and Roots Growth of Turfgrass. Agronomy 2021, 11, 573. [Google Scholar] [CrossRef]
- Kauffman, G.L.; Kneivel, D.P.; Watschke, T.L. Effects of a Biostimulant on the Heat Tolerance Associated with Photosynthetic Capacity, Membrane Thermostability, and Polyphenol Production of Perennial Ryegrass. Crop Sci. 2007, 47, 261–267. [Google Scholar] [CrossRef]
- European Commission. Regulation of the European Parliament and of the Council Laying down Rules on the Making Available on the Market of EU Fertilising Products and Amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009 and Repealing Regulation (EC) No 2003/2003. Off. J. Eur. Union 2019, 2019, 114. [Google Scholar]
- du Jardin, P. Plant Biostimulants: Definition, Concept, Main Categories and Regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Yakhin, O.I.; Lubyanov, A.A.; Yakhin, I.A.; Brown, P.H. Biostimulants in Plant Science: A Global Perspective. Front. Plant Sci. 2017, 7, 2049. [Google Scholar] [CrossRef]
- Ronga, D.; Biazzi, E.; Parati, K.; Carminati, D.; Carminati, E.; Tava, A. Microalgal Biostimulants and Biofertilisers in Crop Productions. Agronomy 2019, 9, 192. [Google Scholar] [CrossRef]
- Guntzer, F.; Keller, C.; Meunier, J.-D. Benefits of plant silicon for crops: A review. Agron. Sustain. Dev. 2012, 32, 201–213. [Google Scholar] [CrossRef]
- Du Jardin, P.; Xu, L.; Geelen, D. Agricultural Functions and Action Mechanisms of Plant Biostimulants (PBs): An Introduction. Chem. Biol. Plant Biostimul. 2020, 1, 3–30. [Google Scholar] [CrossRef]
- Chiaiese, P.; Corrado, G.; Colla, G.; Kyriacou, M.C.; Rouphael, Y. Renewable Sources of Plant Biostimulation: Microalgae as a Sustainable Means to Improve Crop Performance. Front. Plant Sci. 2018, 871, 01782. [Google Scholar] [CrossRef] [PubMed]
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. Agric. 2017, 4, 5. [Google Scholar] [CrossRef]
- Kapoore, R.V.; Wood, E.E.; Llewellyn, C.A. Algae biostimulants: A critical look at microalgal biostimulants for sustainable agricultural practices. Biotechnol. Adv. 2021, 49, 107754. [Google Scholar] [CrossRef]
- Cadoret, J.-P.; Bernard, O. La Production de Biocarburant Lipidique Avec Des Microalgues: Promesses et Défis. J. Soc. Biol. 2008, 202, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Issa, A.A.; Hemida Abd-Alla, M.; Ohyam, T. Nitrogen Fixing Cyanobacteria: Future Prospect. Adv. Biol. Ecol. Nitrogen Fixat. 2014, 1, 24–48. [Google Scholar] [CrossRef]
- Sankaran, R.; Show, P.L.; Nagarajan, D.; Chang, J.S. Exploitation and Biorefinery of Microalgae; Elsevier B.V.: Amsterdam, The Netherlands, 2018; ISBN 9780444639929. [Google Scholar]
- Singh, S. A review on possible elicitor molecules of cyanobacteria: Their role in improving plant growth and providing tolerance against biotic or abiotic stress. J. Appl. Microbiol. 2014, 117, 1221–1244. [Google Scholar] [CrossRef]
- Górka, B.; Korzeniowska, K.; Lipok, J.; Wieczorek, P.P. The Biomass of Algae and Algal Extracts in Agricultural Production. Algae Biomass Charact. Appl. 2018, 8, 103–114. [Google Scholar] [CrossRef]
- Márquez-Rocha, F.J.; Palma-Ramírez, D.; García-Alamilla, P.; López-Hernández, J.F.; Santiago-Morales, I.S.; Flores-Vela, A.I. Microalgae Cultivation for Secondary Metabolite Production. Microalgae Physiol. Appl. 2020, 11, 193–204. [Google Scholar] [CrossRef]
- Vuppaladadiyam, A.K.; Prinsen, P.; Raheem, A.; Luque, R.; Zhao, M. Microalgae cultivation and metabolites production: A comprehensive review. Biofuels Bioprod. Biorefin. 2018, 8, 743. [Google Scholar] [CrossRef]
- Liang, Y.; Kashdan, T.; Sterner, C.; Dombrowski, L.; Petrick, I.; Kröger, M.; Höfer, R. Algal Biorefineries; Elsevier B.V.: Amsterdam, The Netherlands, 2015; ISBN 9780444634535. [Google Scholar]
- Branco-Vieira, M.; Mata, T.; Martins, A.; Freitas, M.; Caetano, N. Economic analysis of microalgae biodiesel production in a small-scale facility. Energy Rep. 2020, 6, 325–332. [Google Scholar] [CrossRef]
- Ummalyma, S.B.; Sahoo, D.; Pandey, A. Microalgal Biorefineries for Industrial Products. In Microalgae Cultivation for Biofuels Production; Academic Press: Cambridge, MA, USA, 2020; pp. 187–195. [Google Scholar] [CrossRef]
- Al-Jabri, H.; Das, P.; Khan, S.; Thaher, M.; AbdulQuadir, M. Treatment of Wastewaters by Microalgae and the Potential Applications of the Produced Biomass—A Review. Water 2020, 13, 27. [Google Scholar] [CrossRef]
- Navarro-López, E.; Ruíz-Nieto, A.; Ferreira, A.; Acién, F.G.; Gouveia, L. Biostimulant Potential of Scenedesmus obliquus Grown in Brewery Wastewater. Molecules 2020, 25, 664. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.; Melkonyan, L.; Carapinha, S.; Ribeiro, B.; Figueiredo, D.; Avetisova, G.; Gouveia, L. Biostimulant and biopesticide potential of microalgae growing in piggery wastewater. Environ. Adv. 2021, 4, 100062. [Google Scholar] [CrossRef]
- Morillas-España, A.; Lafarga, T.; Sánchez-Zurano, A.; Acién-Fernández, F.G.; González-López, C. Microalgae based wastewater treatment coupled to the production of high value agricultural products: Current needs and challenges. Chemosphere 2022, 291, 132968. [Google Scholar] [CrossRef]
- Alling, T.; Funk, C.; Gentili, F.G. Nordic microalgae produce biostimulant for the germination of tomato and barley seeds. Sci. Rep. 2023, 13, 3509. [Google Scholar] [CrossRef]
- Nwuche, C.O.; Ekpo, D.C.; Eze, C.N.; Aoyagi, H.; Ogbonna, J.C. Use of Palm Oil Mill Effluent as Medium for Cultivation of Chlorella sorokiniana. Br. Biotechnol. J. 2014, 4, 305–316. [Google Scholar] [CrossRef]
- Selmani, N.; Mirghani, M.E.S.; Alam, Z. Study the Growth of Microalgae in Palm Oil Mill Effluent Waste Water. IOP Conf. Ser. Earth Environ. Sci. 2013, 16, 12–16. [Google Scholar] [CrossRef]
- Priya, H.; Prasanna, R.; Ramakrishnan, B.; Bidyarani, N.; Babu, S.; Thapa, S.; Renuka, N. Influence of cyanobacterial inoculation on the culturable microbiome and growth of rice. Microbiol. Res. 2015, 171, 78–89. [Google Scholar] [CrossRef]
- Ferreira, A.; Figueiredo, D.; Ferreira, F.; Marujo, A.; Bastos, C.R.; Martin-Atanes, G.; Ribeiro, B.; Štěrbová, K.; Marques-Dos-Santos, C.; Acién, F.G.; et al. From piggery wastewater to wheat using microalgae towards zero waste. Algal Res. 2023, 72. [Google Scholar] [CrossRef]
- Álvarez-González, A.; Uggetti, E.; Serrano, L.; Gorchs, G.; Casas, M.E.; Matamoros, V.; Gonzalez-Flo, E.; Díez-Montero, R. The potential of wastewater grown microalgae for agricultural purposes: Contaminants of emerging concern, heavy metals and pathogens assessment. Environ. Pollut. 2023, 324, 121399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.-W.; Lin, L.-G.; Ye, W.-C. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Xu, G.; Li, S.-P.; Wang, Y.-T.; Fan, T.-P.; Zhao, Q.-S.; Zhang, Q.-W. Optimizing Ultraperformance Liquid Chromatographic Analysis of 10 Diterpenoid Compounds in Salvia miltiorrhiza Using Central Composite Design. J. Agric. Food Chem. 2008, 56, 1164–1171. [Google Scholar] [CrossRef]
- Du, G.; Zhao, H.; Song, Y.; Zhang, Q.; Wang, Y. Rapid simultaneous determination of isoflavones in Radix puerariae using high-performance liquid chromatography-triple quadrupole mass spectrometry with novel shell-type column. J. Sep. Sci. 2011, 34, 2576–2585. [Google Scholar] [CrossRef]
- Michalak, I.; Chojnacka, K. Algal extracts: Technology and advances. Eng. Life Sci. 2014, 14, 581–591. [Google Scholar] [CrossRef]
- Michalak, I.; Chojnacka, K. Algae as production systems of bioactive compounds. Eng. Life Sci. 2015, 15, 160–176. [Google Scholar] [CrossRef]
- Kim, G.M.; Kim, Y.K. Drying Techniques of Microalgal Biomass: A Review. Appl. Chem. Eng. 2022, 33, 145–150. [Google Scholar] [CrossRef]
- Wiltshire, K.H.; Boersma, M.; Möller, A.; Buhtz, H. Extraction of pigments and fatty acids from the green alga Scenedesmus obliquus (Chlorophyceae). Aquat. Ecol. 2000, 34, 119–126. [Google Scholar] [CrossRef]
- Lee, J.Y.; Yoo, C.; Jun, S.Y.; Ahn, C.Y.; Oh, H.M. Comparison of Several Methods for Effective Lipid Extraction from Micro-algae. Bioresour. Technol. 2010, 101, S75–S77. [Google Scholar] [CrossRef]
- Navarro-López, E.; Gallardo-Rodríguez, J.J.; Cerón-García, M.d.C.; Gallego-López, I.; Acién-Fernández, F.G.; Molina-Grima, E. Extraction of phytostimulant molecules from Scenedesmus almeriensis using different extractor systems. J. Appl. Phycol. 2023, 35, 701–711. [Google Scholar] [CrossRef]
- Gitau, M.M.; Farkas, A.; Balla, B.; Ördög, V.; Futó, Z.; Maróti, G. Strain-Specific Biostimulant Effects of Chlorella and Chlamydomonas Green Microalgae on Medicago truncatula. Plants 2021, 10, 1060. [Google Scholar] [CrossRef]
- Bayona-Morcillo, P.J.; Plaza, B.M.; Gómez-Serrano, C.; Rojas, E.; Jiménez-Becker, S. Effect of the foliar application of cyanobacterial hydrolysate (Arthrospira platensis) on the growth of Petunia x hybrida under salinity conditions. J. Appl. Phycol. 2020, 32, 4003–4011. [Google Scholar] [CrossRef]
- Refaay, D.A.; El-Marzoki, E.M.; Abdel-Hamid, M.I.; Haroun, S.A. Effect of foliar application with Chlorella vulgaris, Tetradesmus dimorphus, and Arthrospira platensis as biostimulants for common bean. J. Appl. Phycol. 2021, 33, 3807–3815. [Google Scholar] [CrossRef]
- Supraja, K.V.; Behera, B.; Balasubramanian, P. Efficacy of microalgal extracts as biostimulants through seed treatment and foliar spray for tomato cultivation. Ind. Crop. Prod. 2020, 151, 112453. [Google Scholar] [CrossRef]
- La Bella, E.; Baglieri, A.; Rovetto, E.I.; Stevanato, P.; Puglisi, I. Foliar Spray Application of Chlorella vulgaris Extract: Effect on the Growth of Lettuce Seedlings. Agronomy 2021, 11, 308. [Google Scholar] [CrossRef]
- Puglisi, I.; Barone, V.; Fragalà, F.; Stevanato, P.; Baglieri, A.; Vitale, A. Effect of Microalgal Extracts from Chlorella vulgaris and Scenedesmus quadricauda on Germination of Beta vulgaris Seeds. Plants 2020, 9, 675. [Google Scholar] [CrossRef]
- Rupawalla, Z.; Shaw, L.; Ross, I.L.; Schmidt, S.; Hankamer, B.; Wolf, J. Germination screen for microalgae-generated plant growth biostimulants. Algal Res. 2022, 66, 102784. [Google Scholar] [CrossRef]
- Jafarlou, M.B.; Pilehvar, B.; Modarresi, M.; Mohammadi, M. Performance of Algae Extracts Priming for Enhancing Seed Germination Indices and Salt Tolerance in Calotropis procera (Aiton) W.T. Iran. J. Sci. Technol. Trans. A Sci. 2021, 45, 493–502. [Google Scholar] [CrossRef]
- Van Do, T.C.; Tran, D.T.; Le, T.G.; Nguyen, Q.T. Characterization of Endogenous Auxins and Gibberellins Produced by Chlorella sorokiniana TH01 under Phototrophic and Mixtrophic Cultivation Modes toward Applications in Microalgal Biorefinery and Crop Research. J. Chem. 2020, 2020, 4910621. [Google Scholar] [CrossRef]
- Suchithra, M.; Muniswami, D.M.; Sri, M.S.; Usha, R.; Rasheeq, A.A.; Preethi, B.A.; Dineshkumar, R. Effectiveness of green microalgae as biostimulants and biofertilizer through foliar spray and soil drench method for tomato cultivation. S. Afr. J. Bot. 2021, 146, 740–750. [Google Scholar] [CrossRef]
- El-Eslamboly, A.A.S.A.; El-Wanis, M.M.A.; Amin, A.W. Algal application as a biological control method of root-knot nematode Meloidogyne incognita on cucumber under protected culture conditions and its impact on yield and fruit quality. Egypt. J. Biol. Pest Control. 2019, 29, 18. [Google Scholar] [CrossRef]
- Chanda, M.; Benhima, R.; Elmernissi, N.; Kasmi, Y.; Karim, L.; Sbabou, L.; Youssef, Z.; ElArroussi, H. Screening of microalgae liquid extracts for their bio stimulant properties on plant growth, nutrient uptake and metabolite profile of Solanum lycopersicum L. Sci. Rep. 2020, 10, 2820. [Google Scholar] [CrossRef]
- Ergun, O.; Dasgan, H.; Isık, O. Effects of microalgae Chlorella vulgaris on hydroponically grown lettuce. Acta Hortic. 2020, 1273, 169–175. [Google Scholar] [CrossRef]
- Dasgan, H.Y.; Kacmaz, S.; Arpaci, B.B.; Ikiz, B.; Gruda, N.S. Biofertilizers Improve the Leaf Quality of Hydroponically Grown Baby Spinach (Spinacia oleracea L.). Agronomy 2023, 13, 575. [Google Scholar] [CrossRef]
- Santini, G.; Rodolfi, L.; Biondi, N.; Sampietro, G.; Tredici, M.R. Effects of cyanobacterial-based biostimulants on plant growth and development: A case study on basil (Ocimum basilicum L.). J. Appl. Phycol. 2022, 34, 2063–2073. [Google Scholar] [CrossRef]
- Stirk, W.A.; Bálint, P.; Vambe, M.; Kulkarni, M.G.; van Staden, J.; Ördög, V. Effect of storage on plant biostimulant and bioactive properties of freeze-dried Chlorella vulgaris biomass. J. Appl. Phycol. 2021, 33, 3797–3806. [Google Scholar] [CrossRef]
- Kumar, S.; Korra, T.; Singh, U.B.; Singh, S.; Bisen, K. Microalgal Based Biostimulants as Alleviator of Biotic and Abiotic Stresses in Crop Plants. In New and Future Developments in Microbial Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2022; pp. 195–216. [Google Scholar] [CrossRef]
- Kumari, M.; Swarupa, P.; Kesari, K.K.; Kumar, A. Microbial Inoculants as Plant Biostimulants: A Review on Risk Status. Life 2023, 13, 12. [Google Scholar] [CrossRef]
- Parađiković, N.; Teklić, T.; Zeljković, S.; Lisjak, M.; Špoljarević, M. Biostimulants research in some horticultural plant species—A review. Food Energy Secur. 2019, 8, e00162. [Google Scholar] [CrossRef]
- Bhupenchandra, I.; Devi, S.H.; Basumatary, A.; Dutta, S.; Singh, L.K.; Kalita, P.; Bora, S.S.; Saikia, A.; Sharma, P.; Bhagowati, S.; et al. Biostimulants: Potential and Prospects in Agriculture. Int. Res. J. Pure Appl. Chem. 2020, 21, 20–35. [Google Scholar] [CrossRef]
- Renuka, N.; Guldhe, A.; Prasanna, R.; Singh, P.; Bux, F. Microalgae as multi-functional options in modern agriculture: Current trends, prospects and challenges. Biotechnol. Adv. 2018, 36, 1255–1273. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, B.; Maddela, N.R.; Venkateswarlu, K.; Megharaj, M. Potential of microalgae and cyanobacteria to improve soil health and agricultural productivity: A critical view. Environ. Sci. Adv. 2023, 2, 586–611. [Google Scholar] [CrossRef]
- Tinte, M.M.; Masike, K.; Steenkamp, P.A.; Huyser, J.; van der Hooft, J.J.J.; Tugizimana, F. Computational Metabolomics Tools Reveal Metabolic Reconfigurations Underlying the Effects of Biostimulant Seaweed Extracts on Maize Plants under Drought Stress Conditions. Metabolites 2022, 12, 487. [Google Scholar] [CrossRef] [PubMed]
- Tung, H.; Shen, T. Studies of the Azolla pinnata—Anabaena azollae symbiosis: Concurrent growth of Azolla with rice. Aquat. Bot. 1985, 22, 145–152. [Google Scholar] [CrossRef]
- Craigie, J.S. Seaweed extract stimuli in plant science and agriculture. J. Appl. Phycol. 2011, 23, 371–393. [Google Scholar] [CrossRef]
- López-Padrón, I.; Martínez-González, L.; Pérez-Domínguez, G.; Reyes-Guerrero, Y.; Núñez-Vázquez, M.; Cabrera-Rodríguez, J. Algae and Their Uses in Agriculture. An Update. Cultiv. Trop. 2020, 41, 10. [Google Scholar]
- Nanda, B.; Tripathy, S.K.; Padhi, S. Effect of algalization on seed germination of vegetable crops. World J. Microbiol. Biotechnol. 1991, 7, 622–623. [Google Scholar] [CrossRef]
- Adam, M.S. The Promotive Effect of the Cyanobacterium Nostoc Muscorum on the Growth of Some Crop Plants. Acta Microbiol. Pol. 1999, 48, 163–171. [Google Scholar]
- Llamas, A.; Leon-miranda, E.; Tejada-jimenez, M. Microalgal and Nitrogen-Fixing Bacterial Consortia: From Interaction to Biotechnological Potential. Plants 2023, 12, 2476. [Google Scholar] [CrossRef]
- Caputo, A.; Nylander, J.A.A.; Foster, R.A. The genetic diversity and evolution of diatom-diazotroph associations highlights traits favoring symbiont integration. FEMS Microbiol. Lett. 2019, 366, fny297. [Google Scholar] [CrossRef]
- Gautam, K.; Tripathi, J.K.; Pareek, A.; Sharma, D.K. Growth and secretome analysis of possible synergistic interaction between green algae and cyanobacteria. J. Biosci. Bioeng. 2019, 127, 213–221. [Google Scholar] [CrossRef]
- Maqubela, M.P.; Mnkeni, P.N.S.; Issa, O.M.; Pardo, M.T.; D’acqui, L.P. Nostoc cyanobacterial inoculation in South African agricultural soils enhances soil structure, fertility, and maize growth. Plant Soil 2008, 315, 79–92. [Google Scholar] [CrossRef]
- Mazhar, S.; Cohen, J.D.; Hasnain, S. Auxin producing non-heterocystous Cyanobacteria and their impact on the growth and endogenous auxin homeostasis of wheat. J. Basic Microbiol. 2013, 53, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Shariatmadari, Z.; Riahi, H.; Hashtroudi, M.S.; Ghassempour, A.; Aghashariatmadary, Z. Plant growth promoting cyanobacteria and their distribution in terrestrial habitats of Iran. Soil Sci. Plant Nutr. 2013, 59, 535–547. [Google Scholar] [CrossRef]
- Shariatmadari, Z.; Riahi, H.; Abdi, M.; Hashtroudi, M.S.; Ghassempour, A.R. Impact of cyanobacterial extracts on the growth and oil content of the medicinal plant Mentha piperita L. J. Appl. Phycol. 2015, 27, 2279–2287. [Google Scholar] [CrossRef]
- Dias, G.A.; Rocha, R.H.C.; Araújo, J.L.; Lima, J.F.; Guedes, W.A. Growth, yield, and postharvest quality in eggplant produced under different foliar fertilizer (Spirulina platensis) treatments. Semina Ciênc. Agrár. 2016, 37, 3893–3902. [Google Scholar] [CrossRef]
- El Arroussi, H.; Elmernissi, N.; Benhima, R.; Meftah El Kadmiri, I.; Bendaou, N.; Smouni, A. Microalgae Polysaccharides a Promising Plant Growth Biostimulant. J. Algal Biomass Utln. 2016, 7, 55–63. [Google Scholar]
- Grzesik, M.; Romanowska-Duda, Z.; Kalaji, H.M. Effectiveness of cyanobacteria and green algae in enhancing the photosynthetic performance and growth of willow (Salix viminalis L.) plants under limited synthetic fertilizers application. Photosynthetica 2017, 55, 510–521. [Google Scholar] [CrossRef]
- Mógor, Á.F.; Ördög, V.; Lima, G.P.P.; Molnár, Z.; Mógor, G. Biostimulant properties of cyanobacterial hydrolysate related to polyamines. J. Appl. Phycol. 2018, 30, 453–460. [Google Scholar] [CrossRef]
- El-Baky, H.H.A.; El-Baz, F.K.; El Baroty, G.S. Enhancing antioxidant availability in wheat grains from plants grown under seawater stress in response to microalgae extract treatments. J. Sci. Food Agric. 2009, 90, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Mutale-Joan, C.; Rachidi, F.; Mohamed, H.A.; El Mernissi, N.; Aasfar, A.; Barakate, M.; Mohammed, D.; Sbabou, L.; El Arroussi, H. Microalgae-cyanobacteria–based biostimulant effect on salinity tolerance mechanisms, nutrient uptake, and tomato plant growth under salt stress. J. Appl. Phycol. 2021, 33, 3779–3795. [Google Scholar] [CrossRef]
- Bello, A.S.; Ben-Hamadou, R.; Hamdi, H.; Saadaoui, I.; Ahmed, T. Application of Cyanobacteria (Roholtiella sp.) Liquid Extract for the Alleviation of Salt Stress in Bell Pepper (Capsicum annuum L.) Plants Grown in a Soilless System. Plants 2022, 11, 104. [Google Scholar] [CrossRef] [PubMed]
- Brito, Â.; Rocha, M.; Kaštovský, J.; Vieira, J.; Vieira, C.P.; Ramos, V.; Correia, M.; Santos, M.; Mota, R.; Roque, J.; et al. A new cyanobacterial species with a protective effect on lettuce grown under salinity stress: Envisaging sustainable agriculture practices. J. Appl. Phycol. 2022, 34, 915–928. [Google Scholar] [CrossRef]
- Alharbi, K.; Hafez, E.M.; Omara, A.E.-D.; Osman, H.S. Mitigating Osmotic Stress and Enhancing Developmental Productivity Processes in Cotton through Integrative Use of Vermicompost and Cyanobacteria. Plants 2023, 12, 1872. [Google Scholar] [CrossRef]
- Wijffels, R.H.; Kruse, O.; Hellingwerf, K.J. Potential of industrial biotechnology with cyanobacteria and eukaryotic microalgae. Curr. Opin. Biotechnol. 2013, 24, 405–413. [Google Scholar] [CrossRef]
- Ortiz-Moreno, M.L.; Sandoval-Parra, K.X.; Solarte-Murillo, L.V. Chlorella, ¿un potencial biofertilizante? Orinoquia 2019, 23, 71–78. [Google Scholar] [CrossRef]
- Faheed, F.A.; Abd-El Fattah, Z. Effect of Chlorella Vulgaris as Bio-Fertilizer on Growth Parameters and Metabolic Aspects of Lettuce Plant. AWB J. Agri. Soc. Sci. 2008, 4, 1813–2235. [Google Scholar]
- Chacón Lee, T.L. Efecto de La Aplicación de Soluciones de Chorella Vulgaris y Scenedesmus Obliquus Sobre El Contenido de Compuestos Funcionales En Germinados de Brócoli. Master’s Thesis, Universidad de La Sabana, Cundinamarca, Colombia, 2011. [Google Scholar]
- Garcia-Gonzalez, J.; Sommerfeld, M. Biofertilizer and Biostimulant Properties of the Microlaga Acutodesmus Dimorphus. J. Appl. Phycol. 2016, 28, 1051–1061. [Google Scholar] [CrossRef]
- Puglisi, I.; La Bella, E.; Rovetto, E.I.; Piero, A.R.L.; Baglieri, A. Biostimulant Effect and Biochemical Response in Lettuce Seedlings Treated with A Scenedesmus quadricauda Extract. Plants 2020, 9, 123. [Google Scholar] [CrossRef]
- Mazepa, E.; Malburg, B.V.; Mógor, G.; de Oliveira, A.C.; Amatussi, J.O.; Corrêa, D.O.; Lemos, J.S.; Ducatti, D.R.B.; Duarte, M.E.R.; Mógor, Á.F.; et al. Plant growth biostimulant activity of the green microalga Desmodesmus subspicatus. Algal Res. 2021, 59, 102434. [Google Scholar] [CrossRef]
- Puglisi, I.; La Bella, E.; Rovetto, E.I.; Stevanato, P.; Fascella, G.; Baglieri, A. Morpho biometric and Biochemical Responses in Lettuce Seedlings Treated by Different Application Methods of Chlorella Vulgaris Extract: Foliar Spray or Root Drench? J. Appl. Phycol. 2022, 34, 889–901. [Google Scholar] [CrossRef]
- Dineshkumar, R.; Duraimurugan, M.; Sharmiladevi, N.; Lakshmi, L.P.; Rasheeq, A.A.; Arumugam, A.; Sampathkumar, P. Microalgal liquid biofertilizer and biostimulant effect on green gram (Vigna radiata L) an experimental cultivation. Biomass-Convers. Biorefinery 2020, 12, 3007–3027. [Google Scholar] [CrossRef]
- Park, Y.J.; Park, J.-E.; Truong, T.Q.; Koo, S.Y.; Choi, J.-H.; Kim, S.M. Effect of Chlorella vulgaris on the Growth and Phytochemical Contents of “Red Russian” Kale (Brassica napus var. Pabularia). Agronomy 2022, 12, 2138. [Google Scholar] [CrossRef]
- Kusvuran, S. Microalgae (Chlorella vulgaris Beijerinck) alleviates drought stress of broccoli plants by improving nutrient uptake, secondary metabolites, and antioxidative defense system. Hortic. Plant J. 2021, 7, 221–231. [Google Scholar] [CrossRef]
- Rachidi, F.; Benhima, R.; Kasmi, Y.; Sbabou, L.; El Arroussi, H. Evaluation of microalgae polysaccharides as biostimulants of tomato plant defense using metabolomics and biochemical approaches. Sci. Rep. 2021, 11, 930. [Google Scholar] [CrossRef]
- Martini, F.; Beghini, G.; Zanin, L.; Varanini, Z.; Zamboni, A.; Ballottari, M. The potential use of Chlamydomonas reinhardtii and Chlorella sorokiniana as biostimulants on maize plants. Algal Res. 2021, 60, 102515. [Google Scholar] [CrossRef]
- Sharma, N.; Simon, D.P.; Diaz-Garza, A.M.; Fantino, E.; Messaabi, A.; Meddeb-Mouelhi, F.; Germain, H.; Desgagné-Penix, I. Diatoms Biotechnology: Various Industrial Applications for a Greener Tomorrow. Front. Mar. Sci. 2021, 8, 636613. [Google Scholar] [CrossRef]
- Guzmán-Murillo, M.A.; Ascencio, F.; Larrinaga-Mayoral, J.A. Germination and ROS detoxification in bell pepper (Capsicum annuum L.) under NaCl stress and treatment with microalgae extracts. Protoplasma 2013, 250, 33–42. [Google Scholar] [CrossRef]
- Abd Al Rhman, A.M.; Ahmed, H.; Mohamed, S. Effect of Moringa Extracts and Diatoms Foliar Applications on Washington Navel Orange and Murcott Tangor Transplant Performance. J. Hortic. Sci. Ornam. Plants 2018, 10, 28–40. [Google Scholar] [CrossRef]
- Piotrowski, K.; Romanowska-Duda, Z.; Messyasz, B. Cultivation of energy crops by ecological methods under the conditions of global climate and environmental changes with the use of diatom extract as a natural source of chemical compounds. Acta Physiol. Plant. 2020, 42, 146. [Google Scholar] [CrossRef]
- Woo, S.L.; Pepe, O. Microbial Consortia: Promising Probiotics as Plant Biostimulants for Sustainable Agriculture. Front. Plant Sci. 2018, 9, 7–12. [Google Scholar] [CrossRef]
- Win, T.T.; Barone, G.D.; Secundo, F.; Fu, P. Algal Biofertilizers and Plant Growth Stimulants for Sustainable Agriculture. Ind. Biotechnol. 2018, 14, 203–211. [Google Scholar] [CrossRef]
- Melchor, D.J.H.; Jimenez, J.C.; Lara, M.E.H.; Dendooven, L.; Moreno, R.M.; Villanueva, R.O.C. Phylogenetic and Morpho-logical Identification of a Photosynthetic Microbial Consortium of Potential Biotechnological Interest|Identificación Mor-fológica y Filogenética de un Consorcio Microbiano Fotosintético de Posible Interés Biotecnológico. Hidrobiologica 2016, 26, 311–321. [Google Scholar]
- Palacios, O.A.; López, B.R.; De-Bashan, L.E. Microalga Growth-Promoting Bacteria (MGPB): A formal term proposed for beneficial bacteria involved in microalgal–bacterial interactions. Algal Res. 2022, 61, 102585. [Google Scholar] [CrossRef]
- Tong, C.; Honda, K.; Derek, C. A review on microalgal-bacterial co-culture: The multifaceted role of beneficial bacteria towards enhancement of microalgal metabolite production. Environ. Res. 2023, 228, 115872. [Google Scholar] [CrossRef]
- Singh, S.K.; Sundaram, S.; Sinha, S.; Rahman, A.; Kapur, S. Recent advances in CO2 uptake and fixation mechanism of cyanobacteria and microalgae. Crit. Rev. Environ. Sci. Technol. 2016, 46, 1297–1323. [Google Scholar] [CrossRef]
- Dao, G.-H.; Wu, G.-X.; Wang, X.-X.; Zhang, T.-Y.; Zhan, X.-M.; Hu, H.-Y. Enhanced microalgae growth through stimulated secretion of indole acetic acid by symbiotic bacteria. Algal Res. 2018, 33, 345–351. [Google Scholar] [CrossRef]
- Lopez, B.R.; Palacios, O.A.; Bashan, Y.; Hernández-Sandoval, F.E.; De-Bashan, L.E. Riboflavin and lumichrome exuded by the bacterium Azospirillum brasilense promote growth and changes in metabolites in Chlorella sorokiniana under autotrophic conditions. Algal Res. 2019, 44, 101696. [Google Scholar] [CrossRef]
- Hernandez, J.-P.; De-Bashan, L.E.; Rodriguez, D.J.; Rodriguez, Y.; Bashan, Y. Growth promotion of the freshwater microalga Chlorella vulgaris by the nitrogen-fixing, plant growth-promoting bacterium Bacillus pumilus from arid zone soils. Eur. J. Soil Biol. 2009, 45, 88–93. [Google Scholar] [CrossRef]
- Yilmaz, E.; Sönmez, M. The role of organic/bio–fertilizer amendment on aggregate stability and organic carbon content in different aggregate scales. Soil Tillage Res. 2017, 168, 118–124. [Google Scholar] [CrossRef]
- Gemin, L.G.; Mógor, Á.F.; De Oliveira Amatussi, J.; Mógor, G. Microalgae associated to humic acid as a novel biostimulant improving onion growth and yield. Sci. Hortic. 2019, 256, 108560. [Google Scholar] [CrossRef]
- Horácio, E.H.; Zucareli, C.; Gavilanes, F.Z.; Yunes, J.S.; dos Santos Sanzovo, A.W.; Andrade, D.S. Co-inoculation of rhizobia, azospirilla and cyanobacteria for increasing common bean production. Semin. Ciências Agrárias 2020, 41, 2015–2028. [Google Scholar] [CrossRef]
- Gavilanes, F.Z.; Andrade, D.S.; Zucareli, C.; Horácio, E.H.; Yunes, J.S.; Barbosa, A.P.; Alves, L.A.R.; Cruzatty, L.G.; Maddela, N.R.; de Fátima Guimarães, M. Co-inoculation of Anabaena cylindrica with Azospirillum brasilense increases grain yield of maize hybrids. Rhizosphere 2020, 15, 100224. [Google Scholar] [CrossRef]
- Toribio, A.J.; Suárez-Estrella, F.; Jurado, M.M.; López-González, J.A.; Martínez-Gallardo, M.R.; López, M.J. Design and validation of cyanobacteria-rhizobacteria consortia for tomato seedlings growth promotion. Sci. Rep. 2022, 12, 1–15. [Google Scholar] [CrossRef]
- World Intellectual Property Organization (WIPO). Patent Landscape Report: Microalgae-Related Technologies; World Intellectual Property Organization (WIPO): Geneva, Switzerland, 2016; pp. 1–71. ISBN 978-92-805-2702-5. Available online: https://www.wipo.int/edocs/pubdocs/en/wipo_pub_947_5.pdf (accessed on 9 June 2023).
- Future Market Insights Demand for Microalgae in Fertilizers Sector Outlook (2023 to 2033). Available online: https://www.futuremarketinsights.com/reports/microalgae-fertilizers-sector (accessed on 11 June 2023).
- Murata, M.M.; Ito Morioka, L.R.; Da Silva Marques, J.B.; Bosso, A.; Suguimoto, H.H. What do patents tell us about microalgae in agriculture? AMB Express 2021, 11, 154. [Google Scholar] [CrossRef] [PubMed]
- Lafarga, T.; Pieroni, C.; D’imporzano, G.; Maggioni, L.; Adani, F.; Acién, G. Consumer Attitudes towards Microalgae Production and Microalgae-Based Agricultural Products: The Cases of Almería (Spain) and Livorno (Italy). Chemengineering 2021, 5, 27. [Google Scholar] [CrossRef]
- Ruiz-Nieto, Á.; Gómez-Serrano, C.; Acién, G.; Castro, A.J. Farmers’ Knowledge and Acceptance of Microalgae in Almería Greenhouse Horticulture. Agronomy 2022, 12, 2778. [Google Scholar] [CrossRef]
- US Public Law Agriculture Improvement Act of 2018; 2018, pp. 1–530. Available online: https://www.congress.gov/115/plaws/publ334/PLAW-115publ334.pdf (accessed on 12 June 2023).
- Traon, D.; Amat, L.; Zotz, F.; du Jardin, P. A Legal Framework for Plant Biostimulants and Agronomic Fertiliser Additives in the EU. Arcadia Int. 2014, 133, 1–105. Available online: https://ec.europa.eu/docsroom/documents/5403/attachments/1/translations/en/renditions/pdf (accessed on 14 June 2023).
- Li, J.; Van Gerrewey, T.; Geelen, D. A Meta-Analysis of Biostimulant Yield Effectiveness in Field Trials. Front. Plant Sci. 2022, 13, 836702. [Google Scholar] [CrossRef]

| Green Microalgae Strains | Biomass Growth Medium | Application Methods/Crop Plants or Seeds | Biostimulant Effects | Reference |
|---|---|---|---|---|
| Scenedesmus obliquus | Pretreated brewery wastewater | Centrifuged, ultrasonicated, and enzyme hydrolyzed biomass applied on watercress seeds (Lepidium sativum), mung bean (Vigna radiata), and cucumber (Cucumis sativus L.) |
| [30] |
| Tetradesmus obliquus; Chlorella protothecoides | Piggery wastewater | Fresh microalgal biomass applied on cucumber (Cucumis sativus), barley (Hordeum vulgare), wheat (Triticum aestivum), soybean (Glycine max), watercress (Nasturium officinale), and tomato (Lycopersicon esculentum) |
| [31] |
| Chlorella vulgaris UAL-1; Chlorella sp. UAL-2; Chlorella vulgaris UAL-3; Chlamydopodium fusiforme UAL-4 | Secondary-treated urban wastewater supplemented with concentrate | Aqueous extracts applied on watercress (Lepidium sativum L.), soybean (Glycine max L.), cucumber (Cucumis sativus L.), and wheat (Triticum aestivum L.) seeds |
| [32] |
| Chlorella vulgaris (13–1); Scenedesmus obliquus (B2-2) | Untreated municipal wastewater | Algal biomass (intact and broken cells) and culture supernatant applied on tomato (Solanum lycopersicum) and barely (Hordeum vulgare) seeds |
| [33] |
| Cyanobacteria Strain | Application Method | Crop Plant/Seeds | Biostimulant Effects | Reference |
|---|---|---|---|---|
| Nostoc sp. | Soil inoculation with fresh cyanobacteria biomass | Maize (Zea mays) |
| [79] |
| Chroococcidiopsis SM-04; Synechocystis SM-10 Phormidium SM-14; Leptolyngbya SM-13 | Inoculation with fresh cyanobacterial cultures in a hydroponic growth system | Wheat seeds (Triticum aestivum var Uqab-2000) |
| [80] |
| Anabaena vaginicola ISC90; Nostoc calcicola ISC89 | Spray with 1% cyanobacterial extracts on soil surface | Squash (Cucurbita maxima); Cucumber (Cucumis sativus L.); Tomato (Solanum lycopersicum L.) |
| [81] |
| Anabaena vaginicola ISB42; Cylindrospermum michailovskoense ISB45; Trichormus ellipsosporus ISB44 | Foliar spray with 1% cyanobacterial extracts | Peppermint (Mentha piperita) |
| [82] |
| Spirulina platensis | Foliar application with S. platensis-based commercial product | Eggplants (Solanum melongena) |
| [83] |
| Spirulina platensis | Foliar spray with polysaccharides extract | Pepper (Capsicum annuum var. andalus); Tomato (Solanum lycopersicum L. Var. metro) |
| [84] |
| Microcystis aeruginosa MKR 0105; Anabaena sp. PCC 7120 | Foliar spray with intact cyanobacterial monoculture | Willow plants (Salix viminalis L.) |
| [85] |
| Arthrospira platensis | Foliar applications with aqueous suspensions | Lettuce (Lactuca sativa) |
| [86] |
| Nostoc sp.; Tolypothrix sp.; Leptolyngbya sp. | Foliar spray with cyanobacterial hydrolysates in a hydroponic growth system | Basil (Ocimum basilicum L.) |
| [62] |
| Cyanobacteria Strain | Stress | Crop Plant | Application Method | Biostimulant Effects | Reference |
|---|---|---|---|---|---|
| Spirulina maxima | Salinity stress | Wheat grains (Triticum aestivum L. cv. Giza 94) | Irrigation with S. maxima aqueous extract |
| [87] |
| Aphanothece sp. BEA O935B; Arthrospira maxima MSS001 | Salinity stress | Tomato (Solanum lycopersicum) | Extract formulations applied to plants by soil drench |
| [88] |
| Roholtiella sp. QUC- CCM97 | Salinity stress | Bell pepper (Capsicum annuum L.) | Foliar application with aqueous extract |
| [89] |
| Oculatella lusitanica LEGE 161147 | Salinity stress | Lettuce (Lactuca sativa) | Plants grown in a mixture of vermiculite and perlite supplemented at the top with perlite containing O. lusitanica |
| [90] |
| Arthrospira platensis | Drought stress | Cotton plants (Gossypium barbadense L. cv. Giza 94) | Foliar application with cyanobacterial extract |
| [91] |
| Green Microalgae Strain | Application Method | Crop Plant/Seeds | Biostimulant Effects | Reference |
|---|---|---|---|---|
| Chlorella vulgaris | Fresh and dry biomass mixed with soil | Lettuce (Lactuca sativa) |
| [94] |
| Chlorella vulgaris | Irrigation with freeze dried biomass solutions | Broccoli (Brassica oleracea) |
| [95] |
| Acutodesmus dimorphus | Cellular extracts and dry biomass applied as seed primer and foliar spray | Tomato (Solanum lycopersicum) |
| [96] |
| Scenedesmus quadricauda | Irrigation with Hoagland solution containing S. quadricauda extract | Lettuce (Lactuca sativa) |
| [97] |
| Chlorella vulgaris; Scenedesmus quadricauda | Microalgal extract used as seed soaking solution in Petri dishes | Sugar beet seeds (Beta vulgaris subsp. vulgaris) |
| [53] |
| Chlorella ellipsoidea | Soil drench with an acid hydrolysis extract | Tomato (Solanum lycopersicum) |
| [59] |
| Chlorella vulgaris; Tetradesmus dimorphus | Foliar application of microalgal suspensions in amended soils | Common bean (Phaseolus vulgaris) |
| [50] |
| Desmodesmus subspicatus | Foliar application of aqueous extract | Tomato (Solanum lycopersicum) |
| [98] |
| Chlorella sp. (MACC-360 and MACC-38); Chlamydomonas reinhardtii (cc124) | Soil drench with live microalgae cells | Barrelclover (Medicago truncatula) |
| [48] |
| Chlorella vulgaris | Foliar spray with microalgal extract | Lettuce (Lactuca sativa) |
| [52] |
| Chlorella vulgaris | Foliar spray and soil drench with microalgal extract (with/without cowdung) | Tomato (Solanum lycopersicum) |
| [57] |
| Chlorella vulgaris | Foliar spray and soil drench with microalgal extract | Lettuce (Lactuca sativa) |
| [99] |
| Chlorella vulgaris | Foliar spray with algal cell liquid extract | Green gram (Vigna radiata L.) |
| [100] |
| Chlorella vulgaris | Foliar spray with Chlorella suspension (CS), Chlorella biomass (CB), and Chlorella-free supernatant (CFS) | “Red Russian” Kale (Brassica napus var. Pabularia) |
| [101] |
| Green Microalgae Strain | Stress | Crop Plant | Application Method | Biostimulant Effects | Reference |
|---|---|---|---|---|---|
| Dunaliella salina | Salt stress | Tomato (Solanum lycopersicum) | Foliar spray with polysaccharides extract |
| [84] |
| Chlorella vulgaris | Drought stress | Broccoli (Brassica oleracea) | Foliar spray with microalgal extract |
| [102] |
| Desmodesmus sp.; Dunaliella salina | Biotic stress | Tomato (Solanum lycopersicum) | Injection of microalgal polysaccharides extract |
| [103] |
| Chlorella sorokiniana; Chlamydomonas reinhardtii | Nitrogen deficit and drought stress | Maize (Zea mays) | Seedling soaked in a nutrient solution supplemented with algae freeze-dried biomass |
| [104] |
| Diatom Strain | Application Method | Crop Plant/Seeds | Biostimulant Effects | Reference |
|---|---|---|---|---|
| Phaeodactylum tricornutum | Seeds soaking with polysaccharides extract | Bell pepper (Capsicum annuum L.) |
| [106] |
| Diatom (Unspecified species) | Foliar spray with Diatoms suspension | Washington navel orange (Citrus sinensis) and Murcott Tangor (C. reticulata x sinensis) transplants |
| [107] |
| Navicula sp. | Direct watering, spraying, and watering + spraying with sonicated extract of Navicula sp. | Willow (Salix viminalis); Jerusalem artichoke (Helianthus tuberosus); Virginia mallow (Sida hermaphrodita) |
| [108] |
| Consortia/Combination | Crop Plant | Biostimulant Effects | Reference |
|---|---|---|---|
| Microalgae and cyanobacteria: Chlorella sp. + Scenedesmus sp. + Spirulina sp. + Synechocystis sp. | Tomato (Solanum lycopersicum) |
| [51] |
| Cyanobacteria and diazotrophic bacteria co-inoculation: Anabaena cylindrica + Rhizobium freirei + Rhizobium tropici + Azospirillum brasilense | Common bean (Phaseolus vulgaris) |
| [118] |
| Anabaena cylindrica + Azospirillum brasilense | Maize (Zea mays) |
| [119] |
| Cyanobacteria SAB-B866 (Nostocaceae Family) + Pseudomonas putida-BIO175 + Pantoea cypripedii- BIO175 | Tomato (Solanum lycopersicum San Pedro variety) |
| [120] |
| Microalgae and substances combination: Chlorella sp. and Vermicompost combination | Maize (Zea mays) |
| [121] |
| Scenedesmus subspicatus and humic acids combination | Mung bean (Vigna radiata); Onion (Allium cepa L.) |
| [122] |
| Product | Microalgae | Composition | Application Method | Biostimulant Effects | Country |
|---|---|---|---|---|---|
| AGRIALGAE® Premium Rooting | Microalgae combination |
| Soil application |
| Spain |
| AGRIALGAE® Premium Sprouting | Microalgae combination |
| Foliar application |
| Spain |
| AGRIALGAE® Premium Flowering | Microalgae combination |
| Foliar and soil application |
| Spain |
| AGRIALGAE® Premium Fruit Setting | Microalgae combination |
| Foliar and soil application |
| Spain |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chabili, A.; Minaoui, F.; Hakkoum, Z.; Douma, M.; Meddich, A.; Loudiki, M. A Comprehensive Review of Microalgae and Cyanobacteria-Based Biostimulants for Agriculture Uses. Plants 2024, 13, 159. https://doi.org/10.3390/plants13020159
Chabili A, Minaoui F, Hakkoum Z, Douma M, Meddich A, Loudiki M. A Comprehensive Review of Microalgae and Cyanobacteria-Based Biostimulants for Agriculture Uses. Plants. 2024; 13(2):159. https://doi.org/10.3390/plants13020159
Chicago/Turabian StyleChabili, Amer, Farah Minaoui, Zineb Hakkoum, Mountasser Douma, Abdelilah Meddich, and Mohammed Loudiki. 2024. "A Comprehensive Review of Microalgae and Cyanobacteria-Based Biostimulants for Agriculture Uses" Plants 13, no. 2: 159. https://doi.org/10.3390/plants13020159
APA StyleChabili, A., Minaoui, F., Hakkoum, Z., Douma, M., Meddich, A., & Loudiki, M. (2024). A Comprehensive Review of Microalgae and Cyanobacteria-Based Biostimulants for Agriculture Uses. Plants, 13(2), 159. https://doi.org/10.3390/plants13020159





