1. Introduction
The most important role of seeds is to keep a species in existence. Accordingly, plants have evolved various strategies to ensure successful germination of seeds [
1]. Germination is the first step in plant life history. The success of seed germination and seedling establishment can affect features for the propagation of plant species, which are of both economic and ecologic importance [
2].
Veronicastrum sibiricum, which is endemic to Northeast Asia, is distributed in the central and northern parts of the Korean Peninsula, Far East Russia, Northeast China, Mongolia, and Northern Japan [
3].
V. sibiricum is a perennial herb with a height of 50–90 cm, long, oval leaves, and light purple flowers in racemes blooming from July to August [
3]. The species belongs to the Scrophulariaceae Juss., which includes approximately 220 genera and 4000 species, with 68 species in 25 genera distributed in South Korea. There are two forms of the same species of
Veronicastrum,
V. sibiricum and
V. sibiricum f.
albiflora T. Yamaz in Korea [
4]. The species of
Veronicastrum and its closely related genus
Veronica are widely distributed in the Northern Hemisphere, and in several regions in the Southern Hemisphere [
5]. Both
Veronicastrum and
Veronica are morphologically closely related.
Veronicastrum flowers have short calyx lobes and long corolla tube, whereas
Veronica flowers have long calyx lobes and short corolla tube [
6]. The phylogenetic analysis based on DNA sequences, seed microstructure [
7], and pollen [
8] indicate similarity between the two genera.
Traditionally, in South Korea,
V. sibiricum roots have been used to treat neuralgia, arthritis, and inflammation and the young shoots are used as edible herbs [
4]. Pharmacological studies have indicated the presence of compounds, such as isoferulic acid and 3,4-dimethoxy cinnamic acid, which have anti-inflammatory and analgesic properties [
9], diterpene, which has antioxidant and anticancer properties [
10,
11], and iridoids for treating common cold, leucorrhea, cystitis, and liver [
12].
Seed dormancy is a survival strategy, wherein germination is blocked under favorable environmental conditions [
13,
14]. Global data on seed dormancy and germination of 5250 species in major vegetation zones indicated that seeds of 69.6% of the species are dormant when freshly matured [
15]. Strategies to break seed dormancy differ between species [
16]. The seeds of many native plants in temperate regions have relatively small immature embryos [
17]. If embryo elongation and germination stages in such immature embryos are achieved within 30 d under environmental conditions favorable for germination, the dormancy type is classified as morphological dormancy (MD) [
13]. If there is an additional dormancy mechanism that inhibits the germination of immature embryos, the dormancy type is classified as morphophysiological dormancy (MPD). On the other hand, if a germination-inhibiting mechanism is added to seeds with mature embryos, the dormancy type is classified as physiological dormancy (PD) [
13,
15].
Choi [
18] and Martinez-Ortega and Rico [
19] determined that the genera
Veronica and
Veronicastrum are closely related based on the phylogeny results. Furthermore, the seeds of
Veronica parnkalliana, a species native to South Australia, have immature embryos and show MPD, but 80–90% of the seeds germinated after gibberellic acid (GA) treatment [
20]. Fifteen species of
Veronica were classified as having PD, and
V. biloba and
V. wormskjoldii were found to have non-dormant seeds [
13]. Song et al. [
21] reported that eight species of
Veronica native to the Korean Peninsula had MD or MPD. Some closely related species show the same dormancy type (trait stasis; [
22]), whereas some exhibit significant differences in the type of depth of dormancy (trait divergence; [
23]). This implies that during adaptation to different environments, dormancy patterns are either preserved or changed. Breaking seed dormancy and different germination traits are ecological characteristics and physiological control mechanisms of plants [
22].
To determine the dormancy class, it is important to establish whether seeds contain embryos that are fully developed or underdeveloped at maturity and, therefore, if the embryo must elongate inside the seed before germination. Guerin et al. [
20] and Song et al. [
21] measured embryo growth in the seeds of
V. parnkalliana and other
Veronica species. However, many previous studies have not accurately measured growth (or the absence of growth) of the embryo in the seeds of species of
Veronica and
Veronicastrum [
13]. Accurate classification of seed dormancy can provide a comprehensive understanding of the early stages of the plant life history and further facilitate efficient seed propagation. Therefore, this study aimed to investigate the seed ecology and physiological characteristics of
V. sibiricum native to the Korean peninsula, to classify its seed dormancy, and develop technology for mass propagation based on the acquired ecophysiological information. Additional experiments to analyze the dormancy class of other species, which are closely related to
V. sibiricum, were performed as a comparison.
3. Discussion
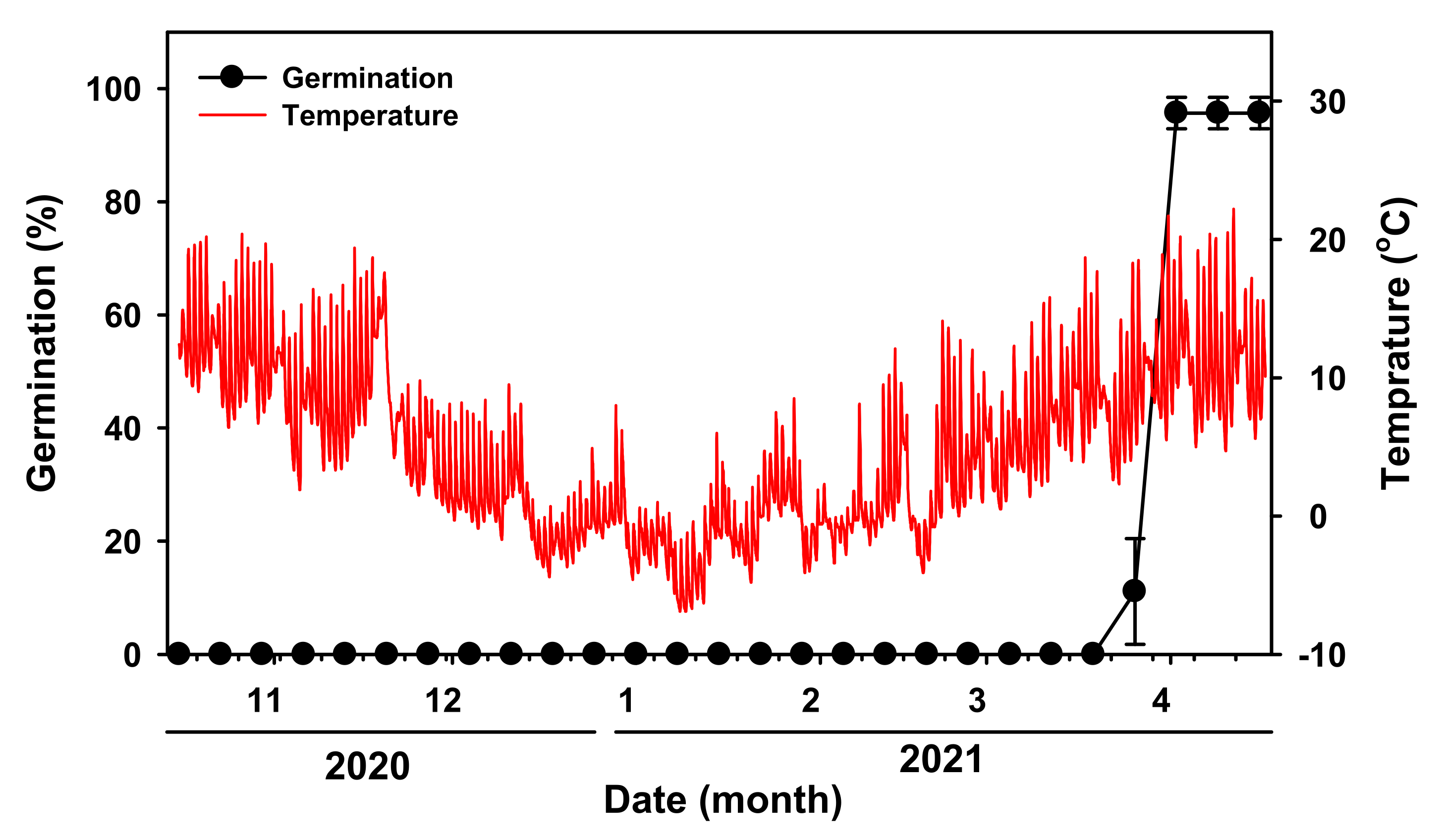
Mature
V. sibiricum seeds were sown in mid-October in phenology experiment. More than 90% of the
V. sibiricum seeds germinated by the end of March in field conditions when the average maximum temperature for one week was 13.1 °C and the minimum temperature was 4.7 °C (
Figure 3).
V. sibiricum seeds did not germinate during winter and germinated in spring of the following year. In the low-temperature cold stratification treatment, germination occurred after two weeks (
Figure 5). Therefore, in the natural conditions, the seeds may be in a state of ‘quiescence’ during winter [
13], germination did not occur until late March of the following year due to unfavorable (low) temperatures for germination.
Soil seed bank is largely divided into transient type and persistent type. Transient type I seeds germinate in the summer or autumn of the year of seed detachment, whereas transient type II seeds spend the winter of the year of seed detachment in a dormant state, and then fully germinate during spring of the following year [
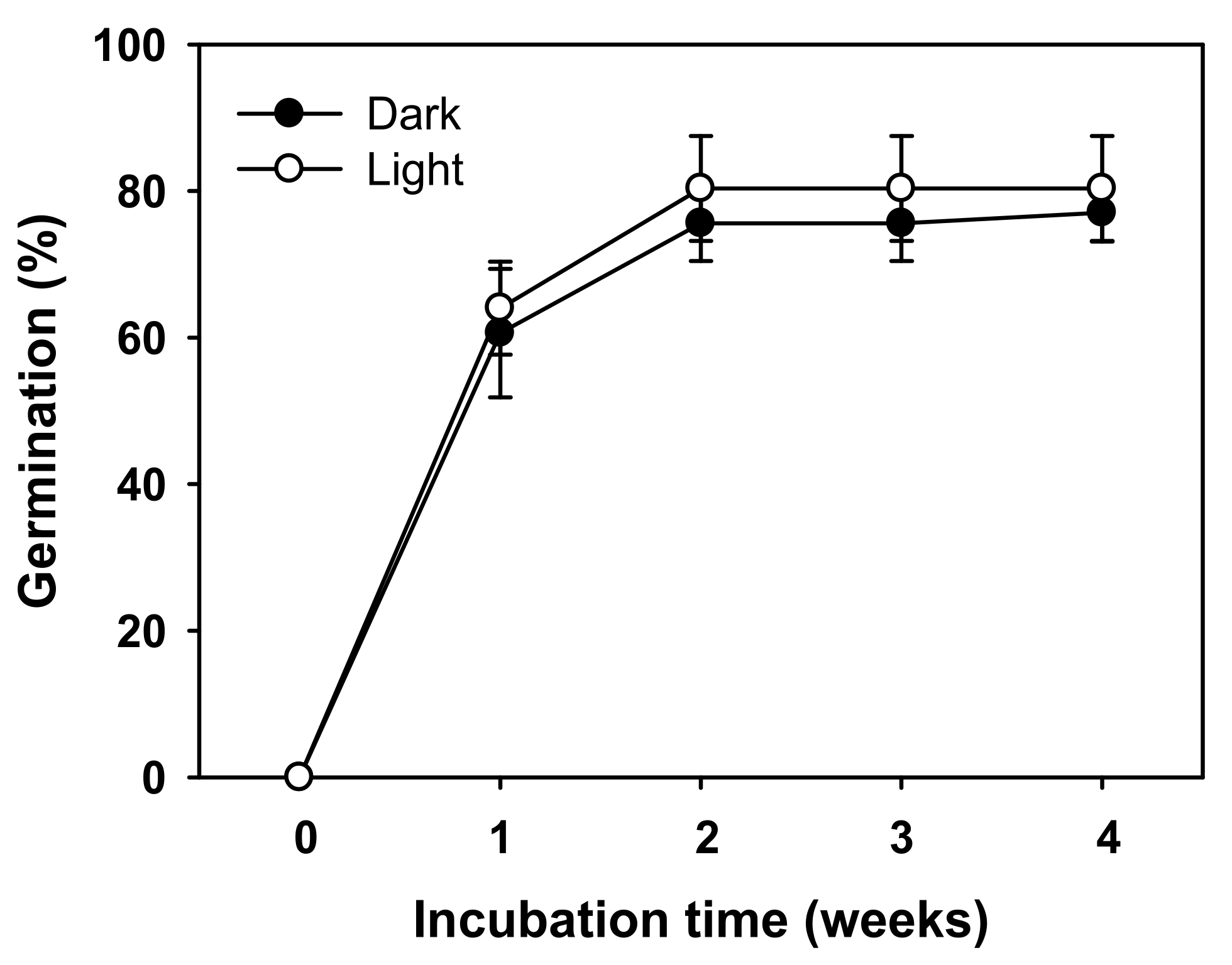
24]. After the dormancy was broken, seeds germinated under dark conditions (
Figure 7). Therefore, a short-term soil seed bank, which was classified as the transient type II, was established by suppressing seed germination in the same year.
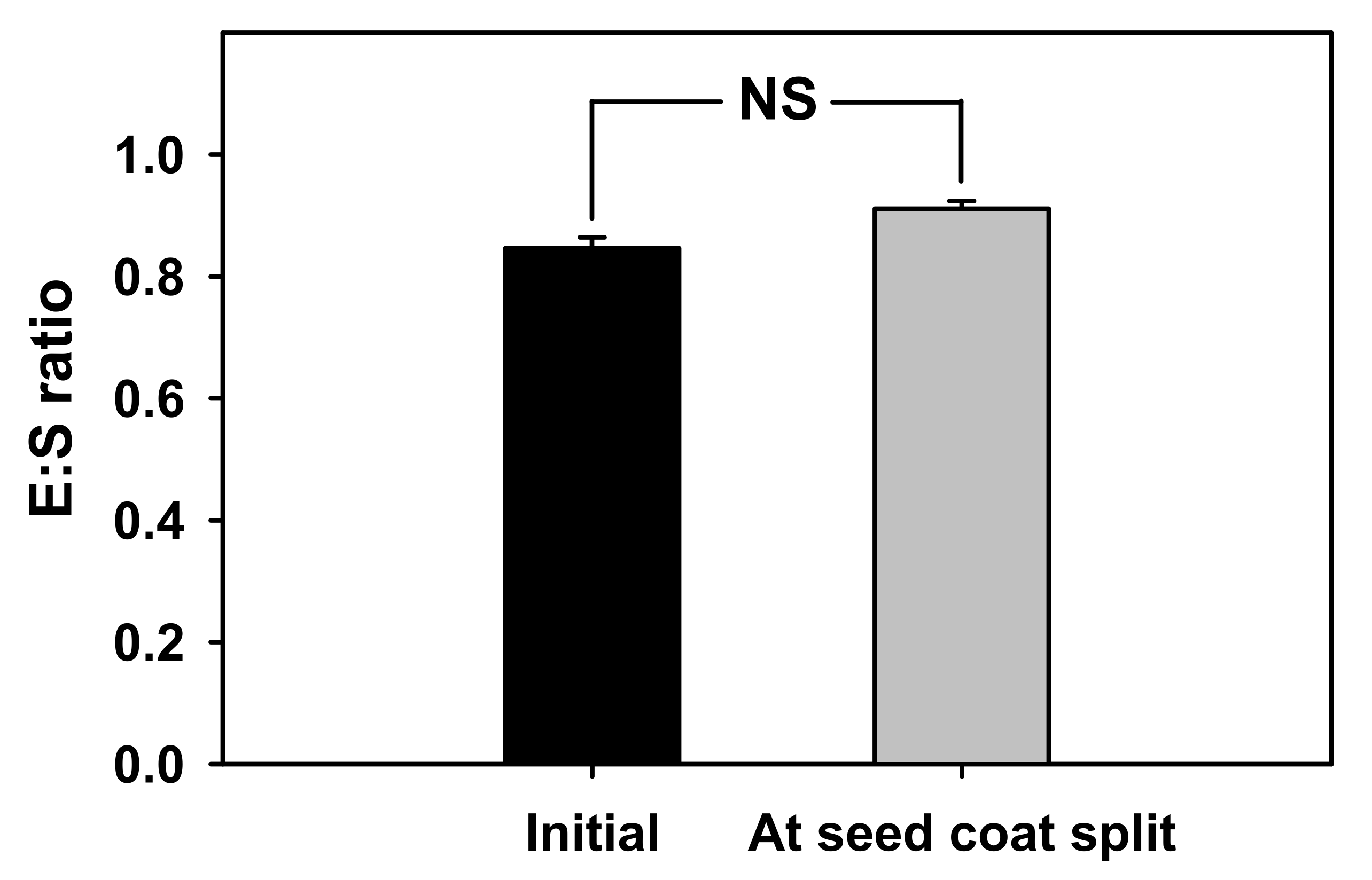
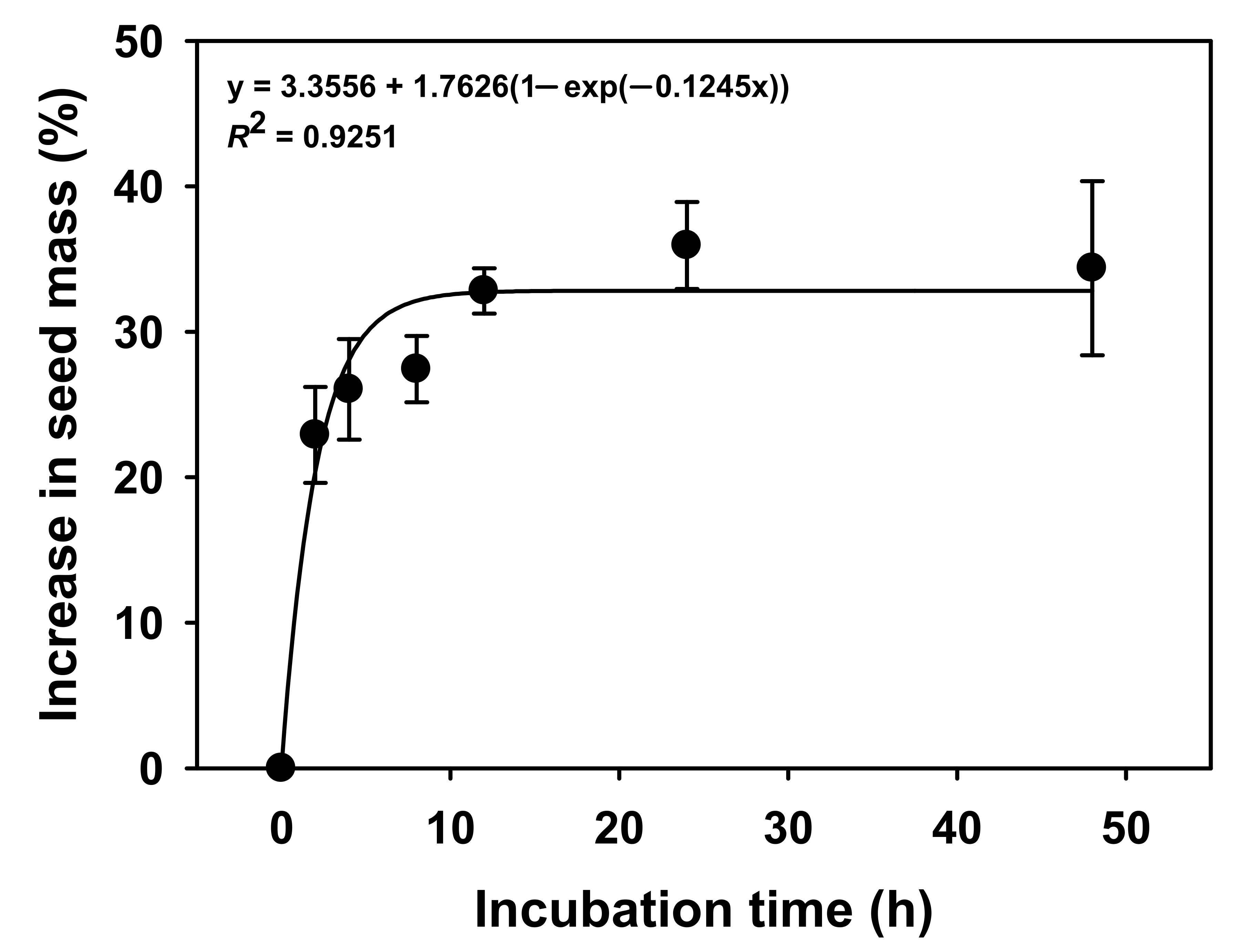
Since mass of the
V. sibiricum seeds increased by approximately 30% or more within 24 h compared with the initial mass due to water absorption (
Figure 1), the seed coat was permeable to water [
25] and the seeds do not have physical dormancy.
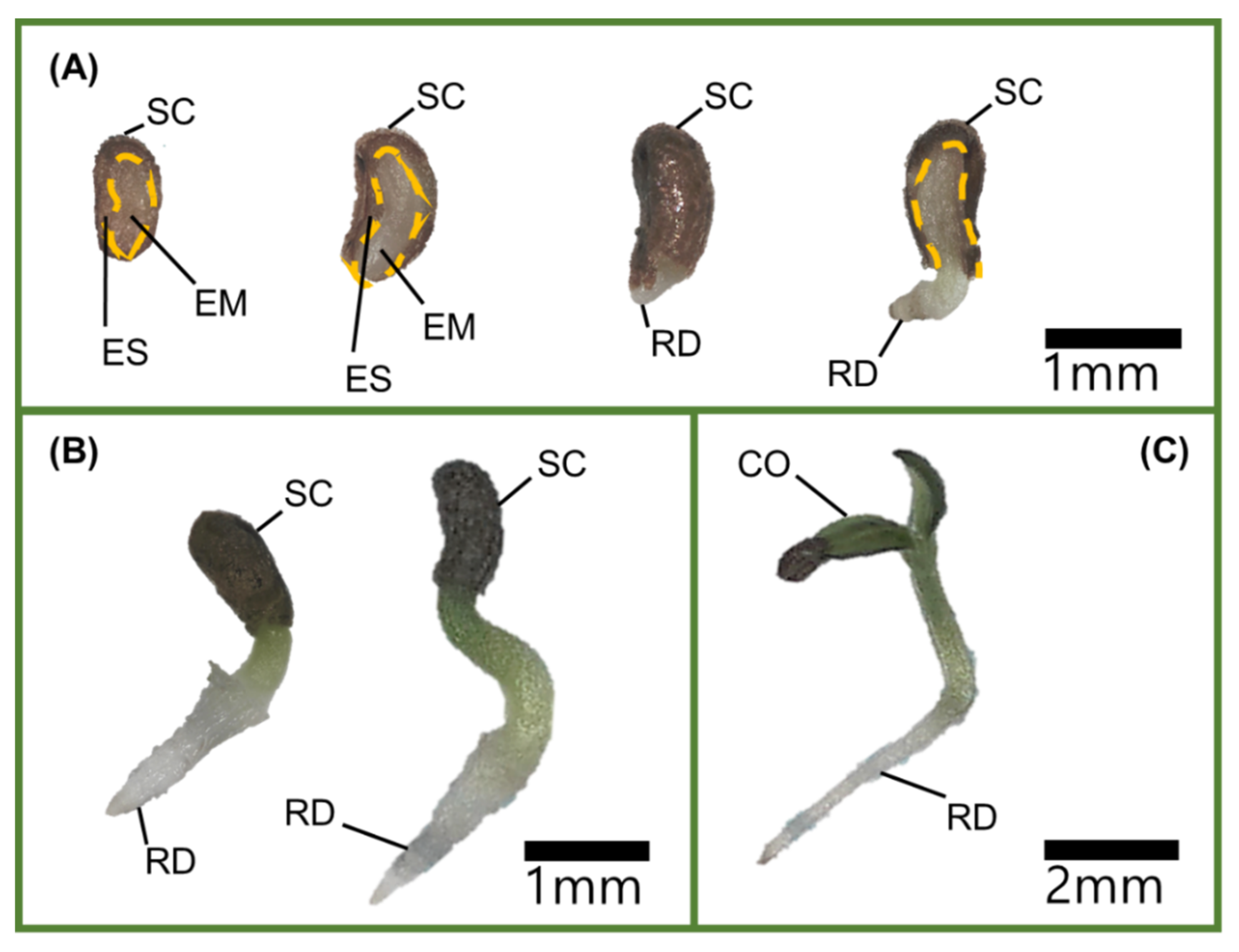
The inner and outer shape of the embryos of
V. sibiricum seeds observed in this study (
Table 1 and
Figure 8) were similar to those of the linear, axile embryos of the genus
Veronica positioned in the center of the seed, which is classified as dwarf (seed size 0.3–2 mm) [
17]. When seeds have an immature embryo that grows within 30 d under favorable germination conditions, they are considered to show MD [
13]. As the difference in the E:S ratios between the initial point of seed detachment and immediately before germination were not significant (
Figure 1), the embryo was considered to be in a mature state during detachment. Therefore, seeds of
V. sibiricum do not have MD.
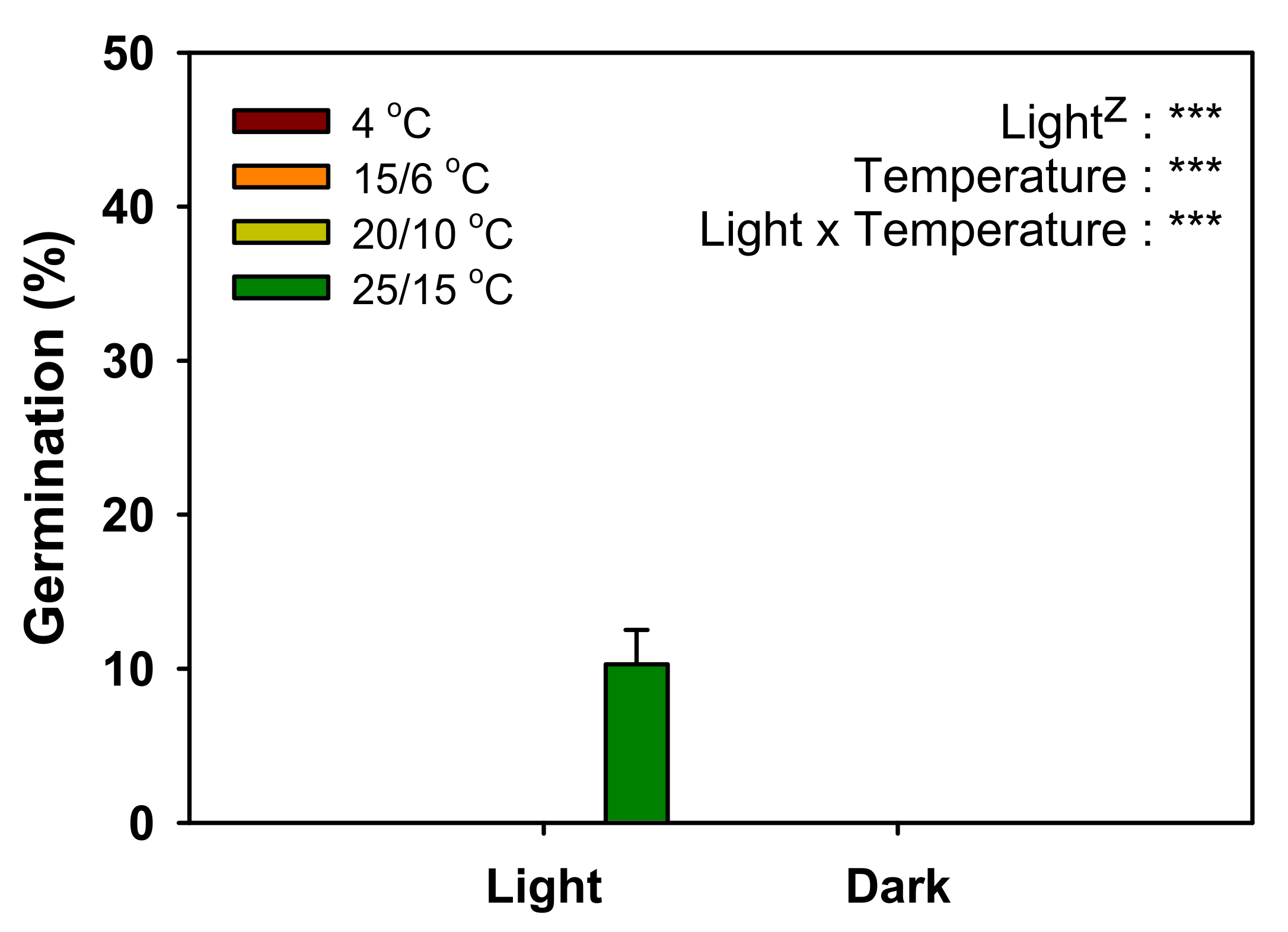
When
V. sibiricum seeds were cultured for four weeks at various temperature conditions, only 10% germination was observed only at 25/15 °C (
Figure 4), indicating that the seeds were dormant under appropriate environmental conditions [
13]. If the seeds with mature embryos without PY or MD do not germinate within 30 d under favorable germination conditions, they are considered to exhibit PD [
26]. Therefore, the dormancy type of
V. sibiricum seeds was judged as PD. Based on the physiological germination inhibition mechanism, PD can be divided into three levels (non-deep, intermediate, and deep) [
27]. Non-deep PD is broken by short-term warm or cold stratification or by GA
3 treatment, intermediate PD is broken by cold stratification for at least two months and GA
3 treatment, while deep PD is broken by cold stratification for 3–4 months but not by GA
3 treatment [
26].
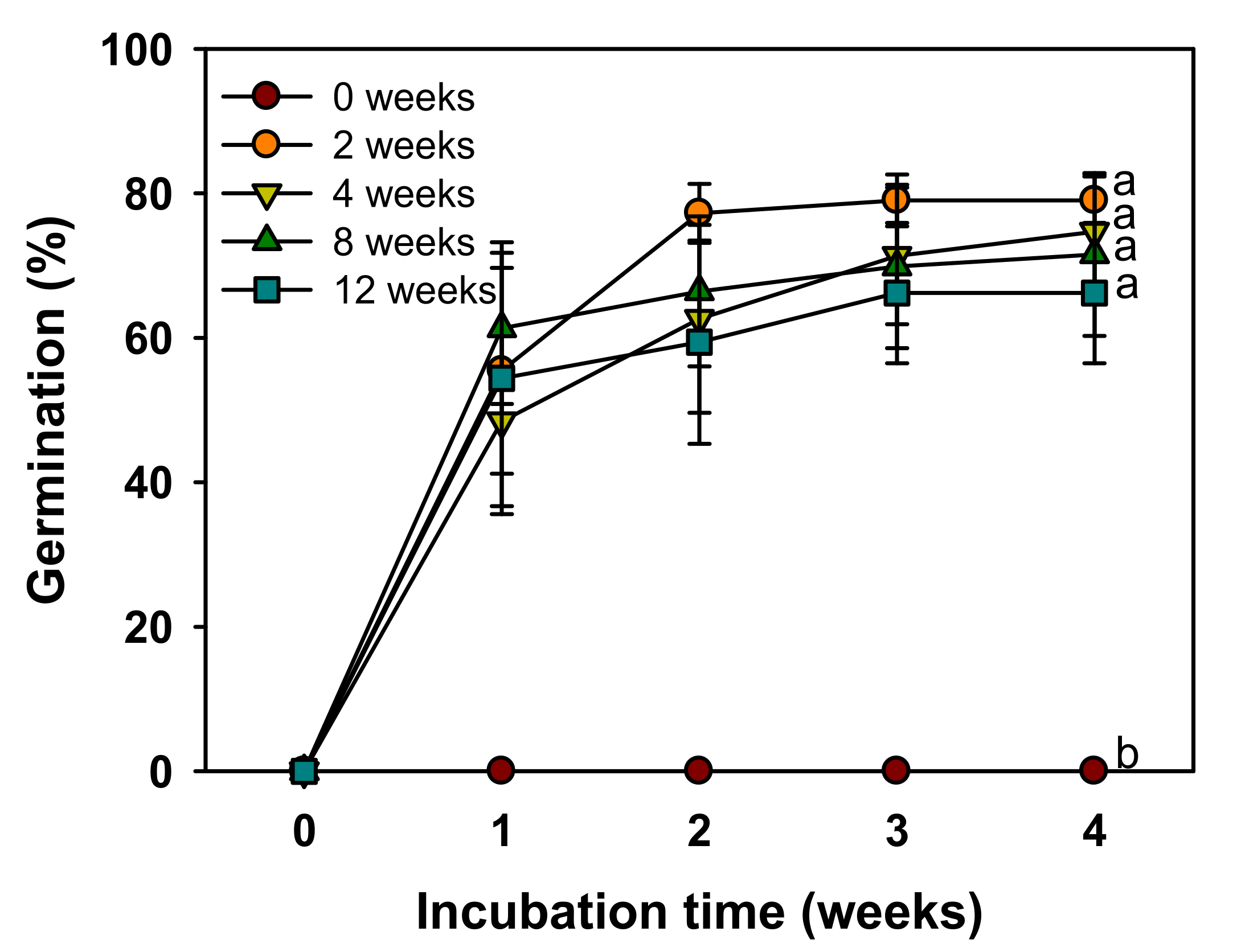
V. sibiricum seeds did not germinate in the control that was not subjected to cold stratification, but germination was more than 60% in the group subjected to cold stratification for two weeks (
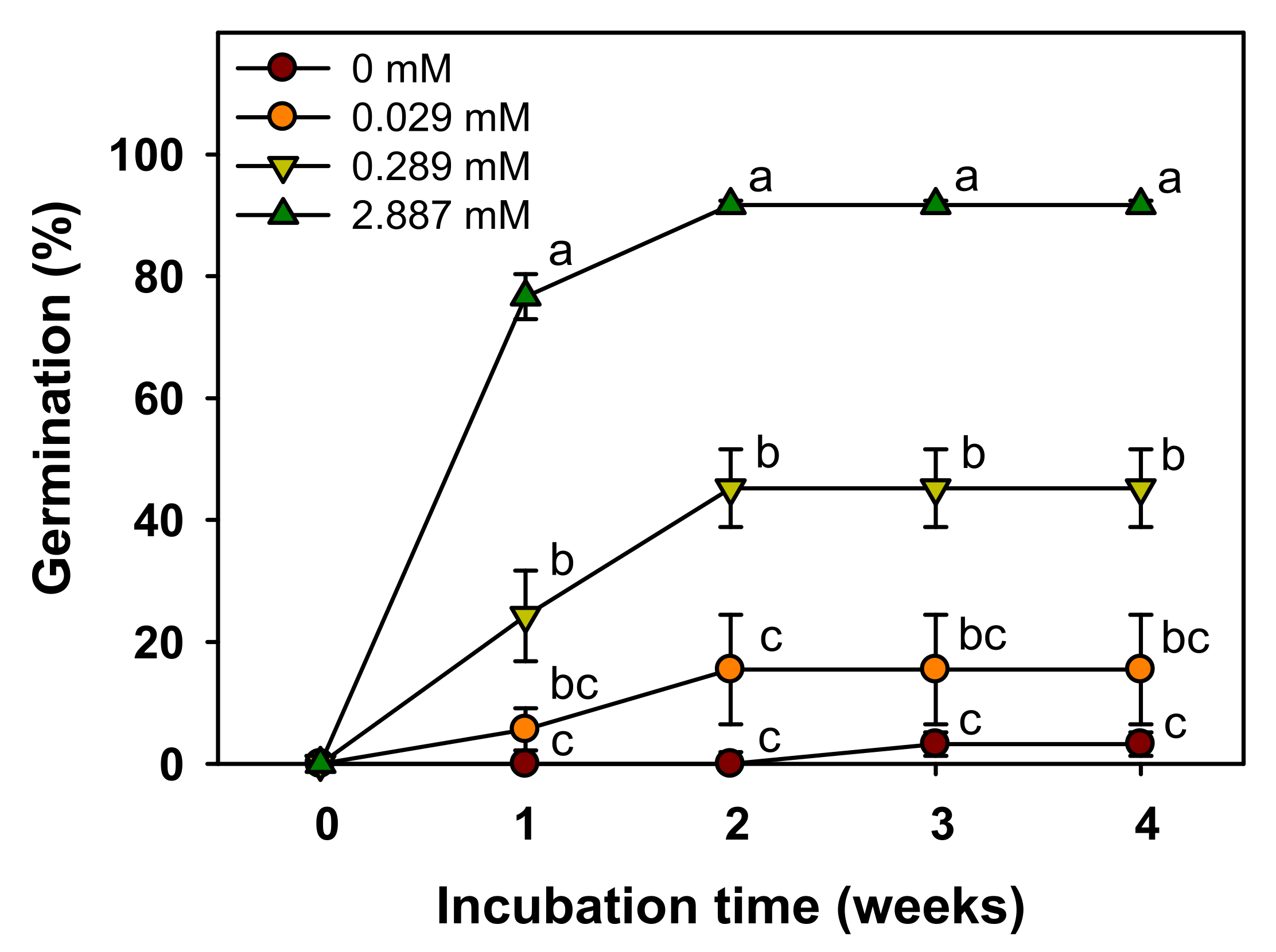
Figure 5). Further, germination increased with the increase in GA
3 concentration, and germination was more than 90% at 2.887 mM GA
3 (
Figure 6). Therefore, the dormancy was classified as non-deep PD, which was broken by a relatively short cold stratification period and by the GA
3 treatment. We used the seeds stored for seven weeks at room temperatures in this study. Many seeds with PD show the effects of dry after-ripening that can release dormancy and promote germination [
28]. Therefore, the depth of dormancy may decrease during storage. As a result of a preliminary experiment immediately after harvesting seeds in 2018, cold stratification for about 2 weeks was also effective in breaking PD of this species. This means that the relatively short cold stratification period is effective in breaking the PD. However, since we presented the results using seeds harvested in 2019 in this study, additional experiments are needed to know the exact depth of dormancy of seeds immediately after harvesting.
The subgenus
Pseudolysimachium in the genus
Veronica is assumed to have originated in Northeast Asia, and the differentiation of the genus may have occurred from Northeast Asia to Korea, Japan, China, and Europe [
18]. Recently, several species of
Veronica such as
V. dahurica,
V. rotunda,
V. kiusiana var.
diamantiaca,
V. pusanensis,
V. rotunda var.
subintegra,
V. nakaiana,
V. pyrethrina, and
V. kiusiana var.
glabrifolia that are native to the Korean Peninsula have been placed in the
Pseudolysisimachion reflecting the results of various molecular data [
29]. In the Cenozoic Era, during which time the genus
Pseudolysisimachium originated, Northeast Asia and North America were separated. Consequently, the subgenus was distributed locally only in the northern hemisphere, except in North America, due to the formation of a large mountain range in the lower regions of Northeast Asia [
18]. Comparative analysis of chloroplast genomes in three species of the genera
Veronica and the genus
Veronicastrum revealed that the species had identical coding genes, tRNA and rRNA [
30]. Therefore, the species of the two genera were considered phylogenetically closely related. We collected information on the class of seed dormancy of species of
Veronica and
Veronicastrum in previous research (
Table 2). In previous studies, seed dormancy of
V. virginicum and
V. parnkalliana was classified as PD and MPD, respectively [
20]. Song et al. [
21] analyzed the ratio of embryo length to seed length (E:S ratio) in eight species of the genus
Veronica (
V. dahurica,
V. rotunda,
V. kiusiana var.
diamantiaca,
V. pusanensis,
V. rotunda var.
subintegra,
V. nakaiana,
V. pyrethrina, and
V. kiusiana var.
glabrifolia) native to the Korean Peninsula and found that the radicles in all eight species emerged after the E:S ratio increased from 18.8% to 58.0%. Among these species, seven had MD, and
V. kiusiana var.
diamantiaca had the characteristics of both MD and MPD at the population level [
21]. Therefore, class of dormancy was differentiated at the species and genus level. Moreover, when the physiological characteristics of a species do not change during adaptation, the phenomenon is defined as “trait stasis.” Conversely, when physiological characteristics change during adaptation, the phenomenon is defined as “adaptation (or trait divergence)” [
22]. Therefore, the findings of this study indicated that divergence occurred during dormancy in the closely related taxa because of the variation in embryo size and the different dormancy breaking requirements in species of the sister genera
Veronicastrum and
Veronica. The results of this study provide important preliminary data for the evolutionary ecological studies on seed dormancy and seed-based mass propagation in
V. sibiricum.
4. Materials and Methods
4.1. Experimental Materials
The V. sibiricum seeds used in this study were harvested from the parent plant grown at the Andong National University affiliated farm (36°32′40″ N 128°48′03″ E) in Andong-si, Gyeongsangbuk-do on 16 October 2019. After harvesting, the seeds were dried in a laboratory at 21–27 °C for seven weeks before storing in a cold storage facility at 0 °C (DOI1815DOP; Winiamando, Gwangju, Korea) for further experimental use. Laboratory experiment was started at 2 weeks after storing the seeds at 0 °C. The seeds for phenology experiments to determine seed dormancy were harvested from the same area on 14 September 2020.
4.2. Analysis of Internal and External Seed Morphology
To observe the internal and external morphology of seeds, the seeds were cut with a stainless steel razor blade (Dorco, Korea), and the resultant cross sections were photographed at 200–210× magnification under a USB microscope (AM3111 Dino-Lite premier; AnMo Electronics Co., Taiwan). Further, to determine whether the photographed seeds comprised immature embryos, embryos at the time of early detachment and just before germination were investigated, and the corresponding E:S length ratio was calculated.
4.3. Water Absorption
Water absorption was examined to identify the presence or absence of physical dormancy in the
V. sibiricum seeds. Twenty seeds were placed in Petri dishes (90 × 15 mm; diameter × height) lined with two sheets of filter paper (ADVANTEC No. 1; Toyo Roshi Kaisha, Ltd., Tokyo, Japan) and added with distilled water (≒15 mL), and then cultured at 21–27 °C; four replicates were prepared per treatment. The seeds were not submerged in this experiment. The reason is that it was able to supply sufficient moisture without being submerged in our preliminary experiment. The distilled water was added as needed to keep the seeds moist. The initial weight of the seeds before water absorption and final weights after 2, 4, 8, 12, 24, and 48 h of water treatment were measured. The water absorption rate was calculated as follows:
where
Ws is relative weight ratio of seeds increased through water absorption,
Wh is the weight per culture time, and
Wi is weight of seeds before water absorption.
4.4. Phenology of Germination
This experiment was started after about 4 weeks of harvesting the seeds on 14 September 2020. The seeds were stored at the laboratory conditions before being used for the phenology experiment. The germination phenology experiment was conducted from 16 October 2020 to 16 April 2021 at the above-mentioned farm. There are several herbaceous plants growing around V. sibiricum in their native habitats. When the V. sibiricum seeds fall to the ground, direct sunlight was blocked by the plants. Therefore, a light-shielding film was installed where the seeds were planted to prevent direct sunlight exposure to the ground surface. Temperature data were collected using a sensor connected to a data logger (3683WD1; Spectrum Technologies, Inc., Aurora, IL, USA). The sensor was buried at a depth of 3 cm during the experiment, and the temperature was recorded every 30 min.
To observe the seasonal germination response of the seeds, the germination was investigated weekly by exhuming the seeds in a phenology plot of the experimental farm. For the germination experiment, the seeds were wrapped in a plastic mesh filled with sand, and buried to a depth of 3 cm by filling the plastic pot with a horticultural substrate (Sunshine Mix #4; SunGro Horticulture, Agawam, MA, USA). This plastic pot was buried in the ground at a height similar to that of the top soil. Four replicates, each of 20 seeds per pot, were prepared. Once the emerged radicle reached a length of 1 mm, the seeds were regarded as germinated and were removed immediately.
4.5. Effect of Light and Temperature on Germination
The seeds to be cultured were sterilized in 500 mg·L−1 diluted solution of benomyl (a fungicide) for 4 h. Petri dishes (90 × 15 mm) were lined with two sheets of filter paper (ADVANTEC No. 1; Toyo Roshi Kaisha, Ltd., Tokyo, Japan), and 20 seeds were placed in each dish; four replicates were prepared per treatment. Distilled water (≒10 mL) was added to prevent the desiccation of the seeds. The seeds were cultured in a multi-room incubator (HB-101-4; HANBAEK-Scientific, Bucheon, Korea) at 25/15, 20/10, and 15/6 °C for 12/12 h of day/night and the photosynthetic photon flux density (PPFD) of 63.46 μmol·m−2·s−1. Culturing at 4 °C was conducted in a growth chamber (HB-603CM; HANBAEK-Scientific, Bucheon, Korea) under PPFD of 2.56 μmol·m−2·s−1. For incubation in dark conditions, petri dishes were wrapped in double aluminum foil to prevent the exposure of seeds to light. The seeds were examined each week and after four weeks, when the emerged radicle reached 1 mm in length, the seeds were considered germinated. Subsequently, the germinated seeds were counted and removed from the petri dish.
4.6. Effect of Cold Stratification
The effects of cold stratification were determined to understand whether the dormancy could be broken by cold stratification. Twenty sterilized seeds per replicate, four replicates per treatment, were placed in Petri dishes (90 × 15 mm) lined with two sheets of filter paper (ADVANTEC No. 1) and transferred to a growth chamber at 4 °C. Cold stratification at 4 °C was conducted in a growth chamber (HB-603CM; HANBAEK-Scientific, Bucheon, Korea) under PPFD of 2.56 μmol·m−2·s−1. After the cold stratification treatment for 0, 2, 4, 8, and 12 weeks, seeds were incubated in light at 20/10 °C.
4.7. Effect of GA3 Treatment
The effects of GA3 on dormancy break were analyzed by immersing the sterilized seeds in 0, 0.029, 0.289, and 2.887 mM GA3 solution for 24 h, after which they were rinsed with distilled water for 3 min and disinfected with benomyl wettable powder (500 mg·L−1) for 4 h. The seeds were sown in Petri dishes as described above and incubated at 20/10 °C. Distilled water was supplied to prevent desiccation.
4.8. Effect of Light on Germination after Cold Stratification
The germination percentages based on varying incubation light conditions (light or dark) were determined. Twenty sterilized seeds per replicates, four replicates per treatment, were placed in Petri dishes and cold stratified at 4 °C for 2 weeks. The dishes of seeds in the dark treatment were wrapped with aluminum foil to protect the seeds from being exposed to any light. After the cold stratification treatment, both treatment groups were incubated at 20/10 °C for 4 weeks with 12/12 h of day/night and the photosynthetic photon flux density (PPFD) of 63.46 μmol·m−2·s−1.
4.9. Statistical Analysis
Analysis of variance was performed on the collected data using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). Significant differences in embryo to seed length ratio (E:fvS ratio) at initial stage and seed coat split stage were determined by paired t-test at p < 0.05. To determine the statistical significance of the percentage germination between different treatments, Tukey’s honestly significant difference (HSD) test at p < 0.05 was used. The graphs were prepared using Sigma Plot 10.0 (Systat Software Inc., San Jose, CA, USA).