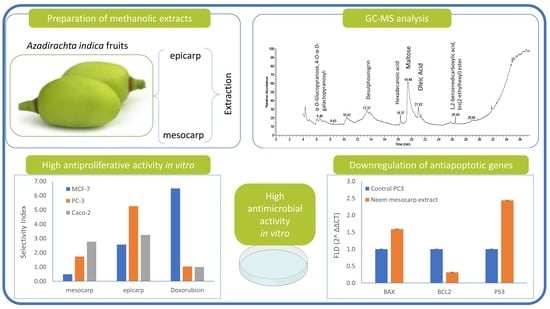
Azadirachta indica A. Juss Fruit Mesocarp and Epicarp Extracts Induce Antimicrobial and Antiproliferative Effects against Prostate (PC-3), Breast (MCF-7), and Colorectal Adenocarcinoma (Caco-2) Cancer Cell Lines through Upregulation of Proapoptotic Genes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Neem Fruit Extracts
2.2. Gas Chromatography–Mass Spectrometry (GC–MS) Analysis of Neem Extract
2.3. Antifungal Activity of Neem Extracts
2.4. Antibacterial Activity of Neem Extracts
2.5. Cytotoxicity Test
2.5.1. Cell Cultures
2.5.2. MTT Assay
2.6. Cell-Cycle Arrest Assessment
2.7. Flow Cytometry Assessment of Apoptotic vs. Necrotic Cells
2.8. Assessment of Apoptosis-Related Gene Expression
2.9. Statistical Analysis
3. Results
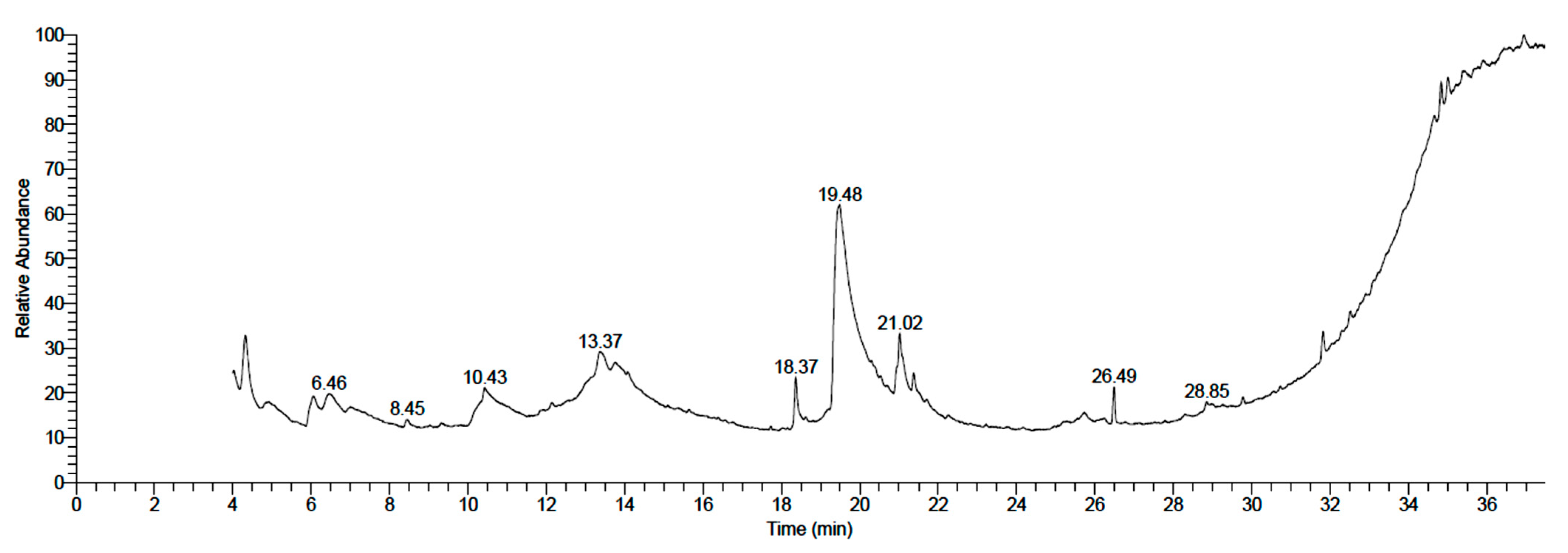
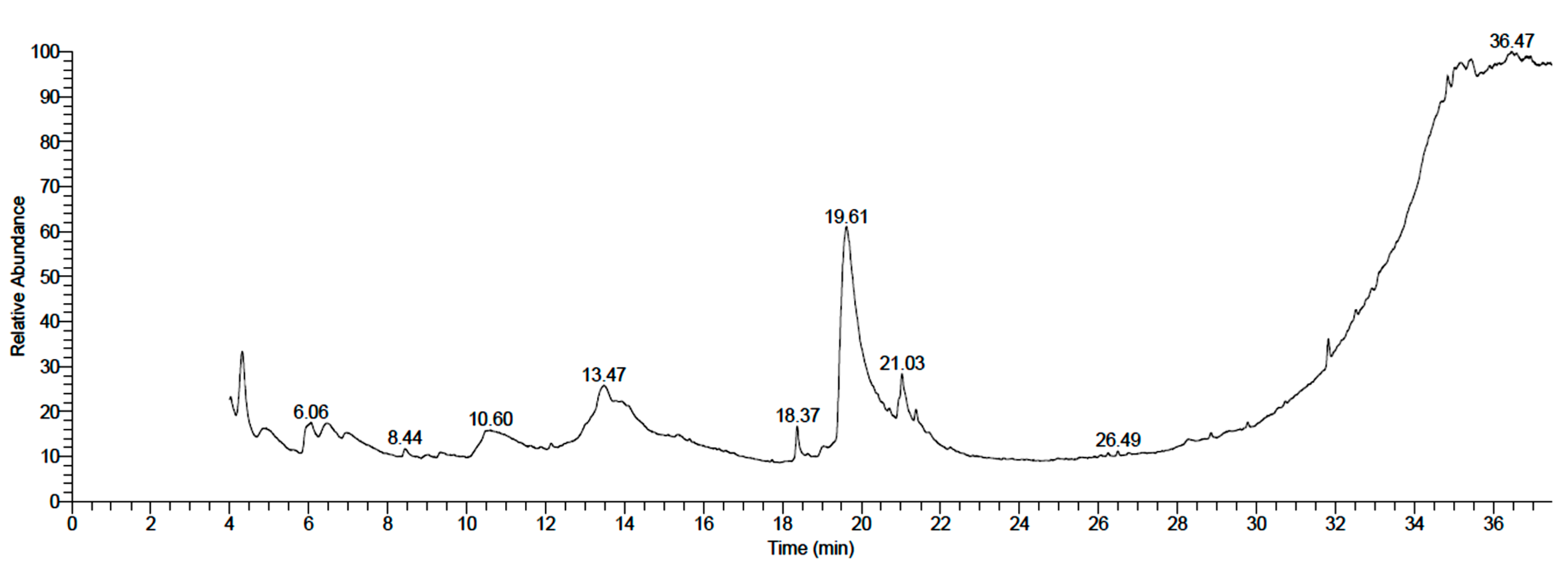
3.1. Chemical Composition of Neem Fruit Mesocarp Extract
3.2. Antimicrobial Activity of Neem Extracts
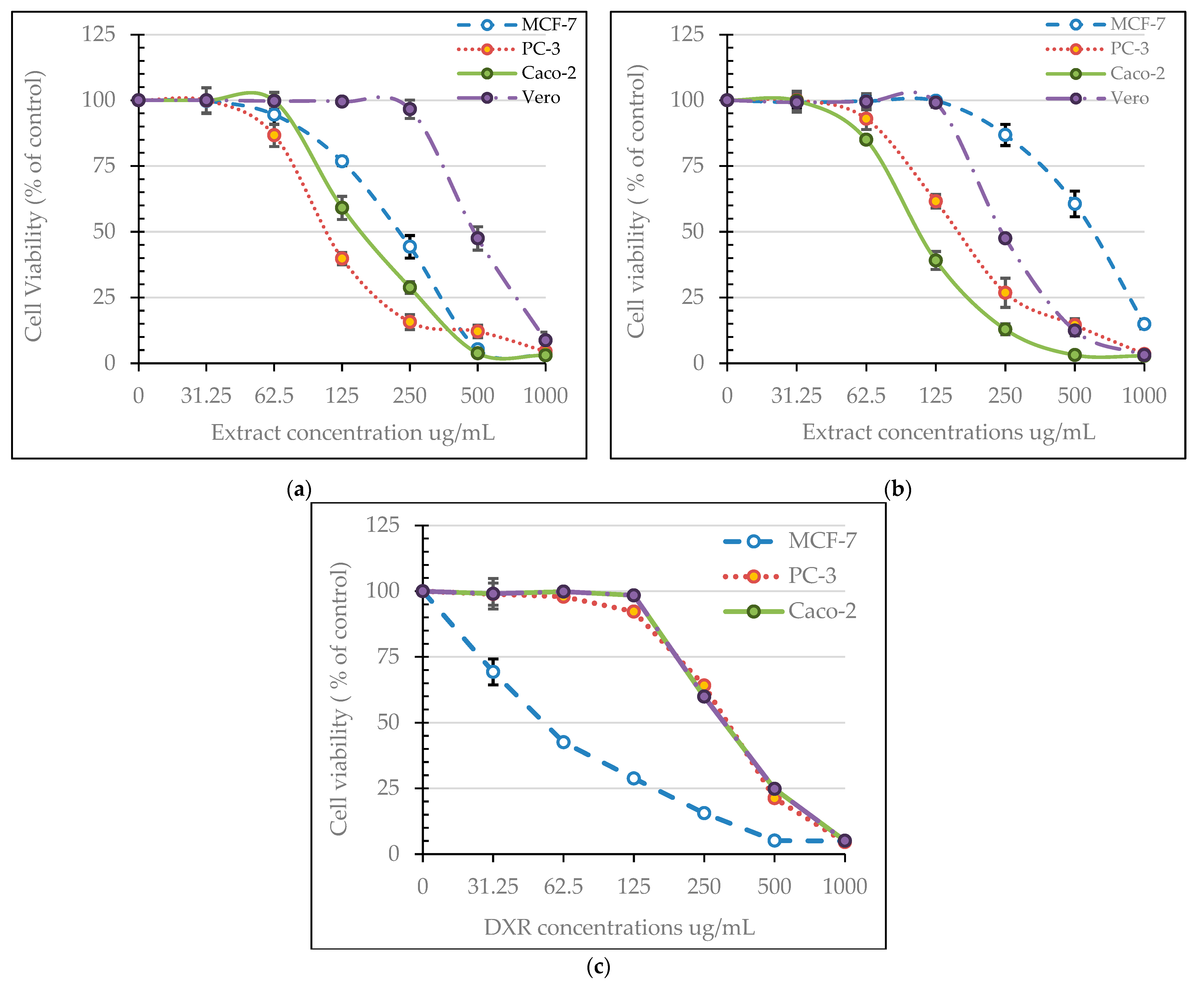
3.3. Cytotoxic Activity of Neem Extracts
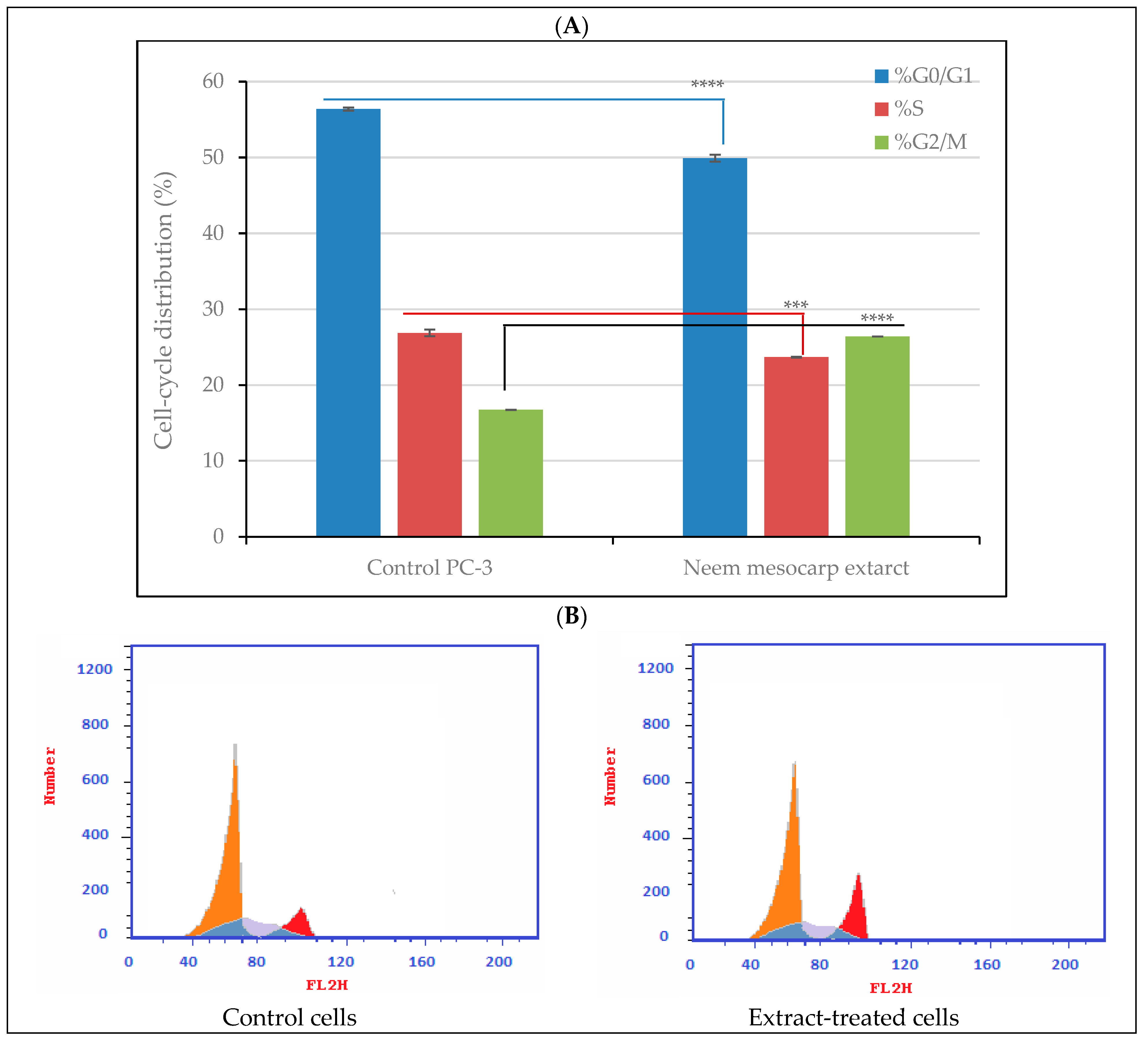
3.4. Effect of Neem Extract on Cell-Cycle Arrest in PC-3 Cells
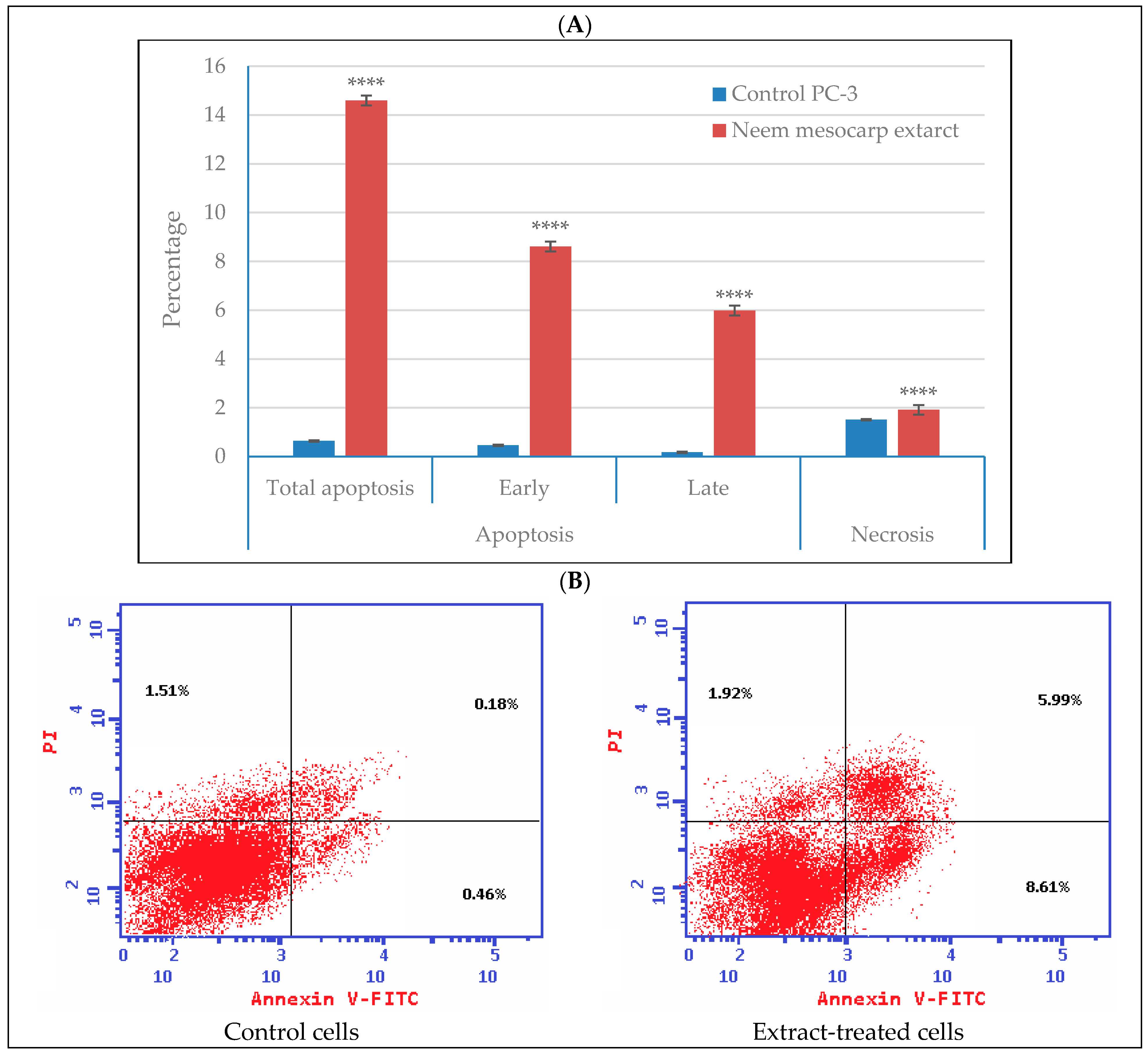
3.5. Effect of Neem Extract on Apoptosis and Necrosis of Cells
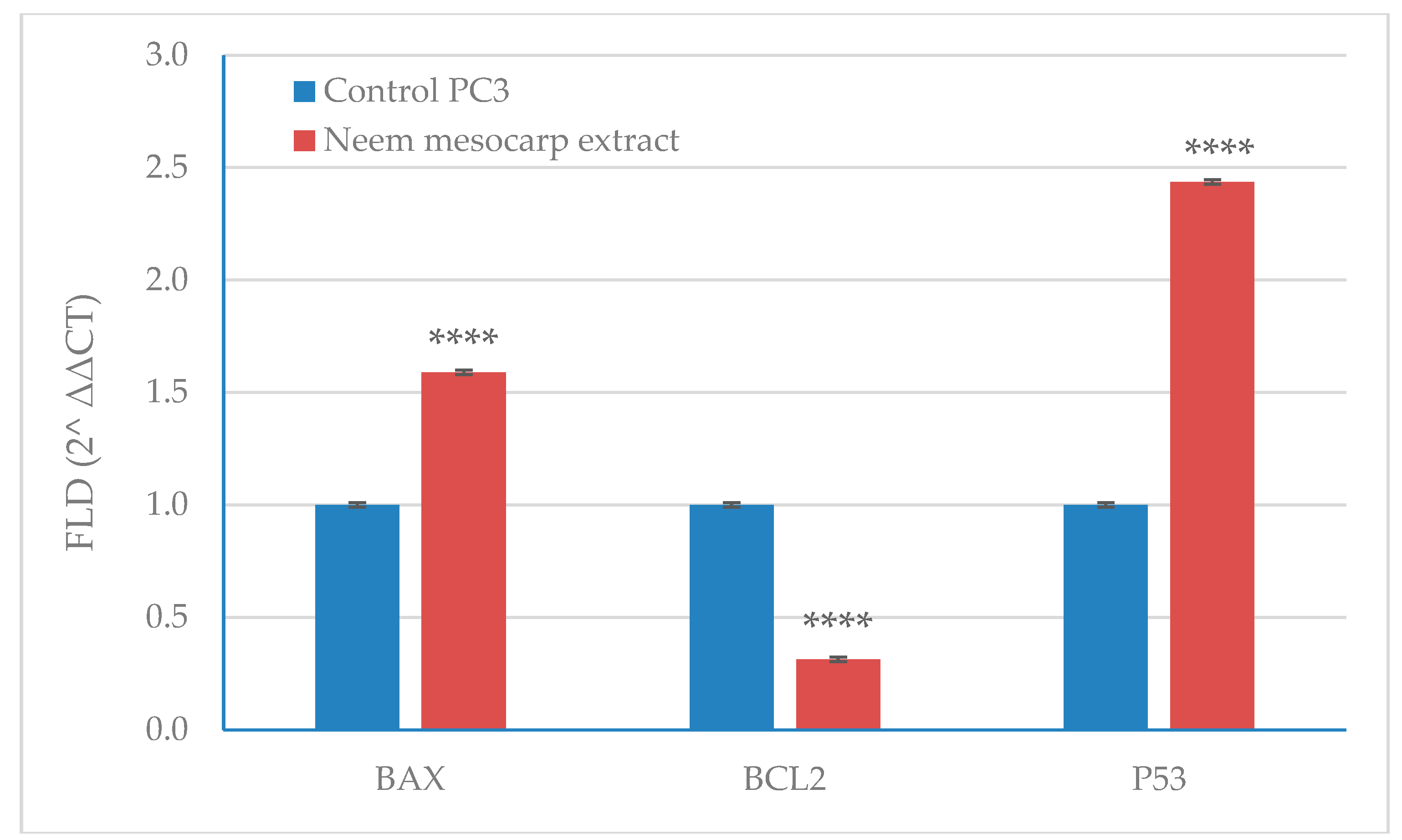
3.6. Effect of Neem Extract on the Expression of Apoptosis-Related Genes in PC-3 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anand, P.; Kunnumakara, A.B.; Sundaram, C.; Harikumar, K.B.; Tharakan, S.T.; Lai, O.S.; Sung, B.; Aggarwal, B.B. Cancer is a preventable disease that requires major lifestyle changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, B.; Akram, M.; Balasubramanian, I.; Tam, K.K.Y.; Koh, K.X.; Yee, M.Q.; Soong, R. Pharmacologic synergy between dual phosphoinositide-3-kinase and mammalian target of rapamycin inhibition and 5-fluorouracil in PIK3CA mutant gastric cancer cells. Cancer Biol. Ther. 2012, 13, 34–42. [Google Scholar] [CrossRef] [Green Version]
- Abdul-Hafeez, E.Y.; Orabi, M.A.A.; Ibrahim, O.H.M.; Ilinskaya, O.; Karamova, N.S. In vitro cytotoxic activity of certain succulent plants against human colon, breast and liver cancer cell lines. S. Afr. J. Bot. 2020, 131, 295–301. [Google Scholar] [CrossRef]
- Castro-Puyana, M.; Pérez-Sánchez, A.; Valdés, A.; Ibrahim, O.H.M.; Suarez-Álvarez, S.; Ferragut, J.A.; Micol, V.; Cifuentes, A.; Ibáñez, E.; García-Cañas, V. Pressurized liquid extraction of Neochloris oleoabundans for the recovery of bioactive carotenoids with anti-proliferative activity against human colon cancer cells. Food Res. Int. 2017, 99, 1048–1055. [Google Scholar] [CrossRef] [Green Version]
- Kumar, V.S.; Navaratnam, V. Neem (Azadirachta indica): Prehistory to contemporary medicinal uses to humankind. Asian Pac. J. Trop. Biomed. 2013, 3, 505–514. [Google Scholar] [CrossRef] [Green Version]
- Chopra, R.N.; Nayar, S.L.; Chopra, I.C. Glossary of Indian Medical Plant with Active Principles; NISC CSIR: New Delhi, India, 1999. [Google Scholar]
- Kurimoto, S.-I.; Takaishi, Y.; Ahmed, F.A.; Kashiwada, Y. Triterpenoids from the fruits of Azadirachta indica (Meliaceae). Fitoterapia 2014, 92, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Trease, G.E.; Evans, W.C. Textbook of Pharmacognosy; Elsevier India Private Limited: New Delhi, India, 2009. [Google Scholar]
- Rahmani, A.; Almatroudi, A.; Alrumaihi, F.; Khan, A. Pharmacological and therapeutic potential of neem (Azadirachta indica). Pharmacogn. Rev. 2018, 12, 250–255. [Google Scholar] [CrossRef]
- Islas, J.F.; Acosta, E.; G-Buentello, Z.; Delgado-Gallegos, J.L.; Moreno-Treviño, M.G.; Escalante, B.; Moreno-Cuevas, J.E. An overview of Neem (Azadirachta indica) and its potential impact on health. J. Funct. Foods 2020, 74, 104171. [Google Scholar] [CrossRef]
- Guchhait, K.C.; Manna, T.; Barai, M.; Karmakar, M.; Nandi, S.K.; Jana, D.; Dey, A.; Panda, S.; Raul, P.; Patra, A. Antibiofilm and anticancer activities of unripe and ripe Azadirachta indica (neem) seed extracts. BMC Complementary Med. Ther. 2022, 22, 42. [Google Scholar] [CrossRef] [PubMed]
- Atta, N.M.; Ismaiel, G.H.; Hashish, A.E.M.S.; Mohamed, E.S. Physical and chemical characteristics of neem oils extracted from seed, whole fruit and flesh. Egypt. J. Agric. Res. 2015, 93, 887–899. [Google Scholar] [CrossRef]
- Ilango, K.; Maharajan, G.; Narasimhan, S. Anti-nociceptive and anti-inflammatory activities of Azadirachta indica fruit skin extract and its isolated constituent azadiradione. Nat. Prod. Res. 2013, 27, 1463–1467. [Google Scholar] [CrossRef] [PubMed]
- Dhakal, S.; Aryal, P.; Aryal, S.; Bashyal, D.; Khadka, D. Phytochemical and antioxidant studies of methanol and chloroform extract from leaves of Azadirachta indica A. Juss. in Tropical region of Nepal. J. Pharmacogn. Phytother. 2016, 8, 203–208. [Google Scholar] [CrossRef] [Green Version]
- Koul, A.; Mohan, V.; Bharati, S. Azadirachta indica mitigates DMBA-induced hepatotoxicity: A biochemical and radiometric study. Indian J. Biochem. Biophys. 2014, 51, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Sunarwidhi, A.L.; Sudarsono, S.; Nugroho, A.E. Hypoglycemic effect of combination of Azadirachta indica A. Juss. and Gynura procumbens (Lour.) Merr. ethanolic extracts standardized by rutin and quercetin in alloxan-induced hyperglycemic rats. Adv. Pharm. Bull. 2014, 4, 613. [Google Scholar] [CrossRef] [PubMed]
- Sithisarn, P.; Supabphol, R.; Gritsanapan, W. Antioxidant activity of Siamese neem tree (VP1209). J. Ethnopharmacol. 2005, 99, 109–112. [Google Scholar] [CrossRef]
- Usha, S.; Mohammed, I.; Mahendra, K. Antioxidant activity and total flavonoids content of different parts of Azadirachta indica A. Juss. J. Med. Plants Res. 2012, 6, 5737–5742. [Google Scholar] [CrossRef]
- Manikandan, P.; Letchoumy, P.V.; Gopalakrishnan, M.; Nagini, S. Evaluation of Azadirachta indica leaf fractions for in vitro antioxidant potential and in vivo modulation of biomarkers of chemoprevention in the hamster buccal pouch carcinogenesis model. Food Chem. Toxicol. 2008, 46, 2332–2343. [Google Scholar] [CrossRef] [PubMed]
- Jóźwiak, M.; Filipowska, A.; Fiorino, F.; Struga, M. Anticancer activities of fatty acids and their heterocyclic derivatives. Eur. J. Pharmacol. 2020, 871, 172937. [Google Scholar] [CrossRef]
- Bharath, B.; Perinbam, K.; Devanesan, S.; AlSalhi, M.S.; Saravanan, M. Evaluation of the anticancer potential of Hexadecanoic acid from brown algae Turbinaria ornata on HT–29 colon cancer cells. J. Mol. Struct. 2021, 1235, 130229. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, W.; He, Q.; Wu, Y.; Lu, Z.; Sun, J.; Liu, Z.; Shao, Y.; Wang, A. Oleic acid induces apoptosis and autophagy in the treatment of tongue squamous cell carcinomas. Sci. Rep. 2017, 7, 11277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, X.; Zhao, M.; Liu, F.; Zeng, S.; Hu, J. Identification of 2, 3-dihydro-3, 5-dihydroxy-6-methyl-4H-pyran-4-one as a strong antioxidant in glucose–histidine Maillard reaction products. Food Res. Int. 2013, 51, 397–403. [Google Scholar] [CrossRef]
- Ban, J.O.; Hwang, I.G.; Kim, T.M.; Hwang, B.Y.; Lee, U.S.; Jeong, H.-S.; Yoon, Y.W.; Kim, D.J.; Hong, J.T. Anti-proliferate and pro-apoptotic effects of 2, 3-dihydro-3, 5-dihydroxy-6-methyl-4H-pyranone through inactivation of NF-κB in human colon cancer cells. Arch. Pharmacal Res. 2007, 30, 1455–1463. [Google Scholar] [CrossRef] [PubMed]
- Brulez, W.; Zeller, W. Seasonal changes of epiphytic Erwinia amylovora on ornamentals in relation to weather conditions and course of infections. Acta Hortic. 1981, 117, 37–43. [Google Scholar] [CrossRef]
- Bahuguna, A.; Khan, I.; Bajpai, V.K.; Kang, S.C. MTT assay to evaluate the cytotoxic potential of a drug. Bangladesh J. Pharmacol. 2017, 12, 115–118. [Google Scholar] [CrossRef]
- Alqahtani, S.A.; Nasr, F.A.; Noman, O.M.; Farooq, M.; Alhawassi, T.; Qamar, W.; El-Gamal, A. Cytotoxic Evaluation and Anti-Angiogenic Effects of Two Furano-Sesquiterpenoids from Commiphora myrrh Resin. Molecules 2020, 25, 1318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nasr, F.A.; Noman, O.M.; Alqahtani, A.S.; Qamar, W.; Ahamad, S.R.; Al-Mishari, A.A.; Alyhya, N.; Farooq, M. Phytochemical constituents and anticancer activities of Tarchonanthus camphoratus essential oils grown in Saudi Arabia. Saudi Pharm. J. 2020, 28, 1474–1480. [Google Scholar] [CrossRef]
- Krishnaveni, M. Docking, simulation studies of desulphosinigrin–cyclin dependent kinase 2, an anticancer drug target. Int. J. Pharm. Sci. Rev. Res. 2015, 30, 115–118. [Google Scholar]
- Kavitha, S.; Lincy, M.L.P.; Kala, S.M.J.; Mohan, V.R.; Maruthupandian, A. GC-MS analysis of ethanol extract of stem of Nothapodytes nimmoniana (Graham) Mabb. World J. Pharm. Sci. 2015, 3, 1145–1150. [Google Scholar]
- Thakur, R.S.; Ahirwar, B. A steroidal derivative from Trigonella foenum graecum L. that induces apoptosis in vitro and in vivo. J. Food Drug Anal. 2019, 27, 231–239. [Google Scholar] [CrossRef] [Green Version]
- Duke, J.; Mj, B. Dr. Duke’s Phytochemical and Ethnobotanical Databases; USDA, Agricultural Research Service: Washington, DC, USA, 1994.
- Harada, H.; Yamashita, U.; Kurihara, H.; Fukushi, E.; Kawabata, J.; Kamei, Y. Antitumor activity of palmitic acid found as a selective cytotoxic substance in a marine red alga. Anticancer. Res. 2002, 22, 2587–2590. [Google Scholar]
- Moni, S.S.; Alam, M.F.; Sultan, M.H.; Makeen, H.A.; Alhazmi, H.A.; Mohan, S.; Alam, M.S.; Rehman, Z.U.; Jabeen, A.; Sanobar, S. Spectral analysis, in vitro cytotoxicity and antibacterial studies of bioactive principles from the leaves of Conocarpus lancifolius, a common tree of Jazan, Saudi Arabia. Braz. J. Biol. 2021, 83, e244479. [Google Scholar] [CrossRef] [PubMed]
- Ezhilan, B.P.; Neelamegam, R. GC-MS analysis of phytocomponents in the ethanol extract of Polygonum chinense L. Pharmacogn. Res. 2012, 4, 11–14. [Google Scholar] [CrossRef] [Green Version]
- Arora, S.; Kumar, G. Phytochemical screening of root, stem and leaves of Cenchrus biflorus Roxb. J. Pharmacogn. Phytochem. 2018, 7, 1445–1450. [Google Scholar]
- Geetha, D.H.; Rajeswari, M.; Jayashree, I. Chemical profiling of Elaeocarpus serratus L. by GC-MS. Asian Pac. J. Trop. Biomed. 2013, 3, 985–987. [Google Scholar] [CrossRef] [Green Version]
- Bremer, E.; Van Dam, G.; Kroesen, B.J.; de Leij, L.; Helfrich, W. Targeted induction of apoptosis for cancer therapy: Current progress and prospects. Trends Mol. Med. 2006, 12, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, C.M.; Singh, A.T.K. Apoptosis: A target for anticancer therapy. Int. J. Mol. Sci. 2018, 19, 448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Natarajan, V.; Venugopal, P.V.; Menon, T. Effect of Azadirachta indica (neem) on the growth pattern of dermatophytes. Indian J. Med. Microbiol. 2003, 21, 98–101. [Google Scholar] [CrossRef]


| Gene | Primer |
|---|---|
| BAX | F: 5’-ATGGACGGGTCCGGGGAG-3’ |
| R: 5’-ATCCAGCCCAACAGCCGC-3’ | |
| BCL2 | F: 5’-AAG CCG GCG ACGACT TCT-3’ |
| R: 5’-GGT GCC GGT TCA GGTACT CA-3’ | |
| p53 | F: 5’-ATGTTTTGCCAACTGGCCAAG -3’ |
| R: 5’-TGAGCAGCGCTCATGGTG-3’ |
| No. | Compound | RT | Area % | Formula | MW | CAS Number |
|---|---|---|---|---|---|---|
| 1 | Octadecanoic acid, 2,3-dihydroxypropyl ester | 4.32 | 9.51 | C21H42O4 | 358 | 123-94-4 |
| 2 | α-D-Glucopyranose, 4-O-α-D-galactopyranosyl- | 6.04 | 5.70 | C12H22O11 | 342 | 5965-66-2 |
| 3 | Desulfosinigrin | 13.36 | 5.98 | C10H17NO6S | 279 | 5115-81-1 |
| 4 | Hexadecanoic acid | 18.36 | 5.84 | C16H32O2 | 256 | 57-10-3 |
| 5 | Maltose | 19.43 | 45.19 | C12H22O11 | 342 | 69-79-4 |
| 6 | 9,12-Octadecadienoic acid (Z,Z)- | 20.94 | 1.83 | C18H32O2 | 280 | 60-33-3 |
| 7 | Oleic Acid | 21.02 | 10.06 | C18H34O2 | 282 | 112-80-1 |
| 8 | Hexadecanoic acid, 2,3-dihydroxypropyl ester | 21.38 | 3.18 | C19H38O4 | 330 | 542-44-9 |
| 9 | 1,2-benzenedicarboxylic acid, bis(2-ethylhexyl) ester | 26.49 | 3.74 | C24H38O4 | 390 | 117-81-7 |
| 10 | 9,12,15-octadecatrienoic acid, 2,3-bis[(trimethylsilyl)oxy]propyl ester, (Z,Z,Z)- | 31.82 | 2.90 | C27H52O4Si2 | 496 | 55521-22-7 |
| 11 | Ethyl iso-allocholate | 34.84 | 6.08 | C26H44O5 | 436 | NA |
| No. | Compound | RT | Area % | Formula | MW | CAS Number |
|---|---|---|---|---|---|---|
| 1 | 4h-pyran-4-one, 2,3-dihydro-3,5-dihydroxy-6-methyl- | 4.32 | 8.07 | C6H8O4 | 144 | 28564-83-2 |
| 2 | 2-amino-5-guanidino-pentanoic acid | 4.83 | 0.82 | C6H14N4O2 | 174 | 74-79-3 |
| 3 | 3,4,5,6tetrahydroxy-2-oxohexanoic Acid | 4.91 | 1.88 | C6H10O7 | 194 | NA |
| 4 | Octadecanoic acid, 3-hydroxy-, methyl ester | 5.94 | 4.80 | C19H38O3 | 314 | 2420-36-2 |
| 5 | α-d-glucopyranose, 4-o-α-d-galactopyranosyl- | 6.47 | 2.61 | C12H22O11 | 342 | 5965-66-2 |
| 6 | L-gala-l-ido-octonic lactone | 8.44 | 0.88 | C8H14O8 | 238 | NA |
| 7 | Digitoxin | 10.48 | 2.26 | C41H64O13 | 764 | 71-63-6 |
| 8 | Desulfosinigrin | 13.45 | 6.04 | C10H17NO6S | 279 | 5115-81-1 |
| 9 | Hexadecanoic acid | 18.37 | 2.65 | C16H32O2 | 256 | 57-10-3 |
| 10 | d-Manno-l-gluco-octonic acid | 19.59 | 43.64 | C8H16O9 | 256 | NA |
| 11 | [1,1’-bicyclopropyl]-2-octanoic acid, 2’-hexyl-, methyl ester | 20.95 | 1.09 | C21H38O2 | 322 | 56687-68-4 |
| 12 | Oleic acid | 21.03 | 5.86 | C18H34O2 | 282 | 112-80-1 |
| 13 | Ethyl iso-allocholate | 29.78 | 7.23 | C26H44O5 | 436 | NA |
| 14 | 9,12,15-octadecatrienoic acid, 2,3-bis[(trimethylsilyl)oxy]propyl ester, (Z,Z,Z)- | 32.91 | 0.64 | C27H52O4Si2 | 496 | 55521-22-7 |
| 15 | Docosanoic acid, 1,2,3-propanetriyl ester | 33.10 | 7.13 | C69H134O6 | 1058 | 18641-57-1 |
| 16 | Oleic acid, eicosyl ester | 34.29 | 0.32 | C38H74O2 | 562 | 22393-88-0 |
| 17 | 9-octadecenoic acid, 1,2,3-propanetriyl ester, (E,E,E)- | 34.66 | 2.35 | C57H104O6 | 884 | 537-39-3 |
| 18 | Psi.,.psi.-carotene, 1,2-dihydro-1-hydroxy- | 36.37 | 1.71 | C40H58O | 554 | 105-92-0 |
| Extract | Concentrations (µg/mL) | Inhibition Zone (mm) | Growth Inhibition (%) | ||||
|---|---|---|---|---|---|---|---|
| Seratia | Acinetobacter | Agrobacterium | Seratia | Acinetobacter | Agrobacterium | ||
| epicarp | 15.6 | 6.0 h | 6.0 h | 6.0 h | 0.00 | 0.00 | 0.00 |
| 31.3 | 6.0 h | 6.0 h | 6.0 h | 0.00 | 0.00 | 0.00 | |
| 62.5 | 6.7 gh | 7.0 gh | 7.7 g | 11.11 | 16.67 | 27.78 | |
| 125 | 7.7 fg | 8.3 fg | 8.0 g | 27.78 | 38.89 | 33.33 | |
| 250 | 12.7 de | 13.7 de | 13.7 de | 111.11 | 127.78 | 127.78 | |
| 500 | 15.7 c | 17.3 c | 17.0 c | 161.11 | 188.89 | 183.33 | |
| 1000 | 18.7 b | 20.3 b | 18.8 b | 211.11 | 238.89 | 216.67 | |
| mesocarp | 15.6 | 6.0 h | 6.0 h | 6.0 h | 0.00 | 0.00 | 0.00 |
| 31.3 | 6.0 h | 6.0 h | 6.0 h | 0.00 | 0.00 | 0.00 | |
| 62.5 | 7.3 gh | 7.3 gh | 7.3 gh | 22.22 | 22.22 | 22.22 | |
| 125 | 9.0 f | 9.3 f | 8.7 g | 50.00 | 55.56 | 44.44 | |
| 250 | 11.7 e | 12.0 e | 11.3 f | 94.44 | 100.00 | 88.89 | |
| 500 | 13.3 d | 14.0 d | 13.0 e | 122.22 | 133.33 | 116.67 | |
| 1000 | 16.3 c | 17.3 c | 14.8 d | 172.22 | 188.89 | 150.00 | |
| Negative control | 6.0 h | 6.00 f | 6.0 h | 0.00 | 0.00 | 0.00 | |
| Amoxicillin (62.5 ppm) | 41.3 a | 38.33 a | 37.0 a | 588.89 | 538.89 | 516.67 | |
| Extract | Concentrations (µg/mL) | Inhibition Zone (mm) | Growth Inhibition (%) | ||||
|---|---|---|---|---|---|---|---|
| Rhizoctonia | Penicillium | Fusarium | Rhizoctonia | Penicillium | Fusarium | ||
| epicarp | 15.6 | 5.8 e | 8.5 b | 6.6 bc | 35.19 | 5.93 | 26.67 |
| 31.3 | 5.8 e | 8.3 bc | 6.6 b | 35.56 | 7.78 | 26.30 | |
| 62.5 | 5.5 f | 8.0 bcd | 6.4 cd | 38.52 | 10.74 | 28.89 | |
| 125 | 5.4 fg | 7.5 ef | 6.3 de | 39.63 | 17.04 | 30.37 | |
| 250 | 5.1 h | 7.5 ef | 6.0 f | 43.70 | 16.67 | 32.96 | |
| 500 | 4.4 i | 7.0 f | 5.8 gh | 50.74 | 21.85 | 35.93 | |
| 1000 | 4.2 j | 6.4 g | 5.0 ij | 53.70 | 28.89 | 44.07 | |
| mesocarp | 15.6 | 6.7 b | 7.9 cde | 6.6 b | 25.56 | 12.59 | 26.30 |
| 31.3 | 6.6 bc | 7.7 de | 6.1 ef | 27.04 | 14.44 | 31.85 | |
| 62.5 | 6.4 c | 7.5 ef | 5.9 fg | 29.26 | 16.67 | 33.70 | |
| 125 | 6.1 d | 6.2 gh | 5.7 h | 31.85 | 31.48 | 36.67 | |
| 250 | 6.0 de | 5.9 h | 5.2 i | 33.33 | 34.07 | 41.85 | |
| 500 | 5.5 f | 5.4 i | 5.1 ij | 38.89 | 39.63 | 42.96 | |
| 1000 | 5.3 gh | 4.2 j | 4.9 j | 41.85 | 54.07 | 45.19 | |
| Negative control | 9.0 a | 9.0 a | 9.0 a | 0.00 | 0.00 | 0.00 | |
| Hymexazol (1000 ppm) | 1.8 k | 2.9 k | 1.6 k | 79.63 | 68.15 | 81.85 | |
| Neem Methanolic Extract | IC50 (µg/mL) | Selectivity Index | |||||
|---|---|---|---|---|---|---|---|
| Vero | MCF-7 | PC-3 | Caco-2 | MCF-7 | PC-3 | Caco-2 | |
| Fruit epicarp | 305.40 | 629.23 | 176.31 | 110.16 | 0.49 | 1.73 | 2.77 |
| Fruit mesocarp | 588.30 | 228.86 | 111.76 | 180.77 | 2.57 | 5.26 | 3.25 |
| Doxorubicin | 35.09 | 5.40 | 34.11 | 35.09 | 6.50 | 1.03 | 1.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibrahim, O.H.M.; Mousa, M.A.A.; Asiry, K.A.; Alhakamy, N.A.; Abo-Elyousr, K.A.M. Azadirachta indica A. Juss Fruit Mesocarp and Epicarp Extracts Induce Antimicrobial and Antiproliferative Effects against Prostate (PC-3), Breast (MCF-7), and Colorectal Adenocarcinoma (Caco-2) Cancer Cell Lines through Upregulation of Proapoptotic Genes. Plants 2022, 11, 1990. https://doi.org/10.3390/plants11151990
Ibrahim OHM, Mousa MAA, Asiry KA, Alhakamy NA, Abo-Elyousr KAM. Azadirachta indica A. Juss Fruit Mesocarp and Epicarp Extracts Induce Antimicrobial and Antiproliferative Effects against Prostate (PC-3), Breast (MCF-7), and Colorectal Adenocarcinoma (Caco-2) Cancer Cell Lines through Upregulation of Proapoptotic Genes. Plants. 2022; 11(15):1990. https://doi.org/10.3390/plants11151990
Chicago/Turabian StyleIbrahim, Omer H. M., Magdi A. A. Mousa, Khalid A. Asiry, Nabil A. Alhakamy, and Kamal A. M. Abo-Elyousr. 2022. "Azadirachta indica A. Juss Fruit Mesocarp and Epicarp Extracts Induce Antimicrobial and Antiproliferative Effects against Prostate (PC-3), Breast (MCF-7), and Colorectal Adenocarcinoma (Caco-2) Cancer Cell Lines through Upregulation of Proapoptotic Genes" Plants 11, no. 15: 1990. https://doi.org/10.3390/plants11151990
APA StyleIbrahim, O. H. M., Mousa, M. A. A., Asiry, K. A., Alhakamy, N. A., & Abo-Elyousr, K. A. M. (2022). Azadirachta indica A. Juss Fruit Mesocarp and Epicarp Extracts Induce Antimicrobial and Antiproliferative Effects against Prostate (PC-3), Breast (MCF-7), and Colorectal Adenocarcinoma (Caco-2) Cancer Cell Lines through Upregulation of Proapoptotic Genes. Plants, 11(15), 1990. https://doi.org/10.3390/plants11151990