Examination of the Effects of Domestic Water Buffalo (Bubalus bubalis) Grazing on Wetland and Dry Grassland Habitats
Abstract
1. Introduction
- (1)
- Whether grazing by water buffalo (Bubalus bubalis) can be applicable for the control of shrub encroachment?
- (2)
- How does grazing by water buffalo (Bubalus bubalis) affect the species composition and structure of the grasslands in a typical Pannonian dry grassland, a typical wet fen and a grassland in a mid-mountain area, mainly agronomically (turf management)?
- (3)
- Is grazing by water buffalo (Bubalus bubalis) effective for the suppression of the invasive Solidago gigantea?
2. Results
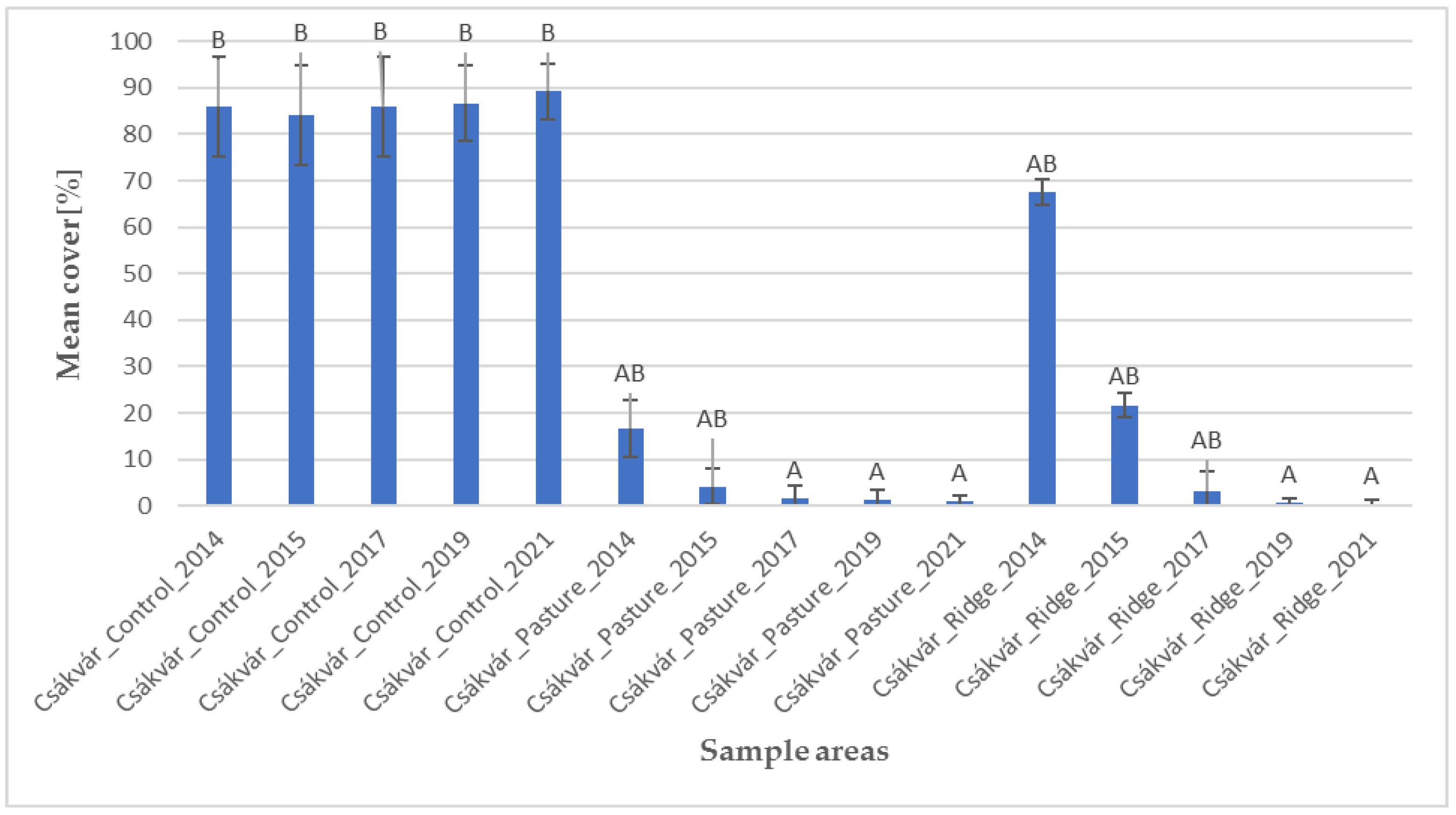
2.1. Species Composition and Structure of Vegetation
2.2. Results of Biomass
3. Discussion
4. Materials and Methods
4.1. Data Collection and Surveyed Areas
- Control, which is left untreated and has a high proportion of individuals of the invasive species;
- Pasture is a fen grassland which is grazed by domestic water buffalo (Bubalus bubalis) since 2013 (stocking rate is 2 buffalo/ha) to manage the cover of the invasive species in order to restore Molinia meadows;
- Ridge is an area where ridge planting used to be practiced and recently has been restored through grazing by buffalo since 2013 (stocking rate is 1 buffalo/ha) to manage the area in order to restore typic Pannonian dry grassland.
- I. sample area was grazed by water buffalo for 2 years;
- II. sample area was grazed by water buffalo for 4 years;
- III. sample area was grazed by water buffalo for 6 years.
- The period of grazing was from the 24 April to the 5 November in the sample areas.
4.2. Analysis of Biomassa
- P: yield [Kg/ha]
- M: grass height [cm]
- s: stubble height [cm]
- BM: grass 400 [kg/ha]; alfalfa 470 [kg/ha]
- b: coverage [%]
4.3. Functional Groups
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tälle, M.; Deák, B.; Poschlod, P.; Valkó, O.; Vesterberg, L.; Milberg, P. Grazing vs. mowing: A meta-analysis of biodiversity benefits for grassland management. Agric. Ecosyst. Environ. 2016, 222, 200–212. [Google Scholar] [CrossRef]
- Klimek, S.; Gen-Kemmermann, A.R.; Hofmann, M.; Isselstein, J. Plant species richness and composition in managed grasslands: The relative importance of field management and environmental factors. Biol. Conserv. 2007, 134, 559–570. [Google Scholar] [CrossRef]
- Török, P.; Kelemen, A.; Valkó, O.; Deák, B.; Lukács, B.; Tóthmérész, B. Lucerne dominated fields recover native grass diversity without intensive management actions. J. Appl. Ecol. 2011, 48, 257–264. [Google Scholar] [CrossRef]
- Török, P.; Penksza, K.; Tóth, E.; Kelemen, A.; Sonkoly, J.; Tóthmérész, B. Vegetation type and grazing intensity jointly shape grazing on grassland biodiversity. Ecol. Evol. 2018, 8, 10326–10335. [Google Scholar] [CrossRef] [PubMed]
- Pywell, R.F.; Bullock, J.M.; Hopkins, A.; Walker, K.J.; Sparks, T.H.; Burke, M.J.W.; Peel, S. Restoration of species, rich grassland on arable land: Assessing the limiting processes using a multi, site experiment. J. Appl. Ecol. 2002, 39, 294–309. [Google Scholar] [CrossRef]
- Szentes, S.; Sutyinszki, Z.; Szabó, G.; Zimmermann, Z.; Házi, J.; Wichmann, B.; Hufnágel, L.; Penksza, K.; Bartha, S. Grazed Pannonian grassland beta-diversity changes due to C4 yellow bluestem. Cent. Eur. J. Biol. 2012, 7, 1055–1065. [Google Scholar] [CrossRef]
- Török, P.; Miglécz, T.; Valkó, O.; Kelemen, A.; Deák, B.; Lengyel, S.; Tóthmérész, B. Recovery of native grass biodiversity by sowing on former croplands: Is weed suppression a feasible goal for grassland restoration? J. Nat. Conserv. 2012, 20, 41–48. [Google Scholar] [CrossRef]
- Hajnáczki, S.; Pajor, F.; Péter, N.; Bodnár, Á.; Penksza, K.; Póti, P. Solidago gigantea Ait. and Calamagrostis epigejos (L) Roth invasive plants as potential forage for goats. Not. Bot. Horti Agrobot. Cluj Napoca 2021, 49, 12197. [Google Scholar] [CrossRef]
- Valkó, O.; Török, P.; Deák, B.; Tóthmérész, B. Prospects and limitations of prescribed burning as a management tool in European grasslands. Basic Appl. Ecol. 2014, 15, 26–33. [Google Scholar] [CrossRef]
- Bajor, Z.; Zimmermann, Z.; Szabó, G.; Fehér, Z.; Járdi, I.; Lampert, R.; Kerény-Nagy, V.; Penksza, P.; Szabó, Z.L.; Székely, Z.; et al. Effect of conservation management practices on sand grassland vegetation in Budapest, Hungary. Appl. Ecol. Environ. Res. 2016, 14, 233–247. [Google Scholar] [CrossRef]
- Szentes, S.; Sutyinszki, Z.; Kiss, T.; Fűrész, A.; Saláta, D.; Harkányiné Székely, Z.; Penksza, K. Verges as Fragments of Loess Grasslands in the Carpathian Basin and Their Festuca Species. Diversity 2022, 14, 510. [Google Scholar] [CrossRef]
- Valkó, O.; Török, P.; Tóthmérész, B.; Matus, G. Restoration potential in seed banks of acidic fen and dry-mesophilous meadows: Can restoration be based on local seed banks? Restor. Ecol. 2011, 19, 9–15. [Google Scholar] [CrossRef]
- Valkó, O.; Török, P.; Matus, G.; Tóthmérész, B. Is regular mowing the most appropriate and cost, effective management maintaining diversity and biomass of target forbs in mountain hay meadows? Flora 2012, 207, 303–309. [Google Scholar] [CrossRef]
- Kiss, T.; Lévai, P.; Ferencz, Á.; Szentes, S.; Hufnagel, L.; Nagy, A.; Balogh, Á.; Pintér, O.; Saláta, D.; Házi, J.; et al. Change of composition and diversity of species and grassland management between different grazing intensity in Pannonian dry and wet grasslands. Appl. Ecol. Environ. Res. 2011, 9, 197–230. [Google Scholar] [CrossRef]
- Török, P.; Valkó, O.; Deák, B.; Kelemen, A.; Tóthmérész, B. Traditional cattle grazing in a mosaic alkali landscape: Effects on grassland biodiversity along a moisture gradient. PLoS ONE 2014, 9, e97095. [Google Scholar] [CrossRef]
- Turcsányi-Járdi, I.; Penksza, K.; Saláta-Falusi, E. Vegetation investigation of cattle pastures in the Ipoly Valley, Dejtár. Gyepgazdálkodási Közlemények 2022, 20, 53–54. [Google Scholar] [CrossRef]
- Penksza, K.; Saláta, D. Study on the changes of vegetation composition of the wood pasture near Cserépfalu, Hungary. Gyepgazdálkodási Közlemények 2022, 22, 41–44. [Google Scholar] [CrossRef]
- Marty, J.T. Effects of Cattle Grazing on Diversity in Ephemeral Wetlands. Conserv. Biol. 2005, 19, 1626–1632. [Google Scholar] [CrossRef]
- Fenu, G.; Melis, A.; Pinna, M.S.; Loi, M.C.; Calderisi, G.; Cogoni, D. Impact of Horse Grazing on Floristic Diversity in Mediterranean Small Standing-Water Ecosystems (SWEs). Plants 2022, 11, 1597. [Google Scholar] [CrossRef] [PubMed]
- Póti, P.; Pajor, F.; Láczó, E. Sustainable grazing in small ruminants. Cereal Res. Commun. 2007, 35, 945–948. [Google Scholar] [CrossRef]
- Yayota, M.; Doi, K. Goat Grazing for Restoring, Managing, and Conserving “Satoyama”, a Unique Socio-Ecological Production Landscape. Front. Sustain. Food Syst. 2020, 4, 541721. [Google Scholar] [CrossRef]
- Schindler, S.; Staska, B.; Adam, M.; Rabitsch, W.; Essl, F. Alien species and public health impacts in Europe: A literature review. NeoBiota 2015, 27, 1–23. [Google Scholar] [CrossRef]
- Fenesi, A.; Vágási, C.I.; Beldean, M.; Földesi, R.; Kolcsár, L.-P.; Shapiro, J.T.; Török, E.; Kovács-Hostyánszki, A. Solidago canadensis impact on native plant and pollintor communities in different-aged old fields. Basic Appl. Ecol. 2015, 16, 335–346. [Google Scholar] [CrossRef]
- Vilà, M.; Espinar, J.L.; Hejda, M.; Hulme, P.E.; Jarošík, V.; Maron, J.L.; Pergl, J.; Schaffner, U.; Sun, Y.; Pyšek, P. Ecological impacts of invasive alien plants: A meta-analysis of their effects on species, communities and ecosystems. Ecol. Lett. 2011, 14, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Pyšek, P.; Richardson, D.M.; Rejmánek, M.; Webster, G.L.; Williamson, M.; Kirschner, J. Alien plants in checklists and floras: Towards better communication between taxonomists and ecologists. Taxon 2004, 53, 131–143. [Google Scholar] [CrossRef]
- De la Fuente, B.; Beck, P.S. Invasive species may disrupt protected area networks: Insights from the pine wood nematode spread in Portugal. Forests 2018, 9, 282. [Google Scholar] [CrossRef]
- Demeter, A.; Saláta, D.; Tormáné Kovács, E.; Szirmai, O.; Trenyik, P.; Meinhardt, S.; Rusvai, K.; Verbényiné Neumann, K.; Schermann, B.; Szegleti, Z.; et al. Effects of the Invasive Tree Species Ailanthus altissima on the Floral Diversity and Soil Properties in the Pannonian Region. Land 2021, 10, 1155. [Google Scholar] [CrossRef]
- Holub, P.; Tůma, I.; Záhora, J.; Fiala, K. Different nutrient use strategies of expansive grasses Calamagrostis epigejos and Arrhenatherum elatius. Biologia 2012, 67, 673–680. [Google Scholar] [CrossRef]
- Weber, E.; Jakobs, G. Biological flora of central Europe: Solidago gigantea Aiton. Flora 2004, 200, 109–118. [Google Scholar] [CrossRef]
- Teixeira, L.H.; Yannelli, F.A.; Ganade, G.; Kollmann, J. Functional Diversity and Invasive Species Influence Soil Fertility in Experimental Grasslands. Plants 2020, 9, 53. [Google Scholar] [CrossRef]
- König, J.; Van Kleunen, M.; Dawson, W. No consistent legacy effects of invasion by giant goldenrod (Solidago gigantea) via soil biota on native plant growth. J. Plant Ecol. 2016, 9, 320–327. [Google Scholar] [CrossRef]
- Weber, E.; Jakobs, G. Biological flora of central Europe: Solidago gigantea Aiton. Flora Morphol. Distrib. Funct. Ecol. Plants 2005, 200, 109–118. [Google Scholar] [CrossRef]
- Baličević, R.; Ravlić, M.; Živković, T. Allelopathic Effect of Invasive Species Giant Goldenrod (Solidago gigantea Ait.) on Crops and Weeds. Herbol. Int. J. Weed Res. Control 2015, 1, 19–29. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Cianfaglione, K.; Nagy, D.U.; Canale, A.; Maggi, F. Evaluation of two invasive plant invaders in Europe (Solidago canadensis and Solidago gigantea) as possible sources of botanical insecticides. J. Pest Sci. 2019, 92, 805–821. [Google Scholar] [CrossRef]
- Bobuľská, L.; Demková, L.; Čerevková, A.; Renčo, M. Invasive Goldenrod (Solidago gigantea) Influences Soil Microbial Activities in Forest and Grassland Ecosystems in Central Europe. Diversity 2019, 11, 134. [Google Scholar] [CrossRef]
- Harkes, P.; van Heumen, L.J.M.; van den Elsen, S.J.J.; Mooijman, P.J.W.; Vervoort, M.T.W.; Gort, G.; Holterman, M.H.M.; van Steenbrugge, J.J.M.; Quist, C.W.; Helder, J. Characterization of the Habitat- and Season-Independent Increase in Fungal Biomass Induced by the Invasive Giant Goldenrod and Its Impact on the Fungivorous Nematode Community. Microorganisms 2021, 9, 437. [Google Scholar] [CrossRef]
- Gazoulis, I.; Antonopoulos, N.; Kanatas, P.; Karavas, N.; Bertoncelj, I.; Travlos, I. Invasive Alien Plant Species—Raising Awareness of a Threat to Biodiversity and Ecological Connectivity (EC) in the Adriatic-Ionian Region. Diversity 2022, 14, 387. [Google Scholar] [CrossRef]
- Hall, R.M.; Urban, B.; Kaul, H. The management success of the invasive late goldenrod (Solidago gigantea Aiton.) in a nature conservation area is strongly related to site, control measures and environmental factors. PeerJ 2022, 10, e13161. [Google Scholar] [CrossRef]
- Smart, A.J.; Larson, G.E.; Bauman, P.J. Grass and Canada Goldenrod (Solidago canadensis) competition and implications for managements for management in northern tallgrass. Prairie Nat. 2012, 45, 4–12. [Google Scholar]
- Szépligeti, M.; Körösi, Á.; Szentirmai, I.; Házi, J.; Bartha, D.; Bartha, S. Evaluating alternative mowing regimes for conservation management of Central European mesic hay meadows: A field experiment. Plant Biosyst. 2018, 152, 90–97. [Google Scholar] [CrossRef]
- Nagy, D.U.; Rauschert, E.S.J.; Callaway, R.M.; Henn, T.; Filep, R.; Pal, R.W. Intense mowing management supresses invader, but shifts competitive resistance by a native to facilitation. Restor. Ecol. 2021, 30, e13483. [Google Scholar] [CrossRef]
- Firn, J.; Martin, T.G.; Chadès, I.; Walters, B.; Hayes, J.; Nicol, S.; Carwardine, J. Priority threat management of non-native plants to maintain ecosystem integrity across heterogeneous landscapes. J. Appl. Ecol. 2015, 52, 1135–1144. [Google Scholar] [CrossRef]
- Nagy, D.U.; Stranczinger, S.; Godi, A.; Weisz, A.; Rosche, C.; Suda, J.; Mariano, M.; Pal, R.W. Does higher ploidy level increase the risk of invasion? A case study with two geo-cytotypes of Solidago gigantea Aiton (Asteraceae). J. Plant Ecol. 2018, 11, 317–327. [Google Scholar] [CrossRef]
- Vanderhoeven, S.; Dassonville, N.; Chapuis-Lardy, L.; Hayez, M.; Meerts, P. Impact of the invasive alien plant Solidago gigantea on primary productivity, plant nutrient content and soil mineral nutrient concentration. Plant Soil 2006, 286, 259–268. [Google Scholar] [CrossRef]
- Nagy, D.U.; Rauschert, E.S.; Henn, T.; Cianfaglione, K.; Stranczinger, S.; Pal, R.W. The more we do, the less we gain? Balancing effort and efficacy in managing the Solidago gigantea invasion. Weed Res. 2020, 60, 232–240. [Google Scholar] [CrossRef]
- Lendl, A.; Reznicek, G. Two New Saponins from Solidago gigantea. Sci. Pharm. 2007, 75, 111–120. [Google Scholar] [CrossRef]
- Mietlińska, K.; Przybyt, M.; Kalemba, D. Polish plants as raw materials for cosmetic purposes. Biotechnol. Food Sci. 2019, 83, 95–106. [Google Scholar] [CrossRef]
- Minervino, A.H.H.; Zava, M.; Vecchio, D.; Borghese, A. Bubalus bubalis: A short story. Front. Vet. Sci. 2020, 7, 1–15. [Google Scholar] [CrossRef]
- Mota-Rojas, D.; Braghieri, A.; Álvarez-Macías, A.; Serrapica, F.; Ramírez-Bribiesca, E.; Cruz-Monterrosa, R.; Masucci, F.; Mora-Medina, P.; Napolitano, F. The use of draught animals in rural labour. Animals 2021, 11, 2683. [Google Scholar] [CrossRef]
- Valente, G.F.; Ferraz, G.A.E.S.; Santana, L.S.; Ferraz, P.F.P.; Mariano, D.D.C.; dos Santos, C.M.; Okumura, R.S.; Simonini, S.; Barbari, M.; Rossi, G. Mapping Soil and Pasture Attributes for Buffalo Management through Remote Sensing and Geostatistics in Amazon Biome. Animals 2022, 12, 2374. [Google Scholar] [CrossRef]
- Silva, J.A.R.D.; Rodrigues, L.S.; Lourenço-Júnior, J.D.B.; Alfaia, C.M.; Costa, M.M.; Castro, V.C.G.D.; Bezerra, A.S.; Almeida, A.M.D.; Prates, J.A.M. Total Lipids, Fatty Acid Composition, Total Cholesterol and Lipid-Soluble Antioxidant Vitamins in the longissimus lumborum Muscle of Water Buffalo (Bubalus bubalis) from Different Production Systems of the Brazilian Eastern Amazon. Animals 2022, 12, 595. [Google Scholar] [CrossRef]
- Georgoudis, A.G.; Papanastasis, V.P.; Boyazoglu, J.G. Use of Water Buffalo for Environmental Conservation of Waterland—Review. Asian-Australas. J. Anim. Sci. 1999, 12, 1324–1331. [Google Scholar] [CrossRef]
- Gulickx, M.M.C.; Beecroft, R.C.; Green, A.C. Introduction of water buffalo Bubalus bubalis to recently created wetlands at Kingfishers. Conserv. Evid. 2007, 4, 43–44. [Google Scholar]
- Mihailou, H.; Massaro, M. An overview of the impacts of feral cattle, water buffalo and pigs on the savannas, wetlands and biota of northern Australia. Austral Ecol. 2021, 46, 699–712. [Google Scholar] [CrossRef]
- Ffoulkes, D.; Bamualin, A. Improving the Nutrition Level of Draught Animals Using Available Feeds. In Draught Animals in Rural Development, Proceedings of the International Research Symposium, Cipanas, Indonesia, 3–7 July 1989; Hoffmann, D., Nari, J., Petheram., R.J., Eds.; ACIAR: Canberra, Australia, 1989; pp. 134–145. [Google Scholar]
- Winugroho, M. The Effect of Work Durations on Voluntary Feed Intake and Digestibility, Liveweights and Physiology of Buffaloes Fed a Grass:Rice Straw Diet. In Proceedings of the Congress of the Sixth Federation of Asian Veterinary Associations (FAVA), Depasar, Bali, Indonesia, 16–19 October 1988. [Google Scholar]
- Escarcha, J.F.; Lassa, J.A.; Palacpac, E.P.; Zander, K.K. Understanding Climate Change Impacts on Water Buffalo Production through Farmers’ Perceptions. Clim. Risk Manag. 2018, 20, 50–63. [Google Scholar] [CrossRef]
- Warriach, H.M.; McGill, D.M.; Bush, R.D.; Wynn, P.C.; Chohan, K.R. A Review of Recent Developments in Buffalo Reproduction—A Review. Asian-Australas. J. Anim. Sci. 2015, 28, 451–455. [Google Scholar] [CrossRef]
- Desta, T.T. Introduction of domestic buffalo (Bubalus bubalis) into Ethiopia would be feasible. Renew. Agric. Food Syst. 2012, 27, 305–313. [Google Scholar] [CrossRef]
- Whitmore, J.S. Drought Management on Farmland; Kluwer Academic Publishers: Amsterdam, The Netherlands, 2000; p. 293. [Google Scholar]
- Antkowiak, I.; Pytlewski, J.; Purczynska, A.; Skrzypek, R. A preliminary study of the behaviour of water buffaloes (Bubalus bubalis) imported to Poland. Arch. Tierz. 2012, 55, 415–419. [Google Scholar] [CrossRef]
- Tsiobani, E.T.; Yiakoulaki, M.D.; Hasanagas, N.D.; Papanikolaou, K.C. Water buffaloes’ diet selection grazing at the area of the Lake Kerkini, Northern Greece. J. Mt. Agric. Balk. 2014, 17, 30–40. [Google Scholar]
- Ørskov, E.B. Conferencia silvopastoral systems: Technical, environmental and socio-economic challenges. Pastos 2005, 28, 5–9. [Google Scholar]
- Galloso-Hernández, M.A.; Rodríguez-Estévez, V.; Alvarez-Díaz, C.A.; Soca-Perez, M.; Dublin, D.R.; Iglesias-Gómez, J.; Guelmes, L.S. Selectivity of Leguminous Trees by Water Buffaloes in Semi-intensive Systems. Front. Vet. Sci. 2020, 7, 542338. [Google Scholar] [CrossRef] [PubMed]
- Tsiobani, E.; Yiakoulaki, M.; Menexes, G. Seasonal variation in water buffaloes’ diet grazing in wet grasslands in Northern Greece. Hacquetia 2019, 18, 201–212. [Google Scholar] [CrossRef]
- Phillips, C. Cattle Behaviour and Welfare, 2nd ed.; Blackwell Science: Hoboken, NJ, USA, 1988; p. 274. [Google Scholar]
- Wiegleb, G.; Krawczynski, R. Biodiversity Management by Water Buffalos in Restored Wetlands. Wald. Landsch. Nat. 2010, 10, 17–22. [Google Scholar]
- Napolitano, F.; Pacelli, C.; Grasso, F.; Braghieri, A.; De Rosa, G. The behaviour and welfare of buffaloes (Bubalus bubalis) in modern dairy enterprises. Animal 2013, 7, 1704–1713. [Google Scholar] [CrossRef]
- Jerrentrup, J.S.; Seither, M.; Petersen, U.; Isselstein, J. Little grazer species effect on the vegetation in a rotational grazing system. Agric. Ecosyst. Environ. 2015, 202, 243–250. [Google Scholar] [CrossRef]
- Tóth, E.; Deák, B.; Valkó, O.; Kelemen, A.; Miglécz, T.; Tóthmérész, B.; Török, P. Livestock type is more crucial than grazing intensity: Traditional cattle and sheep grazing in short-grass steppes. Land Degrad. Dev. 2018, 29, 231–239. [Google Scholar] [CrossRef]
- Duncan, P.; Danell, K.; Bergström, R.; Pastor, J. Large Herbivore Ecology, Ecosystem Dynamics and Conservation; Cambridge University Press: Cambridge, UK, 2006; p. 523. [Google Scholar]
- Braithwaite, R. Pandanus: Then and now. Aus. Nat. Hist. 1994, 24, 24–31. [Google Scholar]
- Fundora, O. Performance of river buffaloes (Bubalus bubalis) from Buffalypso breed in feeding systems based on grazing: Fifteen years of research in the Instituto de ciencia animal. Cuban J. Agric. Sci. 2015, 49, 161–171. Available online: http://cjascience.com/index.php/CJAS/issue/view/35 (accessed on 2 March 2023).
- Laca, E.A.; Distal, R.A.; Griggs, T.C.; Demment, M.W. Effects of canopy structure on patch depression by grazers. Ecology 1994, 75, 705–716. [Google Scholar] [CrossRef]
- Perrin, M.R.; Brereton-Stiles, R. Habitat use and feeding behavior of the buffalo and the white rhinoceros in the Hluhluwe-Umfolozi game reserve. S. Afr. J. Wildl. Res. 1999, 29, 72–80. [Google Scholar]
- Wanapat, M.; Phesatcha, K. Rumen adaptation of swamp buffaloes (Bubalus bubalis) by high level of urea supplementation when fed on rice straw-based diet. Trop Anim. Health Product. 2016, 48, 1135–1140. [Google Scholar] [CrossRef]
- Patra, A.K.; Pal, K.; Lalhriatpuii, M. Prediction of nitrogen excretion in buffalo production systems using dietary and animal variables. Agricult Syst. 2020, 182, 102845. [Google Scholar] [CrossRef]
- Tallowin, J.; Rook, A.; Rutter, S. Impact of grazing management on biodiversity of grasslands. Anim. Sci. 2005, 81, 193–198. [Google Scholar] [CrossRef]
- Házi, J.; Purger, D.; Penksza, K.; Bartha, S. Interaction of Management and Spontaneous Succession Suppresses the Impact of Harmful Native Dominant Species in a 20-Year-Long Experiment. Land 2023, 12, 149. [Google Scholar] [CrossRef]
- Braun-Blanquet, J. Pflanzensoziologie; Wien: New York, NY, USA, 1964; pp. 2–865. [Google Scholar]
- Király, G. (Ed.) Új Magyar Füvészkönyv. Magyarország Hajtásos Növényei. Határozókulcsok [New Hungarian Herbal. The Vascular Plants of Hungary. Identification Key]; ANP Igazgatóság: Jósvafő, Hungary, 2009; pp. 3–456. [Google Scholar]
- Balázs, F. A Gyepek Botanikai és Gazdasági Értékelése [Botanical and Economic Assessment of Grasslands]; Mezőgazdasági kiadó: Budapest, Hungary, 1960; pp. 3–28. [Google Scholar]
- Tasi, J.; Bajnok, M.; Sutyinszki, Z.; Szentes, S. Assessing the quality and quantity of green forage with the help of a three-dimensional method. In Proceedings of the 19th International Scientific Symposium on Nutrition of Farm Animals “Zadravec-erjavec Days”, Radenci, Slovenia, 28–29 October 2010; pp. 152–160. [Google Scholar]
- Klapp, E.; Boeker, P.; König, F.; Stählin, A. Wertzahlen der Grünlandpflanzen. Grünland 1953, 2, 38–40. [Google Scholar]
- Briemle, G.; Nitsche, S.; Nitsche, L. Nutzungswertzahlen für Gefäßpflanzen des Grünlandes. Schriftenreihe für Vegetationskunde; Bundesamt für Naturschutz: Bonn, Germany, 2005; pp. 203–225. [Google Scholar]
- Addinsoft XLSTAT 2016: Data Analysis and Statistical Solution for Microsoft Excel. Long Island, NY, USA. Available online: https://www.xlstat.com/fr/ (accessed on 5 March 2023).






| Control Area | |||||
|---|---|---|---|---|---|
| 2014 | 2015 | 2017 | 2019 | 2021 | |
| Total cover (%) * | 116 ±10.13 A ** | 120 ±12.87 A | 122 ±16.01 A | 128 ±11.86 A | 123 ±10.23 A |
| Total yield (t/ha) | 36.4 ±3.87 A | 36.8 ±3.54 A | 40.1 ±5.23 A | 41.7 ±4.24 A | 40.9 ±3.17 A |
| Feed value | −0.47 ±0.37 A | −0.39 ±0.22 A | −0.30 ±0.29 A | −0.26 ±0.25 A | −0.27 ±0.23 A |
| Pasture | |||||
| 2014 | 2015 | 2017 | 2019 | 2021 | |
| Total cover (%) | 50 ±8.15 A | 148 ±47.94 AB | 161 36.97 AB | 183 ±47.20 B | 201 ±58.24 B |
| Total yield (t/ha) | 6.95 ±1.43 A | 25.8 ±6.98 AB | 26.5 ±6.68 B | 27.3 ±7.25 B | 15.7 ±4.17 B |
| Feed value | 1.63 ±0.39 A | 2.05 ±0.28 AB | 2.19 ±0.27 AB | 2.16 ±0.24 AB | 2.30 ±0.16 B |
| Ridge | |||||
| 2014 | 2015 | 2017 | 2019 | 2021 | |
| Total cover (%) | 123 ±9.87 A | 116 ±24.30 A | 144 ±9.33 AB | 154 ±30.22 AB | 170 ±26.56 B |
| Total yield (t/ha) | 28.8 ±1.82 A | 19.7 ±3.33 A | 19.3 ±1.76 A | 19.9 ±3.46 AB | 21.2 ±3.07 B |
| Feed value | −0.62 ±0.21 A | 1.87 ±0.17 AB | 2.47 ±0.29 B | 2.46 ±0.41 B | 2.60 ±0.25 B |
| 2 Years | 4 Years | 6 Years | |
|---|---|---|---|
| Total cover (%) * | 149 ±13.58 A ** | 130 ±19.51 A | 138 ±19.91 A |
| Total yield (t/ha) | 17 ±1.74 A | 14 ±2.03 A | 14.3 ±1.95 A |
| Feed value | 1.20 ±0.27 A | 2.27 ±0.24 AB | 2.64 ±0.30 B |
| Classification | Quality of the Feed Value of Grassland | K-Value |
|---|---|---|
| Class I. | very good | 4< |
| Class II. | good | 3–4 |
| Class III. | medium | 2–3 |
| Class IV. | poor | 1–2 |
| Class V. | bad | 0–1 |
| Economical Useful Grasses | |||||
|---|---|---|---|---|---|
| Agrostis stolonifera L. | Alopecurus pratensis L. | Arrhenatherum elatius (L.) P. Beauv. ex J. Presl & C. Presl | Brachypodium pinnatum (L.) P. Beauv. | Briza media L. | Bromus erectus Huds. |
| Bromus inermis Leyss. | Dactylis glomerata L. | Deschampsia cespitosa (L.) P. Beauv. | Elymus repens (L.) Gould | Festuca arundinacea Schreb. | Festuca ovina L. |
| Festuca pratensis Huds. | Festuca pseudovina Hack. | Festuca rubra L. | Festuca rupicola Heuff. | Festuca valesiaca Schleich. ex Gaudin | Holcus lanatus L. |
| Molinia caerulea (L.) Moench | Phleum pratense L. | Poa angustifolia L. | Poa humilis Ehrh. ex Hoffm. | Sesleria uliginosa Opiz | |
| Economical non-useful grasses | |||||
| Bothriochloa ischaemum (L.) Keng | Bromus arvensis L. | Bromus ramosus Huds. | Bromus sterilis L. | Calamagrostis epigeios (L.) Roth | Danthonia decumbens (L.) DC. |
| Cynodon dactylon (L.) Pers. | Koeleria cristata (L.) Pers. em. Borbás ex Domin | Melica transsilvanica Schur | Phragmites australis (Cav.) Trin. ex Steud. | Stipa pennata L. | |
| Legume species | |||||
| Dorycnium germanicum (Gremli) Rikli | Dorycnium herbaceum Vill. | Genista tinctoria L. | Lathyrus nissolia L. | Lathyrus pratensis L. | Lathyrus tuberosus L. |
| Lotus corniculatus L. | Lotus tenuis Waldst. & Kit. ex Willd. | Medicago falcata L. | Medicago lupulina L. | Ononis spinosa L. | Tetragonolobus maritimus (L.) Roth |
| Trifolium arvense L. | Trifolium montanum L. | Trifolium ochroleucon Huds. | Trifolium pratense L. | Trifolium repens L. | Vicia angustifolia L. |
| Vicia cracca L. | Vicia tetrasperma (L.) Schreb. | ||||
| Invasive plants in Hungary | |||||
| Solidago gigantea Aiton | |||||
| Shrubs | |||||
| Crataegus monogyna Jacq. | Ligustrum vulgare L. | Prunus spinosa L. | Pyrus achras Gaertn. | Rhamnus cathartica L. | Rosa canina L. |
| Rosa gallica L. | Rosa rubiginosa L. | Rubus caesius L. | |||
| Other species | |||||
| Achillea aspleniifolia Vent | Achillea collina Becker ex Rchb | Achillea nobilis L. | Achillea pannonica Scheele | Achillea setacea Waldst. & Kit. | Acinos arvensis (Lam.) Dandy |
| Agrimonia eupatoria L. | Allium angulosum L. | Allium scorodoprasum L. | Anchusa officinalis L. | Angelica sylvestris L. | Arenaria serpyllifolia L. |
| Calystegia sepium (L.) R. Br. | Carduus acanthoides L. | Carex acutiformis Ehrh. | Carex caryophyllea Latourr. | Carex distans L. | Carex disticha Huds. |
| Carex elata All. | Carex flacca Schreb | Carex hirta L. | Carex melanostachya M. Bieb. ex Willd. | Carex panicea L. | Carex praecox Schreb. |
| Carex tomentosa L. | Centaurea jacea L. | Centaurea pannonica (Heuff.) Hayek | Cerastium vulgatum L. | Cichorium intybus L. | Cirsium arvense (L.) Scop. |
| Cirsium canum (L.) All. | Colchicum autumnale L. | Convolvulus arvensis L. | Daucus carota L. | Dipsacus laciniatus L. | Echium vulgare L. |
| Epilobium parviflorum Schreb. | Equisetum arvense L. | Erigeron canadensis L. | Eryngium campestre L. | Eupatorium cannabinum L. | Euphorbia cyparissias L. |
| Euphorbia virgata Waldst. & Kit. | Filipendula vulgaris Moench | Fragaria vesca L. | Galium lucidum All. | Galium mollugo L. | Galium verum L. |
| Gentiana cruciata L. | Geranium columbinum L. | Hieracium bauhinia Schult ex Besser | Hieracium pilosella L. | Hieracium sabaudum L. | Hypericum perforatum L. |
| Inula britannica L. | Inula ensifolia L. | Inula salicina L. | Iris spuria L. | Juncus compressus Jacq. | Juncus effusus L. |
| Juncus inflexus L. | Lepidium campestre (L.) W. T. Aiton | Lepidium draba L. | Linaria vulgaris Mill. | Lychnis coronaria (L.) Desr. | Lycopus europaeus L. |
| Lysimachia nummularia L. | Lysimachia vulgaris L. | Lythrum salicaria L. | Mentha aquatica L. | Mentha longifolia (L.) L. | Mentha pulegium L. |
| Myosotis arvensis (L.) Hill | Odontites rubra (Baumg.) Opiz | Anacamptis morio (L.) R. M. Bateman | Pridgeon & M. W. Chase | Ornithogalum umbellatum L. | Pastinaca sativa L. |
| Pimpinella saxifraga L. | Plantago lanceolata L. | Plantago major L. | Plantago media L. | Plantago maritima L. | Podospermum canum C.A.Mey. |
| Potentilla anserina L. | Potentilla argentea L. | Potentilla reptans L. | Prunella laciniata (L.) L. | Pulicaria dysenterica (L.) Bernh. | Ranunculus acris L. |
| Ranunculus polyanthemos L. | Ranunculus repens L. | Rumex acetosa L. | Rumex stenophyllus Ledeb. | Sanguisorba officinalis L. | Sanguisorba minor Scop. |
| Scirpoides holoschoenus (L.) Soják | Senecio aquaticus Hill | Serratula tinctoria L. | Seseli annuum L. | Silene alba (Mill.) E.H.L.Krause | Silene bupleuroides L. |
| Silene vulgaris (Moench) Garcke | Sonchus arvensis L. | Succisa pratensis Moench | Taraxacum officinale F. H. Wigg. | Thalictrum lucidum L. | Thesium linophyllon L. |
| Thymus glabrescens Willd. | Tussilago farfara L. | Urtica dioica L. | Valerianella dentata (L.) All. | Verbascum blattaria L. | Verbascum phoeniceum L. |
| Veronica arvensis L. | Veronica orchidea Crantz | Veronica prostrata L. | Veronica spicata L. | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fűrész, A.; Penksza, K.; Sipos, L.; Turcsányi-Járdi, I.; Szentes, S.; Fintha, G.; Penksza, P.; Viszló, L.; Szalai, F.; Wagenhoffer, Z. Examination of the Effects of Domestic Water Buffalo (Bubalus bubalis) Grazing on Wetland and Dry Grassland Habitats. Plants 2023, 12, 2184. https://doi.org/10.3390/plants12112184
Fűrész A, Penksza K, Sipos L, Turcsányi-Járdi I, Szentes S, Fintha G, Penksza P, Viszló L, Szalai F, Wagenhoffer Z. Examination of the Effects of Domestic Water Buffalo (Bubalus bubalis) Grazing on Wetland and Dry Grassland Habitats. Plants. 2023; 12(11):2184. https://doi.org/10.3390/plants12112184
Chicago/Turabian StyleFűrész, Attila, Károly Penksza, László Sipos, Ildikó Turcsányi-Járdi, Szilárd Szentes, Gabriella Fintha, Péter Penksza, Levente Viszló, Ferenc Szalai, and Zsombor Wagenhoffer. 2023. "Examination of the Effects of Domestic Water Buffalo (Bubalus bubalis) Grazing on Wetland and Dry Grassland Habitats" Plants 12, no. 11: 2184. https://doi.org/10.3390/plants12112184
APA StyleFűrész, A., Penksza, K., Sipos, L., Turcsányi-Járdi, I., Szentes, S., Fintha, G., Penksza, P., Viszló, L., Szalai, F., & Wagenhoffer, Z. (2023). Examination of the Effects of Domestic Water Buffalo (Bubalus bubalis) Grazing on Wetland and Dry Grassland Habitats. Plants, 12(11), 2184. https://doi.org/10.3390/plants12112184









