Application of Silicon, Zinc, and Zeolite Nanoparticles—A Tool to Enhance Drought Stress Tolerance in Coriander Plants for Better Growth Performance and Productivity
Abstract
1. Introduction
2. Results
2.1. Vegetative Growth Parameters of Coriander Plants
2.2. Leaf Chlorophyll Content, Photosynthetic Machinery, Photosystem II Efficiency, Chlorophyll Fluorescence, and Total Carbohydrate Content
2.3. Antioxidant Compounds Content and Proline
2.4. Phytohormones, Malondialdehyde, and Catalase
2.5. Macro and Micronutrient Content
2.6. Essential Oil, Fruit Yield, and Its Components in Coriander Plants
2.7. Composition of Essential Oil
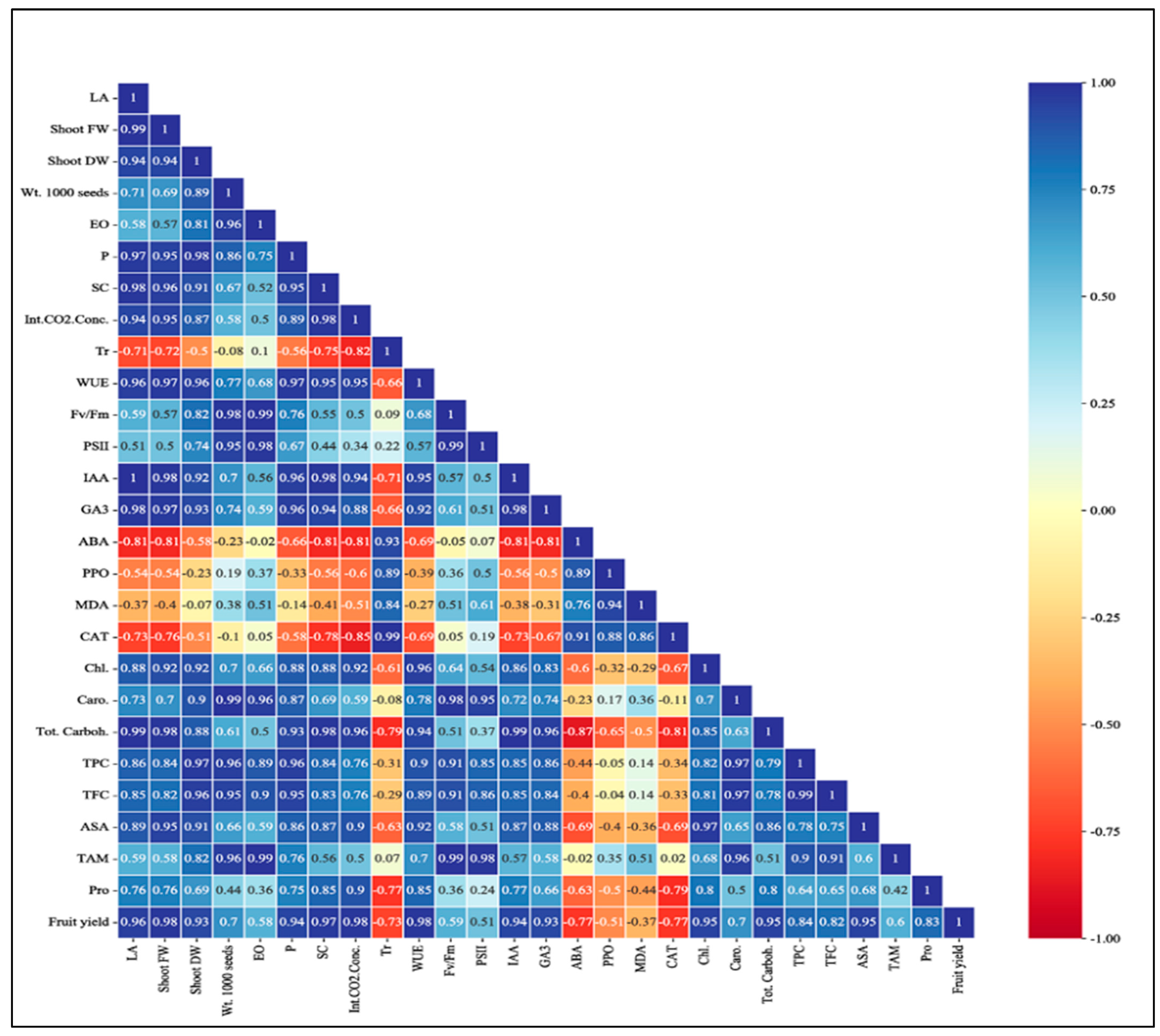
2.8. Correlation Study
3. Discussion
4. Materials and Methods
4.1. Experimental Site, Fertilizers Application, and Plant Materials
4.2. Experimental Design and Treatments Applied
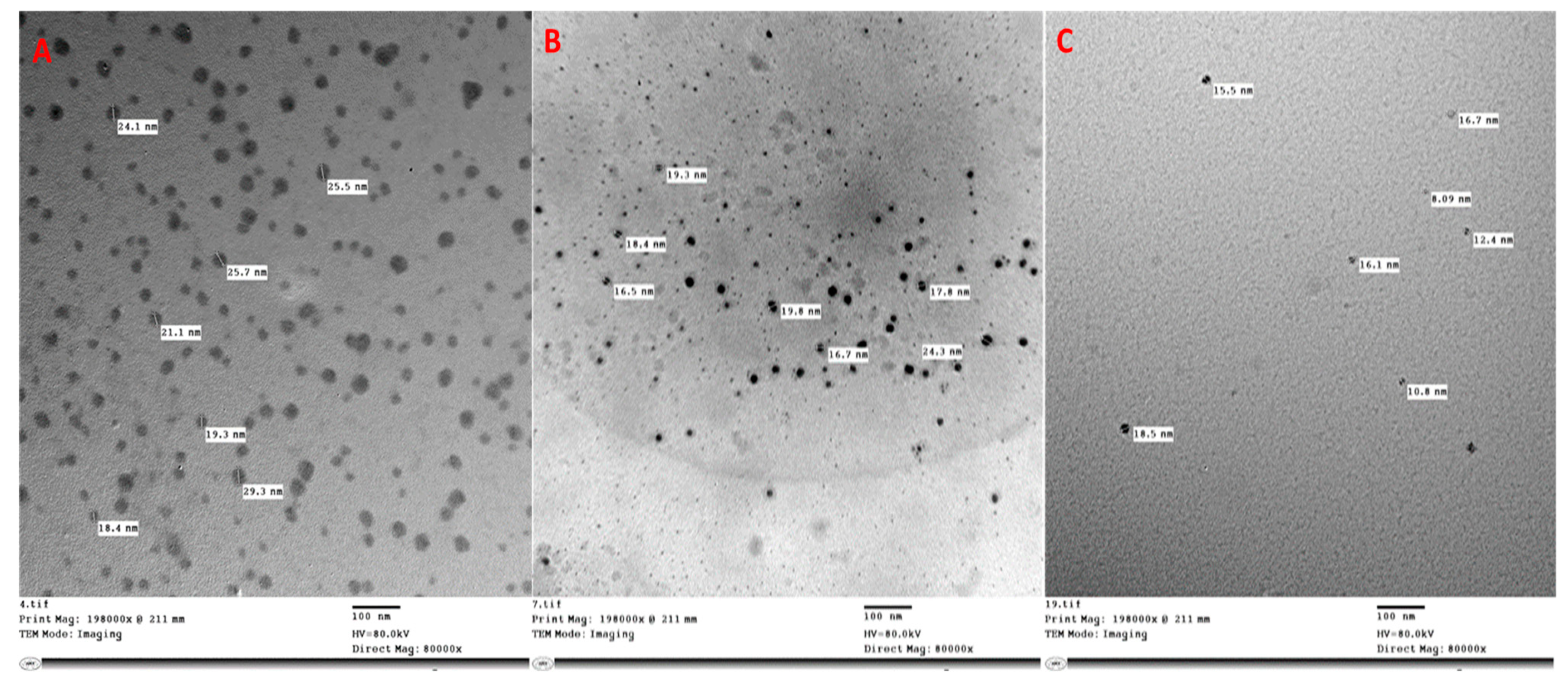
4.3. Synthesis of Zeolite, Zinc, and Silicon Nanoparticles
4.3.1. Zeolite Nanoparticles
4.3.2. Zinc and Silicon Nanoparticles
4.4. Data Recorded
4.4.1. Morphological Characteristics and Yield Components
4.4.2. Leaf Chlorophyll Content, Photosynthetic Apparatus, and Chlorophyll Fluorescence Parameters
Leaf Chlorophyll Content and Photosynthetic Apparatus
Measurement of Chlorophyll Fluorescence Parameters
4.4.3. Total Carbohydrate Content
4.4.4. Phytohormones, Malondialdehyde, and Catalase
4.4.5. Antioxidant Compounds Content and Proline
4.4.6. Plant Nutrient Content
4.4.7. Essential Oil Content
4.4.8. Gas Chromatography-Mass Spectrometry
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Ahmadalipour, A.; Moradkhani, H.; Castelletti, A.; Magliocca, N. Future Drought Risk in Africa: Integrating Vulnerability, Climate Change, and Population Growth. Sci. Total Environ. 2019, 662, 672–686. [Google Scholar] [CrossRef]
- Zhang, C.; Dong, J.; Ge, Q. Mapping 20 Years of Irrigated Croplands in China Using MODIS and Statistics and Existing Irrigation Products. Sci. Data 2022, 9, 407. [Google Scholar] [CrossRef]
- Abdelkhalik, A.; Pascual-Seva, N.; Nájera, I.; Giner, A.; Baixauli, C.; Pascual, B. Yield Response of Seedless Watermelon to Different Drip Irrigation Strategies under Mediterranean Conditions. Agric. Water Manag. 2019, 212, 99–110. [Google Scholar] [CrossRef]
- Besser, H.; Hamed, Y. Environmental Impacts of Land Management on the Sustainability of Natural Resources in Oriental Erg Tunisia, North Africa. Environ. Dev. Sustain. 2021, 23, 11677–11705. [Google Scholar] [CrossRef]
- Abuarab, M.E.; Hafez, S.M.; Shahein, M.M.; Hassan, A.M.; El-Sawy, M.B.; El-Mogy, M.M.; Abdeldaym, E.A. Irrigation scheduling for green beans grown in clay loam soil under a drip irrigation system. Water SA 2020, 46, 573–582. [Google Scholar]
- Toscano, S.; Franzoni, G.; Álvarez, S. Drought Stress in Horticultural Plants. Horticulturae 2023, 9, 7. [Google Scholar] [CrossRef]
- Abdelaziz, M.E.; Atia, M.A.M.; Abdelsattar, M.; Abdelaziz, S.M.; Ibrahim, T.A.A.; Abdeldaym, E.A. Unravelling the Role of Piriformospora indica in Combating Water Deficiency by Modulating Physiological Performance and Chlorophyll Metabolism-Related Genes in Cucumis sativus. Horticulturae 2021, 7, 399. [Google Scholar] [CrossRef]
- Shehata, S.A.; Omar, H.S.; Elfaidy, A.G.; El-Sayed, S.S.; Abuarab, M.E.; Abdeldaym, E.A. Grafting enhances drought tolerance by regulating stress-responsive gene expression and antioxidant enzyme activities in cucumbers. BMC Plant Biol. 2022, 22, 408. [Google Scholar] [CrossRef] [PubMed]
- El-Mogy, M.M.; Atia, M.A.M.; Dhawi, F.; Fouad, A.S.; Bendary, E.S.A.; Khojah, E.; Samra, B.N.; Abdelgawad, K.F.; Ibrahim, M.F.M.; Abdeldaym, E.A. Towards Better Grafting: SCoT and CDDP Analyses for Prediction of the Tomato Rootstocks Performance under Drought Stress. Agronomy 2022, 12, 153. [Google Scholar] [CrossRef]
- Khan, M.B.; Farooq, M.; Hussain, M.; Shanawaz; Shabir, G. Foliar application of micronutrients improves the wheat yield and net economic return. Int. J. Agric. Biol. 2010, 12, 953–956. [Google Scholar]
- EL-Bauome, H.A.; Abdeldaym, E.A.; Abd El-Hady, M.A.M.; Darwish, D.B.E.; Alsubeie, M.S.; El-Mogy, M.M.; Basahi, M.A.; Al-Qahtani, S.M.; Al-Harbi, N.A.; Alzuaibr, F.M.; et al. Exogenous Proline, Methionine, and Melatonin Stimulate Growth, Quality, and Drought Tolerance in Cauliflower Plants. Agriculture 2022, 12, 1301. [Google Scholar] [CrossRef]
- Ahmadian, K.; Jalilian, J.; Pirzad, A. Nano-fertilizers improved drought tolerance in wheat under deficit irrigation. Agric. Water Manag. 2021, 244, 106544. [Google Scholar] [CrossRef]
- Shah, M.N.; Wright, D.L.; Hussain, S.; Koutroubas, S.D.; Seepaul, R.; George, S.; Ali, S.; Naveed, M.; Khan, M.; Altaf, M.T.; et al. Organic fertilizer sources improve the yield and quality attributes of maize (Zea mays L.) hybrids by improving soil properties and nutrient uptake under drought stress. J. King Saud Univ.-Sci. 2023, 35, 102570. [Google Scholar] [CrossRef]
- Haider, I.; Raza, M.A.S.; Iqbal, R.; Aslam, M.U.; Habib-ur-Rahman, M.; Raja, S.; Khan, M.T.; Aslam, M.M.; Waqas, M.; Ahmad, S. Potential effects of biochar application on mitigating the drought stress implications on wheat (Triticum aestivum L.) under various growth stages. J. Saudi Chem. Soc. 2020, 24, 974–981. [Google Scholar] [CrossRef]
- Mphande, W.; Kettlewell, P.S.; Grove, I.G.; Farrell, A.D. The potential of anti-transpirants in drought management of arable crops: A review. Agric. Water Manag. 2020, 236, 106143. [Google Scholar] [CrossRef]
- Xu, Y.; Gao, Y.; Li, W.; Chen, S.; Li, Y.; Shi, Y. Effects of compound water retention agent on soil nutrients and soil microbial diversity of winter wheat in saline-alkali land. Chem. Biol. Technol. Agric. 2023, 10, 2. [Google Scholar] [CrossRef]
- Mahmoud, A.W.M.; Abdeldaym, E.A.; Abdelaziz, S.M.; El-Sawy, M.B.I.; Mottaleb, S.A. Synergetic Effects of Zinc, Boron, Silicon, and Zeolite Nanoparticles on Confer Tolerance in Potato Plants Subjected to Salinity. Agronomy 2020, 10, 19. [Google Scholar] [CrossRef]
- Mahmoud, A.W.M.; Samy, M.M.; Sany, H.; Eid, R.R.; Rashad, H.M.; Abdeldaym, E.A. Nanopotassium, Nanosilicon, and Biochar Applications Improve Potato Salt Tolerance by Modulating Photosynthesis, Water Status, and Biochemical Constituents. Sustainability 2022, 14, 723. [Google Scholar] [CrossRef]
- Karimian, Z.; Samiei, L. ZnO nanoparticles efficiently enhance drought tolerance in Dracocephalum kotschyi through altering physiological, biochemical and elemental contents. Front. Plant Sci. 2023, 14, 1063618. [Google Scholar] [CrossRef]
- Jan, A.U.; Hadi, F.; Ditta, A.; Suleman, M.; Ullah, M. Zinc-induced anti-oxidative defense and osmotic adjustments to enhance drought stress tolerance in sunflower (Helianthus annuus L.). Environ. Exp. Bot. 2022, 193, 104682. [Google Scholar] [CrossRef]
- Ahmad, M.; El-Saeid, M.H.; Akram, M.A.; Ahmad, H.R.; Haroon, H.; Hussain, A. Silicon fertilization—A tool to boost up drought tolerance in wheat (Triticum aestivum L.) crop for better yield. J. Plant Nutr. 2016, 39, 1283–1291. [Google Scholar] [CrossRef]
- Wang, M.; Wang, R.; Mur, L.A.J.; Ruan, J.; Shen, Q.; Guo, S. Functions of silicon in plant drought stress responses. Hortic. Res. 2021, 8, 254. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hakim, S.G.; Shehata, A.S.A.; Moghannem, S.A.; Qadri, M.; El-Ghany, M.F.A.; Abdeldaym, E.A.; Darwish, O.S. Nanoparticulate Fertilizers Increase Nutrient Absorption Efficiency and Agro-Physiological Properties of Lettuce Plant. Agronomy 2023, 13, 691. [Google Scholar] [CrossRef]
- Taran, N.; Storozhenko, V.; Svietlova, N.; Batsmanova, L.; Shvartau, V.; Kovalenko, M. Effect of Zinc and Copper Nanoparticles on Drought Resistance of Wheat Seedlings. Nanoscale Res. Lett. 2017, 12, 60. [Google Scholar] [CrossRef]
- Kandhol, N.; Jain, M.; Tripathi, D.K. Nanoparticles as potential hallmarks of drought stress tolerance in plants. Physiol. Plant. 2022, 174, e13665. [Google Scholar] [CrossRef]
- Weisany, W.; Khosropour, E. Engineered nanomaterials in crop plants drought stress management. In Engineered Nanomaterials for Sustainable Agricultural Production, Soil Improvement and Stress Management; Academic Press: Cambridge, MA, USA, 2023; pp. 183–204. [Google Scholar] [CrossRef]
- Bahador, M.; Tadayon, M.R. Investigating of zeolite role in modifying the effect of drought stress in hemp: Antioxidant enzymes and oil content. Ind. Crops Prod. 2020, 144, 112042. [Google Scholar] [CrossRef]
- Mohammadifard, F.; Tarakemeh, A.; Moghaddam, M.; Zim, M. Bentonite Mitigates the Adverse Effects of Drought Stress in Fenugreek (Trigonella foenum-graecum L.). J. Soil Sci. Plant Nutr. 2022, 22, 1098–1111. [Google Scholar] [CrossRef]
- Al-Shammari, A.M.A.; Abood, M.A.; Hamdi, G.J. Perlite affects some plant indicators and reduces water deficit in tomato. Int. J. Veg. Sci. 2018, 24, 490–500. [Google Scholar] [CrossRef]
- Hazrati, S.; Tahmasebi-Sarvestani, Z.; Mokhtassi-Bidgoli, A.; Modarres-Sanavy, S.A.M.; Mohammadi, H.; Nicola, S. Effects of zeolite and water stress on growth, yield and chemical compositions of Aloe vera L. Agric. Water Manag. 2017, 181, 66–72. [Google Scholar] [CrossRef]
- Hazrati, S.; Khurizadeh, S.; Sadeghi, A.R. Application of zeolite improves water and nitrogen use efficiency while increasing essential oil yield and quality of Salvia officinalis under water-deficit stress. Saudi J. Biol. Sci. 2022, 29, 1707–1716. [Google Scholar] [CrossRef]
- Ghamarnia, H.; Daichin, S. Effect of different water stress regimes on different coriander (Coriander sativum L.) parameters in a semi-arid climate. Int. J. Agron. Plant Prod. 2013, 4, 822–832. [Google Scholar]
- Amirahmadi, E.; Ghorbani, M.; Moudrý, J. Effects of Zeolite on Aggregation, Nutrient Availability, and Growth Characteristics of Corn (Zea mays L.) in Cadmium-Contaminated Soils. Water Air Soil Pollut. 2022, 233, 436. [Google Scholar] [CrossRef]
- Chaudhry, S.; Sidhu, G.P.S. Climate Change Regulated Abiotic Stress Mechanisms in Plants: A Comprehensive Review. Plant Cell Rep. 2021, 41, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Stadnik, B.; Tobiasz-Salach, R.; Mazurek, M. Effect of Silicon on Oat Salinity Tolerance: Analysis of the Epigenetic and Physiological Response of Plants. Agriculture 2023, 13, 81. [Google Scholar] [CrossRef]
- Maghsoudi, K.; Emam, Y.; Ashraf, M. Influence of foliar application of silicon on chlorophyll fluorescence, photosynthetic pigments, and growth in water-stressed wheat cultivars differing in drought tolerance. Turk. J. Bot. 2015, 39, 625–634. [Google Scholar] [CrossRef]
- Singh, A.K.; Dubey, R.S. Changes in chlorophyll a and b contents and activities of photosystems 1 and 2 in rice seedlings induced by NaCl. Photosynthetica 1995, 31, 489–499. [Google Scholar]
- Baca Cabrera, J.C.; Hirl, R.T.; Schäufele, R.; Macdonald, A.; Schnyder, H. Stomatal Conductance Limited the CO2 Response of Grassland in the Last Century. BMC Biol. 2021, 19, 50. [Google Scholar] [CrossRef]
- Franco-Navarro, J.D.; Rosales, M.A.; Cubero-Font, P.; Calvo, P.; Alvarez, R.; Diaz-Espejo, A.; Colmenero-Flores, J.M. Chloride as a Macronutrient Increases Water-use Efficiency by Anatomically Driven Reduced Stomatal Conductance and Increased Mesophyll Diffusion to CO2. Plant J. 2019, 99, 815–831. [Google Scholar]
- Anik, T.R.; Mostofa, M.G.; Rahman, M.M.; Khan, M.A.R.; Ghosh, P.K.; Sultana, S.; Das, A.K.; Hossain, M.S.; Keya, S.S.; Rahman, M.A.; et al. Zn Supplementation Mitigates Drought Effects on Cotton by Improving Photosynthetic Performance and Antioxidant Defense Mechanisms. Antioxidants 2023, 12, 854. [Google Scholar] [CrossRef]
- Pavia, I.; Roque, J.; Rocha, L.; Ferreira, H.; Castro, C.; Carvalho, A.; Silva, E.; Brito, C.; Goncalves, A.; Lima-Brito, J.; et al. Zinc priming and foliar application enhances photoprotection mechanisms in drought-stressed wheat plants during anthesis. Plant Physiol. Biochem. 2019, 140, 27–42. [Google Scholar] [CrossRef]
- Irfan, M.; Maqsood, M.A.; Rehman, H.U.; Mahboob, W.; Sarwar, N.; Hafeez, O.B.A.; Hussain, S.; Ercisli, S.; Akhtar, M.; Aziz, T. Silicon Nutrition in Plants under Water-Deficit Conditions: Overview and Prospects. Water 2023, 15, 739. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, S.H.I.; Gong, H.J.; Zhao, H.L.; Li, H.L.; Hu, Y.H.; Wang, Y.C. Beneficial effects of silicon on photosynthesis of tomato seedlings under water stress. J. Integr. Agric. 2018, 17, 2151–2159. [Google Scholar] [CrossRef]
- Shahsavari, N. Effects of zeolite and zinc on quality of canola (Brassica napus L.) under late season drought stress. Commun. Soil Sci. Plant Anal. 2019, 50, 1117–1122. [Google Scholar] [CrossRef]
- Guidi, L.; Lo Piccolo, E.; Landi, M. Chlorophyll Fluorescence, Photoinhibition and Abiotic Stress: Does It Make Any Difference the Fact to Be a C3 or C4 Species? Front. Plant Sci. 2019, 10, 174. [Google Scholar] [CrossRef]
- Ghorbani, M.; Amirahmadi, E.; Konvalina, P.; Moudrý, J.; Bárta, J.; Kopecký, M.; Teodorescu, R.I.; Bucur, R.D. Comparative Influence of Biochar and Zeolite on Soil Hydrological Indices and Growth Characteristics of Corn (Zea mays L.). Water 2022, 14, 3506. [Google Scholar] [CrossRef]
- Ulloa, M.; Nunes-Nesi, A.; da Fonseca-Pereira, P.; Poblete-Grant, P.; Reyes-Díaz, M.; Cartes, P. The effect of silicon supply on photosynthesis and carbohydrate metabolism in two wheat (Triticum aestivum L.) cultivars contrasting in response to phosphorus nutrition. Plant Physiol. Biochem. 2021, 169, 236–248. [Google Scholar] [CrossRef]
- Sun, L.; Song, F.; Zhu, X.; Liu, S.; Liu, F.; Wang, Y.; Li, X. Nano-ZnO alleviates drought stress via modulating the plant water use and carbohydrate metabolism in maize. Arch. Agron. Soil Sci. 2021, 67, 245–259. [Google Scholar] [CrossRef]
- Othman, E.Z.; El-Attar, A.B.; El-Bahbohy, R.M.; Abd El-Khalek, S.N.; Morgan, S.H.; Mahmoud, A.W.M. Exogenous Appliance of Nano-Zeolite and Nano-Silicon Elevate Solidago canadensis Invasive Plant Tolerance to Water Deficiency. Horticulturae 2023, 9, 172. [Google Scholar] [CrossRef]
- Umair Hassan, M.; Aamer, M.; Umer Chattha, M.; Haiying, T.; Shahzad, B.; Barbanti, L.; Nawaz, M.; Rasheed, A.; Afzal, A.; Liu, Y.; et al. The Critical Role of Zinc in Plants Facing the Drought Stress. Agriculture 2020, 10, 396. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Y.; Hassan, M.J.; Li, Z.; Peng, Y. Indole-3-acetic acid improves drought tolerance of white clover via activating auxin, abscisic acid and jasmonic acid related genes and inhibiting senescence genes. BMC Plant Biol. 2020, 20, 150. [Google Scholar] [CrossRef]
- Zhang, Y.; Luan, Q.; Jiang, J.; Li, Y. Prediction and Utilization of Malondialdehyde in Exotic Pine Under Drought Stress Using Near-Infrared Spectroscopy. Front. Plant Sci. 2021, 12, 735275. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Bhuyan, M.H.M.; Zulfiqar, F.; Raza, A.; Mohsin, S.M.; Mahmud, J.A.; Fujita, M.; Fotopoulos, V. Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator. Antioxidants 2020, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Lahbouki, S.; Ben-Laouane, R.; Anli, M.; Boutasknit, A.; Ait-Rahou, Y.; Ait-El-Mokhtar, M.; El Gabardi, S.; Douira, A.; Wahbi, S.; Outzourhit, A. Arbuscular Mycorrhizal Fungi and/or Organic Amendment Enhance the Tolerance of Prickly Pear (Opuntia ficusindica) under Drought Stress. J. Arid. Environ. 2022, 199, 104703. [Google Scholar] [CrossRef]
- Altaf, M.A.; Shahid, R.; Ren, M.-X.; Naz, S.; Altaf, M.M.; Khan, L.U.; Tiwari, R.K.; Lal, M.K.; Shahid, M.A.; Kumar, R. Melatonin Improves Drought Stress Tolerance of Tomato by Modulating Plant Growth, Root Architecture, Photosynthesis, and Antioxidant Defense System. Antioxidants 2022, 11, 309. [Google Scholar] [CrossRef]
- Liu, C.; Wang, Y.; Pan, K.; Li, W.; Zhang, L.; Shen, X.; Liu, L.; Deng, M. Responses of the antioxidant defense system to drought stress in the leaves of Fargesia denudata seedlings, the staple food of the giant panda. Russ. J. Plant Physiol. 2019, 3, 374–383. [Google Scholar] [CrossRef]
- Ben Ahmed, C.; Ben Rouina, B.; Sensoy, S.; Boukhris, M.; Ben Abdallah, F. Changes in gas exchange, proline accumulation and antioxidative enzyme activities in three olive cultivars under contrasting water availability regimes. Environ. Exp. Bot. 2009, 67, 345–352. [Google Scholar] [CrossRef]
- Anjum, S.A.; Xie, X.; Wang, L.; Saleem, M.F.; Man, C.; Lei, W. Morphological, physiological and biochemical responses of plants to drought stress. Afr. J. Agric. Res. 2011, 6, 2026–2032. [Google Scholar]
- Afshari, M.; Pazoki, A.; Sadeghipour, O. Biochemical Changes of Coriander (Coriandrum sativum L.) Plants under Drought Stress and Foliar Application of Salicylic Acid and Silicon Nanoparticles. J. Med. Plants By-Product. 2022. Available online: https://journals.areeo.ac.ir/article_126516_364874ebbb6b2b661d174c1c688e4ef7.Pdf (accessed on 27 July 2023).
- Avasiloaiei, D.I.; Calara, M.; Brezeanu, P.M.; Murariu, O.C.; Brezeanu, C. On the Future Perspectives of Some Medicinal Plants within Lamiaceae Botanic Family Regarding Their Comprehensive Properties and Resistance against Biotic and Abiotic Stresses. Genes 2023, 14, 955. [Google Scholar] [CrossRef]
- Talebi, M.; Moghaddam, M.; Pirbalouti, A.G. Methyl jasmonate effects on volatile oil compounds and antioxidant activity of leaf extract of two basil cultivars under salinity stress. Acta Physiolo. Plant 2018, 40, 34–40. [Google Scholar] [CrossRef]
- Mirzaie, M.; Ladanmoghadam, A.R.; Hakimi, L.; Danaee, E. Water stress modifies essential oil yield and composition, glandular trichomes and stomatal features of lemongrass (Cymbopogon citratus) inoculated with arbuscular mycorrhizal fungi. J. Agric. Sci. Technol. 2020, 12, 27–36. [Google Scholar]
- Richards, L.S. Diagnosis and Improvement of Saline and Alkaline Soils; U.S. Department 722 Agriculture Handbook; U.S. Government Printing Office: Washington, DC, USA, 1954; Volume 18, p. 348. [Google Scholar]
- Jackson, M.L. Soil Chemical Analysis; Printice-Hall of India: New Delhi, India, 1973; p. 498. [Google Scholar]
- Page, A.I.; Miller, R.H.; Keeney, T.R. Methods of Soil Analysis Part 2; Amer Society of Agronomy: Madison, WI, USA, 1982; p. 595. [Google Scholar]
- Hassan, A.Z.A.; Mahmoud, A.W.M. Hydrothermal synthesis of nano crystals (AM) zeolite using variable temperature programs. J. Nanomater. 731 Mol. Nanotechnol. 2015, 4, 4. [Google Scholar] [CrossRef]
- Li, J.; Wee, C.; Sohn, L. Effect of ammonium-and potassium-loaded zeolite on kale (Brassica alboglabra) growth and soil property. Am. J. Plant Sci. 2013, 4, 1976. [Google Scholar] [CrossRef]
- Daneshvar, N.; Aber, S.; Sayed, D.; Khataee, M.S.; Rasoulifard, A.R. Preparation and investigation of photocatalytic properties of ZnO nanocrystals: Effect of operational parameters and kinetic study. Int. J. Nucl. Quantum Eng. 2007, 1, 24–29. [Google Scholar]
- Zhu, Y.; Gong, H. Beneficial effects of silicon on salt and drought tolerance in plants. Agron. Sustain. Dev. 2014, 34, 455–472. [Google Scholar] [CrossRef]
- Moran, R. Formulae for determination of chlorophyllous pigments extracted with N, N-dimethylformamide. Plant Physiol. 1982, 69, 1376–1381. [Google Scholar] [CrossRef]
- Helrich, K. Official Methods of Analysis, 15th ed.; Association of Official Agricultural Chemist: Arlington, VA, USA, 1990; Volume 1, p. 673. [Google Scholar]
- Fales, H.M.; Jaouni, T.M. Simple device for preparing ethereal diazomrthane without restoring to Codisitillation. Anal. Chem. 1973, 45, 2302–2303. [Google Scholar] [CrossRef]
- Vogel, A.I. Practical Organic Chemistry, 3rd ed.; Longman Group Limited: London, UK, 1974; pp. 969–971. [Google Scholar]
- Hodges, D.M.; Delong, J.M.; Forney, C.; Prange, R.K. Improving the hiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 1999, 207, 604–611. [Google Scholar] [CrossRef]
- Pereira, G.J.G.; Molina, S.M.G.; Lea, P.J.; Azevedo, R. Activity of antioxidant enzymes in response to cadmium in Crotalaria juncea. Plant Soil. 2002, 239, 123–132. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphmolybdic phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar] [CrossRef]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Katoch, R. Analytical Techniques in Biochemistry and Molecular Biology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls and Carotenoids: Measurement and Characterization by UV-VIS Spectroscopy. Curr. Protoc. Food Anal. Chem. 2001, 1, F4.3.1–F4.3.8. [Google Scholar] [CrossRef]
- Abdallah, I.S.; Atia, M.A.M.; Nasrallah, A.K.; El-Beltagi, H.S.; Kabil, F.F.; El-Mogy, M.M.; Abdeldaym, E.A. Effect of New Pre-Emergence Herbicides on Quality and Yield of Potato and Its Associated Weeds. Sustainability 2021, 13, 9796. [Google Scholar] [CrossRef]
- Bates, L.; Waldren, R.; Teare, I. Rapid determination of free proline for water-stress 758 studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Clevenger, J.F. Apparatus for Volatile Oil Determination, Description of New Type. J. Am. Pharm. Assoc. 1912, 17, 345–349. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy; Allured Business Media: Carol Stream, IL, USA, 1995. [Google Scholar]
- SRplot, Online Platform for Data Analysis and Visualization. Available online: https://www.bioinformatics.com.cn/en?keywords=heatmap (accessed on 28 July 2023).
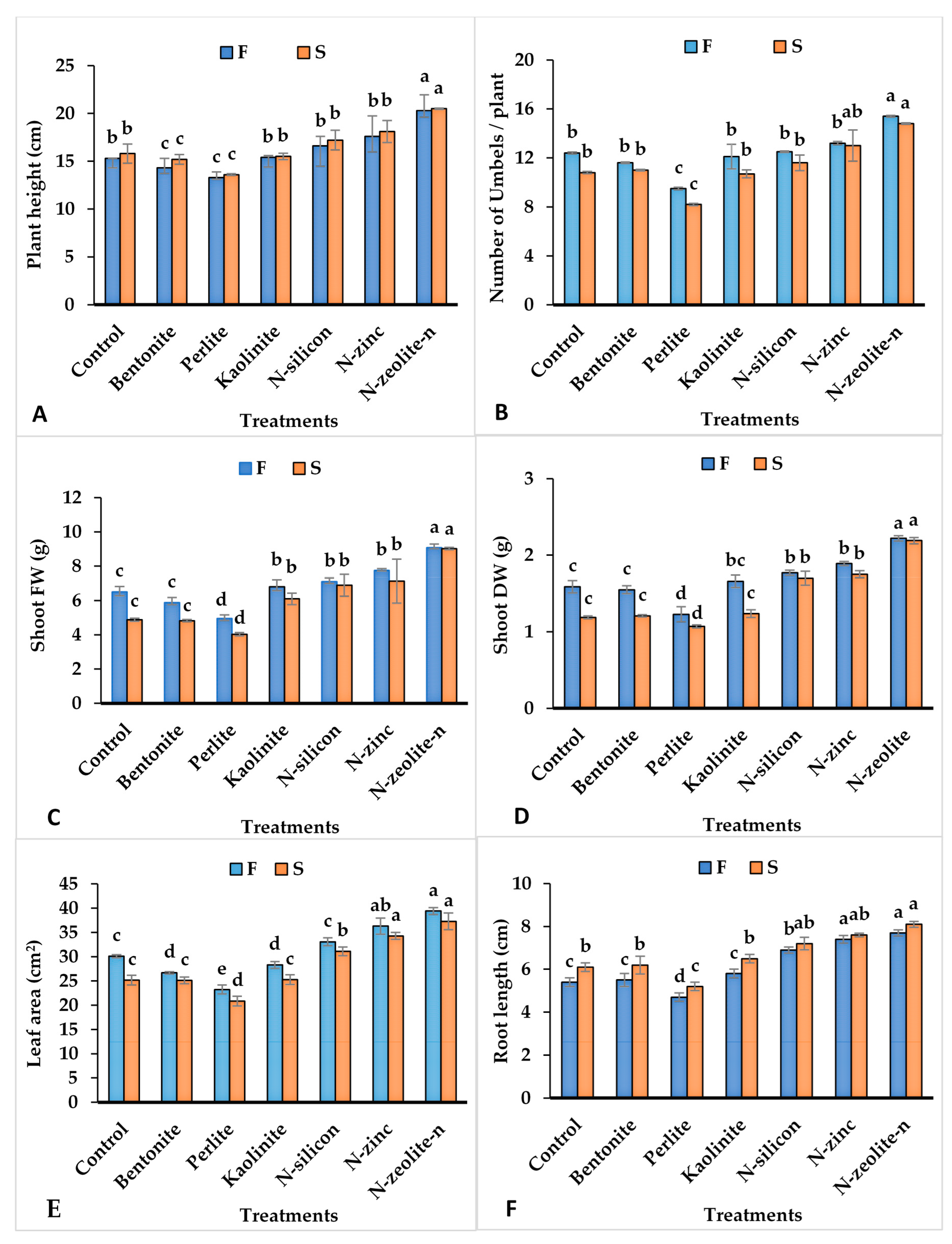
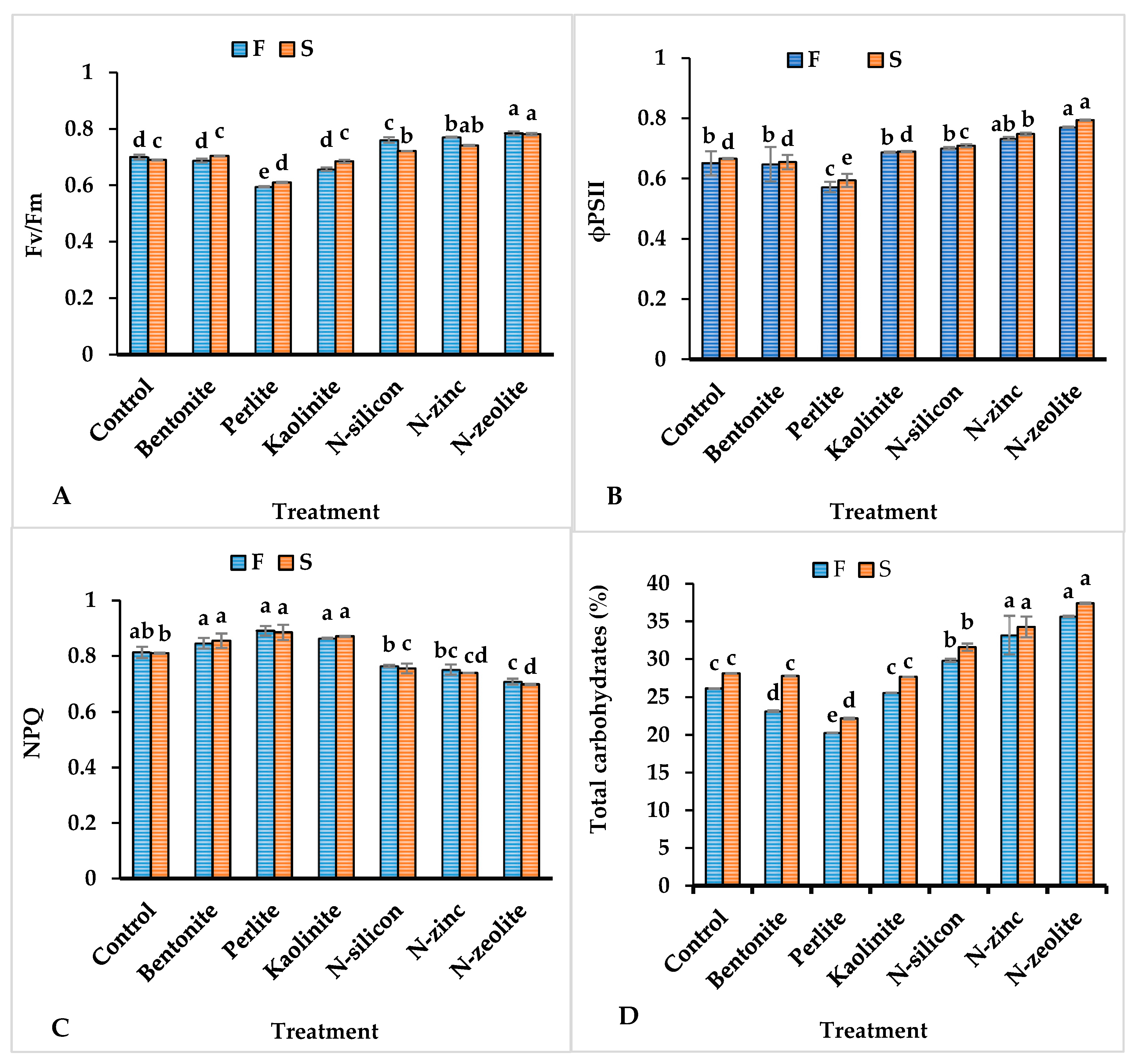

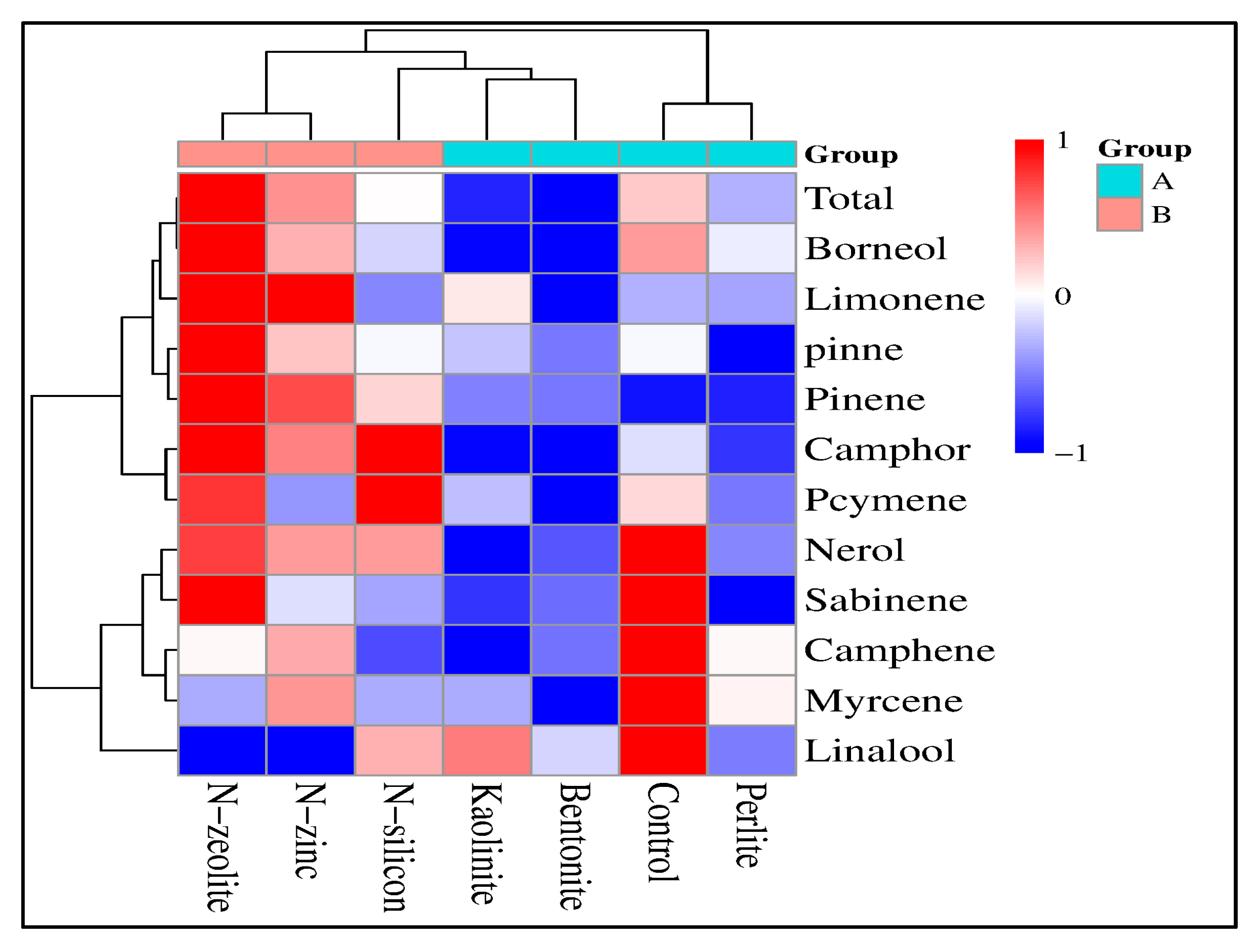
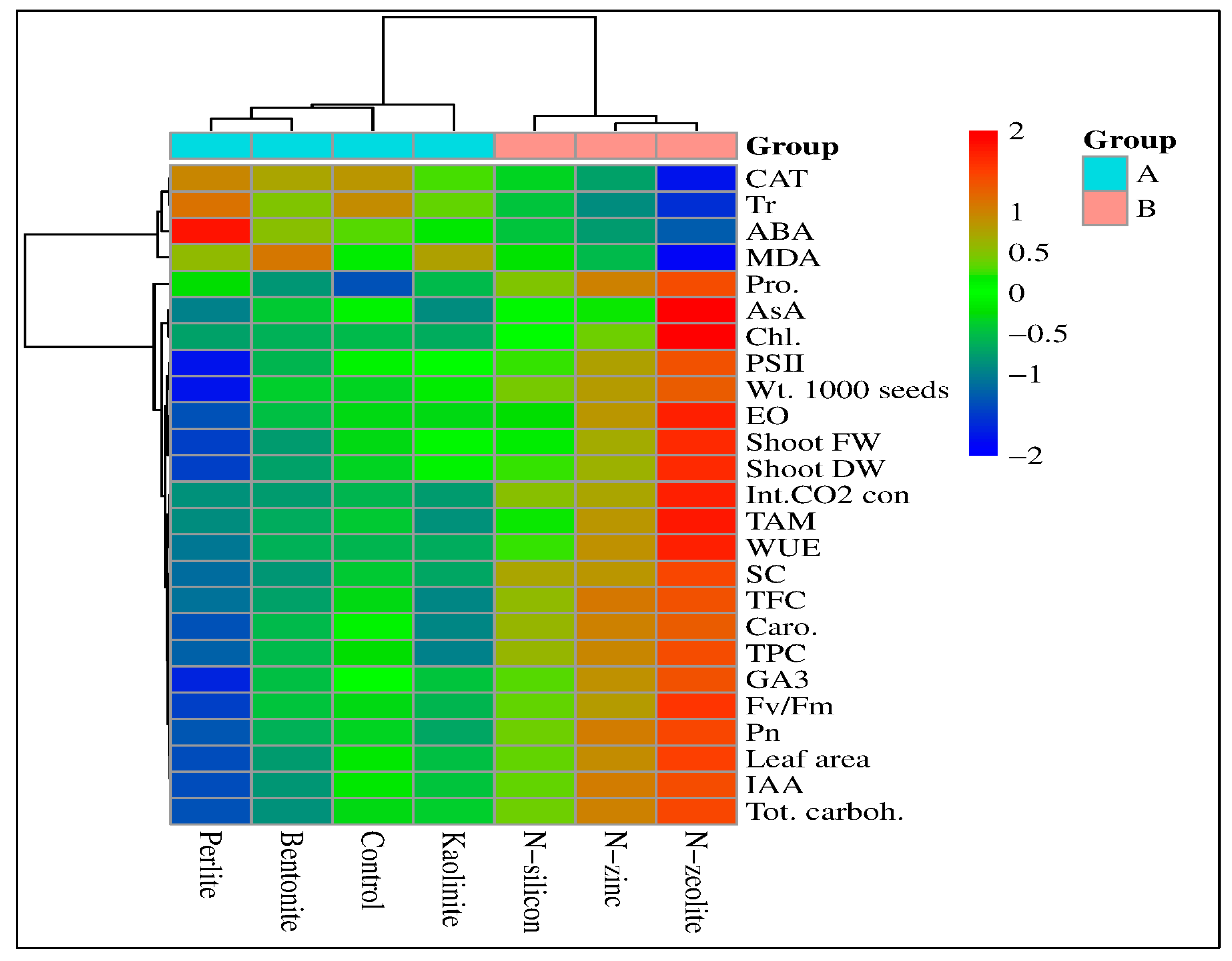
| Treatment | Chlorophyll Content (mg.g−1) | Photosynthetic Rate (µmol m–2s–1) | Stomatal Conductance (mmol m–2s–1) | Intercellular CO2 Concentration (ppm) | Transpiration Rate (mmol m–2s–1) | Water Use Efficiency (μmol mmol−1) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F | S | F | S | F | S | F | S | F | S | F | S | |
| Control | 2.28 c | 2.36 c | 5.309 b | 4.751 d | 33.24 c | 31.22 c | 138.91 c | 125.18 c | 7.721 a | 8.053 a | 0.687 c | 0.589 c |
| Bentonite | 2.26 c | 2.39 c | 4.662 c | 4.110 d | 30.18 c | 32.19 c | 131.45 c | 122.63 c | 7.11 b | 7.690 b | 0.655 c | 0.534 c |
| Perlite | 2.02 d | 2.18 d | 3.204 c | 3.109 d | 27.61 c | 30.28 c | 126.71 c | 115.97 d | 7.948 a | 8.06 a | 0.403 d | 0.393 d |
| Kaolinite | 2.24 c | 2.31 c | 4.530 c | 4.085 d | 31.15 c | 30.92 c | 130.44 c | 123.09 c | 6.981 a | 7.215 b | 0.648 c | 0.566 c |
| N-silicon | 2.45 b | 2.49 b | 6.859 b | 6.937 c | 42.11 b | 43.04 b | 188.29 b | 191.04 b | 5.886 b | 5.210 c | 1.165 b | 1.331 b |
| N-zinc | 2.57 b | 2.69 b | 8.207 a | 7.914 b | 43.21 b | 42.89 b | 196.88 b | 206.37 a | 5.282 b | 5.079 c | 1.553 b | 1.558 b |
| N-zeolite | 3.09 a | 3.11 a | 9.067 a | 8.669 a | 48.05 a | 47.25 a | 241.21 a | 252.06 a | 4.371 c | 3.975 d | 2.074 a | 2.180 a |
| Treatment | IAA (µg/g F.W/Leaves) | GA3 (µg/g F.W/Leaves) | ABA (µg/g F.W/Leaves) | MDA (Units mg−1 Protein) | CAT (Units mg−1 Protein) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| F | S | F | S | F | S | F | S | F | S | |
| Control | 18.901 c | 18.720 c | 36.90 c | 38.115 c | 16.429 b | 18.109 a | 4.261 a | 4.078 a | 14.722 a | 15.051 a |
| Bentonite | 17.953 d | 17.105 d | 37.805 c | 36.332 c | 16.796 b | 17.441 a | 4.815 a | 4.922 a | 14.582 a | 15.307 a |
| Perlite | 15.908 e | 14.880 e | 32.107 d | 28.311 d | 19.028 a | 19.504 a | 4.510 a | 5.200 a | 14.903 a | 15.009 a |
| Kaolinite | 19.401 c | 18.776 c | 38.103 c | 35.109 c | 15.630 b | 16.408 b | 4.616 a | 4.190 a | 13.926 a | 14.622 a |
| N-silicon | 23.404 b | 21.759 b | 41.694 b | 40.810 b | 15.142 b | 15.026 b | 4.069 ab | 4.008 ab | 13.051 b | 12.691 b |
| N-zinc | 25.013 a | 22.105 b | 44.503 a | 42.775 b | 14.582 b | 14.972 b | 3.875 b | 3.290 b | 12.640 b | 12.033 b |
| N-zeolite | 26.428 a | 25.337 a | 46.968 a | 46.115 a | 13.708 c | 13.994 c | 3.103 b | 2.795 b | 10.976 c | 10.351 c |
| Treatment | N (%) | P (%) | K (%) | Mg (%) | Ca (%) | Fe (ppm) | Zn (ppm) | Mn (ppm) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F | S | F | S | F | S | F | S | F | S | F | S | F | S | F | S | |
| Control | 2.57 c | 2.51 c | 0.27 bc | 0.25 c | 3.28 b | 3.21 c | 0.22 c | 0.20 c | 1.48 c | 1.44 c | 161.2 c | 160.5 c | 48.8 bc | 48.2 bc | 10.6 c | 11.c |
| Bentonite | 2.56 c | 2.57 c | 0.27 bc | 0.26 c | 3.25 bc | 3.24 c | 0.23 c | 0.21 c | 1.41 c | 1.42 c | 168.4 c | 165.2 c | 46.61 c | 49.5 bc | 11.3 b | 11.1 c |
| Perlite | 2.32 d | 2.42 d | 0.25 c | 0.23 c | 3.20 d | 3.22 d | 0.20 d | 0.20 d | 1.42 d | 1.29 d | 155.7 d | 152.9 d | 45.2 c | 47.6 c | 10.8 c | 11.0 c |
| Kaolinite | 2.45 c | 2.55 c | 0.25 c | 0.24 c | 3.28 b | 3.26 c | 0.23 c | 0.20 c | 1.47 c | 1.40 c | 167.5 c | 166.3 c | 47.3 c | 46.8 c | 10.2 c | 10.6 c |
| N-silicon | 2.85 b | 2.80 b | 0.30 b | 0.32 b | 3.33 b | 3.29 bc | 0.30 b | 0.32 b | 1.51 b | 1.50 b | 181.2 b | 180.3 b | 52.5 b | 50.4 b | 13.2 b | 13.0 b |
| N-zinc | 2.96 b | 3.12 a | 0.30 b | 0.34 b | 3.43 a | 3.47 b | 0.33 b | 0.35 b | 1.53 b | 1.55 b | 187.5 b | 185.6 b | 56.4 b | 58.2 a | 13.3 b | 13.5 b |
| N-zeolite | 3.11 a | 3.20 a | 0.34 a | 0.37 a | 3.51 a | 3.59 a | 0.38 a | 0.40 a | 1.60 a | 1.63 a | 193.5 a | 196.8 a | 61.5 a | 60.3 a | 15.5 a | 16.8 a |
| Treatment | Fruit Yield/Plant (g) | Weight of 1000 Seeds (g) | Fruit Yield (Kg/fad) | Essential Oil/Plant (mL) | Essential Oil (L/fed.) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| F | S | F | S | F | S | F | S | F | S | |
| Control | 12.16 b | 12.00 b | 8.37 c | 8.01 c | 302.41 c | 300.39 c | 0.050 c | 0.052 c | 3.10 c | 3.09 c |
| Bentonite | 12.15 b | 12.57 b | 8.35 c | 8.02 c | 300.79 c | 298.55 c | 0.051 c | 0.049 c | 3.05 c | 3.03 c |
| Perlite | 11.49 b | 11.16 b | 7.22 d | 7.37 d | 289.63 d | 280.46 cd | 0.042 d | 0.040 d | 2.78 d | 2.72 d |
| Kaolinite | 12.46 b | 12.44 b | 8.19 c | 8.05 c | 306.27 c | 302.62 c | 0.051 c | 0.052 bc | 3.24 c | 3.13 c |
| N-silicon | 13.25 ab | 13.22 ab | 8.89 b | 8.86 b | 328.13 b | 325.10 b | 0.058 b | 0.059 b | 3.26 b | 3.19 b |
| N-zinc | 13.32 ab | 13.29 ab | 9.12 a | 9.10 a | 337.45 b | 335.72 b | 0.060 ab | 0.060 ab | 3.75 ab | 3.70 ab |
| N-zeolite | 14.68 a | 14.53 a | 9.44 a | 9.39 a | 350.25 a | 346.59 a | 0.066 a | 0.064 a | 4.15 a | 4.12 a |
| Fatty Acids Compounds | Control | Bentonite | Perlite | Kaolinite | N-Silicon | N-Zinc | N-Zeolite | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| F | S | F | S | F | S | F | S | F | S | F | S | F | S | |
| β-Pinene | 2.46 | 2.51 | 3.11 | 2.96 | 2.31 | 2.79 | 3.07 | 3.15 | 4.05 | 4.12 | 4.71 | 5.03 | 6.19 | 6.88 |
| Limonene | 3.66 | 3.97 | 2.71 | 2.89 | 3.45 | 4.11 | 4.03 | 4.29 | 3.59 | 3.78 | 5.02 | 4.94 | 5.15 | 5.44 |
| P-cymene | 4.11 | 4.75 | 3.15 | 3.55 | 3.82 | 4.09 | 4.12 | 4.18 | 5.44 | 5.48 | 3.89 | 4.18 | 4.62 | 5.1 |
| α-pinne | 3.29 | 3.41 | 3.02 | 3.13 | 2.08 | 3.16 | 3.21 | 3.27 | 3.32 | 3.37 | 3.46 | 3.52 | 4.15 | 4.63 |
| Camphor | 2.78 | 2.81 | 2.06 | 2.39 | 2.36 | 2.48 | 2.26 | 2.38 | 3.54 | 3.88 | 3.13 | 3.2 | 3.26 | 3.71 |
| Nerol | 0.72 | 0.81 | 0.45 | 0.51 | 0.42 | 0.44 | 0.55 | 0.61 | 0.52 | 0.58 | 0.33 | 0.39 | 0.35 | 0.37 |
| Camphene | 0.35 | 0.36 | 0.25 | 0.21 | 0.22 | 0.26 | 0.3 | 0.32 | 0.28 | 0.3 | 0.27 | 0.31 | 0.29 | 0.33 |
| Sabinene | 0.55 | 0.57 | 0.31 | 0.46 | 0.4 | 0.45 | 0.32 | 0.37 | 0.36 | 0.4 | 0.41 | 0.48 | 0.42 | 0.44 |
| Myrcene | 0.5 | 0.54 | 0.39 | 0.44 | 0.37 | 0.42 | 0.38 | 0.43 | 0.4 | 0.46 | 0.41 | 0.47 | 0.49 | 0.51 |
| Borneol | 0.15 | 0.17 | 0.05 | 0.08 | 0.1 | 0.12 | 0.09 | 0.11 | 0.07 | 0.12 | 0.1 | 0.13 | 0.09 | 0.1 |
| Linalool | 44.17 | 46.22 | 28.74 | 28.96 | 40.26 | 41.36 | 31.36 | 33.48 | 39.24 | 40.52 | 44.80 | 45.16 | 57.39 | 58.26 |
| Total% | 62.74 | 66.12 | 44.24 | 45.58 | 55.79 | 59.68 | 49.69 | 52.59 | 60.81 | 63.01 | 66.53 | 67.81 | 82.4 | 85.77 |
| Parameters | Soil Depth (cm) | |
|---|---|---|
| 0–30 | 30–60 | |
| Particle size distribution (%) | ||
| Sand | 90.10 | 90.00 |
| Silt | 6.90 | 6.50 |
| Clay | 3.00 | 3.50 |
| Textural class | Sand | Sand |
| Saturation water content (cm3 cm−3) | 0.385 | 0.396 |
| Field capacity cm3 cm−3 | 0.213 | 0.218 |
| Permanent wilting point (cm3 cm−3) | 0.057 | 0.057 |
| Available water (cm3 cm−3) | 0.156 | 0.161 |
| Bulk density (mg m−3) | 1.64 | 1.65 |
| Saturated hydraulic conductivity, cm day−1 | 240.00 | 234.00 |
| Organic matter (%) | 0.31 | 0.25 |
| Calcium carbonates (%) | 4.80 | 3.71 |
| pH (1:1, soil: water suspension) | 7.70 | 7.81 |
| EC(1:1, soil: water extract) (dS.m−1) | 1.62 | 1.83 |
| Soluble Cations Cmole(+). Kg−1 soil | ||
| Ca2+ | 13.85 | 13.41 |
| Mg2+ | 12.15 | 10.59 |
| Na+ | 8.10 | 10.25 |
| K+ | 6.00 | 6.05 |
| Soluble Anions, Cmole(−). Kg−1 soil | ||
| CO3− | - | - |
| HCO3− | 11.92 | 9.75 |
| Cl− | 14.00 | 10.50 |
| SO4− | 15.08 | 21.30 |
| Available nutrients mg Kg−1 soil | ||
| N | 16.21 | 13.12 |
| P | 7.78 | 6.21 |
| K | 46.50 | 45.89 |
| Fe | 9.20 | 12.00 |
| Mn | 1.63 | 1.50 |
| Cu | 2.10 | 1.15 |
| Zn | 2.00 | 1.61 |
| B | 0.23 | 0.21 |
| Property | Value |
|---|---|
| Moisture content (%) | 25 |
| PH (1:5) | 7.5 |
| EC (1:5 extract) dsm−1 | 3.1 |
| Organic-C (%) | 33.11 |
| Organic matter (%) | 70 |
| Total-N (%) | 1.82 |
| Total-K (%) | 1.25 |
| C/N ratio | 14:1 |
| Total-P (%) | 1.29 |
| Fe (ppm) | 1019 |
| Mn (ppm) | 111 |
| Cu (ppm) | 180 |
| Zn (ppm) | 280 |
| Total content of bacteria (cfu.g−1) | 2.5 × 107 |
| Phosphate dissolving bacteria (cfu.g−1) | 2.5 × 106 |
| Weed seeds | 0 |
| Chemical Composition (%) | SiO2 | TiO2 | Al2O3 | Fe2O3 | FeO | MnO | MgO | CaO | Na2O | K2O | SrO | P2O3 | N |
| 45.50 | 2.81 | 13.30 | 5.40 | 8.31 | 0.51 | 6.30 | 9.52 | 2.83 | 0.87 | 0.22 | 0.67 | 2.70 | |
| Trace elements (ppm) | Ba | Co | Cr | Se | Cu | Zn | Zr | Nb | Ni | Rb | Y | ||
| 10 | 1.2 | 35 | 0.8 | 19 | 64 | 257 | 13 | 55 | 15 | 22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mahmoud, A.W.M.; Rashad, H.M.; Esmail, S.E.A.; Alsamadany, H.; Abdeldaym, E.A. Application of Silicon, Zinc, and Zeolite Nanoparticles—A Tool to Enhance Drought Stress Tolerance in Coriander Plants for Better Growth Performance and Productivity. Plants 2023, 12, 2838. https://doi.org/10.3390/plants12152838
Mahmoud AWM, Rashad HM, Esmail SEA, Alsamadany H, Abdeldaym EA. Application of Silicon, Zinc, and Zeolite Nanoparticles—A Tool to Enhance Drought Stress Tolerance in Coriander Plants for Better Growth Performance and Productivity. Plants. 2023; 12(15):2838. https://doi.org/10.3390/plants12152838
Chicago/Turabian StyleMahmoud, Abdel Wahab M., Hassan M. Rashad, Sanaa E. A. Esmail, Hameed Alsamadany, and Emad A. Abdeldaym. 2023. "Application of Silicon, Zinc, and Zeolite Nanoparticles—A Tool to Enhance Drought Stress Tolerance in Coriander Plants for Better Growth Performance and Productivity" Plants 12, no. 15: 2838. https://doi.org/10.3390/plants12152838
APA StyleMahmoud, A. W. M., Rashad, H. M., Esmail, S. E. A., Alsamadany, H., & Abdeldaym, E. A. (2023). Application of Silicon, Zinc, and Zeolite Nanoparticles—A Tool to Enhance Drought Stress Tolerance in Coriander Plants for Better Growth Performance and Productivity. Plants, 12(15), 2838. https://doi.org/10.3390/plants12152838







