Identification of Long-Distance Transport Signal Molecules Associated with Plant Maturity in Tetraploid Cultivated Potatoes (Solanum tuberosum L.)
Abstract
:1. Introduction
2. Results
2.1. Effect of Grafting on Maturity
2.2. Grafting Affects Tuber Yield
2.3. Grafting Affected Hormone Contents
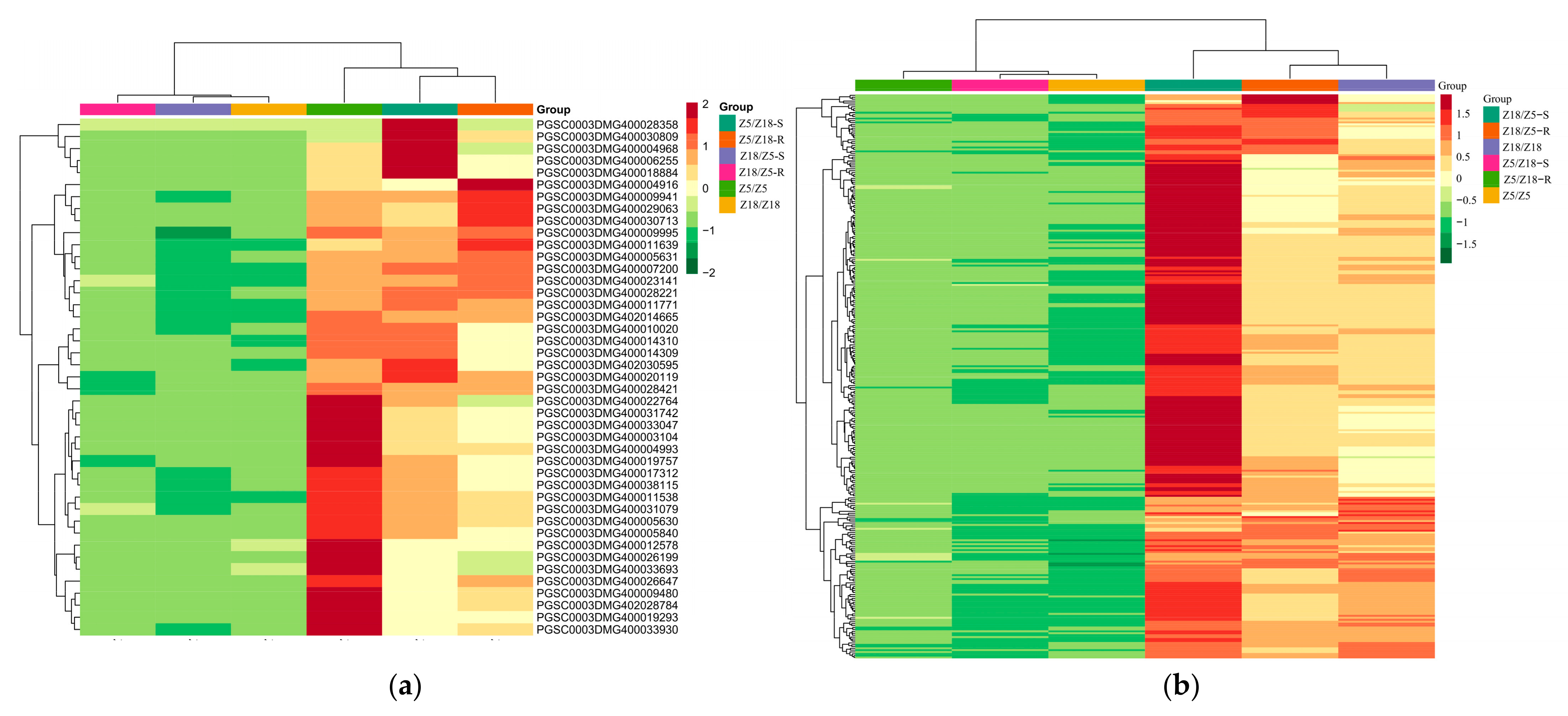
2.4. Identification of Long-Distance Transport mRNAs Associated with Maturity
2.5. GO Enrichment and KEGG Pathway Analysis of the Detected Long-Distance Transport mRNAs
2.6. Identification of Metabolites Associated with Maturity
2.7. KEGG Pathway Analysis of Metabolites Associated with Maturity
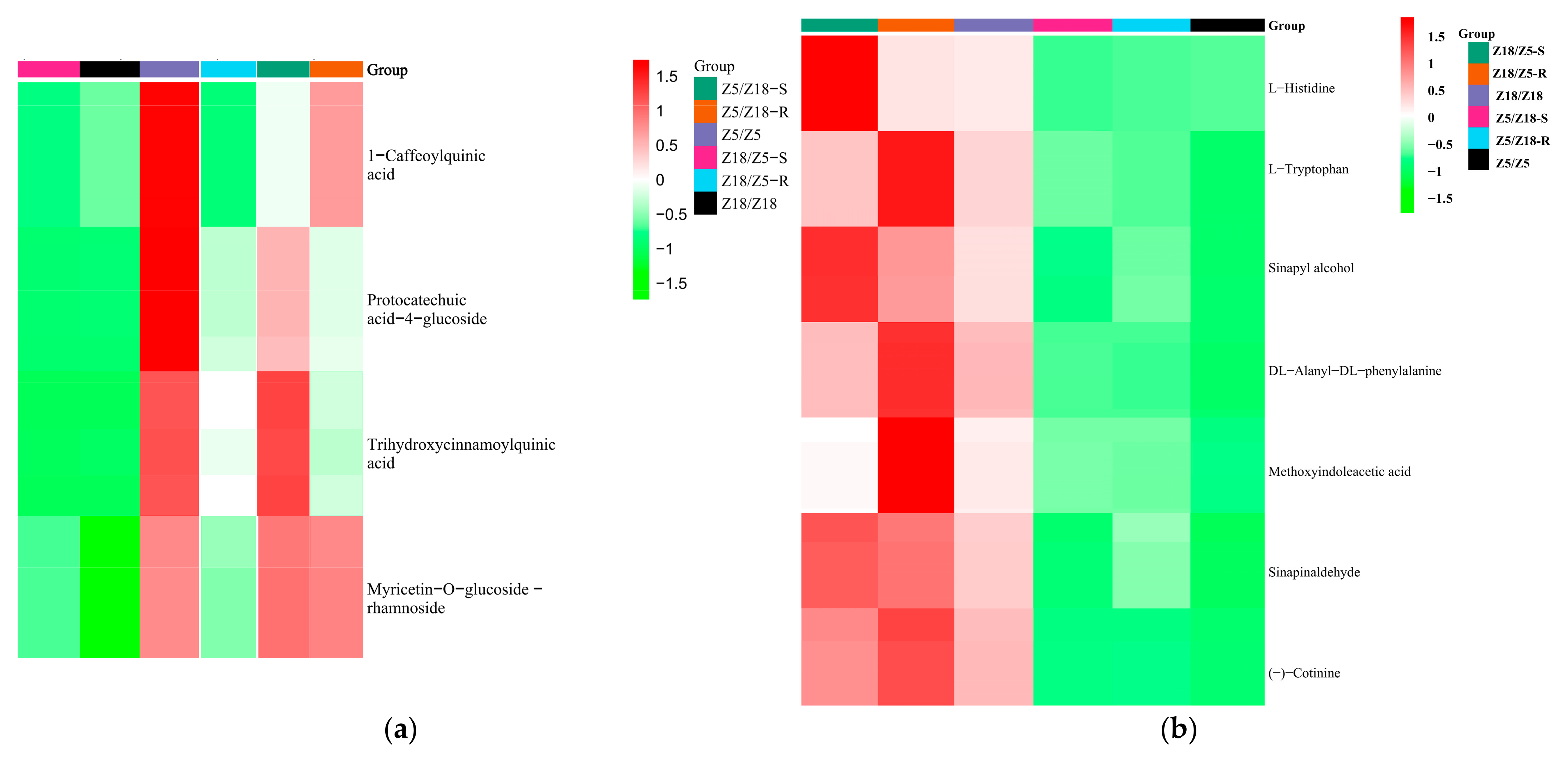
2.8. Identification of Graft-Transmissible Metabolites Associated with Maturity
2.9. Integrated Analysis of Long-Distance Transport mRNAs and Metabolites
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Grafting Procedure
4.3. Sample Preparation
4.4. Identification of Plant Maturity and Tuber Yield of Grafted Plants
4.5. Phytohormone Analysis
4.6. RNAseq Sample and Library Preparation
4.7. Sequencing Data Analysis
4.8. Widely Targeted Metabolome Sample Preparation and Analysis
4.9. Chromatographic Mass Spectrometry Acquisition Conditions
4.10. Metabolomics Data Analysis
4.11. Identification Standards for Long-Distance Transport Signals Associated with Potato Plant Maturity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stokstad, E. The new potato. Science 2019, 363, 574–577. [Google Scholar] [CrossRef] [PubMed]
- Kloosterman, B.; Abelenda, J.A.; Gomez, M.M.C.; Oortwijn, M.; de Boer, J.M.; Kowitwanich, K.; Horvath, B.M.; van Eck, H.J.; Smaczniak, C.; Prat, S.; et al. Naturally occurring allele diversity allows potato cultivation in northern latitudes. Nature 2013, 495, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Hackett, C.A.; Bradshaw, J.E.; Bryan, G.J. QTL mapping in autotetraploids using SNP dosage information. Theor. Appl. Genet. 2014, 127, 1885–1904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Massa, A.N.; Manrique-Carpintero, N.C.; Coombs, J.J.; Zarka, D.G.; Boone, A.E.; Kirk, W.W.; Hackett, C.A.; Bryan, G.J.; Douches, D.S. Genetic linkage mapping of economically important traits in cultivated tetraploid potato (Solanum tuberosum L.). G3 2015, 5, 2357–2364. [Google Scholar] [CrossRef] [Green Version]
- Massa, A.N.; Manrique-Carpintero, N.C.; Coombs, J.J.; Haynes, K.G.; Bethke, P.C.; Brandt, T.L.; Gupta, S.K.; Yencho, G.C.; Novy, R.G.; Douches, D.S. Linkage analysis and QTL mapping in a tetraploid russet mapping population of potato. BMC Genet. 2018, 19, 87. [Google Scholar] [CrossRef] [Green Version]
- Mackerron, D.K.L.; Davies, H.V. Markers for maturity and senescence in the potato crop. Potato Res. 1986, 29, 427–436. [Google Scholar] [CrossRef]
- Turnbull, C.G.N.; Lopez-Cobollo, R.M. Heavy traffic in the fast lane: Long-distance signalling by macromolecules. New Phytol. 2013, 198, 33–51. [Google Scholar] [CrossRef]
- Osaki, M.; Shinano, T.; Matsumoto, M.; Zheng, T.; Tadano, T. A root-shoot interaction hypothesis for high productivity of field crops. Soil Sci. Plant Nutr. 1997, 43 (Suppl. 1), 1079–1084. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y. Plant Senescence (Methods and Protocols); Humana Press: New York, NY, USA, 2018; pp. 3–7. [Google Scholar]
- Santner, A.; Estelle, M. Recent advances and emerging trends in plant hormone signalling. Nature 2009, 459, 1071–1078. [Google Scholar] [CrossRef]
- Shan, X.; Yan, J.; Xie, D. Comparison of phytohormone signaling mechanisms. Curr. Opin. Plant Biol. 2012, 15, 84–91. [Google Scholar] [CrossRef]
- Li, J.; Li, C. Seven-year major research progress in plant hormones by China scholars. Sci Sin Vitae 2019, 49, 1227–1281. [Google Scholar] [CrossRef]
- Mueller-Roeber, B.; Balazadeh, S. Auxin and its role in plant senescence. J. Plant Growth Regul. 2014, 33, 21–33. [Google Scholar] [CrossRef]
- Mayta, M.L.; Lodeyro, A.F.; Guiamet, J.J.; Tognetti, V.B.; Melzer, M.; Hajirezaei, M.R.; Carrillo, N. Expression of a plastid-targeted flavodoxin decreases chloroplast reactive oxygen species accumulation and delays senescence in aging tobacco leaves. Front. Plant Sci. 2018, 9, 1039. [Google Scholar] [CrossRef]
- Meng, M.; Liu, M.; Men, F.; Mao, X.; Li, X. Study on the correlation ship of tuber yield with the endogenous hormone contents of leaves of potato. J. Inn. Mong. Inst. Agric. Anim. Husb. 1997, 4, 8–11. [Google Scholar]
- Kim, T.; Kang, K.; Kim, S.; An, G.; Paek, N. OsWRKY5 promotes rice leaf senescence via senescence-associated NAC and abscisic acid biosynthesis pathway. Int. J. Mol. Sci. 2019, 20, 4437. [Google Scholar] [CrossRef] [Green Version]
- Guo, P.; Li, Z.; Huang, P.; Li, B.; Fang, S.; Chu, J.; Guo, H. A tripartite amplification loop involving the transcription factor WRKY75, salicylic acid, and reactive oxygen species accelerates leaf senescence. Plant Cell 2017, 29, 2854–2870. [Google Scholar] [CrossRef]
- Zhou, M.; Sakuraba, Y.; Yanagisawa, S. A jasmonate-activated MYC2-Dof2.1-MYC2 transcriptional loop promotes leaf senescence in Arabidopsis. Plant Cell 2020, 32, 242–262. [Google Scholar] [CrossRef]
- Makoi, J.H.J.R.; Ndakidemi, P.A. Biological, ecological and agronomic significance of plant phenolic compounds in rhizosphere of the symbiotic legumes. Afr. J. Biotechnol. 2007, 12, 1358–1368. [Google Scholar]
- Wang, Y. Plant grafting and its application in biological research. Chin. Sci. Bull. 2011, 56, 3511–3517. [Google Scholar] [CrossRef] [Green Version]
- Li, B.; Wang, Y.; Zhang, Z.; Wang, B.; Eneji, A.E.; Duan, L.; Li, Z.; Tian, X. Cotton shoot plays a major role in mediating senescence induced by potassium defificiency. J. Plant Physiol. 2012, 169, 327–335. [Google Scholar] [CrossRef]
- Zhang, G.; Mao, Z.; Wang, Q.; Song, J.; Nie, X.; Wang, T.; Zhang, H.; Guo, H. Comprehensive transcriptome profiling and phenotyping heterografting system. Physiol. Plant. 2019, 166, 833–847. [Google Scholar] [CrossRef]
- Banerjee, A.K.; Chatterjee, M.; Yu, Y.; Suh, S.G.; Miller, W.A.; Hannapel, D.J. Dynamics of a mobile RNA of potato involved in a long-distance signaling pathway. Plant Cell 2006, 18, 3443–3457. [Google Scholar] [CrossRef] [Green Version]
- Mahajan, A.; Bhogale, S.; Kang, I.H.; Hannapel, D.J.; Banerjee, A.K. The mRNA of a Knotted1-like transcription factor of potato is phloem mobile. Plant Mol. Biol. 2012, 79, 595–608. [Google Scholar] [CrossRef]
- Ghate, T.H.; Sharma, P.; Kondhare, K.R.; Hannapel, D.J.; Banerjee, A.K. The mobile RNAs, StBEL11 and StBEL29, suppress growth of tubers in potato. Plant Mol. Biol. 2017, 93, 563–578. [Google Scholar] [CrossRef] [PubMed]
- Gilmour, S.; Fowler, S.G.; Thomashow, M.F. Arabidopsis transcriptional activators CBF1, CBF2, and CBF3 have matching functional activities. Plant Mol. Biol. 2004, 54, 767–781. [Google Scholar] [CrossRef] [PubMed]
- An, D.; Ma, Q.; Yan, W.; Zhou, W.; Liu, G.; Zhang, P. Divergent regulation of CBF regulon on cold tolerance and plant phenotype in Cassava overexpressing Arabidopsis CBF3 gene. Front. Plant Sci. 2016, 7, 1866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Causier, B.; Kieffer, M.; Davies, B. MADS-box genes reach maturity. Science 2002, 296, 275–276. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Xu, H.; Ju, Z.; Cao, D.; Zhu, H.; Fu, D.; Grierson, D.; Qin, G.; Luo, Y.; Zhu, B. The RIN-MC fusion of MADS-box transcription factors has transcriptional activity and modulates expression of many ripening genes. Plant Physiol. 2018, 176, 891–909. [Google Scholar] [CrossRef] [Green Version]
- Saito, T.; Bai, S.; Imai, T.; Ito, A.; Nakajima, I.; Moriguchi, T. Histone modification and signaling cascade of the dormancy-associated MADS-box gene, PpMADS13-1, in Japanese pear (Pyrus pylifolia) during endodormancy. Plant Cell Environ. 2015, 6, 1157–1166. [Google Scholar] [CrossRef]
- Niu, Q.; Li, J.; Cai, D.; Qian, M.; Jia, H.; Bai, S.; Hussain, S.; Liu, G.; Teng, Y.; Zheng, X. Dormancy-associated MADS-box genes and microRNAs jointly control dormancy transition in pear (Pyrus pyrifolia white pear group) flower bud. J. Exp. Bot. 2016, 67, 239–257. [Google Scholar] [CrossRef] [Green Version]
- Wisniewski, M.; Norelli, J.; Artlip, T. Overexpression of a peach CBF gene in apple: A model for understanding the integration of growth, dormancy, and cold hardiness in woody plants. Front. Plant Sci. 2015, 6, 85. [Google Scholar] [CrossRef] [Green Version]
- Artlip, T.S.; Wisniewski, M.E.; Arora, R.; Norelli, J.L. An apple rootstock overexpressing a peach CBF gene alters growth and flowering in the scion but does not impact cold hardiness or dormancy. Hortic. Res. 2016, 3, 16006. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Liu, L.; Huang, W.; Yuan, M.; Zhou, F.; Li, X.; Lin, Y. Overexpression of OsSWEET5 in rice causes growth retardation and precocious senescence. PLoS ONE 2014, 9, e94210. [Google Scholar] [CrossRef] [Green Version]
- Ni, J.; Li, J.; Zhu, R.; Zhang, M.; Qi, K.; Zhang, S.; Wu, J. Overexpression of sugar transporter gene PbSWEET4 of pear causes sugar reduce and early senescence in leaves. Gene 2020, 743, 144582. [Google Scholar] [CrossRef]
- Liu, Y.; Lv, J.; Liu, Z.; Wang, J.; Yang, B.; Chen, W.; Ou, L.; Dai, X.; Zhang, Z.; Zou, X. Integrative analysis of metabolome and transcriptome reveals the mechanism of color formation in pepper fruit (Capsicum annuum L.). Food Chem. 2020, 306, 125629. [Google Scholar] [CrossRef]
- Garcia-Lozano, M.; Dutta, S.K.; Natarajan, P.; Tomason, Y.R.; Lopez, C.; Katam, R.; Levi, A.; Nimmakayala, P.; Reddy, U.K. Transcriptome changes in reciprocal grafts involving watermelon and bottle gourd reveal molecular mechanisms involved in increase of the fruit size, rind toughness and soluble solids. Plant Mol. Biol. 2020, 102, 213–223. [Google Scholar] [CrossRef]
- Hoopes, G.; Meng, X.; Hamilton, J.P.; Achakkagari, S.R.; Guesdes, F.d.A.F.; Bolger, M.E.; Coombs, J.J.; Esselink, D.; Kaiser, N.R.; Kodde, L.; et al. Phased, chromosome-scale genome assemblies of tetraploid potato reveal a complex genome, transcriptome, and predicted proteome landscape underpinning genetic diversity. Mol. Plant 2022, 15, 520–536. [Google Scholar] [CrossRef]
- Navarro, C.; Abelenda, J.A.; Cruz-Oro, E.; Cuellar, C.A.; Tamaki, S.; Silva, J.; Shimamoto, K.; Prat, S. Control of flowering and storage organ formation in potato by FLOWERING LOCUS T. Nature 2011, 478, 119–123. [Google Scholar] [CrossRef]
- Plantenga, F.D.M.; Bergonzi, S.; Abelenda, J.A.; Bachem, C.W.B.; Visser, R.G.F.; Heuvelink, E.; Marcelis, L.F.M. The tuberization signal StSP6A represses flower bud development in potato. J. Exp. Bot. 2019, 70, 937–948. [Google Scholar] [CrossRef]
- Thomas, H. Senescence, ageing and death of the whole plant. New Phytol. 2013, 197, 696–711. [Google Scholar] [CrossRef]
- Shahri, W.; Ahmad, S.S.; Tahir, I. Sugar signaling in plant growth and development. In Plant Signaling: Understanding the Molecular Crosstalk; Springer: New Delhi, India, 2014; pp. 93–116. [Google Scholar]
- Yoon, J.; Cho, L.H.; Tun, W.; Jeon, J.S.; An, G. Sucrose signaling in higher plants. Plant Sci. 2021, 302, 110703. [Google Scholar] [CrossRef]
- Kumar, R.; Bishop, E.; Bridges, W.C.; Tharayil, N.; Sekhon, R.S. Sugar partitioning and source-sink interaction are key determinants of leaf senescence in maize. Plant Cell Environ. 2019, 42, 2597–2611. [Google Scholar] [CrossRef]
- Ma, D.; Li, Y.; Zhang, J.; Wang, C.; Qin, H.; Ding, H.; Xie, Y.; Guo, T. Accumulation of phenolic compounds and expression profiles of phenolic acid biosynthesis-related genes in developing grains of white, purple, and red wheat. Front. Plant Sci. 2016, 7, 528. [Google Scholar] [CrossRef]
- Andriana, Y.; Quy, T.N.; Xuan, T.D. Phenolic acids as plant growth inhibitors from Tridax procumbens L. IOP Conf. Ser. Earth Environ. Sci. 2019, 250, 012024. [Google Scholar] [CrossRef] [Green Version]
- Harrison, H.F., Jr.; Mitchell, T.R.; Peterson, J.K.; Wechter, W.P.; Majetich, G.R. Caffeoylquinic acids in storage roots of sixteen sweetpotato genotypes. J. Am. Soc. Hortsci. 2008, 133, 492–500. [Google Scholar] [CrossRef]
- Buer, C.S.; Muday, G.K. The transparent testa4 mutation prevents flavonoid synthesis and alters Auxin transport and the response of Arabidopsis roots to gravit and light. Plant Cell 2004, 16, 1191–1205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peer, W.A.; Bandyopadhyay, A.; Blakeslee, J.J.; Makam, S.N.; Chen, R.J.; Masson, P.H.; Murphy, A.S. Variation in expression and protein localization of the PIN family of Auxin efflux facilitator proteins in flavonoid mutants with altered auxin transport in Arabidopsis thaliana. Plant Cell 2004, 16, 1898–1911. [Google Scholar] [CrossRef] [Green Version]
- Yin, R.; Han, K.; Heller, W.; Albert, A.; Dobrev, P.I.; Zazimalova, E.; Schaffner, A.R. Kaemperol 3-O-rhamnoside-7-O-rhamnoside is an endogenous flavonol inhibitor of polar auxin transport in Arabidopsis shoots. New Phytol. 2014, 201, 466–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.-M.; Kubota, C.; Tsao, S.J.; Bie, Z.; Echevarria, P.H.; Morra, L.; Oda, M. Current status of vegetable grafting: Diffusion, grafting techniques, automation. Sci. Hortic. 2010, 127, 93–105. [Google Scholar] [CrossRef]
- Li, X.; Xu, J.; Duan, S.; Zhang, J.; Bian, C.; Hu, J.; Li, G.; Jin, L. Mapping and QTL analysis of early-maturity traits in tetraploid potato (Solanum tuberosum L.). Int. J. Mol. Sci. 2018, 19, 3065. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Wu, Q.; He, W.; He, T.; Wu, Q.; Miao, Y. Combined De Novo transcriptome and metabolome analysis of common Bean response to Fusarium oxysporum f. sp. phaseoli Infection. Int. J. Mol. Sci. 2019, 20, 6278. [Google Scholar] [CrossRef] [Green Version]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [Green Version]
- Varet, H.; Brillet-Gueguen, L.; Coppee, J.Y.; Dillies, M.A. SARTools: A DESeq2and EdgeR-Based R Pipeline for comprehensive differential analysis of RNA-Seq Data. PLoS ONE 2016, 11, e0157022. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.; Gong, L.; Guo, Z.; Wang, W.; Zhang, H.; Liu, X.; Yu, S.; Xiong, L.; Luo, J. A novel integrated method for large-scale detection, identification, and quantification of widely targeted metablolites: Application in the study of rice metabolomics. Mol. Plant 2013, 6, 1769–1780. [Google Scholar] [CrossRef] [Green Version]




| Graft Combinations | Mean Tuber Yield Per Plant (g) |
|---|---|
| Z18/Z5 | 501.25 ± 300.50 a |
| Z5/Z5 | 278.57 ± 108.54 b |
| Z5/Z18 | 384.44 ± 197.87 ab |
| Z18/Z18 | 555.00 ± 139.96 a |
| Stem Tissues | ABA (ng/g FW) | SA (ng/g FW) | IAA (ng/g FW) |
|---|---|---|---|
| Z5/Z18-S | 1.77 ± 0.78 a | 205.00 ± 137.45 ab | 10.40 ± 1.93 a |
| Z5/Z18-R | 1.39 ± 0.30 a | 109.23 ± 28.43 ab | 11.86 ± 3.32 a |
| Z5/Z5 | 1.83 ± 0.33 a | 250.13 ± 167.13 a | 7.42 ± 3.04 a |
| Z18/Z5-S | 1.74 ± 0.16 a | 131.57 ± 40.34 ab | 27.70 ± 7.12 b |
| Z18/Z5-R | 1.23 ± 0.25 a | 87.30 ± 38.36 b | 23.13 ± 7.71 b |
| Z18/Z18 | 1.18 ± 0.46 a | 139.00 ± 19.00 ab | 21.73 ± 5.37 b |
| Metabolites | Class | The KEGG Pathway |
|---|---|---|
| 1-Caffeoylquinic acid | Phenolic acids | N/A |
| Protocatechuic acid-4-glucoside | Phenolic acids | N/A |
| Trihydroxycinnamoylquinic acid | Phenolic acids | N/A |
| Myricetin-O-glucoside-rhamnoside | Flavonoids | N/A |
| Metabolites | Class | The KEGG Pathway |
|---|---|---|
| L-histidine | Amino-acids and derivatives | Histidine metabolism |
| L-tryptophan | Amino-acids and derivatives | Tryptophan metabolism |
| DL-alanyl-DL-phenylalanine | Amino-acids and derivatives | N/A |
| Sinapyl alcohol | Phenolic acids | Phenylpropanoid biosynthesis |
| Sinapinaldehyde | Phenolic acids | N/A |
| (-)-Cotinine | Alkaloids | N/A |
| Methoxy indole acetic acid | Alkaloids | Tryptophan metabolism |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hui, Z.; Xu, J.; Jian, Y.; Bian, C.; Duan, S.; Hu, J.; Li, G.; Jin, L. Identification of Long-Distance Transport Signal Molecules Associated with Plant Maturity in Tetraploid Cultivated Potatoes (Solanum tuberosum L.). Plants 2022, 11, 1707. https://doi.org/10.3390/plants11131707
Hui Z, Xu J, Jian Y, Bian C, Duan S, Hu J, Li G, Jin L. Identification of Long-Distance Transport Signal Molecules Associated with Plant Maturity in Tetraploid Cultivated Potatoes (Solanum tuberosum L.). Plants. 2022; 11(13):1707. https://doi.org/10.3390/plants11131707
Chicago/Turabian StyleHui, Zhiming, Jianfei Xu, Yinqiao Jian, Chunsong Bian, Shaoguang Duan, Jun Hu, Guangcun Li, and Liping Jin. 2022. "Identification of Long-Distance Transport Signal Molecules Associated with Plant Maturity in Tetraploid Cultivated Potatoes (Solanum tuberosum L.)" Plants 11, no. 13: 1707. https://doi.org/10.3390/plants11131707
APA StyleHui, Z., Xu, J., Jian, Y., Bian, C., Duan, S., Hu, J., Li, G., & Jin, L. (2022). Identification of Long-Distance Transport Signal Molecules Associated with Plant Maturity in Tetraploid Cultivated Potatoes (Solanum tuberosum L.). Plants, 11(13), 1707. https://doi.org/10.3390/plants11131707






