Seed Size, Not Dispersal Syndrome, Determines Potential for Spread of Ricefield Weeds by Gulls
Abstract
1. Introduction
2. Results
2.1. Survival
2.2. Retention Time
2.3. Germinability
2.4. Germination Time
3. Discussion
3.1. Retrieval, Viability and Retention Time of Seeds in Relation to Plant Traits
3.2. Characteristics of LBBG That Make Them Good Seed Dispersal Vectors
3.3. Ecological and Management Implications of Weed Dispersal by LBBG
4. Materials and Methods
4.1. Plant Species and Seed Collection
4.2. Gull Captures
4.3. Feeding Trial
4.4. Seed Retrieval and Germination Experiment
4.5. Statistical Analyses
4.6. Seed Survival
4.7. Retention Time
4.8. Germinability
4.9. Germination Time
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Levin, S.A.; Muller-Landau, H.C.; Nathan, R.; Chave, J. The ecology and evolution of seed dispersal: A theoretical perspective. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 575–604. [Google Scholar] [CrossRef]
- Kendrick, G.A.; Orth, R.J.; Statton, J.; Hovey, R.; Ruiz Montoya, L.; Lowe, R.J.; Krauss, S.L.; Sinclair, E.A. Demographic and genetic connectivity: The role and consequences of reproduction, dispersal and recruitment in seagrasses. Biol. Rev. 2017, 92, 921–938. [Google Scholar] [CrossRef]
- Levine, J.M.; Murrell, D.J. The community-level consequences of seed dispersal patterns. Annu. Rev. Ecol. Evol. Syst. 2003, 34, 549–574. [Google Scholar] [CrossRef]
- Willson, M.F.; Traveset, A. The ecology of seed dispersal. In Seeds: The Ecology of Regeneration in Plant Communities; Fenner, M., Ed.; CABI: Wallingford, UK, 2000; pp. 85–110. [Google Scholar]
- Rogers, H.S.; Donoso, I.; Traveset, A.; Fricke, E.C. Cascading impacts of seed disperser loss on plant communities and ecosystems. Annu. Rev. Ecol. Evol. Syst. 2021, 52, 641–666. [Google Scholar] [CrossRef]
- McConkey, K.R.; Prasad, S.; Corlett, R.T.; Campos-Arceiz, A.; Brodie, J.F.; Rogers, H.; Santamaria, L. Seed dispersal in changing landscapes. Biol. Conserv. 2012, 146, 1–13. [Google Scholar] [CrossRef]
- Sales, L.P.; Kissling, W.D.; Galetti, M.; Naimi, B.; Pires, M.M. Climate change reshapes the eco-evolutionary dynamics of a Neotropical seed dispersal system. Glob. Ecol. Biogeogr. 2021, 30, 1129–1138. [Google Scholar] [CrossRef]
- Wenny, D.G.; Sekercioglu, C.; Cordeiro, N.J.; Rogers, H.S.; Kelly, D. Seed dispersal by fruit-eating birds. In Why Birds Matter: Avian Ecological Function Ecosystem Services; Sekercioglu, Ç.H., Wenny, D.G., Whelan, C.J., Eds.; The University of Chicago Press: Danvers, MA, USA, 2016; pp. 107–145. [Google Scholar]
- Green, A.J.; Baltzinger, C.; Lovas-Kiss, A. Plant dispersal syndromes are unreliable, especially for predicting zoochory and long-distance dispersal. Oikos 2022, 2022, e08327. [Google Scholar] [CrossRef]
- Green, A.J.; Elmberg, J.; Lovas-Kiss, Á. Beyond scatter-hoarding and frugivory: European corvids as overlooked vectors for a broad range of plants. Front. Ecol. Environ. 2019, 7, 133. [Google Scholar] [CrossRef]
- Green, A.J.; Lovas-Kiss, Á.; Reynolds, C.; Sebastián-González, E.; Silva, G.G.; van Leeuwen, C.H.; Wilkinson, D.M. Dispersal of aquatic and terrestrial organisms by waterbirds: A review of current knowledge and future priorities. Freshw. Biol. 2023, 68, 173–190. [Google Scholar] [CrossRef]
- Martín-Vélez, V.; Lovas-Kiss, A.; Sánchez, M.I.; Green, A.J. Endozoochory of the same community of plants lacking fleshy fruits by storks and gulls. J. Veg. Sci. 2021, 32, e12967. [Google Scholar] [CrossRef]
- Martín-Velez, V.; van Leeuwen, C.H.A.; Sánchez, M.I.; Hortas, F.; Shamoun-Baranes, J.; Thaxter, C.B.; Lens, L.; Camphuysen, C.J.; Green, A.J. Spatial patterns of weed dispersal by wintering gulls within and beyond an agricultural landscape. J. Ecol. 2021, 109, 1947–1958. [Google Scholar] [CrossRef]
- Urgyán, R.; Lukács, B.A.; Fekete, R.; Molnár, A.V.; Nagy, A.; Vincze, O.; Green, A.J.; Lovas-Kiss, Á. Plants dispersed by a non-frugivorous migrant change throughout the annual cycle. Glob. Ecol. Biogeogr. 2022, 32, 70–82. [Google Scholar] [CrossRef]
- Reynolds, C.; Cumming, G.S. Seed traits and bird species influence the dispersal parameters of wetland plants. Freshw. Biol. 2016, 61, 1157–1170. [Google Scholar] [CrossRef]
- Lovas-Kiss, A.; Vincze, O.; Kleyheeg, E.; Sramko, G.; Laczko, L.; Fekete, R.; Molnar, A.V.; Green, A.J. Seed mass, hardness, and phylogeny explain the potential for endozoochory by granivorous waterbirds. Ecol. Evol. 2020, 10, 1413–1424. [Google Scholar] [CrossRef]
- Green, A.J. The importance of waterbirds as an overlooked pathway of invasion for alien species. Divers. Distrib. 2016, 22, 239–247. [Google Scholar] [CrossRef]
- Green, A.J.; Soons, M.; Brochet, A.-L.; Kleyheeg, E. Dispersal of plants by waterbirds. In Why Birds Matter: Avian Ecological Function Ecosystem Services; Sekercioglu, Ç.H., Wenny, D.G., Whelan, C.J., Eds.; University of Chicago Press: Chicago, IL, USA, 2016; Volume 147. [Google Scholar]
- García-Álvarez, A.; van Leeuwen, C.H.; Luque, C.J.; Hussner, A.; Vélez-Martín, A.; Pérez-Vázquez, A.; Green, A.J.; Castellanos, E.M. Internal transport of alien and native plants by geese and ducks: An experimental study. Freshw. Biol. 2015, 60, 1316–1329. [Google Scholar] [CrossRef]
- Rendón, M.A.; Green, A.J.; Aguilera, E.; Almaraz, P. Status, distribution and long-term changes in the waterbird community wintering in Doñana, South–West Spain. Biol. Conserv. 2008, 141, 1371–1388. [Google Scholar] [CrossRef]
- Mundkur, T.; Nagy, S. Waterbird Population Estimates: Summary Report; Report no. 9058820009; Wetlands International: Wageningen, The Netherlands, 2012. [Google Scholar]
- Martín-Vélez, V.; Mohring, B.; van Leeuwen, C.H.A.; Shamoun-Baranes, J.; Thaxter, C.B.; Baert, J.M.; Camphuysen, C.J.; Green, A.J. Functional connectivity network between terrestrial and aquatic habitats by a generalist waterbird, and implications for biovectoring. Sci. Total Environ. 2020, 705, 135886. [Google Scholar] [CrossRef]
- Lovas-Kiss, A.; Sánchez, M.I.; Molnar, A.V.; Valls, L.; Armengol, X.; Mesquita-Joanes, F.; Green, A.J. Crayfish invasion facilitates dispersal of plants and invertebrates by gulls. Freshw. Biol. 2018, 63, 392–404. [Google Scholar] [CrossRef]
- Lovas-Kiss, A.; Sánchez, M.I.; Wilkinson, D.M.; Coughlan, N.E.; Alves, J.A.; Green, A.J. Shorebirds as important vectors for plant dispersal in Europe. Ecography 2019, 42, 956–967. [Google Scholar] [CrossRef]
- Calvino-Cancela, M. Gulls (Laridae) as frugivores and seed dispersers. Plant Ecol. 2011, 212, 1149–1157. [Google Scholar] [CrossRef]
- Farmer, J.A.; Webb, E.B.; Pierce, R.A.; Bradley, K.W. Evaluating the potential for weed seed dispersal based on waterfowl consumption and seed viability. Pest Manag. Sci. 2017, 73, 2592–2603. [Google Scholar] [CrossRef] [PubMed]
- Benvenuti, S. Weed seed movement and dispersal strategies in the agricultural environment. Weed Biol. Manag. 2007, 7, 141–157. [Google Scholar] [CrossRef]
- Van Leeuwen, C.H.; Soons, M.B.; Vandionant, L.G.; Green, A.J.; Bakker, E.S. Seed dispersal by waterbirds: A mechanistic understanding by simulating avian digestion. Ecography 2023, 2023, e06470. [Google Scholar] [CrossRef]
- Figuerola, J.; Charalambidou, I.; Santamaria, L.; Green, A.J. Internal dispersal of seeds by waterfowl: Effect of seed size on gut passage time and germination patterns. Naturwissenschaften 2010, 97, 555–565. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Kawakami, K.; Masaki, T. Allometric scaling of seed retention time in seed dispersers and its application to estimation of seed dispersal potentials of theropod dinosaurs. Oikos 2019, 128, 836–844. [Google Scholar] [CrossRef]
- Costea, M.; El Miari, H.; Laczkó, L.; Fekete, R.; Molnár, A.V.; Lovas-Kiss, Á.; Green, A.J. The effect of gut passage by waterbirds on the seed coat and pericarp of diaspores lacking “external flesh”: Evidence for widespread adaptation to endozoochory in angiosperms. PLoS ONE 2019, 14, e0226551. [Google Scholar] [CrossRef]
- Almeida, B.A.; Lukács, B.A.; Lovas-Kiss, Á.; Reynolds, C.; Green, A.J. Functional traits drive dispersal interactions between European waterfowl and seeds. Front. Plant Sci. 2022, 12, 795288. [Google Scholar] [CrossRef]
- Brochet, A.-L.; Guillemain, M.; Gauthier-Clerc, M.; Fritz, H.; Green, A.J. Endozoochory of Mediterranean aquatic plant seeds by teal after a period of desiccation: Determinants of seed survival and influence of retention time on germinability and viability. Aquat. Bot. 2010, 93, 99–106. [Google Scholar] [CrossRef]
- Eriksson, O.; Jakobsson, A. Abundance, distribution and life histories of grassland plants: A comparative study of 81 species. J. Ecol. Environ. 1998, 86, 922–933. [Google Scholar] [CrossRef]
- Jakobsson, A.; Eriksson, O. A comparative study of seed number, seed size, seedling size and recruitment in grassland plants. Oikos 2000, 88, 494–502. [Google Scholar] [CrossRef]
- Phartyal, S.S.; Rosbakh, S.; Ritz, C.; Poschlod, P. Ready for change: Seed traits contribute to the high adaptability of mudflat species to their unpredictable habitat. J. Veg. Sci. 2020, 31, 331–342. [Google Scholar] [CrossRef]
- Kleyheeg, E.; Claessens, M.; Soons, M.B. Interactions between seed traits and digestive processes determine the germinability of bird-dispersed seeds. PLoS ONE 2018, 13, e0195026. [Google Scholar] [CrossRef]
- Kleyheeg, E.; Nolet, B.A.; Otero-Ojea, S.; Soons, M.B. A mechanistic assessment of the relationship between gut morphology and endozoochorous seed dispersal by waterfowl. Ecol. Evol. 2018, 8, 10857–10867. [Google Scholar] [CrossRef] [PubMed]
- Kleyheeg, E.; Van Leeuwen, C.H.; Morison, M.A.; Nolet, B.A.; Soons, M.B. Bird-mediated seed dispersal: Reduced digestive efficiency in active birds modulates the dispersal capacity of plant seeds. Oikos 2015, 124, 899–907. [Google Scholar] [CrossRef]
- Van Rees, C.B.; Aragonés, D.; Bouten, W.; Thaxter, C.B.; Stienen, E.W.; Bustamante, J.; Green, A.J. Dynamic space use of Andalusian rice fields by Lesser Black-Backed Gulls (Larus fuscus) is driven by flooding pattern. Ibis 2021, 163, 1252–1270. [Google Scholar] [CrossRef]
- Bullock, J.M.; Mallada González, L.; Tamme, R.; Götzenberger, L.; White, S.M.; Pärtel, M.; Hooftman, D.A. A synthesis of empirical plant dispersal kernels. J. Ecol. Environ. 2017, 105, 6–19. [Google Scholar] [CrossRef]
- Tamme, R.; Götzenberger, L.; Zobel, M.; Bullock, J.M.; Hooftman, D.A.; Kaasik, A.; Pärtel, M. Predicting species’ maximum dispersal distances from simple plant traits. Ecol. Evol. 2014, 95, 505–513. [Google Scholar] [CrossRef]
- Rock, P.; Spence, I. Lesser Black-backed gull. In The Migration Atlas: Movements of the Birds of Britain and Ireland; Wernham, C., Toms, M., Marchant, J., Clark, J., Siriwardena, G., Eds.; T & AD Poyser: London, UK, 2002; pp. 365–368. [Google Scholar]
- Klaassen, R.H.; Ens, B.J.; Shamoun-Baranes, J.; Exo, K.-M.; Bairlein, F. Migration strategy of a flight generalist, the Lesser Black-Backed Gull Larus fuscus. Behav. Ecol. Sociobiol. 2012, 23, 58–68. [Google Scholar] [CrossRef]
- Klaassen, R.H.; Strandberg, R.; Hake, M.; Alerstam, T. Flexibility in daily travel routines causes regional variation in bird migration speed. Behav. Ecol. Sociobiol. 2008, 62, 1427–1432. [Google Scholar] [CrossRef]
- Baert, J.M.; Stienen, E.W.; Heylen, B.C.; Kavelaars, M.M.; Buijs, R.-J.; Shamoun-Baranes, J.; Lens, L.; Müller, W. High-resolution GPS tracking reveals sex differences in migratory behaviour and stopover habitat use in the Lesser Black-Backed Gull Larus fuscus. Sci. Rep. Ist. Super. Sanita 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Coulson, J.C.; Coulson, B.A. Lesser Black-backed Gulls Larus fuscus nesting in an inland urban colony: The importance of earthworms (Lumbricidae) in their diet. Bird Study 2008, 55, 297–303. [Google Scholar] [CrossRef]
- Gyimesi, A.; Boudewijn, T.J.; Buijs, R.-J.; Shamoun-Baranes, J.Z.; de Jong, J.W.; Fijn, R.C.; van Horssen, P.W.; Poot, M.J. Lesser black-backed gulls Larus fuscus thriving on a non-marine diet. Bird Study 2016, 63, 241–249. [Google Scholar] [CrossRef]
- Figuerola, J.; Green, A.J.; Santamaría, L. Comparative dispersal effectiveness of wigeongrass seeds by waterfowl wintering in South-West Spain: Quantitative and qualitative aspects. J. Ecol. Environ. 2002, 90, 989–1001. [Google Scholar] [CrossRef]
- Van Leeuwen, C.H.; Villar, N.; Mendoza Sagrera, I.; Green, A.J.; Bakker, E.S.; Soons, M.B.; Galetti, M.; Jansen, P.A.; Nolet, B.A.; Santamaría, L. A seed dispersal effectiveness framework across the mutualism–antagonism continuum. Oikos 2022, 2022, e09254. [Google Scholar] [CrossRef]
- Mandak, B.; Zakravsky, P.; Dostal, P.; Plackova, I. Population genetic structure of the noxious weed Amaranthus retroflexus in Central Europe. Flora 2011, 206, 697–703. [Google Scholar] [CrossRef]
- Singh, S. Evaluation of some graminicides against Polypogon monspeliensis (L.) Desf. Ind. J. Weed Sci. 2009, 41, 96–98. [Google Scholar]
- Yadav, D.; Roshan, L.; Punia, S.; Ashok, Y.; Malik, R.; Hasija, R. Efficacy of clodinafop and fenoxaprop in combination with isoproturon against Polypogon monspeliensis and Phalaris minor in wheat. Environ. Ecol. 2011, 29, 1256–1259. [Google Scholar]
- Singh, A.; Bhullar, M.S.; Yadav, R.; Chowdhury, T. Weed management in zero-till wheat. Ind. J. Weed Sci. 2015, 47, 233–239. [Google Scholar]
- López-Granados, F.; Peña-Barragán, J.M.; Jurado-Expósito, M.; Francisco-Fernández, M.; Cao, R.; Alonso-Betanzos, A.; Fontenla-Romero, O. Multispectral classification of grass weeds and wheat (Triticum durum) using linear and nonparametric functional discriminant analysis and neural networks. Weed Res. 2008, 48, 28–37. [Google Scholar] [CrossRef]
- Callaway, J.; Zedler, J.B. Interactions between a salt marsh native perennial (Salicornia virginica) and an exotic annual (Polypogon monspeliensis) under varied salinity and hydroperiod. Wetl. Ecol. Manag. 1997, 5, 179–194. [Google Scholar] [CrossRef]
- Callaway, J.C.; Zedler, J.B. Restoration of urban salt marshes: Lessons from southern California. Urban Ecosyst. 2004, 7, 107–124. [Google Scholar] [CrossRef]
- Holm, L.; Doll, J.; Holm, E.; Pancho, J.V.; Herberger, J.P. World Weeds: Natural Histories and Distribution; John Wiley & Sons: Hoboken, NJ, USA, 1997. [Google Scholar]
- Tranel, P.J.; Trucco, F. 21st-century weed science: A call for Amaranthus genomics. In Weedy and Invasive Plant Genomics; Stewart, C.N., Jr., Ed.; Wiley-Blackwell: Ames, IA, USA, 2009; Volume 53, p. 81. [Google Scholar]
- Yarnia, M.; Benam, M.K.; Tabrizi, E.F.M. Allelopathic effects of sorghum extracts on Amaranthus retroflexus seed germination and growth. J. Food Agric. Environ. 2009, 7, 770–774. [Google Scholar] [CrossRef]
- Marinov-Serafimov, P. Determination of allelopathic effect of some invasive weed species on germination and initial development of grain legume crops. Pestic. Phytomed. 2015, 25, 251–259. [Google Scholar] [CrossRef]
- McKay, A.; Ophel, K.; Reardon, T.; Gooden, J. Livestock deaths associated with Clavibacter toxicus/Anguina sp. infection in seedheads of Agrostis avenacea and Polypogon monspeliensis. Plant Dis. 1993, 77, 635–641. [Google Scholar] [CrossRef]
- Kessell, A.; Boulton, J.; Krebs, G.; Quinn, J. Acute renal failure due to Amaranthus species ingestion by lambs. Aust. Vet. J. 2015, 93, 208–213. [Google Scholar] [CrossRef]
- Weston, P.A.; Gurusinghe, S.; Birckhead, E.; Skoneczny, D.; Quinn, J.C.; Weston, L.A. Chemometric analysis of Amaranthus retroflexus in relation to livestock toxicity in Southern Australia. Phytochemistry 2019, 161, 1–10. [Google Scholar] [CrossRef]
- Wylie, S.J.; Li, H.; Jones, M.G. Yellow tailflower mild mottle virus: A new tobamovirus described from Anthocercis littorea (Solanaceae) in Western Australia. Arch. Virol. 2014, 159, 791–795. [Google Scholar] [CrossRef]
- Bryson, C.T.; Carter, R. The significance of Cyperaceae as weeds. In Sedges: Uses, Diversity, Systematics of the Cyperaceae; Missouri Botanical Garden Press: St. Louis, MO, USA, 2008; Volume 108, p. 15. [Google Scholar]
- Gómez de Barreda, D.; Pardo, G.; Osca, J.M.; Catala-Forner, M.; Consola, S.; Garnica, I.; López-Martínez, N.; Palmerín, J.A.; Osuna, M.D. An overview of rice cultivation in Spain and the management of herbicide-resistant weeds. Agron. J. 2021, 11, 1095. [Google Scholar] [CrossRef]
- Cuba-Díaz, M.; Fuentes, E.; Rondanelli-Reyes, M. Experimental culture of non-indigenous Juncus bufonius from King George Island, South Shetland Island, Antarctica. Adv. Polar Sci. 2015, 26, 24–29. [Google Scholar] [CrossRef]
- Chauhan, B.S.; Matloob, A.; Mahajan, G.; Aslam, F.; Florentine, S.K.; Jha, P. Emerging challenges and opportunities for education and research in weed science. Front. Plant Sci. 2017, 8, 1537. [Google Scholar] [CrossRef] [PubMed]
- Hanson, C. Genetic, biological, chemical, and management approaches to crop protection. In Introduction to Crop Protection; Ennis, W.B., Ed.; American Society of Agronomy: Madison, WI, USA, 1979; pp. 89–151. [Google Scholar]
- Bartel, R.D.; Sheppard, J.L.; Lovas-Kiss, A.; Green, A.J. Endozoochory by mallard in New Zealand: What seeds are dispersed and how far? PeerJ 2018, 6, 19. [Google Scholar] [CrossRef]
- Zedler, J.B.; Kercher, S. Causes and consequences of invasive plants in wetlands: Opportunities, opportunists, and outcomes. Crit. Rev. Plant Sci. 2004, 23, 431–452. [Google Scholar] [CrossRef]
- Chauhan, B.S.; Johnson, D.E. Ecological studies on Cyperus difformis, Cyperus iria and Fimbristylis miliacea: Three troublesome annual sedge weeds of rice. Ann. Appl. Biol. 2009, 155, 103–112. [Google Scholar] [CrossRef]
- Devlaeminck, R.; Bossuyt, B.; Hermy, M. Seed dispersal from a forest into adjacent cropland. Agric. Ecosyst. Environ. 2005, 107, 57–64. [Google Scholar] [CrossRef]
- Julve, P. Baseflor. Index Botanique, Écologique et Chorologique de la flore de France; Institut Catholique de Lille: Lille, France, 1998. [Google Scholar]
- Consejería de Sostenibilidad, Medio Ambiente y Economía Azul. Programa de Emergencias, Control Epidemiológico y Seguimiento de Fauna Silvestre de Andalucía. Seguimiento de Aves Terrestres Amenazadas de Andalucía. Invernada de 2018/2019. Informe Regional; Consejería de Sostenibilidad, Medio Ambiente y Economía Azul, Junta de Andalucía: Seville, Spain, 2020. [Google Scholar]
- Snell, R.; Beckman, N.; Fricke, E. Consequences of intraspecific variation in seed dispersal for plant demography. AOB Plants 2019, 11, plz016. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Magnusson, A.; Skaug, H.; Nielsen, A.; Berg, C.; Kristensen, K.; Maechler, M.; van Bentham, K.; Bolker, B.; Brooks, M.; Brooks, M.M. Package ‘glmmtmb’. R J. 2017, 9, 378–400. [Google Scholar] [CrossRef]
- Lenth, R.V. Emmeans: Estimated Marginal Means, aka Least-Squares Means, R Package Version 1.7.1-1; 2021. Available online: https://cran.r-project.org/web/packages/emmeans/index.html (accessed on 1 October 2022).
- Wickham, H. Data analysis. In ggplot2; Springer: Berlin/Heidelberg, Germany, 2016; pp. 189–201. [Google Scholar]
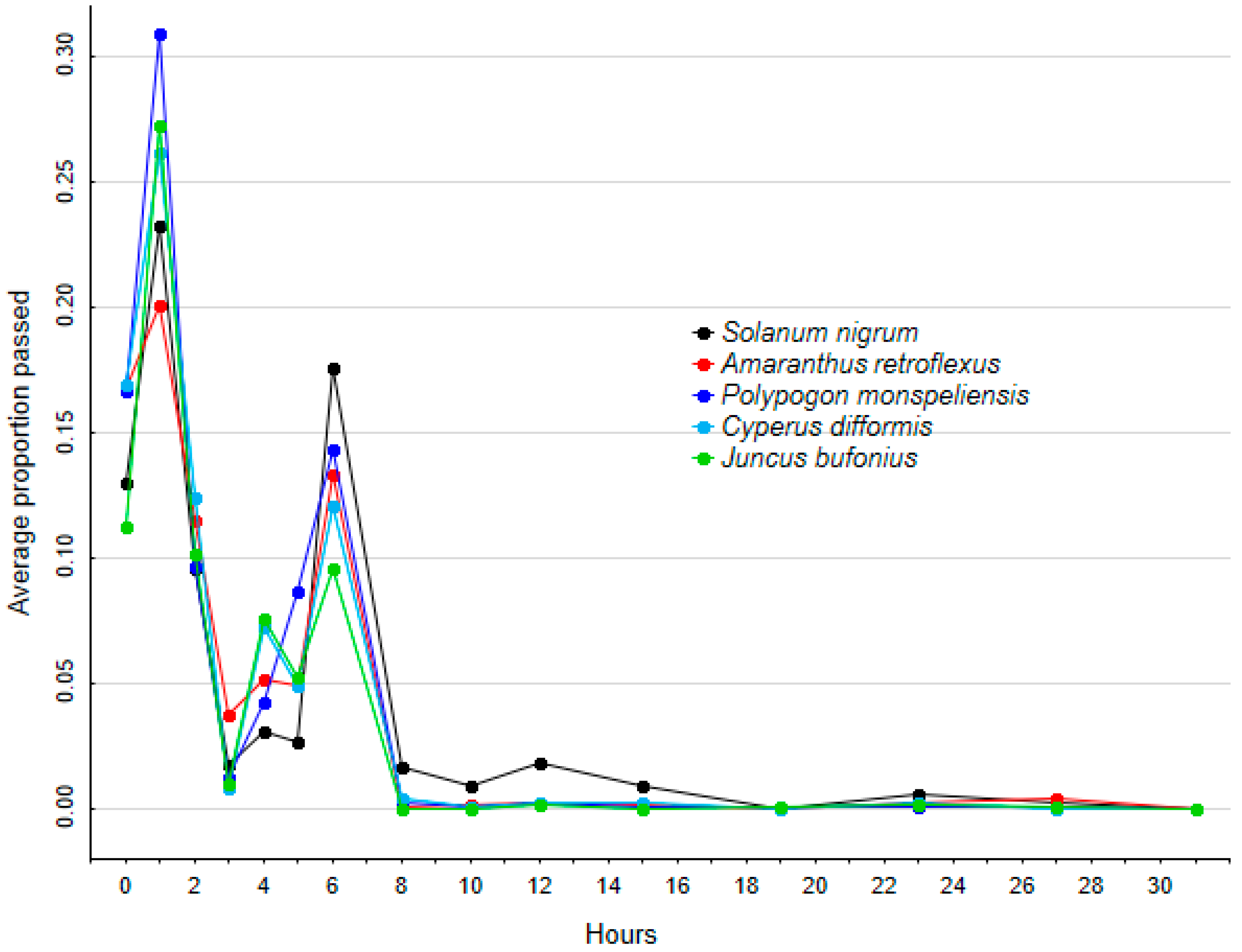
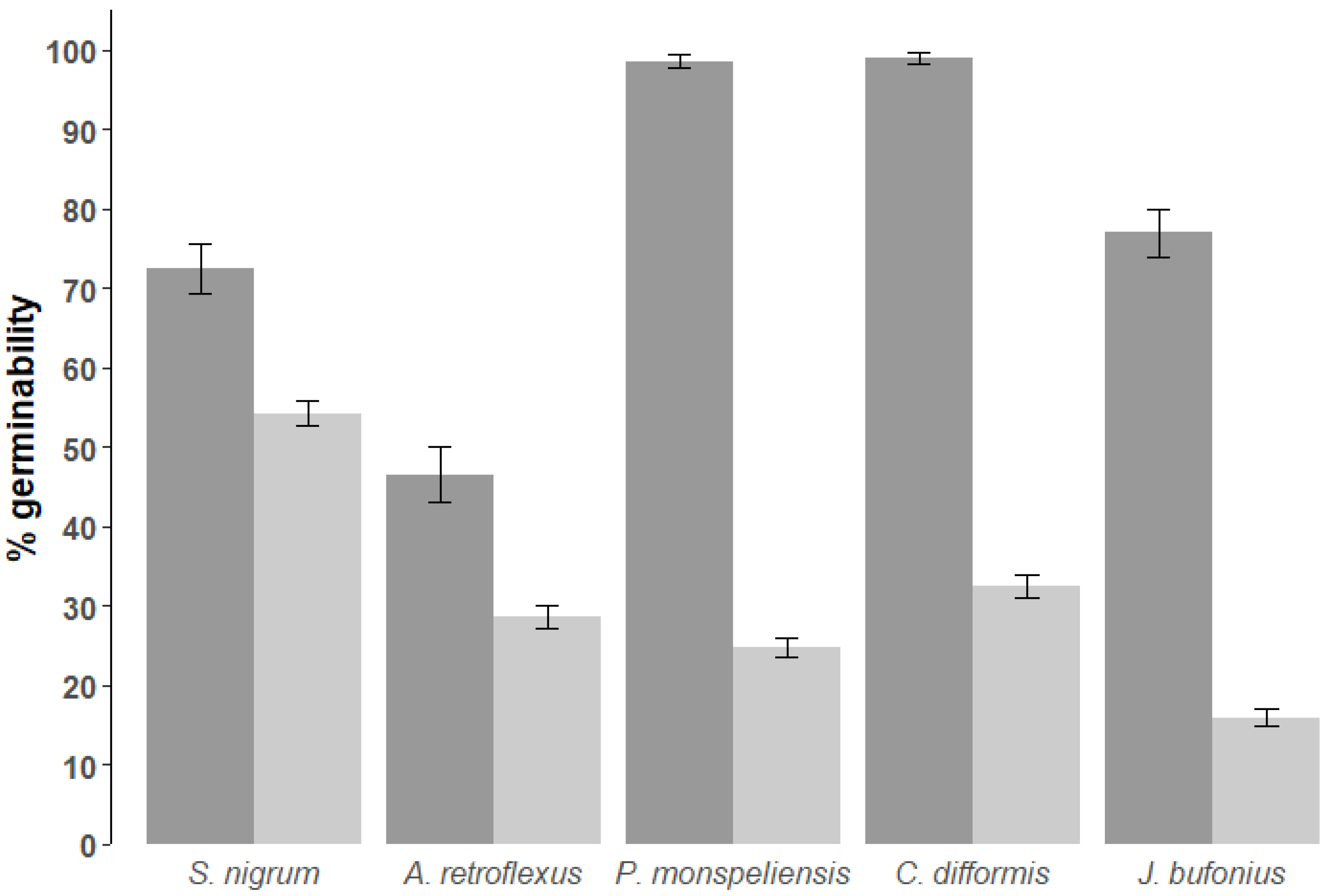
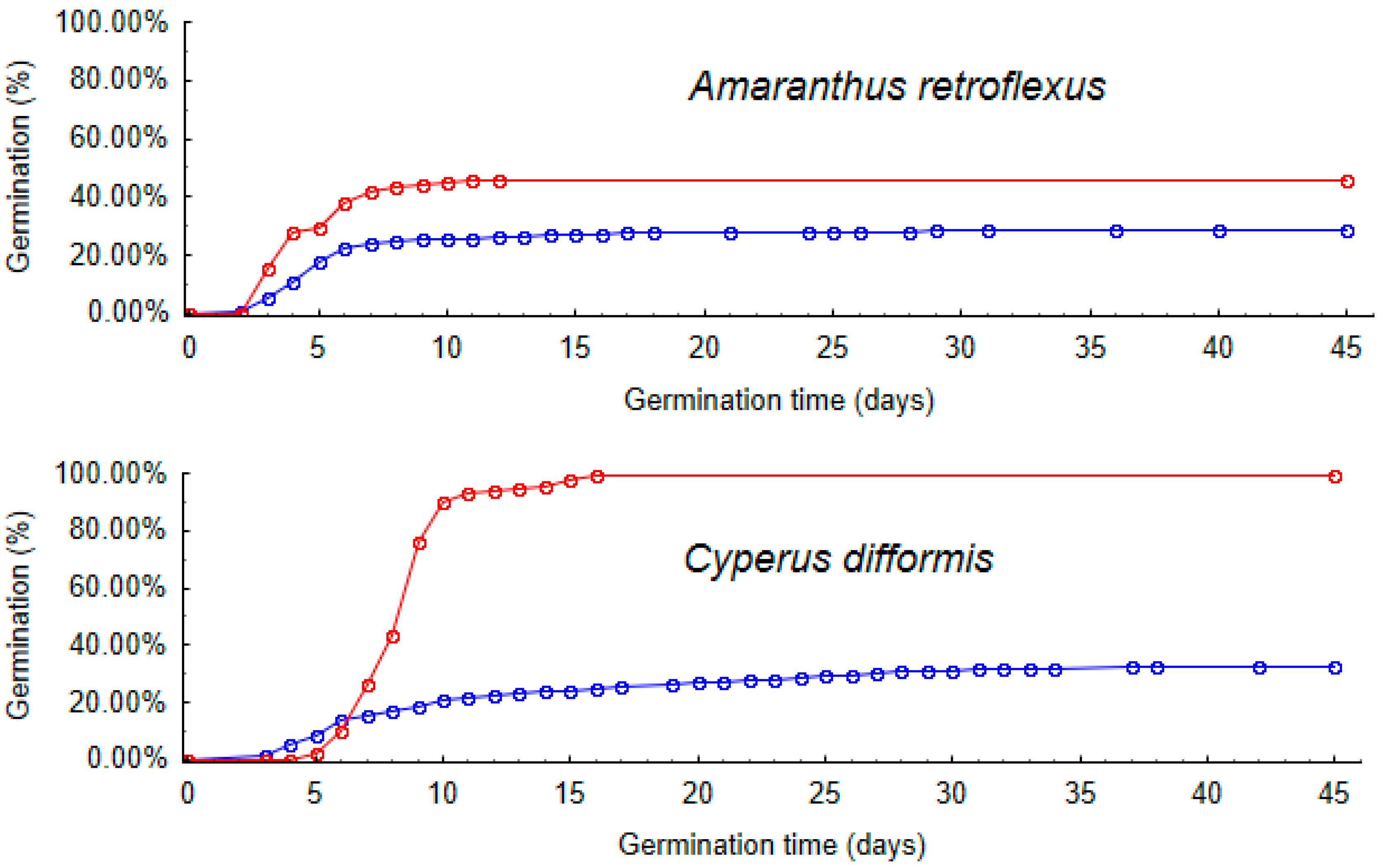


| (a) Survival Rate | (b) Retention Time | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Plant Species | Average | S.D. | Min. | Max. | Median | Average | S.D. | Min. | Max. | Median |
| Amaranthus retroflexus | 76.93 | 6.95 | 64.00 | 85.00 | 78.50 | 2.80 | 3.22 | 0.25 | 27.5 | 2.00 |
| Cyperus difformis | 81.86 | 9.01 | 69.00 | 92.00 | 86.00 | 2.53 | 2.66 | 0.25 | 23.5 | 1.00 |
| Juncus bufonius | 72.43 | 9.48 | 57.50 | 83.00 | 72.00 | 2.47 | 2.45 | 0.25 | 27.5 | 1.00 |
| Polypogon monspeliensis | 87.71 | 6.38 | 80.50 | 96.00 | 85.00 | 2.58 | 2.62 | 0.25 | 27.5 | 1.00 |
| Solanum nigrum | 76.71 | 21.97 | 31.50 | 100.00 | 80.00 | 3.55 | 4.00 | 0.25 | 27.5 | 2.00 |
| Contrast | Estimate | SE | t. Ratio | p Value |
|---|---|---|---|---|
| A. retroflexus—C. difformis | −0.18 | 0.06 | −2.86 | 0.036 |
| A. retroflexus—J. bufonius | −0.05 | 0.06 | −0.73 | 0.949 |
| A. retroflexus—P. monspeliensis | −0.23 | 0.06 | −3.68 | 0.002 |
| A. retroflexus—S. nigrum | 0.20 | 0.06 | 3.18 | 0.014 |
| C. difformis—J. bufonius | 0.13 | 0.06 | 2.09 | 0.228 |
| C. difformis—P. monspeliensis | −0.05 | 0.06 | −0.79 | 0.932 |
| C. difformis—S. nigrum | 0.38 | 0.06 | 6.00 | <0.0001 |
| J. bufonius—P. monspeliensis | −0.18 | 0.06 | −2.88 | 0.034 |
| J. bufonius—S. nigrum | 0.25 | 0.07 | 3.81 | 0.002 |
| P. monspeliensis—S. nigrum | 0.43 | 0.06 | 6.86 | <0.0001 |
| Contrast | Estimate | SE | t. Ratio | p Value |
|---|---|---|---|---|
| A. retroflexus— C. difformis | 0.03 | 0.01 | 2.47 | 0.097 |
| A. retroflexus— J. bufonius | 0.04 | 0.01 | 2.62 | 0.067 |
| A. retroflexus—P. monspeliensis | 0.03 | 0.01 | 2.52 | 0.087 |
| A. retroflexus— S. nigrum | −0.08 | 0.01 | −5.92 | <0.001 |
| C. difformis—J. bufonius | <0.01 | 0.01 | 0.24 | 0.999 |
| C. difformis—P. monspeliensis | <0.01 | 0.01 | −0.01 | 0.999 |
| C. difformis—S. nigrum | −0.11 | 0.01 | −8.40 | <0.001 |
| J. bufonius—P. monspeliensis | <0.01 | 0.01 | −0.25 | 0.999 |
| J. bufonius—S. nigrum | −0.12 | 0.01 | −8.30 | <0.001 |
| P. monspeliensis—S. nigrum | −0.11 | 0.01 | −8.57 | <0.001 |
| Experimental Seeds | Control Seeds | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | Average | SD | CI | N | Average | SD | CI | |||
| Amaranthus retroflexus | 1077 | 28.69 | 45.26 | 25.99 | 31.40 | 200 | 46.50 | 50.00 | 39.53 | 53.47 |
| Cyperus difformis | 1146 | 32.55 | 46.88 | 29.83 | 35.26 | 200 | 99.00 | 9.97 | 97.61 | 100.39 |
| Juncus bufonius | 1014 | 15.98 | 36.66 | 13.72 | 18.24 | 200 | 77.00 | 42.19 | 71.12 | 82.88 |
| Polypogon monspeliensis | 1210 | 24.79 | 43.20 | 22.36 | 27.23 | 200 | 98.50 | 12.18 | 96.80 | 100.20 |
| Solanum nigrum | 1070 | 54.21 | 49.85 | 51.22 | 57.20 | 200 | 72.50 | 44.76 | 66.26 | 78.74 |
| Passage Seeds | Control Seeds | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | A | M | Max | Min | SD | CI | N | A | M | Max | Min | SD | CI | |||
| Amaranthus retroflexus | 309 | 6.51 | 5 | 2 | 40 | 5.55 | 5.15 | 6.03 | 92 | 4.82 | 4 | 3 | 12 | 2.02 | 1.77 | 2.36 |
| Cyperus difformis | 373 | 11.66 | 8 | 3 | 42 | 8.73 | 8.15 | 9.41 | 198 | 8.70 | 9 | 5 | 16 | 1.98 | 1.80 | 2.20 |
| Juncus bufonius | 162 | 14.83 | 7 | 4 | 43 | 13.00 | 11.73 | 14.60 | 154 | 19.55 | 16 | 8 | 45 | 10.21 | 9.18 | 11.50 |
| Polypogon monspeliensis | 300 | 5.76 | 4 | 1 | 41 | 6.76 | 6.26 | 7.35 | 197 | 3.82 | 3 | 2 | 19 | 2.24 | 2.04 | 2.49 |
| Solanum nigrum | 579 | 9.79 | 8 | 3 | 43 | 5.26 | 4.98 | 5.58 | 145 | 14.84 | 15 | 6 | 37 | 6.97 | 6.25 | 7.88 |
| Control | Passage | |||||||
|---|---|---|---|---|---|---|---|---|
| Plant Species | Average | SE | Average | SE | Estimate | SE | Chisq | p |
| Amaranthus retroflexus | 4.82 | 0.21 | 6.51 | 0.32 | 0.30 | 0.05 | 35.21 | <0.001 |
| Cyperus difformis | 8.70 | 0.14 | 11.66 | 0.45 | 0.29 | 0.03 | 110.48 | <0.001 |
| Juncus bufonius | 19.55 | 0.82 | 14.83 | 1.02 | −0.28 | 0.03 | 102.42 | <.001 |
| Polypogon monspeliensis | 3.83 | 0.16 | 5.76 | 0.39 | 0.41 | 0.04 | 92.79 | <0.001 |
| Solanum nigrum | 14.84 | 0.58 | 9.79 | 0.22 | −0.42 | 0.03 | 253.08 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peralta-Sánchez, J.M.; Ansotegui, A.; Hortas, F.; Redón, S.; Martín-Vélez, V.; Green, A.J.; Navarro-Ramos, M.J.; Lovas-Kiss, A.; Sánchez, M.I. Seed Size, Not Dispersal Syndrome, Determines Potential for Spread of Ricefield Weeds by Gulls. Plants 2023, 12, 1470. https://doi.org/10.3390/plants12071470
Peralta-Sánchez JM, Ansotegui A, Hortas F, Redón S, Martín-Vélez V, Green AJ, Navarro-Ramos MJ, Lovas-Kiss A, Sánchez MI. Seed Size, Not Dispersal Syndrome, Determines Potential for Spread of Ricefield Weeds by Gulls. Plants. 2023; 12(7):1470. https://doi.org/10.3390/plants12071470
Chicago/Turabian StylePeralta-Sánchez, Juan Manuel, Albán Ansotegui, Francisco Hortas, Stella Redón, Víctor Martín-Vélez, Andy J. Green, María J. Navarro-Ramos, Adam Lovas-Kiss, and Marta I. Sánchez. 2023. "Seed Size, Not Dispersal Syndrome, Determines Potential for Spread of Ricefield Weeds by Gulls" Plants 12, no. 7: 1470. https://doi.org/10.3390/plants12071470
APA StylePeralta-Sánchez, J. M., Ansotegui, A., Hortas, F., Redón, S., Martín-Vélez, V., Green, A. J., Navarro-Ramos, M. J., Lovas-Kiss, A., & Sánchez, M. I. (2023). Seed Size, Not Dispersal Syndrome, Determines Potential for Spread of Ricefield Weeds by Gulls. Plants, 12(7), 1470. https://doi.org/10.3390/plants12071470








