Influence of Intact Mycelium of Arbuscular Mycorrhizal Fungi on Soil Microbiome Functional Profile in Wheat under Mn Stress
Abstract
1. Introduction
2. Results
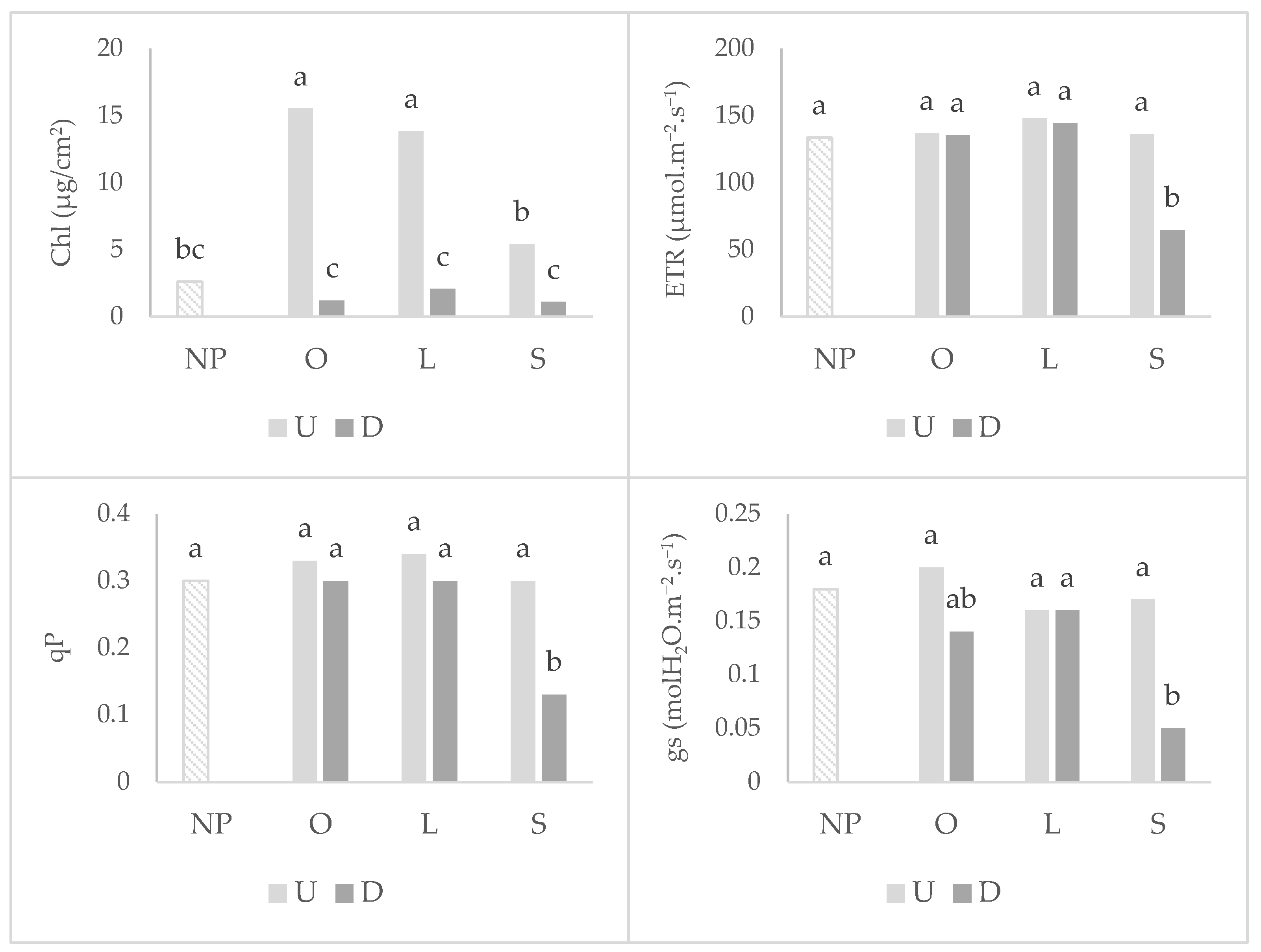
2.1. Wheat Root Colonization, Photosynthetic Parameters, and Shoot Development
2.2. Soil Microbial Activity
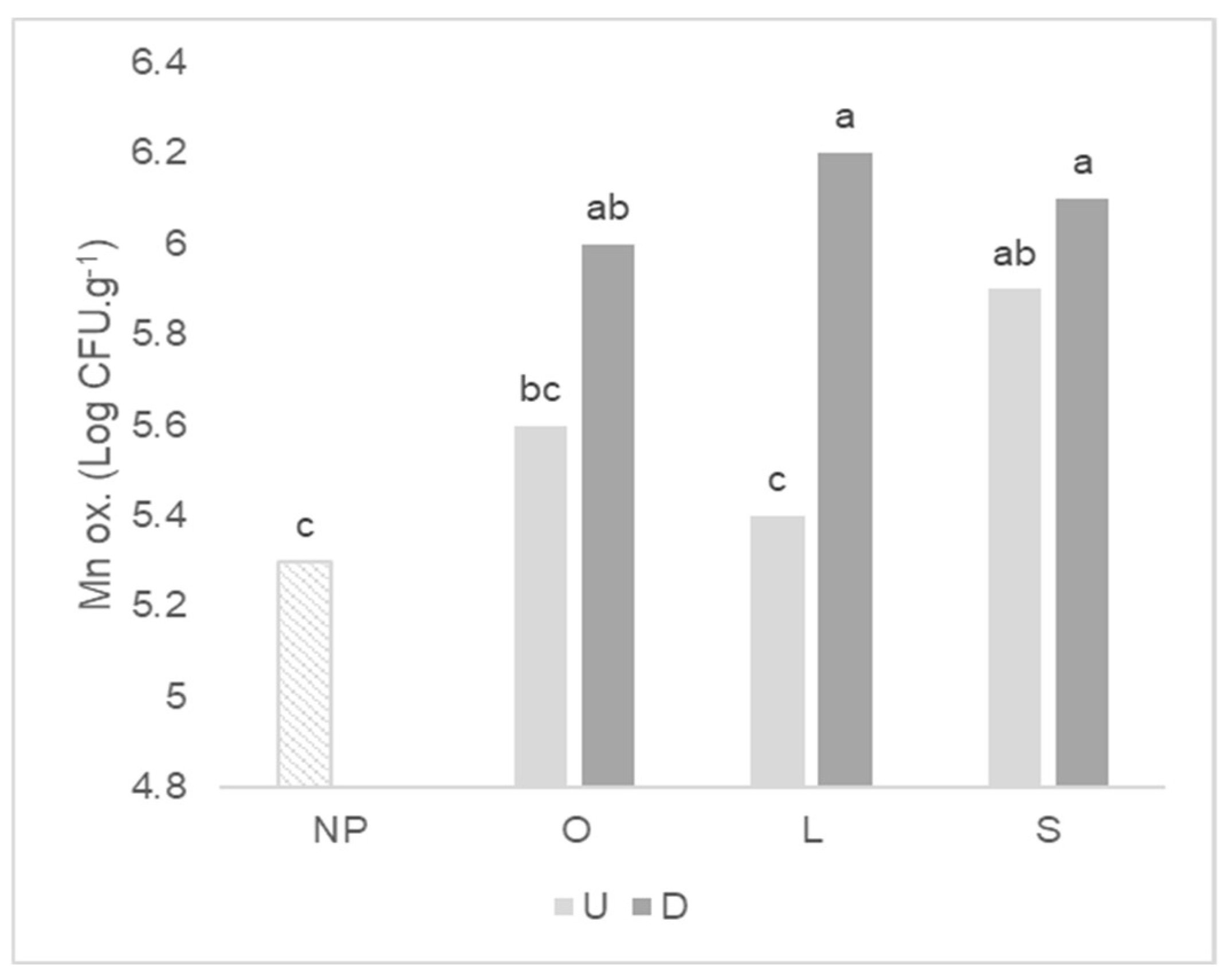
2.3. Functional Groups of Culturable Microorganisms
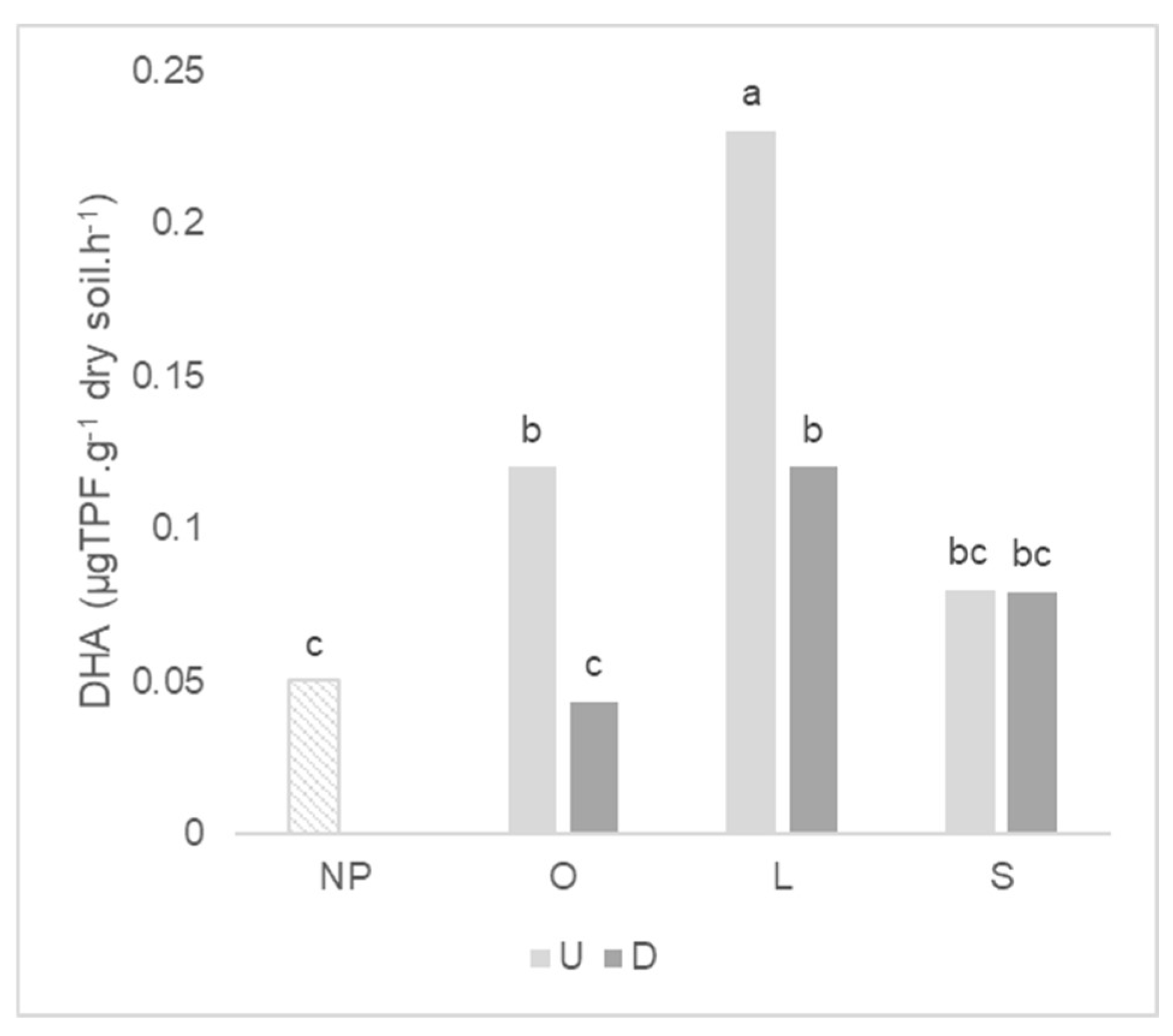
2.4. Enzymatic Activity
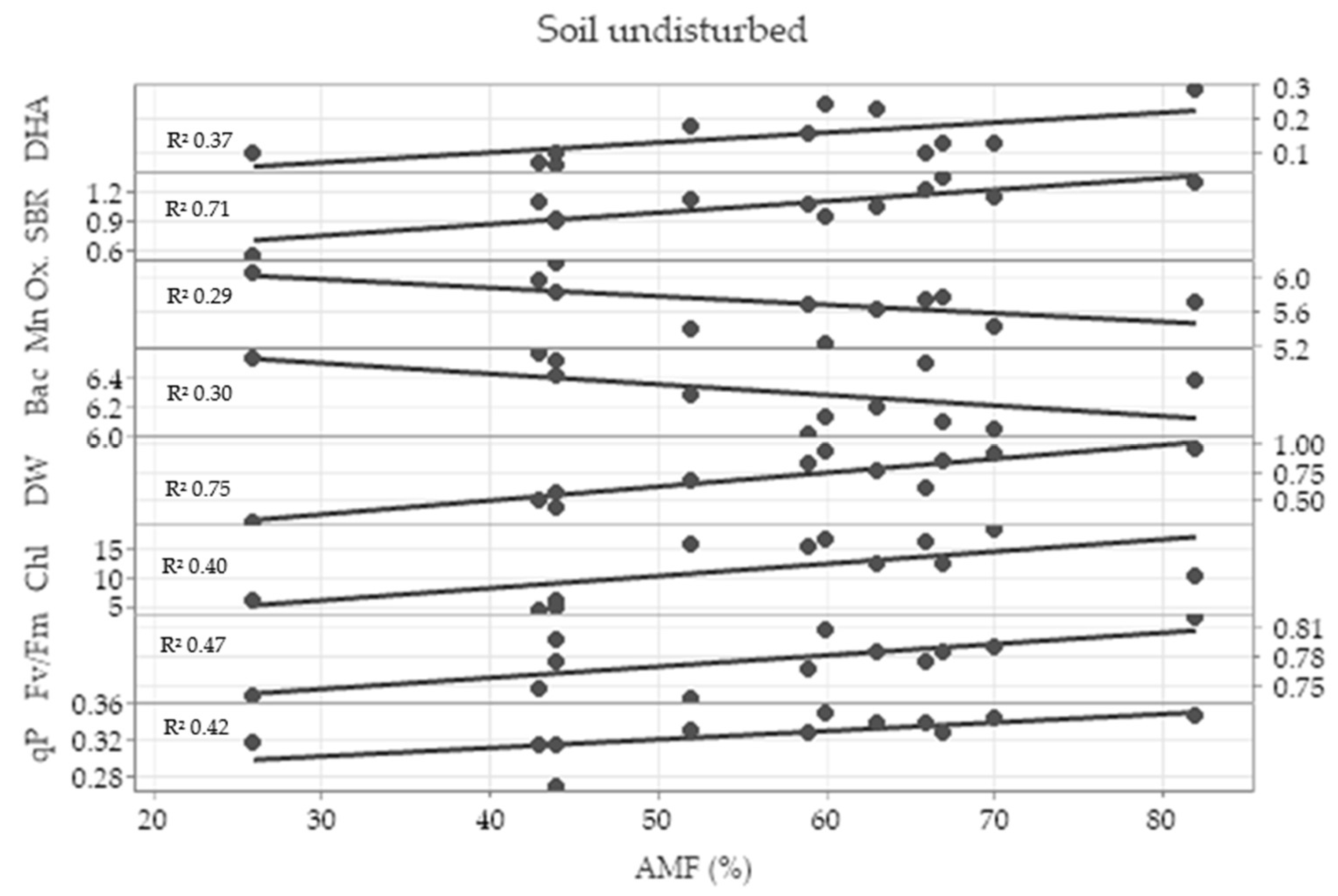
2.5. Correlation Analysis
3. Discussion
4. Materials and Methods
4.1. Experimental Design
4.2. Soil Microbial Activity
4.3. Functional Groups of Culturable Microorganisms
4.4. Enzyme Activity
4.5. Photosynthetic Parameters
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Page, K.L.; Dang, Y.P.; Dalal, R.C. The Ability of Conservation Agriculture to Conserve Soil Organic Carbon and the Subsequent Impact on Soil Physical, Chemical, and Biological Properties and Yield. Front. Sustain. Food Syst. 2020, 4, 32. [Google Scholar] [CrossRef]
- Goss, M.J.; Carvalho, M.; Brito, I. Challenges to Agriculture Systems. In Functional Diversity of Mycorrhiza and Sustainable Agriculture—Management to Overcome Biotic and Abiotic Stresses; Academic Press: Cambridge, MA, USA; Elsevier: London, UK, 2017; p. 231. ISBN 9780128042441. [Google Scholar]
- Barea, J.M. Future Challenges and Perspectives for Applying Microbial Biotechnology in Sustainable Agriculture Based on a Better Understanding of Plant-Microbiome Interactions. J. Soil Sci. Plant Nutr. 2015, 15, 261–282. [Google Scholar] [CrossRef]
- Goss, M.J.; Carvalho, M. Manganese Toxicity: The Significance of Magnesium for the Sensitivity of Wheat Plants. Plant Soil 1992, 139, 91–98. [Google Scholar] [CrossRef]
- Alejandro, S.; Höller, S.; Meier, B.; Peiter, E. Manganese in Plants: From Acquisition to Subcellular Allocation. Front. Plant Sci. 2020, 11, 300. [Google Scholar] [CrossRef]
- Brito, I.; Carvalho, M.; Alho, L.; Goss, M.J. Managing Arbuscular Mycorrhizal Fungi for Bioprotection: Mn Toxicity. Soil Biol. Biochem. 2014, 68, 78–84. [Google Scholar] [CrossRef]
- Alho, L.; Carvalho, M.; Brito, I.; Goss, M.J. The Effect of Arbuscular Mycorrhiza Fungal Propagules on the Growth of Subterranean Clover (Trifolium subterraneum L.) under Mn Toxicity in Ex Situ Experiments. Soil Use Manag. 2015, 31, 337–344. [Google Scholar] [CrossRef]
- Begum, N.; Qin, C.; Ahanger, M.A.; Raza, S.; Khan, M.I.; Ashraf, M.; Ahmed, N.; Zhang, L. Role of Arbuscular Mycorrhizal Fungi in Plant Growth Regulation: Implications in Abiotic Stress Tolerance. Front. Plant Sci. 2019, 10, 1068. [Google Scholar] [CrossRef]
- Brito, I.; Goss, M.J.; Alho, L.; Brígido, C.; van Tuinen, D.; Félix, M.R.; Carvalho, M. Agronomic Management of AMF Functional Diversity to Overcome Biotic and Abiotic Stresses—The Role of Plant Sequence and Intact Extraradical Mycelium. Fungal Ecol. 2019, 40, 72–81. [Google Scholar] [CrossRef]
- Andrade, G.; Mihara, K.L.; Linderman, R.G.; Bethlenfalvay, G.J. Bacteria from Rhizosphere and Hyphosphere Soils of Different Arbuscular-Mycorrhizal Fungi. Plant Soil 1997, 71, 71–79. [Google Scholar] [CrossRef]
- Brito, I.; Carvalho, M.; Goss, M.J. Managing the Functional Diversity of Arbuscular Mycorrhizal Fungi for the Sustainable Intensification of Crop Production. Plants People Planet 2021, 3, 491–505. [Google Scholar] [CrossRef]
- Bini, D.; dos Santos, C.A.; Bernal, L.P.T.; Andrade, G.; Nogueira, M.A. Identifying Indicators of C and N Cycling in a Clayey Ultisol under Different Tillage and Uses in Winter. Appl. Soil Ecol. 2014, 76, 95–101. [Google Scholar] [CrossRef]
- Hsieh, Y.P.; Anderson, G.A.; Miller, R.O.; Nemours, D. Non-Instrumental Real-Time Soil Respiration Rate and Soil Microbial Biomass Carbon Determinations. Commun. Soil Sci. Plant Anal. 2020, 51, 2479–2490. [Google Scholar] [CrossRef]
- Creamer, R.E.; Schulte, R.P.O.; Stone, D.; Gal, A.; Krogh, P.H.; Lo Papa, G.; Murray, P.J.; Pérès, G.; Foerster, B.; Rutgers, M.; et al. Measuring Basal Soil Respiration across Europe: Do Incubation Temperature and Incubation Period Matter? Ecol. Indic. 2014, 36, 409–418. [Google Scholar] [CrossRef]
- Nogueira, M.A.; Albino, U.B.; Brandão-Junior, O.; Braun, G.; Cruz, M.F.; Dias, B.A.; Duarte, R.T.D.; Gioppo, N.M.R.; Menna, P.; Orlandi, J.M.; et al. Promising Indicators for Assessment of Agroecosystems Alteration among Natural, Reforested and Agricultural Land Use in Southern Brazil. Agric. Ecosyst. Environ. 2006, 115, 237–247. [Google Scholar] [CrossRef]
- Alkorta, I.; Aizpurua, A.; Riga, P.; Albizu, I.; Amézaga, I.; Garbisu, C. Soil Enzyme Activities as Biological Indicators of Soil Health. Rev. Environ. Health 2003, 18, 65–73. [Google Scholar] [CrossRef]
- Nannipieri, P.; Ascher, J.; Ceccherini, M.T.; Landi, L.; Pietramellara, G.; Renella, G. Microbial Diversity and Soil Functions. Eur. J. Soil Sci. 2017, 68, 12–26. [Google Scholar] [CrossRef]
- Tate, R.L. Microbial Diversity of Soil Ecosystems. In Soil Microbiology; Tate, R.L., Ed.; Wiley: Hoboken, NJ, USA, 2020; pp. 89–113. ISBN 9781119114314. [Google Scholar]
- Liu, S.; Coyne, M.S.; Grove, J.H. Long-Term Tillage and Nitrogen Fertilization: Consequences for Nitrifier Density and Activity. Appl. Soil Ecol. 2017, 120, 121–127. [Google Scholar] [CrossRef]
- Bolo, P.; Kihara, J.; Mucheru-Muna, M.; Njeru, E.M.; Kinyua, M.; Sommer, R. Application of Residue, Inorganic Fertilizer and Lime Affect Phosphorus Solubilizing Microorganisms and Microbial Biomass under Different Tillage and Cropping Systems in a Ferralsol. Geoderma 2021, 390, 114962. [Google Scholar] [CrossRef]
- Niewiadomska, A.; Majchrzak, L.; Borowiak, K.; Wolna-Maruwka, A.; Waraczewska, Z.; Budka, A.; Gaj, R. The Influence of Tillage and Cover Cropping on Soil Microbial Parameters and Spring Wheat Physiology. Agronomy 2020, 10, 200. [Google Scholar] [CrossRef]
- Bissett, A.; Richardson, A.E.; Baker, G.; Kirkegaard, J.; Thrall, P.H. Bacterial Community Response to Tillage and Nutrient Additions in a Long-Term Wheat Cropping Experiment. Soil Biol. Biochem. 2013, 58, 281–292. [Google Scholar] [CrossRef]
- Hill, G.T.; Mitkowski, N.A.; Aldrich-Wolfe, L.; Emele, L.R.; Jurkonie, D.D.; Ficke, A.; Maldonado-Ramirez, S.; Lynch, S.T.; Nelson, E.B. Methods for assessing the composition and diversity of soil microbial communities. Appl. Soil Ecol. 2000, 15, 25–36. [Google Scholar] [CrossRef]
- Takagi, D.; Ishiyama, K.; Suganami, M.; Ushijima, T.; Fujii, T.; Tazoe, Y.; Kawasaki, M.; Noguchi, K.; Makino, A. Manganese Toxicity Disrupts Indole Acetic Acid Homeostasis and Suppresses the CO2 Assimilation Reaction in Rice Leaves. Sci. Rep. 2021, 11, 20922. [Google Scholar] [CrossRef]
- Liu, P.; Huang, R.; Hu, X.; Jia, Y.; Li, J.; Luo, J.; Liu, Q.; Luo, L.; Liu, G.; Chen, Z. Physiological Responses and Proteomic Changes Reveal Insights into Stylosanthes Response to Manganese Toxicity. BMC Plant Biol. 2019, 19, 212. [Google Scholar] [CrossRef]
- Liang, H.Z.; Zhu, F.; Wang, R.J.; Huang, X.H.; Chu, J.J. Photosystem II of Ligustrum Lucidum in Response to Different Levels of Manganese Exposure. Sci. Rep. 2019, 9, 12568. [Google Scholar] [CrossRef] [PubMed]
- Ribera, A.E.; Reyes-Díaz, M.M.; Alberdi, M.R.; Alvarez-Cortez, D.A.; Rengel, Z.; Mora, M.D.L.L. Photosynthetic Impairment Caused by Manganese Toxicity and Associated Antioxidative Responses in Perennial Ryegrass. Crop Pasture Sci. 2013, 64, 696–707. [Google Scholar] [CrossRef]
- Guidi, L.; Lo Piccolo, E.; Landi, M. Chlorophyll Fluorescence, Photoinhibition and Abiotic Stress: Does It Make Any Difference the Fact to Be a C3 or C4 Species? Front. Plant Sci. 2019, 10, 174. [Google Scholar] [CrossRef]
- Shao, Q.; Wang, H.; Guo, H.; Zhou, A.; Huang, Y.; Sun, Y.; Li, M. Effects of Shade Treatments on Photosynthetic Characteristics, Chloroplast Ultrastructure, and Physiology of Anoectochilus Roxburghii. PLoS ONE 2014, 9, e85996. [Google Scholar] [CrossRef] [PubMed]
- Faria, J.M.; Pinto, A.P.; Teixeira, D.M.; Brito, I.; Barrulas, P.; Carvalho, M. The Protective Biochemical Properties of Arbuscular Mycorrhiza Extraradical Mycelium in Acidic Soils Are Maintained throughout the Mediterranean Summer Conditions. Agronomy 2021, 11, 748. [Google Scholar] [CrossRef]
- Deng, S.P.; Tabatabai, M.A. Effect of Tillage and Residue Management on Enzyme Activities in Soils. Biol. Fertil. Soils 1996, 22, 208–213. [Google Scholar] [CrossRef]
- Madejón, E.; Moreno, F.; Murillo, J.M.; Pelegrín, F. Soil Biochemical Response to Long-Term Conservation Tillage under Semi-Arid Mediterranean Conditions. Soil Tillage Res. 2007, 94, 346–352. [Google Scholar] [CrossRef]
- Pandey, D.; Agrawal, M.; Bohra, J.S. Assessment of Soil Quality under Different Tillage Practices during Wheat Cultivation: Soil Enzymes and Microbial Biomass. Chem. Ecol. 2015, 31, 510–523. [Google Scholar] [CrossRef]
- Nannipieri, P.; Giagnoni, L.; Landi, L.; Renella, G. Role of Phosphatase Enzymes in Soil. In Phosphorus in Action; Soil, Biology; Bünemann, E., Oberson, A., Frossard, E., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 26. [Google Scholar] [CrossRef]
- Marschner, P.; Fu, Q.; Rengel, Z. Manganese Availability and Microbial Populations in the Rhizosphere of Wheat Genotypes Differing in Tolerance to Mn Deficiency. J. Plant Nutr. Soil Sci. 2003, 166, 712–718. [Google Scholar] [CrossRef]
- Yang, W.; Zhang, Z.; Zhang, Z.; Chen, H.; Liu, J.; Ali, M.; Liu, F.; Li, L. Population Structure of Manganese-Oxidizing Bacteria in Stratified Soils and Properties of Manganese Oxide Aggregates under Manganese-Complex Medium Enrichment. PLoS ONE 2013, 8, e73778. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, B.S.; Philippot, L. Insights into the Resistance and Resilience of the Soil Microbial Community. FEMS Microbiol. Rev. 2013, 37, 112–129. [Google Scholar] [CrossRef]
- Yin, B.; Crowley, D.; Sparovek, G.; De Melo, W.J.; Borneman, J. Bacterial Functional Redundancy along a Soil Reclamation Gradient. Appl. Environ. Microbiol. 2000, 66, 4361–4365. [Google Scholar] [CrossRef] [PubMed]
- Brígido, C.; van Tuinen, D.; Brito, I.; Alho, L.; Goss, M.J.; Carvalho, M. Management of the Biological Diversity of AM Fungi by Combination of Host Plant Succession and Integrity of Extraradical Mycelium. Soil Biol. Biochem. 2017, 112, 237–247. [Google Scholar] [CrossRef]
- McGonigle, T.P.; Miller, M.H.; Evans, D.G.; Fairchild, G.L.; Swan, J.A. A New Method Which Gives an Objective Measure of Colonization of Roots by Vesicular-Arbuscular Mycorrhizal Fungi. New Phytol. 1990, 115, 495–501. [Google Scholar] [CrossRef]
- Monteiro, R.T.R.; Frighetto, R.T.S. Determinação Da Umidade, PH e Capacidade de Retenção de Água Do Solo. In Indicadores Biológicos e Bioquímicos da Qualidade do Solo: Manual Técnico; Frighetto, R.T.S., Valarini, P.J., Eds.; EMBRAPA Centro Nacional de Pesquisa de Arroz e Feijão: Jaguariúna, Brazil, 2000; pp. 37–40. [Google Scholar]
- Silva, E.E.; Azevedo, P.H.S.; De-Polli, H. Determinação Da Respiração Basal (RBS) e Quociente Metabólico Do Solo (QCO2). Available online: https://ainfo.cnptia.embrapa.br/digital/bitstream/CNPAB-2010/34390/1/cot099.pdf (accessed on 10 December 2018).
- Vance, E.D.E.D.; Brookes, P.C.; Jenkinson, D.S. An Extraction Method for Measuring Soil Microbial Biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Joergensen, R.G. The Fumigation-Extraction Method to Estimate Soil Microbial Biomass: Calibration of the KEC Value. Soil Biol. Biochem. 1996, 28, 25–31. [Google Scholar] [CrossRef]
- Anderson, T.H.; Domsch, K.H. Application of Eco-Physiological Quotients (QCO2 and QD) on Microbial Biomasses from Soils of Different Cropping Histories. Soil Biol. Biochem. 1990, 22, 251–255. [Google Scholar] [CrossRef]
- Albino, U.B.; Andrade, G. Evaluation of the Functional Group of Microorganisms as Bioindicators on the Rhizosphere Microcosm. In Handbook of Microbial Biofertilizers; Rai, M.K., Ed.; Haworth: New York, NY, USA, 2007; p. 532. [Google Scholar]
- Nogueira, M.A.; Nehls, U.; Hampp, R.; Poralla, K.; Cardoso, E.J.B.N. Mycorrhiza and Soil Bacteria Influence Extractable Iron and Manganese in Soil and Uptake by Soybean. Plant Soil 2007, 298, 273–284. [Google Scholar] [CrossRef]
- Vidyalakshmi, R.; Sridar, R. Isolation and Characterization of Sulphur Oxidizing Bacteria. J. Cult. Collect. 2007, 5, 73–77. [Google Scholar]
- Casida, L.; Klein, D.A.; Santoro, T. Soil Dehydrogenase Activity. Soil Sci. 1964, 98, 371–376. [Google Scholar] [CrossRef]
- ISO 20130; Enzyme Activity Patterns in Soil in Soil Samples Using Colorimetric Substrates in Micro-Well Plates. International Standard Organization: Geneva, Switzerland, 2018.
- Fernandez-Göbel, T.F.; Deanna, R.; Muñoz, N.B.; Robert, G.; Asurmendi, S.; Lascano, R. Redox Systemic Signaling and Induced Tolerance Responses during Soybean–Bradyrhizobium Japonicum Interaction: Involvement of Nod Factor Receptor and Autoregulation of Nodulation. Front. Plant Sci. 2019, 10, 141. [Google Scholar] [CrossRef] [PubMed]
- Minitab, LLC. Minitab. 2021. Available online: https://www.minitab.com (accessed on 1 July 2022).



| Fv/Fm (µg/cm2) | A (µmol CO2 m−2 s−1) | |||||||
|---|---|---|---|---|---|---|---|---|
| Soil Disturbance | Mean Plant | Soil Disturbance | Mean Plant | |||||
| Plant Type | U | D | U | D | ||||
| O. compressus | 0.77 | 0.77 | 0.77 | A | 16.61 | 13.52 | 15.06 | AB |
| L. rigidum | 0.78 | 0.77 | 0.77 | A | 16.44 | 16.33 | 16.39 | A |
| S. gallica | 0.76 | 0.74 | 0.75 | A | 17.48 | 18.97 | 18.22 | A |
| No Plant | 0.7 | 0.70 | B | 0.70 | 0.70 | B | ||
| Mean soil | 0.74 | 0.75 | 15.57 | 15.14 | ||||
| SBR | C-Mic | qCO2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| (mg CO2 Kg−1 Soil h−1) | (mg C Kg−1 Soil) | (mg CO2 mg−1 C-mic h−1) ×10−3 | |||||||||
| Soil Disturbance | Mean Plant | Soil Disturbance | Mean Plant | Soil Disturbance | Mean Plant | ||||||
| Plant Type | U | D | U | D | U | D | |||||
| O. compressus | 1.19 | 1.13 | 1.16 | A | 137.73 | 106.48 | 122.10 | 8.8 | 10.6 | 9.7 | A |
| L. rigidum | 1.09 | 1.08 | 1.09 | AB | 113.42 | 114.58 | 114.00 | 9.9 | 9.7 | 9.8 | A |
| S. gallica | 0.86 | 1.04 | 0.95 | B | 129.63 | 118.05 | 123.84 | 6.9 | 9.3 | 8.1 | AB |
| No Plant | 0.54 | 0.54 | C | 99.53 | 99.53 | 5.5 | 5.5 | B | |||
| Mean soil | 0.92 | 0.95 | 120.08 | 109.66 | 7.8 | 8.8 | |||||
| Total Bacteria | P Solubilizers | Ammonifiers | S Oxidizers | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (Log CFU g−1) | (Log MPN g−1) | |||||||||||||||||||
| Soil Disturbance | Mean Plant | Soil Disturbance | Mean Plant | Soil Disturbance | Mean Plant | Soil Disturbance | Mean Plant | |||||||||||||
| Plant Type | U | D | U | D | U | D | U | D | ||||||||||||
| O. compressus | 6.16 | 6.32 | 6.24 | B | 5.49 | 5.31 | 5.40 | AB | 7.32 | 8.91 | 8.11 | 3.14 | 4.39 | 3.77 | ||||||
| L. rigidum | 6.24 | 6.61 | 6.42 | AB | 5.52 | 5.95 | 5.74 | A | 6.93 | 8.24 | 7.59 | 3.44 | 4.28 | 3.86 | ||||||
| S. gallica | 6.50 | 6.54 | 6.52 | A | 5.16 | 5.37 | 5.26 | B | 7.44 | 8.04 | 7.74 | 3.65 | 4.23 | 3.94 | ||||||
| No Plant | 6.25 | 6.25 | B | 5.66 | 5.66 | AB | 7.30 | 7.30 | 3.63 | 3.63 | ||||||||||
| Mean soil | 6.29 | B | 6.43 | A | 5.46 | 5.57 | 7.25 | B | 8.12 | A | 3.47 | B | 4.13 | A | ||||||
| Arylsulfatase | Phosphatase | β-Glucosidase | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (nmol ρ-Nitrophenol g−1 Dry Soil h−1) | ||||||||||||||
| Soil Disturbance | Mean Plant | Soil Disturbance | Mean Plant | Soil Disturbance | Mean Plant | |||||||||
| Plant Type | U | D | U | D | U | D | ||||||||
| O. compressus | 37.50 | 41.22 | 39.36 | AB | 2091.81 | 2250.25 | 2171.03 | B | 499.09 | 621.40 | 560.24 | AB | ||
| L. rigidum | 42.86 | 41.61 | 42.24 | A | 2255.14 | 2391.39 | 2323.27 | AB | 538.91 | 687.76 | 613.34 | A | ||
| S. gallica | 37.66 | 36.84 | 37.25 | AB | 2430.72 | 2600.24 | 2515.48 | A | 476.75 | 483.34 | 480.04 | B | ||
| No Plant | 33.56 | 33.56 | B | 1908.00 | 1908.00 | C | 548.69 | 548.69 | AB | |||||
| Mean soil | 37.90 | 38.31 | 2171.42 | 2287.47 | 515.86 | B | 585.30 | A | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Conceição, T.A.; Andrade, G.; Brito, I. Influence of Intact Mycelium of Arbuscular Mycorrhizal Fungi on Soil Microbiome Functional Profile in Wheat under Mn Stress. Plants 2022, 11, 2598. https://doi.org/10.3390/plants11192598
Conceição TA, Andrade G, Brito I. Influence of Intact Mycelium of Arbuscular Mycorrhizal Fungi on Soil Microbiome Functional Profile in Wheat under Mn Stress. Plants. 2022; 11(19):2598. https://doi.org/10.3390/plants11192598
Chicago/Turabian StyleConceição, Taiana A., Galdino Andrade, and Isabel Brito. 2022. "Influence of Intact Mycelium of Arbuscular Mycorrhizal Fungi on Soil Microbiome Functional Profile in Wheat under Mn Stress" Plants 11, no. 19: 2598. https://doi.org/10.3390/plants11192598
APA StyleConceição, T. A., Andrade, G., & Brito, I. (2022). Influence of Intact Mycelium of Arbuscular Mycorrhizal Fungi on Soil Microbiome Functional Profile in Wheat under Mn Stress. Plants, 11(19), 2598. https://doi.org/10.3390/plants11192598






