Changes in Organic Carbon Stock in Soil and Whole Tree Biomass in Afforested Areas in Latvia
Abstract
1. Introduction
2. Results
2.1. Soil Bulk Density
2.2. Soil Organic Carbon
2.2.1. Soil Organic Carbon Concentration
2.2.2. Soil Organic Carbon Stock
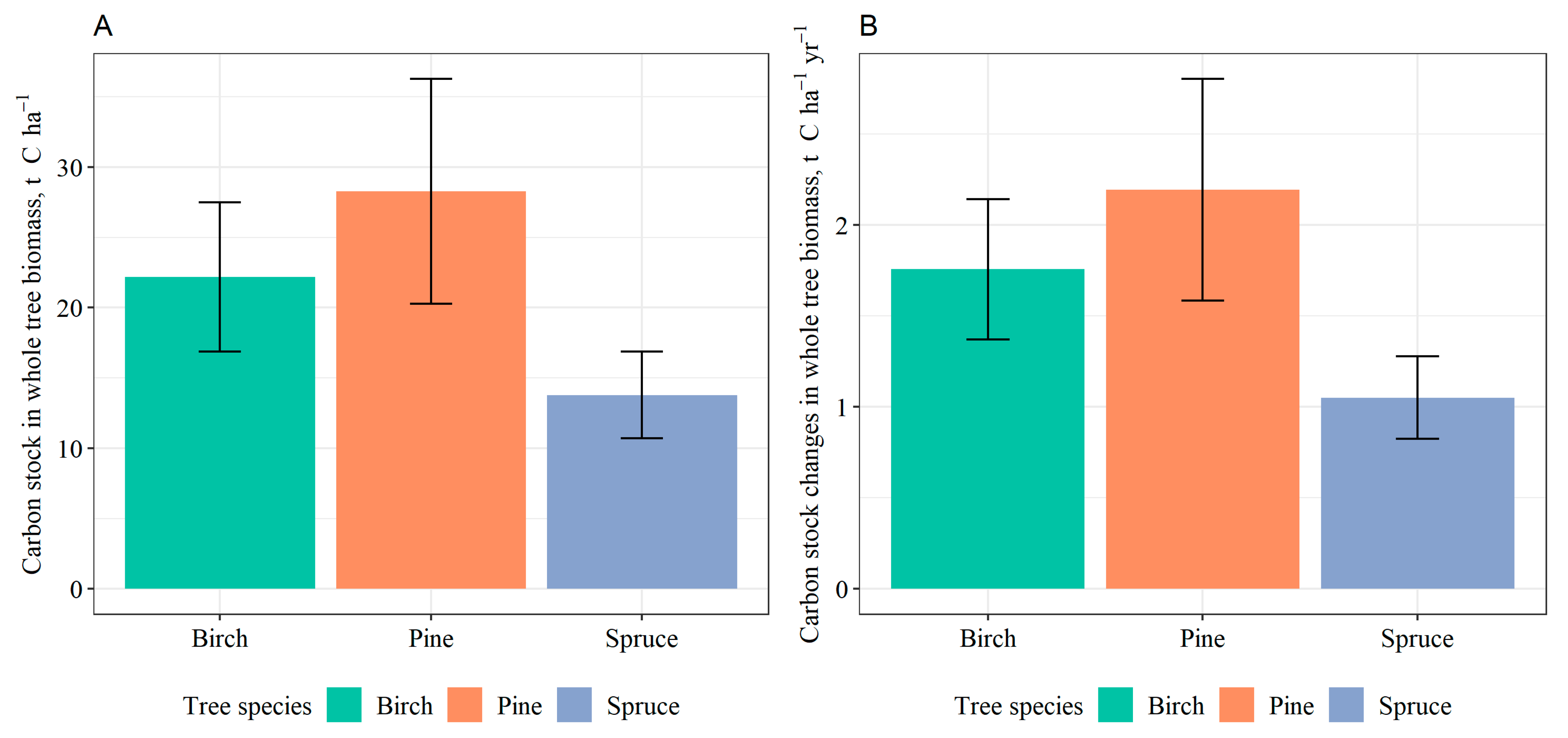
2.3. Tree Biomass
2.4. Total Impact of Afforestation on C Stock in Soil and Tree Biomass
3. Discussion
3.1. Soil Bulk Density
3.2. Soil Organic Carbon Concentration and Stock
3.3. Carbon Stock in Tree Biomass
4. Materials and Methods
4.1. Research Sites
4.2. Soil Sampling and Analyses
4.3. Tree Biomass
- y—dry biomass fraction, kg
- H—stem height, m
- D—diameter at breast height, cm
- a, b, c, d, e—equation parameters
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Climate Change: Better Using EU Forests as Carbon Sinks. Available online: https://www.europarl.europa.eu/news/en/headlines/society/20170711STO79506/climate-change-better-using-eu-forests-as-carbon-sinks (accessed on 2 May 2023).
- Climate Action. Forests and Agriculture. Available online: https://climate.ec.europa.eu/eu-action/forests-and-agriculture_en (accessed on 2 May 2023).
- Latvia. 2022 National Inventory Report (NIR). Available online: https://unfccc.int/documents/461908 (accessed on 2 May 2023).
- Zhang, X.; Adamowski, J.F.; Deo, R.C.; Xu, X.; Zhu, G.; Cao, J. Effects of Afforestation on Soil Bulk Density and pH in the Loess Plateau, China. Water 2018, 10, 1710. [Google Scholar] [CrossRef]
- Podrázský, V.; Holubík, O.; Vopravil, J.; Khel, T.; Moser, W.; Prknová, H. Effects of afforestation on soil structure formation in two climatic regions of the Czech Republic. J. For. Sci. 2015, 61, 225–234. [Google Scholar] [CrossRef]
- Gao, Y.; Cheng, J.; Ma, Z.; Zhao, Y.; Su, J. Carbon storage in biomass, litter, and soil of different plantations in a semiarid temperate region of northwest China. Ann. For. Sci. 2014, 71, 427–435. [Google Scholar] [CrossRef]
- Jandl, R.; Lindner, M.; Vesterdal, L.; Bauwens, B.; Baritz, R.; Hagedorn, F.; Johnson, D.W.; Minkkinen, K.; Byrne, K.A. How strongly can forest management influence soil carbon sequestration? Geoderma 2007, 137, 253–268. [Google Scholar] [CrossRef]
- Valente, F.D.A.; Castro, M.F.; Filho, J.F.L.; Lopes, A.T.; Borges, S.R.; Gomes, L.C.; Neves, J.C.L.; Silva, I.R.; Oliveira, T.S. Soil Use and Management Pollution, Soil Remediation and Recovery of Degraded Areas Litterfall production, decomposition and litter nutrient contents in a mined area revegetated with different forest species. Bras. Cienc. Solo. 2023, 47, e0220122. [Google Scholar] [CrossRef]
- Qiu, L.; Xiao, T.; Bai, T.; Mo, X.; Huang, J.; Deng, W.; Liu, Y. Seasonal Dynamics and Influencing Factors of Litterfall Production and Carbon Input in Typical Forest Community Types in Lushan Mountain, China. Forests 2023, 14, 341. [Google Scholar] [CrossRef]
- Lei, X.; Shen, Y.; Zhao, J.; Huang, J.; Wang, H.; Yu, Y.; Xiao, C. Root Exudates Mediate the Processes of Soil Organic Carbon Input and Efflux. Plants 2023, 12, 630. [Google Scholar] [CrossRef]
- Trivedi, P.; Singh, B.P.; Singh, B.K. Chapter 1—Soil Carbon: Introduction, Importance, Status, Threat, and Mitigation. In Soil Carbon Storage, 1st ed.; Singh, B.K., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 1–28. [Google Scholar]
- Post, W.M.; Kwon, K.C. Soil carbon sequestration and land-use change: Processes and potential. Glob. Chang. Biol. 2000, 6, 317–327. [Google Scholar] [CrossRef]
- Del Galdo, I.; Six, J.; Peressotti, A.; Cotrufo, M.F. Assessing the impact of land-use change on soil C sequestration in agricultural soils by means of organic matter fractionation and stable C isotopes. Glob. Chang. Biol. 2003, 9, 1204–1213. [Google Scholar] [CrossRef]
- Six, J.; Elliott, E.T.; Paustian, K. Soil macroaggregate turnover and microaggregate formation: A mechanism for C sequestration under no-tillage agriculture. Soil Biol. Biochem. 2000, 32, 2099–2103. [Google Scholar] [CrossRef]
- DeGryze, S.; Six, J.; Paustian, K.; Morris, S.J.; Paul, E.A.; Merckx, R. Soil organic carbon pool changes following land-use conversions. Glob. Chang. Biol. 2004, 10, 1120–1132. [Google Scholar] [CrossRef]
- Smal, H.; Olszewska, M. The effect of afforestation with Scots pine (Pinus sylvestris L.) of sandy post-arable soils on their selected properties. II. Reaction, carbon, nitrogen and phosphorus. Plant Soil 2008, 305, 171–187. [Google Scholar] [CrossRef]
- Varnagirytė-Kabašinskienė, I.; Žemaitis, P.; Armolaitis, K.; Stakėnas, V.; Urbaitis, G. Soil organic carbon stocks in afforested agricultural land in Lithuanian hemiboreal forest zone. Forests 2021, 12, 1562. [Google Scholar] [CrossRef]
- Zhang, W.; Yang, K.; Lyu, Z.; Zhu, J. Microbial groups and their functions control the decomposition of coniferous litter: A comparison with broadleaved tree litters. Soil Biol. Biochem. 2019, 133, 196–207. [Google Scholar] [CrossRef]
- Speckert, T.C.; Wiesenberg, G.L.B. Alterations of soil organic matter following 130 years of afforestation assessed by molecular markers. In Proceedings of the EGU General Assembly 2023, Vienna, Austria, 24–28 April 2023. [Google Scholar]
- Lobsey, C.R.; Viscarra Rossel, R.A. Sensing of soil bulk density for more accurate carbon accounting. Eur. J. Soil Sci. 2016, 67, 504–513. [Google Scholar] [CrossRef]
- Indoria, A.K.; Sharma, K.L.; Reddy, K.S. Chapter 18—Hydraulic properties of soil under warming climate. In Climate Change and Soil Interactions, 1st ed.; Prasad, M.N.V., Pietrzykowski, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 473–508. [Google Scholar]
- Yao, W.; Nan, F.; Li, Y.; Li, Y.; Liang, P.; Zhao, C. Effects of Different Afforestation Years on Soil Properties and Quality. Forests 2023, 14, 329. [Google Scholar] [CrossRef]
- Vesterdal, L.; Rosenqist, L.; Salms, C.; Hansen, K.; Groenenberg, B.J.; Johansson, M.B. Carbon sequestration in soil and biomass following afforestation: Experiences from Oak and Norway spruce chronosequences in Denmark, Sweden and the Netherlands. In Environmental Effects of Afforestation in North-Western Europe; Heil, G.W., Muys, B., Hansen, K., Eds.; Kluwer Academic Publishers: Amsterdam, The Netherlands, 2007; pp. 19–51. [Google Scholar]
- De Vries, W.; Reinds, G.J.; Posch, M.; Sanz, M.; Krause, G.; Calatyud, V.; Dupouey, J.; Sterba, H.; Gundersen, P.; Voogd, J.; et al. Intensive Monitoring of Forest Ecosystems in Europe; Technical Report; European Commission UN/ECE: Brussels, Belgium, 2003. [Google Scholar]
- Paul, K.I.; Larmour, J.; Specht, A.; Zerihun, A.; Ritson, P.; Roxburgh, S.; Sochachi, S.; Lewis, T.; Barton, C.; England, J.R.; et al. Testing the generality of below-ground biomass allometry across plant functional types at the continent scale. For. Ecol. Manag. 2019, 432, 102–114. [Google Scholar] [CrossRef]
- Paul, K.I.; England, J.R.; Roxburgh, H.S. Carbon dynamics in tree plantings: How changes in woody biomass impact litter and soil carbon. For. Ecol. Manag. 2022, 521, 120406. [Google Scholar] [CrossRef]
- Liepiņš, J.; Lazdiņš, A.; Kalēja, S.; Liepiņš, K. Species Composition Affects the Accuracy of Stand-Level Biomass Models in Hemiboreal Forests. Land 2022, 11, 1108. [Google Scholar] [CrossRef]
- Pati, P.K.; Kaushik, P.; Khan, M.L.; Khare, P.K. Allometric equations for biomass and carbon stock estimation of small diameter woody species from tropical dry deciduous forests: Support to REDD+. Trees For. People 2022, 9, 100289. [Google Scholar] [CrossRef]
- Wellock, M.L.; LaPerle, C.M.; Kiely, G. What is the impact of afforestation on the carbon stocks of Irish mineral soils? For. Ecol. Manag. 2011, 262, 1589–1596. [Google Scholar] [CrossRef]
- Morris, S.J.; Bohm, S.; Haile-Mariam, S.; Paul, E.A. Evaluation of carbon accrual in afforested agricultural soils. Glob. Chang. Biol. 2007, 13, 1145–1156. [Google Scholar] [CrossRef]
- Kirschbaum, M.U.F.; Guo, L.B.; Gifford, R.M. Why does rainfall affect the trend in soil carbon after converting pastures to forests? A possible explanation based on nitrogen dynamics. For. Ecol. Manag. 2008, 255, 2990–3000. [Google Scholar] [CrossRef]
- Augusto, L.; De Schrijver, A.; Vesterdal, L.; Smolander, A.; Prescott, C.; Ranger, J. Influences of evergreen gymnosperm and deciduous angiosperm tree species on the functioning of temperate and boreal forests. Biol. Rev. 2015, 90, 444–466. [Google Scholar] [CrossRef] [PubMed]
- Dawud, S.M.; Raulund-Rasmussen, K.; Domisch, T.; Finér, L.; Jaroszewic, B.; Vesterdal, L. Is tree species diversity or species identity the more important driver of soil carbon stocks, C/N ratio, and pH? Ecosystems 2016, 19, 645–660. [Google Scholar] [CrossRef]
- Alakukku, L. Subsoil compaction due to wheel traffic. Rev. Agric. Food Sci. Finl. 1999, 8, 333–351. [Google Scholar] [CrossRef]
- Soane, B.D.; Blackwell, P.S.; Dickson, J.W.; Painter, D.J. Compaction by agricultural vehicles: A review. II. Compaction under tyres and other running gear. Soil Tillage Res. 1981, 1, 373–400. [Google Scholar] [CrossRef]
- Suzuki, L.E.A.S.; Reinert, D.J.; Fenner, P.T.; Secco, D.; Reichert, J.M. Prevention of additional compaction in eucalyptus and pasture land uses, considering soil moisture and bulk density. J. S. Am. Earth Sci. 2022, 120, 104113. [Google Scholar] [CrossRef]
- Wang, W.-J.; Qiu, L.; Zu, Y.-G.; Su, D.-X.; An, J.; Wang, H.-Y.; Zheng, G.-Y.; Sun, W.; Chen, X.-Q. Changes in soil organic carbon, nitrogen, pH and bulk density with the development of larch (Larix gmelinii) plantations in China. Glob. Chang. Biol. 2011, 17, 2657–2676. [Google Scholar]
- Mongil-Manso, J.; Navarro-Hevia, J.; San Martín, R. Impact of Land Use Change and Afforestation on Soil Properties in a Mediterranean Mountain Area of Central Spain. Land 2022, 11, 1043. [Google Scholar] [CrossRef]
- Wall, A.; Heiskanen, J. Water-retention characteristics and related physical properties of soil on afforested agricultural land in Finland. For. Ecol. Manag. 2003, 186, 21–32. [Google Scholar] [CrossRef]
- Wall, A.; Hytönen, J. Soil fertility of afforested arable land compared to continuously. Plant Soil 2005, 275, 247–260. [Google Scholar] [CrossRef]
- Olsson, B.A.; Guedes, B.S.; Dahlin, A.S.; Hyvönen, R. Predicted long-term effects of decomposition of leaf litter from Pinus taeda, Eucalyptus cloeziana and deciduous miombo trees on soil carbon stocks. Glob. Ecol. Conserv. 2019, 17, e00587. [Google Scholar] [CrossRef]
- Cornwell, W.K.; Cornelissen, J.H.; Amatangelo, K.; Dorrepaal, E.; Eviner, V.T.; Godoy, O.; Hobbie, S.E.; Hoorens, B.; Kurokawa, H.; Harguindeguy, N.P.; et al. Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol. Lett. 2008, 11, 1065–1071. [Google Scholar] [CrossRef]
- Hou, G.; Delang, C.O.; Lu, X.; Gao, L. A meta-analysis of changes in soil organic carbon stocks after afforestation with deciduous broadleaved, sempervirent broadleaved, and conifer tree species. Ann. For. Sci. 2020, 77, 92. [Google Scholar] [CrossRef]
- Berthrong, S.T.; Jobbágy, E.G.; Jackson, R.B. A global meta-analysis of soil exchangeable cations, pH, carbon, and nitrogen with afforestation. Ecol. Appl. 2009, 19, 2228–2241. [Google Scholar] [CrossRef]
- Laganiére, J.; Angers, D.A.; Paré, D. Carbon accumulation in agricultural soils after afforestation: A meta-analysis. Glob. Chang. Biol. 2010, 16, 439–453. [Google Scholar] [CrossRef]
- Osman, H.E.M.; Elaidarous, A.A.; El-Morsy, M.H.; Eid, E.M.; Keshta, A.E. Soils with more clay and dense vegetation were rich in soil carbon along Wadi Al-Sharaea, Makkah, Saudi Arabia. Heliyon 2023, 9, e12988. [Google Scholar] [CrossRef]
- Strand, L.T.; Fjellstad, W.; Jackson-Blake, L.; De Wit, H.A. Afforestation of a pasture in Norway did not result in higher soil carbon, 50 years after planting. Landsc. Urban Plan. 2021, 207, 104007. [Google Scholar] [CrossRef]
- Poeplau, C.; Don, A.; Vesterdal, L.; Leifeld, J.; van Wesemael, B.A.; Schumacher, J.; Gensior, A. Temporal dynamics of soil organic carbon after land-use change in the temperate zone–carbon response functions as a model approach. Glob. Chang. Biol. 2011, 17, 2415–2427. [Google Scholar] [CrossRef]
- Guo, Y.; Abdalla, M.; Espenberg, M.; Hastings, A.; Hallett, P.; Smith, P. A systematic analysis and review of the impacts of afforestation on soil quality indicators as modified by climate zone, forest type and age. Sci. Total Environ. 2021, 757, 143824. [Google Scholar] [CrossRef] [PubMed]
- Davis, M. Soil properties under pine forest and pasture at two hill country sites in Canterbury. N. Z. J. For. Sci. 2001, 31, 3–17. [Google Scholar]
- Vesterdal, L.; Ritter, E.; Gundersen, P. Change in soil organic carbon following afforestation of former arable land. For. Ecol. Manag. 2002, 169, 137–147. [Google Scholar] [CrossRef]
- Guo, L.B.; Gifford, R. Soil carbon stocks and land use change: A meta analysis. Glob. Chang. Biol. 2002, 8, 345–360. [Google Scholar] [CrossRef]
- Chen, G.; Zhang, M.; Yao, X.; Zhu, Y.; Hu, Y.; Hui, D.; Li, J.; Chen, J.; Deng, Q. Soil Organic Carbon Stock, Source, and Stability after 20-Year Mangrove Afforestation in Southern China. Available online: https://ssrn.com/abstract=4421376 (accessed on 5 June 2023).
- Yu, P.; Li, Y.; Liu, S.; Liu, J.; Ding, Z.; Ma, M.; Tang, X. Afforestation influences soil organic carbon and its fractions associated with aggregates in a karst region of Southwest China. Sci. Total Environ. 2022, 814, 152710. [Google Scholar] [CrossRef]
- Romanyà, J.; Cortina, J.; Falloon, P.; Coleman, K.; Smith, P. Modelling changes in soil organic matter after planting fast-growing Pinus radiata on Mediterranean agricultural soils. Eur. J. Soil Sci. 2000, 51, 627–641. [Google Scholar]
- Davis, M.R.; Condron, L.M. Impact of grassland afforestation on soil carbon in New Zealand: A review of paired-site studies. Aust. J. Soil Res. 2002, 40, 675–690. [Google Scholar] [CrossRef]
- Paul, K.I.; Polglase, P.J.; Nyakuengama, J.G.; Khanna, P.K. Change in soil carbon following afforestation. For. Ecol. Manag. 2002, 168, 241–257. [Google Scholar] [CrossRef]
- Turner, J.; Lambert, M.J.; Johnson, D.W. Experience with patterns of change in soil carbon resulting from forest plantation establishment in eastern Australia. For. Ecol. Manag. 2005, 220, 259–269. [Google Scholar] [CrossRef]
- Mao, R.; Zeng, D.-H.; Hu, Y.-L.; Li, L.-J.; Yang, D. Soil organic carbon and nitrogen stocks in an age-sequence of poplar stands planted on marginal agricultural land in Northeast China. Plant Soil 2010, 332, 277–287. [Google Scholar] [CrossRef]
- Paul, K.L.; Polglase, P.J.; Richards, G.P. Predicted change in soil carbon following afforestation or reforestation, and analysis of controlling factors by linking a C accounting model (CAMFor) to models of forest growth (3PG), litter decomposition (GENDEC) and soil C turnover (RothC). For. Ecol. Manag. 2003, 177, 485–501. [Google Scholar] [CrossRef]
- Risch, A.C.; Jurgensen, M.F.; Page-Dumroese, D.S.; Wildi, O.; Schütz, M. Long-term development of above- and below-ground carbon stocks following land-use change in subalpine ecosystems of the Swiss National Park. Can. J. For. Res. 2008, 38, 1590–1602. [Google Scholar] [CrossRef]
- Mälkönen, E. Annual primary production and nutrient cycle in some Scots pine stands. Commun. Inst. For. Fenn. 1974, 84, 1–87. [Google Scholar]
- Havas, P.; Kubin, E. Structure, growth and organic matter content in the vegetation cover of an old spruce forest in northern Finland. Ann. Bot. Fenn. 1983, 20, 115–149. [Google Scholar]
- Lakida, P.; Nilsson, S.; Shvidenko, A. Estmation of forest phytomass for selected countries of the former European U.S.S.R. Biomass Bioenergy 1996, 11, 371–382. [Google Scholar] [CrossRef]
- Pussinen, A.; Karjalainen, T.; Kellomäki, S. Potential contribution of the forest sector to carbon sequestration in Finland. Biomass Bioenergy 1997, 13, 377–387. [Google Scholar] [CrossRef]
- Shen, X.; Jiang, M.; Lu, X.; Liu, X.; Liu, B.; Zhang, J.; Wang, X.; Tong, S.; Lei, G.; Wang, S.; et al. Aboveground biomass and its spatial distribution pattern of herbaceous marsh vegetation in China. Sci. China Earth Sci. 2021, 64, 1115–1125. [Google Scholar] [CrossRef]
- Mao, P.; Qin, L.; Hao, M.; Zhao, W.; Luo, J.; Qiu, X.; Xu, L.; Xiong, Y.; Ran, Y.; Yan, C.; et al. An improved approach to estimate above-ground volume and biomass of desert shrub communities based on UAV RGB images. Ecol. Indic. 2021, 125, 107494. [Google Scholar] [CrossRef]
- Bārdule, A.; Petaja, G.; Butlers, A.; Purviņa, D.; Lazdiņš, A. Estimation of litter input in hemi-boreal forests with drained organic soils for improvement of GHG inventories. Balt. For. 2021, 27, 534. [Google Scholar] [CrossRef]
- Latvian Environment, Geology and Meteorology Centre. Climate Portal. Available online: https://klimats.meteo.lv/klimats/latvijas_klimats/ (accessed on 30 April 2023).
- Nacionālais Meža Monitorings. Available online: https://www.silava.lv/petnieciba/nacionalais-meza-monitorings (accessed on 1 May 2023).
- Cools, N.; De Vos, B. Part X: Sampling and Analysis of Soil. Version 2020-1. In UNECE ICP Forests Programme Co-Ordinating Centre (Ed.): Manual on Methods and Criteria for Harmonized Sampling, Assessment, Monitoring and Analysis of the Effects of Air Pollution on Forests; Thünen Institute of Forest Ecosystems: Eberswalde, Germany, 2020; 101 p. + Annex; Available online: https://www.icp-forests.org/pdf/manual/2020/ICP_Manual_part10_2020_Soil_version_2020-1.pdf (accessed on 1 May 2023).
- Kārkliņš, A. Augsnes Diagnostika un Apraksts; Latvia University of Agriculture: Jelgava, Latvia, 2008. [Google Scholar]
- Liepiņš, J.; Lazdiņš, A.; Liepiņš, K. Equations for estimating above- and belowground biomass of Norway spruce, Scots pine, birch spp. and European aspen in Latvia. Scand. J. For. Res. 2018, 33, 58–70. [Google Scholar] [CrossRef]
- Mežsaimniecisko Darbību Ietekme uz Siltumnīcefekta Gāzu Emisijām un CO2 (2011–2015. Gads). Available online: https://www.lvm.lv/petijumi-un-publikacijas/mezsaimniecisko-darbibu-ietekmes-uz-siltumnicefekta-gazu-emisijam-un-co-piesaisti (accessed on 1 May 2023).





| Parameter | Method ISO Reference | Principle | Method Description (Analytical Equipment) |
|---|---|---|---|
| Soil moisture | LVS ISO 11465:2006 | Gravimetry | Air-dried and treated soil samples are dried at 105 °C (Sartorius AX224) |
| Content of carbonates | LVS EN ISO 10693:2014 | Volumetry | Samples are treated with 4M HCl solution and volume of produced CO2 is measured (Eijkelkamp calcimeter) |
| Content of total carbon | LVS ISO 10694:2006 ISO 15178:2000 LVS ISO 13878:1998 | Dry incineration | Dry incineration at 950 °C temperature (Elementar EL Cube) |
| Soil preparation | LVS ISO 11464:2006 | Drying and sieving | Drying at 40 °C temperature and separation of fine fraction for chemical analyses |
| Bulk density | LVS ISO 11272:2017 | Soil cylinder method | Dry mass (dried at 105 °C) of 100 cm3 of undisturbed soil samples |
| Coefficient | Pine | Spruce | Birch | |||
|---|---|---|---|---|---|---|
| Above-Ground Biomass | Below-Ground Biomass | Above-Ground Biomass | Below-Ground Biomass | Above-Ground Biomass | Below-Ground Biomass | |
| a | 2273.58 | 553.59 | 1902.43 | 29,714.37 | 1261.84 | 338.25 |
| b | 62.32 | 21.77 | 33.31 | 131.71 | 43.41 | 17.87 |
| c | 31.65 | 10.75 | 15.62 | 31.92 | 20.89 | 16.86 |
| d | 53.86 | 51.97 | 63.39 | 25.56 | 44.34 | 44.45 |
| e | 18.69 | 14.01 | 22.3 | 11.6 | 15.15 | 12.88 |
| Tree Species | Carbon Content, g C kg−1 | Uncertainty, g C kg−1 | ||
|---|---|---|---|---|
| Above-Ground Biomass | Below-Ground Biomass | Above-Ground Biomass | Below-Ground Biomass | |
| Birch | 520.01 | 519.85 | 0.22 | 1.22 |
| Spruce | 525.59 | 526.07 | 1.15 | 0.62 |
| Pine | 531.17 | 548.08 | 0.43 | 0.97 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petaja, G.; Bārdule, A.; Zalmanis, J.; Lazdiņa, D.; Daugaviete, M.; Skranda, I.; Zvaigzne, Z.A.; Purviņa, D. Changes in Organic Carbon Stock in Soil and Whole Tree Biomass in Afforested Areas in Latvia. Plants 2023, 12, 2264. https://doi.org/10.3390/plants12122264
Petaja G, Bārdule A, Zalmanis J, Lazdiņa D, Daugaviete M, Skranda I, Zvaigzne ZA, Purviņa D. Changes in Organic Carbon Stock in Soil and Whole Tree Biomass in Afforested Areas in Latvia. Plants. 2023; 12(12):2264. https://doi.org/10.3390/plants12122264
Chicago/Turabian StylePetaja, Guna, Arta Bārdule, Juris Zalmanis, Dagnija Lazdiņa, Mudrīte Daugaviete, Ilona Skranda, Zaiga Anna Zvaigzne, and Dana Purviņa. 2023. "Changes in Organic Carbon Stock in Soil and Whole Tree Biomass in Afforested Areas in Latvia" Plants 12, no. 12: 2264. https://doi.org/10.3390/plants12122264
APA StylePetaja, G., Bārdule, A., Zalmanis, J., Lazdiņa, D., Daugaviete, M., Skranda, I., Zvaigzne, Z. A., & Purviņa, D. (2023). Changes in Organic Carbon Stock in Soil and Whole Tree Biomass in Afforested Areas in Latvia. Plants, 12(12), 2264. https://doi.org/10.3390/plants12122264






