Development of Microsatellite Markers for Tanacetum cinerariifolium (Trevis.) Sch. Bip., a Plant with a Large and Highly Repetitive Genome
Abstract
1. Introduction
2. Results and Discussion
2.1. Contigs Assembly and Genome-Wide Identification of SSR Markers
2.2. Selection, Testing, and Characterization of SSR Markers
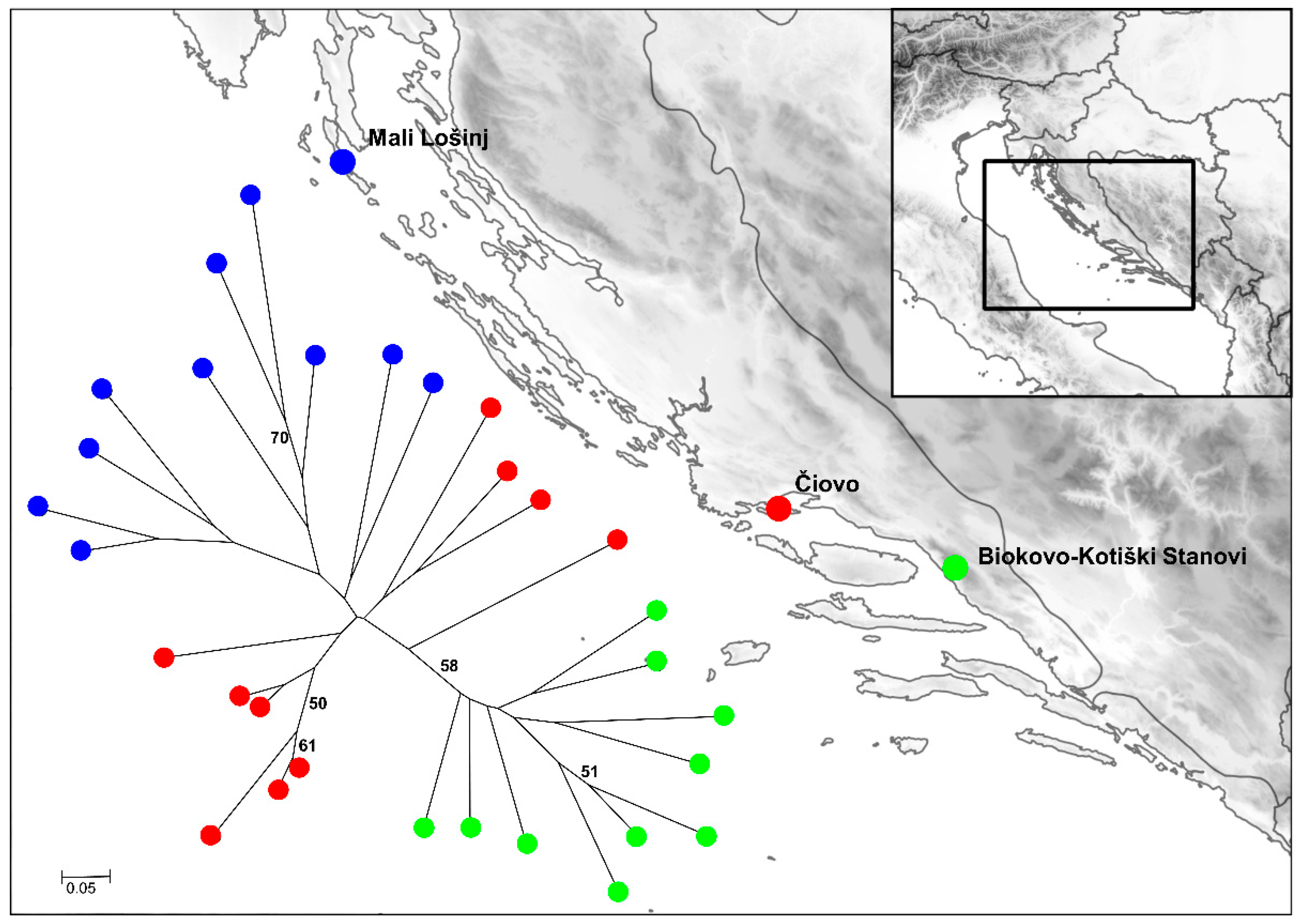
2.3. Preliminary Genetic Diversity Study of Dalmatian Pyrethrum Populations
3. Materials and Methods
3.1. Plant Material
3.2. DNA Isolation
3.3. Next Generation Sequencing, DNA Assembly and SSR Identification
3.4. Testing and Characterization of Developed SSR Markers
3.5. Preliminary Study of Dalmatian Pyrethrum Genetic Diversity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Eastwood, W.J. East Mediterranean Vegetation and Climate Change. In Balkan Biodiversity—Pattern and Process in the European Hotspot; Griffiths, H.I., Kryštufek, B., Reed, J.M., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 25–48. [Google Scholar]
- Grdiša, M.; Carović-Stanko, K.; Kolak, I.; Šatović, Z. Morphological and Biochemical Diversity of Dalmatian Pyrethrum (Tanacetum cinerariifolium (Trevir.) Sch. Bip.). Agric. Conspec. Sci. 2009, 74, 73–80. [Google Scholar]
- Bhat, B.K. Breeding Methodologies Applicable to Pyrethrum. In Pyrethrum Flowers: Production, Chemistry, Toxicology, and Uses; Casida, J.E., Quistad, G.B., Eds.; Oxford University Press: New York, NY, USA, 1995; pp. 67–94. [Google Scholar]
- Davies, T.G.E.; Field, L.M.; Usherwood, P.N.R.; Williamson, M.S. DDT, Pyrethrins, Pyrethroids and Insect Sodium Channels. IUBMB Life 2007, 59, 151–162. [Google Scholar] [CrossRef]
- Bakarić, P. Buhač—Prirodni Insekticid. Gospod. List 2005, 17, 41–45. (In Croatian) [Google Scholar]
- Hitmi, A.; Coudret, A.; Barthomeuf, C. The Production of Pyrethrins by Plant Cell and Tissue Cultures of Chrysanthemum Cinerariaefolium and Tagetes Species. Crit. Rev. Biochem. Mol. Biol. 2000, 35, 317–337. [Google Scholar] [CrossRef] [PubMed]
- Wandahwa, P.; Van Ranst, E.; Van Damme, P. Pyrethrum (Chrysanthemum cinerariaefolium Vis.) Cultivation in West Kenya: Origin, Ecological Conditions and Management. Ind. Crops Prod. 1996, 5, 307–322. [Google Scholar] [CrossRef]
- Ožanić, S. Poljoprivreda Dalmacije u Prošlosti (in Croatian); Agronomic Society NRH: Split, Croatia, 1955. [Google Scholar]
- FAO. Food and Agriculture Organization of the United Nations. Production: Crops; FAO: Rome, Italy, 2020. [Google Scholar]
- BRA. Botanical Resources Australia. Available online: https://www.botanicalresources.com/ (accessed on 14 March 2022).
- Ambrožič Dolinšek, J.; Kovač, M.; Žel, J.; Camloh, M. Pyrethrum (Tanacetum cinerariifolium) from the Northern Adriatic as a Potential Source of Natural Insecticide. Ann. Ser. Hist. Nat. 2007, 17, 39–46. [Google Scholar]
- Grdiša, M.; Babić, S.; Periša, M.; Carović-Stanko, K.; Kolak, I.; Liber, Z.; Jug-Dujaković, M.; Satovic, Z. Chemical Diversity of the Natural Populations of Dalmatian Pyrethrum (Tanacetum cinerariifolium/Trevir./Sch.Bip.) in Croatia. Chem. Biodivers 2013, 10, 460–472. [Google Scholar] [CrossRef]
- Varga, F.; Jeran, N.; Šatović, Z.; Biošić, M.; Grdiša, M. High Diversity of Natural Dalmatian Pyrethrum Based on Pyrethrin Composition at Intra- and Interpopulation Level. Phytochemistry 2021, 192, 112934. [Google Scholar] [CrossRef]
- Morris, S.E.; Davies, N.W.; Brown, P.H.; Groom, T. Effect of Drying Conditions on Pyrethrins Content. Ind. Crops Prod. 2006, 23, 9–14. [Google Scholar] [CrossRef]
- Grdiša, M.; Liber, Z.; Radosavljević, I.; Carović-Stanko, K.; Kolak, I.; Satovic, Z. Genetic Diversity and Structure of Dalmatian Pyrethrum (Tanacetum Cinerariifolium Trevir./Sch./Bip., Asteraceae) within the Balkan Refugium. PLoS ONE 2014, 9, e105265. [Google Scholar] [CrossRef]
- Mlinarec, J.; Skuhala, A.; Jurković, A.; Malenica, N.; McCann, J.; Weiss-Schneeweiss, H.; Bohanec, B.; Besendorfer, V. The Repetitive DNA Composition in the Natural Pesticide Producer Tanacetum Cinerariifolium: Interindividual Variation of Subtelomeric Tandem Repeats. Front. Plant Sci. 2019, 10, 613. [Google Scholar] [CrossRef] [PubMed]
- Ottaro, W.G.W. The Relationship between the Ploidy Level and Certain Morphological Characteristics of Chrysanthemum cinerariaefolium Vis. Pyrethrum Post 1977, 14, 10–14. [Google Scholar]
- Liu, P.L.; Wan, J.N.; Guo, Y.P.; Ge, S.; Rao, G.Y. Adaptive Evolution of the Chrysanthemyl Diphosphate Synthase Gene Involved in Irregular Monoterpene Metabolism. BMC Evol. Biol. 2012, 12, 214. [Google Scholar] [CrossRef] [PubMed]
- Rivera, S.B.; Swedlund, B.D.; King, G.J.; Bell, R.N.; Hussey, C.E.; Shattuck-Eidens, D.M.; Wrobel, W.M.; Peiser, G.D.; Poulter, C.D. Chrysanthemyl Diphosphate Synthase: Isolation of the Gene and Characterization of the Recombinant Non-Head-to-Tail Monoterpene Synthase from Chrysanthemum cinerariaefolium. Proc. Natl. Acad. Sci. USA 2001, 98, 4373–4378. [Google Scholar] [CrossRef] [PubMed]
- Sultana, S.; Hu, H.; Gao, L.; Mao, J.; Luo, J.; Jongsma, M.A.; Wang, C. Molecular Cloning and Characterization of the Trichome Specific Chrysanthemyl Diphosphate/Chrysanthemol Synthase Promoter from Tanacetum cinerariifolium. Sci. Hortic. 2015, 185, 193–199. [Google Scholar] [CrossRef]
- Tang, L.; Li, J.; Khalil, R.; Yang, Y.; Fan, J.; Liu, M.; Li, Z. Cloning and Functional Analysis of CDS_CCI2: A Tanacetum Cinerariaefolium Chrysanthemyl Diphosphate Synthase Gene. Plant Growth Regul. 2012, 67, 161–169. [Google Scholar] [CrossRef]
- Khan, S.; Upadhyay, S.; Khan, F.; Tandon, S.; Shukla, R.K.; Ghosh, S.; Gupta, V.; Banerjee, S.; Ur Rahman, L. Comparative Transcriptome Analysis Reveals Candidate Genes for the Biosynthesis of Natural Insecticide in Tanacetum cinerariifolium. BMC Genom. 2017, 18, 54. [Google Scholar] [CrossRef]
- Yamashiro, T.; Shiraishi, A.; Satake, H.; Nakayama, K. Draft Genome of Tanacetum cinerariifolium, the Natural Source of Mosquito Coil. Sci. Rep. 2019, 9, 18249. [Google Scholar] [CrossRef]
- Vieira, M.L.C.; Santini, L.; Diniz, A.L.; Munhoz, C.d.F. Microsatellite Markers: What They Mean and Why They Are so Useful. Genet. Mol. Biol. 2016, 39, 312–328. [Google Scholar] [CrossRef]
- Vaz Patto, M.C.; Satovic, Z.; Pêgo, S.; Fevereiro, P. Assessing the Genetic Diversity of Portuguese Maize Germplasm Using Microsatellite Markers. Euphytica 2004, 137, 63–72. [Google Scholar] [CrossRef]
- Belaj, A.; Muñoz-Diez, C.; Baldoni, L.; Porceddu, A.; Barranco, D.; Satovic, Z. Genetic Diversity and Population Structure of Wild Olives from the North-Western Mediterranean Assessed by SSR Markers. Ann. Bot. 2007, 100, 449–458. [Google Scholar] [CrossRef]
- Kostia, S.; Varvio, S.L.; Vakkari, P.; Pulkkinen, P. Microsatellite Sequences in a Conifer, Pinus sylvestris. Genome 1995, 38, 1244–1248. [Google Scholar] [CrossRef]
- Pfeiffer, A.; Olivieri, A.M.; Morgante, M. Identification and Characterization of Microsatellites in Norway Spruce (Picea Abies K.). Genome 1997, 40, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Garner, T.W.J. Genome Size and Microsatellites: The Effect of Nuclear Size on Amplification Potential. Genome 2002, 45, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Purwoko, D.; Cartealy, I.C.; Tajuddin, T.; Dinarti, D.; Sudarsono, S. SSR Identification and Marker Development for Sago Palm Based on NGS Genome Data. Breed. Sci. 2019, 69, 1–10. [Google Scholar] [CrossRef]
- Šmarda, P.; Knápek, O.; Březinová, A.; Horová, L.; Grulich, V.; Danihelka, J.; Veselý, P.; Šmerda, J.; Rotreklová, O.; Bureš, P. Genome Sizes and Genomic Guanine + cytosine (GC) Contents of the Czech Vascular Flora with New Estimates for 1700 Species. Preslia 2019, 91, 117–142. [Google Scholar] [CrossRef]
- Baruca Arbeiter, A.; Hladnik, M.; Jakše, J.; Bandelj, D. First Set of Microsatellite Markers for Immortelle (Helichrysum italicum (Roth) G. Don): A Step towards the Selection of the Most Promising Genotypes for Cultivation. Ind. Crops Prod. 2021, 162, 113298. [Google Scholar] [CrossRef]
- Singh, R.; Ming, R.; Yu, Q. Comparative Analysis of GC Content Variations in Plant Genomes. Trop. Plant Biol. 2016, 9, 136–149. [Google Scholar] [CrossRef]
- Cserhati, M. Motif Content Comparison between Monocot and Dicot Species. Genom. Data 2015, 3, 128. [Google Scholar] [CrossRef][Green Version]
- Peng, Y.; Lai, Z.; Lane, T.; Nageswara-Rao, M.; Okada, M.; Jasieniuk, M.; O’geen, H.; Kim, R.W.; Douglas Sammons, R.; Rieseberg, L.H.; et al. De Novo Genome Assembly of the Economically Important Weed Horseweed Using Integrated Data from Multiple Sequencing Platforms. Plant Physiol. 2014, 166, 1241–1254. [Google Scholar] [CrossRef]
- Scaglione, D.; Reyes-Chin-Wo, S.; Acquadro, A.; Froenicke, L.; Portis, E.; Beitel, C.; Tirone, M.; Mauro, R.; Lo Monaco, A.; Mauromicale, G.; et al. The Genome Sequence of the Outbreeding Globe Artichoke Constructed de Novo Incorporating a Phase-Aware Low-Pass Sequencing Strategy of F1 Progeny. Sci. Rep. 2016, 6, 19427. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, A.; Mathur, S.; Lawson, S.S.; LaBonte, N.R.; Lorch, A.; Coggeshall, M.V.; Woeste, K.E. Microsatellite Borders and Micro-Sequence Conservation in Juglans. Sci. Rep. 2019, 9, 3748. [Google Scholar] [CrossRef] [PubMed]
- Staton, M.; Best, T.; Khodwekar, S.; Owusu, S.; Xu, T.; Xu, Y.; Jennings, T.; Cronn, R.; Arumuganathan, A.K.; Coggeshall, M.; et al. Preliminary Genomic Characterization of Ten Hardwood Tree Species from Multiplexed Low Coverage Whole Genome Sequencing. PLoS ONE 2015, 10, e0145031. [Google Scholar] [CrossRef] [PubMed]
- Schoebel, C.N.; Brodbeck, S.; Buehler, D.; Cornejo, C.; Gajurel, J.; Hartikainen, H.; Keller, D.; Leys, M.; Říčanová, Š.; Segelbacher, G.; et al. Lessons Learned from Microsatellite Development for Nonmodel Organisms Using 454 Pyrosequencing. J. Evol. Biol. 2013, 26, 600–611. [Google Scholar] [CrossRef] [PubMed]
- Hildebrand, C.E.; Torney, D.C.; Wagner, R.P. Informativeness of Polymorphic DNA Markers. Los Alamos Sci. 1992, 20, 100–102. [Google Scholar]
- Andrews, S. FastQC—A Quality Control Tool for High Throughput Sequence Data; Babraham Institute: Cambridge, UK, 2010. [Google Scholar]
- Bushnell, B.; Rood, J.; Singer, E. BBMerge—Accurate Paired Shotgun Read Merging via Overlap. PLoS ONE 2017, 12, e0185056. [Google Scholar] [CrossRef]
- Smit, A.; Hubley, R.; Grenn, P. RepeatMasker Open-4.0; Institute for System Biology: Seattle, WA, USA, 2013. [Google Scholar]
- Thiel, T.; Michalek, W.; Varshney, R.K.; Graner, A. Exploiting EST Databases for the Development and Characterization of Gene-Derived SSR-Markers in Barley (Hordeum vulgare L.). Theor. Appl. Genet. 2003, 106, 411–422. [Google Scholar] [CrossRef]
- Zhang, Z.; Schwartz, S.; Wagner, L.; Miller, W. A Greedy Algorithm for Aligning DNA Sequences. J. Comput. Biol. 2000, 7, 203–214. [Google Scholar] [CrossRef]
- Koressaar, T.; Remm, M. Enhancements and Modifications of Primer Design Program Primer3. Bioinformatics 2007, 23, 1289–1291. [Google Scholar] [CrossRef]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3-New Capabilities and Interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef]
- Schuelke, M. An Economic Method for the Fluorescent Labeling of PCR Fragments. Nat. Biotechnol. 2000, 18, 233–234. [Google Scholar] [CrossRef] [PubMed]
- Radosavljević, I.; Jakse, J.; Javornik, B.; Satovic, Z.; Liber, Z. New Microsatellite Markers for Salvia Officinalis (Lamiaceae) and Cross-Amplification in Closely Related Species. Am. J. Bot. 2011, 98, e316–e318. [Google Scholar] [CrossRef] [PubMed]
- Rousset, F. Genepop’007: A Complete Re-Implementation of the Genepop Software for Windows and Linux. Mol. Ecol. Resour. 2008, 8, 103–106. [Google Scholar] [CrossRef]
- Holm, S. A Simple Sequentially Rejective Multiple Test Procedure. Scand. J. Stat. 1979, 6, 65–70. [Google Scholar]
- van Oosterhout, C.; Hutchinson, W.F.; Wills, D.P.M.; Shipely, P. Micro-Checker: Software for Identifying and Correcting Genotyping Errors in Microsatellite Data. Mol. Ecol. Notes 2004, 4, 535–538. [Google Scholar] [CrossRef]
- Kalinowski, S.T.; Taper, M.L.; Marshall, T.C. Revising How the Computer Program CERVUS Accommodates Genotyping Error Increases Success in Paternity Assignment. Mol. Ecol. 2007, 16, 1099–1106. [Google Scholar] [CrossRef]
- Bowcock, A.M.; Ruiz-Linares, A.; Tomfohrde, J.; Minch, E.; Kidd, J.R.; Cavalli-Sforza, L.L. High Resolution of Human Evolutionary Trees with Polymorphic Microsatellites. Nature 1994, 368, 455–457. [Google Scholar] [CrossRef]
- Minch, E.; Ruiz-Linares, A.; Goldstein, D.; Feldman, M.; Cavalli-Sforza, L.L. MICROSAT: A Computer Program for Calculating Various Statistics on Microsatellite Allele Data; Stanford University: Stanford, CA, USA, 1997. [Google Scholar]
- Felsenstein, J. PHYLIP (Phylogenetic Inference Package, Version 3.6b); Department of Genetics, University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
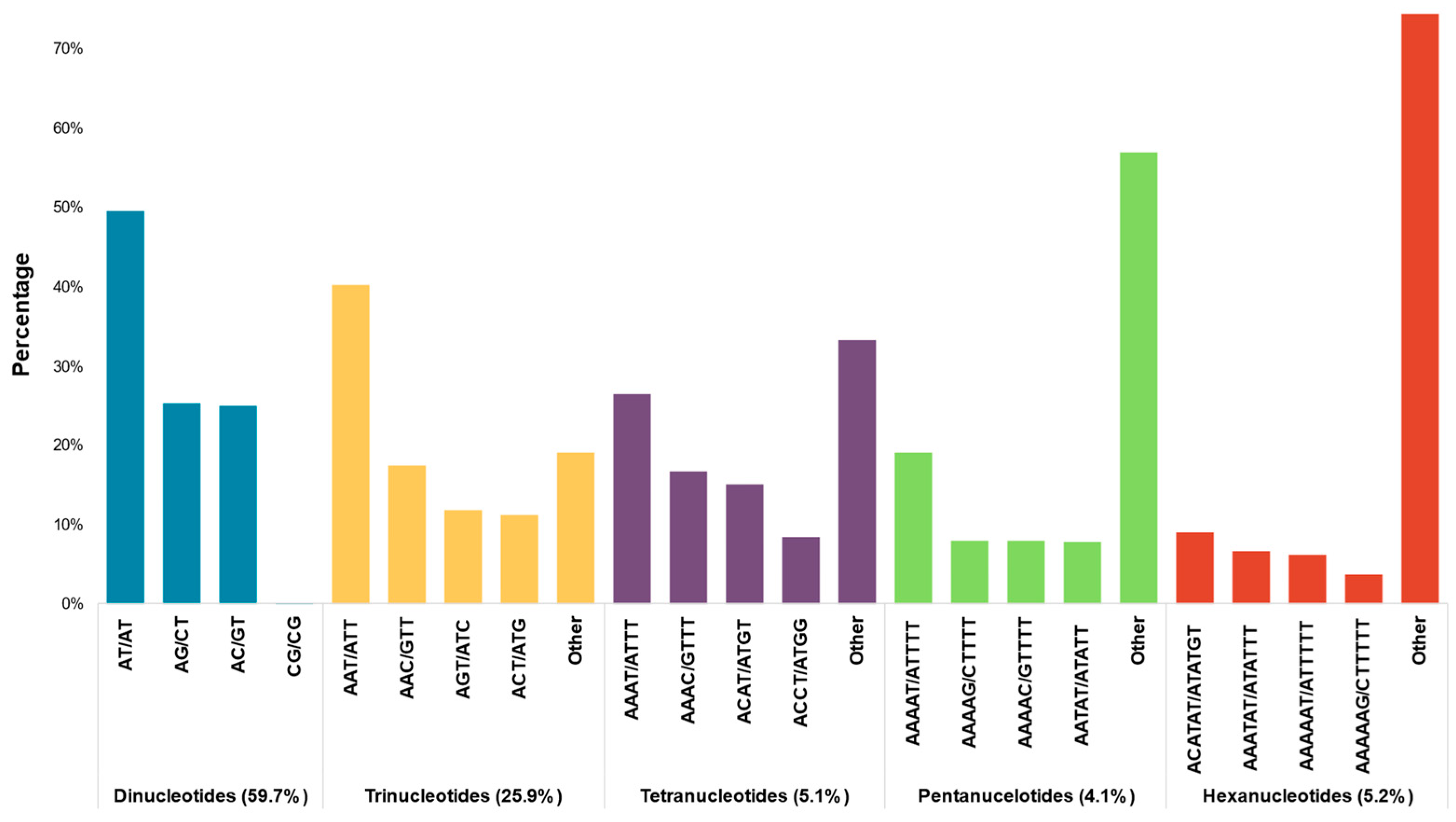
| Category | First Approach | Second Approach | Combined Results |
|---|---|---|---|
| Total number of contigs examined | 923,207 | 5,986,468 | 6,909,675 |
| Total size of examined contigs (bp) | 409,503,869 | 1,311,396,942 | 1,720,900,811 |
| Total number of identified SSRs | 11,652 | 23,904 | 35,556 |
| SSR containing contigs | 9973 | 21,407 | 31,380 |
| Contigs containing more than one SSR | 1267 | 2005 | 3272 |
| GC content (%) | 34.34 | 34.41 | 35.89 |
| Locus Name | GenBank Acc. No. | Primer Sequence (5′ to 3′) | Repeat Motif | Size (bp) |
|---|---|---|---|---|
| TcUniZg004 | MW498263 | F*: TGATCTTTTAAATTTGGAACTGAA | (GT)10 | 248–256 |
| R: GAAAGCTTCCTCTACCTCCTTG | ||||
| TcUniZg005 | MW498264 | F: CCAGATCATTTACTTAAAATGGAACA | (AC)8 | 217–223 |
| R: TACAACACTGGTGGCGTCAT | ||||
| TcUniZg006 | MW498265 | F: CGACGGTTGGTGTGTGTATC | (GA)10 | 224–280 |
| R: CCATACGTGTCTCTTTCTCTTTGA | ||||
| TcUniZg007 | MW498266 | F: GCTTCACATGGTTCGTCTCTG | (CA)8 | 191–203 |
| R: GCTTCACATGGTTCGTCTCTG | ||||
| TcUniZg008 | MW498267 | F: TGCGATGATGATGATTGAGAG | (GT)11 | 116–146 |
| R: ATGGCAGAACATTCAACACAA | ||||
| TcUniZg009 | MW498268 | F: TCTCCTTCTTCCTCCTGCAA | (CA)11 | 106–118 |
| R: GGATGTTTGTTGTGTTCATTGG | ||||
| TcUniZg010 | MW498269 | F: CATACCTCCGCCCTTGATTA | (TG)8 | 180–194 |
| R: CCAAGACCCACTTTTTGGTG | ||||
| TcUniZg012 | MW498270 | F: TCATCATCAACAAAATATCCAAGAA | (CA)10 | 244–254 |
| R: CCACCGACCACCTCATAATC | ||||
| TcUniZg013 | MW498271 | F: ACATAACGTCGGAGGCATCA | (TA)8 | 216–222 |
| R: TGAGTTGGGTGCGTTACAAA | ||||
| TcUniZg014 | MW498272 | F: AGCATAGACTGACTGTTCCTTCA | (TG)12 | 216–230 |
| R: CCATATTCATCACAGCCTACGA | ||||
| TcUniZg017 | MW498273 | F: AAGGCTGCGCTTCTTAACAG | (TA)10 | 258–274 |
| R: TAGCCATGCCTGGGTACTTC | ||||
| TcUniZg019 | MW498274 | F: AATGTGTGACTAATGGTCCTCAGA | (TA)8 | 116–124 |
| R: TGTTACTTAATTATAACATGCGGCCTA | ||||
| TcUniZg020 | MW498275 | F: ACCACCAATACAAATACACCTTC | (CA)7 | 113–117 |
| R: GCAGAGGCTCGAGCTAGGAC | ||||
| TcUniZg023 | MW498276 | F: CACAAATCCTTCACCTGTCAAA | (AC)9 | 240–250 |
| R: GCCAGTGGCAGAAGAGAAGT | ||||
| TcUniZg032 | MW498277 | F: GAAATCAAGTGCGGATACGA | (CAT)8 | 106–115 |
| R: TTTCCATATTGTGTTTTGGGTTC | ||||
| TcUniZg037 | MW498278 | F: GGACGGGATTACAGAAGGTG | (CAA)7 | 249–258 |
| R: TCGACCTCATTATGCTGCTG | ||||
| TcUniZg038 | MW498279 | F: GGAGCCAAATACTAGCCTTCAA | (TTG)6 | 151–163 |
| R: CGTTAGTCATCCGTGAGCAA |
| Locus | Na | HO | HE | FIS | Sign | Pnull | PIC | PI |
|---|---|---|---|---|---|---|---|---|
| TcUniZg004 | 4 | 0.167 | 0.235 | 0.29 | ns | - | 0.219 | 0.605 |
| TcUniZg005 | 4 | 0.304 | 0.449 | 0.322 | ns | - | 0.384 | 0.37 |
| TcUniZg006 | 13 | 0.542 | 0.633 | 0.145 | ns | - | 0.605 | 0.158 |
| TcUniZg007 | 5 | 0.333 | 0.505 | 0.339 | ns | 0.091 | 0.432 | 0.318 |
| TcUniZg008 | 11 | 0.708 | 0.864 | 0.18 | ns | - | 0.824 | 0.043 |
| TcUniZg009 | 6 | 0.583 | 0.774 | 0.246 | ns | 0.111 | 0.716 | 0.098 |
| TcUniZg010 | 6 | 0.667 | 0.757 | 0.12 | ns | - | 0.697 | 0.11 |
| TcUniZg012 | 5 | 0.391 | 0.542 | 0.277 | ns | - | 0.485 | 0.266 |
| TcUniZg013 | 4 | 0.375 | 0.323 | −0.16 | ns | - | 0.288 | 0.495 |
| TcUniZg014 | 6 | 0.542 | 0.531 | −0.021 | ns | - | 0.488 | 0.263 |
| TcUniZg017 | 7 | 0.542 | 0.704 | 0.23 | ns | - | 0.639 | 0.146 |
| TcUniZg019 | 5 | 0.583 | 0.72 | 0.19 | ns | - | 0.654 | 0.137 |
| TcUniZg020 | 3 | 0.083 | 0.197 | 0.576 | ns | 0.111 | 0.178 | 0.668 |
| TcUniZg023 | 3 | 0.458 | 0.433 | −0.059 | ns | - | 0.35 | 0.405 |
| TcUniZg032 | 3 | 0.5 | 0.592 | 0.155 | ns | - | 0.506 | 0.25 |
| TcUniZg037 | 4 | 0.125 | 0.299 | 0.582 | * | 0.146 | 0.266 | 0.528 |
| TcUniZg038 | 5 | 0.542 | 0.702 | 0.228 | ns | - | 0.634 | 0.15 |
| Accession Number a | Location | Elevation (m) | Latitude b | Longitude b |
|---|---|---|---|---|
| MAP02814 | Mali Lošinj | 39 | 44.574 | 14.420 |
| MAP02807 | Čiovo | 211 | 43.498 | 16.303 |
| MAP02809 | Biokovo—Kotiški Stanovi | 1350 | 43.314 | 17.062 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varga, F.; Liber, Z.; Jakše, J.; Turudić, A.; Šatović, Z.; Radosavljević, I.; Jeran, N.; Grdiša, M. Development of Microsatellite Markers for Tanacetum cinerariifolium (Trevis.) Sch. Bip., a Plant with a Large and Highly Repetitive Genome. Plants 2022, 11, 1778. https://doi.org/10.3390/plants11131778
Varga F, Liber Z, Jakše J, Turudić A, Šatović Z, Radosavljević I, Jeran N, Grdiša M. Development of Microsatellite Markers for Tanacetum cinerariifolium (Trevis.) Sch. Bip., a Plant with a Large and Highly Repetitive Genome. Plants. 2022; 11(13):1778. https://doi.org/10.3390/plants11131778
Chicago/Turabian StyleVarga, Filip, Zlatko Liber, Jernej Jakše, Ante Turudić, Zlatko Šatović, Ivan Radosavljević, Nina Jeran, and Martina Grdiša. 2022. "Development of Microsatellite Markers for Tanacetum cinerariifolium (Trevis.) Sch. Bip., a Plant with a Large and Highly Repetitive Genome" Plants 11, no. 13: 1778. https://doi.org/10.3390/plants11131778
APA StyleVarga, F., Liber, Z., Jakše, J., Turudić, A., Šatović, Z., Radosavljević, I., Jeran, N., & Grdiša, M. (2022). Development of Microsatellite Markers for Tanacetum cinerariifolium (Trevis.) Sch. Bip., a Plant with a Large and Highly Repetitive Genome. Plants, 11(13), 1778. https://doi.org/10.3390/plants11131778









