Zinc Biofortification in Vitis vinifera: Implications for Quality and Wine Production
Abstract
1. Introduction
2. Results
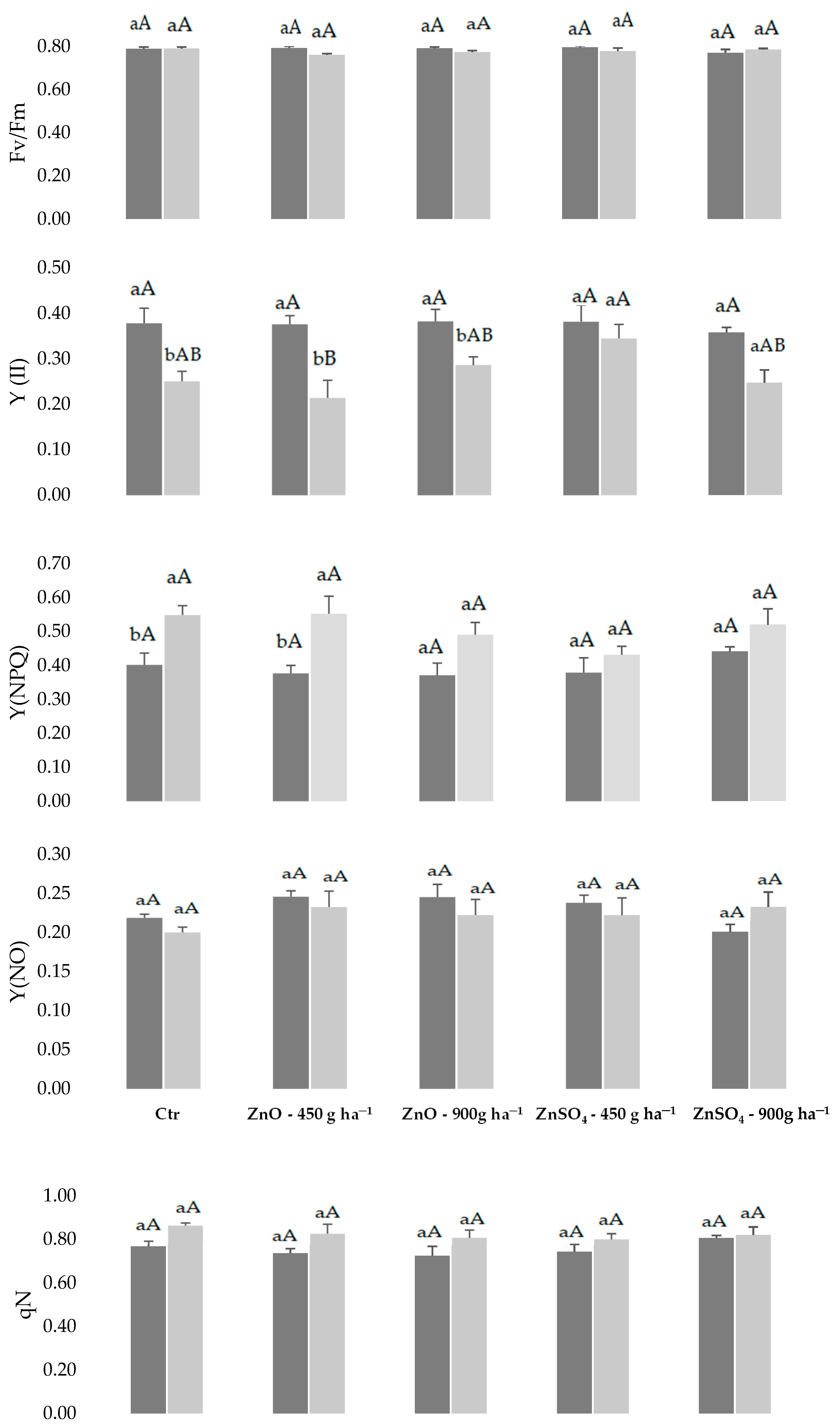
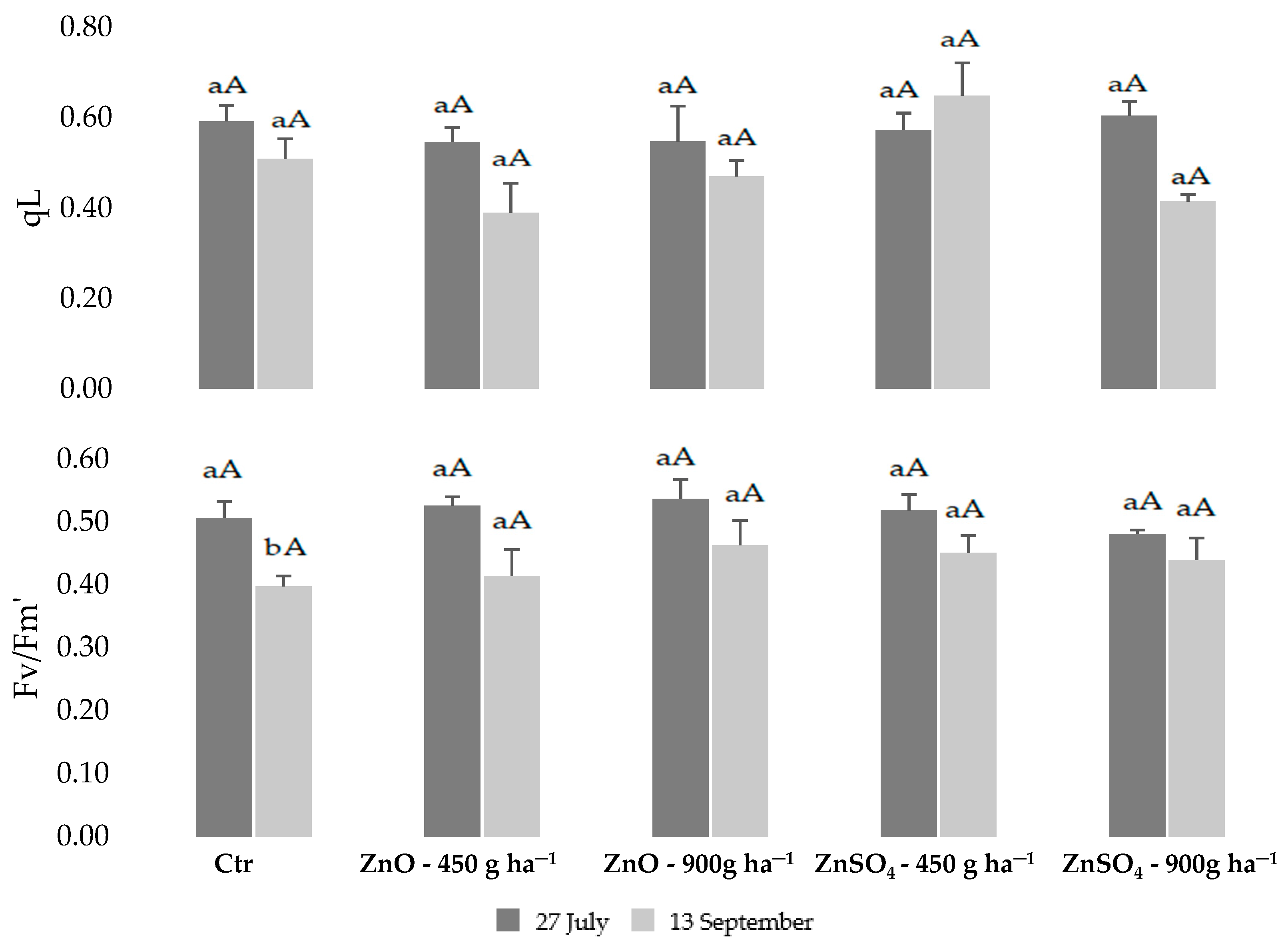
2.1. Physiological Monitoring of Photossintetic Parameters during Biofortification with Zn
2.2. Zinc Content in Leaves, Grapes, and Wine
2.3. Sugar Composition after Zinc Biofortification
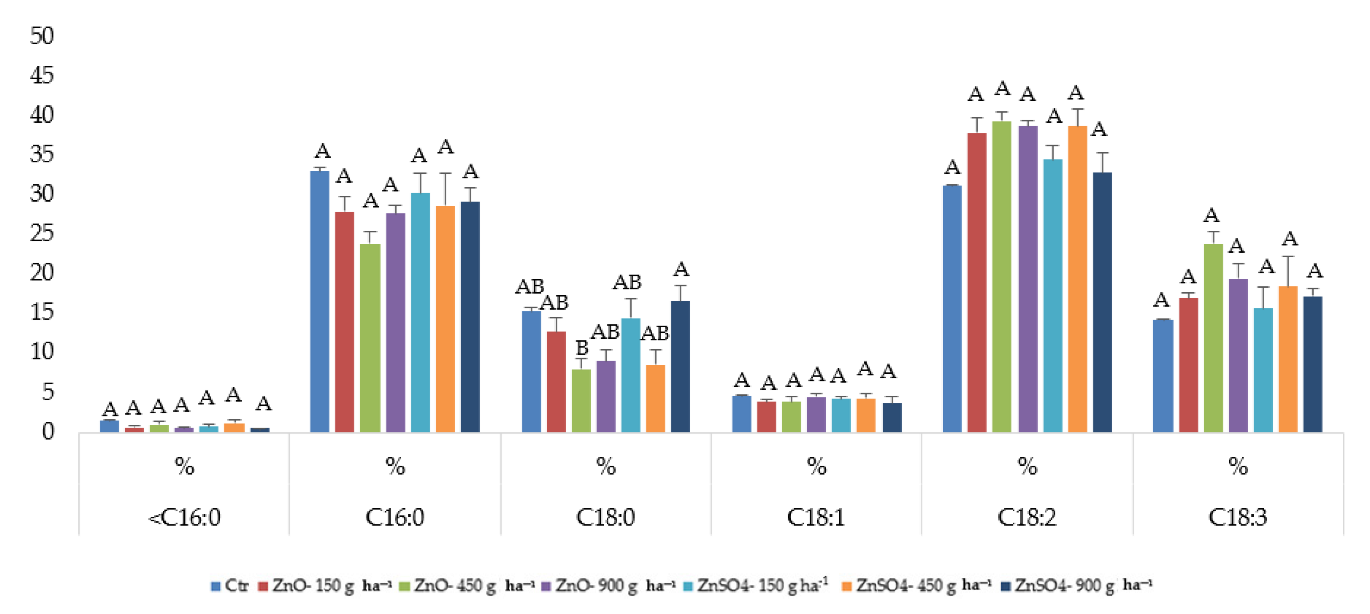
2.4. Fatty Acid Composition after Zinc Agronomic Biofortification
2.5. Colorimetric Parameters after Zinc Agronomic Biofortification
2.6. Physical Characteristics and Sensory Analysis
3. Materials and Methods
3.1. Experimental Fields
3.2. Field Characterization for Zinc Agronomic Biofortification
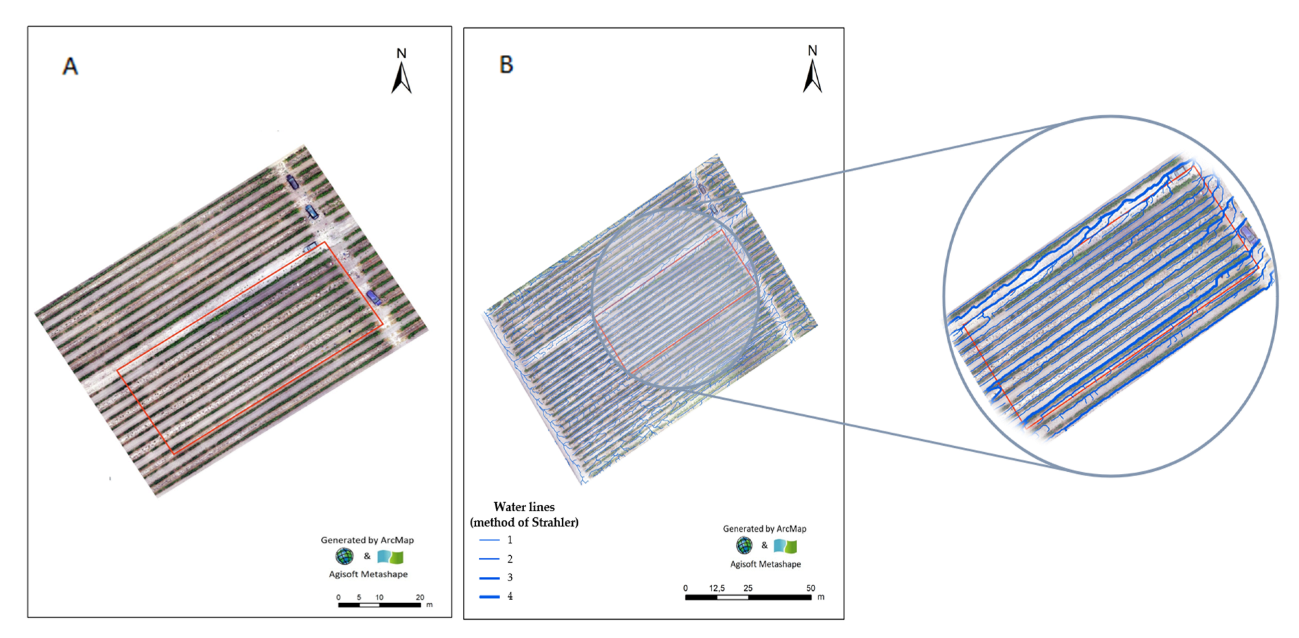
3.3. Orthophotomap and Surface-Water Drainage
3.4. Soil Analysis
3.5. Chlorophyll a Fluorescence
3.6. Analysis of Zn Content in Leaves, Grapes, and Wine
3.7. Sugar Analysis
3.8. Fatty Acid Analysis
3.9. Colorimetric Parameters of Grapes
3.10. Physical and Sensory Parameters of Grapes
3.11. Winemaking
3.12. Statistical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marschner, P. Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Academic Press: San Diego, CA, USA, 2012. [Google Scholar]
- Tsonev, T.; Lidon, F.C. Zinc in plants—An overview. Emir. J. Food Agric. 2012, 24, 322–333. [Google Scholar]
- Rudani, K.; Patel, V.; Prajapati, K. The importance of zinc in plant growth—A review. Int. Res. J. Nat. Appl. Sci. 2018, 5, 38–48. [Google Scholar]
- Hacisalihoglu, G. Zinc (Zn): The last nutrient in the alphabet and shedding light on Zn efficiency for the future of crop production under suboptimal Zn. Plants 2020, 9, 1471. [Google Scholar] [CrossRef]
- Ackova, D.G. Heavy metals and their general toxicity on plants. Plant Sci. Today 2018, 5, 15–19. [Google Scholar] [CrossRef]
- Ghori, N.H.; Ghori, T.; Hayat, M.Q.; Imadi, S.R.; Gul, A.; Altay, V.; Ozturk, M. Heavy metal stress and responses in plants. Int. J. Sci. Environ. Technol. 2019, 16, 1807–1828. [Google Scholar] [CrossRef]
- Reboredo, F. Interaction between copper and zinc and their uptake by Halimione portulacoides (L.) Aellen. Bull. Environ. Contam. Toxicol. 1994, 52, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Krishna, T.P.A.; Maharajan, T.; Roch, G.V.; Ignacimuthu, S.; Ceasar, S.A. Structure, function, regulation and phylogenetic relationship of ZIP family transporters of plants. Front. Plant Sci. 2020, 11, 662. [Google Scholar] [CrossRef]
- Chasapis, C.T.; Ntoupa, P.A.; Spiliopoulou, C.A.; Stefanidou, M.E. Recent aspects of the effects of zinc on human health. Arch. Toxicol. 2020, 94, 1443–1460. [Google Scholar] [CrossRef]
- Chasapis, C.T.; Loutsidou, A.C.; Spiliopoulou, C.A.; Srefanidou, M.E. Zinc and human health: An update. Arch. Toxicol. 2011, 86, 521–534. [Google Scholar] [CrossRef]
- Wessells, K.R.; Brown, K.H. Estimating the global prevalence of zinc deficiency: Results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 2012, 7, e50568. [Google Scholar] [CrossRef]
- Wei, Y.; Shohag, M.J.; Yang, X. Biofortification and bioavailability of rice grain zinc as affected by different forms of foliar zinc fertilization. PLoS ONE 2012, 7, e45428. [Google Scholar] [CrossRef]
- Trijatmiko, K.R.; Dueñas, C.; Tsakirpaloglou, N.; Torrizo, L.; Arines, F.M.; Adeva, C.; Balindong, J.; Oliva, N.; Sapasap, M.V. Biofortified indica rice attains iron and zinc nutrition dietary targets in the field. Sci. Rep. 2016, 6, 19792. [Google Scholar] [CrossRef]
- Poblaciones, M.J.; Rengel, Z. Soil and foliar zinc biofortification in field pea (Pisum sativum L.): Grain accumulation and bioavailability in raw and cooked grains. Food Chem. 2016, 212, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Velu, G.; Singh, R.P.; Huerta-Espino, J.; Peña, R.J. Breeding for enhanced zinc and iron concentration in CIMMYT spring wheat germplasm. Czech J. Genet. Plant Breed. 2011, 47, S174–S177. [Google Scholar] [CrossRef]
- Singh, R.P.; Velu, G. Zinc-Biofortified Wheat: Harnessing Genetic Diversity for Improved Nutritional Quality. Science Brief: Biofortification No. 1 (May 2017), CIMMYT, HarvestPlus, and the Global Crop Diversity Trust. Available online: https://www.harvestplus.org/wp-content/uploads/2017/05/Science-Brief-Biofortification-1_Zinc-Wheat_May2017.pdf (accessed on 24 August 2022).
- Obaid, H.; Shrestha, R.K.; Liu, D.; Elsayed, N.S.; Ni, J.; Ni, C. Biofortification of maize with zinc and its effect on human health. J. Soil Sci. Plant Nutr. 2022, 22, 1792–1804. [Google Scholar] [CrossRef]
- Daccak, D.; Lidon, F.C.; Pessoa, C.C.; Luís, I.C.; Coelho, A.R.F.; Marques, A.C.; Ramalho, J.C.; Silva, M.J.; Rodrigues, A.P.; Guerra, M.; et al. Enrichment of grapes with Zinc-efficiency of foliar fertilization with ZnSO4 and ZnO and implications on winemaking. Plants 2022, 11, 1399. [Google Scholar] [CrossRef]
- Valença, A.W.; Bake, A.; Brouwer, I.D.; Giller, K.E. Agronomic biofortification of crops to fight hidden hunger in sub-Saharan Africa. Glob. Food Sec. 2017, 12, 8–14. [Google Scholar] [CrossRef]
- Di Gioia, F.; Petropoulos, S.A.; Ozores-Hampton, M.; Morgan, K.; Rosskopf, E.N. Zinc and iron agronomic biofortification of Brassicaceae microgreens. J. Agron. 2019, 9, 677. [Google Scholar] [CrossRef]
- Zhao, H.; Xie, X.; Read, P.; Loseke, B.; Gamet, S.; Li, W.; Xu, C. Biofortification with selenium and lithium improves nutraceutical properties of major winery grapes in the Midwestern United States. J. Food Sci. Technol. 2020, 56, 825–837. [Google Scholar] [CrossRef]
- Rehman, A.; Farooq, M.; Ullah, A.; Nadeem, F.; Im, S.Y.; Park, S.K.; Lee, D.J. Agronomic biofortification of zinc in Pakistan: Status, benefits, and constraints. Front. Sustain. Food Syst. 2020, 4, 591722. [Google Scholar] [CrossRef]
- Direcção Geral de Agricultura Desenvolvimento Rural. Carta de Capacidade de Uso do Solo de Portugal—Bases e Normas Adoptadas na sua Elaboração, 6th ed.; Ministério da Economia, Secretaria de Estado da Agricultura, Serviço de Reconhecimento e de Ordenamento Agrário: Lisboa, Portugal, 1972; pp. 25–26. [Google Scholar]
- Reboredo, F.; Simões, M.; Jorge, C.; Mancuso, M.; Martinez, J.; Guerra, M.; Ramalho, J.C.; Pessoa, M.F.; Lidon, F. Metal content in edible crops and agricultural soils due to intensive use of fertilizers and pesticides in Terras da Costa de Caparica (Portugal). Environ. Sci. Pollut. Res. 2019, 26, 2512–2522. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, W.P.; Martins, M.Q.; Fortunato, A.S.; Rodrigues, A.P.; Semedo, J.N.; Simões-Costa, M.C.; Pais, I.P.; Leitão, A.E.; Colwell, F.; Goulão, L.; et al. Long-term air [CO2] strenghtens photosymthetic functioning and mitigates the impact of supra-optimal temperatures in tropical Coffea arabica and C. canephora species. Glob. Change Biol. 2016, 22, 415–431. [Google Scholar] [CrossRef] [PubMed]
- Semedo, J.N.; Rodrigues, A.P.; Lidon, F.C.; Pais, I.P.; Marques, I.; Gouveia, D.; Armengaud, J.; Silva, M.J.; Martins, S.; Semedo, M.C.; et al. Intrinsic non-stomatal resilience to drought of the photosynthetic apparatus in Coffea spp. is strengthened by elevated air [CO2]. Tree Physiol. 2020, 41, 708–727. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zhang, S.B.; Cao, K.F. Cyclic electron flow plays an important role in photoprotection of tropical trees illuminated at temporal chilling temperature. Plant Cell Physiol. 2011, 52, 297–305. [Google Scholar] [CrossRef]
- Klughammer, C.; Schreiber, U. Complementary PS II quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the Saturation Pulse method. PAM Appl. Notes 2008, 1, 27–35. [Google Scholar]
- Kramer, D.M.; Johnson, G.; Kiirats, O.; Edwards, G.E. New flux parameters for the determination of QA redox state and excitation fluxes. Photosynth. Res. 2004, 79, 209–218. [Google Scholar] [CrossRef]
- Krause, G.H.; Jahns, P. Non-photochemical energy dissipation determined by chlorophyll fluorescence quenching: Characterization and function. In Chlorophyll a Fluorescence: A Signature of Photosynthesis; Springer: Dordrecht, The Netherlands, 2004; pp. 463–495. [Google Scholar]
- Schreiber, U. Pulse-Amplitude-Modulation (PAM) fluorometry and saturation pulse method: An overview. In Chlorophyll a Fluorescence: A Signature of Photosynthesis; Springer: Dordrecht, The Netherlands, 2004; pp. 279–319. [Google Scholar]
- Reboredo, F.H.; Pelica, J.; Lidon, F.C.; Pessoa, M.F.; Silva, M.M.; Guerra, M.; Leitão, R.; Ramalho, J.C. The tolerance of Eucalyptus globulus to soil contamination with arsenic. Plants 2021, 10, 627. [Google Scholar] [CrossRef]
- Coelho, A.R.F.; Ramalho, J.C.; Lidon, F.C.; Marques, A.C.; Daccak, D.; Pessoa, C.C.; Luís, I.C.; Guerra, M.; Leitão, R.G.; Semedo, J.M.N.; et al. Foliar spraying of Solanum tuberosum L. with CaCl2 and Ca(NO3)2: Interactions with nutrients accumulation in tubers. Plants 2022, 11, 1725. [Google Scholar] [CrossRef]
- Reboredo, F.H.; Pelica, J.; Lidon, F.C.; Ramalho, J.C.; Pessoa, M.F.; Calvão, T.; Simões, M.; Guerra, M. Heavy metal content of edible plants collected close to an area of intense mining activity (Southern Portugal). Environ. Monit. Assess. 2018, 190, 484. [Google Scholar] [CrossRef]
- Carrondo, M.J.T.; Reboredo, F.; Ganho, R.M.B.; Oliveira, J.F.S. Analysis of sediments for heavy-metals by a rapid electrothermal atomic-absorption procedure. Talanta 1984, 31, 561–564. [Google Scholar] [CrossRef]
- Reboredo, F.H.S.; Ribeiro, C.A.G. Vertical Distribution of Al, Cu, Fe and Zn in the Soil Salt Marshes of the Sado Estuary, Portugal. Int. J. Environ. Stud. 1984, 23, 249–253. [Google Scholar] [CrossRef]
- Medlicott, A.P.; Thompson, A.K. Analysis of sugars and organic acids in ripening mango fruits (Mangifera indica L. var Keitt) by high performance liquid chromatography. J. Sci. Food Agric. 1985, 36, 561–566. [Google Scholar] [CrossRef]
- Vidigal, P.; Duarte, B.; Cavaco, A.R.; Caçador, I.; Figueiredo, A.; Matos, A.R.; Viegas, W.; Monteiro, F. Preliminary diversity assessment of an undervalued tropical bean (Lablab purpureus (L.) Sweet) through fatty acid profiling. Plant Physiol. Biochem. 2018, 132, 508–514. [Google Scholar] [CrossRef]
- Mazliak, P. Plant membrane lipids: Changes and alterations during aging and senescence. In Post-Harvest Physiology and Crop Preservation; Lieberman, M., Ed.; Springer: Boston, MA, USA, 1983; Volume 46, pp. 123–140. [Google Scholar] [CrossRef]
- Marques, A.C.; Lidon, F.C.; Coelho, A.R.F.; Pessoa, C.C.; Luís, I.C.; Scotti-Campos, P.; Simões, M.; Almeida, A.S.; Legoinha, P.; Pessoa, M.F.; et al. Quantification and tissue localization of selenium in rice (Oryza sativa L., Poaceae) grains: A perspective of agronomic biofortification. Plants 2020, 9, 1670. [Google Scholar] [CrossRef] [PubMed]
- Daccak, D.; Pessoa, C.C.; Coelho, A.R.F.; Luís, I.C.; Marques, A.C.; Simões, M.; Reboredo, F.; Pessoa, M.F.; Silva, M.M.; Galhano, C.; et al. Influence of Zinc fertilization for physical and chemical parameters and sensory. In Proceedings of the 1st International Conference on Water Energy Food and Sustainability (ICoWEFS 2021), Leiria, Portugal, 10–12 May 2021; pp. 170–177. [Google Scholar] [CrossRef]
- Ubalde, J.M.; Sort, X.; Zayas, A.; Poch, R.M. Effects of soil and climatic conditions on grape ripening and wine quality of Cabernet Sauvignon. J. Wine Res. 2010, 21, 1–17. [Google Scholar] [CrossRef]
- Van Leeuwen, C.; Roby, J.-P.; de Rességuier, L. Soil-related terroir factors: A review. OENO One 2018, 52, 173–188. [Google Scholar] [CrossRef]
- Van Leeuwen, C.; Trégoat, O.; Choné, X.; Bois, B.; Pernet, D.; Gaudillère, J.-P. Vine water status is a key factor in grape ripening and vintage quality for red Bordeaux wine. How can it be assessed for vineyard management purposes? J. Int. Sci. Vigne Vin 2009, 43, 121–134. [Google Scholar] [CrossRef]
- Ollé, D.; Guiraud, J.L.; Souquet, J.M.; Terrier, N.; Ageorges, A.; Cheynier, V.; Verries, C. Effect of pre- and post-veraison water deficit on proanthocyanidin and anthocyanin accumulation during Shiraz berry development. Aust. J. Grape Wine Res. 2011, 17, 90–100. [Google Scholar] [CrossRef]
- Arnó, J.; Rosell, J.R.; Blanco, R.; Ramos, M.C.; Martínez-Casasnovas, J.A. Spatial variability in grape yield and quality influenced by soil and crop nutrition characteristics. Precis. Agric. 2012, 13, 393–410. [Google Scholar] [CrossRef]
- Bramley, R.G.V.; Ouzman, J.; Boss, P.K. Variation in vine vigour, grape yield and vineyard soils and topography as indicators of variation in the chemical composition of grapes, wine and wine sensory attributes. Aust. J. Grape Wine Res. 2011, 17, 217–229. [Google Scholar] [CrossRef]
- Amorós, J.A.; Bravo, S.; Pérez-de-los-Reyes, C.; Garcia-Navarro, F.J.; Campos, J.A.; Sánchez-Ormeno, M.; Jiménez-Ballesta, R.; Higueras, P. Iron uptake in vineyard soils and relationships with other elements (Zn, Mn and Ca). The case of Castilla-La Mancha, Central Spain. Appl. Geochem. 2018, 88, 17–22. [Google Scholar] [CrossRef]
- Bora, F.D.; Bunea, C.I.; Rusu, T.; Pop, N. Vertical distribution and analysis of micro-macroelements and heavy metals in the system soil-grapevine-wine in vineyard from North-West Romania. Chem. Cent. J. 2015, 9, 19. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kalis, E.J.; Temminghoff, E.J.; Town, R.M.; Unsworth, E.R.; van Riemsdijk, W.H. Relationship between metal speciation in soil solution and metal adsorption at the root surface of ryegrass. J. Environ. Qual. 2008, 37, 2221–2231. [Google Scholar] [CrossRef]
- Wang, R.; Sun, Q.; Chang, Q. Soil types effect on grape and wine composition in Helan Mountain area of Ningxia. PLoS ONE 2015, 10, e0116690. [Google Scholar] [CrossRef]
- Almeida, C.M.R.; Vasconcelos, M.T.S.D. Multielement composition of wines and their precursors including provenance soil and their potentialities as fingerprints of wine origin. J. Agric. Food Chem. 2003, 51, 4788–4798. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, D.E.; Christy, A.G. The role of soil chemistry in wine grape quality and sustainable soil management in vineyards. Water Sci. Technol. 2005, 51, 27–37. [Google Scholar] [CrossRef]
- Hartmann, T.E.; Wollmann, I.; You, Y.; Müller, T. Sensitivity of three phosphate extraction methods to the application of phosphate species differing in immediate plant availability. Agronomy 2019, 9, 29. [Google Scholar] [CrossRef]
- Catarino, S.; Madeira, M.; Monteiro, F.; Caldeira, I.; Sousa, R.B.; Curvelo-Garcia, A. Mineral composition through soil-wine system of Portuguese vineyards and its potential for wine traceability. Beverages 2018, 4, 85. [Google Scholar] [CrossRef]
- Oliver, D.P.; Bramley, R.G.V.; Riches, D.; Porter, I.; Edwards, J. Soil physical and chemical properties as indicators of soil quality in Australian viticulture. Aust. J. Grape Wine Res. 2013, 19, 129–139. [Google Scholar] [CrossRef]
- Longbottom, M. (Ed.) Managing Grapevine Nutrition in a Changing Environment, Research to Practice Manual; Australian Wine Research Institute: Adelaide, Australia, 2009. [Google Scholar]
- Hazelton, P.; Murphy, B.; NSW Department of Natural Resources. Interpreting Soil Test Results: What Do All the Numbers Mean? 2nd ed.; CSIRO Publishing: Clayton, Australia, 2007; 169p.
- Ozcan, M.M. Mineral contents of several grape seeds. Asian J. Chem. 2010, 22, 6480–6488. [Google Scholar]
- Cepo, D.V.; Karoglan, M.; Borgese, L.; Depero, L.E.; Marguí, E.; Jablan, J. Application of benchtop total-reflection X-ray fluorescence spectrometry and chemometrics in classification of origin and type of Croatian wines. Food Chem. X 2022, 13, 100209. [Google Scholar] [CrossRef]
- Greenough, J.D.; Longerich, H.P.; Jackson, S.E. Element fingerprinting of Okanagan Valley wines using ICP-MS: Relationships between wine composition vineyard and wine colour. Aust. J. Grape Wine Res. 1997, 3, 75–83. [Google Scholar] [CrossRef]
- Garrido, A.; Vos, R.C.H.D.; Conde, A.; Cunha, A. Light microclimate-driven changes at transcriptional level in photosynthetic grape berry tissues- photosynthetic grape berry tissues. Plants 2021, 10, 1769. [Google Scholar] [CrossRef] [PubMed]
- Gambetta, J.M.; Holzapfel, B.P.; Stoll, M.; Friedel, M. Sunburn in grapes: A review. Front. Plant Sci. 2021, 11, 604691. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, L.C.; Coito, J.L.; Gonçalves, E.F.; Chaves, M.M.; Amâncio, S. Differential physiological response of the grapevine varieties Touriga Nacional and Trincadeira to combined heat, drought and light stresses. Plant Biol. 2016, 18, 101–111. [Google Scholar] [CrossRef]
- Glenn, D.M.; Yuri, J.A. Photosynthetically active radiation (PAR) × ultraviolet radiation (UV) interact to initiate solar injury in apple. Sci. Hortic. 2013, 162, 117–124. [Google Scholar] [CrossRef]
- Mittler, R.; Vanderauwera, S.; Gollery, M.; Breusegem, F.V. Reactive oxygen gene network of plants. Trends Plant Sci. 2004, 9, 490–498. [Google Scholar] [CrossRef]
- Hussain, T.; Koyro, H.W.; Zhang, W.; Liu, X.; Gul, B.; Liu, X. Low salinity improves photosynthetic performance in panicum antidotale under drought Stress. Front. Plant Sci. 2020, 11, 481. [Google Scholar] [CrossRef]
- Gutiérrez-Gamboa, G.; Garde-Cerdán, T.; Souza-Da Costa, B.; Moreno-Simunovic, Y. Strategies for the improvement of fruit set in Vitis vinifera L. cv. ‘Carménère’ through different foliar biostimulants in two different locations. Ciênc. Téc. Vitiviníc. 2018, 33, 177–183. [Google Scholar] [CrossRef]
- Christensen, L.P. Additives don’t improve zinc uptake in grapevines. Hilgardia 1986, 40, 22–23. [Google Scholar] [CrossRef]
- Trad, M.; Boge, M.; Hamda, B.H.; Renard, C.M.; Harbi, M. The glucose-fructose ratio of wild Tunisian grapes. Cogent Food Agric. 2017, 3, 1374156. [Google Scholar] [CrossRef]
- Walker, R.P.; Bonghi, C.; Varotto, S.; Battistelli, A.; Burbidge, C.A.; Castellarin, S.D.; Chen, Z.H.; Darriet, P.; Moscatello, S.; Rienth, M.; et al. Sucrose metabolism and transport in grapevines, with emphasis on berries and leaves, and insights gained from a cross-species comparison. Int. J. Mol. Sci. 2021, 22, 7794. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D. Comparison of sugars, organic acids and aroma components of five table grapes in Xinjiang. IOP Conf. Ser. Earth Environ. Sci. 2021, 792, 012029. [Google Scholar] [CrossRef]
- Sadras, V.O.; McCarthy, M.G. Quantifying the dynamics of sugar concentration in berries of Vitis vinifera cv. Shiraz: A novel approach based on allometric analysis. Aust. J. Grape Wine Res. 2007, 13, 66–71. [Google Scholar] [CrossRef]
- Jordão, A.M.; Vilela, A.; Cosme, F. From sugar of grape to alcohol of wine: Sensorial impact of alcohol in wine. Beverages 2015, 1, 292–310. [Google Scholar] [CrossRef]
- Ju, Y.-L.; Liu, M.; Zhao, H.; Meng, J.-F.; Fang, Y.-L. Effect of exogenous abscisic acid and methyl jasmonate on anthocyanin composition, fatty acids, and volatile compounds of Cabernet Sauvignon (Vitis vinifera L.) grape berries. Molecules 2016, 21, 1354. [Google Scholar] [CrossRef]
- Yi, C.; Shi, J.; Kramer, J.; Xue, S.; Jiang, Y.; Zhang, M.; Ma, Y.; Pohorly, J. Fatty acid composition and phenolic antioxidants of winemaking pomace powder. Food Chem. 2009, 114, 570–576. [Google Scholar] [CrossRef]
- Szabó, É.; Marosvölgyi, T.; Szilágyi, G.; Kőrösi, L.; Schmidt, J.; Csepregi, K.; Márk, L.; Bóna, Á. Correlations between total antioxidant capacity, polyphenol and fatty acid content of native grape seed and pomace of four different grape varieties in Hungary. Antioxidants 2021, 10, 1101. [Google Scholar] [CrossRef]
- Rocchi, L.; Rustioni, L.; Failla, O. Chlorophyll and carotenoid quantifications in white grape (Vitis vinifera L.) skins by reflectance spectroscopy. Vitis 2016, 55, 11–16. [Google Scholar] [CrossRef]
- Underhill, A.N.; Hirsch, C.D.; Clark, M.D. Evaluating and mapping grape color using image-based phenotyping. Plant Phenomics 2020, 2020, 8086309. [Google Scholar] [CrossRef]
- Qian, X.; Sun, L.; Xu, X.Q.; Zhu, B.Q.; Xu, H.Y. Differential expression of VvLOXA diversifies C6 volatile profiles in some Vitis vinifera table grape cultivars. Int. J. Mol. Sci. 2017, 18, 2705. [Google Scholar] [CrossRef] [PubMed]
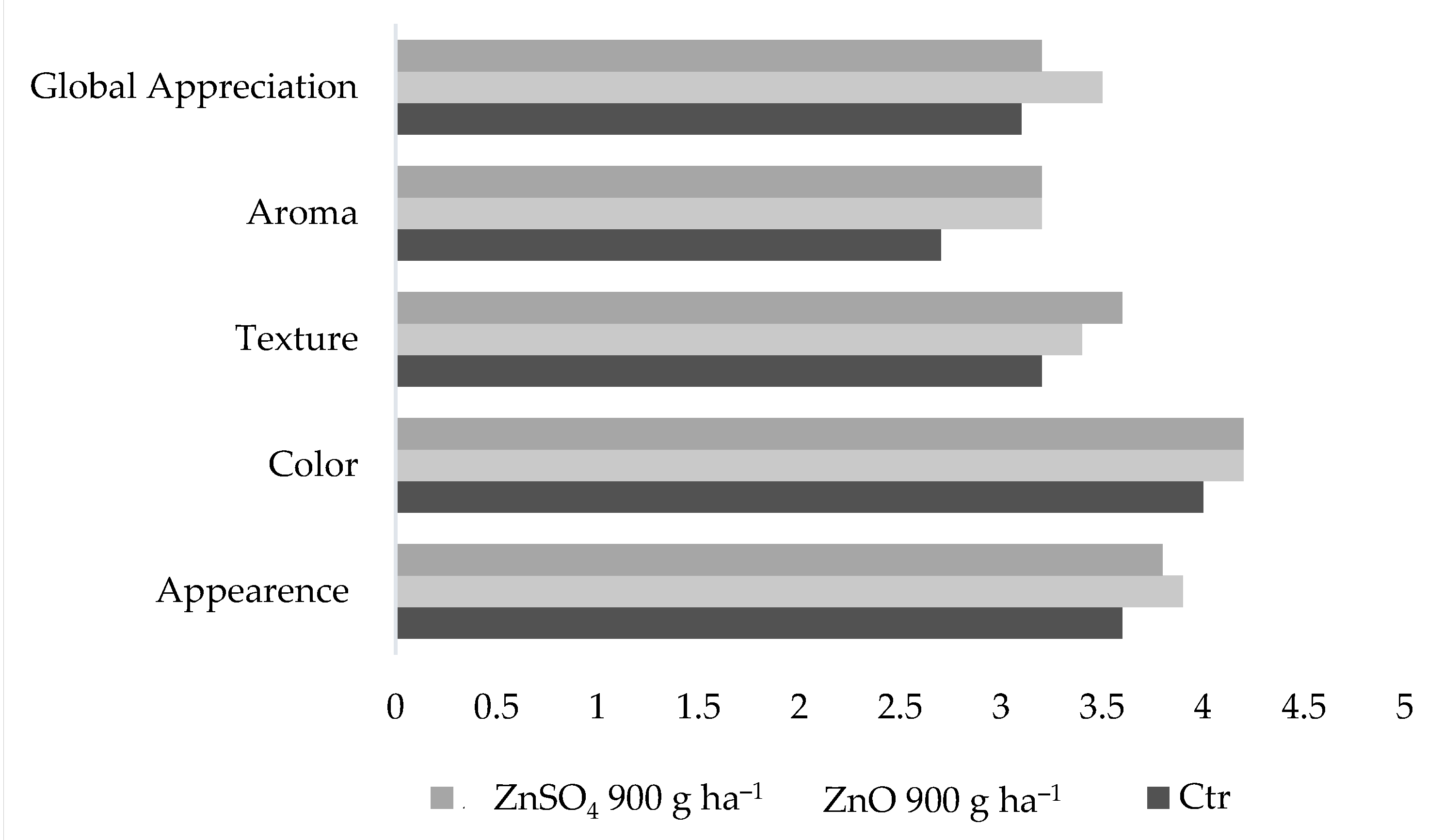
| Aptitude to Accumulate or Drain Surface Water | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Slope Class (%) | Surface Drainage | Area (m2) | Área (%) | ||||||
| 1—[0–5%] | Low | 682.7 | 34.43 | ||||||
| 2—[5–20%] | Moderate | 1224.3.4 | 61.74 | ||||||
| 3—>20% | High | 76.1 | 3.84 | ||||||
| Soil Analysis (0–30 cm deep) (n = 28) | |||||||||
| pH | Electrical Conductivity | Organic Matter | Ca | K | P | Fe | S | Zn | Mn |
| μS cm −1 | % | ppm | |||||||
| 6.85 ± 0.07 | 93.4 ± 17.78 | 1.36 ± 0.15 | 0.54 ± 0.15 | 3.53 ± 0.09 | 0.17 ± 0.02 | 0.35 ± 0.02 | 305.6 ± 117.8 | 58.9 ± 8.99 | 299.0 ± 43.3 |
| Zn Content | ||||
|---|---|---|---|---|
| Leaves (ppm) | Grapes (ppm) | Wine (mg L−1) | ||
| Treatments | Seed | Skin | ||
| Ctr | 32.76 ± 4.36 E | 24.6 ± 1.23 D | 23.4 ± 1.17 D | 0.98 ± 0.01 B |
| ZnO (150 g ha−1) | 110.30 ± 1.39 C,D | 17.1 ± 0.85 E | 37.5 ± 1.88 C | - |
| ZnO (450 g ha−1) | 176.98 ± 32.37 C | 37.1 ± 1.85 B,C | 54.4 ± 2.72 A,B | 1.45 ± 0.05 A |
| ZnO (900 g ha−1) | 337.72 ± 0.00 B | 44.7 ± 2.24 A | 59.4 ± 2.97 A | 1.53 ± 0.03 A |
| ZnSO4 (150 g ha−1) | 91.75 ± 13.45 D,E | 49.9 ± 2.49 A | 54.2 ± 2.71 A,B | - |
| ZnSO4 (450 g ha−1) | 297.47 ± 20.03 B | 42.9 ± 2.15 A,B | 50.0 ± 2.50 B | 1.56 ± 0.02 A |
| ZnSO4 (900 g ha−1) | 496.22 ± 4.57 A | 30.1 ± 1.5 C,D | 39.8 ± 1.99 C | 1.44 ± 0.11 A |
| Sugar Content in Grapes (mg g−1 Fresh Weight) | |||
|---|---|---|---|
| Treatments | Sucrose | Glucose | Fructose |
| Ctr | 2.13 ± 0.61 A | 102.12 ± 21.34 B | 102.78 ± 21.88 A |
| ZnO (150 g ha−1) | 1.90 ± 0.20 A | 111.07 ± 25.24 A,B | 116.96 ± 26.04 A |
| ZnO (450 g ha−1) | 2.85 ± 0.28 A | 88.95 ± 5.490 B | 94.20 ± 6.45 A |
| ZnO (900 g ha−1) | 2.77 ± 0.45 A | 132.92 ± 20.82 A,B | 135.20 ± 19.77 A |
| ZnSO4 (150 g ha−1) | 3.35 ± 0.51 A | 182.51 ± 11.25 A | 158.84 ± 20.23 A |
| ZnSO4 (450 g ha−1) | 2.87 ± 0.58 A | 130.18 ± 11.07 A,B | 135.43 ± 14.24 A |
| ZnSO4 (900 g ha−1) | 2.72 ± 1.19 A | 134.85 ± 6.91 A,B | 140.19 ± 5.79 A |
| Treatments | TFA | DBI |
|---|---|---|
| Ctr | 0.91 ± 0.05 A | 2.21 ± 0.01 A |
| ZnO (150 g ha−1) | 1.05 ± 0.11 A | 3.21 ± 0.34 A |
| ZnO (450 g ha−1) | 1.35 ± 0.21 A | 4.73 ± 0.21 A |
| ZnO (900 g ha−1) | 1.12 ± 0.18 A | 3.83 ± 0.43 A |
| ZnSO4 (150 g ha−1) | 0.81 ± 0.04 A | 2.76 ± 0.57 A |
| ZnSO4 (450 g ha−1) | 1.06 ± 0.07 A | 4.00 ± 1.30 A |
| ZnSO4 (900 g ha−1) | 0.54 ± 0.04 A | 2.69 ± 0.42 A |
| Transmittance (nm) | ||||||
|---|---|---|---|---|---|---|
| Treatments | 450 | 500 | 550 | 570 | 600 | 650 |
| Ctr | 616.00 ± 9.54 A | 522.67 ± 5.21 A | 886.00 ± 10.41 A | 465.33 ± 3.93 A | 546.00 ± 4.16 A | 902.33 ± 6.69 A |
| ZnO (150 g ha−1) | 491.00 ± 106.17 A | 390.33 ± 100.83 A | 679.00 ± 141.57 A | 362.33 ± 71.67 A | 458.33 ± 68.41 A | 783.00 ± 96.17 A |
| ZnO (450 g ha−1) | 555.67 ± 12.99 A | 467.00 ± 9.64 A | 808.00 ± 19.30 A | 415.33 ± 2.73 A | 498.33 ± 8.29 A | 842.67 ± 8.57 A |
| ZnO (900 g ha−1) | 451.00 ± 71.14 A | 362.33 ± 61.20 A | 671.67 ± 42.78 A | 344.00 ± 28.84 A | 382.00 ± 29.50 A | 702.33 ± 69.63 A |
| ZnSO4 (150 g ha−1) | 568.33 ± 44.28 A | 466.00 ± 47.43 A | 767.00 ± 115.11 A | 405.00 ± 52.21 A | 470.00 ± 62.39 A | 839.67 ± 42.45 A |
| ZnSO4 (450 g ha−1) | 576.00 ± 42.36 A | 482.00 ± 36.30 A | 867.67 ± 37.26 A | 458.67 ± 24.01 A | 517.00 ± 30.07 A | 873.00 ± 42.51 A |
| ZnSO4 (900 g ha−1) | 530.00 ± 62.60 A | 422.67 ± 58.81 A | 720.67 ± 36.20 A | 377.33 ± 28.70 A | 436.67 ± 29.08 A | 779.33 ± 54.81 A |
| Physical Characteristics | |||
|---|---|---|---|
| Treatments | Firmness [N] | Penetration Force [N·s] | Pulp Hardness [N] |
| Ctr | 3.47 ± 0.32 A | 15.57 ± 4.47 A | 0.13 ± 0.03 A |
| ZnO (900 g ha−1) | 3.27 ± 0.62 A | 11.46 ± 2.15 A | 0.13 ± 0.05 A |
| ZnSO4 (900 g ha−1) | 2.93 ± 0.47 A | 8.94 ± 2.19 A | 0.17 ± 0.05 A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daccak, D.; Lidon, F.C.; Luís, I.C.; Marques, A.C.; Coelho, A.R.F.; Pessoa, C.C.; Caleiro, J.; Ramalho, J.C.; Leitão, A.E.; Silva, M.J.; et al. Zinc Biofortification in Vitis vinifera: Implications for Quality and Wine Production. Plants 2022, 11, 2442. https://doi.org/10.3390/plants11182442
Daccak D, Lidon FC, Luís IC, Marques AC, Coelho ARF, Pessoa CC, Caleiro J, Ramalho JC, Leitão AE, Silva MJ, et al. Zinc Biofortification in Vitis vinifera: Implications for Quality and Wine Production. Plants. 2022; 11(18):2442. https://doi.org/10.3390/plants11182442
Chicago/Turabian StyleDaccak, Diana, Fernando C. Lidon, Inês Carmo Luís, Ana Coelho Marques, Ana Rita F. Coelho, Cláudia Campos Pessoa, João Caleiro, José C. Ramalho, António E. Leitão, Maria José Silva, and et al. 2022. "Zinc Biofortification in Vitis vinifera: Implications for Quality and Wine Production" Plants 11, no. 18: 2442. https://doi.org/10.3390/plants11182442
APA StyleDaccak, D., Lidon, F. C., Luís, I. C., Marques, A. C., Coelho, A. R. F., Pessoa, C. C., Caleiro, J., Ramalho, J. C., Leitão, A. E., Silva, M. J., Rodrigues, A. P., Guerra, M., Leitão, R. G., Campos, P. S., Pais, I. P., Semedo, J. N., Alvarenga, N., Gonçalves, E. M., Silva, M. M., ... Reboredo, F. H. (2022). Zinc Biofortification in Vitis vinifera: Implications for Quality and Wine Production. Plants, 11(18), 2442. https://doi.org/10.3390/plants11182442




















