Total Soil CO2 Efflux from Drained Terric Histosols
Abstract
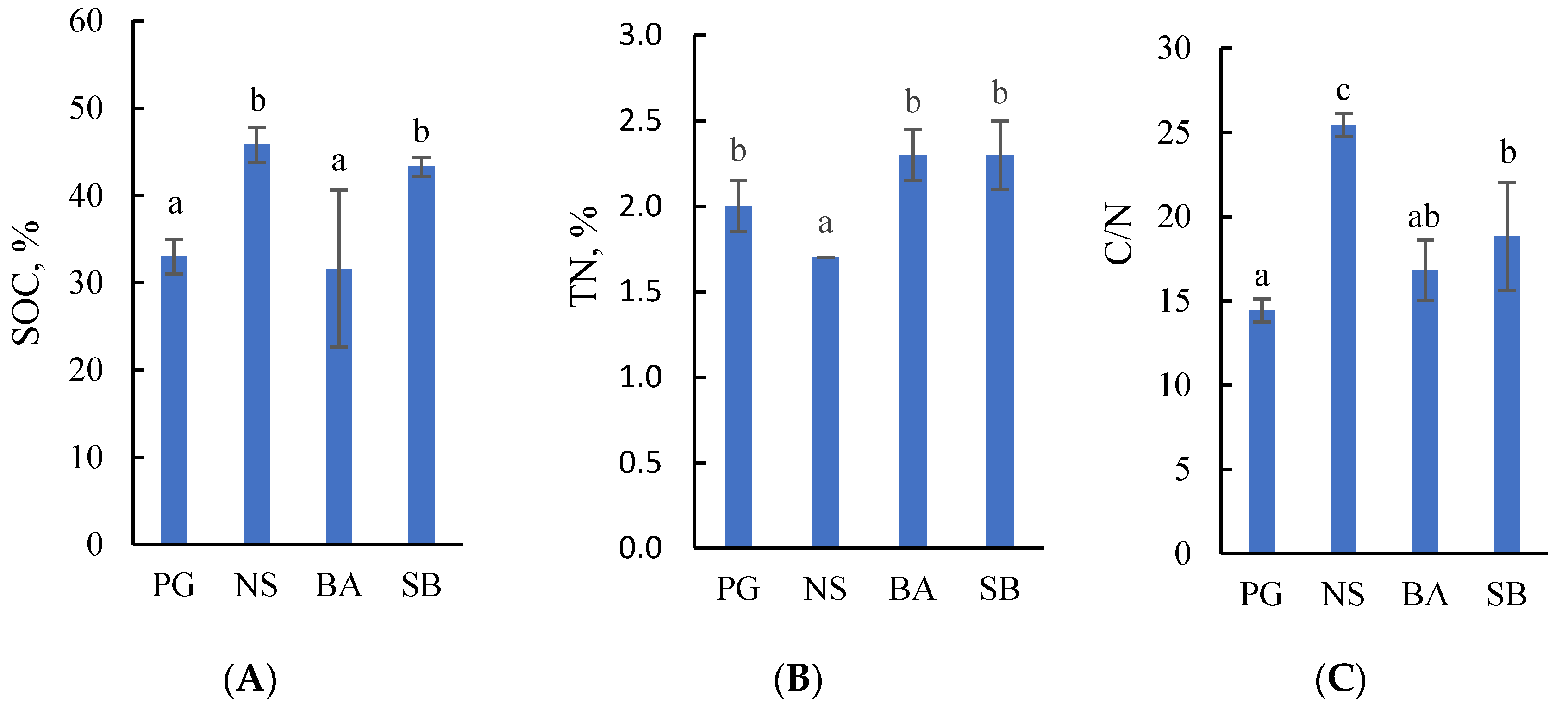
1. Introduction
2. Results
2.1. Concentrations of Soil Organic Carbon and Total Nitrogen in Drained Terric Histosols
2.2. Total Soil CO2 Efflux under Forest and Grassland Sites
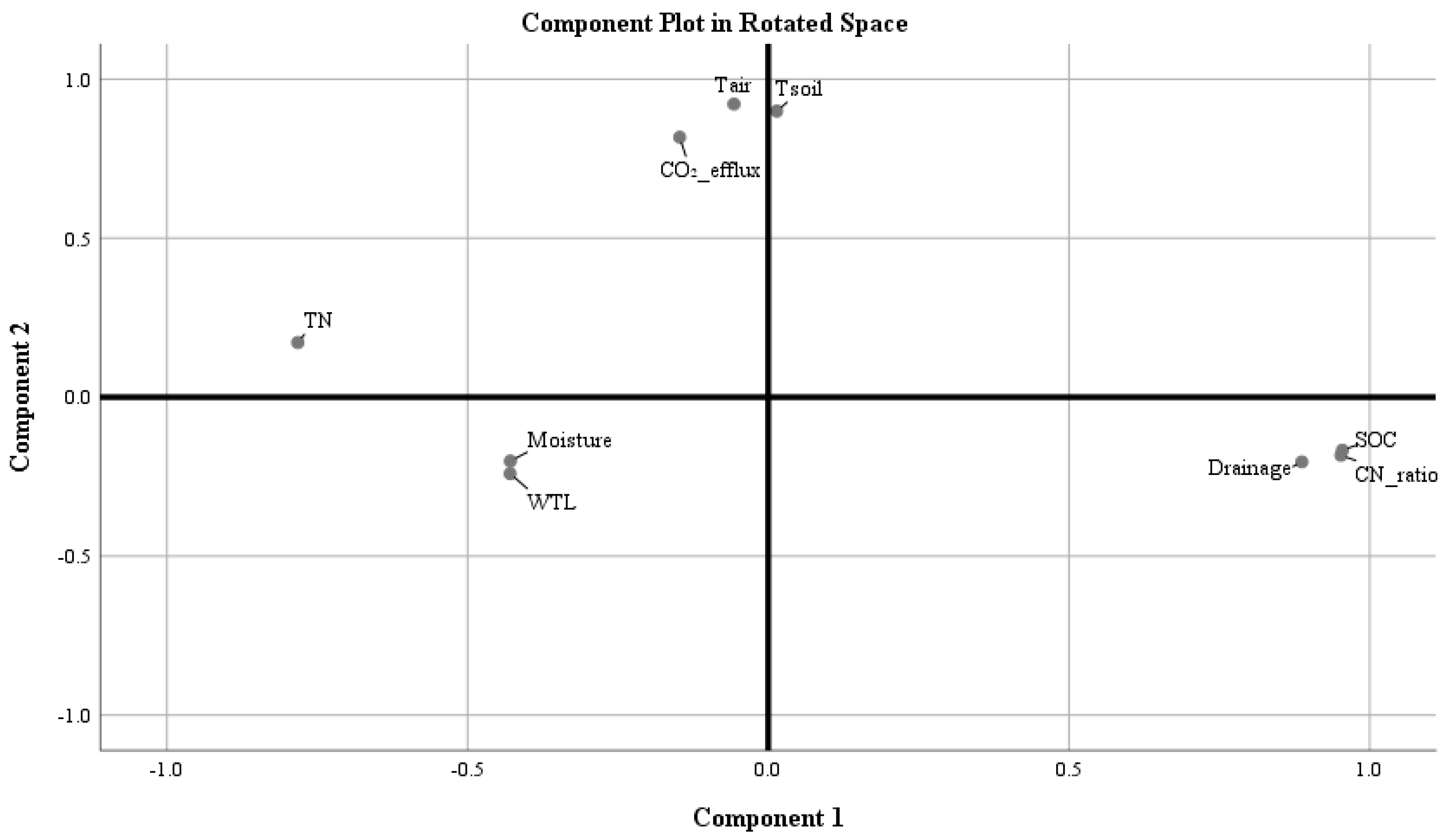
2.3. The Influence of Environmental Factors on Total Soil CO2 Efflux
3. Discussion
4. Material and Methods
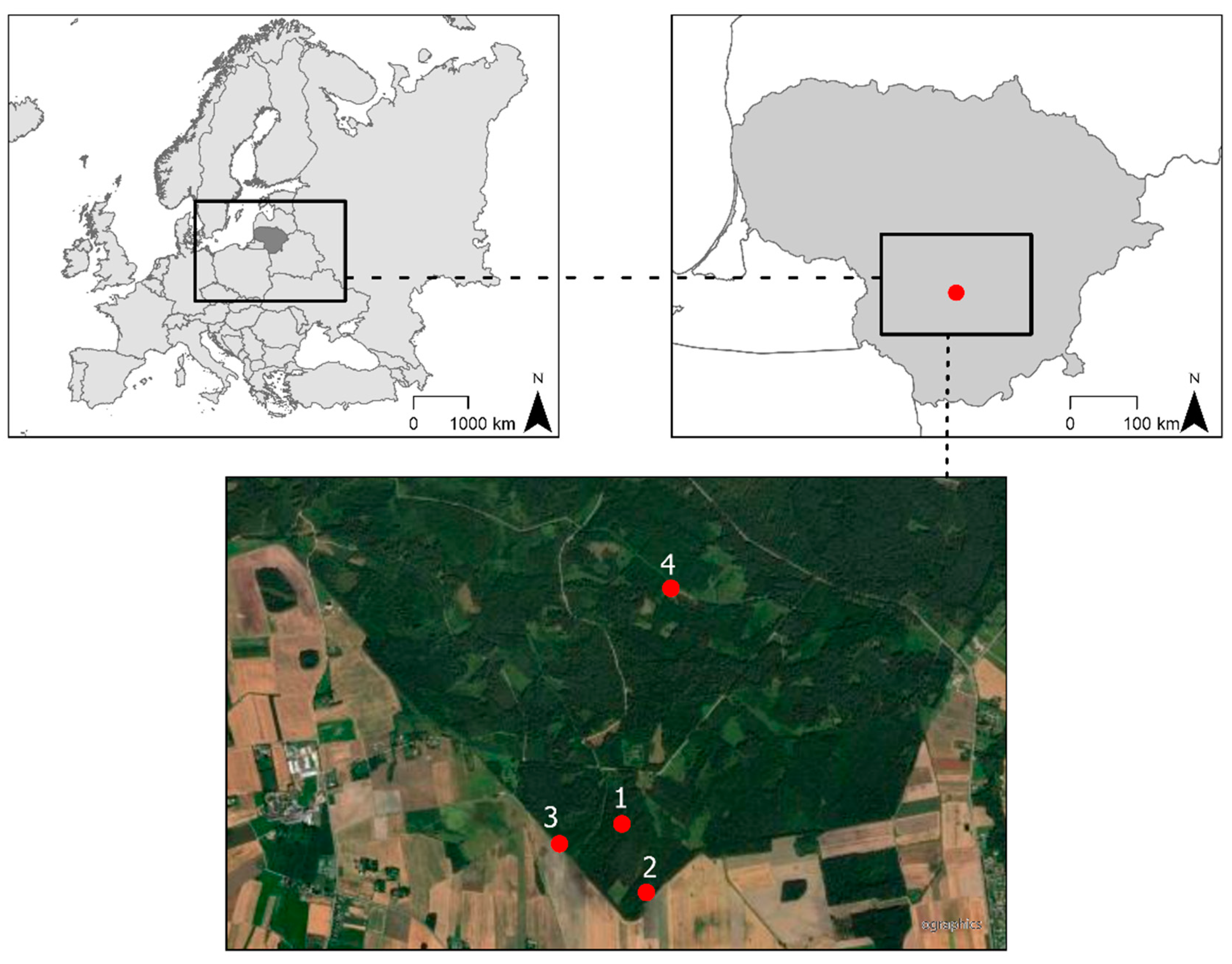
4.1. Study Sites
4.2. Measurements of Total Soil CO2 Efflux and Environmental Factors
4.3. Soil Sampling and Chemical Analysis
4.4. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Turunen, J.; Tomppo, E.; Tolonen, K.; Reinikainen, A. Estimating carbon accumulation rates of undrained mires in Finland—Application to boreal and subarctic regions. Holocene 2002, 12, 69–80. [Google Scholar] [CrossRef]
- Limpens, J.; Berendse, F.; Blodau, C.; Canadell, J.G.; Freeman, C.; Holden, J.; Roulet, N.; Rydin, H.; Schaepman-Strub, G. Peatlands and the carbon cycle: From local processes to global implications—A synthesis. Biogeosciences 2008, 5, 1475–1491. [Google Scholar] [CrossRef]
- Frolking, S.J.; Talbot, M.C.; Jones, C.C.; Treat, J.B.; Kauffman, E.S.; Tuittila, E.S.; Roulet, N. Peatlands in the Earth’s 21st century coupled climate-carbon system. Environ. Rev. 2011, 19, 371–396. [Google Scholar] [CrossRef]
- Tubiello, F.N.; Biancalani, R.; Salvatore, M.; Rossi, S.; Conchedda, G. A worldwide assessment of greenhouse gas emissions from drained organic soils. Sustainability 2016, 8, 371. [Google Scholar] [CrossRef]
- O’Brolchain, N.; Peters, J.; Tanneberger, F. CAP Policy Brief Peatlands in the New European Union Version 4.8. 2020. Available online: https://www.eurosite.org/wp-content/uploads/CAP-Policy-Brief-Peatlands-in-the-new-European-Union-Version-4.8.pdf (accessed on 15 March 2023).
- Taminskas, J.; Pileckas, M.; Šimanauskienė, R.; Linkevičienė, R. Lietuvos šlapynės: Klasifikacija ir sklaida. Baltica 2011, 24, 151–162. [Google Scholar]
- Slepetiene, A.; Amaleviciute-Volunge, K.; Slepetys, J.; Liaudanskiene, I.; Volungevicius, J. The Status of Pachiterric Histosols as Influence by Different Land Use. In Peat; Topcuoğlu, B., Turan, M., Eds.; IntechOpen: Rijeka, Croatia, 2018; pp. 49–73. [Google Scholar]
- Valatka, S.; Stoškus, A.; Pileckis, M. Lietuvos Durpynai: Kiek Jų Turime, Ar Racionaliai Naudojame? Gamtos Paveldo Fondas: Vilnius, Lithuania, 2018; p. 91. [Google Scholar]
- Buivydaitė, V.; Vaičys, M. Naujoji Lietuvos Dirvožemių Klasifikacija; Lietuvos Mokslas: Vilnius, Lithuania, 2001; p. 282. [Google Scholar]
- WRB (World Reference Base) of Soil Resources 2014 (Update 2015). International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; Food and Agriculture Organization of the United Nations: Rome, Italy, 2015; p. 203. [Google Scholar]
- Karazija, S. Lietuvos Miškų Tipai; Mokslas: Vilnius, Lithuania, 1988; p. 211. [Google Scholar]
- Armolaitis, K.; Beniušis, R.; Urbaitis, G. Sodiname Mišką; Lithuanian Research Centre for Agriculture And Forestry: Kaunas, Lithuania, 2016; p. 31. [Google Scholar]
- MoE/EPA/SFS. Lithuanian’s National Inventory Report 2019: Greenhouse Gas Emissions 1991–2017; The Ministry of Environment of the Republic of Lithuania: Vilnius, Lithuania, 2019; p. 565. [Google Scholar]
- IPCC. IPCC Guidelines for National Greenhouse Gas Inventories. In The Intergovernmental Panel on Climate Change; The National Greenhouse Gas Inventories Programme; Eggleston, H.S., Buendia, L., Miwa, K., Ngara, T., Tanabe, K., Eds.; IPCC: Hayama, Japan, 2006. [Google Scholar]
- IPCC. Climate Change 2014: Synthesis Report; Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Core Writing Team, Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014; p. 151. [Google Scholar]
- Bond-Lamberty, B.; Thomson, A. A global database of soil respiration data. Biogeosciences 2018, 7, 1915–1926. [Google Scholar] [CrossRef]
- Wang, C.; Yang, J. Rhizospheric and heterotrophic components of soil respiration in six Chinese temperate forests. Glob. Chang. Biol. 2007, 13, 123–131. [Google Scholar] [CrossRef]
- Ryan, M.G.; Lavigne, M.B.; Gower, S.T. Annual carbon cost of autotrophic respiration in boreal forest ecosystems in relation to species and climate. J. Geophys. Res. Atmos. 1997, 102, 28871–28883. [Google Scholar] [CrossRef]
- Dhital, D.; Manandhar, R.; Manandhar, P.; Maharjan, S. Soil CO2 efflux dynamics and its relationship with the environmental variables in a sub-tropical mixed forest. OJF 2022, 12, 312–336. [Google Scholar] [CrossRef]
- Swails, E.E.; Ardón, M.; Krauss, K.W.; Peralta, A.L.; Emanuel, R.E.; Helton, A.M.; Morse, J.L.; Gutenberg, L.; Cormier, N.; Shoch, D.; et al. Response of soil respiration to changes in soil temperature and water table level in drained and restored peatlands of the southeastern United States. Carbon Balance Manag. 2022, 17, 18. [Google Scholar] [CrossRef]
- Rustad, L.E.; Huntington, T.G.; Boone, R.D. Controls on soil respiration: Implications for climate change. Biogeochemistry 2000, 48, 1–6. [Google Scholar] [CrossRef]
- Ruehr, N.K.; Knohl, A.; Buchmann, N. Environmental variables controlling soil respiration on diurnal, seasonal and annual time-scales in a mixed mountain forest in Switzerland. Biogeochemistry 2010, 98, 153–170. [Google Scholar] [CrossRef]
- Oertel, C.; Matschullat, J.; Zurba, K.; Zimmermann, F.; Erasmi, S. Greenhouse gas emissions from soils—A review. Geochemistry 2016, 76, 327–352. [Google Scholar] [CrossRef]
- Vanags-Duka, M.; Bardule, A.; Butlers, A.; Upenieks, E.M.; Lazdinš, A.; Purvina, D.; Līcīte, I. GHG emissions from drainage ditches in peat extraction sites and peatland forests in hemiboreal Latvia. Land 2022, 11, 2233. [Google Scholar] [CrossRef]
- Conchedda, G.; Tubiello, F.N. Drainage of organic soils and GHG emissions: Validation with country data. Earth Syst. Sci. Data 2020, 12, 3113–3137. [Google Scholar] [CrossRef]
- Chen, R.; Senbayram, M.; Blagodatsky, S.; Myachina, O.; Dittert, K.; Lin, X.; Blagodatskaya, E.; Kuzyakov, Y. Soil C and N availability determine the priming effect: Microbial N mining and stoichiometric decomposition theories. Glob. Chang. Biol. 2014, 20, 2356–2367. [Google Scholar] [CrossRef]
- Craine, J.M.; Morrow, C.; Fierer, N. Microbial nitrogen limitation increases decomposition. Ecology 2007, 88, 2105–2113. [Google Scholar] [CrossRef]
- Vesterdal, L.; Schmidt, I.K.; Callesen, I.; Nilsson, L.O.; Gundersen, P. Carbon and nitrogen in forest floor and mineral soil under six common European tree species. For. Ecol. Manag. 2008, 255, 35–48. [Google Scholar] [CrossRef]
- Gregorich, E.G.; Carter, M.R.; Angers, D.A.; Monreal, C.M.; Ellert, B.H. Towards a minimum data set to assess soil organic matter quality in agricultural soils. Can. J. Soil Sci. 1994, 74, 367–385. [Google Scholar] [CrossRef]
- Burst, M.; Chauchard, S.; Dambrine, E.E.; Dupouey, J.; Amiaud, B. Distribution of soil properties along forest-grassland interfaces: Influence of permanent environmental factors or land-use after-effects? Agric. Ecosyst. Environ. 2020, 289, 106739. [Google Scholar] [CrossRef]
- Loisel, J.Z.; Yu, D.; Beilman, P.; Camill, J.; Alm, M.J.; Amesbury, D.; Anderson, S.; Andersson, C.; Bochicchio, K.; Barber, L.R.; et al. A database and synthesis of northern peatland soil properties and Holocene carbon and nitrogen accumulation. Holocene 2014, 24, 1028–1042. [Google Scholar] [CrossRef]
- Montanarella, L.; Jones, R.J.A.; Hiederer, R. The distribution of peatland Europe. Mires Peat. 2006, 1, 1–10. [Google Scholar]
- Küttim, M.; Küttim, L.; Pajula, R. The current state and ecological restoration of peatlands in Estonia. Dyn. Environnementales 2018, 342–349. [Google Scholar] [CrossRef]
- Aleksāns, O.; Ābelīte, G.; Bebre, I.; Brūniņa, L.; Bukovska, I.; Celma, S.; Dreimanis, I.; Grīnberga, L.; Jeņina, A.; Kalniņa, L.; et al. Life Restore Project Contribution to the Green House Emission Accounts in Latvia. In Sustainable And Responsible After-Use of Peat Extraction Areas; Priede, A., Gancone, A., Eds.; Baltijas Krasti: Riga, Latvia, 2019; pp. 21–54. [Google Scholar]
- Bekken, M.; Pirk, N.; Vatne, A.; Tallaksen, L.; Westermann, S.; Larsen, P.; Ibrom, A.; Steenberg Larsen, K.; Knutson, J.; Dörsch, P. Carbon dynamics of a controlled peatland restoration experiment in Norway. In Proceedings of the EGU General Assembly Conference Abstracts, Vienna, Austria, 24−28 April 2023; p. EGU-15754. [Google Scholar]
- Glukhova, T.; Ilyasov, D.; Vompersky, S.; Golovchenko, A.; Manucharova, N.; Stepanov, A. Soil respiration in alder swamp (Alnus glutinosa) in southern taiga of European Russia depending on microrelief. Forests 2021, 12, 496. [Google Scholar] [CrossRef]
- Ojanen, P.; Minkkinen, K.; Alm, J.; Penttilä, T. Soil–atmosphere CO2, CH4 and N2O fluxes in boreal forestry-drained peatlands. For. Ecol. Manag. 2010, 260, 411–421. [Google Scholar] [CrossRef]
- Maljanen, M.; Komulainen, V.M.; Hytönen, J.; Martikainen, P.; Laine, J. Carbon dioxide, nitrous oxide and methane dynamics in boreal organic agricultural soils with different soil characteristics. Soil Biol. Biochem. 2004, 36, 1801–1808. [Google Scholar] [CrossRef]
- Nykänen, H.; Alm, J.; Lång, K.; Silvola, J.; Martikainen, P. Emissions of CH4, N2O and CO2 from a virgin fen and a fen drained for grassland in Finland. J. Biogeo. 1995, 22, 1149–1155. [Google Scholar] [CrossRef]
- Berglund, Ö.; Kätterer, T.; Meurer, K.H.E. Emissions of CO2, N2O and CH4 from Cultivated and Set Aside Drained Peatland in Central Sweden. Front. Environ. Sci. 2021, 9, 630721. [Google Scholar] [CrossRef]
- Bārdule, A.; Gerra-Inohosa, L.; Kļaviņš, I.; Kļaviņa, Z.; Bitenieks, K.; Butlers, A.; Lazdiņš, A.; Lībiete, Z. Variation in the Mercury Concentrations and Greenhouse Gas Emissions of Pristine and Managed Hemiboreal Peatlands. Land 2022, 11, 1414. [Google Scholar] [CrossRef]
- Alm, J.; Saarnio, S.; Nykänen, H.; Silvola, J.; Martikainen, P. Winter CO2, CH4 and N2O fluxes on some natural and drained boreal peatlands. Biogeochemistry 1999, 44, 163–186. [Google Scholar] [CrossRef]
- Kochiieru, M.; Veršulienė, A.; Feiza, V.; Feizienė, D. Trend for soil CO2 efflux in grassland and forest land in relation with meteorological conditions and root parameters. Sustainability 2023, 15, 7193. [Google Scholar] [CrossRef]
- Peichl, M.; Leava, N.A.; Kiely, G. Above- and belowground ecosystem biomass, carbon and nitrogen allocation in recently afforested grassland and adjacent intensively managed grassland. Plant Soil 2012, 350, 281–296. [Google Scholar] [CrossRef]
- Curiel, Y.J.; Janssens, I.A.; Carrara, A.; Meiresonne, L.; Ceulemans, R. Interactive effects of temperature and precipitation on soil respiration in a temperate maritime pine forest. Tree Physiol. 2003, 23, 1263–1270. [Google Scholar]
- Valujeva, K.; Pilecka-Ulcugaceva, J.; Liepa, S.; Grinfelde, I. Towards An Improved Understanding Of Greenhouse Gas Emissions From Organic Soils Used For Agricultural Production. In Proceedings of the 21st International Multidisciplinary Scientific GeoConference SGEM 2021, Albena, Bulgaria, 16–22 August 2021; pp. 561–567. [Google Scholar]
- Hursh, A.; Ballantyne, A.; Cooper, L.; Maneta, M.; Kimball, J.; Watts, J. The sensitivity of soil respiration to soil temperature, moisture, and carbon supply at the global scale. Glob. Chang. Biol. 2017, 23, 2090–2103. [Google Scholar] [CrossRef] [PubMed]
- Pietikäinen, J.; Pettersson, M.; Bååth, E. Comparison of temperature effects on soil respiration and bacterial and fungal growth rates. FEMS Microbiol. Ecol. 2005, 52, 49–58. [Google Scholar] [CrossRef]
- Vose, J.; Bolstad, P. Biotic and abiotic factors regulating forest floor CO2 flux across a range of forest age classes in the southern Appalachians. Pedobiologia 2007, 50, 577–587. [Google Scholar] [CrossRef]
- Galvonaite, A.; Valiukas, D.; Kilpys, J.; Kitrienė, Z.; Misiūnienė, M. Climate Atlas of Lithuania; Lithuanian Hydrometeorological Service: Vilnius, Lithuania, 2013; p. 175. [Google Scholar]
- World Imagery. Available online: https://services.arcgisonline.com/ArcGIS/rest/services/World_Imagery/MapServer (accessed on 9 August 2023).
- Efrain Maps. Available online: http://tapiquen-sig.jimdo.com (accessed on 9 August 2023).
- Beniušis, R.; Karazija, S.; Kuliešis, A.; Raguotis, A.; Rutkauskas, A.; Vaičys, M. Miško Augaviečių Tipai; Lututė: Kaunas, Lithuania, 2006; p. 95. [Google Scholar]
- Lovelock, C.E.; Ruess, R.W.; Feller, I.C. CO2 efflux from cleared mangrove peat. PLoS ONE 2011, 6, e21279. [Google Scholar] [CrossRef]
- Alm, J.; Shurpali, N.J.; Tuittila, E.-S.; Laurila, T.; Maljanen, M.; Saarnio, S.; Minkkinen, K. Methods for determining emission factors for the use of peat and peatlands—Flux measurements and modelling. Boreal Environ. Res. 2007, 12, 85–100. [Google Scholar]
- ISO 10694; Soil Quality-Determination of Organic and Total Carbon after Dry Combustion (Elementary Analysis). International Organization for Standardization: Geneva, Switzerland, 1995; p. 7.
- ISO 13878; Soil Quality—Determination of Total Nitrogen Content by Dry Combustion (Elemental Analysis). International Organization for Standardization: Geneva, Switzerland, 1998; p. 5.
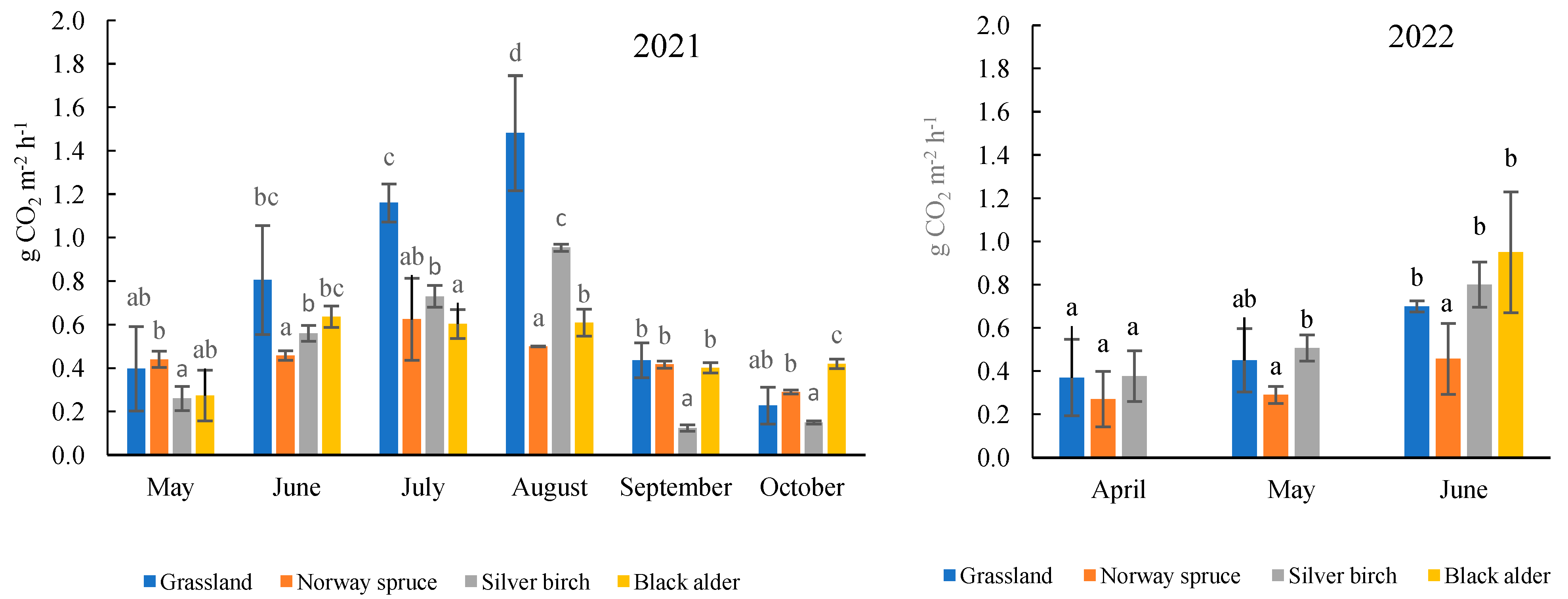

| 2021 | 2022 | ||||
|---|---|---|---|---|---|
| Spring a | Summer b | Autumn c | Spring d | Summer e | |
| Norway spruce | |||||
| Air temperature, °C | 17 ± 0 | 26 ± 2 | 12 ± 1 | 14 ± 3 | 15 ± 0 |
| Soil temperature, °C | 11 ± 1.0 | 19 ± 1 | 10 ± 1 | 10 ± 2 | 14 ± 0 |
| Soil moisture, % | 20 ± 5 | 16 ± 4 | 19 ± 5 | 20 ± 7 | 22 ± 2 |
| WTL, cm | −40 ± 4 | −91 ± 9 | −95 ± 9 | −67 ± 9 | −42 ± 15 |
| Silver birch | |||||
| Air temperature, °C | 19 ± 0 | 26 ± 4 | 16 ± 1 | 13 ± 9 | 25 ± 0 |
| Soil temperature, °C | 12.6 ± 0.2 | 17.3 ± 2.5 | 11.0 ± 2.8 | 6 ± 6 | 15 ± 4 |
| Soil moisture, % | 22 ± 3 | 19 ± 5 | 20 ± 6 | 28 ± 1 | 16 ± 4 |
| WTL, cm | −100 ± 0 | −140 ± 0 | −130 ± 0 | −130 ± 0 | −88 ± 1 |
| Black alder | |||||
| Air temperature, °C | 15 ± 0 | 27 ± 3 | 17 ± 2 | 15 ± 8 | 25 ± 0 |
| Soil temperature, °C | 13 ± 0 | 19 ± 2 | 13 ± 4 | 10 ± 1 | 14 ± 0 |
| Soil moisture, % | 29 ± 5 | 24 ± 1 | 23 ± 1 | 37 ± 12 | 30 ± 1 |
| WTL, cm | −40 ± 15 | −87 ± 32 | −101 ± 14 | −47 ± 9 | −30 ± 7 |
| Perennial grassland | |||||
| Air temperature, °C | 19 ± 0 | 29 ± 4 | 18 ± 6 | 15 ± 2 | 19 ± 0 |
| Soil temperature, °C | 7 ± 1 | 18 ± 3 | 13 ± 5 | 6 ± 2 | 10 ± 2 |
| Soil moisture, % | 80 ± 35 | 32 ± 15 | 64 ± 21 | 82 ± 20 | 100 ± 0 |
| WTL, cm | −52 ± 10 | −67 ± 20 | −46 ± 19 | −28 ± 11 | 0 ± 0 |
| CO2 Efflux | T Air | T Soil | WTL | Moisture | SOC | TN | C/N | |
|---|---|---|---|---|---|---|---|---|
| CO2 efflux | 1.0 | 0.7 * | 0.7 * | −0.3 * | −0.4 * | −0.2 | 0.2 | −0.2 |
| T air | 1.0 | 0.8 * | −0.1 | −0.1 | −0.2 * | 0.2 * | −0.2 * | |
| T soil | 1.0 | −0.1 * | −0.1 | −0.2 * | 0.1 | −0.2 * | ||
| WTL | 1.0 | 0.4 * | −0.3 * | 0.2 | −0.2 * | |||
| Moisture | 1.0 | −0.4* | 0.2 * | −0.2 * | ||||
| SOC | 1.0 | −0.7 * | 0.9 * | |||||
| TN | 1.0 | 0.9 | ||||||
| C/N | 1.0 |
| Forest Zone | Country | Vegetation Type | Study Period | Soil CO2 Efflux | Reference |
|---|---|---|---|---|---|
| Boreal | Finland | Grassland | Growing | 4.84–7.00 t CO2 ha−1 y−1 (from ~0.50 to ~1.00 g CO2 m−2 h−1) | [39] |
| Boreal | Finland | Grassland | Winter | 130–150 kg CO2 ha−1 y−1 (from ~0.00 to ~0.40 g CO2 m−2 h−1) | |
| Boreal | Finland | Grassland | Growing | 0.35 g CO2 m−2 h−1 | [38] |
| Boreal | Finland | Cropland | Growing | 0.33 g CO2 m−2 h−1 | |
| Boreal | Russia | Forest | Growing | 0.33 g CO2 m−2 h−1 | [36] |
| Boreal | Finland | Forest | Annual | 2000 g CO2 m−2 y−1 (0.23 g CO2 m−2 h−1) | [37] |
| Boreal | Finland | Forest | Annual | 3000 g CO2 m−2 y−1 (0.34 g CO2 m−2 h−1) | |
| Hemi-boreal | Sweden | Grassland | Growing | 0.74 g CO2 m−2 h−1 | [40] |
| Hemi-boreal | Sweden | Grassland | Spring | 0.90 g CO2 m−2 h−1 | |
| Hemi-boreal | Sweden | Grassland | Summer | 1.12 g CO2 m−2 h−1 | |
| Hemi-boreal | Sweden | Grassland | Autumn | 0.40 g CO2 m−2 h−1 | |
| Hemi-boreal | Latvia | Grassland | May-August | 0.35 g CO2-C m−2 h−1 (1.28 g CO2 m−2 h−1) | [41] |
| Hemi-boreal | Latvia | Forest | May-August | 0.23 CO2-C m−2 h−1 (0.84 g CO2 m−2 h−1) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vigricas, E.; Čiuldienė, D.; Armolaitis, K.; Valujeva, K.; Laiho, R.; Jauhiainen, J.; Schindler, T.; Bārdule, A.; Lazdiņš, A.; Butlers, A.; et al. Total Soil CO2 Efflux from Drained Terric Histosols. Plants 2024, 13, 139. https://doi.org/10.3390/plants13010139
Vigricas E, Čiuldienė D, Armolaitis K, Valujeva K, Laiho R, Jauhiainen J, Schindler T, Bārdule A, Lazdiņš A, Butlers A, et al. Total Soil CO2 Efflux from Drained Terric Histosols. Plants. 2024; 13(1):139. https://doi.org/10.3390/plants13010139
Chicago/Turabian StyleVigricas, Egidijus, Dovilė Čiuldienė, Kęstutis Armolaitis, Kristine Valujeva, Raija Laiho, Jyrki Jauhiainen, Thomas Schindler, Arta Bārdule, Andis Lazdiņš, Aldis Butlers, and et al. 2024. "Total Soil CO2 Efflux from Drained Terric Histosols" Plants 13, no. 1: 139. https://doi.org/10.3390/plants13010139
APA StyleVigricas, E., Čiuldienė, D., Armolaitis, K., Valujeva, K., Laiho, R., Jauhiainen, J., Schindler, T., Bārdule, A., Lazdiņš, A., Butlers, A., Kazanavičiūtė, V., Belova, O., Kamil-Sardar, M., & Soosaar, K. (2024). Total Soil CO2 Efflux from Drained Terric Histosols. Plants, 13(1), 139. https://doi.org/10.3390/plants13010139






