Abstract
Microalgae are used in various biotechnological processes, such as biofuel production due to their high biomass yields, agriculture as biofertilizers, production of high-value-added products, decontamination of wastewater, or as biological models for carbon sequestration. The number of these biotechnological applications is increasing, and as such, any advances that contribute to reducing costs and increasing economic profitability can have a significant impact. Nitrogen fixing organisms, often called diazotroph, also have great biotechnological potential, mainly in agriculture as an alternative to chemical fertilizers. Microbial consortia typically perform more complex tasks than monocultures and can execute functions that are challenging or even impossible for individual strains or species. Interestingly, microalgae and diazotrophic organisms are capable to embrace different types of symbiotic associations. Certain corals and lichens exhibit this symbiotic relationship in nature, which enhances their fitness. However, this relationship can also be artificially created in laboratory conditions with the objective of enhancing some of the biotechnological processes that each organism carries out independently. As a result, the utilization of microalgae and diazotrophic organisms in consortia is garnering significant interest as a potential alternative for reducing production costs and increasing yields of microalgae biomass, as well as for producing derived products and serving biotechnological purposes. This review makes an effort to examine the associations of microalgae and diazotrophic organisms, with the aim of highlighting the potential of these associations in improving various biotechnological processes.
1. Introduction
Algae are defined as a highly diverse group of eukaryotic organisms that undergo photosynthesis, but lack true roots, stems, leaves, and other specialized multicellular structures found in higher plants. Algae are considered one of the largest primary producers on our planet [1]. Within the algae group, the unicellular ones are referred to as microalgae. Although primarily abundant in aquatic ecosystems, microalgae can also thrive in various other habitats, such as terrestrial environments, within the crowded rhizosphere [2], in lichens [3], and even inside animal as in coral reefs [4]. Additionally, microalgae have been found on glaciers and ice sheets [5]. Microalgae exhibit a wide range of architectures, morphologies, and cell sizes. Their lifestyles are highly diverse and can be categorized as photoautotrophic, heterotrophic, or mixotrophic. Interestingly, microalgae have a broad metabolic capacity, some of which are unique and thus attract scientific research attention [6].
Symbiosis is a phenomenon widely utilized in nature, taking on multiple forms and serving various objectives that can be beneficial for only one of the organisms (commensalism, parasitism) or for both (mutualism) [7]. It has been observed that microalgae are capable of establishing symbiotic interactions with a diverse range of organisms, including plants, animals, fungi, and bacteria [8,9]. The term “phycosphere” refers to the area beyond the microalgal cell membrane, including the extracellular aqueous zone where bacteria interact with the microalgae. This region has no precise boundary and can extend to different distances around the microalgae [10]. In this area, bacterial growth is stimulated by the extracellular products of the microalgae, and vice versa. However, negative interactions between microalgae and bacteria mediated by various secondary metabolites (phycotoxins) have also been described [11]. In order to understand the interactions between microalgae and bacteria, several studies have focused on characterizing the microbiota of the phycosphere. It has been observed that the bacterial communities in the phycosphere are taxonomically distinct from those present in environments without microalgae [12]. Microalgae release significant amounts of dissolved organic matter that accumulates in the phycosphere, and these molecules are utilized as food or energy by the organisms that inhabit this niche [13]. Generally, the microalgae provide their partners with oxygen and a carbon source fixed through photosynthesis (photosynthates). In exchange, the microalgae receive some molecules necessary for their survival in their habitat, usually vitamins, hormones or a source of nitrogen [14,15]. The molecular and taxonomic diversity of mutualistic interactions between microalgae and their partners is significant. It is worth noting that the ability of organisms to adapt through mutations plays a critical role in the establishment of these interactions [16].
Nitrogen (N) is abundant in the atmosphere in the form of a dinitrogen gas (N2), but only a small number of prokaryotic organisms, known as diazotrophs, are capable of using it directly as a nitrogen source through biological nitrogen fixation (BNF) [17]. BNF is a highly regulated process due to the high energetic demands of the main enzyme involved in this reaction, nitrogenase. Nitrogenase requires 16 moles of ATP and 8 moles of electrons to reduce one mole of N2 to ammonium [18]. Several organisms rely directly or indirectly on diazotrophs and establish symbiotic interactions with them for proper growth. One of the most extensively studied interactions is the endosymbiotic relationship established between the roots of legumes and diazotrophic organisms called rhizobia, which provide fixed nitrogen to plants in exchange for protection, nutrients, and energy [19]. Mimicking this natural process, various diazotrophic organisms are also being tested as biofertilizers in non-leguminous plants [20]. The phycosphere of different green microalgae with great potential in the industry as Chlamydomonas reinhardtii (hereafter Chlamydomonas) [21], Chlorella vulgaris [22], Scenedesmus quadricauda [23] and Botryococcus braunii [24] has been characterized, and interestingly, some of them contain diazotrophic organisms such as Rhizobium sp. [21,24].
Over the past few years, there has been an exponential increase in the use of microalgae for various biotechnological applications [25]. Microalgae are crucial to sustaining the ecosystems, however occasionally they also produce harmful effects, like algal blooms that can cause a great ecological, economical and health impact [26]. In recent years, several studies have evaluated the role of naturally occurring diazotrophic association in organisms where microalgae play an essential function, such as corals and lichens. Additionally, studies have shown that the use of microalgae and diazotrophic organisms in consortia is very convenient for certain biotechnological applications.
In this review, we aim to gather the main available information on the different interactions studied between microalgae and N-fixing bacteria. Firstly, we present the interactions of microalgae with non-photosynthetic diazotrophic organisms, with various species of Azotobacter and Azospirillum standing out. Next, we move on to photosynthetic diazotrophic organisms, primarily focusing on cyanobacteria. Studies conducted with corals and lichens are also presented, where in some cases, the interactions between diazotrophs and microalgae also seem to play an important role. At the end, studies on different types of biotechnological applications utilizing the microalgae and N-fixing bacteria consortium are gathered and presented. The main objective is to draw attention to the fact that optimizing these consortia can lead to a significant decrease in the economic costs of cultivating these microorganisms.
2. Microalgae Interaction with Diazotrophic Bacteria
Plant growth-promoting bacteria (PGPB) enhance plant growth through various mechanisms, mainly by improving nutrient absorption in the soil. These bacteria have the ability to solubilize phosphorus, fix nitrogen, synthesize siderophores, produce phytostimulant substances such as auxins, gibberellins, cytokinins, and act as stress controllers in plants, among others [27]. An analogous concept, although less studied, is that of microalgae growth-promoting bacteria (MGPB), which are bacteria that enhance the growth of microalgae through several mechanisms [28]. Next, studies will be presented in which a free-living diazotrophic organism, naturally or artificially induced under laboratory conditions, acts as MGPB and improves the growth or certain characteristics of microalgae. This type of interaction has been divided into, on one hand, interactions between microalgae and photosynthetic or non-photosynthetic diazotrophic bacteria, and on the other hand, interactions between microalgae and diazotrophic bacteria in corals and lichens.
2.1. Microalgae Interaction with Non-Photosynthetic Diazotrophic Bacteria
The most numerous studies investigating interactions between microalgae and non-photosynthetic diazotrophic organisms have been conducted on Azotobacter spp. and Azospirillum spp. Therefore, we will pay special attention to these interactions.
2.1.1. Microalgae Interaction with Azotobacter spp.
The obligate aerobe Azotobacter sp. is a model organism for studying BNF. Azotobacter spp. are organisms that thrive in aerobic conditions. However, they have developed several mechanisms to adapt and carry out strictly anaerobic pathways, such as the BNF. Some of these mechanisms include increased respiration and high exopolysaccharide production [29]. This makes Azotobacter an ideal organism to co-culture with microalgae since it does not require growth in anaerobic environments. However, wild-type free-living diazotrophs like Azotobacter mainly fix enough N for their own needs and do not excrete significant amounts of fixed N products. In this sense, Azotobacter has evolved enzymes and transporters to minimize the loss of fixed N into the surrounding environment [30]. However, under certain conditions, some release of ammonium must occur since studies have shown that the N-fixing capacity of certain Azotobacter species (e.g., A. vinelandii, A. agilis, A. beljerinckii, A. chroococcum) can support the growth of different microalgal species like Chlamydomonas in media with no added N source, solely by using N from the air [31,32]. Indeed, to establish and maintain these co-culture for several years, it was critical to use nitrogen-free and carbon-free media [33]. That is why the absence of carbon must also be essential. Interestingly, the microalgal photosynthetic efficiency was similar to those growing with added N, which strongly suggests that Azotobacter, through its N-fixing capacity, provides Chlamydomonas with readily available N sources [34]. In return, it is clear that Chlamydomonas provides Azotobacter with fixed carbon, while Azotobacter provides the microalgae with fixed N. However, the precise identity of the exchanged molecules is still unknown, and only indirect clues are available. Azotobacter can excrete up to 17 different types of amino acids into the media [33], and Chlamydomonas can use some of these amino acids as N source [35]. In this regard, Chlamydomonas can also secrete different carbon sources as pyruvate, glycolate, oxalate [36], several keto acids [37], acetate [37], various fermentation products like malic acid, acetic acid, ethanol and formic acid [38] and amino acids and sugars [39]. Some of these compounds are known to be carbon sources for Azotobacter and, for this reason, may be the ones chosen for N exchange, although it is unknown which ones in particular.
This Chlamydomonas-Azotobacter association was extracellular. However, it has been described that Azotobacter and Chlamydomonas can fuse their membranes in the presence of polyethylene glycol, which can transform this association from extracellular to endosymbiotic while maintaining the ability to grow in N-deficient medium for several years [40]. In this forced endosymbiotic association between Chlamydomonas and Azotobacter, the diazotrophic cells were found not only in the Chlamydomonas periplasmic space and across the cell wall, but also in organelle-like vesicles located in the Chlamydomonas cytoplasm, which resembled true organelles. The number of endosymbiotic bacteria present in the Chlamydomonas cytoplasm varied from 1 to 8 per cell, with an average of 1–2 per cell. Also, the number of endosymbiotic bacteria and the volume ratio of Chlamydomonas-Azotobacter were kept nearly constant, indicating that these two parameters were critical for stabilizing the interaction [41]. When a third component such as the mycorrhizal fungus Alternaria sp. was introduced into this Chlamydomonas-Azotobacter consortia, it was observed that the presence of the fungus had a positive impact on the survival of the association in the absence of added N. It has been suggested that the fungus may contribute by releasing sulfur-containing amino acids, such as cystathionine, which could be utilized as a source of sulfur by both the microalgae and the bacteria [42].
Furthermore, certain strains of Azotobacter vinelandii have been genetically modified to secrete a certain amount of fixed N2 in the form of ammonium. In Azotobacter the nitrogenase expression is controlled by the two-component operon system NifL-NifA, where NifA serves as a transcriptional activator for nitrogenase, while NifL acts as an anti-activator by sensing the cell’s redox signals and energy status (Figure 1). The precise deletion of almost the entire NifL gene while leaving NifA intact (in the Azotobacter vinelandii AV3 mutant) resulted in constitutive expression of nitrogenase. This mutant presents a deregulated phenotype that produces levels of ammonium higher than the growth requirements of Azotobacter vinelandii, resulting in the release of excess ammonium into the growth medium [43] (Figure 1). The microalgae strains Chlorella sorokiniana RP, Pseukirchneriella sp. C1D, and Scenedesmus obliquus C1S were able to utilize this ammonium released by the Azotobacter vinelandii AV3 mutant to grow without requiring any other N source. The nitrogenase is a molybdoenzyme. In this context, the increase in molybdate present in the medium increases the nitrogenase activity and the release of ammonium [44,45] (Figure 1). However, its role in microalgae growth has not been studied yet. Furthermore, studies have shown that the deletion of NifL in Azotobacter vinelandii accompanied by increased expression of rnf1, which encodes an electron transport complex, can enhance the excretion of ammonium by the bacteria, possibly by increasing the availability of reducing equivalents to support nitrogenase catalysis [46] (Figure 1). The amount of carbon source was found to be a critical factor in sustaining high levels of ammonium release by Azotobacter vinelandii NifL. It was determined that 2 moles of sucrose were necessary to release one mole of ammonium [45].
It has been shown that mutations in the glutamine synthetase (GlnA) can also increase the ammonium excretion in Azotobacter vinelandii (Figure 1). Specifically, the Azotobacter vinelandii GlnA mutant (AV6) was found to release a significant amount of ammonium, and the double mutant of NifL and GlnA (AV7) also exhibited considerable ammonium release. The Azotobacter vinelandii strains AV6 and AV7 supported better growth of the microalgae Chlorella sorokiniana strain RP compared to Azotobacter vinelandii AV3 when atmospheric N2 was used as the sole source of nitrogen. This suggests that the increased ammonium excretion resulting from mutations in the GlnA and NifL genes has a positive impact on the growth of microalgae [47]. These results demonstrate that restricting amino acids synthesis and bacterial biomass through the GlnA inhibition resulted in a more energy-efficient strategy for the biological production of ammonium than deregulating nitrogenase through NifL mutation. One of the drawbacks of using Azotobacter vinelandii GlnA mutants is their poor growth under diazotrophic conditions. To address this issue, Ambrosio and collaborators developed the Azotobacter vinelandii AV11 strain, which has an inducible GlnA promoter that can be activated by isopropyl-β-thiogalactoside (IPTG) [48] (Figure 1). Azotobacter vinelandii AV11 requires IPTG to grow diazotrophically, but upon removal of IPTG from the medium, it exhibits a significant release of ammonium, up to 20 mM, which promotes robust growth of the microalgae S. obliquus and Chlorella sorokiniana [49]. Other approaches have been carried out to increase the production of extracellular ammonium by Azotobacter vinelandii. In this regard, the mutation of the ammonium transporter gene amtB in Azotobacter vinelandii resulted in the production of sufficient levels of extracellular ammonium to support the growth of Chlorella sorokiniana [50] (Figure 1). Interestingly, the authors also mutated the urease enzyme system (ureABC). This mutation increased the levels of extracellular urea; however, the concentration released was not sufficient to support the growth of Chlorella sorokiniana.
It has been observed that Azotobacter promotes the growth of microalgae using other strategies. In this regard, Azotobacter vinelandii in diazotrophic conditions provides the microalgae Neochloris oleoabundans and Scenedesmus sp. BA03 with azotobactin, a peptide siderophore, as a N source to support their growth [51]. This interaction appears to be commensalistic since Azotobacter vinelandii is apparently not benefiting from the microalgae. One of the challenges with using Azotobacter vinelandii mutants that accumulate ammonium is that any mutation that improves the growth rate while no longer fixing nitrogen will result in a subpopulation of cheaters, taking advantage of the accumulated ammonium. In this sense, some laboratories have reported that the mutation in the NifL gene is not very stable and over time spontaneous populations of revertants appear [49,52]. A question that needs to be resolved, especially if it is intended to cultivate these strains of Azotobacter and microalgae for a long time.
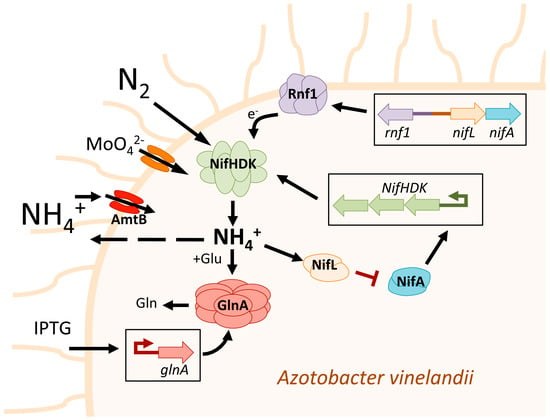
Figure 1.
Schematic representation of the different strategies followed in Azotobacter vinelandii to achieve high ammonium excretion. The operon NifHDK refers to a set of three genes, nifH, nifD, and nifK, that encode for the three subunits of nitrogenase. In Azotobacter vinelandii, nitrogenase expression is controlled by the two-component operon system NifL-NifA, where NifA is an activator and NifL is an anti-activator of nitrogenase. In the Azotobacter vinelandii AV3 mutant, NifL is mutated in a specific manner that prevents the inactivation of NifA by high intracellular ammonium concentration. This mutation leads to constitutive expression of nitrogenase and the release of excess ammonium into the growth medium [43]. Adjacent to operon NifL-NifA is the operon rnf1 that is transcribed divergently from nifLA promoter. Rnf1 is a membrane-bound complex involved in electron transport to nitrogenase. It is supposed that the increased expression of rnf1 enhances the electron (e−) supply to nitrogenase, thereby increasing its activity and, consequently, the production of ammonium [46]. The increase in molybdate present in the medium is supposed to produce higher molybdate transport, which increases nitrogenase activity and its ammonium production [44,45]. It has been demonstrated that a mutation in the glutamine synthetase gene (GlnA) in the Azotobacter vinelandii AV6 mutant prevents the conversion of ammonium to glutamine, leading to increased ammonium excretion. This is because the ammonium produced by the nitrogenase cannot be incorporated into glutamate (Glu) to form glutamine (Gln) [47]. The Azotobacter vinelandii AV11 mutant has an inducible GlnA promoter that can be activated by IPTG. When IPTG is removed from the medium, GlnA is not expressed, and Azotobacter vinelandii AV11 shows a significant increase in ammonium release [49]. The mutation of the ammonium transporter gene amtB prevents the re-entry of excreted ammonium into the cell interior, resulting in an increase in the presence of ammonium in the media [50].
Figure 1.
Schematic representation of the different strategies followed in Azotobacter vinelandii to achieve high ammonium excretion. The operon NifHDK refers to a set of three genes, nifH, nifD, and nifK, that encode for the three subunits of nitrogenase. In Azotobacter vinelandii, nitrogenase expression is controlled by the two-component operon system NifL-NifA, where NifA is an activator and NifL is an anti-activator of nitrogenase. In the Azotobacter vinelandii AV3 mutant, NifL is mutated in a specific manner that prevents the inactivation of NifA by high intracellular ammonium concentration. This mutation leads to constitutive expression of nitrogenase and the release of excess ammonium into the growth medium [43]. Adjacent to operon NifL-NifA is the operon rnf1 that is transcribed divergently from nifLA promoter. Rnf1 is a membrane-bound complex involved in electron transport to nitrogenase. It is supposed that the increased expression of rnf1 enhances the electron (e−) supply to nitrogenase, thereby increasing its activity and, consequently, the production of ammonium [46]. The increase in molybdate present in the medium is supposed to produce higher molybdate transport, which increases nitrogenase activity and its ammonium production [44,45]. It has been demonstrated that a mutation in the glutamine synthetase gene (GlnA) in the Azotobacter vinelandii AV6 mutant prevents the conversion of ammonium to glutamine, leading to increased ammonium excretion. This is because the ammonium produced by the nitrogenase cannot be incorporated into glutamate (Glu) to form glutamine (Gln) [47]. The Azotobacter vinelandii AV11 mutant has an inducible GlnA promoter that can be activated by IPTG. When IPTG is removed from the medium, GlnA is not expressed, and Azotobacter vinelandii AV11 shows a significant increase in ammonium release [49]. The mutation of the ammonium transporter gene amtB prevents the re-entry of excreted ammonium into the cell interior, resulting in an increase in the presence of ammonium in the media [50].
2.1.2. Microalgae Interaction with Azospirillum spp.
The free-living diazotrophs belonging to the Azospirillum genus are a group of plant rhizosphere bacteria that have been widely recognized as model PGPB due to their positive impact on plant growth, crop yields, and nitrogen content [53]. Bacteria belonging to this genus synthesize one polar flagellum during growth in liquid medium, primarily used for swimming towards optimal oxygen concentrations, a behavior known as aerotaxis [54]. Azospirillum does not excrete significant amounts of ammonium under diazotrophic growth. Therefore, in addition to biological nitrogen fixation, its plant stimulatory effect has also been attributed to several other mechanisms, which are reviewed in detail in [55]. Among these mechanisms, several stand out, including the secretion of the auxin phytohormone indole-3-acetic acid (IAA) [56], polyamine and trehalose production, an increase in the rate of mineral uptake by the plant [57], and the production of bioactive volatile organics, such as 2,3-butanediol and acetoin [58]. Apart from its well-known interaction with plants, several studies have also shown that Azospirillum is capable of establishing different interactions with microalgae mainly due to its auxin-production capacity [59]. In this regard, it has been described that Azospirillum brasilense in consortia with Chlorella species could almost completely mitigate the oxidative stress in the microalgae derived from N limitation [60], enhance removal of ammonium and phosphorus in wastewater [61], and increase the ammonium [62], polyphosphate [63], starch [64,65] and the fatty acid intracellular accumulation of Chlorella [66]. It has also been studied that Azospirillum brasilense maintains a mutualistic interaction with the microalgae Scenedesmus obliquus, Chlorella vulgaris, and Chlamydomonas reinhardtii, supported by the exchange of IAA and tryptophan, respectively. This interaction increases the microalgal CO2 fixation [67]. In addition to tryptophan, it has been described that thiamine released by Chlorella sorokiniana also plays a role in mutualism with Azospirillum brasilense [68].
It has been described that the N fixation by Azospirillum only occurs under microaerophilic conditions [69] which are unlikely to exist in the hyper-oxygenated environment of a microalgae culture. For this reason, its N-fixing capacity has not been considered important in any of the interactions between Azospirillum and microalgae described so far. However, some of these studies have been performed by co-immobilizing the microalgae C. vulgaris and Chlorella sorokiniana with Azospirillum brasilense in alginate beads [61,70,71,72,73,74]. Light microscopy revealed that both microorganisms colonized the same cavities inside the beads [75]. Moreover, it has been described that the interior of large alginate beads may become oxygen deficient [76]. Therefore, it is possible that in these microaerophilic spaces, Azospirillum could be fixing nitrogen and releasing it into the bead where the microalgae reside. As a result, the nitrogen fixation carried out by Azospirillum may be contributing more to these interactions than previously believed. Further experiments to test this hypothesis would be highly valuable.
2.1.3. Microalgae Interaction with Other Diazotrophic Bacteria
Apart from Azotobacter and Azospirillum, few diazotrophic bacterial have been studied for their interactions with microalgae. Co-culturing Chlorella vulgaris with the N2 fixing bacteria Bacillus pumilus ES4 has been shown to enhance the growth of microalgae, likely due to the N fixing ability of Bacillus pumilus, which leads to the accumulation of ammonium in the medium. This growth increase is noticeable only when no N is added to the media, suggesting that the microalgae may be assimilating some of the N fixed by Bacillus pumilus [77]. Nannochloropsis oceanica is a marine microalga known for its high production of eicosapentaenoic acid (EPA), making it one of the most commonly cultivated microalgae for aquaculture and food supplements. In a study, 10 bacteria were isolated and co-cultivated with Nannochloropsis oceanica, resulting in increased biomass and EPA production. The analysis showed that out of the bacteria tested, seven were capable of fixing N. Among them, Nitratireductor sp., a diazotrophic bacterium, caused the greatest increase in biomass [78].
2.2. Microalgae Interaction with Photosynthetic Diazotrophic Bacteria
Cereibacter sphaeroides (previously known as Rhodobacter sphaeroides) is an aerobic, photosynthetic [79] nitrogen fixing bacterium that secretes polysaccharides to avoid inactivation of nitrogenase, which leads to a decrease in the partial pressure of oxygen [80]. Cereibacter sphaeroides and the microalga Coelastrella sp. KGU-HN001 have been shown to maintain a mutualistic exchange that allows both organisms to grow in media with air as the only source of nitrogen and carbon. In the coculture, Coelastrella secreted several saccharides, and the authors propose that maltose is likely the carbon source used by Cereibacter sphaeroides [81]. Cyanobacteria is a wide and diversified phylum of photosynthetic bacteria. Some of them are capable of both oxygenic photosynthesis and N fixation. However, their N-fixing enzyme, the nitrogenase, is sensitive to oxygen [82]. Therefore, these organisms usually separate N fixation from photosynthesis in specialized cells called heterocysts. Heterocysts provide cyanobacteria vegetative cells with fixed N. In return, the vegetative cells provide the heterocysts with reduced carbon through photosynthesis, which is needed to support N fixation [83]. One of the characteristics of cyanobacteria, especially those forming heterocysts, is their high propensity for symbioses. Cyanobacteria establish various types of symbiosis with the host with variable degrees of integration. Most of these interactions have been described with terrestrial plants but also with hornworts, liverworts, ferns, cycads, cyanolichens, and angiosperms [84]. Additionally, interactions between N fixing cyanobacteria and microalgae, particularly diatoms (Bacillariophyceae), have been described. Diatoms are an ecologically and morphologically diverse group of microalgae that have a cell wall that incorporates silica as a main component. These interactions are called diatom-diazotroph associations (DDAs) and provide an essential contribution to oceanic CO2 fixation (primary productivity) supported by N fixation [85]. In DDAs, symbiotic diazotrophs form trichomes where one cell is specialized in N fixation, the heterocyst, while the remaining cells, the vegetative cells, are phototrophic and divide, whereas heterocysts cannot [86]. In marine environment trichomes receive essential nutrients that allow them to grow more efficiently in symbiosis [87], being unable to grow in the laboratory [88]. Unlike terrestrial systems, the interaction between marine diatoms and cyanobacteria is less characterized. In the DDAs of the diatom microalgae Hemiaulus hauckii and heterocystous cyanobacteria Richelia intracellularis, it has been shown that carbon transfer from the diatom enables faster growth and N fixation rate by the trichomes. The authors discovered that N fixation is 5.5 times higher during symbiosis, and that 25% of the fixed carbon from the host diatom is transferred to the symbiotic trichomes to support the high rate of N fixation. In turn, 82% of the N fixed by the trichomes ends up in the host. Richelia intracellularis can be located inside the cytoplasm of Hemiaulus hauckii, either partially (between the diatom plasma membrane and the cell wall) or completely external. Moreover, this location is related to the age of the association, with the older symbionts being more internal [89]. The associations of the diatoms Rhizosolenia Brightwell and Hemiaulus Ehrenberg, with Richelia intracellularis is hypothesized to have a critical role in the development of microalgae blooms in N limited region [90].
Measurements of nitrogen and carbon fixation rates have been reported for symbiotic associations of microalgae and cyanobacteria in oceans, but the majority of these measurements are based on bulk or plankton cell concentrates [91,92]. However, there are studies conducted in oligotrophic oceans where the rate of N fixation by symbiotic filamentous cyanobacteria Richelia intracellularis and Calothrix rhizosoleniae, and the transfer of N to their diatom partners (Hemiaulus spp., Rhizosolenia spp., and Chaetoceros compressus) have been measured [93]. The N fixation rates calculated for Calothrix and Richelia symbionts were 171–420 times higher when the cells were in symbiosis with the diatoms, compared to the rates estimated for the cells living independently. Furthermore, up to 97.3% of the fixed N is transferred to the diatom partners, and not assimilated by the symbionts themselves, nor released into the environment. Interestingly, having more trichomes in a single host diatom reduces the demand for N fixation per trichome, thereby decreasing their cost of carbon [87]. In agreement with that the diatom partners also have a positive influence on the metabolism and growth of their cyanobacterial symbionts [93]. In another study, also involving the diatom-Richelia system, the authors investigated how the partners coordinate their carbon fixation and how the diatom ensures that Richelia maintains a high rate of N fixation well above its needs. To achieve this, the researchers inhibited diatom photosynthesis with inhibitors, resulting in a decrease in the N fixation rate of Richelia. These findings suggest that despite the Richelia ability to fix their own carbon, it still relies on the diatoms for carbon fixation. This is likely because Richelia requires an additional supply of carbon to support the increased N fixation, which is provided by the photosynthetic activity of the diatom. Supporting this idea, up to 22% of the carbon assimilated by Richelia comes from the diatom [94].
As mentioned earlier, the heterocyst-forming cyanobacterium Richelia intracellularis provides fixed N to the diatom Hemiaulus hauckii, and hence there must also be mechanism(s) for molecular transfer of fixed N from the cyanobacterium to the diatom. To study this, an electron microscopy analysis was performed on the relationship between Hemiaulus hauckii and Richelia intracellularis, which revealed that the filaments of the Richelia intracellularis symbiont, typically composed of one terminal heterocyst and three or four vegetative cells, are located in the diatom’s cytoplasm and are not enclosed by a host membrane [95]. Interestingly, numerous membrane vesicles were detected in the vegetative cells of Richelia intracellularis. These vesicles can export cytoplasmic material from the bacterium, leading the authors to suggest that they represent a vehicle for the transfer of fixed N from Richelia intracellularis to the diatom’s cytoplasm. It is worth noting that the genome of Richelia intracellularis, which is 3.2 Mbp, is much smaller than the genome of most free-living heterocyst-forming cyanobacteria, which is typically 7–9 Mbp [96]. Therefore, the authors hypothesize that these vesicles, apart from transporting N, could also serve as a vehicle for gene transfer. Additionally, the authors observed a possible association of the cyanobacteria with the mitochondria of Hemiaulus hauckii and proposed that this association may be important for protecting the N-fixing enzyme, nitrogenase, from oxygen produced during photosynthesis. The oxygen-respiring mitochondria would help to decrease the level of intracellular oxygen in the vicinity of the cyanobacterium [95].
The amount and chemical form (ammonium, amino acids, etc.) in which N is transferred in the DDAs is unknown, as well as the role of the host in potentially providing glutamate or other C skeletons to the symbionts, and its influence on the N-fixation in the symbiont. Certain transporters encoded in the genomes of cyanobacteria have been proposed as responsible for transferring specific compounds between the symbiont and host. To investigate this, membrane transporters have been studied in three different types of DDAs according to the cellular location of the symbiont: the Hemiaulus hauckii- Richelia intracellularis RintHH01 symbiosis (internal association), Rhizosolenia clevei- Richelia intracellularis RintHM0 (partial association), and Chaetoceros compresus-Calothrix rhizosoleniae CalSC01 (external association) [97]. The truly internal Richelia symbionts of Hemiaulus spp. are proposed to be renamed as Richelia euintracellularis and the externally attached symbionts of Chaetoceros compressus are removed from the genus Calothrix and renamed Richelia rhizosoleniae [98]. When the symbiont is internal or partially internal, it contains a similar array of transporters. In contrast, the external symbiont has transporters that are similar to those of endosymbionts, as well as other transporters that are useful for life in a free-living form. The authors hypothesized that glutamine and arginine are transferred from the symbiont to the host in these DDAs, but the presence of ammonium transporter proteins, specifically in Calothrix rhizosoleniae CalSC01, and the presence of the NKCC1-type cation transporter in all three symbionts, suggest that ammonium may be a possible N vehicle in at least some DDAs.
The symbiotic association between three N-fixing cyanobacteria Anabaena variabilis, Westiellopsis prolifica and Nostoc muscorum with three green microalgae Chlorella vulgaris, Scenedesmus obliquus and Botryococcus braunii was studied under N-deficient conditions. Out of the nine interactions studied, the synergism between Botryococcus braunii and Nostoc muscorum was the best established, with a 50% enhancement in N fixation in co-culture. Analysis of their secretome revealed the presence of new secondary metabolites that have roles in quorum signaling, carbon metabolism, N fixation, lipid metabolism, and antimicrobial activity [99]. Some of these compounds may have interesting roles, such as 9-octadecenamide, a known phytohormone [100] and 1,30-triacontanediol, which improves glucose-lipid metabolism and exhibits antimicrobial activity [101].
A small number of marine microalgae have acquired cyanobacterial endosymbionts with the ability to fix nitrogen, allowing them to thrive in nitrogen-limited environments. These microalgae harbor the N-fixing cyanobacteria as organelles of endosymbiotic origin, known as spheroid bodies. Recently, these spheroid bodies have been propose to be renamed as ‘diazoplasts’ [102]. These organelles are distinctive from all other unicellular N-fixing cyanobacteria in that they only fix N in the presence of light [103]. The diazoplasts encode genes for N fixation and have the capacity to fix molecular N2, but they have lost the ability to perform photosynthesis. These organelles have been identified in the diatom microalga Rhopalodia gibba, being the closest free-living relatives diazotrophic cyanobacteria of the Cyanothece sp. group [103]. The unicellular cyanobacteria Crocosphaera spp. [104] appears to be highly favored for this type of endosymbiotic interaction. In fact, permanent endosymbionts that are closely related to the Crocosphaera genus, particularly Crocosphaera subtropica, are found in various microalgae including the diatoms Epithemia spp. and Climacodium frauenfeldianum [105], as well as the coccolithophore Braarudosphaera bigelowii [106]. Despite the diverse morphology of Epithemia spp., it is believed that their N-fixing endosymbiont originate from a solitary endosymbiotic event with a cyanobacteria belonging to the Crocosphaera genus [107]. The carbohydrate catabolism in the diazoplast implies that both the oxidative pentose pathway and oxidative phosphorylation work together to produce ATP and reducing equivalents, while consuming oxygen to facilitate the nitrogenase activity. The diazoplast has a reduced capacity to utilize alternate sources of N, in contrast to its increased nitrogenase activity [102].
Other example studied is the endosymbiotic cyanobacterium Candidatus Atelocyanobacterium thalassa (UCYN-A), which are obligate endosymbionts of single-celled haptophyte microalgae [108,109]. It has been described that UCYN-A has an endosymbiotic association with several picoeukaryotic microalgae of the Prymnesiophyceae class such as Emiliania huxleyi. The partnership is mutualistic because Emiliania. huxleyi receive fixed N from UCYN-A, and in exchange, transfer fixed carbon to UCYN-A [110]. It has been characterized that UCYN-A also establishes an endosymbiotic relationship with the microalga Braarudosphaera bigelowii [106]. Another interaction studied is the endosymbiotic relationship in marine environments between cyanobacteria of the UCYN-C group, closely related to Crocosphaera, and two diatoms from the Rhopalodiaceae family, Epithemia pelagica sp. and Epithemia catenata sp. These symbioses were challenging to detect because the endosymbionts lack fluorescent of the pigments involved in photosynthesis, have nitrogenase gene sequence similar to free-living cyanobacteria, and are only formed in N-deficient media [111]. Interestingly, it has recently been demonstrated in the oceans that species belonging to the UCYN groups are significant exporters of organic matter in the form of nitrogen and carbon, despite their small size, which is fixed in the deep sea. In this study, UCYN-B and UCYN-C cells were found to be embedded in large organic aggregates comprised of tens to hundreds of cells connected by an extracellular matrix, presumably facilitating this process [112].
2.3. Microalgae Interaction with Diazotrophic Bacteria in the Corals
Corals provide a habitat for many different organisms and are considered as multipartite symbiotic organisms, or holobionts, formed by the animal host (Cnidarians) and a diverse microbiota consisting mainly of microalgae from the genus Symbiodinium, as well as bacteria, archaea, fungi, and viruses [113]. The symbiotic association between the coral host and photoautotrophic microalgae of the genus Symbiodinium, commonly referred to as zooxanthellae, has been well studied in corals, see reviews [114,115]. These microalgae commonly reside in the endoderm of corals, where the coral host provides inorganic nutrients in exchange for photosynthetically fixed carbon and amino acids. Nitrogen assimilation is of particular importance for corals living in oligotrophic reef waters, as it is a major factor that limits their net primary productivity. Interestingly, N fixation activity have been detected in corals [116] and diazotrophic organism have been identified as their main responsible [117,118]. The coral’s acquisition of N through this process is called diazotroph-derived nitrogen (DDN). In this sense, a wide variety of cyanobacterial and non-cyanobacterial diazotrophic communities have been discovered in coral tissue, within the surface mucus layer, or in the coral skeleton [119]. These diazotrophic organisms, such as Cyanothece, have been shown to be taken up by the corals from the surrounding seawater [120,121]. The dominant diazotrophic organisms in corals are closely related to the bacterial group rhizobia, therefore it has been hypothesized that, as in terrestrial plants, these diazotrophs have developed a mutualistic relationship with corals and contribute to fixing N for the microalgae Symbiodinium [122]. Interestingly, It has been found that in Montipora corals, the abundances of Symbiodinium and diazotrophic bacteria closely related to the Vibrio genus were positively correlated [123].
It has been shown that the DDN is assimilated not only by the animal host but also by Symbiodinium, as highlighted in the review [124]. In relation to that, experiments have shown that in tropical corals such as Pocillopora damicornis and Stylophora pistillata as well as in Hawaiian corals of the genus Montipora, DDN notably increases the N enrichment of microalgae Symbiodinium [120,125]. This suggests that diazotrophs are an important source of N for the microalgae and increase the standing stock of their population. Interestingly, DDN has been linked to the ability of corals to deal with different types of stress. In this sense, it has been shown that DDN could trigger coral bleaching by generating an imbalanced nutrient supply, leading to phosphorus starvation of Symbiodinium [126]. During heat stress, the enrichment of dissolved organic carbon increases the abundances of diazotrophs in Xenia umbellata and Pinnigorgia flava corals, which increases N availability and destabilizes the coral-algal symbiosis [127]. However, beneficial effects of DDN in coral bleaching have also been reported. In the coral Oculina Patagonica, DDN has been described to facilitate the rapid proliferation of microalgae, which provide an alternative carbon source for bleached corals, helping them to recover [128]. DDN has been shown to increase as a mechanism for the coral to deal with thermal stress [129]. However, whether this N input is beneficial or not for the microalgae-host symbiotic association to deal with thermal increase remains to be resolved.
2.4. Microalgae Interaction with Diazotrophic Bacteria in the Lichens
Lichens are regarded as highly successful life forms due to their self-sustaining ecosystems that involve a diverse microbiota comprising various kingdoms of life in intricate, albeit not fully comprehended, associations [130,131]. Lichens are fascinating organisms in which a fungus (mycobiont) lives in an intimate relationship with at least one photosynthetic organism (photobiont). The photosynthetic organism in lichens can be either a microalgae or a filamentous alga (known as chlorolichens), a cyanobacterium (cyanolichens), or both (known as tripartite lichens or photosymbiodemes) [132,133]. Although lichens are generally defined as bipartite or tripartite associations, a wide array of bacterial communities are also present in a stable and host-specific manner. However, the precise role of these bacteria is still unknown [134]. The presence of these bacteria has been linked to the difficulty of resynthesizing or recreating some lichens under laboratory conditions through the coculture of their fungal and algal partners [135]. The microalgae Trebouxia, Coccomyxa and the filamentous alga Trentepohlia are the most commonly recruited green microalgae that form symbiotic relationships with the mycobiont [136]. In the context of lichen symbiosis, while there is a wealth of information on the interaction between the fungus and the photosynthetic partner, there has been relatively little research on the relationship between the microalgae and cyanobacteria in tripartite lichens. This stable symbiotic relationship enables chlorolichens to be C-autotrophs and cyanolichens to be C-autotrophs and N-autotrophs. These characteristics enable lichens to colonize nutrient-poor areas [137]. In cyanolichens, the cyanobacterium is referred to as the cyanobiont. In tripartite lichens, the cyanobiont is mainly responsible for N fixation [138] and it has been observed that lichen fitness increases through the specialization of the cyanobiont in nitrogen fixation [139]. To prevent the inhibition of nitrogenase by oxygen, the fungus partner of the lichen creates an environment with reduced oxygen levels and harbors the cyanobiont cells within internal structures called cephalodia, which are gall-like in appearance [140].
The cyanobiont in lichens is typically filamentous and capable of forming heterocysts (e.g., Calothrix, Nostoc). Nitrogen fixation is beneficial not only to the cyanobiont but also to both the fungal and, interestingly, the algal partners. The N-fixation capacity of the cyanobiont strongly and positively correlates with the N content and the maximum photosynthetic rate of the green alga [141]. It has been observed that the cyanobiont is dependent on the algal population for the majority of its carbon income [142]. Furthermore, this dependency is stronger in lichens with internal cephalodia such as Placopsis pycnotheca, which have lower exposure to light [143]. It should be noted that a direct physical interaction between cyanobacteria and green microalgae has never been observed [144]. Interestingly, the frequency of heterocysts respect to the total cells in bipartite lichens varies between 2–8%, whereas in tripartite lichens it varies between 10–55%, with higher rates of N fixation [139]. Nevertheless, in tripartite lichens, the number of cyanobionts is always kept lower than that of the algal photobiont, although the reason for this is unknown. Microalgae could potentially receive multiple benefits from this fixed N. Cyanolichens show higher concentrations of organic N compared to chlorolichens. Therefore, it has been concluded that cyanolichens may have an advantage in inhabiting special ecological niches, such as extremely oligotrophic habitats [145]. It is worth noting that the association with diazotrophic cyanobacteria has allowed lichens to surpass size limitations in oligotrophic environments [146].
It has been described that non-cyanobacterial diazotrophs are also present and are capable of performing N-fixation in association with lichens, particularly Azotobacter spp., Actinomycetes or lichen-associated Rhizobiales [147,148,149,150]. However, studies investigating the relationship between these non-cyanobacterial diazotrophs and the photobiont green microalgae are limited. Tripartite lichens have been found to synthesize a unique class of chemicals known as mycosporine-like amino acids [151], but their involvement in the association with the green microalgae requires further investigation.
3. Biotechnological Potential
The microalgae have a huge metabolic diversity, fast growth, and low production costs, do not compete for arable lands, can be cultivated throughout the year, and usually are more productive per unit of land area than any plant system, characteristics that make these organisms very useful in biotechnological applications aimed at meeting urgent needs in industry and agriculture [152]. Cyanobacteria also possess a series of favorable characteristics that have led them to be widely used in various biotechnological applications, as highlighted in the review [153]. For instance, microalgae are used in wastewater treatment, as sustainable sources to produce high-value compounds, fertilizers, hydrogen, biofuel, or livestock feed, as well as a way of carbon sequestration to avoid CO2 increase and global warming [154]. Microalgae possess a carbon fixation capacity that is 10–50 times higher than that of terrestrial plants [155]. In this regard, the potential of microalgae to mitigate global warming by reducing atmospheric CO2 through carbon sequestration, based on their rapid growth rate and biomass production, has been reviewed [156]. To enhance this carbon fixation capacity, an increase in N supply is necessary to provide the N required for nucleotide or protein biosynthesis, among other functions. Nevertheless, from a biotechnological point of view, there are still some inefficient procedures that can be improve by means of their interaction with other microorganisms, mainly bacteria and great efforts are being made to improve them [157].
Many of the biotechnological applications of microalgae rely on axenic cultures, but numerous strains of microalgae isolated from nature cannot be grown under axenic laboratory conditions. This means that these strains have become dependent on other organisms due to persistent interactions in their natural habitat. This reliance on other microorganisms is likely due to their long-term co-evolution [158]. Moreover, axenic cultures are more susceptible to contamination, predation, and collapse. Thus, the utilization of co-cultures consisting of multiple species that can adapt to specific conditions could solve this problem [157]. Open ponds are more prone to contamination; however, they are more cost-effective and easier to build than enclosed photobioreactors, so they are preferred for large-scale microalgae cultivation. A scrutiny has been carried out to see which bacterium increase the biomass production of the microalga Chlorella sorokiniana in open ponds. Interestingly, the bacterium that produced the best results was the N-fixing Rhizobium sp. [159]. Thus, it is possible that the N-fixing capacity of Rhizobium sp. provides a selective advantage to microalgae in open ponds. More studies are needed to corroborate this hypothesis, which would improve the cost of microalgae production for many biotechnological applications (Figure 2).
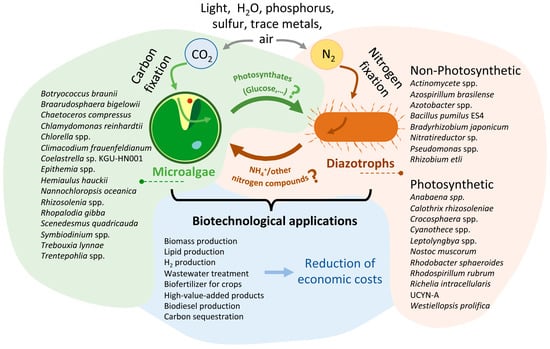
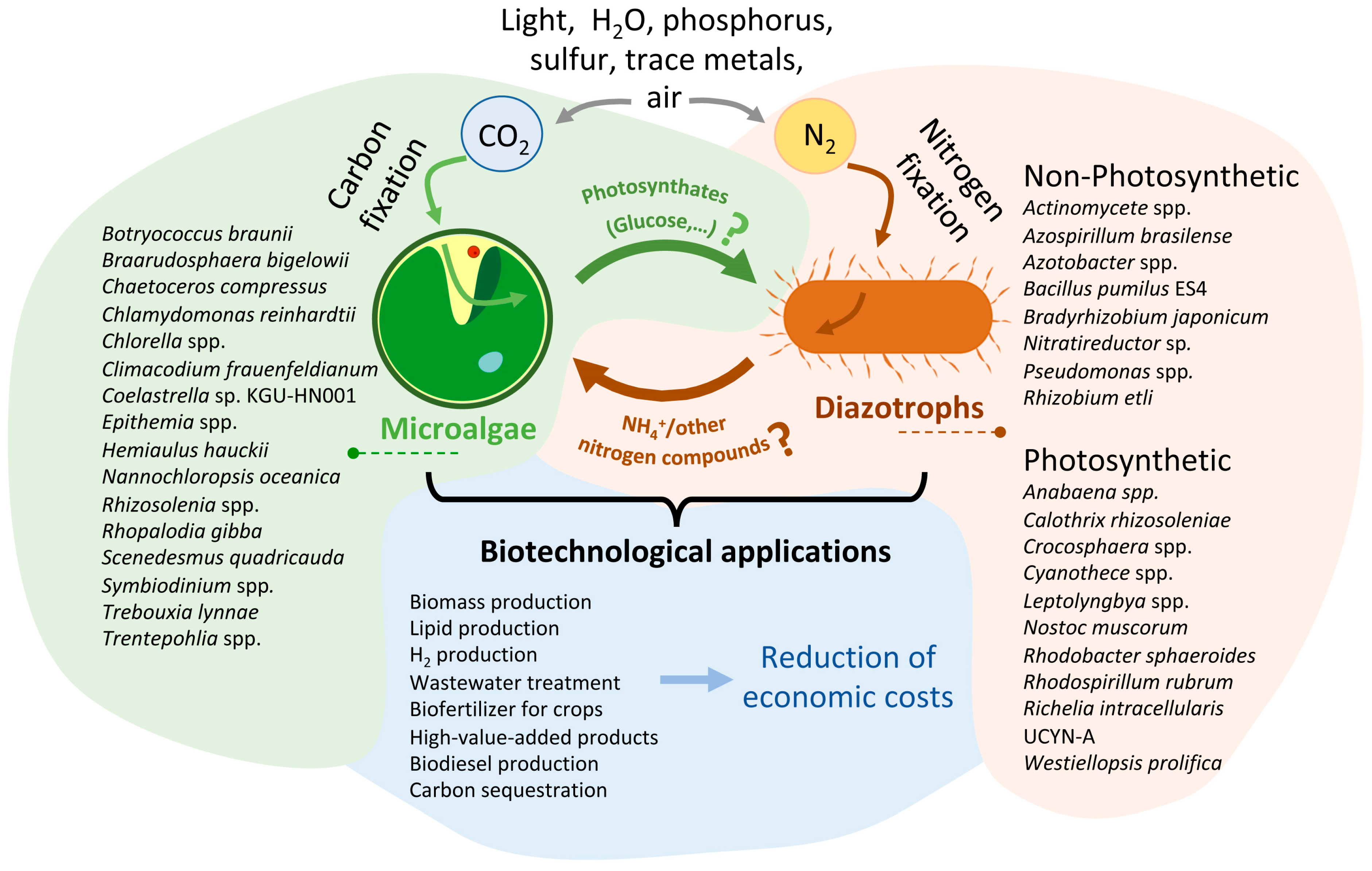
Figure 2.
Conceptual diagram of the different associations and biotechnological application between microalgae and diazotrophic organisms presented in this review. This figure represents a theoretical model of a carbon-nitrogen exchange system that utilizes air, light, water, phosphorus, sulfur, and trace metals to produce biomass and other biotechnological products. On the left, there is a drawing showing a microalga, and on the right, another drawing representing a diazotrophic organism. Diazotrophic photosynthetic organisms can also fix CO2, so there should be an arrow indicating this fact. However, to ensure that the figure clearly conveys the intended message, this fact is indicated here. The names of the main species of organisms described in this article are illustrated next to each organism. Next to the arrows, the main molecules exchanged during the interaction between both microorganisms are indicated. As indicated in the manuscript, the compounds exchanged between the two microorganisms have been more numerous (hormones as IAA, vitamins, etc.). However, in the figure, only those produced directly from the fixation of CO2 (photosynthates) and N2 (ammonium or other nitrogen compound) are represented. The question mark indicates that, as described in the article, in many cases, the specific photosynthate released is unknown. Additionally, not all of the diazotroph species mentioned have been reported to release ammonia or other N sources; for some of them, this is merely a hypothesis. The bottom part of the figure shows the main biotechnological applications described for these consortia. In the future, genetic manipulation engineering could significantly reduce the economic cost of producing such compounds through these consortia. For more details, refer to the text.
The use of microalgae biomass as a source of biofuel has been explored extensively [8]. However, the high cost of N fertilizers used in the cultivation process remains a major challenge. Eutrophication caused by the abusive use of chemical fertilizers has a huge environmental impacts and therefore is a current global problem [160]. Microalgae cocultivation with diazotrophic organisms has been seen to increase biomass and lipid production. In this sense, Azotobacter chroococcum can simultaneously enhance lipid and biomass productivity by promoting growth and inducing regulatory changes in the lipid metabolism of Chlamydomonas [161]. A study found that the co-culture of Chlorella vulgaris with Azotobacter Mesorhizobium sp. in N-deficient conditions increased the biomass and lipid of Chlorella vulgaris [162]. Growth, lipid, and biomass productivity of C. vulgaris increased remarkably in coculture with the cyanobacteria Actinomycet sp. [163], however there is no data about nitrogenase activity in this study, therefore the growth promotion could also be attributed to their production of phytohormones and vitamins [164]. Although it cannot be ruled out that increased growth could also be explained by the presence of N-rich bacterial debris in the system. Based on these data, it can be concluded that the symbiosis between N-fixing organisms and microalgae can be considered an alternative strategy for mass cultivation of microalgae with low N costs, for sustainable production of next-generation biofuels (Figure 2).
Hydrogen production by microorganisms is an area of active research, and there are several different approaches being explored. One approach is to use microorganisms that are capable of fermenting organic matter to produce H2. Some of these microorganisms are diazotrophic. In this regard, it has been observed that the utilization of microalgae, such as Chlamydomonas, can improve the efficiency of H2 production by different diazotrophic species, such as Cereibacter sphaeroides KD131 and Rhodospirillum rubrum [165,166]. On the other hand, some microalgae are capable of producing H2 by themselves, and one of the best-studied examples is Chlamydomonas. In this regard, it has been shown that H2 production by Chlamydomonas can be improved in the presence of certain diazotrophic bacteria. These bacteria include, among others, the soil bacteria Pseudomonas stutzeri, Rhizobium etli, Pseudomonas fluorescens, Azotobacter chroococcum, Mesorhizobium sangaii, Bradyrhizobium japonicum, see reviews [9,167]. However, until now, it has not been studied in detail whether this increased production of H2 is related to N fixation.
Bacteria are widely used in wastewater treatment; however, this has at least two drawbacks, they release the greenhouse gas CO2 and need large O2 supply which make this procedure not environmentally friendly and economically expensive. Therefore, the co-cultivation with oxygen-producing and CO2 fixating microalgae has become a promising way to eliminate those inconveniences [168]. However, studies focusing on the role of N-fixing organisms in these consortia are scarce. In a study, it has been shown that the N-fixing bacteria Azotobacter beijerinckii enhanced the biomass production and improved the wastewater treatment in co-culture with Auxenochlorella pyrenoidosa [169].
The green biotechnology in agriculture is based on the use of microalgae biomass as soil biofertilizer for crop cultivation and phytoremediation, reviewed in [170]. Furthermore, there is increasing interest in the use of N-fixing bacteria as a biofertilizer for crops [18]. Free-living aerobic diazotroph like Azotobacter spp. has been widely used as plant biofertilizer, successfully increasing the yields of a wide variety of crop plants [20]. Combining both strategies, the association of microalgae with N-fixing bacteria, can provide a sustainable solution by reducing the cost and environmental impact of fertilizers (Figure 2). However, the studies investigating whether the consortium of microalgae and N-fixing bacteria enhances this action are limited. It has been described that the diazotrophic cyanobacterium Anabaena azotica in co-culture with Auxenochlorella pyrenoidosa can be used as biofertilizers increasing the content of unsaturated fatty acid, and improve the content of nutrients in the fruits of Crataegi fructus [171]. Although more studies are needed to validate these results, inducing symbiosis between microalgae and N-fixing bacteria may have great potential in the next generation of biofertilizers in agriculture.
4. Concluding Remarks
Microalgae-based products have not been extensively developed due to the high costs associated with microalgae harvesting, maintenance, and extraction of their compounds. One of the biggest limitations is the economic cost of the N source used in these cultures. This review summarizes studies that investigate associations between microalgae and N-fixing organisms, whether induced artificially in the laboratory or naturally present in the environment. Overall, it can be concluded that the mechanisms mediating these interactions are increasingly being understood, and the more we learn, the closer we come to being able to cultivate microalgae exclusively using the N supplied by these organisms. This would result in a significant reduction in production costs. As mentioned, microalgae, through photosynthesis, fix atmospheric CO2 into carbon compounds or photosynthates. Another field in which future research could improve the economic profitability of such associations would be to enable a portion of these photosynthates produced by the microalgae to be released and donated to the diazotrophic bacteria in exchange for the N fixed by them. This would result in co-cultures of microalgae and diazotrophic bacteria using air as the sole source of carbon and nitrogen. Microalgae widely used as model organisms such as Chlamydomonas and Chlorella, along with aerobic diazotrophic bacteria such as Azotobacter, appear to be the best positioned for future research to achieve this goal.
Author Contributions
A.L. conceptualization and original draft preparation; A.L. and M.T.-J. wrote the paper; E.L.-M. review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by Gobierno de España, Ministerio de Ciencia e Innovacion (Grant PID2020-118398GB-I00), Junta de Andalucía (Grant ProyExcel_00483), the “Plan Propio” from University of Cordoba, and a grant awarded by the Torres-Gutierrez foundation.
Data Availability Statement
All data required to evaluate the conclusions of this paper are included in the main text.
Acknowledgments
This paper is dedicated to Emilio Fernandez Reyes for his more than 35 years using Chlamydomonas reinhardtii as a reference organism for nitrogen assimilation. He was the driving force behind the research on nitrogen assimilation in Chlamydomonas, the pillar that has allowed its advancement, and our great teacher whom we will never be able to repay for all the learnings received.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fabris, M.; Abbriano, R.M.; Pernice, M.; Sutherland, D.L.; Commault, A.S.; Hall, C.C.; Labeeuw, L.; McCauley, J.I.; Kuzhiuparambil, U.; Ray, P.; et al. Emerging Technologies in Algal Biotechnology: Toward the Establishment of a Sustainable, Algae-Based Bioeconomy. Front. Plant Sci. 2020, 11, 279. [Google Scholar] [CrossRef]
- Quintas-Nunes, F.; Brandão, P.R.; Barreto Crespo, M.T.; Glick, B.R.; Nascimento, F.X. Plant Growth Promotion, Phytohormone Production and Genomics of the Rhizosphere-Associated Microalga, Micractinium rhizosphaerae sp. nov. Plants 2023, 12, 651. [Google Scholar] [CrossRef]
- Barreno, E.; Muggia, L.; Chiva, S.; Molins, A.; Bordenave, C.; García-Breijo, F.; Moya, P. Trebouxia lynnae sp. nov. (Former Trebouxia Sp. TR9): Biology and Biogeography of an Epitome Lichen Symbiotic Microalga. Biology 2022, 11, 1196. [Google Scholar] [CrossRef]
- Scharfenstein, H.J.; Chan, W.Y.; Buerger, P.; Humphrey, C.; van Oppen, M.J.H. Evidence for de Novo Acquisition of Microalgal Symbionts by Bleached Adult Corals. ISME J. 2022, 16, 1676–1679. [Google Scholar] [CrossRef]
- Winkel, M.; Trivedi, C.B.; Mourot, R.; Bradley, J.A.; Vieth-Hillebrand, A.; Benning, L.G. Seasonality of Glacial Snow and Ice Microbial Communities. Front. Microbiol. 2022, 13, 876848. [Google Scholar] [CrossRef] [PubMed]
- Umen, J.; Herron, M.D. Green Algal Models for Multicellularity. Annu. Rev. Genet. 2021, 55, 603–632. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.Y.; Honda, K.; Derek, C.J.C. A Review on Microalgal-Bacterial Co-Culture: The Multifaceted Role of Beneficial Bacteria towards Enhancement of Microalgal Metabolite Production. Environ. Res. 2023, 228, 115872. [Google Scholar] [CrossRef]
- Ray, A.; Nayak, M.; Ghosh, A. A Review on Co-Culturing of Microalgae: A Greener Strategy towards Sustainable Biofuels Production. Sci. Total Environ. 2022, 802, 149765. [Google Scholar] [CrossRef] [PubMed]
- Calatrava, V.; Tejada-Jimenez, M.; Sanz-Luque, E.; Fernandez, E.; Galvan, A.; Llamas, A. Chlamydomonas Reinhardtii, a Reference Organism to Study Algal–Microbial Interactions: Why Can’t They Be Friends? Plants 2023, 12, 788. [Google Scholar] [CrossRef] [PubMed]
- Bell, W.; Mitchell, R. Chemotactic and Growth Responses of Marine Bacteria to Algal Extracellular Products. Biol. Bull. 1972, 143, 265–277. [Google Scholar] [CrossRef]
- Mathew, M.M.; Khatana, K.; Vats, V.; Dhanker, R.; Kumar, R.; Dahms, H.U.; Hwang, J.S. Biological Approaches Integrating Algae and Bacteria for the Degradation of Wastewater Contaminants—A Review. Front. Microbiol. 2022, 12, 801051. [Google Scholar] [CrossRef] [PubMed]
- Durán, P.; Flores-Uribe, J.; Wippel, K.; Zhang, P.; Guan, R.; Melkonian, B.; Melkonian, M.; Garrido-Oter, R. Shared Features and Reciprocal Complementation of the Chlamydomonas and Arabidopsis Microbiota. Nat. Commun. 2022, 13, 406. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, N.; Ahmad, F.; Lu, Y. Synergy between Microalgae and Microbiome in Polluted Waters. Trends Microbiol. 2023, 31, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Bunbury, F.; Deery, E.; Sayer, A.P.; Bhardwaj, V.; Harrison, E.L.; Warren, M.J.; Smith, A.G. Exploring the Onset of B 12-Based Mutualisms Using a Recently Evolved Chlamydomonas Auxotroph and B12-Producing Bacteria. Environ. Microbiol. 2022, 24, 3134–3147. [Google Scholar] [CrossRef]
- Smith, M.J.; Francis, M.B. A Designed A. vinelandii-S. elongatus Coculture for Chemical Photoproduction from Air, Water, Phosphate, and Trace Metals. ACS Synth. Biol. 2016, 5, 955–961. [Google Scholar] [CrossRef]
- Venkataram, S.; Kuo, H.-Y.; Hom, E.F.Y.; Kryazhimskiy, S. Mutualism-Enhancing Mutations Dominate Early Adaptation in a Two-Species Microbial Community. Nat. Ecol. Evol. 2023, 7, 143–154. [Google Scholar] [CrossRef]
- Threatt, S.D.; Rees, D.C. Biological Nitrogen Fixation in Theory, Practice, and Reality: A Perspective on the Molybdenum Nitrogenase System. FEBS Lett. 2023, 597, 45–58. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, Y.; Huang, K.; Wang, F.; Mei, Z. Molecular Mechanism and Agricultural Application of the NifA–NifL System for Nitrogen Fixation. Int. J. Mol. Sci. 2023, 24, 907. [Google Scholar] [CrossRef]
- Burghardt, L.T.; diCenzo, G.C. The Evolutionary Ecology of Rhizobia: Multiple Facets of Competition before, during, and after Symbiosis with Legumes. Curr. Opin. Microbiol. 2023, 72, 102281. [Google Scholar] [CrossRef]
- Aasfar, A.; Aasfar, A.; Bargaz, A.; Yaakoubi, K.; Hilali, A.; Bennis, I.; Zeroual, Y.; Meftah Kadmiri, I. Nitrogen Fixing Azotobacter Species as Potential Soil Biological Enhancers for Crop Nutrition and Yield Stability. Front. Microbiol. 2021, 12, 628379. [Google Scholar] [CrossRef]
- Kim, B.H.; Ramanan, R.; Cho, D.H.; Oh, H.M.; Kim, H.S. Role of Rhizobium, a Plant Growth Promoting Bacterium, in Enhancing Algal Biomass through Mutualistic Interaction. Biomass Bioenergy 2014, 69, 95–105. [Google Scholar] [CrossRef]
- Wirth, R.; Pap, B.; Böjti, T.; Shetty, P.; Lakatos, G.; Bagi, Z.; Kovács, K.L.; Maróti, G. Chlorella vulgaris and Its Phycosphere in Wastewater: Microalgae-Bacteria Interactions during Nutrient Removal. Front. Bioeng. Biotechnol. 2020, 8, 557572. [Google Scholar] [CrossRef] [PubMed]
- Astafyeva, Y.; Gurschke, M.; Qi, M.; Bergmann, L.; Indenbirken, D.; de Grahl, I.; Katzowitsch, E.; Reumann, S.; Hanelt, D.; Alawi, M.; et al. Microalgae and Bacteria Interaction—Evidence for Division of Diligence in the Alga Microbiota. Microbiol. Spectr. 2022, 10, 633. [Google Scholar] [CrossRef]
- Blifernez-Klassen, O.; Klassen, V.; Wibberg, D.; Cebeci, E.; Henke, C.; Rückert, C.; Chaudhari, S.; Rupp, O.; Blom, J.; Winkler, A.; et al. Phytoplankton Consortia as a Blueprint for Mutually Beneficial Eukaryote-Bacteria Ecosystems Based on the Biocoenosis of Botryococcus Consortia. Sci. Rep. 2021, 11, 1726. [Google Scholar] [CrossRef]
- Oliveira, C.Y.B.; Jacob, A.; Nader, C.; Oliveira, C.D.L.; Matos, Â.P.; Araújo, E.S.; Shabnam, N.; Ashok, B.; Gálvez, A.O. An Overview on Microalgae as Renewable Resources for Meeting Sustainable Development Goals. J. Environ. Manag. 2022, 320, 115897. [Google Scholar] [CrossRef]
- Kazmi, S.S.U.H.; Yapa, N.; Karunarathna, S.C.; Suwannarach, N. Perceived Intensification in Harmful Algal Blooms Is a Wave of Cumulative Threat to the Aquatic Ecosystems. Biology 2022, 11, 852. [Google Scholar] [CrossRef]
- Pereira, J.F.; Oliveira, A.L.M.; Sartori, D.; Yamashita, F.; Mali, S. Perspectives on the Use of Biopolymeric Matrices as Carriers for Plant-Growth Promoting Bacteria in Agricultural Systems. Microorganisms 2023, 11, 467. [Google Scholar] [CrossRef]
- Palacios, O.A.; Espinoza-Hicks, J.C.; Camacho-Dávila, A.A.; López, B.R.; de-Bashan, L.E. Differences in Exudates Between Strains of Chlorella Sorokiniana Affect the Interaction with the Microalga Growth-Promoting Bacteria Azospirillum brasilense. Microb. Ecol. 2022, 22, 02026. [Google Scholar] [CrossRef] [PubMed]
- Sabra, W.; Zeng, A.-P.; Lünsdorf, H.; Lünsdorf, L.; Deckwer, W.-D. Effect of Oxygen on Formation and Structure of Azotobacter vinelandii Alginate and Its Role in Protecting Nitrogenase. Appl. Environ. Microbiol. 2000, 66, 4037–4044. [Google Scholar] [CrossRef]
- Alleman, A.B.; Mus, F.; Peters, J.W. Metabolic Model of the Nitrogen-Fixing Obligate Aerobe Azotobacter vinelandii Predicts Its Adaptation to Oxygen Concentration and Metal Availability. MBio 2021, 12, e02593. [Google Scholar] [CrossRef]
- Gyurjan, I.; Turtoczky, I.; Toth, G.; Paless, G.; Nghia, N.H. Intercellular Symbiosis of Nitrogen-Fixing Bacteria and Green Alga. Acta Bot. Hung 1984, 30, 249–256. [Google Scholar]
- Gyurjan, I.; Nghia, N.H.; Tóth, G.; Turtozky, I.; Stefanovits, P. Photosynthesis, Nitrogen Fixation and Enzyme Activities in Chlamydomonas-Azotobacter Symbioses. Biochem. und Physiol. der Pflanz. 1986, 181, 147–153. [Google Scholar] [CrossRef]
- Preininger, E.; Ponyi, T.; Sarkadi, L.; Nyitrai, P.; Gyurjan, I. Long-Living Azotobacter-Chlamydomonas Association as a Model System for Plant-Microbe Interactions. Symbiosis 2006, 42, 45–50. [Google Scholar]
- Nghia, N.H.; Gyurján, I.; Stefanovits, P.; Turtóczky, I. Establishment of Nitrogen-Fixing Chlamydomonas-Azotobacter Symbioses: Physiological, Biochemical and Morphological Examinations. Endocyt. Cell Res. 1986, 3, 179–188. [Google Scholar]
- Calatrava, V.; Hom, E.F.Y.; Llamas, A.; Fernández, E.; Galvan, A. Nitrogen Scavenging from Amino Acids and Peptides in the Model Alga Chlamydomonas reinhardtii. The Role of Extracellular L-Amino Oxidase. Algal Res. 2019, 38, 101395. [Google Scholar] [CrossRef]
- Allen, M.B. Excretion of Organic Compounds by Chlamydomonas. Arch. Fiir Mikrobiol. 1956, 24, 163–168. [Google Scholar] [CrossRef]
- Collins, R.P.; Kalnins, K. Keto Acids Produced by Chlamydomonas reinhardtii. Can. J. Microbiol. 1967, 13, 995–999. [Google Scholar] [CrossRef]
- Mus, F.; Dubini, A.; Seibert, M.; Posewitz, M.C.; Grossman, A.R. Anaerobic Acclimation in Chlamydomonas reinhardtii. J. Biol. Chem. 2007, 282, 25475–25486. [Google Scholar] [CrossRef]
- Vogel, S.L.; Frisch, H.L.; Gotham, I.J. Qualitative Assay of Dissolved Amino Acids and Sugars Excreted by Chlamydomonas reinhardtii (Chlorophyceae) and Euglena gracilis (Euglenophyceae). J. Phycol. 1978, 14, 403–406. [Google Scholar] [CrossRef]
- Nghia, N.H.; Gyurján, I.; Stefanovits, P.; Paless, G.; Turtóczky, I. Uptake of Azotobacters by Somatic Fusion of Cell-Wall Mutants of Chlamydomonas reinhardtii. Biochem. Physiol. Pflanz. 1986, 181, 347–357. [Google Scholar] [CrossRef]
- Gyurjan, I.; Koranyi, P.; Paless, G.Y. Ultrastructural Analysis of an Artificial Alga-Bacterium Endosymbiosis After Prolonged Cultivation. Symbiosis 1992, 14, 475–484. [Google Scholar]
- Lorincz, Z.; Preininger, E.; Kósa, A.; Pónyi, T.; Nyitrai, P.; Sarkadi, L.; Kovács, G.M.; Böddi, B.; Gyurján, I. Artificial Tripartite Symbiosis Involving a Green Alga (Chlamydomonas), a Bacterium (Azotobacter) and a Fungus (Alternaria): Morphological and Physiological Characterization. Folia Microbiol. 2010, 55, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Marquez, J.C.F.; Nascimento, M.D.; Dublan, M.d.l.A.; Curatti, L. Association with an Ammonium-Excreting Bacterium Allows Diazotrophic Culture of Oil-Rich Eukaryotic Microalgae. Appl. Environ. Microbiol. 2012, 78, 2345–2352. [Google Scholar] [CrossRef]
- Barney, B.M.; Plunkett, M.H.; Natarajan, V.; Mus, F.; Knutson, C.M.; Peters, J.W. Transcriptional Analysis of an Ammonium-Excreting Strain of Azotobacter vinelandii Deregulated for Nitrogen Fixation. Appl. Environ. Microbiol. 2017, 83, 01534. [Google Scholar] [CrossRef]
- Plunkett, M.H.; Knutson, C.M.; Barney, B.M. Key Factors Affecting Ammonium Production by an Azotobacter Vinelandii Strain Deregulated for Biological Nitrogen Fixation. Microb. Cell Fact. 2020, 19, 107. [Google Scholar] [CrossRef]
- Mus, F.; Khokhani, D.; Rugoli, E.; Dixon John, R.; Ané, J.-M.; Peters, J. Genetic Determinants of Ammonia Excretion in NifL Mutants of Azotobacter vinelandii. Appl. Environ. Microbiol. 2022, 88, e01876. [Google Scholar] [CrossRef]
- Ortiz-Marquez, J.C.F.; Do Nascimento, M.; Curatti, L. Metabolic Engineering of Ammonium Release for Nitrogen-Fixing Multispecies Microbial Cell-Factories. Metab. Eng. 2014, 23, 154–164. [Google Scholar] [CrossRef]
- Ambrosio, R.; Ortiz-Marquez, J.C.F.; Curatti, L. Metabolic Engineering of a Diazotrophic Bacterium Improves Ammonium Release and Biofertilization of Plants and Microalgae. Metab. Eng. 2017, 40, 59–68. [Google Scholar] [CrossRef]
- Ambrosio, R.; Curatti, L. Deferred Control of Ammonium Cross-Feeding in a N2-Fixing Bacterium-Microalga Artificial Consortium. Appl. Microbiol. Biotechnol. 2021, 105, 2937–2950. [Google Scholar] [CrossRef]
- Barney, B.M.; Eberhart, L.J.; Ohlert, J.M.; Knutson, C.M.; Plunkett, M.H. Gene Deletions Resulting in Increased Nitrogen Release by Azotobacter vinelandii: Application of a Novel Nitrogen Biosensor. Appl. Environ. Microbiol. 2015, 81, 4316–4328. [Google Scholar] [CrossRef] [PubMed]
- Villa, J.A.; Ray, E.E.; Barney, B.M. Azotobacter vinelandii Siderophore Can Provide Nitrogen to Support the Culture of the Green Algae Neochloris oleoabundans and Scenedesmus sp. BA032. FEMS Microbiol. Lett. 2014, 351, 70–77. [Google Scholar] [CrossRef]
- Barney, B.M. Aerobic Nitrogen-Fixing Bacteria for Hydrogen and Ammonium Production: Current State and Perspectives. Appl. Microbiol. Biotechnol. 2020, 104, 1383–1399. [Google Scholar] [CrossRef]
- Vuolo, F.; Novello, G.; Bona, E.; Gorrasi, S.; Gamalero, E. Impact of Plant-Beneficial Bacterial Inocula on the Resident Bacteriome: Current Knowledge and Future Perspectives. Microorganisms 2022, 10, 2462. [Google Scholar] [CrossRef]
- Elmas, M.; Alexiades, V.; O’neal, L.; Alexandre, G. Modeling Aerotaxis Band Formation in Azospirillum brasilense. BMC Microbiol. 2019, 19, 101. [Google Scholar] [CrossRef] [PubMed]
- Fukami, J.; Cerezini, P.; Hungria, M. Azospirillum: Benefits That Go Far beyond Biological Nitrogen Fixation. AMB Express 2018, 8, 73. [Google Scholar] [CrossRef] [PubMed]
- Cassan, F.D.; Coniglio, A.; Amavizca, E.; Maroniche, G.; Cascales, E.; Bashan, Y.; de-Bashan, L.E. The Azospirillum brasilense Type VI Secretion System Promotes Cell Aggregation, Biocontrol Protection against Phytopathogens and Attachment to the Microalgae Chlorella sorokiniana. Environ. Microbiol. 2021, 23, 6257–6274. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Hernández, M.A.; Mendoza-Herrera, A.; Bocanegra-García, V.; Rivera, G. Azospirillum spp. from Plant Growth-Promoting Bacteria to Their Use in Bioremediation. Microorganisms 2022, 10, 1057. [Google Scholar] [CrossRef] [PubMed]
- Amavizca, E.; Bashan, Y.; Ryu, C.M.; Farag, M.A.; Bebout, B.M.; De-Bashan, L.E. Enhanced Performance of the Microalga Chlorella Sorokiniana Remotely Induced by the Plant Growth-Promoting Bacteria Azospirillum brasilense and Bacillus pumilus. Sci. Rep. 2017, 7, 41310. [Google Scholar] [CrossRef]
- Barbosa-Nuñez, J.A.; Palacios, O.A.; de-Bashan, L.E.; Snell-Castro, R.; Corona-González, R.I.; Choix, F.J. Active Indole-3-Acetic Acid Biosynthesis by the Bacterium Azospirillum brasilense Cultured under a Biogas Atmosphere Enables Its Beneficial Association with Microalgae. J. Appl. Microbiol. 2022, 132, 3650–3663. [Google Scholar] [CrossRef]
- Peng, H.; de-Bashan, L.E.; Higgins, B.T. Azospirillum brasilense Reduces Oxidative Stress in the Green Microalgae Chlorella sorokiniana under Different Stressors. J. Biotechnol. 2021, 325, 179–185. [Google Scholar] [CrossRef]
- de-Bashan, L.E.; Hernandez, J.P.; Morey, T.; Bashan, Y. Microalgae Growth-Promoting Bacteria as “Helpers” for Microalgae: A Novel Approach for Removing Ammonium and Phosphorus from Municipal Wastewater. Water Res. 2004, 38, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Meza, B.; de-Bashan, L.E.; Bashan, Y. Involvement of Indole-3-Acetic Acid Produced by Azospirillum brasilense in Accumulating Intracellular Ammonium in Chlorella vulgaris. Res. Microbiol. 2015, 166, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Meza, B.; de-Bashan, L.E.; Hernandez, J.P.; Bashan, Y. Accumulation of Intra-Cellular Polyphosphate in Chlorella vulgaris Cells Is Related to Indole-3-Acetic Acid Produced by Azospirillum brasilense. Res. Microbiol. 2015, 166, 399–407. [Google Scholar] [CrossRef]
- Palacios, O.A.; Choix, F.J.; Bashan, Y.; de-Bashan, L.E. Influence of Tryptophan and Indole-3-Acetic Acid on Starch Accumulation in the Synthetic Mutualistic Chlorella sorokiniana-Azospirillum brasilense System under Heterotrophic Conditions. Res. Microbiol. 2016, 167, 367–379. [Google Scholar] [CrossRef]
- Choix, F.J.; Bashan, Y.; Mendoza, A.; De-Bashan, L.E. Enhanced Activity of ADP Glucose Pyrophosphorylase and Formation of Starch Induced by Azospirillum brasilense in Chlorella vulgaris. J. Biotechnol. 2014, 177, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Leyva, L.A.; Bashan, Y.; Mendoza, A.; de-Bashan, L.E. Accumulation Fatty Acids of in Chlorella vulgaris under Heterotrophic Conditions in Relation to Activity of Acetyl-CoA Carboxylase, Temperature, and Co-Immobilization with Azospirillum brasilense. Naturwissenschaften 2014, 101, 819–830. [Google Scholar] [CrossRef]
- Choix, F.J.; Guadalupe López-Cisneros, C.; Oscar Méndez-Acosta, H. Azospirillum brasilense Increases CO2 Fixation on Microalgae Scenedesmus obliquus, Chlorella vulgaris, and Chlamydomonas reinhardtii Cultured on High CO2 Concentrations. Microb. Ecol. 2018, 76, 430–442. [Google Scholar] [CrossRef]
- Palacios, O.A.; Gomez-Anduro, G.; Bashan, Y.; de-Bashan, L.E. Tryptophan, Thiamine and Indole-3-Acetic Acid Exchange between Chlorella sorokiniana and the Plant Growth-Promoting Bacterium Azospirillum brasilense. FEMS Microbiol. Ecol. 2016, 92, fiw077. [Google Scholar] [CrossRef]
- Holguin, G.; Bashan, Y. Nitrogen-Fixation by Azospirillum brasilense Cd Is Promoted When Co-Cultured with a Mangrove Rhizosphere Bacterium (Staphylococcus sp.). Soil Bid. Eiochem 1996, 28, 1651–1660. [Google Scholar] [CrossRef]
- de-Bashan, L.E.; Moreno, M.; Hernandez, J.-P.; Bashan, Y. Removal of Ammonium and Phosphorus Ions from Synthetic Wastewater by the Microalgae Chlorella vulgaris Coimmobilized in Alginate Beads with the Microalgae Growth-Promoting Bacterium Azospirillum brasilense. Water Res. 2002, 36, 2941–2948. [Google Scholar] [CrossRef]
- Choix, F.J.; de-Bashan, L.E.; Bashan, Y. Enhanced Accumulation of Starch and Total Carbohydrates in Alginate-Immobilized Chlorella spp. Induced by Azospirillum brasilense: II. Heterotrophic Conditions. Enzym. Microb. Technol. 2012, 51, 300–309. [Google Scholar] [CrossRef] [PubMed]
- de-Bashan, L.E.; Schmid, M.; Rothballer, M.; Hartmann, A.; Bashan, Y. Cell-Cell Interaction in the Eukaryote-Prokaryote Model of the Microalgae Chlorella vulgaris and the Bacterium Azospirillum brasilense Immobilized in Polymer Beads. J. Phycol. 2011, 47, 1350–1359. [Google Scholar] [CrossRef]
- Covarrubias, S.A.; De-Bashan, L.E.; Moreno, M.; Bashan, Y. Alginate Beads Provide a Beneficial Physical Barrier against Native Microorganisms in Wastewater Treated with Immobilized Bacteria and Microalgae. Appl. Microbiol. Biotechnol. 2012, 93, 2669–2680. [Google Scholar] [CrossRef]
- Pham, T.M.; Bui, X.D.; Trang, L.V.K.; Le, T.M.; Nguyen, M.L.; Trinh, D.M.; Phuong, N.T.D.; Khoo, K.S.; Chew, K.W.; Show, P.L. Isolation of Indole-3-Acetic Acid-Producing Azospirillum brasilense from Vietnamese Wet Rice: Co-Immobilization of Isolate and Microalgae as a Sustainable Biorefinery. J. Biotechnol. 2022, 349, 12–20. [Google Scholar] [CrossRef]
- Gonzalez, L.E.; Bashan, Y. Increased Growth of the Microalga Chlorella vulgaris When Coimmobilized and Cocultured in Alginate Beads with the Plant-Growth-Promoting Bacterium Azospirillum brasilense. Appl. Environ. Microbiol. 2000, 66, 1527–1531. [Google Scholar] [CrossRef]
- O’reilly, A.M.; Scott, J.A. Defined Coimmobilization of Mixed Microorganism Cultures. Enzym. Microb. Technol. 1995, 17, 636–646. [Google Scholar] [CrossRef]
- Hernandez, J.P.; de-Bashan, L.E.; Rodriguez, D.J.; Rodriguez, Y.; Bashan, Y. Growth Promotion of the Freshwater Microalga Chlorella vulgaris by the Nitrogen-Fixing, Plant Growth-Promoting Bacterium Bacillus pumilus from Arid Zone Soils. Eur. J. Soil Biol. 2009, 45, 88–93. [Google Scholar] [CrossRef]
- Liu, B.; Eltanahy, E.E.; Liu, H.; Chua, E.T.; Thomas-Hall, S.R.; Wass, T.J.; Pan, K.; Schenk, P.M. Growth-Promoting Bacteria Double Eicosapentaenoic Acid Yield in Microalgae. Bioresour. Technol. 2020, 316, 123916. [Google Scholar] [CrossRef]
- Niederman, R.A. Development and Dynamics of the Photosynthetic Apparatus in Purple Phototrophic Bacteria. Biochim. Biophys. Acta-Bioenerg. 2016, 1857, 232–246. [Google Scholar] [CrossRef] [PubMed]
- Augimeri, R.V.; Varley, A.J.; Strap, J.L. Establishing a Role for Bacterial Cellulose in Environmental Interactions: Lessons Learned from Diverse Biofilm-Producing Proteobacteria. Front. Microbiol. 2015, 6, 1282. [Google Scholar] [CrossRef] [PubMed]
- Aburai, N.; Tsukagoshi, T.; Sekiguchi, S.; Arakawa, H.; Imamura, Y.; Abe, K. Mutual Supply of Carbon and Nitrogen Sources in the Co-Culture of Aerial Microalgae and Nitrogen-Fixing Bacteria. Algal Res. 2023, 70, 103001. [Google Scholar] [CrossRef]
- Burén, S.; Jiménez-Vicente, E.; Echavarri-Erasun, C.; Rubio, L.M. Biosynthesis of Nitrogenase Cofactors. Chem. Rev. 2020, 120, 4921–4968. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Zhang, C.-C. The Making of a Heterocyst in Cyanobacteria. Annu. Rev. Microbiol. 2022, 76, 597–618. [Google Scholar] [CrossRef]
- Kollmen, J.; Strieth, D. The Beneficial Effects of Cyanobacterial Co-Culture on Plant Growth. Life 2022, 12, 223. [Google Scholar] [CrossRef]
- Gao, M.; Armin, G.; Inomura, K. Low-Ammonium Environment Increases the Nutrient Exchange between Diatom–Diazotroph Association Cells and Facilitates Photosynthesis and N2 Fixation—A Mechanistic Modeling Analysis. Cells 2022, 11, 2911. [Google Scholar] [CrossRef] [PubMed]
- Flores, E.; Nieves-Morión, M.; Mullineaux, C.W. Cyanobacterial Septal Junctions: Properties and Regulation. Life 2019, 9, 1. [Google Scholar] [CrossRef]
- Inomura, K.; Follett, C.L.; Masuda, T.; Eichner, M.; Prášil, O.; Deutsch, C. Carbon Transfer from the Host Diatom Enables Fast Growth and High Rate of N2 Fixation by Symbiotic Heterocystous Cyanobacteria. Plants 2020, 9, 192. [Google Scholar] [CrossRef]
- Pyle, A.E.; Johnson, A.M.; Villareal, T.A. Isolation, Growth, and Nitrogen Fixation Rates of the Hemiaulus-Richelia (Diatom-Cyanobacterium) Symbiosis in Culture. PeerJ 2020, 8, 10115. [Google Scholar] [CrossRef]
- Caputo, A.; Nylander, J.A.A.; Foster, R.A. The Genetic Diversity and Evolution of Diatom-Diazotroph Associations Highlights Traits Favoring Symbiont Integration. FEMS Microbiol. Lett. 2019, 366, fny297. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.E.; Wilson, C.; Knap, A.H.; Villareal, T.A. Summer Diatom Blooms in the Eastern North Pacific Gyre Investigated with a Long-Endurance Autonomous Surface Vehicle. PeerJ 2018, 6, 5387. [Google Scholar] [CrossRef]
- Carpenter, E.J.; Montoya, J.P.; Burns, J.; Mulholland, M.R.; Subramaniam, A.; Capone, D.G. Extensive Bloom of a N2-Fixing Diatom/Cyanobacterial Association in the Tropical Atlantic. Source Mar. Ecol. Prog. Ser. 1999, 185, 273–283. [Google Scholar] [CrossRef]
- Karl, D.M.; Church, M.J.; Dore, J.E.; Letelier, R.M.; Mahaffey, C. Predictable and Efficient Carbon Sequestration in the North Pacific Ocean Supported by Symbiotic Nitrogen Fixation. Proc. Natl. Acad. Sci. USA 2012, 109, 1842–1849. [Google Scholar] [CrossRef]
- Foster, R.A.; Kuypers, M.M.M.; Vagner, T.; Paerl, R.W.; Musat, N.; Zehr, J.P. Nitrogen Fixation and Transfer in Open Ocean Diatom-Cyanobacterial Symbioses. ISME J. 2011, 5, 1484–1493. [Google Scholar] [CrossRef]
- Foster, R.A.; Tienken, D.; Littmann, S.; Whitehouse, M.J.; Kuypers, M.M.M.; White, A.E. The Rate and Fate of N2 and C Fixation by Marine Diatom-Diazotroph Symbioses. ISME J. 2021, 16, 477–487. [Google Scholar] [CrossRef]
- Flores, E.; Romanovicz, D.K.; Nieves-Morión, M.; Foster, R.A.; Villareal, T.A. Adaptation to an Intracellular Lifestyle by a Nitrogen-Fixing, Heterocyst-Forming Cyanobacterial Endosymbiont of a Diatom. Front. Microbiol. 2022, 13, 799362. [Google Scholar] [CrossRef]
- Hilton, J.A.; Foster, R.A.; James Tripp, H.; Carter, B.J.; Zehr, J.P.; Villareal, T.A. Genomic Deletions Disrupt Nitrogen Metabolism Pathways of a Cyanobacterial Diatom Symbiont. Nat. Commun. 2013, 4, 1767. [Google Scholar] [CrossRef] [PubMed]
- Nieves-Morión, M.; Flores, E.; Foster, R.A. Predicting Substrate Exchange in Marine Diatom-Heterocystous Cyanobacteria Symbioses. Environ. Microbiol. 2020, 22, 2027–2052. [Google Scholar] [CrossRef]
- Foster, R.A.; Villareal, T.A.; Lundin, D.; Waterbury, J.B.; Webb, E.A.; Zehr, J.P. Richelia. In Bergey’s Manual of Systematics of Archaea and Bacteria; Wiley: Hoboken, NJ, USA, 2022; pp. 1–17. [Google Scholar]
- Gautam, K.; Tripathi, J.K.; Pareek, A.; Sharma, D.K. Growth and Secretome Analysis of Possible Synergistic Interaction between Green Algae and Cyanobacteria. J. Biosci. Bioeng. 2019, 127, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Meenakshi, V.K.; Gomathy, S.; Senthamarai, S.; Paripooranaselvi, M.; Chamundeswari, K.P. GC-MS Determination of the Bioactive Components of Microcosmus exasperatus Heller, 1878. J. Curr. Chem. Pharm. Sc. 2012, 2, 271–276. [Google Scholar]
- Zhai, X.; Lenon, G.B.; Xue, C.C.L.; Li, C.G. Euonymus alatus: A Review on Its Phytochemistry and Antidiabetic Activity. Evidence-Based Complement. Altern. Med. 2016, 2016, 9425714. [Google Scholar] [CrossRef]
- Moulin, S.L.Y.; Frail, S.; Doenier, J.; Braukmann, T.; Yeh, E. The Endosymbiont of Epithemia clementina Is Specialized for Nitrogen Fixation within a Photosynthetic Eukaryote. bioRxiv 2023, preprint. [Google Scholar] [CrossRef]
- Kneip, C.; Voß, C.; Lockhart, P.J.; Maier, U.G. The Cyanobacterial Endosymbiont of the Unicellular Algae Rhopalodia gibba Shows Reductive Genome Evolution. BMC Evol. Biol. 2008, 8, 30. [Google Scholar] [CrossRef]
- Zehr, J.P.; Foster, R.A.; Waterbury, J.; Webb, E.A. Crocosphaera. In Bergey’s Manual of Systematics of Archaea and Bacteria; Wiley: Hoboken, NJ, USA, 2022; pp. 1–9. [Google Scholar]
- Carpenter, E.J.; Janson, S. Intracellular Cyanobacterial Symbionts in the Marine Diatom Climacodium frauenfeldianum (Bacillariophyceae). J. Phycol. 2000, 36, 540–544. [Google Scholar] [CrossRef] [PubMed]
- Hagino, K.; Onuma, R.; Kawachi, M.; Horiguchi, T. Discovery of an Endosymbiotic Nitrogen-Fixing Cyanobacterium UCYN-A in Braarudosphaera bigelowii (Prymnesiophyceae). PLoS ONE 2013, 8, e81749. [Google Scholar] [CrossRef]
- Nakayama, T.; Ikegami, Y.; Nakayama, T.; Ishida, K.I.; Inagaki, Y.; Inouye, I. Spheroid Bodies in Rhopalodiacean Diatoms Were Derived from a Single Endosymbiotic Cyanobacterium. J. Plant Res. 2011, 124, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Turk-Kubo, K.A.; Mills, M.M.; Arrigo, K.R.; van Dijken, G.; Henke, B.A.; Stewart, B.; Wilson, S.T.; Zehr, J.P. UCYN-A/Haptophyte Symbioses Dominate N2 Fixation in the Southern California Current System. ISME Commun. 2021, 1, 42. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Marín, M.D.C.; Magasin, J.D.; Zehr, J.P. Open Ocean and Coastal Strains of the N2-Fixing Cyanobacterium UCYN-A Have Distinct Transcriptomes. PLoS ONE 2023, 18, e0272674. [Google Scholar] [CrossRef]
- Thompson, A.W.; Foster, R.A.; Krupke, A.; Carter, B.J.; Musat, N.; Vaulot, D.; Kuypers, M.M.M.; Zehr, J.P. Unicellular Cyanobacterium Symbiotic with a Single-Celled Eukaryotic Alga. Science 2012, 337, 1546–1554. [Google Scholar] [CrossRef] [PubMed]
- Schvarcz, C.R.; Wilson, S.T.; Caffin, M.; Stancheva, R.; Li, Q.; Turk-Kubo, K.A.; White, A.E.; Karl, D.M.; Zehr, J.P.; Steward, G.F. Overlooked and Widespread Pennate Diatom-Diazotroph Symbioses in the Sea. Nat. Commun. 2022, 13, 799. [Google Scholar] [CrossRef]
- Bonnet, S.; Benavides, M.; Le Moigne, F.A.C.; Camps, M.; Torremocha, A.; Grosso, O.; Dimier, C.; Spungin, D.; Berman-Frank, I.; Garczarek, L.; et al. Diazotrophs Are Overlooked Contributors to Carbon and Nitrogen Export to the Deep Ocean. ISME J. 2023, 17, 47–58. [Google Scholar] [CrossRef]
- Bove, C.B.; Valadez-Ingersoll, M.; Davies, S.W. Help Me, Symbionts, You’re My Only Hope: Approaches to Accelerate Our Understanding of Coral Holobiont Interactions. Integr. Comp. Biol. 2022, 62, 1756–1769. [Google Scholar] [CrossRef]
- Thirukanthan, C.S.; Azra, M.N.; Lananan, F.; Sara’, G.; Grinfelde, I.; Rudovica, V.; Vincevica-Gaile, Z.; Burlakovs, J. The Evolution of Coral Reef under Changing Climate: A Scientometric Review. Animals 2023, 13, 949. [Google Scholar] [CrossRef]
- Benavides, M.; Bednarz, V.N.; Ferrier-Pagès, C. Diazotrophs: Overlooked Key Players within the Coral Symbiosis and Tropical Reef Ecosystems? Front. Mar. Sci. 2017, 4, 10. [Google Scholar] [CrossRef]
- Mague, T.H.; Holm-Hansen, O. Nitrogen Fixation on a Coral Reef. Phycologia 1975, 14, 87–92. [Google Scholar] [CrossRef]
- Shashar, N.; Cohen, Y.; Loya, Y.; Sar, N. Nitrogen Fixation (Acetylene Reduction) in Stony Corals: Evidence for Coral-Bacteria Interactions. Mar. Ecol. Prog. Ser. 1994, 111, 259–264. [Google Scholar] [CrossRef]
- Morrow, K.M.; Pankey, M.S.; Lesser, M.P. Community Structure of Coral Microbiomes Is Dependent on Host Morphology. Microbiome 2022, 10, 113. [Google Scholar] [CrossRef] [PubMed]
- Lema, K.A.; Willis, B.L.; Bourneb, D.G. Corals Form Characteristic Associations with Symbiotic Nitrogen-Fixing Bacteria. Appl. Environ. Microbiol. 2012, 78, 3136–3144. [Google Scholar] [CrossRef]
- Benavides, M.; Houlbreque, F.; Camps, M.; Lorrain, A.; Grosso, O.; Bonnet, S. Diazotrophs: A Non-Negligible Source of Nitrogen for the Tropical Coral Stylophora pistillata. J. Exp. Biol. 2016, 219, 2608–2612. [Google Scholar] [CrossRef]
- Meunier, V.; Geissler, L.; Bonnet, S.; Rädecker, N.; Perna, G.; Grosso, O.; Lambert, C.; Rodolfo-Metalpa, R.; Voolstra, C.R.; Houlbrèque, F. Microbes Support Enhanced Nitrogen Requirements of Coral Holobionts in a High CO2 Environment. Mol. Ecol. 2021, 30, 5888–5899. [Google Scholar] [CrossRef] [PubMed]
- Camps, M.; Benavides, M.; Lema, K.A.; Bourne, D.G.; Grosso, O.; Bonnet, S. Released Coral Mucus Does Not Enhance Planktonic N2 Fixation Rates. Aquat. Microb. Ecol. 2016, 77, 51–63. [Google Scholar] [CrossRef]
- Olson, N.D.; Ainsworth, T.D.; Gates, R.D.; Takabayashi, M. Diazotrophic Bacteria Associated with Hawaiian Montipora Corals: Diversity and Abundance in Correlation with Symbiotic Dinoflagellates. J. Exp. Mar. Bio. Ecol. 2009, 371, 140–146. [Google Scholar] [CrossRef]
- Bednarz, V.N.; Grover, R.; Maguer, J.F.; Fine, M.; Ferrier-Pagès, C. The Assimilation of Diazotroph-Derived Nitrogen by Scleractinian Corals Depends on Their Metabolic Status. mBio 2017, 8, e02058. [Google Scholar] [CrossRef] [PubMed]
- Ceh, J.; Kilburn, M.R.; Cliff, J.B.; Raina, J.B.; Van Keulen, M.; Bourne, D.G. Nutrient Cycling in Early Coral Life Stages: Pocillopora damicornis Larvae Provide Their Algal Symbiont (Symbiodinium) with Nitrogen Acquired from Bacterial Associates. Ecol. Evol. 2013, 3, 2393–2400. [Google Scholar] [CrossRef]
- Pogoreutz, C.; Rädecker, N.; Cárdenas, A.; Gärdes, A.; Voolstra, C.R.; Wild, C. Sugar Enrichment Provides Evidence for a Role of Nitrogen Fixation in Coral Bleaching. Glob. Chang. Biol. 2017, 23, 3838–3848. [Google Scholar] [CrossRef] [PubMed]
- Xiang, N.; Meyer, A.; Pogoreutz, C.; Rädecker, N.; Voolstra, C.R.; Wild, C.; Gärdes, A. Excess Labile Carbon Promotes Diazotroph Abundance in Heat-Stressed Octocorals. R. Soc. Open Sci. 2023, 10, 221268. [Google Scholar] [CrossRef]
- Bednarz, V.N.; van de Water, J.A.J.M.; Rabouille, S.; Maguer, J.F.; Grover, R.; Ferrier-Pagès, C. Diazotrophic Community and Associated Dinitrogen Fixation within the Temperate Coral Oculina patagonica. Environ. Microbiol. 2019, 21, 480–495. [Google Scholar] [CrossRef]
- Cardini, U.; van Hoytema, N.; Bednarz, V.N.; Rix, L.; Foster, R.A.; Al-Rshaidat, M.M.D.; Wild, C. Microbial Dinitrogen Fixation in Coral Holobionts Exposed to Thermal Stress and Bleaching. Environ. Microbiol. 2016, 18, 2620–2633. [Google Scholar] [CrossRef]
- Grimm, M.; Grube, M.; Schiefelbein, U.; Zühlke, D.; Bernhardt, J.; Riedel, K. The Lichens’ Microbiota, Still a Mystery? Front. Microbiol. 2021, 12, 623839. [Google Scholar] [CrossRef]
- Almer, J.; Resl, P.; Gudmundsson, H.; Warshan, D.; Andrésson, Ó.S.; Werth, S. Symbiont-Specific Responses to Environmental Cues in a Threesome Lichen Symbiosis. Mol. Ecol. 2023, 32, 1045–1061. [Google Scholar] [CrossRef]
- Jung, P.; Brust, K.; Schultz, M.; Büdel, B.; Donner, A.; Lakatos, M. Opening the Gap: Rare Lichens With Rare Cyanobionts—Unexpected Cyanobiont Diversity in Cyanobacterial Lichens of the Order Lichinales. Front. Microbiol. 2021, 12, 728378. [Google Scholar] [CrossRef]
- Lindgren, H.; Moncada, B.; Lücking, R.; Magain, N.; Simon, A.; Goffinet, B.; Sérusiaux, E.; Nelsen, M.P.; Mercado-Díaz, J.A.; Widhelm, T.J.; et al. Cophylogenetic Patterns in Algal Symbionts Correlate with Repeated Symbiont Switches during Diversification and Geographic Expansion of Lichen-Forming Fungi in the Genus sticta (Ascomycota, Peltigeraceae). Mol. Phylogenet. Evol. 2020, 150, 106860. [Google Scholar] [CrossRef]
- Sigurbjörnsdóttir, M.A.; Andrésson, Ó.S.; Vilhelmsson, O. Nutrient Scavenging Activity and Antagonistic Factors of Non-Photobiont Lichen-Associated Bacteria: A Review. World J. Microbiol. Biotechnol. 2016, 32, 68. [Google Scholar] [CrossRef] [PubMed]
- Muggia, L.; Fernández-Brime, S.; Grube, M.; Wedin, M. Schizoxylon as an Experimental Model for Studying Interkingdom Symbiosis. FEMS Microbiol. Ecol. 2016, 92, fiw165. [Google Scholar] [CrossRef]
- Friedl, T.; Büdel, B. Photobionts. In Lichen Biology; Nash, T.H., Ed.; Cambridge University Press: Cambridge, UK, 2008; pp. 9–26. [Google Scholar]
- Lawrey, J.D. Biological Role of Lichen Substances. Source Bryol. Summer 1986, 89, 111–122. [Google Scholar] [CrossRef]
- Millbank, J.W.; Kershaw, K.A. Nitrogen Metabolism in Lichens: I. Nitrogen Fixation in the Cephalodia of Peltigera aphthosa. New Phytol. 1969, 68, 721–729. [Google Scholar] [CrossRef]
- Hyvärinen, M.; Härdling, R.; Tuomi, J. Cyanobacterial Lichen Symbiosis: The Fungal Partner as an Optimal Harvester. Oikos 2002, 98, 498–504. [Google Scholar] [CrossRef]
- Cornejo, C.; Scheidegger, C. New Morphological Aspects of Cephalodium Formation in the Lichen Lobaria pulmonaria (Lecanorales, Ascomycota). Lichenologist 2013, 45, 77–87. [Google Scholar] [CrossRef]
- Finger-Higgens, R.; Duniway, M.C.; Fick, S.; Geiger, E.L.; Hoover, D.L.; Pfennigwerth, A.A.; Van Scoyoc, M.W.; Belnap, J. Decline in Biological Soil Crust N-Fixing Lichens Linked to Increasing Summertime Temperatures. Proc. Natl. Acad. Sci. USA 2022, 119, e2120975119. [Google Scholar] [CrossRef]
- Raggio, J.; Green, T.G.A.; Crittenden, P.D.; Pintado, A.; Vivas, M.; Pérez-Ortega, S.; De Los Ríos, A.; Sancho, L.G. Comparative Ecophysiology of Three Placopsis Species, Pioneer Lichens in Recently Exposed Chilean Glacial Forelands. Symbiosis 2012, 56, 55–66. [Google Scholar] [CrossRef]
- De los Ríos, A.; Raggio, J.; Pérez-Ortega, S.; Vivas, M.; Pintado, A.; Green, T.G.A.; Ascaso, C.; Sancho, L.G. Anatomical, Morphological and Ecophysiological Strategies in Placopsis pycnotheca (Lichenized Fungi, Ascomycota) Allowing Rapid Colonization of Recently Deglaciated Soils. Flora 2011, 206, 857–864. [Google Scholar] [CrossRef]
- Rai, A.N.; Bergman, B. Cyanolichens. Proc. R. Irish Acad. 2002, 102, 19–22. [Google Scholar] [CrossRef]
- Palmqvist, K.; Dahlman, L.; Valladares, F.; Tehler, A.; Sancho, L.G.; Mattsson, J.E. CO2 Exchange and Thallus Nitrogen across 75 Contrasting Lichen Associations from Different Climate Zones. Oecologia 2002, 133, 295–306. [Google Scholar] [CrossRef]
- Schneider, K.; Resl, P.; Spribille, T. Escape from the Cryptic Species Trap: Lichen Evolution on Both Sides of a Cyanobacterial Acquisition Event. Mol. Ecol. 2016, 25, 3453–3468. [Google Scholar] [CrossRef]
- Grube, M.; Berg, G. Microbial Consortia of Bacteria and Fungi with Focus on the Lichen Symbiosis. Fungal Biol. Rev. 2009, 23, 72–85. [Google Scholar] [CrossRef]
- Almendras, K.; García, J.; Carú, M.; Orlando, J. Nitrogen-Fixing Bacteria Associated with Peltigera Cyanolichens and Cladonia Chlorolichens. Molecules 2018, 23, 3077. [Google Scholar] [CrossRef] [PubMed]
- Swamy, C.T.; Gayathri, D.; Devaraja, T.N.; Bandekar, M.; D’Souza, S.E.; Meena, R.M.; Ramaiah, N. Plant Growth Promoting Potential and Phylogenetic Characteristics of a Lichenized Nitrogen Fixing Bacterium, Enterobacter cloacae. J. Basic Microbiol. 2016, 56, 1369–1379. [Google Scholar] [CrossRef] [PubMed]
- Eymann, C.; Lassek, C.; Wegner, U.; Bernhardt, J.; Fritsch, O.A.; Fuchs, S.; Otto, A.; Albrecht, D.; Schiefelbein, U.; Cernava, T.; et al. Symbiotic Interplay of Fungi, Algae, and Bacteria within the Lung Lichen Lobaria pulmonaria L. Hoffm. as Assessed by State-of-the-Art Metaproteomics. J. Proteome Res. 2017, 16, 2160–2173. [Google Scholar] [CrossRef]
- Shukla, V.; Kumari, R.; Patel, D.K.; Upreti, D.K. Characterization of the Diversity of Mycosporine-like Amino Acids in Lichens from High Altitude Region of Himalaya. Amino Acids 2016, 48, 129–136. [Google Scholar] [CrossRef]
- Sivaramakrishnan, R.; Suresh, S.; Kanwal, S.; Ramadoss, G.; Ramprakash, B.; Incharoensakdi, A. Microalgal Biorefinery Concepts’ Developments for Biofuel and Bioproducts: Current Perspective and Bottlenecks. Int. J. Mol. Sci. 2022, 23, 2623. [Google Scholar] [CrossRef]
- Żymańczyk-Duda, E.; Samson, S.O.; Brzezińska-Rodak, M.; Klimek-Ochab, M. Versatile Applications of Cyanobacteria in Biotechnology. Microorganisms 2022, 10, 2318. [Google Scholar] [CrossRef]
- Amaro, H.M.; Salgado, E.M.; Nunes, O.C.; Pires, J.C.M.; Esteves, A.F. Microalgae Systems-Environmental Agents for Wastewater Treatment and Further Potential Biomass Valorisation. J. Environ. Manag. 2023, 337, 117678. [Google Scholar] [CrossRef] [PubMed]
- Vuppaladadiyam, A.K.; Yao, J.G.; Florin, N.; George, A.; Wang, X.; Labeeuw, L.; Jiang, Y.; Davis, R.W.; Abbas, A.; Ralph, P.; et al. Impact of Flue Gas Compounds on Microalgae and Mechanisms for Carbon Assimilation and Utilization. ChemSusChem 2018, 11, 334–355. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Li, J.; Qian, J.; Wang, B.; Liu, J.; Xu, R.; Chen, P.; Zhou, W. Recent Advances in CO2 Fixation by Microalgae and Its Potential Contribution to Carbon Neutrality. Chemosphere 2023, 319, 137987. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Li, W.; Guo, Y.; Zhang, Z.; Shi, W.; Cui, F.; Lens, P.N.L.; Tay, J.H. Microalgal-Bacterial Consortia: From Interspecies Interactions to Biotechnological Applications. Renew. Sustain. Energy Rev. 2020, 118, 109563. [Google Scholar] [CrossRef]
- Ramanan, R.; Kim, B.-H.; Cho, D.-H.; Oh, H.-M.; Kim, H.-S. Algae-Bacteria Interactions: Evolution, Ecology and Emerging Applications. Biotechnol. Adv. 2016, 34, 14–29. [Google Scholar] [CrossRef]
- Zhou, Z.; Li, Q.; Song, K.; Wang, R.; Wen, S.; Zhang, D.; Cong, W. Exploration of Applying Growth-Promotion Bacteria of Chlorella sorokiniana to Open Cultivation Systems. Bioprocess Biosyst. Eng. 2021, 44, 1567–1576. [Google Scholar] [CrossRef]
- Jin, Z.; Xiao, J.; Ye, H.; Fu, G.; Li, G.; Wu, W.; Li, F. Determination of Nitrogen Sources and Losses in Surface Runoff from Different Lands at a Watershed Scale. Environ. Sci. Pollut. Res. 2023, 23, 26459. [Google Scholar] [CrossRef]
- Xu, L.; Cheng, X.; Wang, Q. Enhanced Lipid Production in Chlamydomonas reinhardtii by Co-Culturing with Azotobacter chroococcum. Front. Plant Sci. 2018, 9, 741. [Google Scholar] [CrossRef]
- Wei, Z.J.; Xiao, L.I.; Wang, H.N.; Yin, Y.H.; Jun, X.L.; Sheng, G.B. Enhanced Biomass Production and Lipid Accumulation by Co-Cul-Tivation of Chlorella vulgaris with Azotobacter mesorhizobium sp. China Biotechnol. 2019, 39, 56–64. [Google Scholar]
- Silaban, A.; Bai, R.; Gutierrez-Wing, M.T.; Negulescu, I.I.; Rusch, K.A. Effect of Organic Carbon, C: N Ratio and Light on the Growth and Lipid Productivity of Microalgae/Cyanobacteria Coculture. Eng. Life Sci. 2014, 14, 47–56. [Google Scholar] [CrossRef]
- Padmaperuma, G.; Kapoore, R.V.; Gilmour, D.J.; Vaidyanathan, S. Microbial Consortia: A Critical Look at Microalgae Co-Cultures for Enhanced Biomanufacturing. Crit. Rev. Biotechnol. 2018, 38, 690–703. [Google Scholar] [CrossRef]
- Kim, M.S.; Baek, J.S.; Yun, Y.S.; Jun Sim, S.; Park, S.; Kim, S.C. Hydrogen Production from Chlamydomonas reinhardtii Biomass Using a Two-Step Conversion Process: Anaerobic Conversion and Photosynthetic Fermentation. Int. J. Hydrogen Energy 2006, 31, 812–816. [Google Scholar] [CrossRef]
- Melis, A.; Melnicki, M.R. Integrated Biological Hydrogen Production. Int. J. Hydrogen Energy 2006, 31, 1563–1573. [Google Scholar] [CrossRef]
- Fakhimi, N.; Gonzalez-Ballester, D.; Fernández, E.; Galván, A.; Dubini, A. Algae-Bacteria Consortia as a Strategy to Enhance H2 Production. Cells 2020, 9, 1353. [Google Scholar] [CrossRef]
- Oruganti, R.K.; Katam, K.; Show, P.L.; Gadhamshetty, V.; Upadhyayula, V.K.K.; Bhattacharyya, D. A Comprehensive Review on the Use of Algal-Bacterial Systems for Wastewater Treatment with Emphasis on Nutrient and Micropollutant Removal. Bioengineered 2022, 13, 10412–10453. [Google Scholar] [CrossRef]
- Dong, H.; Liu, W.; Zhang, H.; Wang, Z.; Feng, F.; Zhou, L.; Duan, H.; Xu, T.; Li, X.; Ma, J. Enhanced Biomass Production and Wastewater Treatment in Attached Co-Culture of Chlorella pyrenoidosa with Nitrogen-Fixing Bacteria Azotobacter beijerinckii. Bioprocess Biosyst. Eng. 2023, 46, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Osorio-Reyes, J.G.; Valenzuela-Amaro, H.M.; Pizaña-Aranda, J.J.P.; Ramírez-Gamboa, D.; Meléndez-Sánchez, E.R.; López-Arellanes, M.E.; Castañeda-Antonio, M.D.; Coronado-Apodaca, K.G.; Gomes Araújo, R.; Sosa-Hernández, J.E.; et al. Microalgae-Based Biotechnology as Alternative Biofertilizers for Soil Enhancement and Carbon Footprint Reduction: Advantages and Implications. Mar. Drugs 2023, 21, 93. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Hu, B.; Zheng, C.; Deng, Z.; Duan, J.; Qin, K.; Cui, X.; Li, S. Application of Anabaena azotica- And Chlorella pyrenoidosa-Based Algal Biotechnology in Green Production of Algae-Rich Crataegi fructus. J. Healthc. Eng. 2022, 2022, 424890. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).