Crepidium sect. Crepidium (Orchidaceae, Malaxidinae)—Chemical and Morphological Study of Flower Structures in the Context of Pollination Processes
Abstract
:1. Introduction
2. Results
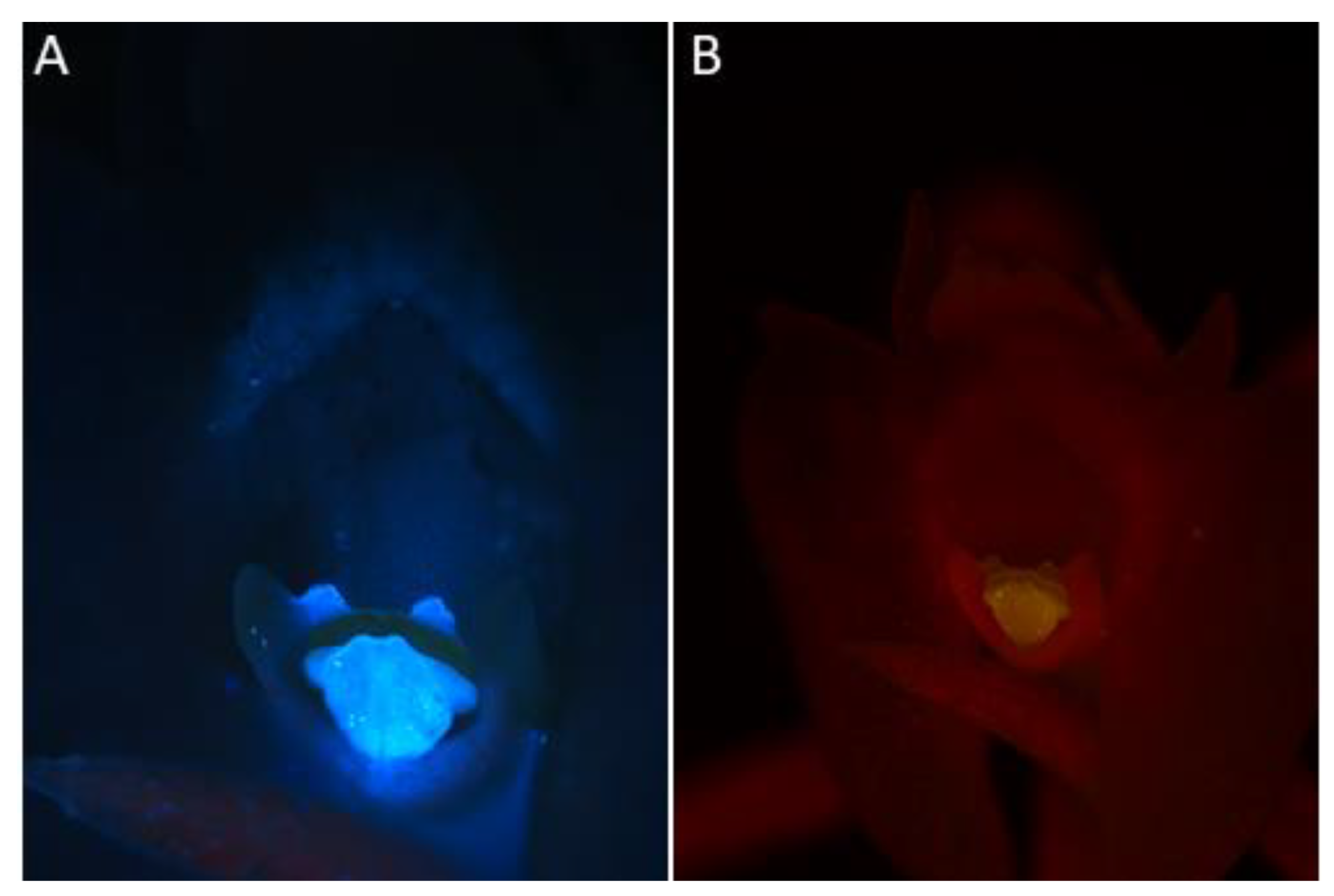
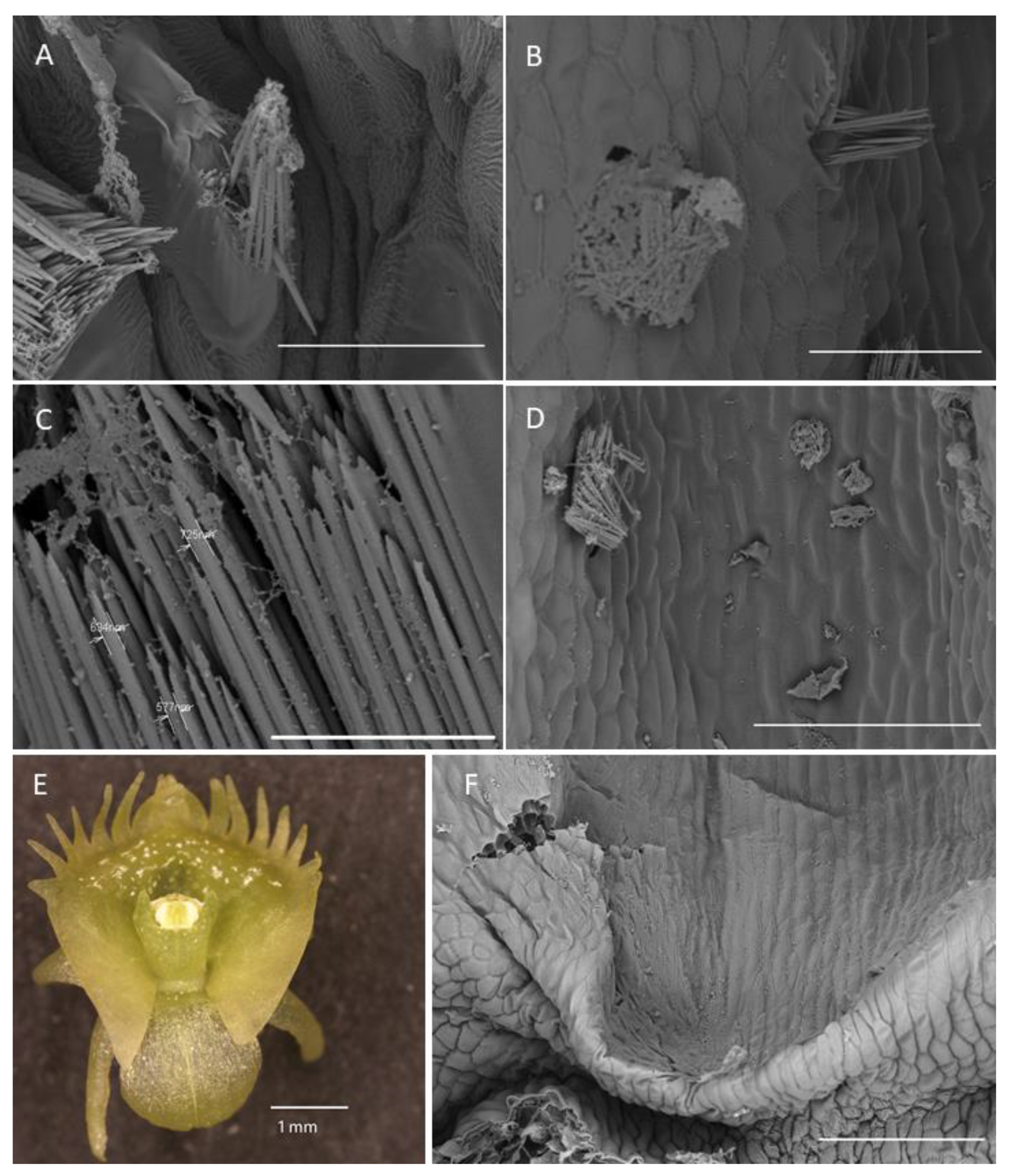
- Under UV light (Nikon UV-2A filter, excitation 330–380 nm, blue fluorescence)—soft reflection of light was observed over the entire surface of the flower, but was reflected particularly strongly by the anther, viscidia, living idioblasts with raphides, and raphides dispersed when idioblasts were damaged (Figure 4A)
- Under blue light (Nikon B-2A filter, long pass emission, excitation 450–490 nm, green fluorescence)—light was reflected particularly strongly by the anther, living idioblasts with raphides, and raphides dispersed when idioblasts were damaged (Figure 4B)
- Under green light (Nikon G-2A filter, excitation 510–590 nm, red fluorescence)—light was strongly reflected only by the anther.
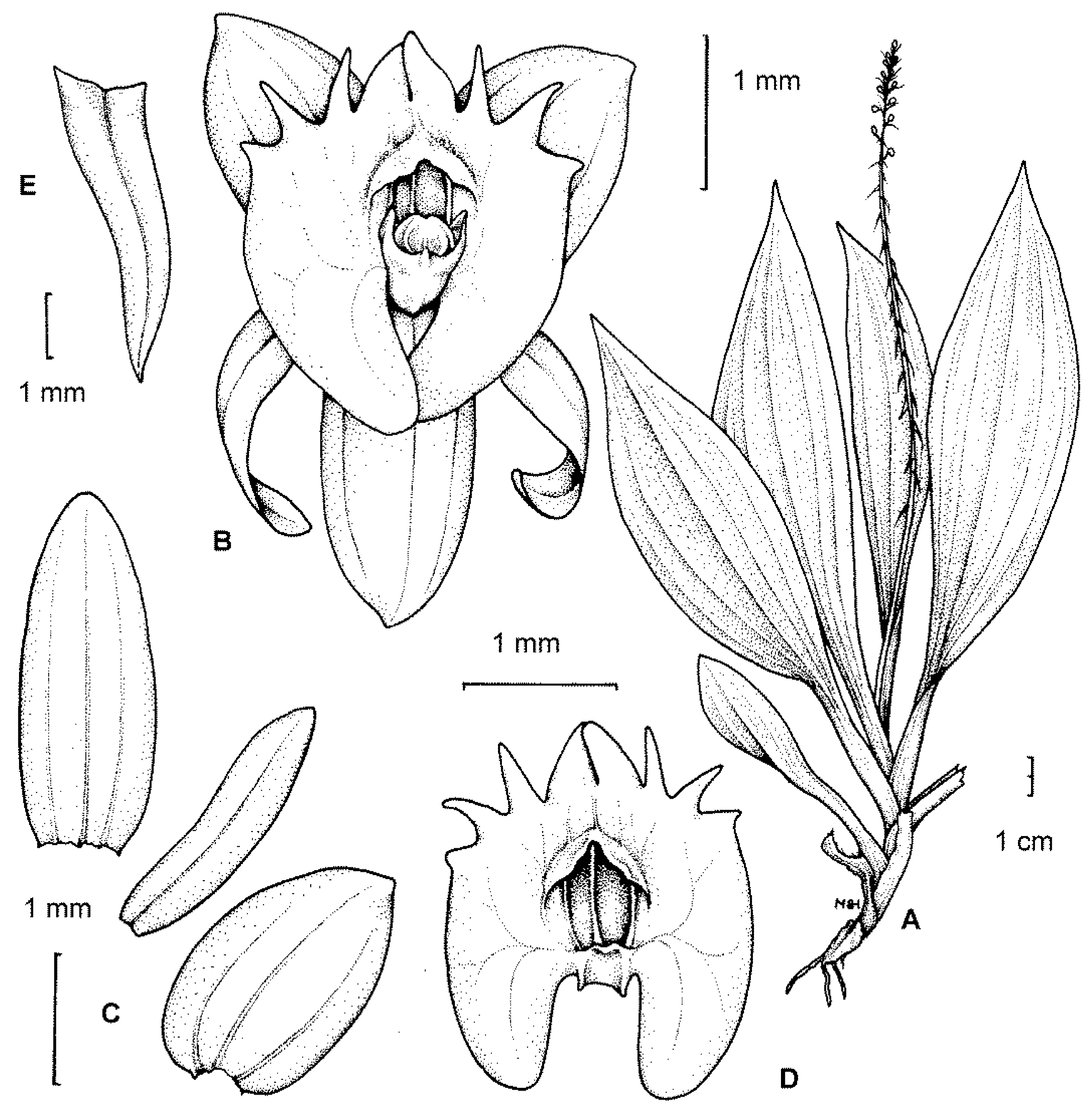
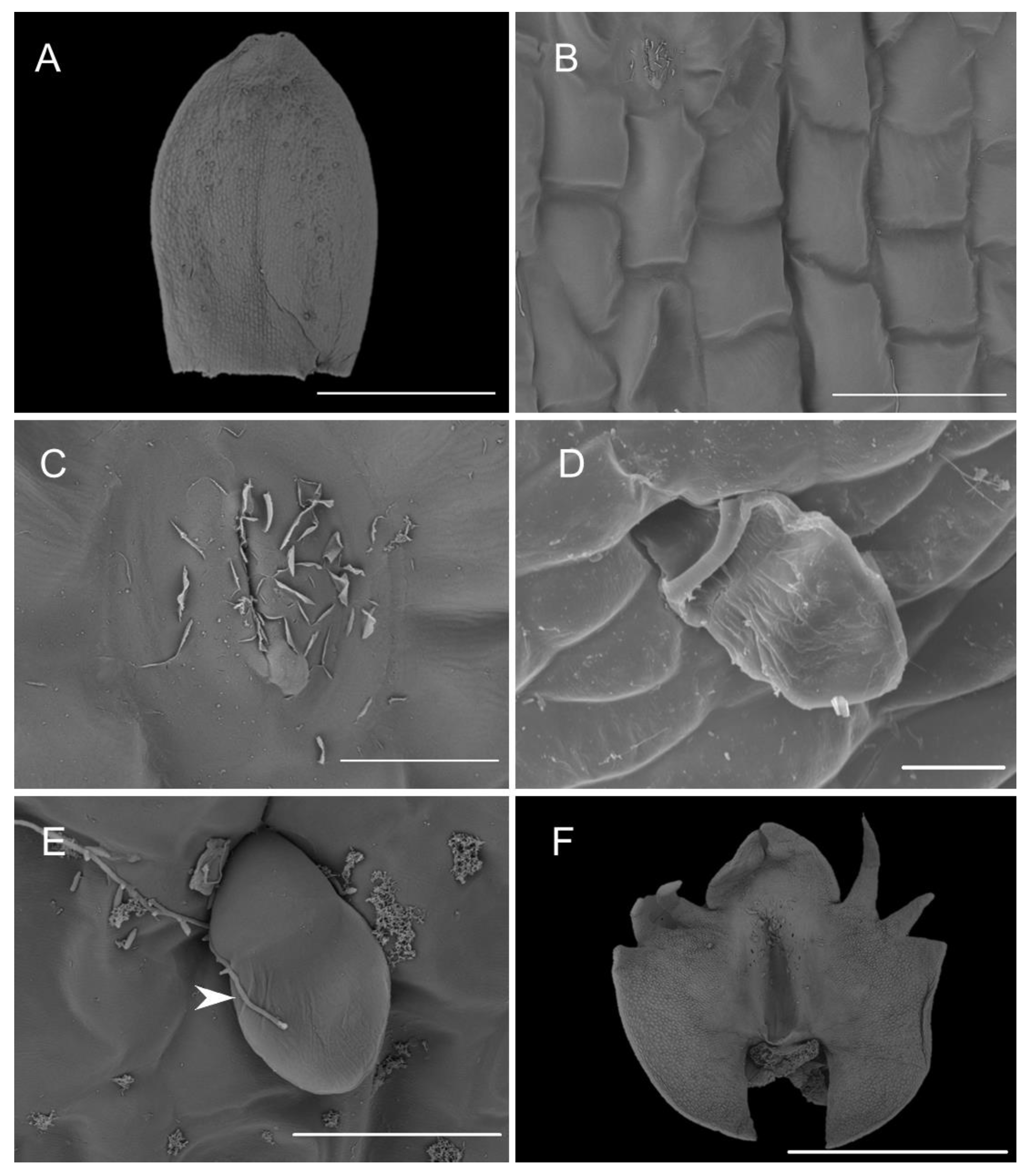
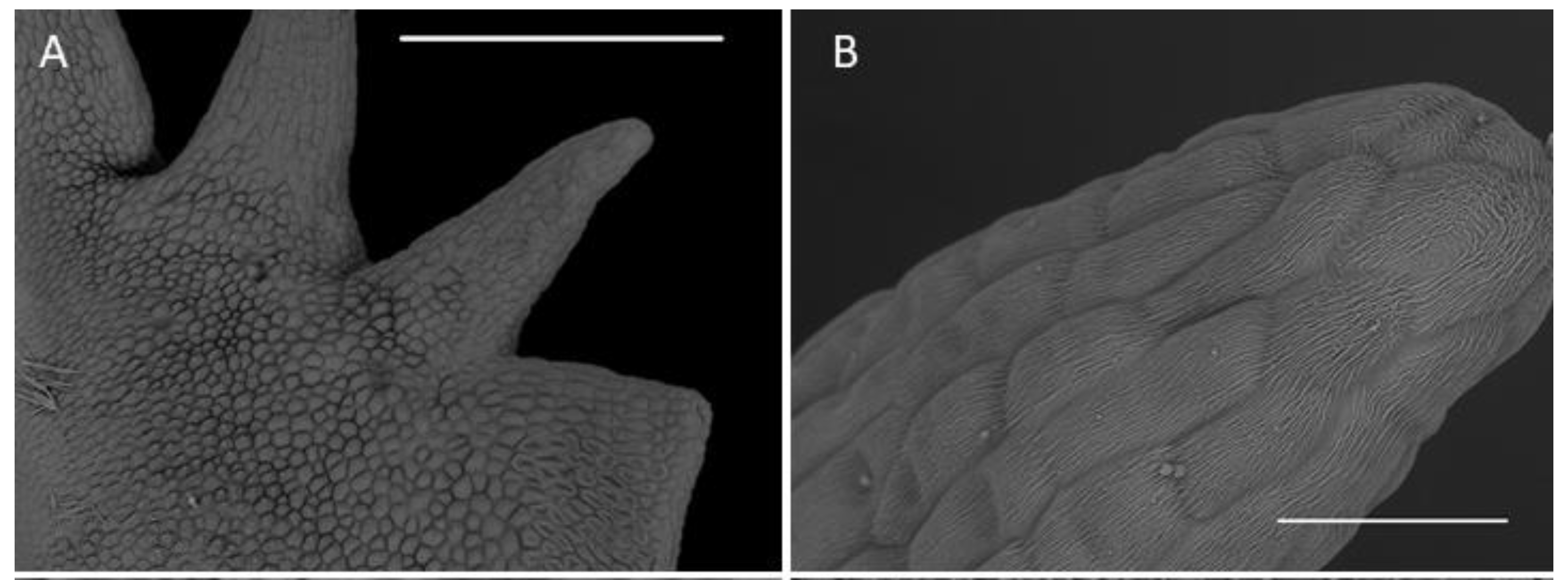
2.1. Scanning Electron Microscopy (SEM)
2.1.1. Sepals
2.1.2. Petals
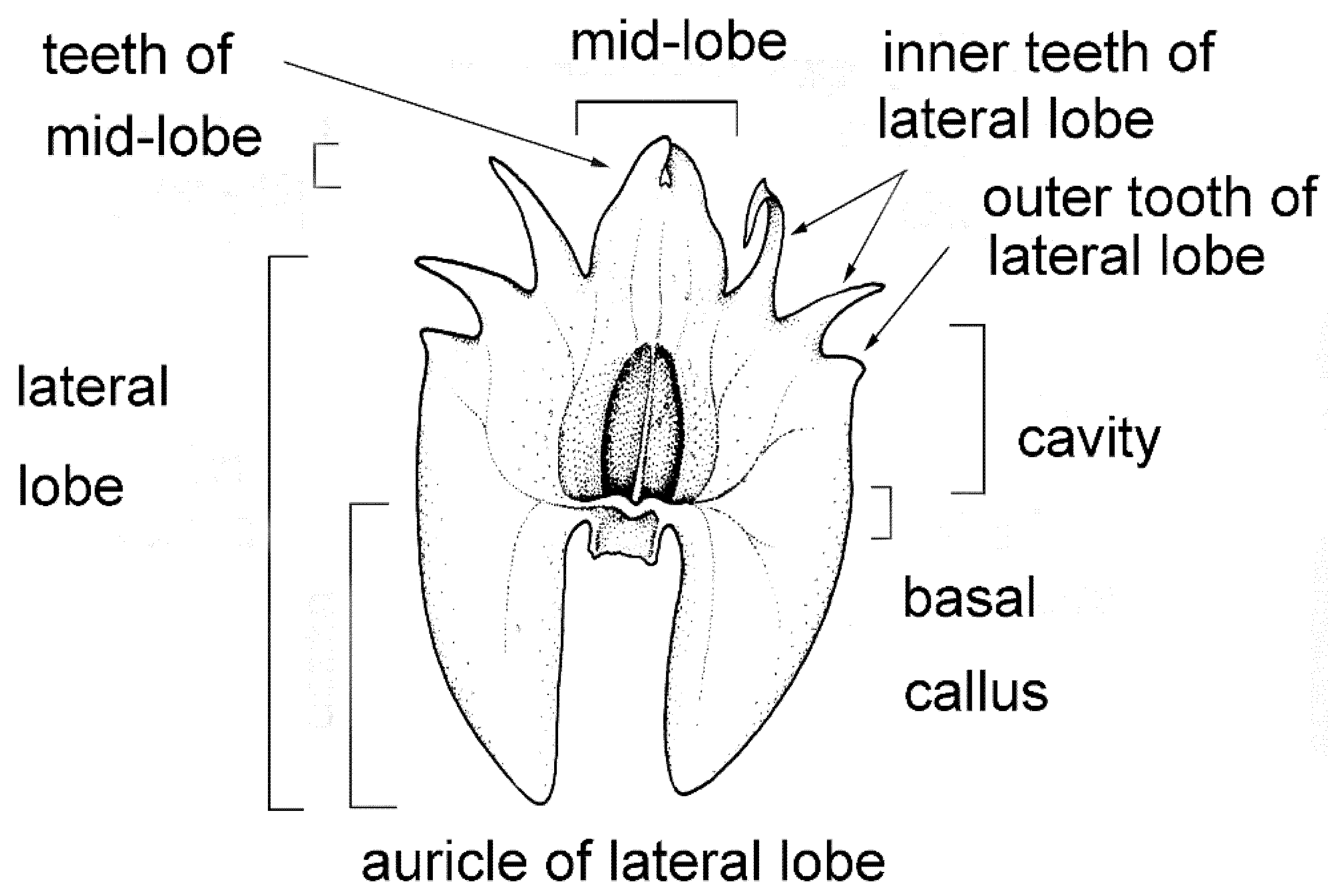
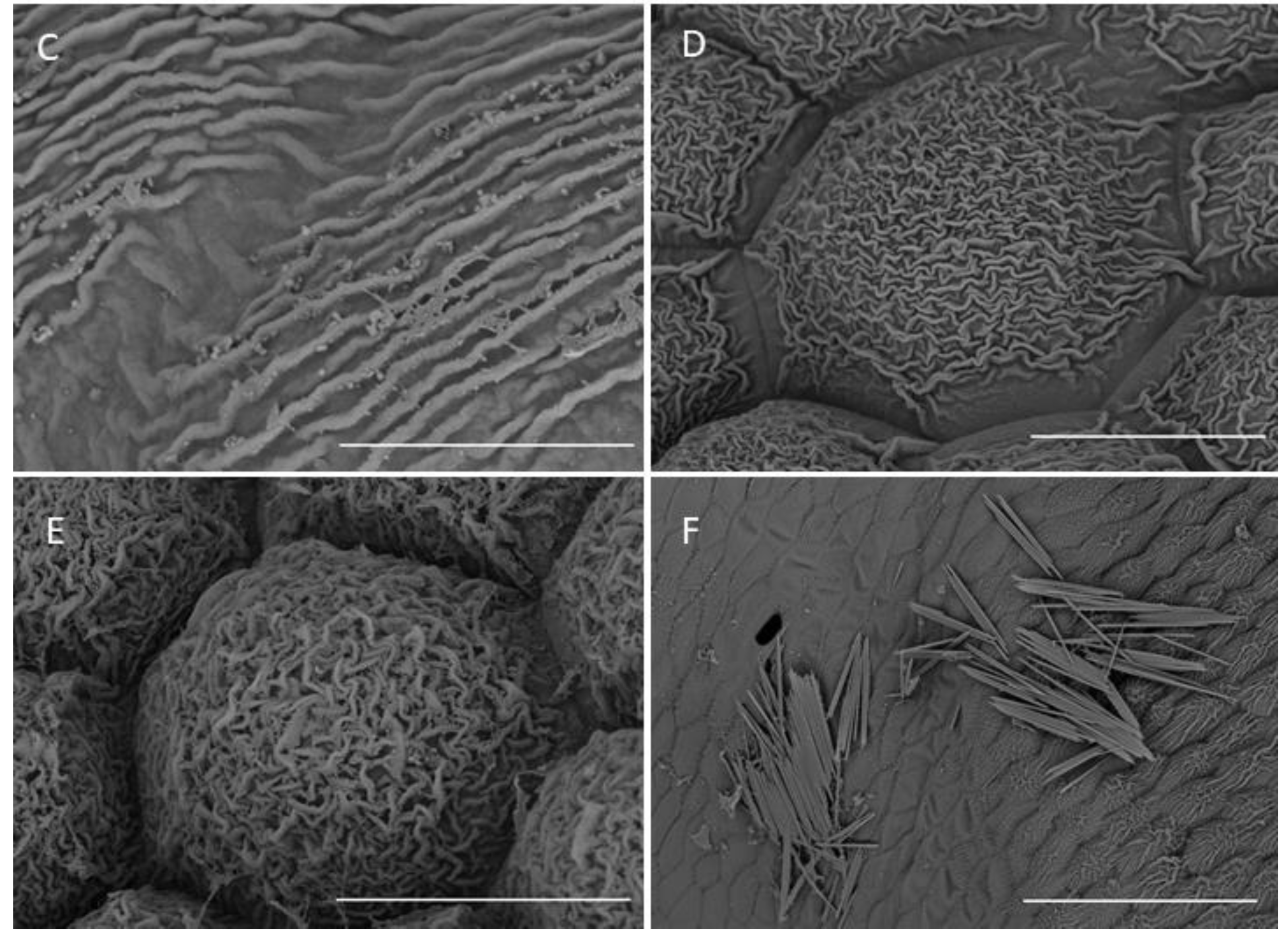
2.1.3. Labellum
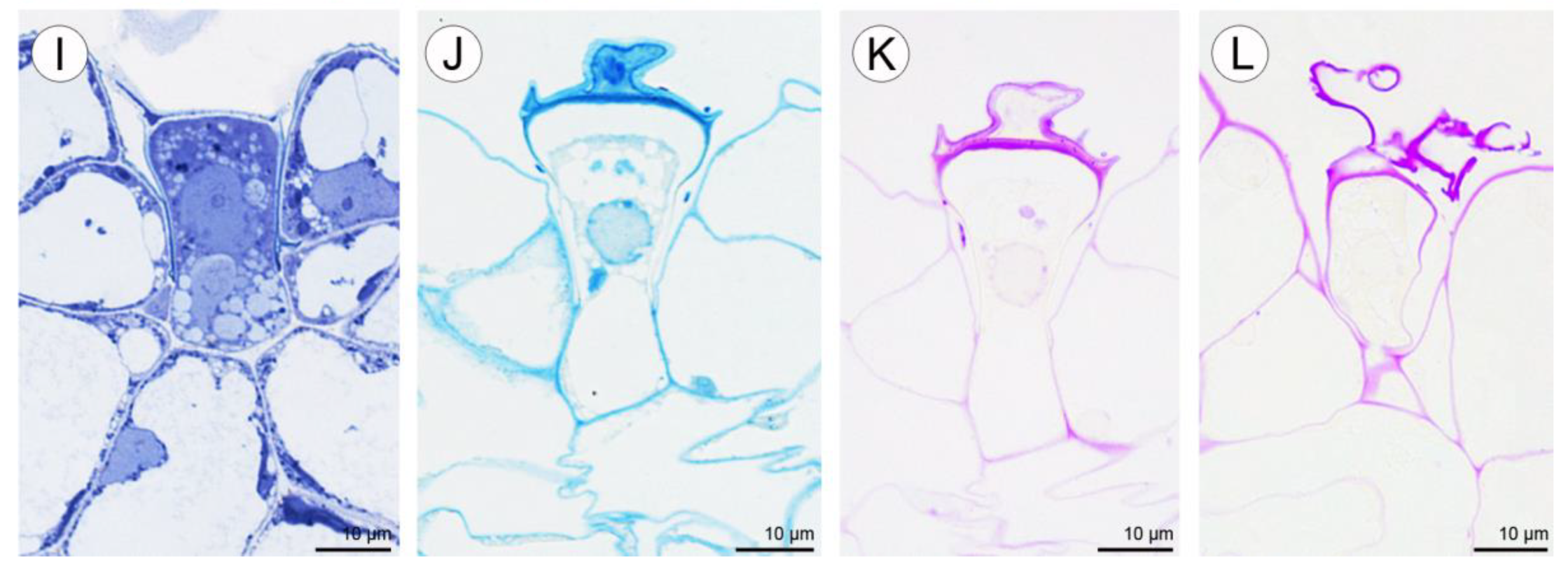
2.1.4. Labellar Cavity (Pseudonectary)
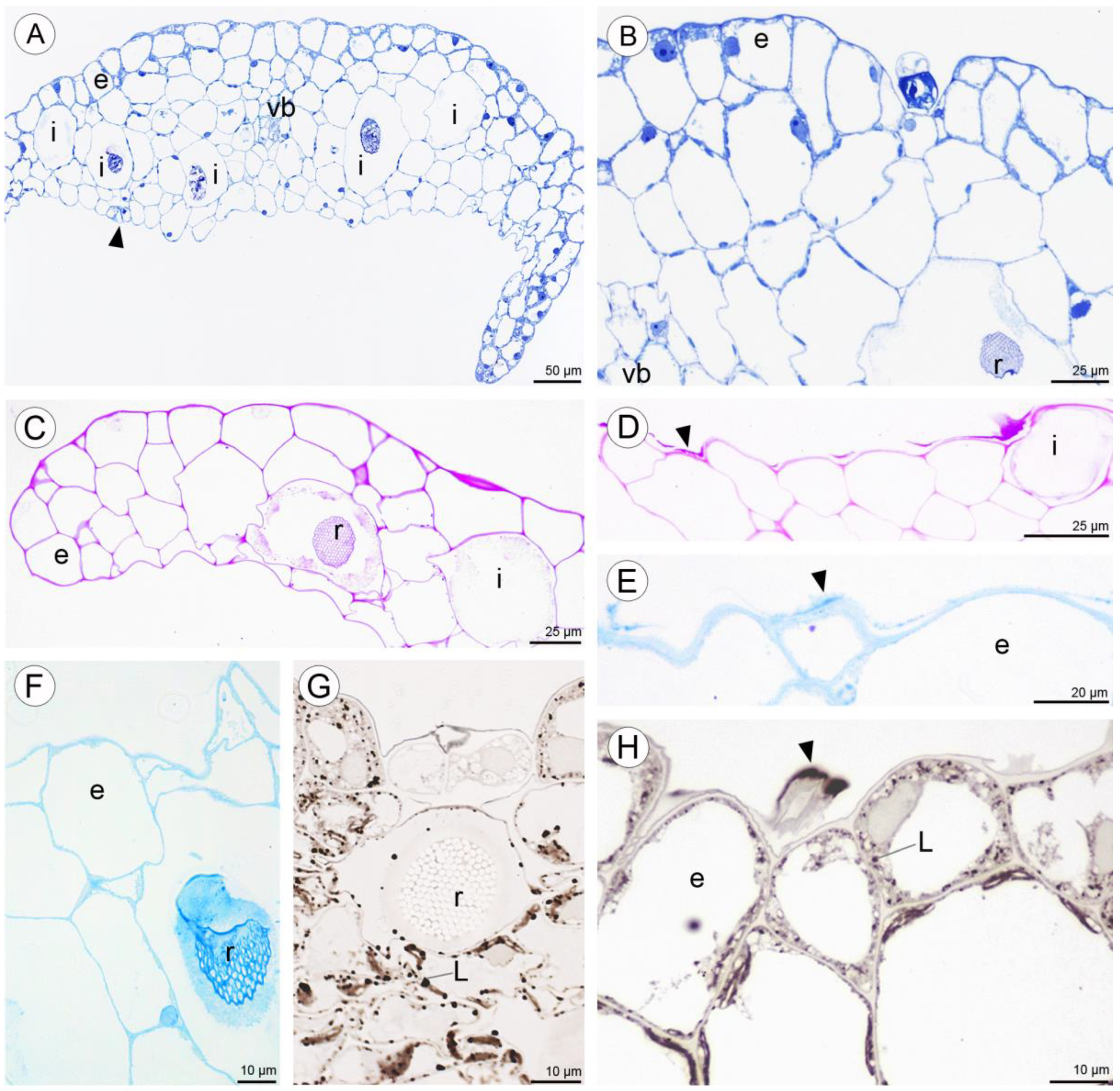
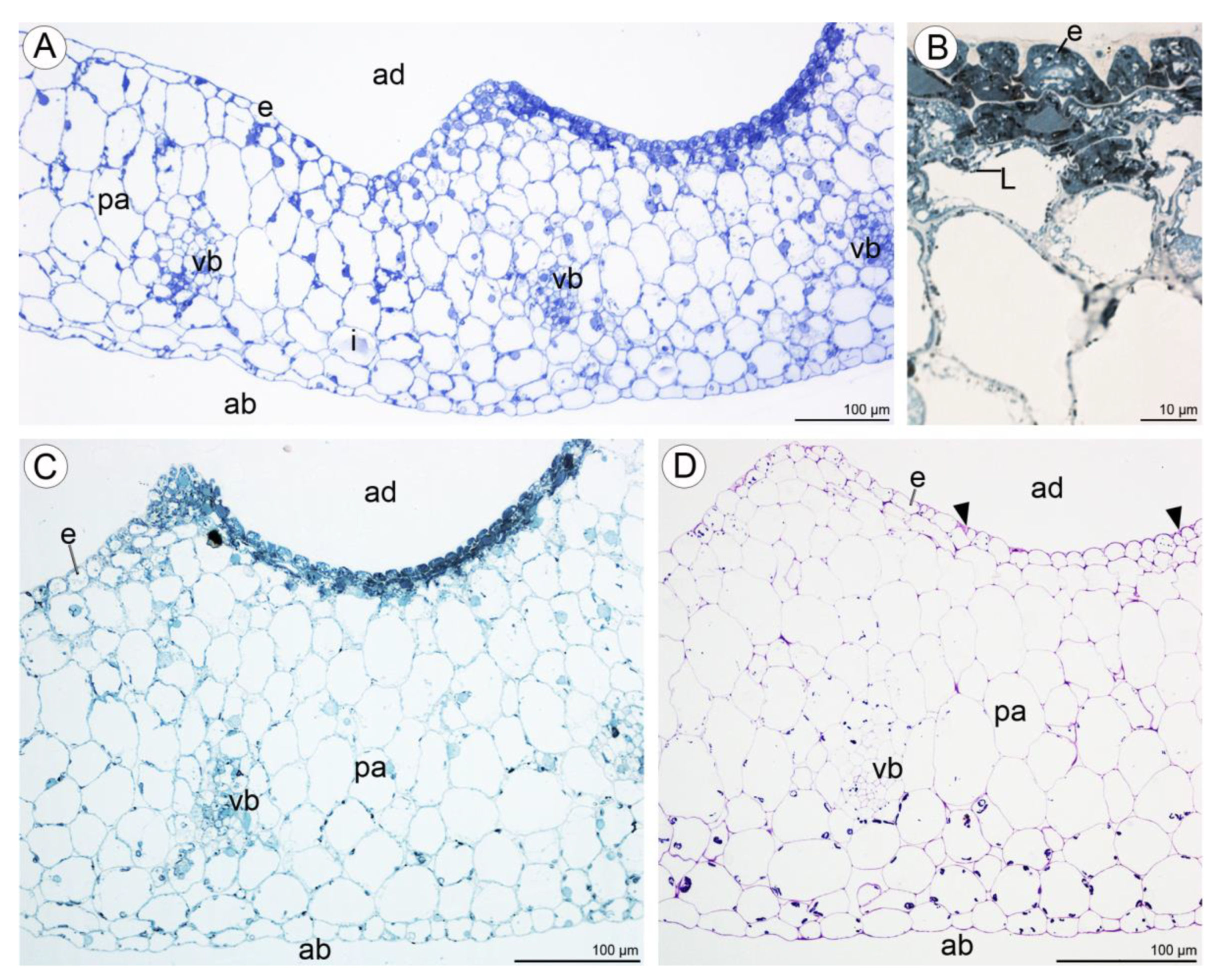
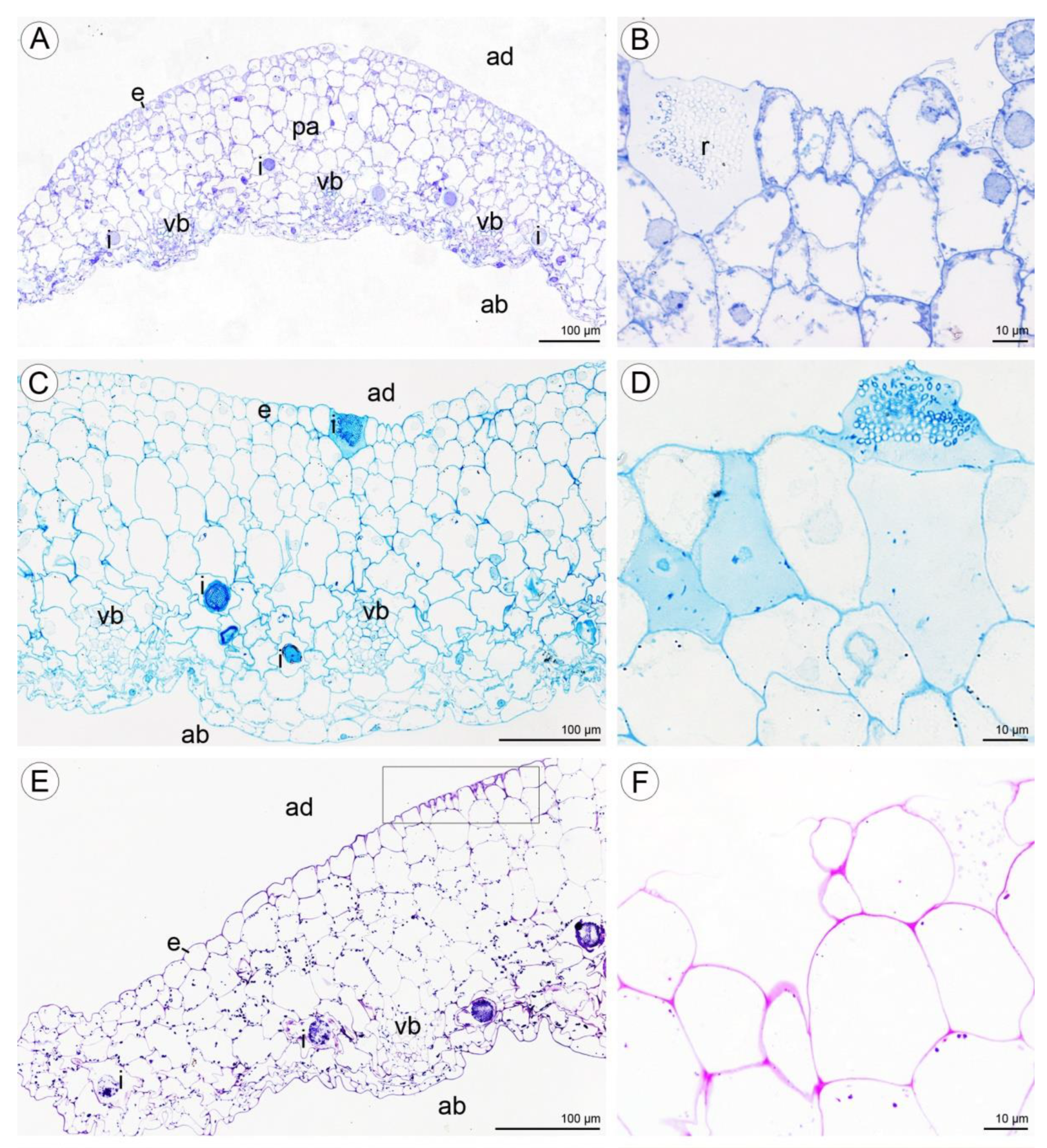
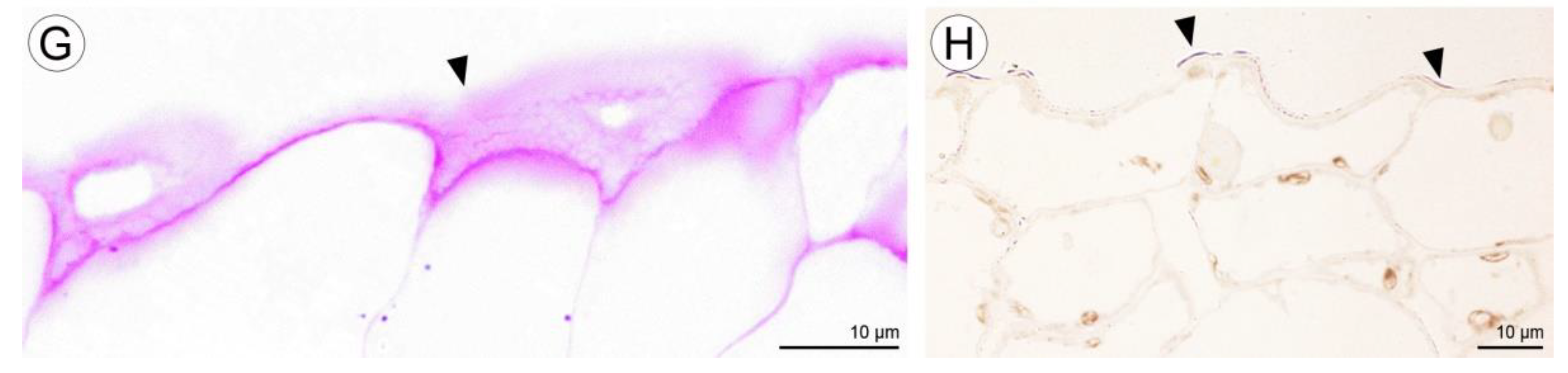
2.2. Anatomical and Histochemical Analysis
2.2.1. Tepals (Petals and Sepals)
2.2.2. Labellum
2.3. Ultrastructural Analysis (TEM)
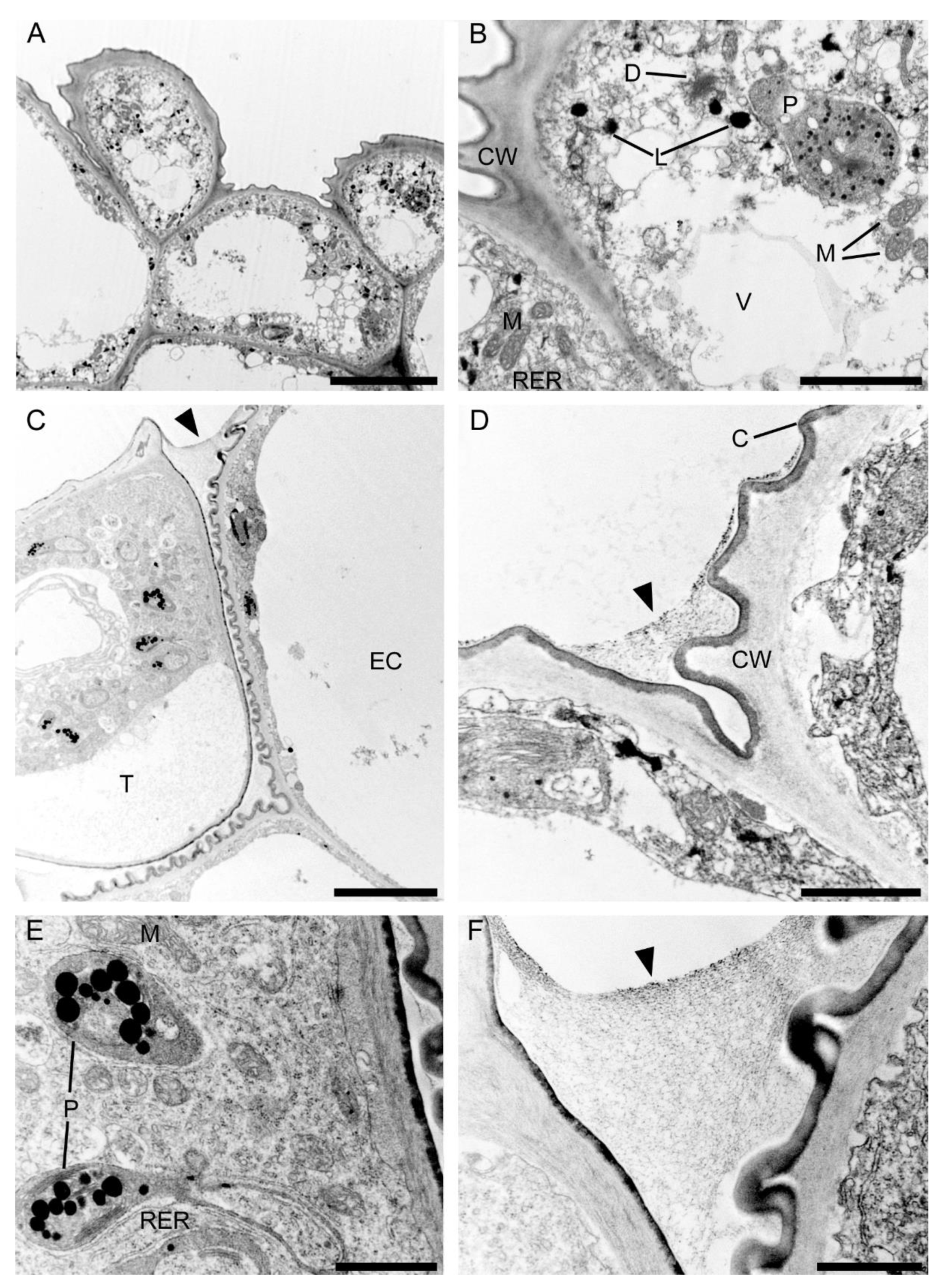
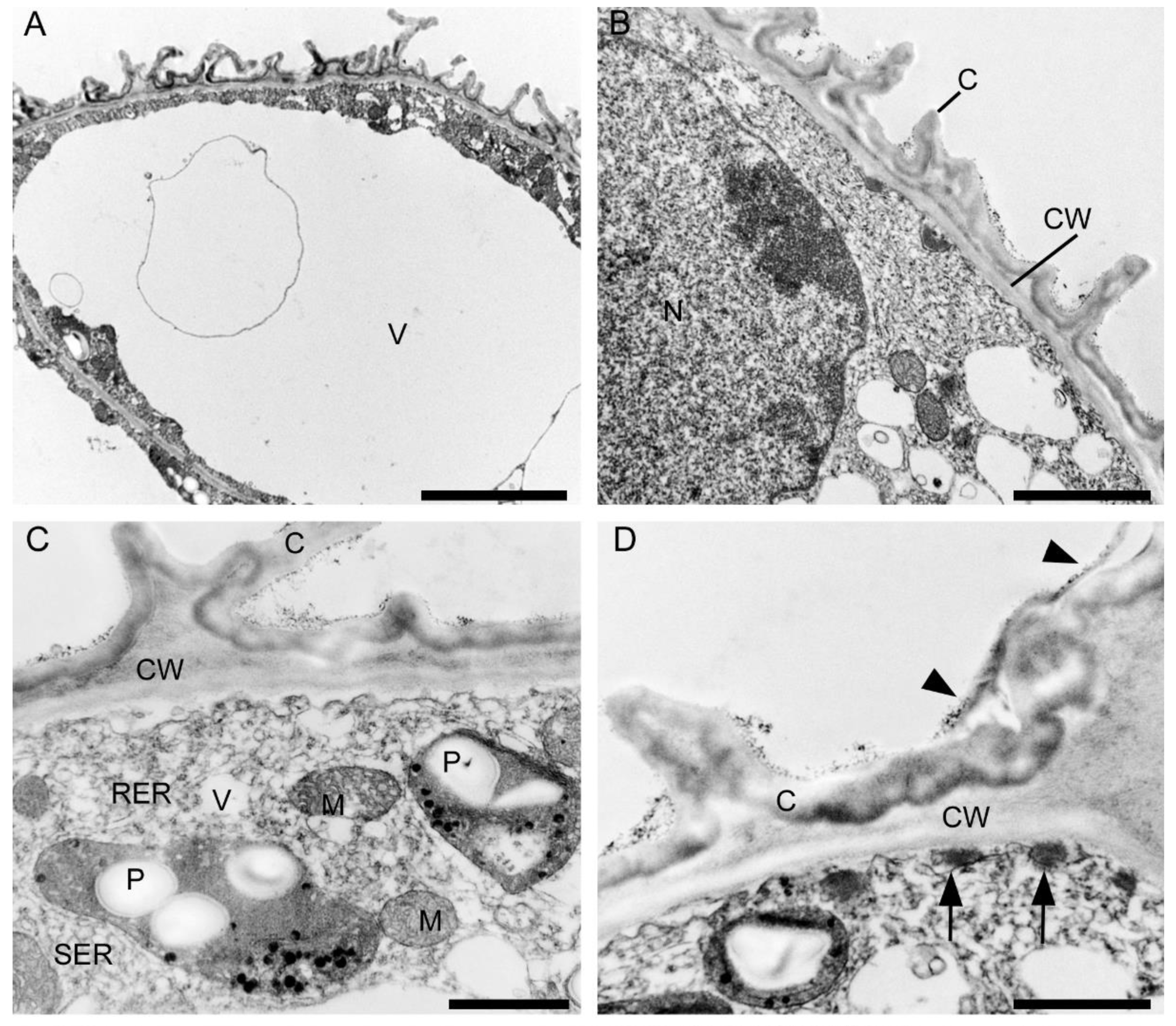
2.3.1. Tepals (Petals and Sepals)
2.3.2. Labellum
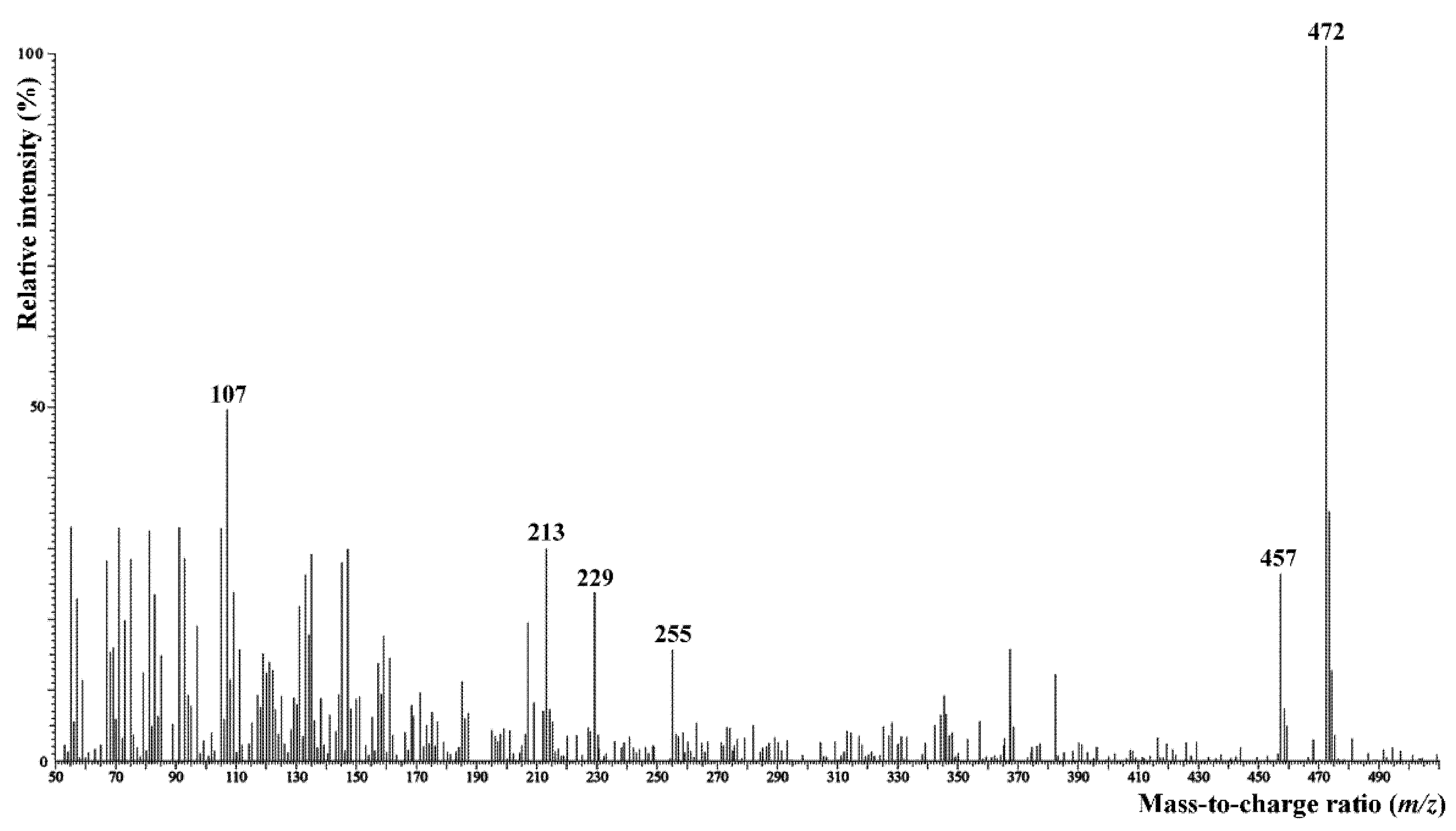
2.4. Chemical Analysis
The Chemical Composition of Sugar Fraction and Surface Lipids
3. Discussion and Conclusions
4. Materials and Methods
4.1. Light Microscopy and Histochemical Analysis
4.2. Scanning Electron Microscopy (SEM)
4.3. Transmission Electron Microscopy (TEM)
4.4. Epifluorescence Analysis
4.5. Chemical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Margońska, H.B.; Kowalkowska, A.K.; Górniak, M.; Rutkowski, P. Taxonomic Redefinition of Subtribe Malaxidinae (Orchidales, Orchidaceae); Koeltz Scientific Books: Koenigstein, Germany, 2012; pp. 1–606. [Google Scholar]
- Schiestl, F.P. On the success of a swindle: Pollination by deception in orchids. Naturwissenschaften 2005, 92, 255–264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Endara, L.; Grimaldi, D.A.; Roy, B.A. Lord of the flies: Pollination of Dracula orchids. Lankesteriana 2010, 10, 1–11. [Google Scholar] [CrossRef] [Green Version]
- van der Pijl, L.; Dodson, C.H. Orchid Flowers: Their Pollination and Evolution; University of Miami Press: Coral Gables, FL, USA, 1966; pp. 1–214. [Google Scholar]
- Catling, P.M. Auto-pollination in the Orchidaceae. In Orchid biology: Reviews and Perspectives; Arditti, J., Ed.; Timber Press: Portland, OR, USA, 1990; Volume V, pp. 121–158. [Google Scholar]
- Comber, J.B. Orchids of Java; Bentham Moxon Trust, Royal Botanic Gardens: Kew, UK, 1990; pp. 1–407. [Google Scholar]
- Comber, J.B. Orchids of Sumatra; Natural History Publications: Kota Kinabalu, Malaysia; Royal Botanic Garden: Kew, UK, 2001; pp. 1–1036. [Google Scholar]
- Brooks, C.J.W.; Middleditch, B.S. Some aspects of mass spectrometry in steroid analysis. In Modern Methods of Steroid Analysis; Heftmann, E., Ed.; Academic Press: London, UK, 1973; pp. 139–197. [Google Scholar]
- Margońska, H.B. Subtribes Malaxidinae and Liparidinae (Orchidaceae, Malaxideae)—Taxonomic divergency as effect of pollination isolation. In Proceedings of the 18th European Orchid Council Conference and Exhibition—What Future for Orchids? Paris, France, 23–25 March 2018; volume 9, pp. 68–69. [Google Scholar]
- Kowalkowska, A.K.; Margońska, H.B.; Kozieradzka-Kiszkurno, M.; Bohdanowicz, J. Studies on the ultrastructure of a three spurred fumeauxiana form of Anacamptis pyramidalis. Plant Syst. Evol. 2012, 298, 1025–1035. [Google Scholar] [CrossRef] [Green Version]
- Pacini, E.; Nepi, M. Nectar production and presentation. In Nectaries and Nectar; Nicolson, S.W., Nepi, M., Pacini, E., Eds.; Springer: Rotterdam, The Netherlands, 2007; pp. 167–214. [Google Scholar]
- Stpiczyńska, M.; Davies, K.L. Evidence for the dual role of floral secretory cells in Bulbophyllum. Acta Biol. Crac. Ser. Bot. 2016, 58, 57–69. [Google Scholar] [CrossRef] [Green Version]
- Pais, M.; Figueiredo, A.C.S. Floral nectaries from Limodorum abortivum (L.) Sw. and Epipactis atropurpurea Rafin (Orchidaceae): Ultrastructural changes in plastids during the secretory process. Apidologie 1994, 25, 615–626. [Google Scholar] [CrossRef] [Green Version]
- Wiśniewska, N.; Lipińska, M.M.; Gołębiowski, M.; Kowalkowska, A.K. Labellum structure of Bulbophyllum echinolabium J.J. Sm. (section Lepidorhiza Schltr., Bulbophyllinae Schltr., Orchidaceae Juss.). Protoplasma 2019, 256, 1185–1203. [Google Scholar] [CrossRef] [Green Version]
- Święczkowska, E.; Kowalkowska, A.K. Floral nectary anatomy and ultrastructure in mycoheterotrophic plant, Epipogium aphyllum Sw. (Orchidaceae). Sci. World J. 2015, 2015, 201702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parham, R.A.; Kaustinen, H.M. On the site of tannin synthesis in plant cells. Bot. Gazet. 1977, 138, 465–467. [Google Scholar] [CrossRef]
- Kowalkowska, A.K.; Kozieradzka-Kiszkurno, M.; Turzyński, S. Morphological, histological and ultrastructural features of osmophores and nectary of Bulbophyllum wendlandianum (Kraenzl.) Dammer (B. section Cirrhopetalum Lindl., Bulbophyllinae Schltr., Orchidaceae). Plant Syst. Evol. 2015, 301, 609–622. [Google Scholar] [CrossRef] [Green Version]
- Kowalkowska, A.K.; Turzyński, S.; Kozieradzka-Kiszkurno, M.; Wiśniewska, N. Floral structure of two species of Bulbophyllum section Cirrhopetalum Lindl: B. weberi Ames and B. cumingii (Lindl.) Rchb. f. (Bulbophyllinae Schltr., Orchidaceae). Protoplasma 2017, 254, 143–149. [Google Scholar] [CrossRef]
- Franceschi, V.R.; Nakata, P.A. Calcium oxalate in plants: Formation and function. Annu. Rev. Plant Biol. 2005, 56, 41–71. [Google Scholar] [CrossRef] [PubMed]
- Meve, U.; Liede, S. Floral biology and pollination in stapeliads—New results and a literature review. Plant Syst. Evol. 1994, 192, 99–116. [Google Scholar] [CrossRef]
- Kowalkowska, A.K.; Margońska, H.B. Diversity of labellar micromorphological structures in selected species of Malaxidinae (Orchidales). Acta Soc. Bot. Polon. 2009, 78, 141–150. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, A.F.M.; Meirelles, S.T.; Salatino, A. Epicuticular waxes from caatinga and cerrado species and their efficiency against water loss. An. Acad. Bras. Ciênc. 2003, 75, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Ginzel, M.D.; Moreira, J.A.; Ray, A.M.; Millar, J.G.; Hanks, L.M. (Z)-9-Nonacosene—Major component of the contact sex pheromone of the beetle Megacyllene caryae. J. Chem. Ecol. 2006, 32, 435–451. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Han, R.; Wang, Y.; Li, X.; Zhang, M.; Yan, Y. Fungicidal activity of a medium-chain fatty acids mixture comprising caprylic, pelargonic and capric acids. Plant Pathol. J. 2014, 13, 65–70. [Google Scholar] [CrossRef]
- Jensen, W.A. Botanical Histochemistry; Freeman: San Francisco, CA, USA, 1962; pp. 1–408. [Google Scholar]
- Bronner, R. Simultaneous demonstration of lipid and starch in plant tissues. Stain. Technol. 1975, 50, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, E.S. The use of lead citrate at high pH as an electron- opaque stain in electron microscopy. J. Cell Biol. 1963, 17, 208–212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz-Matute, A.I.; Soria, A.C.; Martinez-Castro, I.; Sanz, M.L. A new methodology based on GC-MS to detect honey adulteration with commercial syrups. J. Agric. Food Chem. 2007, 55, 7264–7269. [Google Scholar] [CrossRef] [PubMed]





| Compound | C. hoi | C. acutangulum | C. luniferum | |||
|---|---|---|---|---|---|---|
| Water | Methanol | Water | Methanol | Water | Methanol | |
| Fructose | 53.82 | 34.76 | 27.27 | 39.07 | 29.22 | Nd |
| Glucose | 46.18 | 25.49 | 3.97 | 15.05 | 29.78 | Nd |
| Sucrose | Nd | 39.75 | 68.75 | 45.89 | 41.00 | 100.00 |
| Compound | C. hoi | C. acutangulum | C. luniferum |
|---|---|---|---|
| Glycerol | 11.40 | 13.44 | 11.45 |
| Octadecanol | 0.12 | ||
| Tetracosanol | 3.08 | 0.75 | |
| Pentacosanol | 0.40 | ||
| Hexacosanol | 5.12 | ||
| Heptacosanol | 0.75 | ||
| Octacosanol | 7.24 | 0.68 | |
| Nonacosanol | 0.68 | ||
| Triacontanol | 4.37 | 1.64 | |
| Total alcohols | 33.16 | 13.44 | 14.52 |
| Nonanoic acid | 0.38 | 1.92 | 1.47 |
| Decanoic acid | 0.11 | 0.53 | |
| Dodecanoic acid | 0.15 | ||
| Tetradecanoic acid | 0.37 | 0.29 | 0.37 |
| Pentadecanoic acid | 0.18 | 0.47 | |
| Hexadecanoic acid* | 1.16 | 15.62 | 22.06 |
| Octadecadienoic acid | 0.25 | 0.77 | 2.28 |
| Octadecaenoic acid | 0.74 | 2.82 | |
| Octadecanoic acid* | 0.35 | 26.73 | 20.04 |
| Tetracosanoic acid | 0.60 | ||
| Octacosanoic acid | 1.13 | ||
| Triacontanoic acid | 2.84 | ||
| Dotriacontanoic acid | 1.26 | ||
| Total fatty acids | 2.93 | 46.61 | 55.35 |
| Tetradecane | 1.48 | 1.47 | |
| Pentadecane | |||
| Hexadecane | 0.71 | 2.03 | 1.49 |
| Octadecane | 0.65 | 1.50 | 1.08 |
| Eicosane | 0.31 | ||
| Docosane | 0.41 | ||
| Tricosene | 1.17 | 0.90 | |
| Tricosane | 0.93 | 1.72 | 0.69 |
| Tetracosane | 1.03 | ||
| Pentacosene | 0.69 | 0.79 | 0.32 |
| Pentacosane | 4.29 | 3.38 | 1.01 |
| Hexacosane | 1.37 | 1.78 | 0.67 |
| Heptacosene | 1.60 | 0.47 | 0.58 |
| Heptacosane | 14.56 | 10.75 | 2.77 |
| Octacosane | 2.61 | 2.83 | 1.76 |
| Nonacosene | 1.70 | 1.25 | 1.43 |
| Nonacosane | 13.58 | 5.40 | 6.00 |
| Triacontane | 0.63 | ||
| Hentriacontane | 0.72 | ||
| Total hydrocarbons (saturated) (unsaturated) | 45.93 40.77 5.16 | 34.41 31.89 2.51 | 20.18 16.95 3.22 |
| Campesterol | 3.62 | 1.12 | |
| 5α-ergost-8(14)-en-3β-ol | 0.86 | ||
| Stigmasterol | 1.03 | 1.62 | 4.66 |
| Sitosterol | 2.97 | 3.93 | 2.50 |
| Sitostanol | 4.37 | ||
| 24-Methylenecycloartanol | 1.68 | ||
| Total sterols | 12.85 | 5.55 | 9.96 |
| Unidentified | 5.12 |
| Species of Crepidium | ||||||||
|---|---|---|---|---|---|---|---|---|
| Character | C. acutangulum | C. hoi | C. luniferum | C. resupinatum | C. rheedii | C. metallicum | C. taurinum | C. tixierii |
| Where observed | in situ ex situ | in situ | in situ ex situ | in situ ex situ | in situ ex situ | in situ ex situ | in situ | in situ |
| Diameter of flowers (cm) | 0.80–1.20 | ca. 0.60 | 0.47–0.56 | 0.80–1.20 | 0.50–0.70 | 0.90–1.40 | 0.55–0.95 | 0.42–0.55 |
| Relative size of flowers for genus | large | Small | small | large | small | large | medium | small |
| Main colour of mature, adult flower | white | yellow, pink-purple centrally | white | dark red to maroon, violet | purple to purple-red | cream-yellow to purple | yellow, ochre, reddish to purplish | green |
| Scent perceptible to humans | − | − | + | − | − | − | − | − |
| Labellar sectretion | + | + | + | + | + | + | + | + |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Margońska, H.B.; Kozieradzka-Kiszkurno, M.; Brzezicka, E.; Haliński, Ł.P.; Davies, K.L.; Lipińska, M.M. Crepidium sect. Crepidium (Orchidaceae, Malaxidinae)—Chemical and Morphological Study of Flower Structures in the Context of Pollination Processes. Plants 2021, 10, 2373. https://doi.org/10.3390/plants10112373
Margońska HB, Kozieradzka-Kiszkurno M, Brzezicka E, Haliński ŁP, Davies KL, Lipińska MM. Crepidium sect. Crepidium (Orchidaceae, Malaxidinae)—Chemical and Morphological Study of Flower Structures in the Context of Pollination Processes. Plants. 2021; 10(11):2373. https://doi.org/10.3390/plants10112373
Chicago/Turabian StyleMargońska, Hanna B., Małgorzata Kozieradzka-Kiszkurno, Emilia Brzezicka, Łukasz P. Haliński, Kevin L. Davies, and Monika M. Lipińska. 2021. "Crepidium sect. Crepidium (Orchidaceae, Malaxidinae)—Chemical and Morphological Study of Flower Structures in the Context of Pollination Processes" Plants 10, no. 11: 2373. https://doi.org/10.3390/plants10112373
APA StyleMargońska, H. B., Kozieradzka-Kiszkurno, M., Brzezicka, E., Haliński, Ł. P., Davies, K. L., & Lipińska, M. M. (2021). Crepidium sect. Crepidium (Orchidaceae, Malaxidinae)—Chemical and Morphological Study of Flower Structures in the Context of Pollination Processes. Plants, 10(11), 2373. https://doi.org/10.3390/plants10112373








