Analysis of the Orchidaceae Diversity in the Pululahua Reserve, Ecuador: Opportunities and Constraints as Regards the Biodiversity Conservation of the Cloud Mountain Forest
Abstract
:1. Introduction
2. Materials and Methods
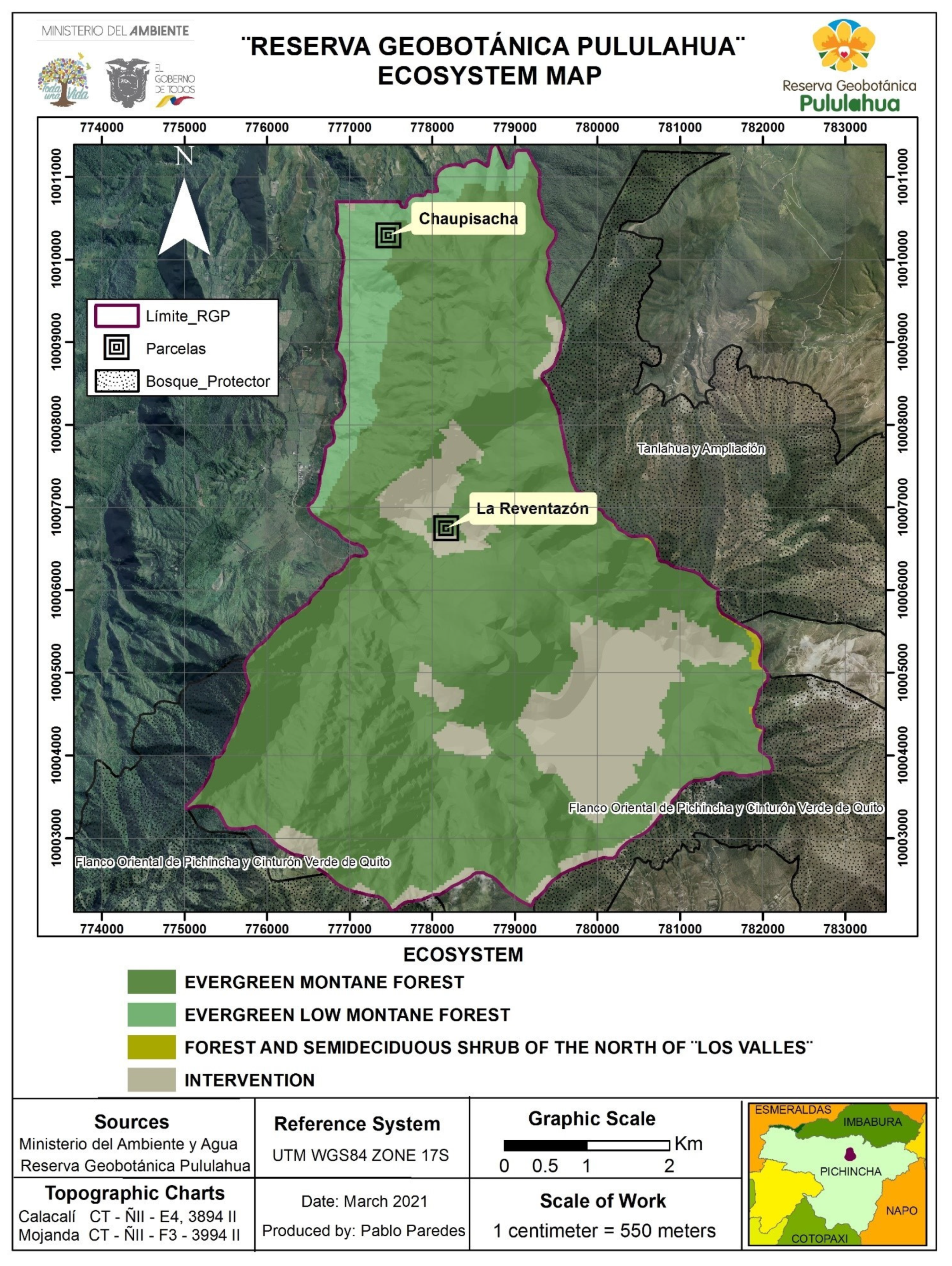
2.1. Study Area
2.2. Data Collection and Analysis
2.3. Diversity
2.4. Similarity and Dissimilarity
- Xij is the number of individuals of species i in location j,
- Xik is the number of individuals of species i in location k,
- and n is the total number of species.
2.5. Conservation of Orchids in Natural Settings Following the IUCN Scale
3. Results
3.1. Diversity of the Forest in the Chaupisacha and La Reventazón Sectors
3.2. Similarity between the Forests in the Chaupisacha and La Reventazón Sectors
3.3. State of Conservation
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Chaupisacha | IUCN Status | Frequency |
|---|---|---|
| Acronia cordata (Ruiz & Pav.) Luer | - | 1 |
| Acronia sp. | - | 2 |
| Byrsella angulata (Rchb. f.) Luer | NT, VU | 2 |
| Corymborkis flava (Sw.) Kuntze | - | 28 |
| Cyclopogon sp. | - | 2 |
| Cyrthochilium sp. | - | 2 |
| Cyrtochilummeirax(Rchb. f.) Dalström | - | 1 |
| Cyrtochilum serratum (Lindl.) Kraenzl. | - | 58 |
| Cyrtochilum tricostatum Kraenzl. | - | 9 |
| Dichaea morrisii Fawc. & Rendle | - | 13 |
| Dracula felix (Luer) Luer | NT, VU, | 15 |
| Elleanthuscapitatus(R. Br.) Cogn. | - | 6 |
| Elleanthus gastroglottis Schltr. | - | 3 |
| Epidendrum ornithoglossum Schltr. | CR, VU, | 7 |
| Epidendrum embreei Dodson | - | 209 |
| Epidendrum fimbriatum Vell. | - | 1 |
| Epidendrum hymenodes Rchb. f. | - | 1 |
| Epidendrum leucochilum Link, Klotzsch& Otto ex Klotzsch | - | 37 |
| Epidendrum macroophorum Hágsater & Dodson | - | 7 |
| Epidendrum porphyreum Lindl. | - | 18 |
| Epidendrum sp. 1 | - | 1 |
| Epidendrum sp. 2 | - | 2 |
| Epidendrum sp. 3 | - | 1 |
| Epidendrum sp. 4 | - | 4 |
| Epidendrum sp. 5 | - | 22 |
| Epidendrum sp. 6 | - | 3 |
| Epidendrum sp. 7 | - | 2 |
| Kefersteinia sanguinolenta Rchb. f. | - | 12 |
| Malaxis sp1. | - | 6 |
| Malaxis sp2. | - | 5 |
| Maxillaria graminifolia (Kunth) Rchb. f. | - | 20 |
| Maxillariella pardalina (Garay)M.A.Blanco & Carnevali | - | 24 |
| Oncidium cimiciferum (Rchb. f.) Rchb. f. ex Lindl. | - | 3 |
| Oncidium pentadactylon Lindl. | - | 17 |
| Oncidium sp. 1 | - | 9 |
| Oncidium sp. 2 | - | 11 |
| Pelexia sp. | - | 6 |
| Pleurothallis anceps Luer | - | 3 |
| Pleurothallis restrepioides Lindl. | - | 1 |
| Pleurothallis ruscifolia (Jacq.) R. Br. | - | 6 |
| Pleurothallis sclerophylla Lindl. | - | 1 |
| Pleurothallis sp. 1 | - | 17 |
| Pleurothallis sp. 2 | - | 2 |
| Pleurothallis sp. 3 | - | 1 |
| Scelochilus luerae Dodson | VU, | 2 |
| Stanhopea impressa Rolfe | - | 1 |
| Stanhopea sp. | - | 9 |
| Stelisvillosa (Knowles &Westc.) Pridgeon& M. W. Chase. | - | 5 |
| Stelis argentata Lindl. | - | 30 |
| Stelis columnaris Lindl. | - | 1 |
| Stelis striolata Lindl. | -DD (EX), | 24 |
| Sudamerlycaste fimbriata (Poepp. &Endl.) Archila | - | 19 |
| Symphyglossumsanguineum(Rchb. f.) Schltr. | - | 4 |
| Trichopilia fragrans (Lindl.) Rchb. f. | - | 77 |
| Xylobiumleontoglossum(Rchb. f.) Rolfe | - | 149 |
| TOTAL | 922 |
Appendix B
| La Reventazón | UICN Status | Frequency |
|---|---|---|
| Aa sp. | 6 | |
| Acronia cordata (Ruiz & Pav.) Luer | - | 2 |
| Anathallissclerophylla (Lindl.) Pridgeon& M.W. Chase | - | 343 |
| Barbosella cucullata (Lindl.) Schltr. | - | 168 |
| Buccella ventricosa (Schltr.) Luer | - | 11 |
| Byrsella angulata (Rchb. f.) Luer | NT, VU | 18 |
| Cyclopogon sp. | - | 1 |
| Cyrtochilum cimiciferum (Rchb. f.) Dalström | - | 24 |
| Cyrtochilum serratum (Lindl.) Kraenzl. | - | 184 |
| Dichaea muricata (Sw.) Lindl. | - | 62 |
| Dracula felix (Luer) Luer | NT, VU | 2 |
| Elleanthuscapitatus (Poepp. & Endl.) Rchb. f. | - | 991 |
| Elleanthus gastroglottis Schltr. | - | 188 |
| Elleanthusmyrosmatis (Rchb. f.) Rchb. f. | - | 1293 |
| Epidendrum aff. secundum Jacq. | - | 3 |
| Epidendrum aff. sophronitoides F. Lehm. & Kraenzl. | - | 2 |
| Epidendrumbrachystele Schltr. | NT | 1 |
| Epidendrumcoryophorum (Kunth) Rchb. f. | - | 33 |
| Epidendrumdiothonaeoides Schltr. | LC | 3 |
| Epidendrumgeminiflorum Kunth | - | 6 |
| Epidendrumjamiesonis Lindl. | - | 703 |
| Epidendrummacroöphorum Hágsater & Dodson | - | 26 |
| Epidendrumpolyanthogastrium Hágsater & Dodson | VU, | 4 |
| Malaxis sp. | - | 3 |
| Maxillaria aff. pardalina | - | 1 |
| Maxillaria calantha Schltr. | - | 10 |
| Maxillariella arbuscula (Lindl.) M.A.Blanco & Carnevali | - | 332 |
| Maxillariella graminifolia (Kunth) M.A.Blanco & Carnevali | - | 158 |
| Maxillariella pardalina (Garay) M.A.Blanco & Carnevali | - | 1108 |
| Oncidium pentadactylon Lindl. | - | 4 |
| Ornithidium aggregatum (Kunth) Rchb.f. | - | 601 |
| Phragmipediumlindenii (Lindl.) Dressler & N.H. Williams | LC | 9 |
| Pleurothallis macra Lindl. | LC | 3 |
| Pleurothallis ruscifolia (Jacq.) R.Br. | - | 4 |
| Pleurothallis pulchella (Kunth) Lindl. | - | 2664 |
| Prosthecheapamplonensis (Rchb. f.) W.E. Higgins | - | 3 |
| Restrepia guttulata Lindl. | LC | 2 |
| Restrepiopsis tubulosa (Lindl.) Luer | - | 1 |
| Sobralia sp. | - | 78 |
| Stelis argentata Lindl. | - | 5 |
| Stelis pusilla Kunth | - | 1 |
| Stelisrestrepioides(Lindl.) Pridgeon&M.W.Chase | - | 80 |
| Stelis sp. 1 | - | 25 |
| Stelis sp. 2 | - | 11 |
| Trichopilia fragrans (Lindl.) Rchb. f. | - | 4 |
| Zootrophion hypodiscus (Rchb.f.) Luer | - | 15 |
| TOTAL | 9196 |
References
- Oña Sánchez, E.P.; Mites Cadena, M.d.J.; Reina Moreno, D. Inventario de la Familia Orchidaceae en la Reserva Geobotánica Pululahua. Rev. Cient. Hallazgos21 2018, 3, 2–13. [Google Scholar]
- Endara, L. Orchidaceae. In Libro Rojo de las Plantas Endémicas del Ecuador, 2nd ed.; León-Yánez, S., Valencia, R., Pitman, N., Endara, L., Ulloa, C., Navarrete, H., Eds.; Publicaciones del Herbario QCA, Pontificia Universidad Católica del Ecuador: Quito, Ecuador, 2011; pp. 441–702. [Google Scholar]
- Radice, M.; Vidari, G. Caracterización fitoquímica de la especie Ilex guayusa Loes. y elaboración de un prototipo de fitofármaco de interés comercial. La Granja 2007, 6, 6–11. [Google Scholar] [CrossRef]
- Jiménez León, M. Orquídeas del Ecuador-Número de especies, endemismo, especies amenazadas y su manejo adecuado. Actualidad 2014, 1, 1–3. [Google Scholar]
- Tirira, D.G. Lista Roja de los mamíferos del Ecuador. In Libro Rojo de los Mamíferos del Ecuador, 2nd ed.; Tirira, D.G., Ed.; Publicación especial sobre los mamíferos del Ecuador 8; Fundación Mamíferos y Conservación, Pontificia Universidad Católica del Ecuador y Ministerio del Ambiente del Ecuador: Quito, Ecuador, 2011. [Google Scholar]
- Dodson, C.; Escobar, R. Orquídeas Nativas del Ecuador; Editorial Colina: Medellín, Colombia, 1994; pp. 49–51. [Google Scholar]
- León-Yánez, S.; Valencia, R.; Pitman, N.; Endara, L.; Ulloa, C.U.; Navarrete, H. Libro Rojo de las Plantas Endémicas del Ecuador; Publicaciones del Herbario QCA, Pontificia Universidad Católica del Ecuador: Quito, Ecuador, 2018. [Google Scholar]
- Dodson, C. Orchidaceae. In Catalogue of the Vascular Plants of Ecuador; Monographs on Systematics Botany; Missouri Botanical Garden: St. Louis, MO, USA, 1999; Volume 75, pp. 630–770. [Google Scholar]
- Hodgson, M.; Paine, R.; Anderson, N. A Guide to the Orchids of the World; Angus & Robertson Book: Sydney, NSW, Australia, 1991; pp. 1–23. [Google Scholar]
- Jadán, O.; Cedillo, H.; Zea, P.; Quichimbo, P.; Peralta, A.; Vaca, C. Relación entre la deforestación y variables topográficas en un contexto agrícola ganadero, cantón Cuenca. Bosques Latid. Cero 2016, 6, 1–13. [Google Scholar]
- Endara, L.; Williams, H.; León-Yánez, S. Explorando los patrones de endemismo de las orquídeas ecuatorianas: Implicaciones para su conservación. In Proceedings of the Actas del X Congreso Latinoamericano de Botánica, La Serena, Chile, 4–10 October 2010. [Google Scholar]
- Newman, B.J.; Ladd, P.; Batty, A.; Dixon, K. Ecology of orchids in urban bushland reserves—Can orchids be used as indicators of vegetation condition? Lankesteriana 2007, 7, 313–315. [Google Scholar] [CrossRef]
- Whigham, D.F.; Willems, J.H. Demographic studies and life-history strategies of temperate terrestrial orchids as a basis for conservation. In Orchid Conservation; Dixon, K., Kell, S., Barret, R., Crib, P., Eds.; Natural History Publications: Kota Kinabalu, Malaysia, 2003; Chapter 8; pp. 137–158. [Google Scholar]
- Ministerio del Ambiente del Ecuador. Plan de Manejo de la Reserva Geobotánica Pululahua; Dirección Nacional de Biodiversidad: Quito, Ecuador, 2011.
- Bland, L.M.; Keith, D.A.; Miller, R.M.; Murray, N.J.; Rodríguez, J.P. Guidelines for the application of IUCN Red List of Ecosystems Categories and Criteria, version 1.1; International Union for the Conservation of Nature: Gland, Switzerland, 2017. [Google Scholar]
- Ministerio del Ambiente del Ecuador. Modelo Bioclimático para la Representación Cartográfica de Ecosistemas del Ecuador Continental; Ministerio del Ambiente del Ecuador: Quito, Ecuador, 2013.
- Gentry, A.H.; Dodson, C. Contribution of nontrees to species richness of a tropical rain forest. St. Louis. Biotropica 1987, 19, 149–156. [Google Scholar] [CrossRef]
- Oña, P.; Reina, D.; Baquero, L. Orquídeas de la Reserva Geobotánica Pululahua (Volume 1). Fieldguides.fieldmuseum.org. 2019. Available online: https://fieldguides.fieldmuseum.org/sites/default/files/rapid-color-guides-pdfs/958_ecuador_orchids_of_the_pululahua_geobotanical_reserve.pdf (accessed on 5 January 2022).
- Simpson, E.H. Measurement of diversity. Nature 1949, 163, 688. [Google Scholar] [CrossRef]
- Ludwig, J. Statistical Ecology; A Primer on Methods and Computing; Willey—Interscience Publication: New York, NY, USA, 1988; pp. 89–95. [Google Scholar]
- Lande, R. Statistics and partitioning of species diversity, and similarity among multiple communities. Oikos 1996, 76, 5–13. [Google Scholar] [CrossRef]
- Shannon, C.E.; Weaver, W. A mathematical theory of communication. Bell Sys. Tech. J. 1948, 27, 379–423, 623–656. [Google Scholar] [CrossRef] [Green Version]
- Zar, J.H. Biostatistical Analysis, 5th ed.; Pearson: London, UK, 2014. [Google Scholar]
- Pielou, E.C. The measurement of diversity in different types of biological collections. J. Theor. Biol. 1966, 13, 131–144. [Google Scholar] [CrossRef]
- Jaccard, P. Étude comparative de la distribution florale dans une portion des alpes et des jura. Bull. Soc. Vaudoise Sci. Nat. 1901, 37, 547–579. [Google Scholar]
- Krebs, C.J. Ecological Methodology, 3rd ed.; Addison Wesley Longman Inc.: Reading, MA, USA, 2014. [Google Scholar]
- Bray, J.R.; Curtis, J.T. An ordination of the upland forest communities of southern Wisconsin. Ecol. Monogr. 1957, 27, 325–349. [Google Scholar] [CrossRef]
- IUCN. Categorías y Criterios de la Lista Roja de la UICN; Version 3.1; IUCN: Gland, Switzerland; Cambridge, UK, 2012. [Google Scholar]
- Committee, P. Guidelines for using the IUCN red list categories and criteria. Eur. J. Taxon. 2022, 15, 42–113. [Google Scholar] [CrossRef]
- Holdridge, L.R. Life Zone Ecology; Tropical Science Center: San Jose, Costa Rica, 1967. [Google Scholar]
- Damian, A. Diversity and altitudinal distribution of terrestrial species of the family Orchidaceae in a montane forest inside Yanachaga Chemillen National Park (Pasco-Peru). Arnaldoa 2013, 20, 103–116. [Google Scholar]
- Dodson, C.; Gentry, A.H. Biological extinction in western Ecuador. Ann. Missouri Bot. Gard. 1991, 78, 273–295. [Google Scholar] [CrossRef]
- Krömer, T.; Gradstein, S.R.; Acebey, A. Diversidad y ecología de epífitas vasculares en bosques montanos primarios y secundarios de Bolivia. Ecol. Boliv. 2007, 42, 23–33. [Google Scholar]
- Atwood, J.T. The size of the Orchidaceae and the systematic distribution of epiphytic orchids. Selbyana 1986, 9, 171–186. [Google Scholar]
- López Ardila, M. Diversidad de Orquídeas en Áreas Silvestres y de uso Antrópico de la Región del Tequendama, Cordillera Oriental de Colombia. Undergraduate Thesis, Pontificia Universidad Javeriana, Bogotá, Colombia, 2018. [Google Scholar]
- Pielou, E.C. Mathematical Ecology; Wiley: New York, NY, USA, 1977. [Google Scholar]
- Calatayud, G. Diversity of Orchidaceae family in the montane forest in San Ignacio (Cajamarca, Peru). Rev. Per. Biol. 2005, 12, 309–316. [Google Scholar]
- Winkler, M.; Hulber, K.; Hietz, P. Population dynamics of epiphytic orchids in a metapopulation context. Ann. Bot. 2009, 104, 995–1004. [Google Scholar] [CrossRef]
- Martínez-Meléndez, N.; Pérez-Ferrera, M.A.; Flores-Palacios, A. Vertical stratification in host preference by vascular epiphytes in a Chiapas, Mexico, cloud forest. Rev. Biol. Trop. 2008, 56, 2069–2686. [Google Scholar]
- OrtaPozo, S.; López Trabanco, P. Patterns which characterized the relationships host-orchid in the Biosphere Reserve “Sierra del Rosario”. Rev. Cient. Avances 2013, 15, 254–264. [Google Scholar]
- Hietz, P.; Hietz-seifert, U. Composition and ecology of vascular epiphyte communities along an altitudinal gradient in central Veracruz, Mexico. J. Veget. Sci. 1995, 6, 487–498. [Google Scholar] [CrossRef] [Green Version]
- Tupac, O.J.; Flanagan, N.S. Above ground orchid interactions: Pollination and mycorrhizae in tropical epiphytic orchids. In Proceedings of the 31st New Phytologist Symposium, Orchid Symbioses: Models for Evolutionary Ecology, Rende, Italy, 14–16 May 2013; p. 17. [Google Scholar]
- Batygina, T.; Bragi, E.; Vasilyeva, V. El sistema reproductivo y germinación de las orquìdeas. Acta Biol. Cracov. Series Botanica 2003, 45, 21–34. [Google Scholar]
- McCormick, M.K.; Taylor, D.L.; Whigham, D.F.; O’neil, J.P. Distribution of orchid populations, a matter of fungi? In Proceedings of the 31st New Phytologist Symposium, Orchid Symbioses: Models for Evolutionary Ecology, Rende, Italy, 14–16 May 2013; p. 12. [Google Scholar]
- Mites, M. Catálogo de Orquídeas; Bosque El Cerro: Chone, Ecuador; Pontifica Universidad Católica del Ecuador: Portoviejo, Ecuador, 2015. [Google Scholar]
- Castillo-Pérez, L.J.; Martínez-Soto, D.; Maldonado-Miranda, J.J.; Alonso-Castro, A.J.; Carranza-Álvarez, C. The endemic orchids of Mexico: A review. Biologia 2019, 74, 1–13. [Google Scholar] [CrossRef]

| Parameter | Chaupisacha | La Reventazón |
|---|---|---|
| Plot surface (m2) | 10,000 | 10,000 |
| No. orchid individuals | 922 | 9196 |
| No. exclusive species | 41 | 32 |
| No. shared species | 14 | |
| Density (ind./100 m2) | 0.96 | 122 |
| Simpson’s diversity (1 − λ) | 0.90 | 0.85 |
| Shannon-Weaner (H′) * | 1.29 a | 1.02 b |
| Equity (Pielou’s J′) | 0.78 | 0.61 |
| Index | Index Value |
|---|---|
| Jaccard | 0.161 |
| Euclidean distance | 1490 |
| Mean Euclidean distance | 398 |
| Bray-Curtis (dissimilarity) | 0.906 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mites, M.; García-Mozo, H.; Galán, C.; Oña, E. Analysis of the Orchidaceae Diversity in the Pululahua Reserve, Ecuador: Opportunities and Constraints as Regards the Biodiversity Conservation of the Cloud Mountain Forest. Plants 2022, 11, 698. https://doi.org/10.3390/plants11050698
Mites M, García-Mozo H, Galán C, Oña E. Analysis of the Orchidaceae Diversity in the Pululahua Reserve, Ecuador: Opportunities and Constraints as Regards the Biodiversity Conservation of the Cloud Mountain Forest. Plants. 2022; 11(5):698. https://doi.org/10.3390/plants11050698
Chicago/Turabian StyleMites, Mariana, Herminia García-Mozo, Carmen Galán, and Edwin Oña. 2022. "Analysis of the Orchidaceae Diversity in the Pululahua Reserve, Ecuador: Opportunities and Constraints as Regards the Biodiversity Conservation of the Cloud Mountain Forest" Plants 11, no. 5: 698. https://doi.org/10.3390/plants11050698
APA StyleMites, M., García-Mozo, H., Galán, C., & Oña, E. (2022). Analysis of the Orchidaceae Diversity in the Pululahua Reserve, Ecuador: Opportunities and Constraints as Regards the Biodiversity Conservation of the Cloud Mountain Forest. Plants, 11(5), 698. https://doi.org/10.3390/plants11050698








