High Temporal Variability in Late Blight Pathogen Diversity, Virulence, and Fungicide Resistance in Potato Breeding Fields: Results from a Long-Term Monitoring Study
Abstract
1. Introduction
2. Results
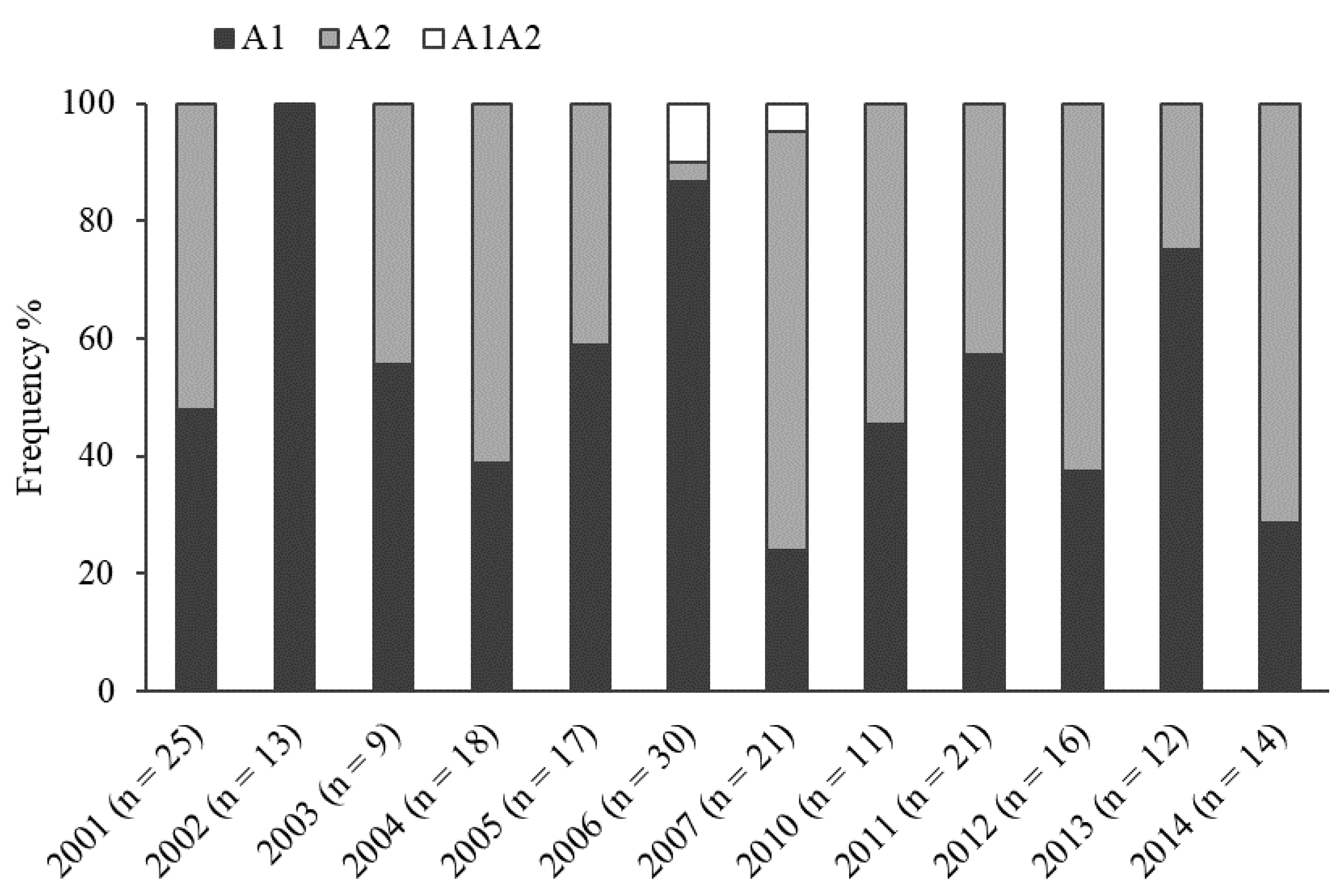
2.1. Mating Type
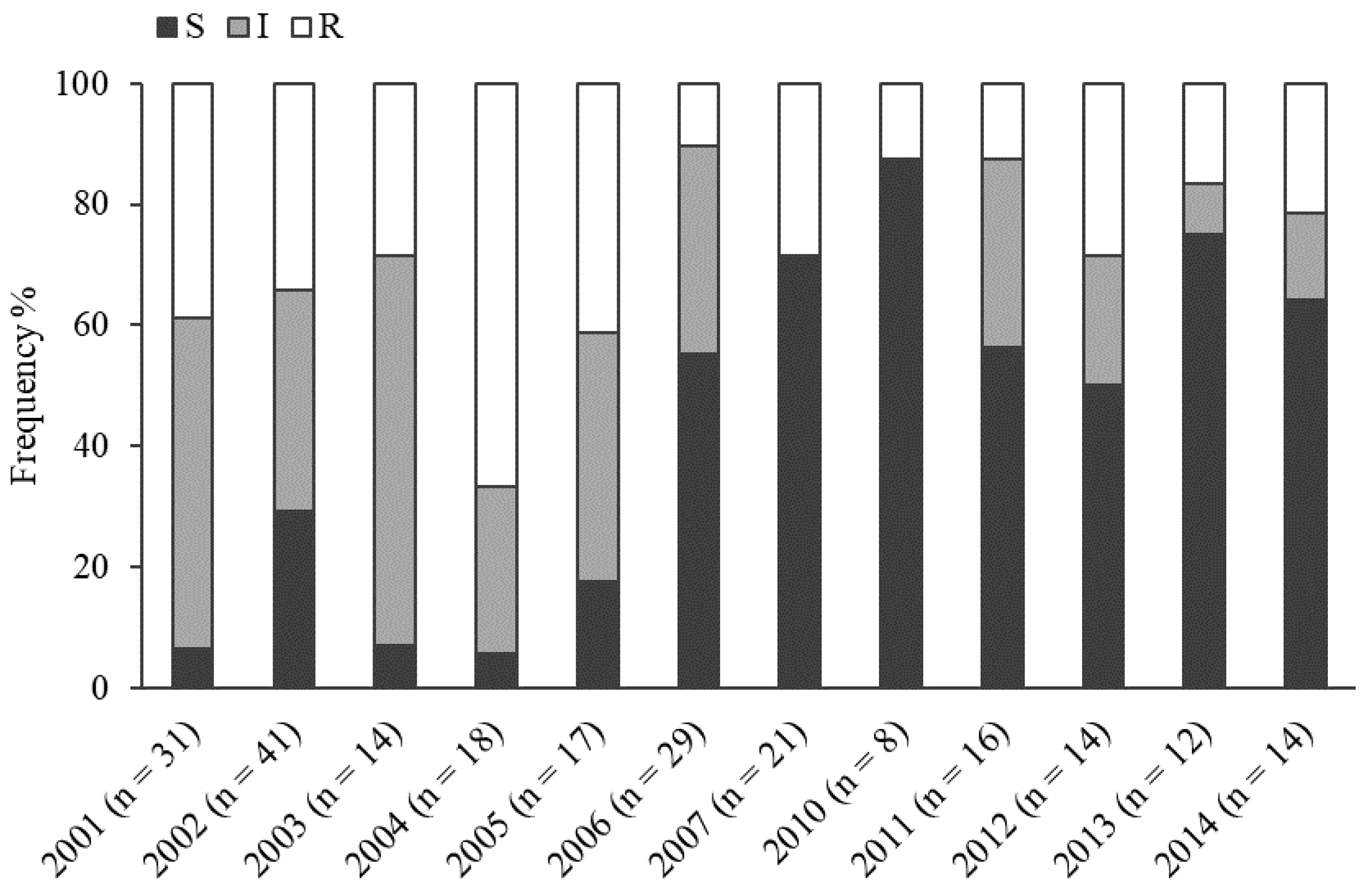
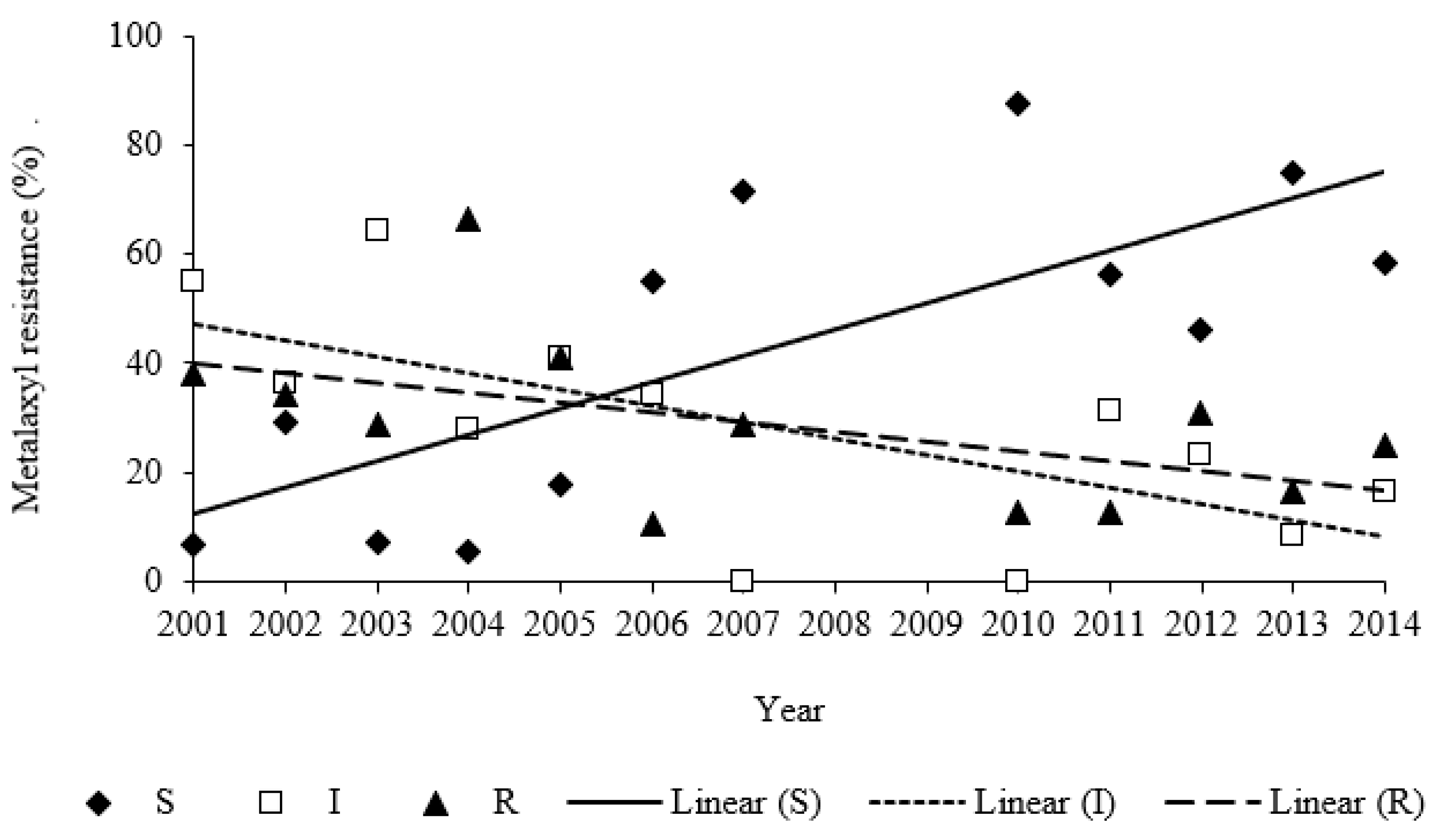
2.2. Metalaxyl Resistance
2.3. Pathotype
3. Discussion
4. Materials and Methods
4.1. Collection and Isolation of P. infestans Isolates
4.2. Phenotypic Analyses
4.3. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fry, W.E.; Birch, P.R.J.; Judelson, H.S.; Grünwald, N.J.; Danies, G.; Everts, K.L.; Gevens, A.J.; Gugino, B.K.; Johnson, D.A.; Johnson, S.B.; et al. Five reasons to consider Phytophthora infestans a reemerging pathogen. Phytopathology 2015, 105, 966–981. [Google Scholar] [CrossRef]
- Fry, W.E. Phytophthora infestans: The itinerant invader; “late blight”: The persistent disease. Phytoparasitica 2020, 48, 87–94. [Google Scholar] [CrossRef]
- Haverkort, A.J.; Boonekamp, P.M.; Hutten, R.; Jacobsen, E.; Lotz, L.A.P.; Kessel, G.J.T.; Visser, R.G.F.; Van der Vossen, E.A.G. Societal costs of late blight in potato and prospects of durable resistance through cisgenic modification. Potato Res. 2008, 51, 47–57. [Google Scholar] [CrossRef]
- Cooke, D.E.L.; Cano, L.M.; Raffaele, S.; Bain, R.A.; Cooke, L.R.; Etherington, G.J.; Deahl, K.L.; Farrer, R.A.; Gilroy, E.M.; Goss, E.M.; et al. Genome analyses of an aggressive and invasive lineage of the Irish potato famine pathogen. PLoS Pathog. 2012, 8, e1002940. [Google Scholar] [CrossRef]
- Mariette, N.; Androdias, A.; Mabon, R.; Corbière, R.; Marquer, B.; Montarry, J.; Andrivon, D. Local adaptation to temperature in populations and clonal lineages of the Irish potato famine pathogen Phytophthora infestans. Ecol. Evol. 2016, 6, 6320–6331. [Google Scholar] [CrossRef]
- Lehsten, V.; Wiik, L.; Hannukkala, A.; Andreasson, E.; Chen, D.; Ou, T.; Liljeroth, E.; Lankinen, Å.; Grenville-Briggs, L. Earlier occurrence and increased explanatory power of climate for the first incidence of potato late blight caused by Phytophthora infestans in Fennoscandia. PLoS ONE 2017, 12, e0177580. [Google Scholar] [CrossRef]
- Janiszewska, M.; Sobkowiak, S.; Stefańczyk, E.; Śliwka, J. Population structure of Phytophthora infestans from a single location in Poland over a long period of time in context of weather conditions. Microb. Ecol. 2021, 81, 746–757. [Google Scholar] [CrossRef]
- Gisi, U.; Walder, F.; Resheat-Eini, Z.; Edel, D.; Sierotzki, H. Changes of genotype, sensitivity and aggressiveness in Phytophthora infestans isolates collected in European countries in 1997, 2006 and 2007. J. Phytopathol. 2011, 159, 223–232. [Google Scholar] [CrossRef]
- Brylinska, M.; Sobkowiak, S.; Stefanczyk, E.; Śliwka, J. Potato cultivation system affects population structure of Phytophthora infestans. Fungal Ecol. 2016, 20, 132–143. [Google Scholar] [CrossRef]
- Hansen, Z.R.; Everts, K.L.; Fry, W.E.; Gevens, A.J.; Grünwald, N.J.; Gugino, B.K.; Johnson, D.A.; Johnson, S.B.; Judelson, H.S.; Knaus, B.J.; et al. Genetic variation within clonal lineages of Phytophthora infestans revealed through genotyping-by-sequencing, and implications for late blight epidemiology. PLoS ONE 2016, 11, e0165690. [Google Scholar] [CrossRef]
- Mariette, N.; Mabon, R.; Corbière, R.; Boulard, F.; Glais, I.; Marquer, B.; Pasco, C.; Montarry, J.; Andrivon, D. Phenotypic and genotypic changes in French populations of Phytophthora infestans: Are invasive clones the most aggressive? Plant Pathol. 2016, 65, 577–586. [Google Scholar] [CrossRef]
- Stroud, J.A.; Shaw, D.S.; Hale, M.D.; Steele, K.A. SSR assessment of Phytophthora infestans populations on tomato and potato in British gardens demonstrates high diversity but no evidence for host specialization. Plant Pathol. 2016, 65, 334–341. [Google Scholar] [CrossRef]
- Zhu, W.; Shen, L.L.; Fang, Z.G.; Yang, L.N.; Zhang, J.F.; Sun, D.L.; Zhan, J. Increased frequency of self-fertile isolates in Phytophthora infestans may attribute to their higher fitness relative to the A1 isolates. Sci. Rep. 2016, 6, 29428. [Google Scholar] [CrossRef]
- Dey, T.; Saville, A.; Myers, K.; Tewari, S.; Cooke, D.E.L.; Tripathy, S.; Fry, W.E.; Ristaino, J.B.; Roy, S.G. Large sub-clonal variation in Phytophthora infestans from recent severe late blight epidemics in India. Sci. Rep. 2018, 8, 4429. [Google Scholar] [CrossRef]
- Saville, A.; Ristaino, J.B. Genetic structure and subclonal variation of extant and recent US lineages of Phytophthora infestans. Phytopathology 2019, 109, 1614–1627. [Google Scholar] [CrossRef]
- Beninal, L.; Bouznad, Z.; Corbiere, R.; Belkhiter, S.; Mabon, R.; Taoutaou, A.; Keddad, A.; Runno-Paurson, E.; Andrivon, D. Distribution of major clonal lineages EU_13_A2, EU_2_A1, and EU_23_A1 of Phytophthora infestans associated with potato late blight across crop seasons and regions in Algeria. Plant Pathol. 2022, 71, 458–469. [Google Scholar] [CrossRef]
- Göre, M.E.; Altin, N.; Myers, K.; Cooke, D.E.L.; Fry, W.E.; Özer, G. Population structure of Phytophthora infestans in Turkey reveals expansion and spread of dominant clonal lineages and virulence. Plant Pathol. 2021, 70, 898–911. [Google Scholar] [CrossRef]
- Saville, A.C.; La Spada, F.; Faedda, R.; Migheli, Q.; Scanu, B.; Ermacora, P.; Gilardi, G.; Fedele, G.; Rossi, V.; Lenzi, N.; et al. Population structure of Phytophthora infestans collected on potato and tomato in Italy. Plant Pathol. 2021, 70, 2165–2178. [Google Scholar] [CrossRef]
- Runno-Paurson, E.; Hansen, M.; Kotkas, K.; Williams, I.H.; Niinemets, Ü.; Einola, A. Evaluation of late blight foliar resistance of potato cultivars in northern Baltic conditions. Zemdirbyste 2019, 106, 45–52. [Google Scholar] [CrossRef]
- Runno-Paurson, E.; Lääniste, P.; Eremeev, V.; Tähtjärv, T.; Kaurilind, E.; Tosens, T.; Niinemets, Ü.; Williams, I.H. Does winter oilseed rape as a winter cover crop influence potato late blight development in an organic crop rotation? Biol. Agric. Hortic. 2020, 36, 71–83. [Google Scholar] [CrossRef]
- Cooke, L.R.; Schepers, H.T.M.; Hermansen, A.; Bain, R.A.; Bradshaw, N.J.; Ritchie, F.; Shaw, D.S.; Evenhuis, A.; Kessel, G.J.T.; Wander, J.G.N.; et al. Epidemiology and integrated control of potato late blight in Europe. Potato Res. 2011, 54, 183–222. [Google Scholar] [CrossRef]
- Schepers, H.T.A.M.; Evenhuis, A.; Spits, H.G. Strategies to control late blight in potatoes in Europe. Acta Hortic. 2009, 834, 79–82. [Google Scholar] [CrossRef]
- Schepers, H.T.A.M.; Kessel, G.J.T.; Lucca, F.; Förch, M.G.; van den Bosch, G.B.M.; Topper, C.G.; Evenhuis, A. Reduced efficacy of fluazinam against Phytophthora infestans in the Netherlands. Eur. J. Plant Pathol. 2018, 151, 947–960. [Google Scholar] [CrossRef]
- Schepers, H.; Hausladen, H.; Vanhavebeke, P. Epidemics and control of early & late blight, 2017 and 2018 in Europe. PPO Spec. Rep. 2019, 19, 11–33. [Google Scholar]
- Schepers, H.; Hausladen, H.; Vanhaverbeke, P. Epidemics and control of early and late blight, 2015 and 2016 in Europe. PPO Spec. Rep. 2017, 18, 11–32. [Google Scholar]
- Hansen, J.G.; Andersson, B.; Bain, R.; Schmiedl, J.; Soellinger, J.; Ritchie, F.; Bucena, L.; Cakir, E.; Cooke, L.; Dubois, L.; et al. The development and control of late blight (Phytophthora infestans) in Europe in 2007 and 2008. PPO Spec. Rep. 2009, 13, 11–30. [Google Scholar]
- Dowley, L.J.; O’Sullivan, E. Metalaxyl resistant strains of Phytophthora infestans (Mont.) de Bary in Ireland. Potato Res. 1981, 24, 417–421. [Google Scholar] [CrossRef]
- Davidse, L.C.; Danial, D.L.; van Westen, C.J. Resistance to metalaxyl in Phytophthora infestans in the Netherlands. Neth. J. Plant Pathol. 1983, 89, 1–20. [Google Scholar] [CrossRef]
- Hannukkala, A.O. History and Consequences of Migrations, Changes in Epidemiology and Population Structure of Potato Late Blight, Phytophthora infestans, in Finland from 1845 to 2011. Ph.D. Thesis, University of Helsinki, Helsinki, Finland, 2012. [Google Scholar]
- Kiiker, R.; Hansen, M.; Williams, I.H.; Cooke, D.E.L.; Runno-Paurson, E. Outcome of sexual reproduction in the Phytophthora infestans population in Estonian potato fields. Eur. J. Plant Pathol. 2018, 152, 395–407. [Google Scholar] [CrossRef]
- Mazákova, J.; Zouhar, M.; Ryšánek, P.; Taborsky, V.; Hausvater, E.; Doležal, P. Sensitivity to fungicides in the isolates of Phytophthora infestans (Mont.) de Bary in the Czech Republic from 2003 to 2008. Plant Prot. Sci. 2011, 47, 5–12. [Google Scholar] [CrossRef]
- Puidet, B.; Mabon, R.; Guibert, M.; Kiiker, R.; Soonvald, L.; Le, V.H.; Eikemo, H.; Dewaegeneire, P.; Saubeau, G.; Chatot, C.; et al. Examining phenotypic traits contributing to the spread in northern European potato crops of EU_41_A2, a new clonal lineage of Phytophthora infestans. Phytopathology 2022, 112, 414–421. [Google Scholar] [CrossRef]
- Hansen, J.G. Results of the EuroBlight Potato Late Blight Monitoring in 2020. 2021. Available online: https://agro.au.dk/forskning/internationale-platforme/euroblight/currently/news/nyhed/artikel/results-of-the-euroblight-potato-late-blight-monitoring-in-2020 (accessed on 15 March 2022).
- Guyomard, H.; Bureau, J.C.; Chatellier, V.; Détang-Dessendre, C.; Dupraz, P.; Jacquet, F.; Reboud, X.; Réquillart, V.; Soler, L.G.; Tysebaert, M. Research for AGRI Committee—The Green Deal and the CAP: Policy Implications to Adapt Farming Practices and to Preserve the EU’s Natural Resources; European Parliament, Policy Department for Structural and Cohesion Policies: Brussels, Belgium, 2020; Available online: https://www.europarl.europa.eu/RegData/etudes/STUD/2020/629214/IPOL_STU(2020)629214_EN.pdf (accessed on 20 June 2022).
- Runno-Paurson, E.; Williams, I.H.; Metspalu, L.; Kaart, T.; Mand, M. Current potato varieties are too susceptible to late blight to be grown without chemical control under North-East European conditions. Acta Agric. Scand. B Soil Plant Sci. 2013, 63, 80–88. [Google Scholar] [CrossRef]
- Armstrong, M.R.; Vossen, J.; Lim, T.Y.; Hutten, R.C.B.; Xu, J.; Strachan, S.M.; Harrower, B.; Champouret, N.; Gilroy, E.M.; Hein, I. Tracking disease resistance deployment in potato breeding by enrichment sequencing. Plant Biotechnol. J. 2019, 17, 540–549. [Google Scholar] [CrossRef]
- Schönhals, E.N.; Ortega, E.M.; Barandalla, L.; Aragones, A.; Ruiz de Galarreta, J.I.; Liao, J.C.; Sanetomo, R.; Walkemeier, B.; Tacke, E.; Ritter, E.; et al. Identification and reproducibility of diagnostic DNA markers for tuber starch and yield optimization in a novel association mapping population of potato (Solanum tuberosum L.). Theor. Appl. Genet. 2016, 129, 767–785. [Google Scholar] [CrossRef]
- Morris, W.L.; Taylor, M.A. Improving flavor to increase consumption. Am. J. Potato Res. 2019, 96, 195–200. [Google Scholar] [CrossRef]
- Sarv, J. The problem of disease resistance in potato growing and variety breeding in the Estonian SSR. In Daily Problems of Potato Growing 2; Sinijärv, K., Ed.; Valgus: Tallinn, Estonia, 1974; pp. 161–168. (In Estonian) [Google Scholar]
- Tähtjärv, T. Cultivar resistance and population studies of late blight pathogen in potato breeding in Estonia. Ph.D. Thesis, Estonian University of Life Sciences, Tartu, Estonia, 2016; 156p. [Google Scholar]
- Eriksson, D.; Carlson-Nilsson, U.; Ortiz, R.; Andreasson, E. Overview and breeding strategies of table potato production in Sweden and the Fennoscandian region. Potato Res. 2016, 59, 279–294. [Google Scholar] [CrossRef]
- Aav, A.; Skrabule, I.; Bimšteine, G.; Kaart, T.; Williams, I.H.; Runno-Paurson, E. The structure of mating type, metalaxyl resistance and virulence of Phytophthora infestans isolates collected from Latvia. Zemdirbyste 2015, 102, 335–342. [Google Scholar] [CrossRef]
- Runno-Paurson, E.; Hannukkala, A.; Kotkas, K.; Koppel, M.; Williams, I.H.; Mänd, M. Population changes and phenotypic diversity of Phytophthora infestans isolates from Estonia and Finland. J. Plant Pathol. 2014, 96, 85–95. [Google Scholar]
- Runno-Paurson, E.; Ronis, A.; Hansen, M.; Aav, A.; Williams, I.H. Lithuanian populations of Phytophthora infestans revealed a high phenotypic diversity. J. Plant Dis. Prot. 2015, 122, 57–65. [Google Scholar] [CrossRef]
- Lehtinen, A.; Hannukkala, A.; Rantanen, T.; Jauhiainen, L. Phenotypic and genetic variation in Finnish potato-late blight populations, 1997−2000. Plant Pathol. 2007, 56, 480–491. [Google Scholar] [CrossRef]
- Chmielarz, M.; Sobkowiak, S.; Dębski, K.; Cooke, D.E.L.; Brurberg, M.B.; Śliwka, J. Diversity of Phytophthora infestans from Poland. Plant Pathol. 2014, 63, 203–211. [Google Scholar] [CrossRef]
- Sedlák, P.; Mazáková, J.; Sedláková, V.; Ryšánek, P.; Vejl, P.; Doležal, P. Virulence and mating type of Phytophthora infestans isolates in the Czech Republic. Sci. Agric. Bohem. 2017, 48, 185–192. [Google Scholar] [CrossRef]
- Stellingwerf, J.S.; Phelan, S.; Doohan, F.M.; Ortiz, V.; Griffin, D.; Bourke, A.; Hutten, R.C.B.; Cooke, D.E.L.; Kildea, S.; Mullins, E. Evidence for selection pressure from resistant potato genotypes but not from fungicide application within a clonal Phytophthora infestans population. Plant Pathol. 2018, 67, 1528–1538. [Google Scholar] [CrossRef]
- Alor, N.; Tierno, R.; Cooke, D.E.L.; Ruiz de Galarreta, J.I. Characterisation of Phytophthora infestans isolates of potato crops from Spain. Potato Res. 2019, 62, 456–463. [Google Scholar] [CrossRef]
- Runno-Paurson, E.; Agho, C.A.; Zoteyeva, N.; Koppel, M.; Hansen, M.; Hallikma, T.; Cooke, D.E.L.; Nassar, H.; Niinemets, Ü. Highly diverse Phytophthora infestans populations infecting potato crops in Pskov region, North-West Russia. J. Fungi 2022, 8, 472. [Google Scholar] [CrossRef]
- Kuznetsova, M.A.; Statsyuk, N.V.; Rogozhin, A.N.; Ulanova, T.I.; Morozova, E.V.; Demidova, V.N. Monitoring of Phytophthora infestans strains isolated from potato and tomato in Moscow Region (2009−2017). Dostizheniya Nauk. I Tekhniki APK 2018, 32, 28–33. [Google Scholar]
- Kanetis, L.; Pittas, L.; Nikoloudakis, N.; Cooke, D.E.; Ioannou, N. Characterization of Phytophthora infestans populations in Cyprus, the southernmost potato-producing European country. Plant Dis. 2021, 105, 7–417. [Google Scholar] [CrossRef]
- EuroBlight. A Potato Late Blight Network for Europe. Available online: https://agro.au.dk/forskning/internationale-platforme/euroblight (accessed on 20 June 2022).
- Rekad, F.Z.; Cooke, D.E.L.; Puglisi, I.; Randall, E.; Guenaoui, Y.; Bouznad, Z.; Evoli, M.; Pane, A.; Schena, L.; di San Lio, G.M.; et al. Characterization of Phytophthora infestans populations in northwestern Algeria during 2008−2014. Fungal Biol. 2017, 121, 467–477. [Google Scholar] [CrossRef]
- Raza, W.; Ghazanfar, M.U.; Sullivan, L.; Cooke, D.E.L.; Cooke, L.R. Mating type and aggressiveness of Phytophthora infestans (Mont.) de Bary in potato-growing areas of Punjab, Pakistan, 2017–2018 and identification of genotype 13_A2 in 2019–2020. Potato Res. 2020, 64, 115–129. [Google Scholar] [CrossRef]
- Hansen, J.G.; Lassen, P.; Cooke, D.E.L.; Lees, A.; Kessel, G.; Andrivon, D.; Corbiere, R. Results of the EuroBlight Potato Late Blight Monitoring in 2021. 2022. Available online: https://agro.au.dk/forskning/internationale-platforme/euroblight/currently/news/nyhed/artikel/monitoring-2021 (accessed on 16 June 2022).
- Cooke, D.E.L.; Kessel, G.; Hansen, J.G. The Evolving Population of Phytophthora Infestans; Global Context and Consequences for IPM. 2022. Available online: https://agro.au.dk/fileadmin/euroblight/Workshops/Monte_Verita/Presentations/1_2_Cook.pdf. (accessed on 15 June 2022).
- Andrivon, D. Invasions Threaten Sustainable Late Blight Control. Open Access News. Agriculture News. 2021. Available online: https://www.openaccessgovernment.org/invasions-threaten-sustainable-late-blight-control/104928/ (accessed on 11 May 2022).
- Lehtinen, A.; Hannukkala, A.; Andersson, B.; Hermansen, A.; Le, V.H.; Naerstad, R.; Brurberg, M.B.; Nielsen, B.J.; Hansen, J.G.; Yuen, J. Phenotypic variation in Nordic populations of Phytophthora infestans in 2003. Plant Pathol. 2008, 57, 227–234. [Google Scholar] [CrossRef]
- Mazákova, J.; Zouhar, M.; Sedlák, P.; Zusková, E.; Ryšánek, P.; Hausvater, E. Sensitivity to fungicides and essential oils in Czech isolates of Phytophthora infestans. Sci. Agric. Bohem. 2018, 49, 69–77. [Google Scholar]
- Brurberg, M.B.; Elameen, A.; Le, V.H.; Naerstad, R.; Hermansen, A.; Lehtinen, A.; Hannnukkala, A.; Nielsen, B.; Hansen, J.; Andersson, B.; et al. Genetic analysis of Phytophthora infestans populations in the Nordic European countries reveals high genetic variability. Fungal Biol. 2011, 115, 335–342. [Google Scholar] [CrossRef]
- Sjöholm, L.; Andersson, B.; Högberg, N.; Widmark, A.K.; Yuen, J. Genotypic diversity and migration patterns of Phytophthora infestans in the Nordic countries. Fungal Biol. 2013, 117, 722–730. [Google Scholar] [CrossRef]
- Runno-Paurson, E.; Kiiker, R.; Joutsjoki, T.; Hannukkala, A. High genotypic diversity found among population of Phytophthora infestans collected in Estonia. Fungal Biol. 2016, 120, 385–392. [Google Scholar] [CrossRef]
- Kiiker, R.; Skrabule, I.; Ronis, A.; Cooke, D.E.L.; Hansen, J.G.; Williams, I.H.; Mänd, M.; Runno-Paurson, E. Diversity of populations of Phytophthora infestans in relation to patterns of potato crop management in Latvia and Lithuania. Plant Pathol. 2019, 68, 1207–1214. [Google Scholar] [CrossRef]
- Pobedinskaya, M.A.; Elansky, S.N.; Statsyuk, N.V.; Plyakhnevich, M.P. Fungicide resistance of Russian Phytophthora infestans strains. PPO Spec. Rep. 2012, 15, 243–248. [Google Scholar]
- Statsyuk, N.V.; Kozlovskaya, I.N.; Koslovsky, B.E.; Ulanova, T.I.; Morozova, E.V.; Kuznetsova, M. Changes in phenotypic characteristics of the Moscow Phytophthora infestans population in the period of 2000–2011. In Proceedings of the 4th International Symposium “Agrosym 2013”, Jahorina, Bosnia and Herzegovina, 3–6 October 2013; Kovačević, D., Ed.; Faculty of Agriculture, University of East Sarajevo: Jahorina, Bosnia and Herzegovina, 2013; pp. 607–613. [Google Scholar]
- Janiszewska, M.; Sobkowiak, S.; Ludwiczewska, M.; Cooke, D.E.L.; Lenartowicz, T.; Śliwka, J. Role of Seed Potatoes in Local Migrations of Phytophthora Infestans. 2022. Available online: https://agro.au.dk/fileadmin/euroblight/Workshops/Monte_Verita/Presentations/2_6_Sliwka.pdf (accessed on 25 May 2022).
- Chowdappa, P.; Nirmal Kumar, B.J.; Madhura, S.; Mohan Kumar, S.P.; Myers, K.L.; Fry, W.E.; Cooke, D.E.L. Severe outbreaks of late blight on potato and tomato in South India caused by recent changes in the Phytophthora infestans population. Plant Pathol. 2015, 64, 191–199. [Google Scholar] [CrossRef]
- Kessel, G.; Moene, A.; van Valkengoed, E.; van der Voet, P.; Michielsen, J.M.; Ahsan, H.; Schmelzer, T.; Maroof, M.; Syed, A.; Hengsdijk, H. Geodata to control potato late blight in Bangladesh. PAGV Spec. Rep. 2017, 18, 59–60. [Google Scholar]
- Cooke, L.R.; Lasserre, C.; Maurer, A. AsiaBlight: Past, present and future. PAGV Spec. Rep. 2019, 19, 37–44. [Google Scholar]
- Runno-Paurson, E.; Fry, W.E.; Remmel, T.; Mänd, M.; Myers, K.L. Phenotypic and genotypic characterisation of Estonian isolates of Phytophthora infestans in 2004–2007. J. Plant Pathol. 2010, 92, 375–384. [Google Scholar]
- Runno-Paurson, E.; Kiiker, R.; Aav, A.; Hansen, M.; Williams, I.H. Distribution of mating types, metalaxyl sensitivity and virulence races of Phytophthora infestans in Estonia. Agron. Res. 2016, 14, 220–227. [Google Scholar]
- Alvarez-Morezuelas, A.; Alor, N.; Barandalla, L.; Ritter, E.; Ruiz de Galarreta, J.I. Virulence of Phytophthora infestans isolates from potato in Spain. Plant Prot. Sci. 2021, 57, 279–288. [Google Scholar]
- Jakobson, L.; (Estonian Crop Research Institute, Jõgeva, Estonia). Unpublished work. 2022.
- Hermansen, A.; Hannukkala, A.; Hafskjold Nærstad, R.; Brurberg, M.B. Variation in populations of Phytophthora infestans in Finland and Norway: Mating type, metalaxyl resistance and virulence phenotype. Plant Pathol. 2000, 49, 11–22. [Google Scholar] [CrossRef]
- Zoteyeva, N.M.; Patrikeeva, M.V. Phenotypic characteristics of North-West Russian populations of Phytophthora infestans (2003−2008). PPO Spec. Rep. 2010, 14, 213–216. [Google Scholar]
- Śliwka, J.; Sobkowiak, S.; Lebecka, R.; Avendano-Corcoles, J.; Zimnoch-Guzowska, E. Mating type, virulence, aggressiveness and metalaxyl resistance of isolates of Phytophthora infestans in Poland. Potato Res. 2006, 49, 155–166. [Google Scholar] [CrossRef]
- Michalska, M.A.; Sobkowiak, S.; Flis, B.; Zimnoch-Guzowska, E. Virulence and aggressiveness of Phytophthora infestans isolates collected in Poland from potato and tomato plants identified no strong specificity. Eur. J. Plant Pathol. 2016, 144, 325–336. [Google Scholar] [CrossRef]
- Flier, W.G.; Kroon, L.P.N.M.; Hermansen, A.; van Raaij, H.M.G.; Speiser, B.; Tamm, L.; Fuchs, J.G.; Lambion, J.; Razzaghian, J.; Andrivon, D.; et al. Genetic structure and pathogenicity of populations of Phytophthora infestans from organic potato crops in France, Norway, Switzerland and the United Kingdom. Plant Pathol. 2007, 56, 562–572. [Google Scholar] [CrossRef]
- Blandón-Díaz, J.U.; Widmark, A.K.; Hannukkala, A.; Andersson, B.; Högberg, N.; Yuen, J.E. Phenotypic variation within a clonal lineage of Phytophthora infestans infecting both tomato and potato in Nicaragua. Phytopathology 2012, 102, 323–330. [Google Scholar] [CrossRef]
- Gibson, A.K.; Nguyen, A.E. Does genetic diversity protect host populations from parasites? A meta-analysis across natural and agricultural systems. Evol. Lett. 2021, 5, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Corredor-Moreno, P.; Saunders, D.G.O. Expecting the unexpected: Factors influencing the emergence of fungal and oomycete plant pathogens. New Phytol. 2020, 225, 118–125. [Google Scholar] [CrossRef]
- Andrivon, D.; Montarry, J.; Fournet, S. Plant health in a one health world: Missing links and hidden treasures. Plant Pathol. 2022, 71, 23–29. [Google Scholar] [CrossRef]
- Tähtjärv, T.; (Estonian Crop Research Institute, Jõgeva, Estonia). Unpublished work. 2022.
- Oliver, R.P.; Hewitt, H.G. Fungicides in Crop Protection, 2nd ed.; CABI: Wallingford, UK, 2014; 190p. [Google Scholar]
- Mboup, M.K.; Sweigard, J.W.; Carroll, A.; Jaworska, G.; Genet, J.L. Genetic mechanism, baseline sensitivity and risk of resistance to oxathiapiprolin in oomycetes. Pest Manag. Sci. 2022, 78, 905–913. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Green Deal: Pioneering Proposals to Restore Europe’s Nature by 2050 and Halve Pesticide Use by 2030. Available online: https://ec.europa.eu/commission/presscorner/detail/en/ip_22_3746 (accessed on 22 June 2022).
- Casañas, F.; Simó, J.; Casals, J.; Prohens, J. Toward an Evolved Concept of Landrace. Front. Plant Sci. 2017, 8, 145. [Google Scholar] [CrossRef] [PubMed]
- Lázaro, E.; Makowski, D.; Vicent, A. Decision support systems halve fungicide use compared to calendar-based strategies without increasing disease risk. Commun. Earth Environ. 2021, 2, 224. [Google Scholar] [CrossRef]
- Jeger, M.; Beresford, R.; Bock, C.; Brown, N.; Fox, A.; Newton, A.; Vicent, A.; Xu, X.; Yuen, J. Global challenges facing plant pathology: Multidisciplinary approaches to meet the food security and environmental challenges in the mid-twenty-first century. CABI Agric. Biosci. 2021, 2, 1–18. [Google Scholar] [CrossRef]
- Peltonen-Sainio, P.; Juvonen, J.; Korhonen, N.; Parkkila, P.; Sorvali, J.; Gregow, H. Climate change, precipitation shifts and early summer drought: An irrigation tipping point for Finnish farmers? Clim. Risk Manag. 2021, 33, 100334. [Google Scholar] [CrossRef]
- Peltonen-Sainio, P.; Sorvali, J.; Kaseva, J. Finnish farmers’ views towards fluctuating and changing precipitation patterns pave the way for the future. Agric. Water Manag. 2021, 255, 107011. [Google Scholar] [CrossRef]
- Rakosy-Tican, E.; Thieme, R.; Nachtigall, M.; Molnar, I.; Denes, T.E. The recipient potato cultivar influences the genetic makeup of the somatic hybrids between five potato cultivars and one cloned accession of sexually incompatible species Solanum bulbocastanum Dun. Plant Cell Tissue Organ Cult. 2015, 122, 395–407. [Google Scholar] [CrossRef]
- Haverkort, A.J.; Struik, P.C.; Visser, R.G.F.; Jacobsen, E. Applied biotechnology to combat late blight in potato caused by Phytophthora infestans. Potato Res. 2009, 52, 29–264. [Google Scholar] [CrossRef]
- Jo, K.R.; Zhu, S.; Bai, Y.; Hutten, R.C.B.; Kessel, G.J.T.; Vleeshouwers, V.G.A.A.; Jacobsen, E.; Visser, R.G.F.; Vossen, J.H. Towards sustainable potato late blight resistance by cisgenic R gene pyramiding. In Plant Pathogen Resistance Biotechnology; Collinge, D.B., Ed.; John Wiley and Sons: Hoboken, NJ, USA, 2016; pp. 171–191. [Google Scholar]
- Rakosy-Tican, E.; Thieme, R.; Konig, J.; Nachtigall, M.; Hammann, T.; Denes, T.E.; Kruppa, K.; Molnar-Lang, M. Introgression of two broad-spectrum late blight resistance genes, Rpi-Blb1 and Rpi-Blb3, from Solanum bulbocastanum Dun plus race-specific R genes into potato pre-breeding lines. Front. Plant Sci. 2020, 11, 699. [Google Scholar] [CrossRef]
- Fernando, D.W.G.; Oghenekaro, A.O.; Tucker, J.R.; Badea, A. Building on a foundation: Advances in epidemiology, resistance breeding, and forecasting research for reducing the impact of fusarium head blight in wheat and barley. Can. J. Plant Pathol. 2021, 43, 495–526. [Google Scholar] [CrossRef]
- Hermeziu, M. The relation between technologies for late blight (Phytophthora infestans) and the yield components (biomass) of different potato varieties. Rom. Agric. Res. 2021, 38, 155–162. [Google Scholar]
- Islam, H.; Masud, M.; Jannat, M.; Hossain, M.I.; Islam, S.; Alam, Z.; Serneels, F.J.B.; Islam, I. Potentiality of formulated bioagents from lab to field: A sustainable alternative for minimizing the use of chemical fungicide in controlling potato late blight. Sustainability 2022, 14, 4383. [Google Scholar] [CrossRef]
- Runno-Paurson, E.; Hannukkala, A.; Williams, I.H.; Koppel, M.; Mand, M. The structure of mating type, virulence, metalaxyl resistance of Phytophthora infestans in a long-term phenotypic study in distinct location in Eastern Estonia. J. Plant Dis. Prot. 2012, 119, 45–52. [Google Scholar] [CrossRef]
- Runno-Paurson, E.; Fry, W.E.; Myers, K.L.; Koppel, M.; Mänd, M. Characterisation of Phytophthora infestans isolates collected from potato in Estonia during 2002–2003. Eur. J. Plant Pathol. 2009, 124, 565–575. [Google Scholar] [CrossRef]
- Malcolmson, J.F.; Black, W. New R genes in Solanum demissum lindl. And their complementary races of Phytophthora infestans (Mont.) de bary. Euphytica 1966, 15, 199–203. [Google Scholar] [CrossRef]
- Sheldon, A.L. Equitability indices: Dependence on the species count. Ecology 1969, 50, 466–467. [Google Scholar] [CrossRef]
| Year | Virulence to Potato Resistance Gene (%) | Number of Virulence Factors per Isolate | Number of Tested Isolates | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 | R9 | R10 | R11 | |||
| 2001 | 91 | 71 | 83 | 97 | 14 | 69 | 100 | 54 | 46 | 97 | 100 | 8.2 c* | 35 |
| 2002 | 93 | 46 | 100 | 83 | 7 | 61 | 98 | 44 | 20 | 71 | 49 | 6.7 b | 41 |
| 2003 | 64 | 50 | 86 | 71 | 7 | 50 | 64 | 79 | 21 | 86 | 86 | 6.6 b | 14 |
| 2004 | 84 | 63 | 89 | 84 | 5 | 32 | 100 | 42 | 11 | 95 | 100 | 7.1 bc | 19 |
| 2005 | 100 | 94 | 100 | 100 | 56 | 67 | 100 | 56 | 17 | 100 | 100 | 8.9 d | 18 |
| 2006 | 90 | 60 | 83 | 93 | 7 | 53 | 97 | 47 | 3 | 83 | 100 | 7.2 bc | 30 |
| 2007 | 100 | 78 | 91 | 96 | 17 | 35 | 100 | 43 | 0 | 100 | 96 | 7.6 bc | 23 |
| 2010 | 64 | 45 | 91 | 64 | 9 | 36 | 82 | 27 | 9 | 73 | 64 | 5.6 a | 11 |
| 2011 | 81 | 81 | 81 | 71 | 10 | 62 | 76 | 14 | 10 | 86 | 81 | 6.6 b | 21 |
| 2012 | 87 | 93 | 67 | 87 | 0 | 60 | 100 | 0 | 13 | 87 | 100 | 6.9 b | 15 |
| 2013 | 100 | 100 | 100 | 100 | 0 | 33 | 100 | 83 | 0 | 100 | 100 | 8.2 c | 12 |
| 2014 | 100 | 83 | 100 | 100 | 0 | 8 | 100 | 25 | 17 | 100 | 100 | 7.3 bc | 12 |
| Total | 89 | 69 | 89 | 88 | 12 | 51 | 94 | 43 | 16 | 88 | 87 | 7.3 ± 0.7 | 251 |
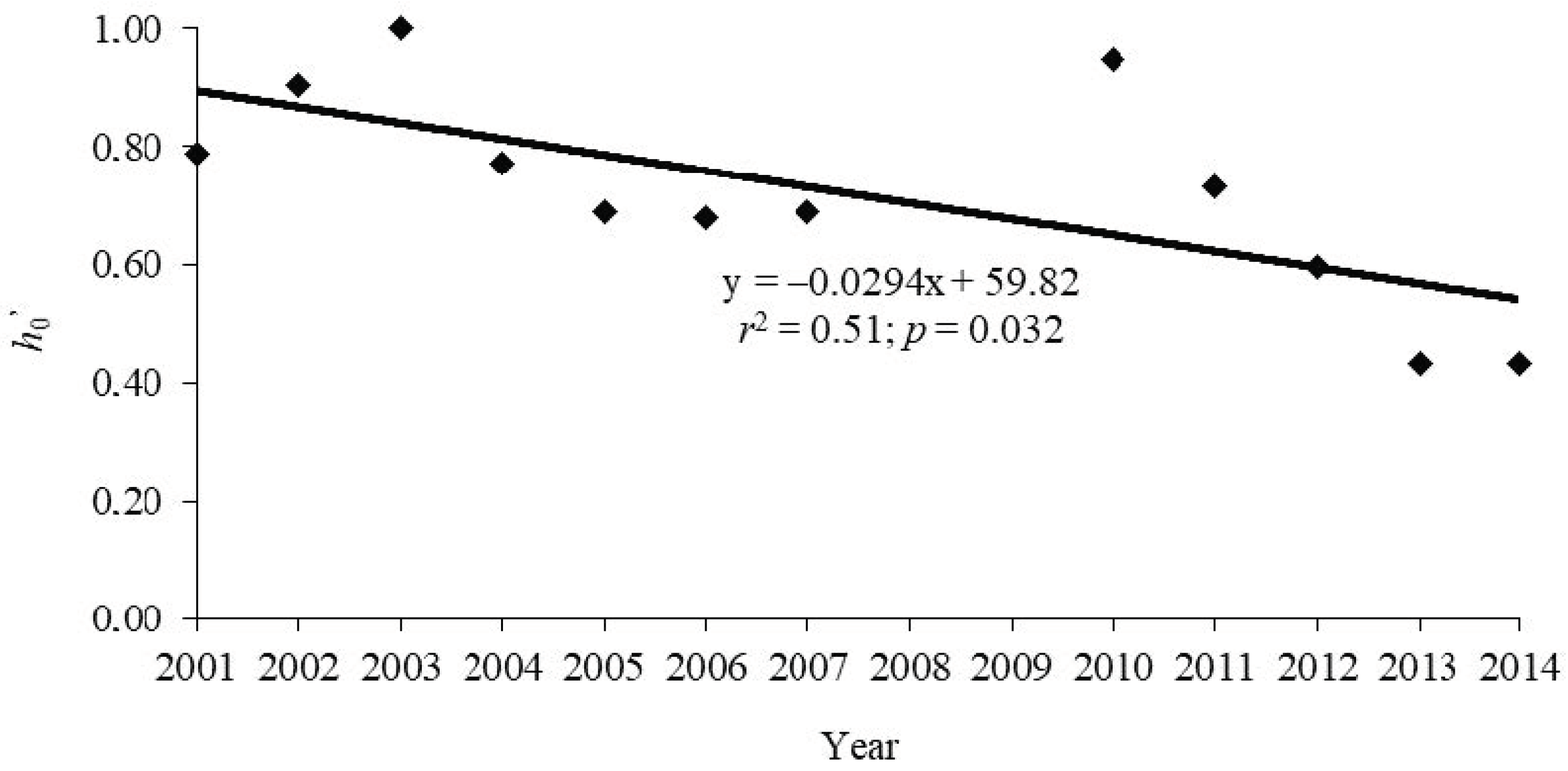
| Year | h0’ |
|---|---|
| 2001 | 0.79 |
| 2002 | 0.91 |
| 2003 | 1.00 |
| 2004 | 0.77 |
| 2005 | 0.69 |
| 2006 | 0.68 |
| 2007 | 0.69 |
| 2010 | 0.95 |
| 2011 | 0.78 |
| 2012 | 0.60 |
| 2013 | 0.43 |
| 2014 | 0.57 |
| Grand Total | 0.66 |
| Isolate Number Tested for | ||||
|---|---|---|---|---|
| Year | Potato Breeding Lines/Cultivars | Mating Type | Metalaxyl Resistance | Virulence |
| 2001 | Breeding lines (359, 386, 476, 477, 569, 1370-94, 1572-98, 458-98, 522-98, 93-BXY-1)/Cultivars (Ando, Anti, Ants, Danva, Folva, Impala, Kuras, Latona, Oleva Sarme, Sava, Van Gogh) | 25 | 31 | 35 |
| 2002 | Breeding lines (391-93, 405-98, 92-BVU-2, 93-BXL-11, R437-98, R989-93, R992-95)/Cultivars (Ando, Anti, Ants, Asterix, Danva, Kuras, Maret, Ofelia, Oleva, Piret, Sante, Sarme) | 13 | 41 | 41 |
| 2003 | Cultivars (Ants, Berber, Bintje, Folva, Latona, Oleva, Sarme, Van Gogh) | 9 | 14 | 14 |
| 2004 | Cultivars (Bintje, Fresco, Impala, Latona, Milva, Piret, Platina, Remarka, Agrie dzeltenie/Varajane kollane, Victora) | 18 | 18 | 19 |
| 2005 | Cultivars (Alpha, Anti, Ants, Evita, Juku, Oleva, Picasso, Piret, Raja, Sarme) | 17 | 17 | 18 |
| 2006 | Cultivars (Ando, Anti, Ants, Asterix, Berber, Granola, Juku, Maret, Princess, Sante, Sarme, Satina, Sinora, Van Gogh) | 30 | 29 | 30 |
| 2007 | Cultivars (Ando, Anti, Ants, Fontane, Juku, Latona, Maret, Secura, Agrie dzeltenie/Varajane kollane) | 21 | 21 | 23 |
| 2010 | Breeding lines (R1067-05, R3456-06; R458-07)/Cultivar (Asterix) | 11 | 8 | 11 |
| 2011 | Breeding lines (R1003-05, R3456-06)/Cultivar (Anti) | 21 | 16 | 21 |
| 2012 | Cultivars (Anti, Certo, Evolution, Agrie dzeltenie/Jõgeva Kollane, Sarme) | 16 | 14 | 15 |
| 2013 | Cultivars (Ambition, Arielle, Birgit, Evolution, Milva, Rosella) | 12 | 12 | 12 |
| 2014 | Breeding line (127-12)/Cultivars (Arielle, Flavia, Red Lady, Solist) | 14 | 14 | 12 |
| Total | 207 | 235 | 251 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Runno-Paurson, E.; Nassar, H.; Tähtjärv, T.; Eremeev, V.; Hansen, M.; Niinemets, Ü. High Temporal Variability in Late Blight Pathogen Diversity, Virulence, and Fungicide Resistance in Potato Breeding Fields: Results from a Long-Term Monitoring Study. Plants 2022, 11, 2426. https://doi.org/10.3390/plants11182426
Runno-Paurson E, Nassar H, Tähtjärv T, Eremeev V, Hansen M, Niinemets Ü. High Temporal Variability in Late Blight Pathogen Diversity, Virulence, and Fungicide Resistance in Potato Breeding Fields: Results from a Long-Term Monitoring Study. Plants. 2022; 11(18):2426. https://doi.org/10.3390/plants11182426
Chicago/Turabian StyleRunno-Paurson, Eve, Helina Nassar, Terje Tähtjärv, Viacheslav Eremeev, Merili Hansen, and Ülo Niinemets. 2022. "High Temporal Variability in Late Blight Pathogen Diversity, Virulence, and Fungicide Resistance in Potato Breeding Fields: Results from a Long-Term Monitoring Study" Plants 11, no. 18: 2426. https://doi.org/10.3390/plants11182426
APA StyleRunno-Paurson, E., Nassar, H., Tähtjärv, T., Eremeev, V., Hansen, M., & Niinemets, Ü. (2022). High Temporal Variability in Late Blight Pathogen Diversity, Virulence, and Fungicide Resistance in Potato Breeding Fields: Results from a Long-Term Monitoring Study. Plants, 11(18), 2426. https://doi.org/10.3390/plants11182426









