New Low Morphine Opium Poppy Genotype Obtained by TILLING Approach
Abstract
1. Introduction
2. Results
2.1. Bioinformatical Selection of Marker Genes
2.2. Mutational Analysis of Selected Gene Markers
2.3. HPLC Analysis of Main Alkaloid Content in Selected M3 Plants
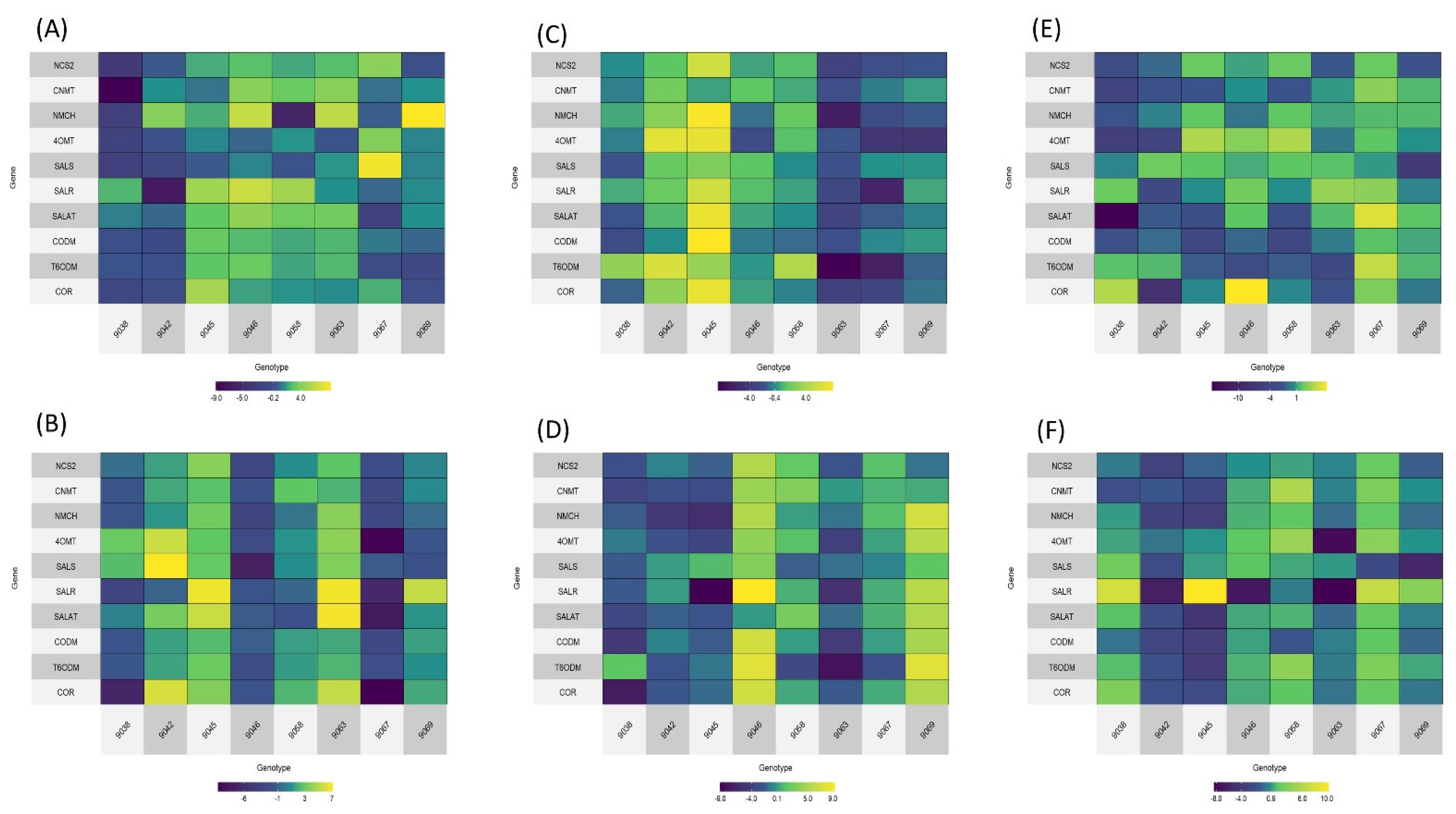
2.4. Gene Expression Analysis of Selected Genotypes
3. Discussion
4. Materials and Methods
4.1. Chemical Mutagenesis and Seed Selection
4.2. Mutation Screening
4.3. Field Cultivation
4.4. RNA Preparation for Gene Expression Measurements
4.5. Gene Expression Profiling
4.6. HPLC Assessment of Main Alkaloids
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Verma, N.; Jena, S.N.; Shukla, S.; Yadav, K. Genetic Diversity, Population Structure and Marker Trait Associations for Alkaloid Content and Licit Opium Yield in India-Wide Collection of Poppy (Papaver somniferum L.). Plant Gene 2016, 7, 26–41. [Google Scholar] [CrossRef]
- Labanca, F.; Ovesnà, J.; Milella, L. Papaver somniferum L. Taxonomy, Uses and New Insight in Poppy Alkaloid Pathways. Phytochem. Rev. 2018, 17, 853–871. [Google Scholar] [CrossRef]
- Lal, R.K.; Chanotiya, C.S.; Gupta, P. Induced Mutation Breeding for Qualitative and Quantitative Traits and Varietal Development in Medicinal and Aromatic Crops at CSIR-CIMAP, Lucknow (India): Past and Recent Accomplishment. Int. J. Radiat. Biol. 2020, 96, 1513–1527. [Google Scholar] [CrossRef] [PubMed]
- Gümüşçü, A.; Arslan, N.; Sarıhan, E.O. Evaluation of Selected Poppy (Papaver somniferum L.) Lines by Their Morphine and Other Alkaloids Contents. Eur. Food Res. Technol. 2008, 226, 1213–1220. [Google Scholar] [CrossRef]
- Beaudoin, G.A.W.; Facchini, P.J. Benzylisoquinoline Alkaloid Biosynthesis in Opium Poppy. Planta 2014, 240, 19–32. [Google Scholar] [CrossRef]
- Singh, A.; Menéndez-Perdomo, I.M.; Facchini, P.J. Benzylisoquinoline Alkaloid Biosynthesis in Opium Poppy: An Update. Phytochem. Rev. 2019, 18, 1457–1482. [Google Scholar] [CrossRef]
- Svoboda, P.; Vašek, J.; Vejl, P.; Ovesná, J. Genetic Features of Czech Blue Poppy (Papaver somniferum L.) Revealed by DNA Polymorphism. Czech J. Food Sci. 2020, 38, 198–202. [Google Scholar] [CrossRef]
- Dubey, M.K.; Shasany, A.K.; Dhawan, O.P.; Shukla, A.K.; Khanuja, S.P.S. Lipid Peroxidation and Antioxidant Activities Involved in Resistance Response against Downy Mildew in Opium Poppy. J. Phytopathol. 2010, 158, 88–92. [Google Scholar] [CrossRef]
- Kundratova, K.; Bartas, M.; Pecinka, P.; Hejna, O.; Rychla, A.; Curn, V.; Cerven, J. Transcriptomic and Proteomic Analysis of Drought Stress Response in Opium Poppy Plants during the First Week of Germination. Plants 2021, 10, 1878. [Google Scholar] [CrossRef] [PubMed]
- Mahdavi-Damghani, A.; Kamkar, B.; Al-Ahmadi, M.J.; Testi, L.; Munoz-Ledesma, F.J.; Villalobos, F.J. Water Stress Effects on Growth, Development and Yield of Opium Poppy (Papaver somniferum L.). Agric. Water Manag. 2010, 97, 1582–1590. [Google Scholar] [CrossRef]
- Pinke, G.; Toth, K.; Kovacs, A.J.; Milics, G.; Varga, Z.; Blazsek, K.; Gal, K.E.; Botta-Dukat, Z. Use of Mesotrione and Tembotrione Herbicides for Post-Emergence Weed Control in Alkaloid Poppy (Papaver somniferum). Int. J. Pest Manag. 2014, 60, 187–195. [Google Scholar] [CrossRef]
- Hu, Y.; Zhao, R.; Xu, P.; Jiao, Y. The Genome of Opium Poppy Reveals Evolutionary History of Morphinan Pathway. Genom. Proteom. Bioinform. 2018, 16, 460–462. [Google Scholar] [CrossRef]
- Li, Y.; Winzer, T.; He, Z.; Graham, I.A. Over 100 Million Years of Enzyme Evolution Underpinning the Production of Morphine in the Papaveraceae Family of Flowering Plants. Plant Commun. 2020, 1, 100029. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Winzer, T.; Yang, X.; Li, Y.; Ning, Z.; He, Z.; Teodor, R.; Lu, Y.; Bowser, T.A.; Graham, I.A.; et al. The Opium Poppy Genome and Morphinan Production. Science 2018, 362, 343–347. [Google Scholar] [CrossRef]
- Mishra, B.K.; Pathak, S.; Sharma, A.; Trivedi, P.K.; Shukla, S. Modulated Gene Expression in Newly Synthesized Auto-Tetraploid of Papaver somniferum L. S. Afr. J. Bot. 2010, 76, 447–452. [Google Scholar] [CrossRef]
- Li, Q.; Ramasamy, S.; Singh, P.; Hagel, J.M.; Dunemann, S.M.; Chen, X.; Chen, R.; Yu, L.; Tucker, J.E.; Facchini, P.J.; et al. Gene Clustering and Copy Number Variation in Alkaloid Metabolic Pathways of Opium Poppy. Nat. Commun. 2020, 11, 1190. [Google Scholar] [CrossRef]
- Gao, C. Genome Engineering for Crop Improvement and Future Agriculture. Cell 2021, 184, 1621–1635. [Google Scholar] [CrossRef]
- Uslu, T. Advantages, Risks and Legal Perspectives of GMOs in 2020s. Plant Biotechnol. Rep. 2021, 15, 741–751. [Google Scholar] [CrossRef]
- Chen, L.; Hao, L.; Parry, M.A.J.; Phillips, A.L.; Hu, Y.-G. Progress in TILLING as a Tool for Functional Genomics and Improvement of Crops. J. Integr. Plant Biol. 2014, 56, 425–443. [Google Scholar] [CrossRef] [PubMed]
- Irshad, A.; Guo, H.; Zhang, S.; Liu, L. TILLING in Cereal Crops for Allele Expansion and Mutation Detection by Using Modern Sequencing Technologies. Agronomy 2020, 10, 405. [Google Scholar] [CrossRef]
- Rashid, M.; He, G.; Guanxiao, Y.; Khurram, Z. Relevance of Tilling in Plant Genomics. Aust. J. Crop Sci. 2011, 5, 411–420. [Google Scholar]
- Tadele, Z. Mutagenesis and TILLING to Dissect Gene Function in Plants. Curr. Genom. 2016, 17, 499–508. [Google Scholar] [CrossRef]
- Taheri, S.; Abdullah, T.L.; Jain, S.M.; Sahebi, M.; Azizi, P. TILLING, High-Resolution Melting (HRM), and next-Generation Sequencing (NGS) Techniques in Plant Mutation Breeding. Mol. Breed. 2017, 37, 40. [Google Scholar] [CrossRef]
- McCallum, C.; Comai, L.; Greene, E.; Henikoff, S. Targeting Induced Local Lesions in Genomes (TILLING) for Plant Functional Genomics. Plant Physiol. 2000, 123, 439–442. [Google Scholar] [CrossRef]
- Colbert, T.; Till, B.; Tompa, R.; Reynolds, S.; Steine, M.; Yeung, A.; McCallum, C.; Comai, L.; Henikoff, S. High-Throughput Screening for Induced Point Mutations. Plant Physiol. 2001, 126, 480–484. [Google Scholar] [CrossRef]
- Tsai, H.; Howell, T.; Nitcher, R.; Missirian, V.; Watson, B.; Ngo, K.J.; Lieberman, M.; Fass, J.; Uauy, C.; Tran, R.K.; et al. Discovery of Rare Mutations in Populations: TILLING by Sequencing. Plant Physiol. 2011, 156, 1257–1268. [Google Scholar] [CrossRef]
- Slade, A.; Fuerstenberg, S.; Loeffler, D.; Steine, M.; Facciotti, D. A Reverse Genetic, Nontransgenic Approach to Wheat Crop Improvement by TILLING. Nat. Biotechnol. 2005, 23, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Lakhssassi, N.; Zhou, Z.; Liu, S.; Colantonio, V.; AbuGhazaleh, A.; Meksem, K. Characterization of the FAD2 Gene Family in Soybean Reveals the Limitations of Gel-Based TILLING in Genes with High Copy Number. Front. Plant Sci. 2017, 8, 324. [Google Scholar] [CrossRef]
- Liscombe, D.K.; O’Connor, S.E. A Virus-Induced Gene Silencing Approach to Understanding Alkaloid Metabolism in Catharanthus Roseus. Phytochemistry 2011, 72, 1969–1977. [Google Scholar] [CrossRef]
- Bird, D.A.; Franceschi, V.R.; Facchini, P.J. A Tale of Three Cell Types: Alkaloid Biosynthesis Is Localized to Sieve Elements in Opium Poppy. Plant Cell 2003, 15, 2626–2635. [Google Scholar] [CrossRef]
- Ozber, N.; Facchini, P.J. Phloem-Specific Localization of Benzylisoquinoline Alkaloid Metabolism in Opium Poppy. J. Plant Physiol. 2022, 271, 153641. [Google Scholar] [CrossRef]
- Watkins, J.L.; Facchini, P.J. Compartmentalization at the Interface of Primary and Alkaloid Metabolism. Curr. Opin. Plant Biol. 2022, 66, 102186. [Google Scholar] [CrossRef]
- Tejklová, E. Curly Stem—An Induced Mutation in Flax (Linum usitatissimum L.). Czech J. Genet. Plant Breed. 2012, 38, 125–128. [Google Scholar] [CrossRef]
- Ye, J.; Coulouris, G.; Zaretskaya, I.; Cutcutache, I.; Rozen, S.; Madden, T.L. Primer-BLAST: A Tool to Design Target-Specific Primers for Polymerase Chain Reaction. BMC Bioinform. 2012, 13, 134. [Google Scholar] [CrossRef]
- Seemann, T. Snippy: Fast Bacterial Variant Calling from NGS Reads; GitHub: San Francisco, CA, USA, 2015. [Google Scholar]
- The Galaxy Community. The Galaxy Platform for Accessible, Reproducible and Collaborative Biomedical Analyses: 2022 Update. Nucleic Acids Res. 2022, 50, W345–W351. [Google Scholar] [CrossRef] [PubMed]
- Aranda, P.S.; LaJoie, D.M.; Jorcyk, C.L. Bleach Gel: A Simple Agarose Gel for Analyzing RNA Quality. Electrophoresis 2012, 33, 366–369. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
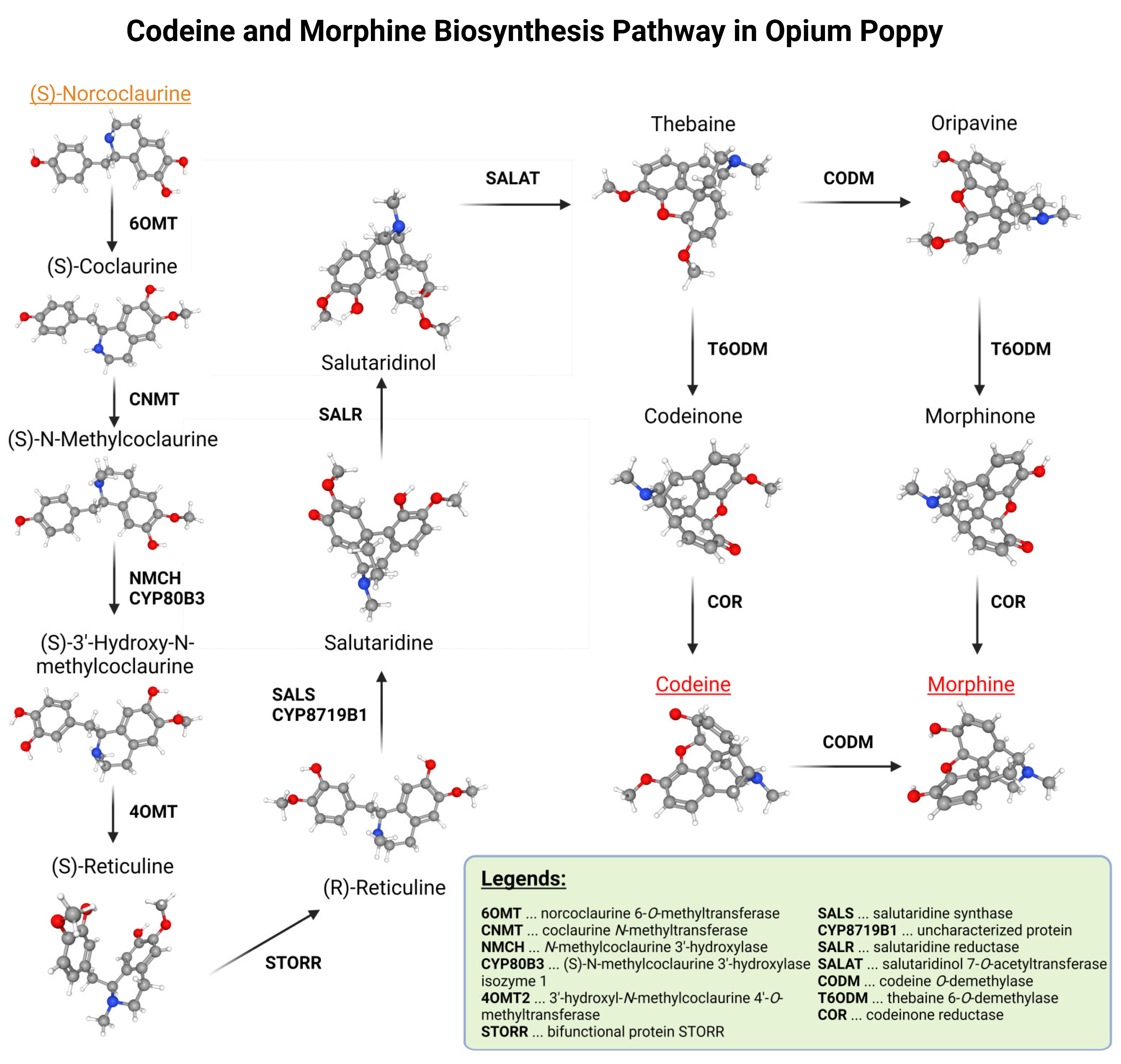
| Gene ID | Gene Name | Shortcut | Gene Length | CDS Length | Chromosome | Location |
|---|---|---|---|---|---|---|
| 113321914 | salutaridine synthase | SalS | 1959 | 1518 | 11 | NC_039368.1 (128249007..128250966) |
| 113339274 | salutaridine synthase-like | SalS | 1966 | 1521 | Unplaced | NW_020631041.1 (8601134..8603100) |
| 113322262 | salutaridine reductase | SalR | 1694 | 939 | 11 | NC_039368.1 (128330274..128331968) |
| 113340174 | salutaridine reductase | SalR | 1762 | 942 | Unplaced | NW_020631041.1 (8574428..8576190) |
| 113321357 | salutaridinol 7-O-acetyltransferase | SalAT | 2305 | 1425 | 11 | NC_039368.1 (128319439..128321744) |
| 113347789 | thebaine 6-O-demethylase | T6ODM | 2464 | 1095 | 2 | NC_039359.1 (59955747..59958211) |
| 113347787 | thebaine 6-O-demethylase-like | T6ODM | 2532 | 1095 | 2 | NC_039359.1 (59933446..59935978) |
| 113347785 | thebaine 6-O-demethylase-like | T6ODM | 2532 | 1095 | 2 | NC_039359.1 (59911109..59913641) |
| 113311621 | codeine O-demethylase | CODM | 1966 | 1083 | 1 | NC_039358.1 (198606455..198608421) |
| 113311630 | codeine O-demethylase | CODM | 1912 | 1083 | 1 | NC_039358.1 (198677764..198679676) |
| 113328201 | NADPH-dependent codeinone reductase 1–5 | COR | 2000 | 966 | Unplaced | NW_020619603.1 (7219550..7221550) |
| 113294469 | NADPH-dependent codeinone reductase 1–4 | COR | 1840 | 966 | 7 | NC_039364.1 (2467198..2469038) |
| 113340282 | S-norcoclaurine synthase 2 | NCS2 | 2912 | 2103 | Unplaced | NW_020631041.1 (12957304..12960215) |
| 113283898 | (RS)-norcoclaurine 6-O-methyltransferase | 6OMT | 1355 | 1041 | 5 | NC_039362.1 (183999440..184000794) |
| 113294670 | (S)-coclaurine N-methyltransferase | CNMT | 2208 | 1056 | 7 | NC_039364.1 (4136348..4138555) |
| 113314340 | (S)-N-methylcoclaurine 3′-hydroxylase | NMCH | 1799 | 1464 | 1 | NC_039358.1 (208684986..208686784) |
| 113328451 | (S)-N-methylcoclaurine 3′-hydroxylase | NMCH | 2033 | 1467 | Unplaced | NW_020619603.1 (9494942..9496974) |
| 113283892 | (S)-N-methylcoclaurine 3′-hydroxylase | NMCH | 1802 | 1464 | 5 | NC_039362.1 (183738532..183740333) |
| 113339850 | 3’-hydroxy-N-methyl-(S)-coclaurine 4’-O-methyltransferase 2 | 4OMT | 1493 | 1074 | Unplaced | NW_020631041.1 (11170945..11172437) |
| 113327792 | 3’-hydroxy-N-methyl-(S)-coclaurine 4’-O-methyltransferase 1 | 4OMT | 1589 | 1065 | Unplaced | NW_020619603.1 (9452525..9454113) |
| Mutation Found | Percent of Reads | Progeny Plants Name |
|---|---|---|
| SalAT 2011 30nt ins | 90 | 9038–9043 |
| CNMT 1205 A->G | 6 | 9044–9045 |
| CNMT 1211 T->A | 6 | 9046–9049 |
| CNMT 1256 A->T | 7 | 9050–9055 |
| CNMT 1456 A->T | 6 | 9056–9060 |
| CNMT 1695 G->A | 5 | 9061–9070 |
| Sample/Plant Name | Morphine | Codeine | Thebaine | Sample/Plant Name | Morphine | Codeine | Thebaine |
|---|---|---|---|---|---|---|---|
| Control O-P-P19 original variety | 1.453 | 0.110 | 0.296 | 9054 | 0.561 | 0.056 | 0.204 |
| 9038 | 0.434 | 0.114 | 0.206 | 9055 | 0.743 | 0.095 | 0.153 |
| 9039 | 0.551 | 0.070 | 0.210 | 9056 | 0.673 | 0.073 | 0.224 |
| 9040 | 0.507 | 0.079 | 0.380 | 9057 | 0.750 | 0.067 | 0.224 |
| 9041 | 0.636 | 0.094 | 0.304 | 9058 | 0.200 | 0.117 | 0.145 |
| 9042 | 0.100 | 0.064 | 0.525 | 9059 | 0.762 | 0.063 | 0.287 |
| 9043 | 0.635 | 0.096 | 0.235 | 9060 | 0.922 | 0.073 | 0.284 |
| 9044 | 0.972 | 0.077 | 0.300 | 9061 | 0.629 | 0.067 | 0.256 |
| 9045 | 0.369 | 0.055 | 0.212 | 9062 | 0.983 | 0.075 | 0.337 |
| 9046 | 0.452 | 0.059 | 0.222 | 9063 | 0.571 | 0.055 | 0.199 |
| 9047 | 0.868 | 0.085 | 0.278 | 9064 | 0.709 | 0.070 | 0.306 |
| 9048 | 0.527 | 0.104 | 0.304 | 9065 | 0.857 | 0.080 | 0.301 |
| 9049 | 0.471 | 0.101 | 0.210 | 9066 | 1.062 | 0.084 | 0.390 |
| 9050 | 0.485 | 0.076 | 0.324 | 9067 | 0.378 | 0.094 | 0.166 |
| 9051 | 0.763 | 0.088 | 0.336 | 9068 | 0.856 | 0.061 | 0.219 |
| 9052 | 0.513 | 0.064 | 0.224 | 9069 | 0.615 | 0.044 | 0.202 |
| 9053 | 0.892 | 0.091 | 0.338 | 9070 | 0.420 | 0.095 | 0.219 |
| SALAT-F-C6 | GCAGTCGAACATGTAGCTGACTCAGGTCACAAGAGGGGTATTCTATTCGGTGA |
| SALAT-R-C6 | TGGATCACTTGTGCAAGCATCACATCGTAGGGATCATCTACCCGAAAACAACC |
| NCS2-F-C6 | GCAGTCGAACATGTAGCTGACTCAGGTCACAAACACTCGGAACCGCAAGT |
| NCS2-R-C6 | TGGATCACTTGTGCAAGCATCACATCGTAGGAAAAATCCTGAACCTTAGAGTGGA |
| CNMT-F-C6 | GCAGTCGAACATGTAGCTGACTCAGGTCACGCTTGGGTTGATACCGGACC |
| CNMT-R-C6 | TGGATCACTTGTGCAAGCATCACATCGTAGCGATGGTAAACAACACACAAAACG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Červeň, J.; Vrbovský, V.; Horáček, J.; Bartas, M.; Endlová, L.; Pečinka, P.; Čurn, V. New Low Morphine Opium Poppy Genotype Obtained by TILLING Approach. Plants 2023, 12, 1077. https://doi.org/10.3390/plants12051077
Červeň J, Vrbovský V, Horáček J, Bartas M, Endlová L, Pečinka P, Čurn V. New Low Morphine Opium Poppy Genotype Obtained by TILLING Approach. Plants. 2023; 12(5):1077. https://doi.org/10.3390/plants12051077
Chicago/Turabian StyleČerveň, Jiří, Viktor Vrbovský, Jiří Horáček, Martin Bartas, Lenka Endlová, Petr Pečinka, and Vladislav Čurn. 2023. "New Low Morphine Opium Poppy Genotype Obtained by TILLING Approach" Plants 12, no. 5: 1077. https://doi.org/10.3390/plants12051077
APA StyleČerveň, J., Vrbovský, V., Horáček, J., Bartas, M., Endlová, L., Pečinka, P., & Čurn, V. (2023). New Low Morphine Opium Poppy Genotype Obtained by TILLING Approach. Plants, 12(5), 1077. https://doi.org/10.3390/plants12051077











