Moroccan Citrus clementina Peels: Optimization of Pectin Extraction and Determination of Chemical and Functional Properties
Abstract
:1. Introduction
2. Results and Discussion
2.1. Experimental Design
2.2. Estimated Model
2.3. Statistical Analysis and Validation of the Model
2.4. Interpretation of the Response Surface Model
2.5. Determination of Optimum Conditions
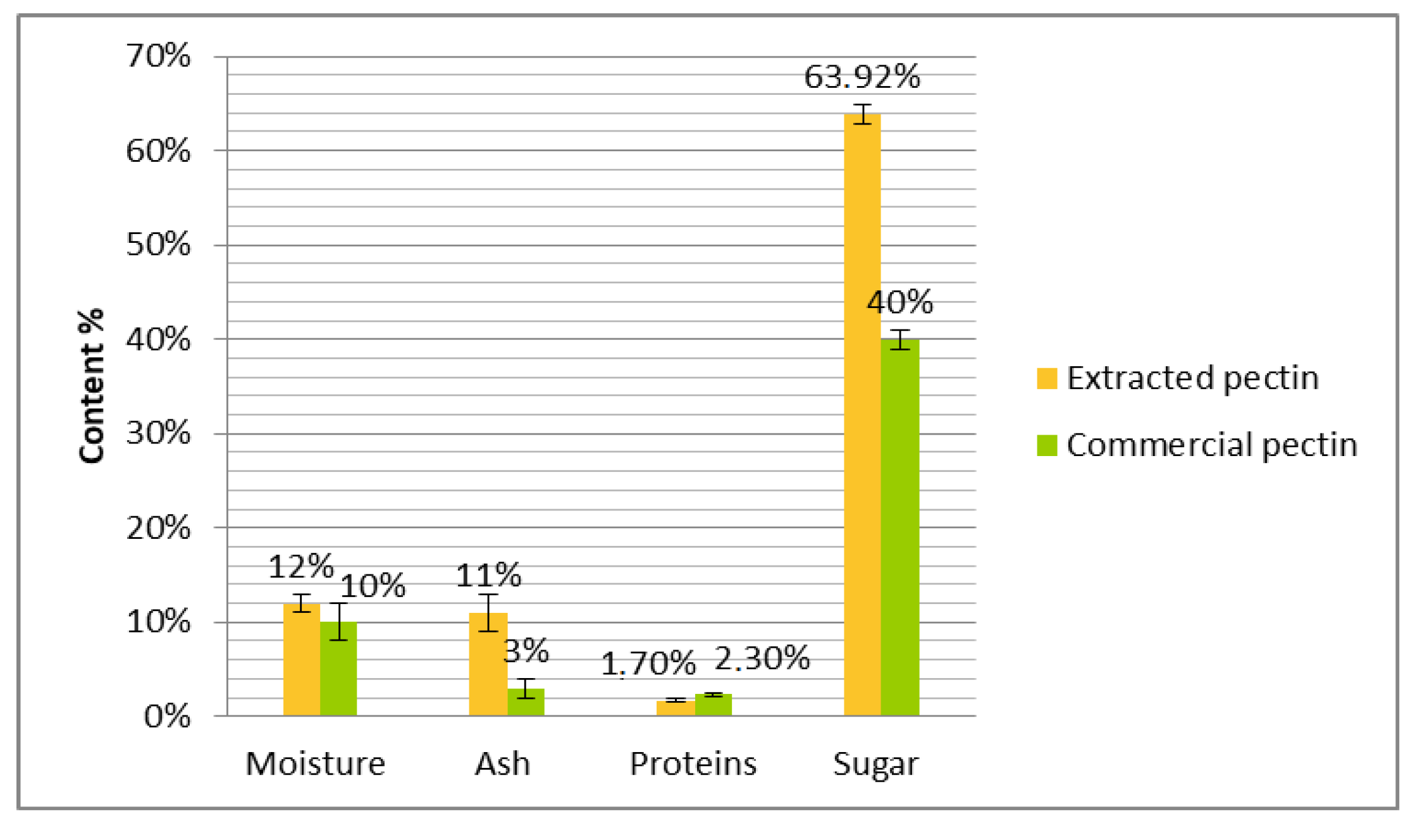
2.6. Chemical Characteristics
2.7. Functional Properties
3. Materials and Methods
3.1. Plant Material
3.2. Pectin Extraction
3.3. Experimental Methodology
3.4. Validation of the Model
3.5. Chemical Characterization and Pectin Yield
3.5.1. Moisture Content
3.5.2. Ash Content
3.5.3. Protein Content
3.5.4. Carbohydrate Content
3.5.5. Pectin Yield
3.6. Functional Properties
3.6.1. Determination of Gelling Power
3.6.2. Emulsifying Activity Analysis
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- MAMPDREF. Ministry of Agriculture, Maritime Fishing, Rural Development and Water and Forests. Morocco. 2020. Available online: https://www.agriculture.gov.ma/fr/filiere/agrumicole (accessed on 22 September 2022).
- Masmoudi, M.A.; Souhail, B.; Moncef, C.; Christelle, R.; Michel, P.; Christophe, B.; Hamadi, A. Optimization of pectin extraction from lemon by-product with acidified date juice using response surface methodology. Carbohydr. Polym. 2008, 74, 185–192. [Google Scholar] [CrossRef]
- Bicu, I.; Mustata, F. Cellulose extraction from orange peel using sulfite digestion reagents. Bioresour. Technol. 2011, 102, 10013–10019. [Google Scholar] [CrossRef] [PubMed]
- Fishman, M.L.; Jen, J.J. Chemistry and Function of Pectins; American Chemical Society: Washington, DC, USA, 1986. [Google Scholar]
- Prakash Maran, J.; Sivakumar, V.; Thirugnanasambandham, K.; Sridhar, R. Microwave assisted extraction of pectin from waste Citrullus lanatus fruit rinds. Carbohydr. Polym. 2014, 101, 786–791. [Google Scholar] [CrossRef] [PubMed]
- Sumathraa, M.; Govindaraja, D.; Jeyarajb, M.; Al Arfajc, A.; Munusamyc, M.A.; Selvaraj Suresh, K.S.; Rajana, M. Sustainable pectin fascinating hydroxyapatite nanocomposite scaffolds to enhance tissue regeneration. Sustain. Chem. Pharm. 2017, 5, 46–53. [Google Scholar] [CrossRef]
- Pang, S.F.; Yusoff, M.M.; Gimbun, J. Assessment of phenolic compounds stability and retention during spray drying of Orthosiphon stamineusextracts. Food Hydrocoll. 2014, 37, 159–165. [Google Scholar] [CrossRef]
- Yeoh, S.; Shi, J.; Langrish, T.A.G. Comparisons between different techniques for water-based extraction of pectin from orange peels. Desalination 2008, 218, 229–237. [Google Scholar] [CrossRef]
- Maran, J.P.; Sivakumar, V.; Thirugnanasambandham, K.; Sridhar, R. Optimization of microwave assisted extraction of pectin from orange peel. Carbohydr. Polym. 2013, 97, 703–709. [Google Scholar] [CrossRef]
- Minkov, S.; Minchev, A.; Paev, K. Modelling of the hydrolysis and extraction of apple pectin. J. Food Eng. 1996, 29, 107–113. [Google Scholar] [CrossRef]
- Raji, Z.; Khodaiyan, F.; Rezaei, K.; Kiani, H.; Hosseini, S.S. Extraction optimization and physicochemical properties of pectin from melon peel. Int. J. Biol. Macromol. 2017, 98, 709–716. [Google Scholar] [CrossRef]
- Wu, Y.; Cui, S.W.; Tang, J.; Gu, X. Optimization of extraction process of crude polysaccharides from boat-fruited sterculia seeds by response surface methodology. Food Chem. 2007, 105, 1599–1605. [Google Scholar] [CrossRef]
- Box George, E.P.; Hunter William, G.; Hunter, J.S. Statistics for Experimenters, 2nd ed.; John Wiley and Sons: New York, NY, USA, 2005; p. 633. [Google Scholar]
- Aina, V.O.; Barau, M.; Mamman, O.A.; Zakari, A.; Haruna, H.; Abba, Y. Extraction and Characterization of Pectin from Peels of Grape Fruit (Citrus paradisi), Sweet Orange (Citrus limetta) and Lemon (Citrus limon). Br. J. Pharmacol. Toxicol. 2012, 3, 259–262. [Google Scholar]
- Kanmani, P.; Dhivya, E.; Aravind, J.; Kumaresan, K. Extraction and Analysis of Pectin from Citrus Peels: Augmenting the Yield from Citrus limon Using Statistical Experimental Design. Iran. J. Energy Environ. 2014, 5, 303–312. [Google Scholar] [CrossRef]
- El-Nawawi, S.A.; Shehata, F.R. Extraction of pectin from egyptian orange peel. Factors affecting the extraction. Biol. Wastes 1987, 20, 281–290. [Google Scholar] [CrossRef]
- Moorthy, I.G.; Maran, J.P.; Surya, S.M.; Naganyashree, S.; Shivamathi, C.S. Response surface optimization of ultrasound assisted extraction of pectin from pomegranate peel. Int. J. Biol. Macromol. 2015, 72, 1323–1328. [Google Scholar] [CrossRef]
- Pasandide, B.; Khodaiyan, F.; Mousavi, Z.; Hosseini, S.S. Pectin extraction from citron peel: Optimization by Box–Behnken response surface design. Food Sci. Biotechnol. 2018, 27, 997–1005. [Google Scholar] [PubMed]
- Canteri-Schemin, M.H.; Fertonani, H.C.R.; Waszczynskyj, N.; Wosiacki, G. Extraction of pectin from apple pomace. Braz. Arch. Biol. Technol. 2005, 48, 259–266. [Google Scholar] [CrossRef]
- Yapo, B.M.; Robert, C.; Etienne, I.; Wathelet, B.; Paquot, M. Effect of extraction conditions on the yield, purity and surface properties of sugar beet pulp pectin extracts. Food Chem. 2007, 100, 1356–1364. [Google Scholar] [CrossRef]
- Yang, Z.; Zhai, W. Optimization of microwave-assisted extraction of anthocyanins from purple corn (Zea mays L.) cob and identification with HPLC-MS. Innov. Food Sci. Emerg. Technol. 2010, 11, 470–476. [Google Scholar]
- Pagan, J.; Ibarz, A.; Llorca, M.; Pagan, A.; Barbosa-Canovas, G.V. Extraction and characterization of pectin from stored peach pomace. Food Res. Int. 2001, 34, 605–612. [Google Scholar] [CrossRef]
- Ferguson, K.; da Cruz, M.A.; Ferrarezi, R.; Dorado, C.; Bai, J.; Cameron, R.G. Impact of Huanglongbing (HLB) on grapefruit pectin yield and quality during grapefruit maturation. Food Hydrocoll. 2021, 113, 106553. [Google Scholar] [CrossRef]
- Chua, B.L.; Tang, S.F.; Ali, A.; Chow, Y.H. Optimisation of pectin production from dragon fruit peels waste: Drying, extraction and characterisation studies. SN Appl. Sci. 2020, 2, 621. [Google Scholar] [CrossRef]
- Baississe, S. Extraction et appréciation des Pectines à Partir d’écorces d’oranges, de Pulpes d’abricots et de Pommes. Master’s Dissertation, Faculty of Science, University Hadj Lakhdar, Batna, Algeria, 2009. [Google Scholar]
- Khotimchenko, M.; Kovalev, V.; Khotimchenko, Y. Equilibrium studies of sorption of lead (II) ions by different pectin compounds. J. Hazard. Mater. 2007, 149, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Herbstreith & Fox. Chances and Limits for the Use of Pectin as Emulsifier. In Proceedings of the Lecture of the Master Class on Emulsion Technology Held at FI Food Ingredients Europe, Frankfurt, Germany, 3–5 November 1998. [Google Scholar]
- Massiot, L.; Renard, C.M.G.C. Composition physicochemical properties and enzymatic degradation of fibres prepared from different tissues of apple. Lebensm. Wiss. U. Technol. 1997, 30, 800–806. [Google Scholar] [CrossRef]
- Lekbir, A. Extraction et appréciation des pectines à partir des écorces d’oranges et de dattes. Master’s Dissertation, Faculty of Science, University Hadj Lakhdar, Batna, Algeria, 2008. [Google Scholar]
- Kurz, C.; Carle, R.; Schieber, A. Characterisation of cell wall polysaccharide profiles of apricots (Prunus armeniaca L.), Peaches (Prunus persica L.), and pumpkins (Cucurbita sp.) for the evaluation of fruit product authenticity. Food Chem. 2008, 106, 421–430. [Google Scholar] [CrossRef]
- Thang, P.T.N. Ripening Behaviour of Capsicum (Capsicum annum L.) Fruit. Ph.D. Thesis, Faculty of Science, Adelaide University, Adelaide, Australia, 2007. [Google Scholar]
- Benchabane, A. Rapport de Synthèse de L’atelier “Technologie et Qualité de la Datte”. Ciheam–Options Mediterranèennes; Institut National Agronomique ElHarrach16200 Alger: Alger, Algeria, 1994. [Google Scholar]
- Sahari, M.A.; Akbarian, A.M.; Hamedi, M. Effect of variety and acid washing method on extraction yield and quality of sunflower head pectin. Food Chem. 2003, 83, 43–47. [Google Scholar] [CrossRef]
- Leroux, J.; Langendorff, V.; Schick, G.; Vaishnav, V.; Mazyer, J. Emulsion stabilising properties of pectin. Food Hydrocoll. 2003, 1, 455–462. [Google Scholar] [CrossRef]
- Kamoun, A.; Samet, B.; Bouaziz, J.; Chaabouni, M. Application of a rotatable orthogonal central composite design to the optimization of the formulation and utilization of an useful plasticizer for cement. Analysis 1999, 27, 91–96. [Google Scholar] [CrossRef]
- Mc Ceady, R.M. Pectin, Methods in Food Analysis, 2nd ed.; Joslyn, M.A., Ed.; Academic Press: New York, NY, USA; London, UK, 1970; pp. 565–599. [Google Scholar]
- AOAC. Official Methods of Analysis, 14th ed.; Association of Official Analytical Chemists: Washington, DC, USA, 1984. [Google Scholar]
- AOAC. Official Methods of Analysis, 12th ed.; Association of Official Analytical Chemists (AOAC): Washington, DC, USA, 1995. [Google Scholar]
- Dubois, M.K. Use of phenol reagent for the determination of total sugar. Anal. Chem. 1956, 28, 350. [Google Scholar] [CrossRef]
- Yapo, B.M. Biochemical Characteristics and Gelling Capacity of Pectin from Yellow Passion Fruit Rind as Affected by Acid Extractant Nature. J. Agric. Food Chem. 2009, 57, 1572–1578. [Google Scholar] [CrossRef]
- Hosseini, S.S.; Khodaiyan, F.; Yarmand, M.S. Optimization of microwave assisted extraction of pectin from sour orange peel and its physicochemical properties. Carbohydr. Polym. 2016, 140, 59–65. [Google Scholar] [CrossRef]


| Factor 1 | Factor 2 | Factor 3 | Response 1 | |
|---|---|---|---|---|
| Std | X1: Particle Size | X2: pH | X3: Temperature | Yield |
| mm | °C | % | ||
| 1 | 0.1 | 1.5 | 80 | 25.4 |
| 2 | 1 | 1.5 | 80 | 18.7 |
| 3 | 0.1 | 3 | 80 | 10.5 |
| 4 | 1 | 3 | 80 | 3.8 |
| 5 | 0.1 | 2.25 | 60 | 17.4 |
| 6 | 1 | 2.25 | 60 | 10.8 |
| 7 | 0.1 | 2.25 | 100 | 18.7 |
| 8 | 1 | 2.25 | 100 | 19.3 |
| 9 | 0.55 | 1.5 | 60 | 21.5 |
| 10 | 0.55 | 3 | 60 | 6.5 |
| 11 | 0.55 | 1.5 | 100 | 22.8 |
| 12 | 0.55 | 3 | 100 | 7.8 |
| 13 | 0.55 | 2.25 | 80 | 14.7 |
| 14 | 0.55 | 2.25 | 80 | 14.6 |
| Source | Sequential p-Value | Lack-of-Fit p-Value | Adjusted R2 | Predicted R2 | |
|---|---|---|---|---|---|
| Linear | <0.0001 | 0.0271 | 0.9130 | 0.8556 | Suggested |
| 2FI | 0.3571 | 0.0271 | 0.9194 | 0.7681 | |
| Quadratic | 0.4610 | 0.0247 | 0.9212 | 0.6123 | |
| Cubic | 0.0247 | 0.9999 | Aliased |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Linear | 36.83 | 9 | 4.09 | 818.41 | 0.0271 | Suggested |
| 2FI | 23.87 | 6 | 3.98 | 795.62 | 0.0271 | |
| Quadratic | 13.33 | 3 | 4.44 | 888.67 | 0.0247 | |
| Cubic | 0.0000 | 0 | Aliased | |||
| Pure Error | 0.0050 | 1 | 0.0050 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 513.27 | 3 | 171.09 | 46.45 | <0.0001 | Significant |
| X1—Particle size | 47.05 | 1 | 47.05 | 12.77 | 0.0051 | |
| X2—pH | 447.01 | 1 | 447.01 | 121.36 | <0.0001 | |
| X3—Temperature | 19.22 | 1 | 19.22 | 5.22 | 0.0455 | |
| Residual | 36.83 | 10 | 3.68 | |||
| Lack of Fit | 36.83 | 9 | 4.09 | 818.41 | 0.0271 | Significant |
| Pure Error | 0.0050 | 1 | 0.0050 | |||
| Cor Total | 550.10 | 13 |
| Std. dev. | 1.92 | R2 | 0.9330 |
| Mean | 15.18 | Adjusted R2 | 0.9130 |
| C.V. % | 12.64 | Predicted R2 | 0.8556 |
| Adeq. Precision | 19.3009 |
| Factor | Coefficient Estimate | df | Standard Error | 95% CI Low | 95% CI High | VIF |
|---|---|---|---|---|---|---|
| Intercept | 15.18 | 1 | 0.5129 | 14.04 | 16.32 | |
| X1—Particle size | −2.43 | 1 | 0.6785 | −3.94 | −0.9131 | 1.0000 |
| X2—pH | −7.48 | 1 | 0.6785 | −8.99 | −5.96 | 1.0000 |
| X3—Temperature | 1.55 | 1 | 0.6785 | 0.0381 | 3.06 | 1.0000 |
| Gelling Power | Emulsifying Activity | |
|---|---|---|
| Extracted pectin | 164° ± 2° SAG a | 38.46% ± 1.66% a |
| Commercial pectin | 150° ± 1° SAG a | 51% ± 2% b |
| Factors | Lower Level | Higher Level | Medium Level | |
|---|---|---|---|---|
| X1 | Particle size (mm) | 0.1 | 1 | 0.55 |
| X2 | pH | 1.5 | 3 | 2.25 |
| X3 | Temperature (°C) | 60 | 100 | 80 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azzouzi, H.; Elhajji, L.; Achchoub, M.; Salmaoui, S.; Ammadi, A.; Harrak, H.; Touzani, R.; Noutfia, Y.; Elfazazi, K. Moroccan Citrus clementina Peels: Optimization of Pectin Extraction and Determination of Chemical and Functional Properties. Plants 2023, 12, 3417. https://doi.org/10.3390/plants12193417
Azzouzi H, Elhajji L, Achchoub M, Salmaoui S, Ammadi A, Harrak H, Touzani R, Noutfia Y, Elfazazi K. Moroccan Citrus clementina Peels: Optimization of Pectin Extraction and Determination of Chemical and Functional Properties. Plants. 2023; 12(19):3417. https://doi.org/10.3390/plants12193417
Chicago/Turabian StyleAzzouzi, Hanane, Loubna Elhajji, Mouad Achchoub, Souad Salmaoui, Abdelillah Ammadi, Hasnaa Harrak, Rachid Touzani, Younes Noutfia, and Kaoutar Elfazazi. 2023. "Moroccan Citrus clementina Peels: Optimization of Pectin Extraction and Determination of Chemical and Functional Properties" Plants 12, no. 19: 3417. https://doi.org/10.3390/plants12193417
APA StyleAzzouzi, H., Elhajji, L., Achchoub, M., Salmaoui, S., Ammadi, A., Harrak, H., Touzani, R., Noutfia, Y., & Elfazazi, K. (2023). Moroccan Citrus clementina Peels: Optimization of Pectin Extraction and Determination of Chemical and Functional Properties. Plants, 12(19), 3417. https://doi.org/10.3390/plants12193417







