Metabolic Effects of Elicitors on the Biosynthesis of Tropane Alkaloids in Medicinal Plants
Abstract
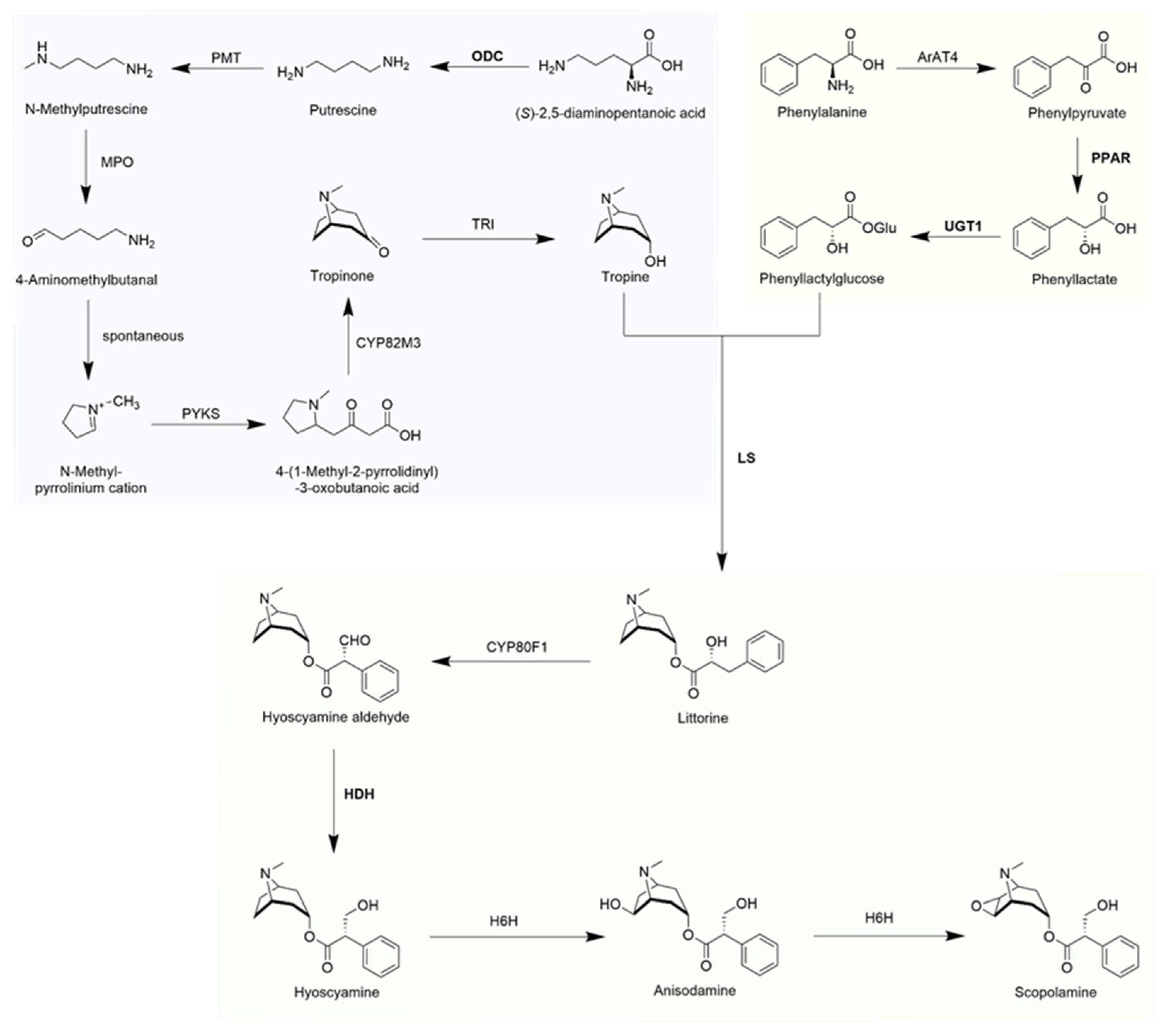
:1. Introduction
2. Elicitors
3. Effects of Biotic Elicitors on TAs
3.1. Sugars, Proteins, and Their Precursors and Derivatives
3.2. Yeast Extract
3.3. Fungi
3.4. Bacteria
3.5. Viruses
4. Effects of Abiotic Elicitors on TAs
4.1. Physical Elicitors
4.2. Chemical Elicitors
4.3. Plant Hormones and Growth Regulators
4.4. New Types
5. Effects of Combined Elicitation on TAs
6. Combination of Elicitors with Other Strategies to Increase TAs
7. Prosperity
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- O’Hagan, D. Pyrrole, Pyrrolidine, Pyridine, Piperidine and Tropane Alkaloids. Nat. Prod. Rep. 2000, 17, 435–446. [Google Scholar] [CrossRef]
- Huang, J.P.; Wang, Y.J.; Tian, T.; Wang, L.; Yan, Y.; Huang, S.X. Tropane Alkaloid Biosynthesis: A Centennial Review. Nat. Prod. Rep. 2021, 38, 1634–1658. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The Traditional Medicine and Modern Medicine from Natural Products. Molecules 2016, 21, 559. [Google Scholar] [CrossRef] [PubMed]
- Xia, K.; Liu, X.; Zhang, Q.; Qiang, W.; Guo, J.; Lan, X.; Chen, M.; Liao, Z. Promoting Scopolamine Biosynthesis in Transgenic Atropa belladonna Plants with Pmt and H6h Overexpression under Field Conditions. Plant Physiol. Biochem. 2016, 106, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Kai, G.Y.; Lu, B.B.; Zhang, H.M.; Tang, K.X.; Jiang, J.H.; Chen, W.S. Metabolic Engineering of Tropane Alkaloid Biosynthesis in Plants. J. Integr. Plant Biol. 2005, 47, 136–143. [Google Scholar] [CrossRef]
- Ramachandra Rao, S.; Ravishankar, G.A. Plant Cell Cultures: Chemical Factories of Secondary Metabolites. Biotechnol. Adv. 2002, 20, 101–153. [Google Scholar] [CrossRef] [PubMed]
- Boller, T.; Felix, G. A Renaissance of Elicitors: Perception of Microbe-Associated Molecular Patterns and Danger Signals by Pattern-Recognition Receptors. Annu. Rev. Plant Biol. 2009, 60, 379–406. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor Signal Transduction Leading to Production of Plant Secondary Metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef]
- Hammerschmidt, R. PHYTOALEXINS: What Have We Learned After 60 Years? Annu. Rev. Phytopathol. 1999, 37, 285–306. [Google Scholar] [CrossRef]
- Boller, T. Chemoperception of Microbial Signals in Plant Cells. Annu. Rev. Plant Physiol. Mol. Biol. 1995, 46, 189–214. [Google Scholar] [CrossRef]
- Halder, M.; Sarkar, S.; Jha, S. Elicitation: A Biotechnological Tool for Enhanced Production of Secondary Metabolites in Hairy Root Cultures. Eng. Life Sci. 2019, 19, 880–895. [Google Scholar] [CrossRef] [PubMed]
- Guru, A.; Dwivedi, P.; Kaur, P.; Pandey, D.K. Exploring the Role of Elicitors in Enhancing Medicinal Values of Plants under in Vitro Condition. S. Afr. J. Bot. 2022, 149, 1029–1043. [Google Scholar] [CrossRef]
- Chen, H.; Chena, F.; Chiu, F.C.K.; Lo, C.M.Y. The Effect of Yeast Elicitor on the Growth and Secondary Metabolism of Hairy Root Cultures of Salvia miltiorrhiza. Enzym. Microb. Technol. 2001, 28, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.S.; Chattopadhyay, T.; Thakur, D.; Kumar, N.; Kumar, T.; Singh, P.K. Hairy Root Culture for In Vitro Production of Secondary Metabolites: A Promising Biotechnological Approach. In Biotechnological Approaches for Medicinal and Aromatic Plants: Conservation, Genetic Improvement and Utilization; Kumar, N., Ed.; Springer: Singapore, 2018; pp. 235–250. ISBN 9789811305351. [Google Scholar]
- Georgiev, M.I.; Agostini, E.; Ludwig-Müller, J.; Xu, J. Genetically Transformed Roots: From Plant Disease to Biotechnological Resource. Trends Biotechnol. 2012, 30, 528–537. [Google Scholar] [CrossRef]
- Halder, T.; Ghosh, B. Hairy Root Cultures of Physalis minima L.—An Alternative Source of Withaferin A Production. Plant Cell Tissue Organ Cult. 2023, 152, 31–44. [Google Scholar] [CrossRef]
- Frankfater, C.R.; Dowd, M.K.; Triplett, B.A. Effect of Elicitors on the Production of Gossypol and Methylated Gossypol in Cotton Hairy Roots. Plant Cell Tissue Organ Cult. 2009, 98, 341–349. [Google Scholar] [CrossRef]
- Häkkinen, S.T.; Moyano, E.; Cusidó, R.M.; Oksman-Caldentey, K.M. Exploring the Metabolic Stability of Engineered Hairy Roots after 16 Years Maintenance. Front. Plant Sci. 2016, 7, 1486. [Google Scholar] [CrossRef]
- Jeong, G.T.; Park, D.H.; Ryu, H.W.; Hwang, B.; Woo, J.C.; Kim, D.; Kim, S.W. Production of Antioxidant Compounds by Culture of Panax ginseng C.A. Meyer Hairy Roots. In Twenty-Sixth Symposium on Biotechnology for Fuels and Chemicals; Davison, B.H., Evans, B.R., Finkelstein, M., McMillan, J.D., Eds.; Humana Press: Totowa, NJ, USA, 2005; pp. 1147–1157. ISBN 978-1-59259-991-2. [Google Scholar]
- Wiktorowska, E.; Długosz, M.; Janiszowska, W. Significant Enhancement of Oleanolic Acid Accumulation by Biotic Elicitors in Cell Suspension Cultures of Calendula officinalis L. Enzym. Microb. Technol. 2010, 46, 14–20. [Google Scholar] [CrossRef]
- Patil, J.G.; Ahire, M.L.; Nitnaware, K.M.; Panda, S.; Bhatt, V.P.; Kishor, P.B.K.; Nikam, T.D. In Vitro Propagation and Production of Cardiotonic Glycosides in Shoot Cultures of Digitalis purpurea L. by Elicitation and Precursor Feeding. Appl. Microbiol. Biot. 2013, 97, 2379–2393. [Google Scholar] [CrossRef]
- Rothe, G.; Dräger, B. Tropane Alkaloids—Metabolic Response to Carbohydrate Signal in Root Cultures of Atropa belladonna. Plant Sci. 2002, 163, 979–985. [Google Scholar] [CrossRef]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Shafi, M.E.; Zabermawi, N.M.; Arif, M.; Batiha, G.E.; Khafaga, A.F.; Abd El-Hakim, Y.M.; Al-Sagheer, A.A. Antimicrobial and Antioxidant Properties of Chitosan and Its Derivatives and Their Applications: A Review. Int. J. Biol. Macromol. 2020, 164, 2726–2744. [Google Scholar] [CrossRef] [PubMed]
- Stasińska-Jakubas, M.; Hawrylak-Nowak, B. Protective, Biostimulating, and Eliciting Effects of Chitosan and Its Derivatives on Crop Plants. Molecules 2022, 27, 2801. [Google Scholar] [CrossRef] [PubMed]
- El Hadrami, A.; Adam, L.R.; El Hadrami, I.; Daayf, F. Chitosan in Plant Protection. Mar. Drugs 2010, 8, 968–987. [Google Scholar] [CrossRef] [PubMed]
- Pitta-Alvarez, S.I.; Giulietti, A.M. Influence of Chitosan, Acetic Acid and Citric Acid on Growth and Tropane Alkaloid Production in Transformed Roots of Brugmansia candida Effect of Medium PH and Growth Phase. Plant Cell Tissue Organ Cult. 1999, 59, 31–38. [Google Scholar] [CrossRef]
- Lee, K.T.; Yamakawa, T.; Kodama, T.; Shimomura, K. Effects of Chemicals on Alkaloid Production by Transformed Roots of Belladonna. Phytochemistry 1998, 49, 2343–2347. [Google Scholar] [CrossRef]
- Rothe, G.; Garske, U.; Dräger, B. Calystegines in Root Cultures of Atropa belladonna Respond to Sucrose, Not to Elicitation. Plant Sci. 2001, 160, 1043–1053. [Google Scholar] [CrossRef]
- Hong, M.L.K.; Bhatt, A.; Ping, N.S.; Keng, C.L. Detection of Elicitation Effect on Hyoscyamus niger L. Root Cultures for the Root Growth and Production of Tropane Alkaloids. Rom. Biotechnol. Lett. 2012, 17, 7340–7351. [Google Scholar]
- Benedetti, M.; Mattei, B.; Pontiggia, D.; Salvi, G.; Savatin, D.V.; Ferrari, S. Methods of Isolation and Characterization of Oligogalacturonide Elicitors. In Plant Pattern Recognition Receptors: Methods and Protocols; Shan, L., He, P., Eds.; Springer: New York, NY, USA, 2017; pp. 25–38. ISBN 978-1-4939-6859-6. [Google Scholar]
- Cabrera, J.C.; Boland, A.; Messiaen, J.; Cambier, P.; Van Cutsem, P. Egg Box Conformation of Oligogalacturonides: The Time-Dependent Stabilization of the Elicitor-Active Conformation Increases Its Biological Activity. Glycobiology 2008, 18, 473–482. [Google Scholar] [CrossRef]
- Zabetakis, I.; Edwards, R.; O’Hagan, D. Elicitation of Tropane Alkaloid Biosynthesis in Transformed Root Cultures of Datura stramonium. Phytochemistry 1999, 50, 53–56. [Google Scholar] [CrossRef]
- Tauzin, A.S.; Giardina, T. Sucrose and Invertases, a Part of the Plant Defense Response to the Biotic Stresses. Front. Plant Sci. 2014, 5, 293. [Google Scholar] [CrossRef]
- Tun, W.; Yoon, J.; Vo, K.T.X.; Cho, L.H.; Hoang, T.V.; Peng, X.; Kim, E.J.; Win, K.T.Y.S.; Lee, S.W.; Jung, K.H.; et al. Sucrose Preferentially Promotes Expression of OsWRKY7 and OsPR10a to Enhance Defense Response to Blast Fungus in Rice. Front. Plant Sci. 2023, 14, 1117023. [Google Scholar] [CrossRef]
- Morkunas, I.; Marczak, Ł.; Stachowiak, J.; Stobiecki, M. Sucrose-Induced Lupine Defense against Fusarium oxysporum. Sucrose-Stimulated Accumulation of Isoflavonoids as a Defense Response of Lupine to Fusarium oxysporum. Plant Physiol. Biochem. 2005, 43, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmson, A.; Häkkinen, S.T.; Kallio, P.T.; Oksman-Caldentey, K.M.; Nuutila, A.M. Heterologous Expression of Vitreoscilla Hemoglobin (VHb) and Cultivation Conditions Affect the Alkaloid Profile of Hyoscyamus muticus Hairy Roots. Biotechnol. Prog. 2006, 22, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Oksman-Caldentey, K.M.; Sevón, N.; Vanhala, L.; Hiltunen, R. Effect of Nitrogen and Sucrose on the Primary and Secondary Metabolism of Transformed Root Cultures of Hyoscyamus muticus. Plant Cell Tissue Organ Cult. 1994, 38, 263–272. [Google Scholar] [CrossRef]
- Matsubayashi, Y.; Takagi, L.; Sakagami, Y. Phytosulfokine-α, a Sulfated Pentapeptide, Stimulates the Proliferation of Rice Cells by Means of Specific High- and Low-Affinity Binding Sites. Proc. Natl. Acad. Sci. USA 1997, 94, 13357–13362. [Google Scholar] [CrossRef]
- Yu, L.; Di, Q.; Zhang, D.; Liu, Y.; Li, X.; Mysore, K.S.; Wen, J.; Yan, J.; Luo, L. A Legume-Specific Novel Type of Phytosulfokine, PSK-δ, Promotes Nodulation by Enhancing Nodule Organogenesis. J. Exp. Bot. 2022, 73, 2698–2713. [Google Scholar] [CrossRef]
- Sasaki, K.; Ishise, T.; Shimomura, K.; Kobayashi, T.; Matsubayashi, Y.; Sakagami, Y.; Umetsu, H.; Kamada, H. Effects of Phytosulfokine-α on Growth and Tropane Alkaloid Production in Transformed Roots of Atropa belladonna. Plant Growth Regul. 2002, 36, 87–90. [Google Scholar] [CrossRef]
- Haile, S.; Ayele, A. Pectinase from Microorganisms and Its Industrial Applications. Sci. World J. 2022, 2022, e1881305. [Google Scholar] [CrossRef]
- Płażek, A.; Hura, K.; Żur, I. Reaction of Winter Oilseed Rape Callus to Different Concentrations of Elicitors: Pectinase or Chitosan. Acta Physiol. Plant. 2003, 25, 83–89. [Google Scholar] [CrossRef]
- Pitta Alvarez, S.; Marconi, P.L.; Giulietti, A. Comparison of the Influence of Different Elicitors on Hyoscyamine and Scopolamine Content in Hairy Root Cultures of Brugmansia candida. In Vitro Cell. Dev. Biol.-Plant 2003, 39, 640–644. [Google Scholar] [CrossRef]
- Pitta-Alvarez, S.I.; Spollansky, T.C.; Giulietti, A.M. Scopolamine and Hyoscyamine Production by Hairy Root Cultures of Brugmansia candida: Influence of Calcium Chloride, Hemicellulase and Theophylline. Biotechnol. Lett. 2000, 22, 1653–1656. [Google Scholar] [CrossRef]
- Kozioł, A.; Cybulska, J.; Pieczywek, P.M.; Zdunek, A. Changes of Pectin Nanostructure and Cell Wall Stiffness Induced in Vitro by Pectinase. Carbohyd. Polym. 2017, 161, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.; Yuan, H.; Liu, M.; Liu, Q.; Zhang, S.; Liu, H.; Jiang, Y.; Huang, D.; Wang, T. Yeast Extract: Characteristics, Production, Applications and Future Perspectives. J. Microbiol. Biotechnol. 2023, 33, 151–166. [Google Scholar] [CrossRef] [PubMed]
- Maqsood, M.; Abdul, M. Yeast Extract Elicitation Increases Vinblastine and Vincristine Yield in Protoplast Derived Tissues and Plantlets in Catharanthus roseus. Rev. Bras. Farm. 2017, 27, 549–556. [Google Scholar] [CrossRef]
- Chen, H.; Chen, F. Effect of Yeast Elicitor on the Secondary Metabolism of Ti-Transformed Salvia miltiorrhiza Cell Suspension Cultures. Plant Cell Rep. 2000, 19, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Yang, Y.; Liu, X.; Wu, N.B. Mechanism exploration on nitrogen metabolism and secondary metabolism in Atropa belladonna hairy roots treated with yeast extract. China J. Chin. Mater. Medica 2018, 43, 1610–1617. (In Chinese) [Google Scholar] [CrossRef]
- Hedayati, A.; Hemmaty, S.; Nourozi, E.; Amirsadeghi, A. Effect of Yeast Extract on H6h Gene Expression and Tropane Alkaloids Production in Atropa belladonna L. Hairy Roots. Russ. J. Plant Physiol. 2021, 68, 102–109. [Google Scholar] [CrossRef]
- Pitta–Alvarez, S.I.; Spollansky, T.C.; Giulietti, A.M. The Influence of Different Biotic and Abiotic Elicitors on the Production and Profile of Tropane Alkaloids in Hairy Root Cultures of Brugmansia candida. Enzym. Microb. Technol. 2000, 26, 252–258. [Google Scholar] [CrossRef]
- Cardillo, A.B.; Perassolo, M.; Sartuqui, M.; Rodríguez Talou, J.; Giulietti, A.M. Production of Tropane Alkaloids by Biotransformation Using Recombinant Escherichia Coli Whole Cells. Biochem. Eng. J. 2017, 125, 180–189. [Google Scholar] [CrossRef]
- Jaremicz, Z.; Luczkiewicz, M.; Kokotkiewicz, A.; Krolicka, A.; Sowinski, P. Production of Tropane Alkaloids in Hyoscyamus niger (Black Henbane) Hairy Roots Grown in Bubble-Column and Spray Bioreactors. Biotechnol. Lett. 2014, 36, 843–853. [Google Scholar] [CrossRef]
- Guo, J.; Cheng, Y. Advances in Fungal Elicitor-Triggered Plant Immunity. Int. J. Mol. Sci. 2022, 23, 12003. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Motaal, F.F.; Nassar, M.S.M.; El-Zayat, S.A.; El-Sayed, M.A.; Ito, S. Responses of Fungi to Tropane Alkaloids Produced by a Medicinal Plant Hyoscyamus muticus (Egyptian Henbane). Folia Microbiol. 2009, 54, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Dabur, R.; Singh, H.; Chhillar, A.K.; Ali, M.; Sharma, G.L. Antifungal Potential of Indian Medicinal Plants. Fitoterapia 2004, 75, 389–391. [Google Scholar] [CrossRef]
- Shafique, S.; Shafique, S. Antifungal Activity of N-Hexane Extracts of Datura metel against Ascochyta rabiei. Mycopath 2008, 6, 31–35. [Google Scholar]
- Ballica, R.; Ryu, D.D.Y.; Kado, C.I. Tropane Alkaloid Production in Datura stramonium Suspension Cultures: Elicitor and Precursor Effects. Biotechnol. Bioeng. 1993, 41, 1075–1081. [Google Scholar] [CrossRef] [PubMed]
- Ajungla, L.; Patil, P.P.; Barmukh, R.B.; Nikam, T.D. Influence of Biotic and Abiotic Elicitors on Accumulation of Hyoscyamine and Scopolamine in Root Cultures of Datura metel L. Indian J. Biotechnol. 2009, 8, 317–322. [Google Scholar]
- Kang, S.M.; Min, J.Y.; Kim, Y.D.; Karigar, C.S.; Kim, S.W.; Goo, G.H.; Choi, M.S. Effect of Biotic Elicitors on the Accumulation of Bilobalide and Ginkgolides in Ginkgo biloba Cell Cultures. J. Biotechnol. 2009, 139, 84–88. [Google Scholar] [CrossRef]
- Jung, H.Y.; Kang, S.M.; Kang, Y.M.; Kang, M.J.; Yun, D.J.; Bahk, J.D.; Yang, J.K.; Choi, M.S. Enhanced Production of Scopolamine by Bacterial Elicitors in Adventitious Hairy Root Cultures of Scopolia parviflora. Enzym. Microb. Technol. 2003, 33, 987–990. [Google Scholar] [CrossRef]
- Moussous, A.; Paris, C.; Khelifi-Slaoui, M.; Bekhouche, M.; Zaoui, D.; Rosloski, S.M.; Makhzoum, A.; Desobry, S.; Khelifi, L. Pseudomonas spp. Increases Root Biomass and Tropane Alkaloid Yields in Transgenic Hairy Roots of Datura spp. In Vitro Cell. Dev. Biol.-Plant 2018, 54, 117–126. [Google Scholar] [CrossRef]
- Lan, H.; Lai, B.; Zhao, P.; Dong, X.; Wei, W.; Ye, Y.; Wu, Z. Cucumber Mosaic Virus Infection Modulated the Phytochemical Contents of Passiflora edulis. Microb. Pathog. 2020, 138, 103828. [Google Scholar] [CrossRef]
- Wink, M. Plant Breeding: Importance of Plant Secondary Metabolites for Protection against Pathogens and Herbivores. Theoret. Appl. Genet. 1988, 75, 225–233. [Google Scholar] [CrossRef]
- Mihálik, D.; Hančinský, R.; Kaňuková, Š.; Mrkvová, M.; Kraic, J. Elicitation of Hyoscyamine Production in Datura stramonium L. Plants Using Tobamoviruses. Plants 2022, 11, 3319. [Google Scholar] [CrossRef] [PubMed]
- Min, J.Y.; Jung, H.Y.; Kang, S.M.; Kim, Y.D.; Kang, Y.M.; Park, D.J.; Prasad, D.T.; Choi, M.S. Production of Tropane Alkaloids by Small-Scale Bubble Column Bioreactor Cultures of Scopolia parviflora Adventitious Roots. Bioresour. Technol. 2007, 98, 1748–1753. [Google Scholar] [CrossRef]
- Shakeran, Z.; Keyhanfar, M.; Ghanadian, M. Biotic Elicitation for Scopolamine Production by Hairy Root Cultures of Datura metel. Mol. Biol. Res. Commun. 2017, 6, 169–179. [Google Scholar] [CrossRef]
- Nowak, M.; Selmar, D. Cellular Distribution of Alkaloids and Their Translocation via Phloem and Xylem: The Importance of Compartment PH. Plant Biol. 2016, 18, 879–882. [Google Scholar] [CrossRef]
- Phyo, P.; Gu, Y.; Hong, M. Impact of Acidic PH on Plant Cell Wall Polysaccharide Structure and Dynamics: Insights into the Mechanism of Acid Growth in Plants from Solid-State NMR. Cellulose 2019, 26, 291–304. [Google Scholar] [CrossRef]
- Angelova, M.I.; Bitbol, A.F.; Seigneuret, M.; Staneva, G.; Kodama, A.; Sakuma, Y.; Kawakatsu, T.; Imai, M.; Puff, N. PH Sensing by Lipids in Membranes: The Fundamentals of PH-Driven Migration, Polarization and Deformations of Lipid Bilayer Assemblies. Biochim. Biophys. Acta-Biomembr. 2018, 1860, 2042–2063. [Google Scholar] [CrossRef] [PubMed]
- Kohnen-Johannsen, K.L.; Kayser, O. Tropane Alkaloids: Chemistry, Pharmacology, Biosynthesis and Production. Molecules 2019, 24, 796. [Google Scholar] [CrossRef]
- Yukimune, Y.; Yamagata, H.; Hara, Y.; Yamada, Y. Effects of Oxygen on Nicotine and Tropane Alkaloid Production in Cultured Roots of Duboisia myoporoides. Biosci. Biotechnol. Biochem. 1994, 58, 1824–1827. [Google Scholar] [CrossRef]
- Williams, G.R.C.; Doran, P.M. Hairy Root Culture in a Liquid-Dispersed Bioreactor: Characterization of Spatial Heterogeneity. Biotechnol. Prog. 2000, 16, 391–401. [Google Scholar] [CrossRef]
- Sauerwein, M.; Wink, M.; Shimomura, K. Influence of Light and Phytohormones on Alkaloid Production in Transformed Root Cultures of Hyoscyamus albus. J. Plant Physiol. 1992, 140, 147–152. [Google Scholar] [CrossRef]
- Baricevic, D.; Umek, A.; Kreft, S.; Maticic, B.; Zupancic, A. Effect of Water Stress and Nitrogen Fertilization on the Content of Hyoscyamine and Scopolamine in the Roots of Deadly Nightshade (Atropa belladonna). Environ. Exp. Bot. 1999, 42, 17–24. [Google Scholar] [CrossRef]
- Halperin, S.J.; Flores, H.E. Hyoscyamine and Proline Accumulation in Water-Stressed Hyoscyamus muticus ‘Hairy Root’ Cultures. In Vitro Cell. Dev. Biol.-Plant 1997, 33, 240–244. [Google Scholar] [CrossRef]
- Zhang, W.J.; Björn, L.O. The Effect of Ultraviolet Radiation on the Accumulation of Medicinal Compounds in Plants. Fitoterapia 2009, 80, 207–218. [Google Scholar] [CrossRef]
- Qin, B.; Ma, L.; Wang, Y.; Chen, M.; Lan, X.; Wu, N.; Liao, Z. Effects of Acetylsalicylic Acid and UV-B on Gene Expression and Tropane Alkaloid Biosynthesis in Hairy Root Cultures of Anisodus luridus. Plant Cell Tissue Organ Cult. 2014, 117, 483–490. [Google Scholar] [CrossRef]
- Petersen, O.H.; Michalak, M.; Verkhratsky, A. Calcium Signalling: Past, Present and Future. Cell Calcium 2005, 38, 161–169. [Google Scholar] [CrossRef]
- Kadota, Y.; Kuchitsu, K. Regulation of Elicitor-Induced Defense Responses by Ca2+ Channels and the Cell Cycle in Tobacco BY-2 Cells. In Tobacco BY-2 Cells: From Cellular Dynamics to Omics; Nagata, T., Matsuoka, K., Inzé, D., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 207–221. ISBN 978-3-540-32674-8. [Google Scholar]
- Amdoun, R.; Khelifi, L.; Khelifi-Slaoui, M.; Amroune, S.; Asch, M.; Assaf-Ducrocq, C.; Gontier, E. Optimization of the Culture Medium Composition to Improve the Production of Hyoscyamine in Elicited Datura stramonium L. Hairy Roots Using the Response Surface Methodology (RSM). Int. J. Mol. Sci. 2010, 11, 4726–4740. [Google Scholar] [CrossRef]
- Gontier, E.; Sangwan, B.S.; Barbotin, J.N. Effects of Calcium, Alginate, and Calcium-Alginate Immobilization on Growth and Tropane Alkaloid Levels of a Stable Suspension Cell Line of Datura innoxia Mill. Plant Cell Rep. 1994, 13, 533–536. [Google Scholar] [CrossRef]
- Murthy, H.N.; Dandin, V.S.; Zhong, J.J.; Paek, K.Y. Strategies for Enhanced Production of Plant Secondary Metabolites from Cell and Organ Cultures. In Production of Biomass and Bioactive Compounds Using Bioreactor Technology; Paek, K.Y., Murthy, H.N., Zhong, J.J., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 471–508. ISBN 978-94-017-9223-3. [Google Scholar]
- Harfi, B.; Khelifi-Slaoui, M.; Bekhouche, M.; Benyammi, R.; Hefferon, K.; Makhzoum, A.; Khelifi, L. Hyoscyamine Production in Hairy Roots of Three Datura Species Exposed to High-Salt Medium. In Vitro Cell. Dev. Biol.-Plant 2016, 52, 92–98. [Google Scholar] [CrossRef]
- Piñol, M.T.; Palazón, J.; Cusidó, R.M.; Ribó, M. Influence of Calcium Ion-Concentration in the Medium on Tropane Alkaloid Accumulation in Datura stramonium Hairy Roots. Plant Sci. 1999, 141, 41–49. [Google Scholar] [CrossRef]
- Karimi, F.; Khataee, E. Aluminum Elicits Tropane Alkaloid Production and Antioxidant System Activity in Micropropagated Datura innoxia Plantlets. Acta Physiol. Plant. 2012, 34, 1035–1041. [Google Scholar] [CrossRef]
- Hall, J.L. Cellular Mechanisms for Heavy Metal Detoxification and Tolerance. J. Exp. Bot. 2002, 53, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Furze, J.M.; Rhodes, M.J.C.; Parr, A.J.; Robins, R.J.; Withehead, I.M.; Threlfall, D.R. Abiotic Factors Elicit Sesquiterpenoid Phytoalexin Production but Not Alkaloid Production in Transformed Root Cultures of Datura stramonium. Plant Cell Rep. 1991, 10, 111–114. [Google Scholar] [CrossRef]
- Kai, G.; Yang, S.; Zhang, Y.; Luo, X.; Fu, X.; Zhang, A.; Xiao, J. Effects of Different Elicitors on Yield of Tropane Alkaloids in Hairy Roots of Anisodus acutangulus. Mol. Biol. Rep. 2012, 39, 1721–1729. [Google Scholar] [CrossRef] [PubMed]
- Vakili, B.; Karimi, F.; Sharifi, M.; Behmanesh, M. Chromium-Induced Tropane Alkaloid Production and H6H Gene Expression in Atropa belladonna L. (Solanaceae) in Vitro-Propagated Plantlets. Plant Physiol. Biochnol. 2012, 52, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Verma, V.; Ravindran, P.; Kumar, P.P. Plant Hormone-Mediated Regulation of Stress Responses. BMC Plant Biol. 2016, 16, 86. [Google Scholar] [CrossRef] [PubMed]
- Mou, W.; Kao, Y.T.; Michard, E.; Simon, A.A.; Li, D.; Wudick, M.M.; Lizzio, M.A.; Feijó, J.A.; Chang, C. Ethylene-Independent Signaling by the Ethylene Precursor ACC in Arabidopsis Ovular Pollen Tube Attraction. Nat. Commun. 2020, 11, 4082. [Google Scholar] [CrossRef]
- Moradi, A.; Sharifi, M.; Mousavi, A. Induced Production of Tropane Alkaloids, and Expression of Hyoscyamine 6β-Hydroxylase (h6h) and Putrescine N-Methyl Transferase (pmt2) Genes in Hairy Roots and Propagated Plantlets of Atropa belladonna L. Elicited by Methyl Jasmonate. S. Afr. J. Bot. 2020, 131, 328–334. [Google Scholar] [CrossRef]
- Kang, S.M.; Jung, H.Y.; Kang, Y.M.; Yun, D.J.; Bahk, J.D.; Yang, J.; Choi, M.S. Effects of Methyl Jasmonate and Salicylic Acid on the Production of Tropane Alkaloids and the Expression of PMT and H6H in Adventitious Root Cultures of Scopolia parviflora. Plant Sci. 2004, 166, 745–751. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, B.; Lu, B.; Kai, G.; Wang, Z.; Xia, Y.; Ding, R.; Zhang, H.; Sun, X.; Chen, W.; et al. Tropane Alkaloids Production in Transgenic Hyoscyamus niger Hairy Root Cultures Over-Expressing Putrescine N-Methyltransferase Is Methyl Jasmonate-Dependent. Planta 2007, 225, 887–896. [Google Scholar] [CrossRef]
- Biondi, S.; Fornalé, S.; Oksman-Caldentey, K.M.; Eeva, M.; Agostani, S.; Bagni, N. Jasmonates Induce Over-Accumulation of Methylputrescine and Conjugated Polyamines in Hyoscyamus muticus L. Root Cultures. Plant Cell Rep. 2000, 19, 691–697. [Google Scholar] [CrossRef]
- Sun, J.W.; Zhang, H.; Wang, F.Y.; Sun, Y.M.; Sun, M. Effects of methyl jasmonate on accumulation and release of main tropane alkaloids in liquid cultures of Datura stramonium hairy root. China J. Chin. Mater. Medica 2013, 38, 1712–1718. (In Chinese) [Google Scholar]
- Zehra, A.; Raytekar, N.A.; Meena, M.; Swapnil, P. Efficiency of Microbial Bio-Agents as Elicitors in Plant Defense Mechanism under Biotic Stress: A Review. Curr. Res. Microb. Sci. 2021, 2, 100054. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.; Kachroo, P.; Klessig, D.F. The Arabidopsis ssi1 Mutation Restores Pathogenesis-Related Gene Expression in npr1 Plants and Renders Defensin Gene Expression Salicylic Acid Dependent. Plant Cell 1999, 11, 191–206. [Google Scholar] [CrossRef]
- Harfi, B.; Khelifi, L.; Khelifi-Slaoui, M.; Assaf-Ducrocq, C.; Gontier, E. Tropane Alkaloids GC/MS Analysis and Low Dose Elicitors’ Effects on Hyoscyamine Biosynthetic Pathway in Hairy Roots of Algerian Datura Species. Sci. Rep. 2018, 8, 17951. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Hirano, H.; Yamakawa, T.; Kodama, T.; Igarashi, Y.; Shimomura, K. Responses of Transformed Root Culture of Atropa belladonna to Salicylic Acid Stress. J. Biosci. Bioeng. 2001, 91, 586–589. [Google Scholar] [CrossRef]
- Vanhala, L.; Eeva, M.; Lapinjoki, S.; Hiltunen, R.; Oksman-Caldentey, K.M. Effect of Growth Regulators on Transformed Root Cultures of Hyoscyamus muticus. J. Plant Physiol. 1998, 153, 475–481. [Google Scholar] [CrossRef]
- Hashimoto, T.; Yukimune, Y.; Yamada, Y. Tropane Alkaloid Production in Hyoscyamus Root Cultures. J. Plant Physiol. 1986, 124, 61–75. [Google Scholar] [CrossRef]
- Hashimoto, T.; Yamada, Y. Effects of Culture Conditions on Tropane Alkaloid Formation in Hyoscyamus niger Suspension Cultures. Agric. Biol. Chem. 1987, 51, 2769–2774. [Google Scholar] [CrossRef]
- Sáenz-Carbonell, L.; Loyola-Vargas, V.M. Datura stramonium Hairy Roots Tropane Alkaloid Content as a Response to Changes in Gamborg’s B5 Medium. Appl. Biochem. Biotechnol. 1997, 61, 321–337. [Google Scholar] [CrossRef]
- Fahad, S.; Hussain, S.; Bano, A.; Saud, S.; Hassan, S.; Shan, D.; Khan, F.A.; Khan, F.; Chen, Y.; Wu, C.; et al. Potential Role of Phytohormones and Plant Growth-Promoting Rhizobacteria in Abiotic Stresses: Consequences for Changing Environment. Environ. Sci. Pollut. Res. 2015, 22, 4907–4921. [Google Scholar] [CrossRef] [PubMed]
- Pitta-Alvarez, S.I.; Giulietti, A.M. Effects of Gibberellin GA7 on Kinetics of Growth and Tropane Alkaloid Accumulation in Hairy Roots of Brugmansia candida. In Vitro Cell. Dev. Biol.-Plant 1997, 33, 147–153. [Google Scholar] [CrossRef]
- Luo, X.; Weng, S.; Ni, X.; Wang, X.; Fu, X.; Xiao, J.; Kai, G. The Effects of Elicitation on the Expression of Key Enzyme Genes and on Production of Tropane Alkaloids in Anisodus acutangulus Plant. Biologia 2012, 67, 352–359. [Google Scholar] [CrossRef]
- Dinalli, R.P.; Buzetti, S.; Gazola, R.d.N.; de Castilho, R.M.M.; Jalal, A.; Galindo, F.S.; Teixeira Filho, M.C.M. Nitrogen Fertilization and Glyphosate as a Growth Regulator: Effects on the Nutritional Efficiency and Nutrient Balance in Emerald Grass. Agronomy 2022, 12, 2473. [Google Scholar] [CrossRef]
- Deng, F. Effects of Glyphosate, Chlorsulfuron, and Methyl Jasmonate on Growth and Alkaloid Biosynthesis of Jimsonweed (Datura stramonium L.). Pestic. Biochem. Phys. 2005, 82, 16–26. [Google Scholar] [CrossRef]
- Xu, J.; Zhou, Y.; Xu, Z.; Chen, Z.; Duan, L. Physiological and Transcriptome Profiling Analyses Reveal Important Roles of Coronatine in Improving Drought Tolerance of Tobacco. J. Plant Growth Regul. 2020, 39, 1346–1358. [Google Scholar] [CrossRef]
- Alcalde, M.A.; Perez-Matas, E.; Escrich, A.; Cusido, R.M.; Palazon, J.; Bonfill, M. Biotic Elicitors in Adventitious and Hairy Root Cultures: A Review from 2010 to 2022. Molecules 2022, 27, 5253. [Google Scholar] [CrossRef]
- Fattahi, F.; Shojaeiyan, A.; Palazon, J.; Moyano, E.; Torras-Claveria, L. Methyl-β-Cyclodextrin and Coronatine as New Elicitors of Tropane Alkaloid Biosynthesis in Atropa acuminata and Atropa belladonna Hairy Root Cultures. Physiol. Plant. 2021, 172, 2098–2111. [Google Scholar] [CrossRef]
- Jiao, Q.; Li, L.; Mu, Q.; Zhang, Q. Immunomodulation of Nanoparticles in Nanomedicine Applications. Biomed Res. Int. 2014, 2014, e426028. [Google Scholar] [CrossRef]
- Asl, K.R.; Hosseini, B.; Sharafi, A.; Palazon, J. Influence of Nano-Zinc Oxide on Tropane Alkaloid Production, H6h Gene Transcription and Antioxidant Enzyme Activity in Hyoscyamus reticulatus L. Hairy Roots. Eng. Life Sci. 2019, 19, 73–89. [Google Scholar] [CrossRef]
- Dev, A.; Srivastava, A.K.; Karmakar, S. Nanomaterial Toxicity for Plants. Environ. Chem. Lett. 2018, 16, 85–100. [Google Scholar] [CrossRef]
- Tripathi, D.K.; Shweta; Singh, S.; Singh, S.; Pandey, R.; Singh, V.P.; Sharma, N.C.; Prasad, S.M.; Dubey, N.K.; Chauhan, D.K. An Overview on Manufactured Nanoparticles in Plants: Uptake, Translocation, Accumulation and Phytotoxicity. Plant Physiol. Biochnol. 2017, 110, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Moharrami, F.; Hosseini, B.; Sharafi, A.; Farjaminezhad, M. Enhanced Production of Hyoscyamine and Scopolamine from Genetically Transformed Root Culture of Hyoscyamus reticulatus L. Elicited by Iron Oxide Nanoparticles. In Vitro Cell. Dev. Biol.-Plant 2017, 53, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Sepehri, E.; Hosseini, B.; Hedayati, A. The Effect of Iron Oxide Nano-Particles on the Production of Tropane Alkaloids, h6h Gene Expression and Antioxidant Enzyme Activity in Atropa belladonna Hairy Roots. Russ. J. Plant Physiol. 2022, 69, 122. [Google Scholar] [CrossRef]
- Hedayati, A.; Hosseini, B.; Palazon, J.; Maleki, R. Improved Tropane Alkaloid Production and Changes in Gene Expression in Hairy Root Cultures of Two Hyoscyamus Species Elicited by Silicon Dioxide Nanoparticles. Plant Physiol. Biochnol. 2020, 155, 416–428. [Google Scholar] [CrossRef]
- Lamattina, L.; García-Mata, C.; Graziano, M.; Pagnussat, G. Nitric Oxide: The Versatility of an Extensive Signal Molecule. Annu. Rev. Plant Biol. 2003, 54, 109–136. [Google Scholar] [CrossRef]
- Zhang, B.; Zheng, L.P.; Wang, J.W. Nitric Oxide Elicitation for Secondary Metabolite Production in Cultured Plant Cells. Appl. Microbiol. Biotechnol. 2012, 93, 455–466. [Google Scholar] [CrossRef]
- Khezerluo, M.; Hosseini, B.; Amiri, J. Sodium Nitroprusside Stimulated Production of Tropane Alkaloids and Antioxidant Enzymes Activity in Hairy Root Culture of Hyoscyamus reticulatus L. Acta Biol. Hung. 2018, 69, 437–448. [Google Scholar] [CrossRef]
- Endo, T.; Yamada, Y. Alkaloid Production in Cultured Roots of Three Species of Duboisia. Phytochemistry 1985, 24, 1233–1236. [Google Scholar] [CrossRef]
- Flores, H.E.; Dai, Y.; Cuello, J.L.; Maldonado-Mendoza, I.E.; Loyola-Vargas, V.M. Green Roots: Photosynthesis and Photoautotrophy in an Underground Plant Organ. Plant Physiol. 1993, 101, 363–371. [Google Scholar] [CrossRef]
- Zeynali, Z.; Hosseini, B.; Rezaei, E. Effect of Elicitation on Antioxidant Activity and Production of Tropane Alkaloids in Hyoscyamus reticulatus Hairy Root Cultures. J. Pharmacogn. 2016, 3, 43–53. [Google Scholar]
- el Jaber-Vazdekis, N.; Barres, M.L.; Ravelo, Á.G.; Zárate, R. Effects of Elicitors on Tropane Alkaloids and Gene Expression in Atropa baetica Transgenic Hairy Roots. J. Nat. Prod. 2008, 71, 2026–2031. [Google Scholar] [CrossRef] [PubMed]
- Ghorbanpour, M.; Hatami, M.; Hatami, M. Activating Antioxidant Enzymes, Hyoscyamine and Scopolamine Biosynthesis of Hyoscyamus niger L. Plants with Nano-Sized Titanium Dioxide and Bulk Application. Acta Agric. Slov. 2015, 105, 23–32. [Google Scholar] [CrossRef]
- Szejtli, J. Past, Present and Futute of Cyclodextrin Research. Pure Appl. Chem. 2004, 76, 1825–1845. [Google Scholar] [CrossRef]
- Sabater-Jara, A.B.; Onrubia, M.; Moyano, E.; Bonfill, M.; Palazón, J.; Pedreño, M.A.; Cusidó, R.M. Synergistic Effect of Cyclodextrins and Methyl Jasmonate on Taxane Production in Taxus x Media Cell Cultures. Plant Biotechnol. J. 2014, 12, 1075–1084. [Google Scholar] [CrossRef]
- Ghorbanpour, M.; Hatami, M.; Khavazi, K. Role of Plant Growth Promoting Rhizobacteria on Antioxidant Enzyme Activities and Tropane Alkaloid Production of Hyoscyamus niger under Water Deficit Stress. Turk. J. Biol. 2013, 37, 350–360. [Google Scholar] [CrossRef]
- Yuwono, T.; Handayani, D.; Soedarsono, J.; Yuwono, T.; Handayani, D.; Soedarsono, J. The Role of Osmotolerant Rhizobacteria in Rice Growth under Different Drought Conditions. Aust. J. Agric. Res. 2005, 56, 715–721. [Google Scholar] [CrossRef]
- Khanam, N.; Khoo, C.; Khan, A.G. Effects of Cytokinin/Auxin Combinations on Organogenesis, Shoot Regeneration and Tropane Alkaloid Production in Duboisia myoporoides. Plant Cell Tissue Organ Cult. 2000, 62, 125–133. [Google Scholar] [CrossRef]
- Lin, G.D.; Griffin, W.J. Scopolamine Content of a Duboisia Hybrid in Callus Cultures. Phytochemistry 1992, 31, 4151–4153. [Google Scholar] [CrossRef]
- Saito, K.; Yamazaki, M.; Kawaguchi, A.; Murakoshi, I. Metabolism of Solanaceous Alkaloids in Transgenic Plant Teratomas Integrated with Genetically Engineered Genes. Tetrahedron 1991, 47, 5955–5968. [Google Scholar] [CrossRef]
- Koo, Y.M.; Heo, A.Y.; Choi, H.W. Salicylic Acid as a Safe Plant Protector and Growth Regulator. Plant Pathol. J. 2020, 36, 1–10. [Google Scholar] [CrossRef]
- Amdoun, R.; Khelifi, L.; Khelifi-Slaoui, M.; Amroune, S.; Benyoussef, E.H.; Thi, D.V.; Assaf-Ducrocq, C.; Gontier, E. Influence of Minerals and Elicitation on Datura stramonium L. Tropane Alkaloid Production: Modelization of the in Vitro Biochemical Response. Plant Sci. 2009, 177, 81–87. [Google Scholar] [CrossRef]
- Yang, C.Q.; Fang, X.; Wu, X.M.; Mao, Y.B.; Wang, L.J.; Chen, X.Y. Transcriptional Regulation of Plant Secondary MetabolismF. J. Integr. Plant Biol. 2012, 54, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Madani, H.; Escrich, A.; Hosseini, B.; Sanchez-Muñoz, R.; Khojasteh, A.; Palazon, J. Effect of Polyploidy Induction on Natural Metabolite Production in Medicinal Plants. Biomolecules 2021, 11, 899. [Google Scholar] [CrossRef] [PubMed]
- Murthy, H.N.; Lee, E.J.; Paek, K.Y. Production of Secondary Metabolites from Cell and Organ Cultures: Strategies and Approaches for Biomass Improvement and Metabolite Accumulation. Plant Cell Tissue Organ Cult. 2014, 118, 1–16. [Google Scholar] [CrossRef]
- Belabbassi, O.; Khelifi-Slaoui, M.; Zaoui, D.; Benyammi, R.; Khalfallah, N.; Malik, S.; Makhzoum, A.; Khelifi, L. Synergistic Effects of Polyploidization and Elicitation on Biomass and Hyoscyamine Content in Hairy Roots of Datura stramonium. Biotechnol. Agron. Soc. Environ. 2016, 20, 408–416. [Google Scholar] [CrossRef]
- Boitel-Conti, M.; Laberche, J.C.; Lanoue, A.; Ducrocq, C.; Sangwan-Norreel, B.S. Influence of Feeding Precursors on Tropane Alkaloid Production during an Abiotic Stress in Datura innoxia Transformed Roots. Plant Cell Tissue Organ Cult. 2000, 60, 131–137. [Google Scholar] [CrossRef]
- Gao, S.L.; Zhu, D.N.; Cai, Z.H.; Xu, D.R. Autotetraploid Plants from Colchicine-Treated Bud Culture of Salvia miltiorrhiza Bge. Plant Cell Tissue Organ Cult. 1996, 47, 73–77. [Google Scholar] [CrossRef]
- Komaraiah, P.; Ramakrishna, S.V.; Reddanna, P.; Kavi Kishor, P.B. Enhanced Production of Plumbagin in Immobilized Cells of Plumbago rosea by Elicitation and in Situ Adsorption. J. Biotechnol. 2003, 101, 181–187. [Google Scholar] [CrossRef]
- Miao, G.P.; Zhu, C.S.; Yang, Y.Q.; Feng, M.X.; Ma, Z.Q.; Feng, J.T.; Zhang, X. Elicitation and in Situ Adsorption Enhanced Secondary Metabolites Production of Tripterygium wilfordii Hook. f. Adventitious Root Fragment Liquid Cultures in Shake Flask and a Modified Bubble Column Bioreactor. Bioproc. Biosyst. Eng. 2014, 37, 641–650. [Google Scholar] [CrossRef]
- Kim, D.J.; Chang, H.N. Increased Shikonin Production in Lithospermum erythrorhizon Suspension Cultures within Situ Extraction and Fungal Cell Treatment (Elicitor). Biotechnol. Lett. 1990, 12, 443–446. [Google Scholar] [CrossRef]

| Elicitor | Host Plant | Elicitation Tissue | Effect | Reference | ||
|---|---|---|---|---|---|---|
| Hyoscyamine | Scopolamine | |||||
| Sugar, protein, and their precursor and derivative | Chitosan | Brugmansia candida | Hairy roots |  |  | [26] |
| Atropa belladonna |  |  | [27,28] | |||
| Hyoscyamus niger |  |  | [29] | |||
| Oligogalacturonides | Datura stramonium |  |  | [32] | ||
| Sucrose | Hyoscyamus muticus | Complex | [37] | |||
| Psk-α | Atropa belladonna |  |  | [40] | ||
| Casein hydrolysate | Hyoscyamus niger |  |  | [29] | ||
| Pectinase | Brugmansia candida |  |  | [43] | ||
| Hemicellulase | Brugmansia candida |  |  | [44] | ||
| YE | YE | Atropa belladonna |  |  | [49] | |
| Brugmansia candida |  |  | [51] | |||
| Hyoscyamus niger |  |  | [29] | |||
| Datura metel |  |  | [59] | |||
| Fungi | Aspergillus niger | Datura metel |  |  | [59] | |
| Alternaria sp. |  |  | [59] | |||
| Fusarium monoliforme |  |  | [59] | |||
| Hormonema ssp. | Brugmansia candida | Complex | Complex | [43] | ||
| Bacteria | Bacillus cereus KCTC 1012 | Scopolia parviflora |  | [61] | ||
| Staphylococcus Aureus KCTC 1916 and 1750 |  | [61] | ||||
| Pseudomonas putida PP01 | Datura stramonium, Datura tatula, and Datura innoxia |  |  | [62] | ||
| Pseudomonas fluorescens P64, P66, and C7R12 |  |  | [62] | |||
| Staphylococus aureuse | Scopolia parviflor |  |  | [66] | ||
| Bacillus cereus | Datura metel |  | [67] | |||
| Staphylococcus aureus |  | [67] | ||||
| Virus | PMMoV, TMV, and ToMV | Datura stramonium | Plants |  | [65] | |
| Fungi | Phytophthora megasperma | Datura stramonium | Cell cultures |  |  | [58] |
| Elicitor | Host Plant | Elicitation Tissue | Effect | Reference | ||
|---|---|---|---|---|---|---|
| Hyoscyamine | Scopolamine | |||||
| Physical elicitor | Acetic acid | Brugmansia candida | Hairy roots |  |  | [26] |
| Citric acid |  |  | [26] | |||
| Oxygen | Duboisia myoporoides |  |  | [72,124] | ||
| Light | Hyoscyamus albus |  | [74] | |||
| Datura innoxia |  | [125] | ||||
| Atropa belladonna |  | [22] | ||||
| WDS | Hyoscyamus muticus |  | [76] | |||
| Atropa belladonna |  |  | [75] | |||
| UV-B | Anisodus luridus |  |  | [78] | ||
| Hyoscyamus reticulatus |  |  | [126] | |||
| Chemical elicitor | Ca2+ | Datura innoxia | Cell cultures |  |  | [82] |
| Brugmansia candida | Hairy roots |  |  | [44] | ||
| Datura stramonium | Complex | [84,85] | ||||
| Al | Datura innoxia | Plants |  |  | [86] | |
| CdCl2 | Atropa belladonna and Datura stramonium | Hairy roots |  |  | [27] | |
| CuCl2 |  |  | [88] | |||
| CdCl2 | Brugmansia candida |  |  | [51] | ||
| Ag+ | Anisodus acutangulus | Complex | Complex | [89] | ||
| Cr | Atropa belladonna | Plants |  |  | [90] | |
| Plant hormone and growth regulator | Methyl jasmonate (MeJA) | Anisodus acutangulus | Hairy roots |  |  | [89] |
| Atropa belladonna |  |  | [93] | |||
| Scopolia parviflora |  |  | [94] | |||
| Hyoscyamus niger |  |  | [53,95] | |||
| Datura stramonium |  |  | [32,97] | |||
| JA | Hyoscyamus muticus |  |  | [96] | ||
| SA | Scopolia parviflora |  | [94] | |||
| Brugmansia candida |  |  | [99] | |||
| Anisodus acutangulus |  |  | [89] | |||
| Atropa belladonna | Complex | Complex | [101] | |||
| Atropa baetica |  | [127] | ||||
| Acetylsalicylic acid (ASA) | Anisodus luridus |  |  | [78] | ||
| Atropa baetica |  | [127] | ||||
| SA | Datura stramnium, Datura tatula and Datura innoxia |  | [100] | |||
| ASA |  | [100] | ||||
| Indole butyric acid (IBA) | Hyoscyamus niger |  |  | [103] | ||
| Naphtalic acetic acid (NAA) | Hyoscyamus niger | Cell cultures |  | [104] | ||
| NAA | Datura stramonlum | Hairy roots |  |  | [105] | |
| 2,4-Dichlorophenoxyacetic acid (2,4-D) |  |  | [105] | |||
| Auxin (IAA) | Hyoscyamus muticus |  |  | [102] | ||
| NAA |  |  | [102] | |||
| Gibberellin A7 (GA7) | Brugmansia candida |  |  | [107] | ||
| GA3 | Hyoscyamus muticus |  | [102] | |||
| Abscisic acid (ABA) |  | [102] | ||||
| ABA | Datura stramonlum |  |  | [105] | ||
| Anisodus acutangulus | Plants | Complex | Complex | [108] | ||
| Glyphosate | Datura stramonium | Seedings |  |  | [110] | |
| Coronarin (COR) | Atropa acuminata | Hairy roots | Complex | Complex | [113] | |
| New type | Iron oxide nanoparticles | Hyoscyamus reticulatus |  |  | [118] | |
| Atropa belladonna |  | [119] | ||||
| Zinc oxide nanoparticles | Hyoscyamus reticulatus |  |  | [115] | ||
| Silicon dioxide nanoparticles |  |  | [120] | |||
| Titanium dioxide nanoparticles | Hyoscyamus niger | Plants |  |  | [128] | |
| NO | Hyoscyamus reticulatus | Hairy roots |  |  | [123] | |
| Combined Elicitation | Host Plant | Elicitation Tissue | Effect | Reference | |
|---|---|---|---|---|---|
| Hyoscyamine | Scopolamine | ||||
| Ca2+ + JA | Datura stramonium | Hairy roots |  | [137] | |
| IAA + SA | Datura metel |  | [59] | ||
| NAA + benzyladenine (BA) | Duboisia myoporoides | Rootless shoots |  |  | [133] |
| IBA + BA |  |  | [133] | ||
| Methyl-β-cyclodextrin (β-CD) + COR | Atropa acuminata | Hairy roots |  |  | [113] |
| β-CD + COR | Atropa belladonna |  |  | [113] | |
| Plant growth promoting rhizobacteria (PGPR) + WDS | Hyoscyamus niger |  |  | [131] | |
| Elicitation + Other Strategy | Host Plant | Elicitation Tissue | Effect | Reference | |
|---|---|---|---|---|---|
| Hyoscyamine | Scopolamine | ||||
| MeJA/ASA + Overexpression H6H | Atropa baetica | Hairy roots |  | [127] | |
| SA + Overexpression H6H | Atropa baetica |  | [127] | ||
| MeJA + Overexpression PMT | Hyoscyamus niger |  | [95] | ||
| SA/ASA + Tetraploidy | Datura stramonium |  | [141] | ||
| Tween 20 + L-phenylalanine/DL-β-phenyllactic acid | Datura innoxia |  | [142] | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wen, Y.; Liao, Y.; Tang, Y.; Zhang, H.; Zhang, J.; Liao, Z. Metabolic Effects of Elicitors on the Biosynthesis of Tropane Alkaloids in Medicinal Plants. Plants 2023, 12, 3050. https://doi.org/10.3390/plants12173050
Wen Y, Liao Y, Tang Y, Zhang H, Zhang J, Liao Z. Metabolic Effects of Elicitors on the Biosynthesis of Tropane Alkaloids in Medicinal Plants. Plants. 2023; 12(17):3050. https://doi.org/10.3390/plants12173050
Chicago/Turabian StyleWen, Yuru, Yiran Liao, Yueli Tang, Hongbo Zhang, Jiahui Zhang, and Zhihua Liao. 2023. "Metabolic Effects of Elicitors on the Biosynthesis of Tropane Alkaloids in Medicinal Plants" Plants 12, no. 17: 3050. https://doi.org/10.3390/plants12173050
APA StyleWen, Y., Liao, Y., Tang, Y., Zhang, H., Zhang, J., & Liao, Z. (2023). Metabolic Effects of Elicitors on the Biosynthesis of Tropane Alkaloids in Medicinal Plants. Plants, 12(17), 3050. https://doi.org/10.3390/plants12173050









