Development of a Real-Time Loop-Mediated Isothermal Amplification Assay for the Rapid Detection of Olea Europaea Geminivirus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material Collection
2.2. DNA Extraction and Sample Preparation
2.3. Preliminary Screening of OEGV by End-Point PCR
2.4. LAMP Primers Design
2.5. OEGV Real-Time LAMP Assay Optimization
2.6. Features of Real-Time LAMP Assay: Sensitivity and Comparison to Conventional PCR, Reaction Time and Specificity
2.7. Set up of a Rapid Sample Preparation Method Suitable for the Real-Time LAMP Assay
2.8. Spread of OEGV in Different Cultivars
3. Results
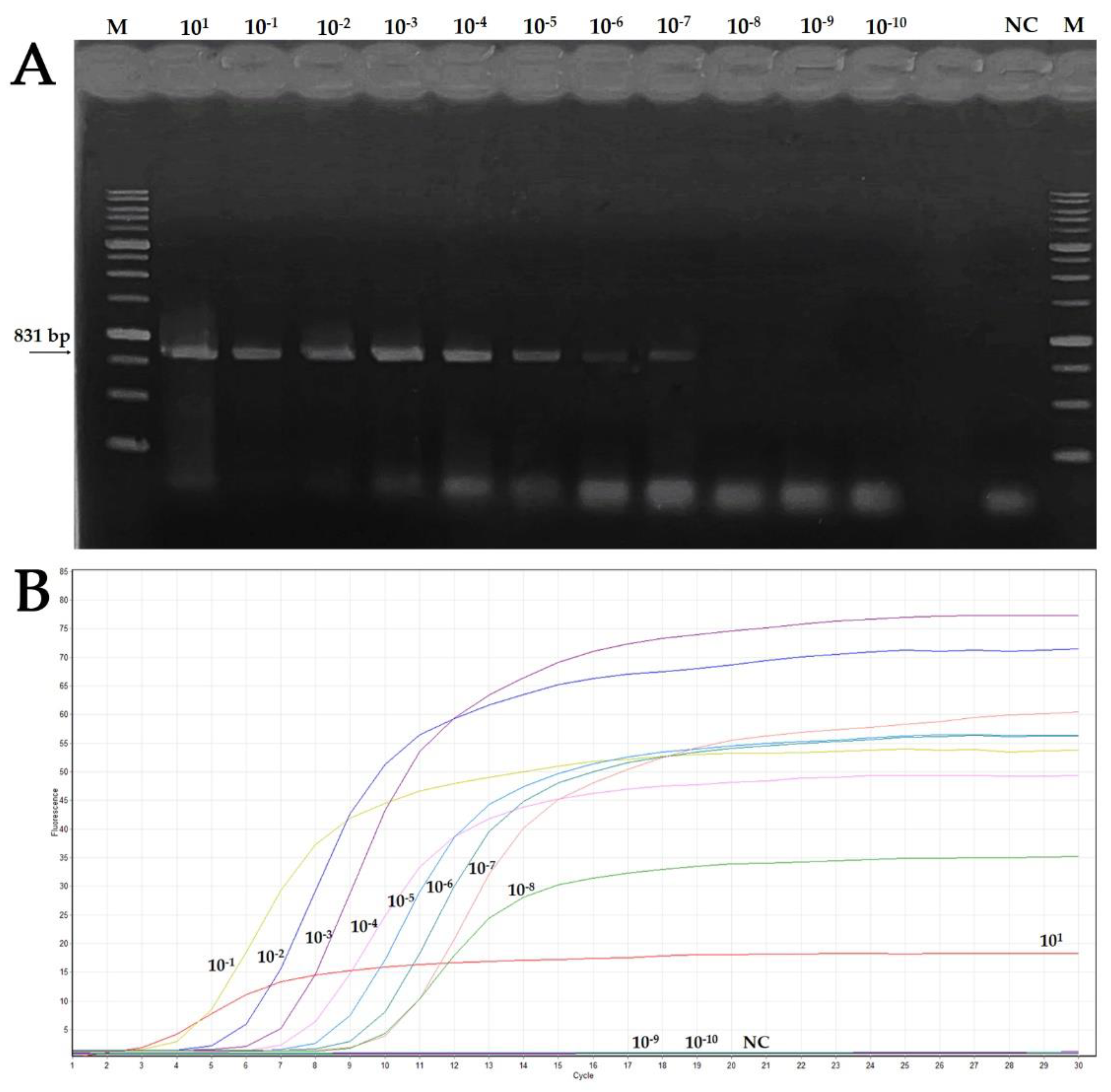
3.1. OEGV Detection by End-Point PCR
3.2. OEGV Real-Time LAMP Primer Design
3.3. OEGV Real-Time LAMP Assay Optimization
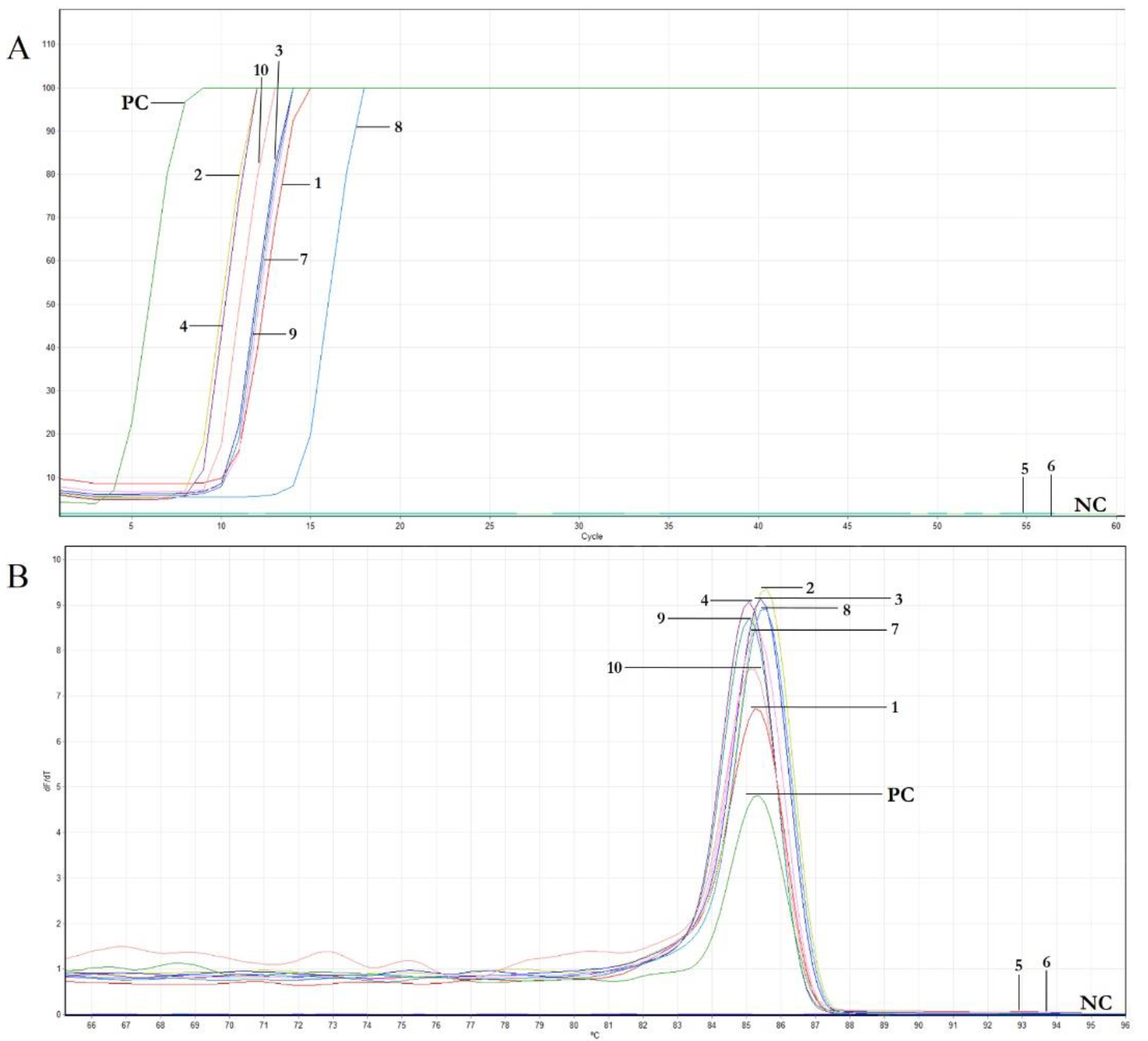
3.4. Features of Real-Time LAMP Assay: Sensitivity and Comparison to Conventional PCR, Reaction Time and Specificity
3.5. Set up of a Rapid Sample Preparation Method Suitable for the Real-Time LAMP Assay
3.6. Spread of OEGV in Sicily
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Terral, J.F. Exploitation and management of the olive tree during prehistoric times in Mediterranean France and Spain. J. Archaeol. Sci. 2000, 27, 127–133. [Google Scholar] [CrossRef]
- Goor, A. The place of the olive in the holy land and its history through the ages. Econ. Bot. 1966, 20, 223–243. [Google Scholar] [CrossRef]
- de Graaff, J.; Eppink, L.A.A.J. Olive oil production and soil conservation in southern Spain, in relation to EU subsidy policies. Land Use Policy 1999, 16, 259–267. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). Available online: http://www.fao.org/faostat/en/#home (accessed on 8 January 2022).
- ISTAT–Istituto Nazionale di Statistica–Agricoltura. Available online: https://www.istat.it/it/agricoltura/dati (accessed on 8 January 2022).
- Caruso, T.; Marra, F.P.; Costa, F.; Campisi, G.; Macaluso, L.; Marchese, A. Genetic diversity and clonal variation within the main Sicilian olive cultivars based on morphological traits and microsatellite markers. Sci. Hortic. 2014, 180, 130–138. [Google Scholar] [CrossRef]
- Las Casas, G.; Scollo, F.; Distefano, G.; Continella, A.; Gentile, A.; La Malfa, S. Molecular characterization of olive (Olea europaea L.) Sicilian cultivars using SSR markers. Biochem. Syst. Ecol. 2014, 57, 15–19. [Google Scholar] [CrossRef]
- Marra, F.P.; Caruso, T.; Costa, F.; Di Vaio, C.; Mafrica, R.; Marchese, A. Genetic relationships, structure and parentage simulation among the olive tree (Olea europaea L. subsp. europaea) cultivated in Southern Italy revealed by SSR markers. Tree Genet. Genomes 2013, 9, 961–973. [Google Scholar] [CrossRef]
- Faggioli, F.; Ferretti, L.; Albanese, G.; Sciarroni, R.; Pasquini, G.; Lumia, V.; Barba, M. Distribution of olive tree viruses in Italy as revealed by one-step RT-PCR. J. Plant Pathol. 2005, 87, 49–55. [Google Scholar]
- Piccolo, S.L.; Mondello, V.; Giambra, S.; Conigliaro, G.; Torta, L.; Burruano, S. Arthrinium phaeospermum, Phoma cladoniicola and Ulocladium consortiale, New Olive Pathogens in Italy. J. Phytopathol. 2014, 162, 258–263. [Google Scholar] [CrossRef]
- Saponari, M.; Boscia, D.; Nigro, F.; Martelli, G.P. Identification of DNA sequences related to Xylella fastidiosa in oleander, almond and olive trees exhibiting leaf scorch symptoms in Apulia (Southern Italy). J. Plant Pathol. 2013, 95. [Google Scholar] [CrossRef]
- Martinelli, F.; Marchese, A.; Giovino, A.; Marra, F.P.; Della Noce, I.; Caruso, T.; Dandekar, A.M. In-field and early detection of Xylella fastidiosa infections in olive using a portable instrument. Front. Plant Sci. 2019, 9, 2007. [Google Scholar] [CrossRef]
- Morelli, M.; García-Madero, J.M.; Jos, Á.; Saldarelli, P.; Dongiovanni, C.; Kovacova, M.; Saponari, M.; Baños Arjona, A.; Hackl, E.; Webb, S.; et al. Xylella fastidiosa in Olive: A Review of Control Attempts and Current Management. Microorganisms 2021, 9, 1771. [Google Scholar] [CrossRef]
- Xylogianni, E.; Margaria, P.; Knierim, D.; Sareli, K.; Winter, S.; Chatzivassiliou, E.K. Virus Surveys in Olive Orchards in Greece Identify Olive Virus T, a Novel Member of the Genus Tepovirus. Pathogens 2021, 10, 574. [Google Scholar] [CrossRef] [PubMed]
- Materatski, P.; Jones, S.; Patanita, M.; Campos, M.D.; Dias, A.B.; Félix, M.D.R.; Varanda, C.M. A Bipartite Geminivirus with a Highly Divergent Genomic Organization Identified in Olive Trees May Represent a Novel Evolutionary Direction in the Family Geminiviridae. Viruses 2021, 13, 2035. [Google Scholar] [CrossRef] [PubMed]
- Chiumenti, M.; Greco, C.; De Stradis, A.; Loconsole, G.; Cavalieri, V.; Altamura, G.; Zicca, S.; Saldarelli, P.; Saponari, M. Olea Europaea Geminivirus: A Novel Bipartite Geminivirid Infecting Olive Trees. Viruses 2021, 13, 481. [Google Scholar] [CrossRef]
- Alabi, O.J.; Diaz-Lara, A.; Erickson, T.M.; Al Rwahnih, M. Olea europaea geminivirus is present in a germplasm repository and in California and Texas olive (Olea europaea L.) groves. Arch. Virol. 2021, 166, 3399–3404. [Google Scholar] [CrossRef]
- Ruiz-García, A.B.; Canales, C.; Morán, F.; Ruiz-Torres, M.; Herrera-Mármol, M.; Olmos, A. Characterization of Spanish Olive Virome by High Throughput Sequencing Opens New Insights and Uncertainties. Viruses 2021, 13, 2233. [Google Scholar] [CrossRef] [PubMed]
- ICTV–International Committee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/taxonomy/ (accessed on 8 January 2022).
- Ma, Y.; Navarro, B.; Zhang, Z.; Lu, M.; Zhou, X.; Chi, S.; Di Serio, F.; Li, S. Identification and molecular characterization of a novel monopartite geminivirus associated with mulberry mosaic dwarf disease. J. Gen. Virol. 2015, 96, 2421–2434. [Google Scholar] [CrossRef] [PubMed]
- Gottwald, T.R.; Hughes, G. A new survey method for Citrus tristeza virus disease assessment. In Proceedings of the XIV International Organization of Citrus Virologists (IOCV), Sao Paulo, Brazil, 14–21 July 2000; pp. 77–87. [Google Scholar]
- Davino, S.; Panno, S.; Arrigo, M.; La Rocca, M.; Caruso, A.G.; Bosco, G.L. Planthology: An application system for plant diseases management. Chem. Eng. Trans. 2017, 58, 619–624. [Google Scholar] [CrossRef]
- Panno, S.; Caruso, A.G.; Troiano, E.; Luigi, M.; Manglli, A.; Vatrano, T.; Iacono, G.; Marchione, S.; Bertin, S.; Tomassoli, L.; et al. Emergence of tomato leaf curl New Delhi virus in Italy: Estimation of incidence and genetic diversity. Plant Pathol. 2019, 68, 601–608. [Google Scholar] [CrossRef]
- Davino, S.; Napoli, C.; Dellacroce, C.; Miozzi, L.; Noris, E.; Davino, M.; Accotto, G.P. Two new natural begomovirus recombinants associated with the tomato yellow leaf curl disease co-exist with parental viruses in tomato epidemics in Italy. Virus Res. 2009, 143, 15–23. [Google Scholar] [CrossRef]
- Davino, S.; Napoli, C.; Davino, M.; Accotto, G.P. Spread of Tomato yellow leaf curl virus in Sicily: Partial displacement of another geminivirus originally present. Eur. J. Plant Pathol. 2006, 114, 293–299. [Google Scholar] [CrossRef]
- Panno, S.; Caruso, A.G.; Davino, S. The nucleotide sequence of a recombinant tomato yellow leaf curl virus strain frequently detected in Sicily isolated from tomato plants carrying the Ty-1 resistance gene. Arch. Virol. 2018, 163, 795–797. [Google Scholar] [CrossRef] [PubMed]
- Sabanadzovic, S.; Abou-Ghanem, N.; La Notte, P.; Savino, V.; Scarito, G.; Martelli, G.P. Partial molecular characterization and RT-PCR detection of a putative closterovirus associated with olive leaf yellowing. J. Plant Pathol. 1999, 81, 37–45. [Google Scholar]
- Varanda, C.M.; Santos, S.; Clara, M.I.E.; do Rosário Félix, M. Olive mild mosaic virus transmission by Olpidium virulentus. Eur. J. Plant Pathol. 2015, 142, 197–201. [Google Scholar] [CrossRef] [Green Version]
- Martelli, G. A brief outline of infectious diseases of olive. Palest. Tech. Univ. Res. J. 2013, 1, 10. [Google Scholar] [CrossRef]
- Martelli, G.P. Infectious diseases and certification of olive: An overview. EPPO Bull. 1999, 29, 127–133. [Google Scholar] [CrossRef]
- Loconsole, G.; Saponari, M.; Faggioli, F.; Albanese, G.; Bouyahia, H.; Elbeaino, T.; Materazzi, A.; Nuzzaci, M.; Prota, V.; Romanazzi, G.; et al. Inter-laboratory validation of PCR-based protocol for detection of olive viruses. EPPO Bull. 2010, 40, 423–428. [Google Scholar] [CrossRef]
- Albanese, G.; Saponari, M.; Faggioli, F. Phytosanitary certification. In Olive Germoplasm-The Olive Cultivation, Table Olive and Olive Oil Industry in Italy, 1st ed.; Muzzalupo, I., Ed.; InTech: Rijeka, Croatia, 2012; pp. 107–132. [Google Scholar]
- Roschetti, A.; Ferretti, L.; Muzzalupo, I.; Pellegrini, F.; Albanese, G.; Faggioli, F. Evaluation of the possibile effect of virus infections on olive propagation. Petria 2009, 19, 18–28. [Google Scholar]
- Pesante, A. On a previously unknown disease of olive. Boll. Della Stn. Patol. Veg. Roma 1938, 18, 401–428. [Google Scholar]
- Caglayan, K.; Fidan, U.; Tarla, G.; Gazel, M. First report of olive viruses in Turkey. J. Plant Pathol. 2004, 86, 91. [Google Scholar]
- Barba, M. Viruses and virus-like diseases of olive 1. EPPO Bull. 1993, 23, 493–497. [Google Scholar] [CrossRef]
- Savino, V.; Barba, M.; Galitelli, G.; Martelli, G.P. Two nepoviruses isolated from olive in Italy. Phytopathol. Mediterr. 1979, 18, 135–142. [Google Scholar]
- Savino, V.; Gallitelli, D. Cherry leafroll virus in olive. Phytopathol. Mediterr. 1981, 20, 202–203. [Google Scholar]
- Savino, V.; Gallitelli, D.; Barba, M. Olive latent ringspot virus, a newly recognised virus infecting olive in Italy. Ann. Appl. Boil. 1983, 103, 243–249. [Google Scholar] [CrossRef]
- Martelli, G.P.; Sabanadzovic, S.; Savino, V.; Abu-Zurayk, A.R.; Masannat, M.C.F.A. Virus-like diseases and viruses of olive in Jordan. Phytopathol. Mediterr. 1995, 34, 133–136. [Google Scholar]
- Ortega, S.F.; Tomlinson, J.; Hodgetts, J.; Spadaro, D.; Gullino, M.L.; Boonham, N. Development of loop-mediated isothermal amplification assays for the detection of seedborne fungal pathogens Fusarium fujikuroi and Magnaporthe oryzae in rice seed. Plant Dis. 2018, 102, 1549–1558. [Google Scholar] [CrossRef] [Green Version]
- Panno, S.; Matić, S.; Tiberini, A.; Caruso, A.G.; Bella, P.; Torta, L.; Stassi, R.; Davino, S. Loop mediated isothermal amplification: Principles and applications in plant virology. Plants 2020, 9, 461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, S.; Nakamura, K.; Kayahara, H. Analysis of phenolic compounds in white rice, brown rice, and germinated brown rice. J. Agric. Food Chem. 2004, 543, 4808–4813. [Google Scholar] [CrossRef] [PubMed]
- Schrader, C.; Schielke, A.; Ellerbroek, L.; Johne, R. PCR inhibitors–occurrence, properties and removal. J. Appl. Microbial. 2012, 113, 1014–1026. [Google Scholar] [CrossRef]
- Boonham, N.; Glover, R.; Tomlinson, J.; Mumford, R. Exploiting generic platform technologies for the detection and identification of plant pathogens. Sustain. Dis. Manag. Eur. Context 2008, 121, 355–363. [Google Scholar] [CrossRef]
- Kaneko, H.; Kawana, T.; Fukushima, E.; Suzutani, T. Tolerance of loop-mediated isothermal amplification to a culture medium.and biological substances. J. Biochem. Biophys. Methods 2007, 70, 499–501. [Google Scholar] [CrossRef] [PubMed]
- Panno, S.; Ruiz-Ruiz, S.; Caruso, A.G.; Alfaro-Fernandez, A.; San Ambrosio, M.I.F.; Davino, S. Real-time reverse transcription polymerase chain reaction development for rapid detection of Tomato brown rugose fruit virus and comparison with other techniques. PeerJ 2019, 7, e7928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davino, S.; Panno, S.; Rangel, E.A.; Davino, M.; Bellardi, M.G.; Rubio, L. Population genetics of cucumber mosaic virus infecting medicinal, aromatic and ornamental plants from northern Italy. Arch. Virol. 2012, 157, 739–745. [Google Scholar] [CrossRef]
- Davino, S.; Panno, S.; Iacono, G.; Sabatino, L.; D’Anna, F.; Iapichino, G.; Olmos, A.; Scuderi, G.; Rubio, L.; Tomassoli, L.; et al. Genetic variation and evolutionary analysis of Pepino mosaic virus in Sicily: Insights into the dispersion and epidemiology. Plant Pathol. 2017, 66, 368–375. [Google Scholar] [CrossRef] [Green Version]
- Ferriol, I.; Rubio, L.; Pérez-Panadés, J.; Carbonell, E.A.; Davino, S.; Belliure, B. Transmissibility of Broad bean wilt virus 1 by aphids: Influence of virus accumulation in plants, virus genotype and aphid species. Ann. Appl. Biol. 2013, 162, 71–79. [Google Scholar] [CrossRef]
- Tiberini, A.; Tomlinson, J.; Micali, G.; Fontana, A.; Albanese, G.; Tomassoli, L. Development of a reverse transcription-loop-mediated isothermal amplification (LAMP) assay for the rapid detection of Onion yellow dwarf virus. J. Virol. Methods 2019, 271, 113680. [Google Scholar] [CrossRef] [PubMed]
- Waliullah, S.; Ling, K.S.; Cieniewicz, E.J.; Oliver, J.E.; Ji, P.; Ali, M.E. Development of loop-mediated isothermal amplification assay for rapid detection of Cucurbit leaf crumple virus. Int. J. Mol. Sci. 2020, 21, 1756. [Google Scholar] [CrossRef] [Green Version]
- Jeger, M.J.; Holt, J.; Van Den Bosch, F.; Madden, L.V. Epidemiology of insect-transmitted plant viruses: Modelling disease dynamics and control interventions. Physiol. Entomol. 2004, 29, 291–304. [Google Scholar] [CrossRef]
- Davino, S.; Calari, A.; Davino, M.; Tessitori, M.; Bertaccini, A.; Bellardi, M.G. Virescence of tenweeks stock associated to phytoplasma infection in Sicily. Bull. Insectol. 2007, 60, 279–280. [Google Scholar]
- Panno, S.; Ferriol, I.; Rangel, E.A.; Olmos, A.; Han, C.; Martinelli, F.; Rubio, L.; Davino, S. Detection and identification of Fabavirus species by one-step RT-PCR and multiplex RT-PCR. J. Virol. Methods 2014, 197, 77–82. [Google Scholar] [CrossRef]
- Panno, S.; Caruso, A.G.; Barone, S.; Lo Bosco, G.; Rangel, E.A.; Davino, S. Spread of tomato brown rugose fruit virus in Sicily and evaluation of the spatiotemporal dispersion in experimental conditions. Agronomy 2020, 10, 834. [Google Scholar] [CrossRef]

| Cultivar | No. Positive/Tested Samples | Percentage of Positive Samples (%) |
|---|---|---|
| Cavalieri Standard | 8/8 | 100 |
| Cerasuola Nilo Paceco | 8/8 | 100 |
| Cerasuola Standard | 8/8 | 100 |
| Giarraffa | 0/8 | 0 |
| Nocellara del Belice Giafalione | 8/8 | 100 |
| Pizzutella | 8/8 | 100 |
| Salicina Vassallo | 3/8 | 37.5 |
| Uovo di piccione | 1/8 | 12.5 |
| Vaddara | 0/8 | 0 |
| Zaituna Florida | 0/8 | 0 |
| Total | 44/80 | 55 |
| Primer Name | Sequence (5′-3′) | Amplicon Size (bp) |
|---|---|---|
| F3-OEGV | CGATACGAGACATACCCAG | 209 |
| B3-OEGV | TCCATGTTGATCATCCAAGT | |
| FIP-OEGV | CAGCCACTGCTTCATATTATGAACACGAATTGTGCTTAACGGTT | - |
| BIP-OEGV | GATGTGGCTCGTGTATGATAGACGTCTGGATCCCGACTTTCC | |
| LF-OEGV | GGCTTCGCTAGTCAACTTAACTG | - |
| LB-OEGV | TCCCGGTAATTCTAATCCCAGAG |
| Cultivar | No. of Different Samples Analyzed | ID Sample | Reaction Time (min) |
|---|---|---|---|
| Cavalieri Standard | 2 | 1 | 10 |
| 2 | 7 | ||
| Cerasuola Standard | 2 | 3 | 10 |
| 4 | 7 | ||
| Giarraffa | 2 | 5 | - |
| 6 | - | ||
| Nocellara del Belice Giafalione | 2 | 7 | 10 |
| 8 | 13 | ||
| Pizzutella | 2 | 9 | 10 |
| 10 | 9 | ||
| Positive control | 1 | PC | 3 |
| Negative control | 1 | NC | - |
| Starting DNA Concentration (80.9 ng/μL) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Assay | 101 | 10−1 | 10−2 | 10−3 | 10−4 | 10−5 | 10−6 | 10−7 | 10−8 | 10−9 | 10−10 |
| End-point PCR | + | + | + | + | + | + | + | + | − | − | − |
| Real-time LAMP | + | + | + | + | + | + | + | + | + | − | − |
| Reaction Time (min) | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 10 | − | − |
| Cultivar | No. Samples Analyzed | Time Value | ||
|---|---|---|---|---|
| ID Sample | Total DNA Extraction by Commercial Kit (min) | Membrane Spot Crude Extract (min) | ||
| Cavalieri Standard | 2 | 1 | 10 | 14 |
| 2 | 7 | 12 | ||
| Cerasuola Standard | 2 | 3 | 10 | 16 |
| 4 | 7 | 10 | ||
| Giarraffa | 2 | 5 | - | - |
| 6 | - | - | ||
| Nocellara del Belice Giafalione | 2 | 7 | 10 | 15 |
| 8 | 14 | 24 | ||
| Pizzutella | 2 | 9 | 10 | 16 |
| 10 | 9 | 14 | ||
| Positive control | 1 | PC | 3 | 12 |
| Negative control | 1 | NC | - | - |
| Cultivar Analyzed | Real-Time LAMP | |
|---|---|---|
| Cultivar Batch | Positive Plants/Tested Plants | |
| Abunara | + | 8/8 |
| Aitana | − | NT |
| Arbequina | + | 8/8 |
| Bariddara | + | 8/8 |
| Biancolilla Caltabellotta | − | NT |
| Biancolilla Caltabellotta TA PC | + | 8/8 |
| Biancolilla Iacapa | − | NT |
| Biancolilla Napoletana | − | NT |
| Biancolilla Pantelleria | − | NT |
| Biancolilla Schimmenti | − | NT |
| Biancolilla Siracusana | − | NT |
| Biancuzza | − | NT |
| Bottone di Gallo Vassallo | − | NT |
| Brandofino | − | NT |
| Calamignara | − | NT |
| Calatina | + | 3/8 |
| Carasuola Cappuccia | + | 8/8 |
| Castricianella Rapparina | + | 8/8 |
| Cavalieri Standard | + | 8/8 |
| Cerasuola 1 Clone 2 | + | 8/8 |
| Cerasuola Nilo Paceco | + | 8/8 |
| Cerasuola Standard | + | 8/8 |
| Conservolia | − | NT |
| Crastu Collesano | − | NT |
| Galatina | − | NT |
| Giarraffa | − | NT |
| Gordales | − | NT |
| Iacona | + | 8/8 |
| Indemoniata | − | NT |
| Koroneiki | + | 8/8 |
| Leucocarpa | − | NT |
| Lunga di Vassallo | + | 8/8 |
| Manzanilla | − | NT |
| Minna di Vacca | − | NT |
| Minuta | + | 8/8 |
| Monaca | + | 8/8 |
| Moresca | − | NT |
| Murtiddara Vassallo | + | 8/8 |
| Nasitana | + | 8/8 |
| Nocellara del Belice Giafalione | + | 8/8 |
| Nocellara del Belice Clone 1 | − | NT |
| Nocellara del Belice Clone 7 | − | NT |
| Nocellara del Belice Mazara del Vallo | − | NT |
| Nocellara del Belice Standard | − | NT |
| Nocellara Etnea | − | NT |
| Nocellara Messinese Ricciardi | − | NT |
| Nocellara Messinese Romana | − | NT |
| Ogliara Maltese | − | NT |
| Oliva Longa | − | NT |
| Olivo di Mandanici | + | 8/8 |
| Olivo di Monaci | + | 8/8 |
| Opera Pia | + | 8/8 |
| Passalunara di Lascari | − | NT |
| Picholine | − | NT |
| Piricuddara | + | 8/8 |
| Pizzo di Corvo | − | NT |
| Pizzuta d’Olio | + | 8/8 |
| Pizzutella | + | 8/8 |
| Salicina Vassallo | − | NT |
| Tonda Iblea | − | NT |
| Tortella Motticiana | − | NT |
| Tunnilidda | − | NT |
| Uovo di Piccione | − | NT |
| Vaddara | − | NT |
| Vaddarica | + | 8/8 |
| Verdella | + | 8/8 |
| Verdella Frutto Grosso | + | 8/8 |
| Verdello | + | 8/8 |
| Vetrana | + | 8/8 |
| Zaituna Floridia | − | NT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertacca, S.; Caruso, A.G.; Trippa, D.; Marchese, A.; Giovino, A.; Matic, S.; Noris, E.; Ambrosio, M.I.F.S.; Alfaro, A.; Panno, S.; et al. Development of a Real-Time Loop-Mediated Isothermal Amplification Assay for the Rapid Detection of Olea Europaea Geminivirus. Plants 2022, 11, 660. https://doi.org/10.3390/plants11050660
Bertacca S, Caruso AG, Trippa D, Marchese A, Giovino A, Matic S, Noris E, Ambrosio MIFS, Alfaro A, Panno S, et al. Development of a Real-Time Loop-Mediated Isothermal Amplification Assay for the Rapid Detection of Olea Europaea Geminivirus. Plants. 2022; 11(5):660. https://doi.org/10.3390/plants11050660
Chicago/Turabian StyleBertacca, Sofia, Andrea Giovanni Caruso, Daniela Trippa, Annalisa Marchese, Antonio Giovino, Slavica Matic, Emanuela Noris, Maria Isabel Font San Ambrosio, Ana Alfaro, Stefano Panno, and et al. 2022. "Development of a Real-Time Loop-Mediated Isothermal Amplification Assay for the Rapid Detection of Olea Europaea Geminivirus" Plants 11, no. 5: 660. https://doi.org/10.3390/plants11050660
APA StyleBertacca, S., Caruso, A. G., Trippa, D., Marchese, A., Giovino, A., Matic, S., Noris, E., Ambrosio, M. I. F. S., Alfaro, A., Panno, S., & Davino, S. (2022). Development of a Real-Time Loop-Mediated Isothermal Amplification Assay for the Rapid Detection of Olea Europaea Geminivirus. Plants, 11(5), 660. https://doi.org/10.3390/plants11050660









