Impact of Leaf Removal on Phenolics and Antioxidant Activity of Trebbiano Berries (Vitis vinifera L.)
Abstract
1. Introduction
2. Results
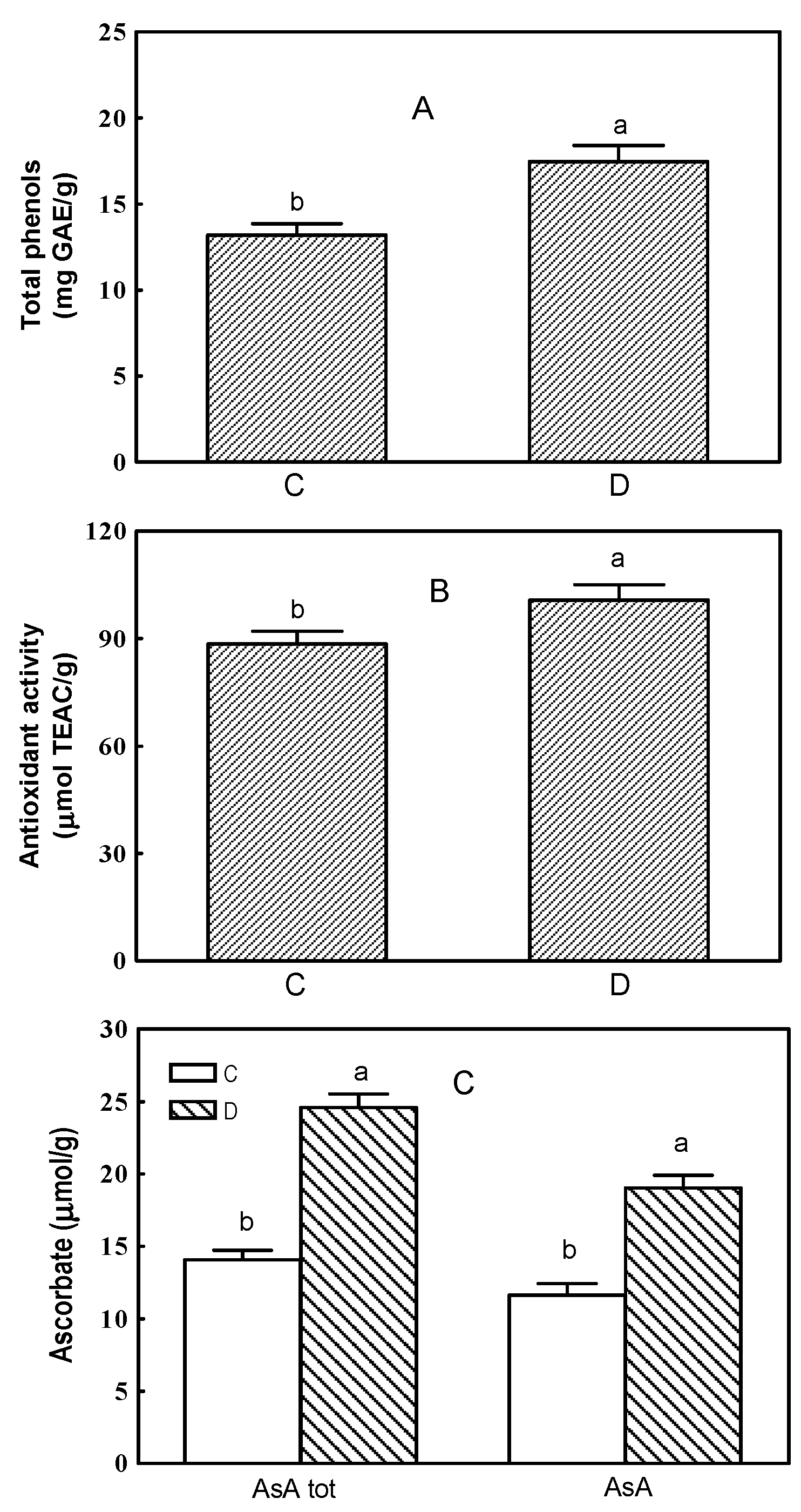
2.1. Total Phenols, Antioxidant Activity and Ascorbate
2.2. Phenolic Acids and Flavonols
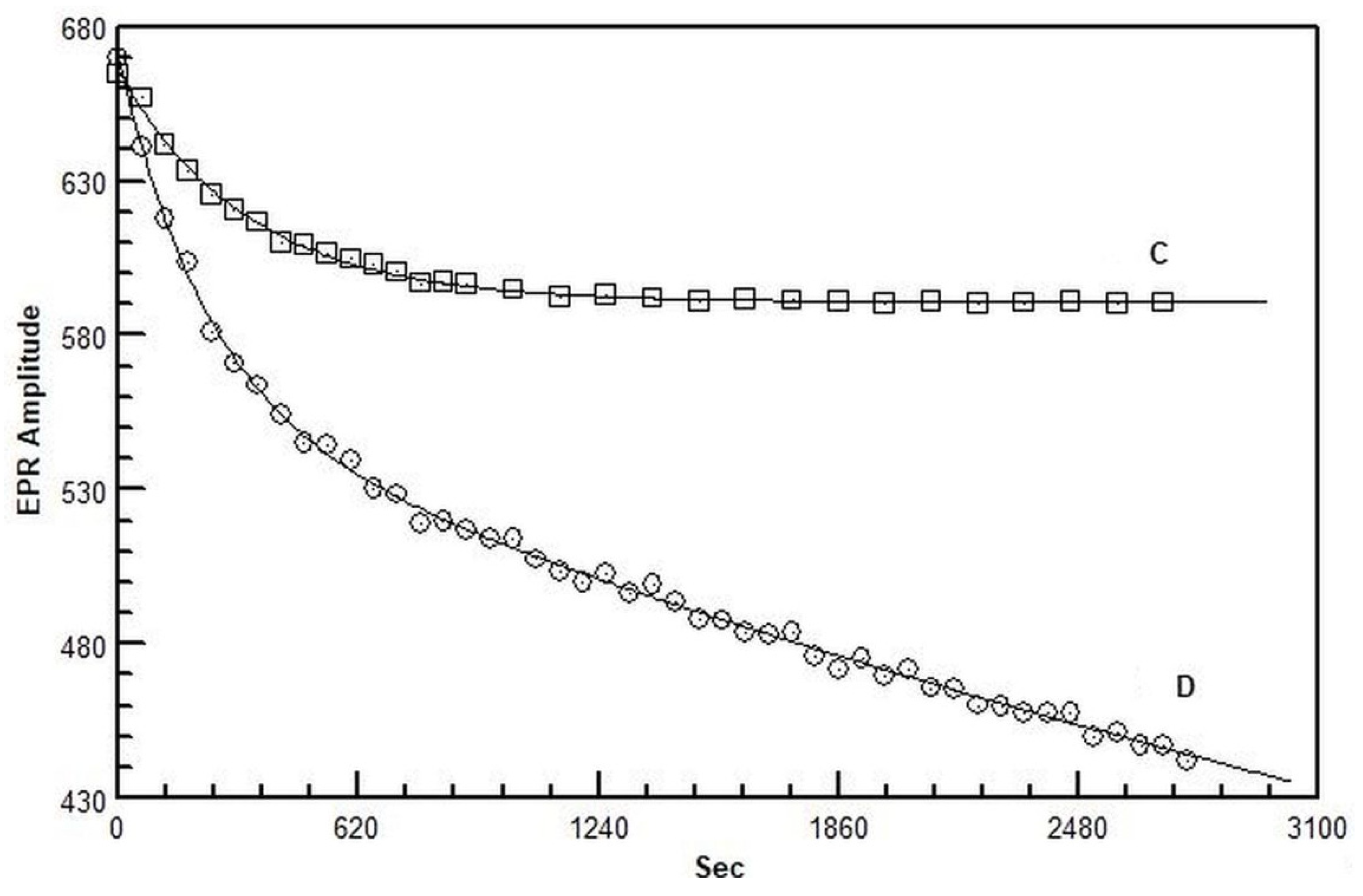
2.3. Electron Paramagnetic Resonance
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Experimental Setup
4.3. Ascorbate
4.4. Sample Extracts
4.5. Total Phenols
4.6. Antioxidant Activity
4.7. Phenolic Acids
4.8. Flavonoids
4.9. EPR Decay Kinetics
4.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pérez-Lόpez, U.; Pinzino, C.; Quartacci, M.F.; Ranieri, A.; Sgherri, C. Phenolic composition and related antioxidant properties in differently colored lettuces: A study by electron paramagnetic resonance (EPR) kinetics. J. Agric. Food Chem. 2014, 62, 12001–12007. [Google Scholar] [CrossRef]
- Cassino, C.; Gianotti, V.; Bonello, F.; Tsolakis, C.; Cravero, M.C.; Osella, D. Antioxidant composition of a selection of Italian red wines and their corresponding free-radical scavenging ability. J. Chem. 2016, 2016, 4565391. [Google Scholar] [CrossRef]
- Teixeira, A.; Eiras-Dias, J.; Castellarin, S.D.; Gerós, H. Berry phenolics of grapevine under challenging environments. Int. J. Mol. Sci. 2013, 14, 18711–18739. [Google Scholar] [CrossRef]
- Downey, M.O.; Harvey, J.S.; Robinson, S.P. Synthesis of flavonols and expression of flavonol synthase genes in the developing grape berries of Shiraz and Chardonnay (Vitis vinifera L.). Aust. J. Grape Wine Res. 2003, 9, 110–121. [Google Scholar] [CrossRef]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects-A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Flamini, R.; Mattivi, F.; de Rosso, M.; Arapitsas, P.; Bavaresco, L. Advanced knowledge of three important classes of grape phenolics: Anthocyanins, stilbenes and flavonols. Int. J. Mol. Sci. 2013, 14, 19651–19669. [Google Scholar] [CrossRef]
- Rodrigo, R.; Miranda, A.; Vergara, L. Modulation of endogenous antioxidant system by wine polyphenols in human disease. Clin. Chim. Acta 2011, 412, 410–424. [Google Scholar] [CrossRef]
- Downey, M.O.; Dokoozlian, N.K.; Krstic, M.P. Cultural practice and environmental impacts on the flavonoid composition of grapes and wine: A review of recent research. Am. J. Enol. Vitic. 2006, 57, 257–268. [Google Scholar]
- Pirker, K.F.; Oliveira, J.; De Freitas, V.; Goodman, B.A.; Mateus, N. Antiradical properties of red wine portisins. J. Agric. Food Chem. 2011, 59, 11833–11837. [Google Scholar] [CrossRef]
- Stavíková, L.; Polovka, M.; Hohnová, B.; Karásek, P.; Roth, M. Antioxidant activity of grape skin aqueous extracts from pressurized hot water extraction combined with electron paramagnetic resonance spectroscopy. Talanta 2011, 85, 2233–2240. [Google Scholar] [CrossRef]
- Sivilotti, P.; Herrera, J.C.; Lisjak, K.; Baš;a Česnik, H.; Sabbatini, P.; Peterlunger, E.; Castellarin, S.D. Impact of leaf removal, applied before and after flowering, on anthocyanin, tannin, and methoxypyrazine concentrations in ‘Merlot’ (Vitis vinifera L.) grapes and wines. J. Agric. Food Chem. 2016, 64, 4487–4496. [Google Scholar] [CrossRef] [PubMed]
- Anić, M.; Osrečak, M.; Andabaka, Ž.; Tomaz, I.; Večenaj, Ž.; Jelić, D.; Kozina, B.; Kontić, J.K.; Karoglan, M. The effect of leaf removal on canopy microclimate, vine performance and grape phenolic composition of Merlot (Vitis vinifera L.) grapes in the continental part of Croatia. Sci. Hort. 2021, 285, 110161. [Google Scholar] [CrossRef]
- Dokoozlian, N.K.; Kliewer, W.M. Influence of light on grape berry growth and composition varies during fruit development. Am. J. Enol. Vitic. 1996, 5, 869–874. [Google Scholar] [CrossRef]
- Diago, M.P.; Ayestarán, B.; Guadalupe, Z.; Poni, S.; Tardáguila, J. Impact of prebloom and fruit set basal leaf removal on the flavonol and anthocyanin composition of Tempranillo grapes. Am. J. Enol. Vitic. 2012, 63, 367–376. [Google Scholar] [CrossRef]
- Mijowska, K.; Ochmian, I.; Oszmiánski, J. Impact of cluster zone leaf removal on grapes cv. Regent polyphenol content by the UPLC-PDA/MS method. Molecules 2016, 21, 1688. [Google Scholar] [CrossRef]
- de Oliveira, A.F.; Mercenaro, L.; Azzena, M.; Nieddu, G. Effects of pre- and post-veraison water deficit on Vermentino cluster microclimate and berry composition. BIO Web Conf. 2019, 13, 04015. [Google Scholar] [CrossRef][Green Version]
- Moreno, D.; Intrigliolo, D.S.; Vilanova, M.; Castel, J.R.; Gamero, E.; Valdés, E. Phenolic profile of grapevine cv. Tempranillo skins is affected by timing and severity of early defoliation. Span. J. Agric. Res. 2021, 19, e0905. [Google Scholar] [CrossRef]
- Bubola, M.; Lukic, I.; Radeka, S.; Sivilotti, P.; Grozic, K.; Vanzo, A.; Bavcar, D.; Lisjak, K. Enhancement of Istrian Malvasia wine aroma and hydroxycinnamate composition by hand and mechanical leaf removal. J. Sci. Food Agric. 2019, 99, 904–914. [Google Scholar] [CrossRef]
- Hickey, C.C.; Kwasniewski, M.T.; Wolf, T.K. Leaf removal effects on Cabernet franc and Petit Verdot: II. Grape carotenoids, phenolics and wine sensory analysis. Am. J. Enol. Vitic. 2018, 69, 231–246. [Google Scholar] [CrossRef]
- Sgherri, C.; Pinzino, C.; Quartacci, M.F. Antioxidant potential in lipophilic and hydrophilic extracts from medicinal herbs (Salvia officinalis and Echinacea angustifolia). A comparison between assays based on electron paramagnetic resonance and spectrophotometry. Am. J. Agric. Biol. Sci. 2012, 7, 417–424. [Google Scholar]
- Sgherri, C.; Pinzino, C.; Navari-Izzo, F.; Izzo, R. Contribution of major lipophilic antioxidants to the antioxidant activity of basil extracts: An EPR study. J. Sci. Food Agric. 2011, 91, 1128–1134. [Google Scholar] [CrossRef]
- Espín, J.C.; Soler-Rivas, C.; Wichers, H.J. Characterization of the total free radical scavenger capacity of vegetable oils and oil fractions using 2,2-diphenyl-1-picrylhydrazyl radical. J. Agric. Food Chem. 2000, 48, 648–656. [Google Scholar] [CrossRef]
- Lu, H.C.; Wang, Y.; Cheng, C.F.; Chen, W.; Li, S.D.; He, F.; Duan, C.Q.; Wang, J. Distal leaf removal made balanced source-sink vines, delayed ripening, and increased flavonol composition in Cabernet Sauvignon grapes and wines in the semi-arid Xinjiang. Food Chem. 2022, 366, 130582. [Google Scholar] [CrossRef]
- Gregan, S.M.; Wargent, J.J.; Liu, L.; Shinkle, J.; Hofmann, R.; Winefield, C.; Trought, M.; Jordan, B. Effects of solar ultraviolet radiation and canopy manipulation on the biochemical composition of Sauvignon Blanc grapes. Aust. J. Grape Wine Res. 2012, 18, 227–238. [Google Scholar] [CrossRef]
- Merzlyak, M.N.; Chivkunova, O.B.; Solovchenko, A.E.; Naqvi, K.R. Light absorption by anthocyanins in juvenile, stressed, and senescing leaves. J. Exp. Bot. 2008, 59, 3903–3911. [Google Scholar] [CrossRef]
- Rice-Evans, C.; Miller, N.J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
- Rescic, J.; Mikulic-Petkovsek, M.; Rusjan, D. The impact of canopy managements on grape and wine composition of cv. ‘Istrian Malvasia’ (Vitis vinifera L.). J. Sci. Food Agric. 2016, 96, 4724–4735. [Google Scholar] [CrossRef]
- Martínez-Lüscher, J.; Brillante, L.; Kurtural, S.K. Flavonol profile is a reliable indicator to assess canopy architecture and the exposure of red wine grapes to solar radiation. Front. Plant Sci. 2019, 10, 10. [Google Scholar] [CrossRef]
- Radovanović, B.; Radovanović, A. Free radical scavenging activity and anthocyanin profile of Cabernet Sauvignon wines from the Balkan region. Molecules 2010, 15, 4213–4226. [Google Scholar] [CrossRef]
- Pavic, V.; Kujundzic, T.; Kopic, M.; Jukic, V.; Braun, U.; Schwander, F.; Drenjancevic, M. Effects of defoliation on phenolic concentrations, antioxidant and antibacterial activity of grape skin extracts of the varieties Blaufrankisch and Merlot (Vitis vinifera L.). Molecules 2019, 24, 2444. [Google Scholar] [CrossRef]
- Harborne, J.B. Variation in and Functional Significance of Phenolic Conjugation in Plants. In Biochemistry of Plant Phenolics. Recent Advances in Phytochemistry; Swain, T., Harbone, J.B., Van Sumere, C.F., Eds.; Springer: Boston, MA, USA, 1979; Volume 12, pp. 457–474. [Google Scholar]
- Subba Rao, M.V.; Muralikrishna, G. Evaluation of the antioxidant properties of free and bound phenolic acids from native and malted finger millet (ragi, Eleusine coracana Indaf-15). J. Agric. Food Chem. 2002, 50, 889–892. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Anee, T.I.; Parvin, K.; Nahar, K.; Mahmud, J.A.; Fujita, M. Regulation of ascorbate-glutathione pathway in mitigating oxidative damage in plants under abiotic stress. Antioxidants 2019, 8, 384. [Google Scholar] [CrossRef]
- Xia, E.Q.; Deng, G.F.; Guo, Y.J.; Li, H.B. Biological activities of polyphenols from grapes. Int. J. Mol. Sci. 2010, 11, 622–646. [Google Scholar] [CrossRef]
- Arts, I.C.; Hollman, P.C. Polyphenols and disease risk in epidemiologic studies. Am. J. Clin. Nutr. 2005, 81, 317S–325S. [Google Scholar] [CrossRef]
- Wang, S.; Jiao, H.J.; Faust, M. Changes in ascorbate, glutathione and related enzyme activities during thidiazuron-induced bud break apple. Physiol. Plant. 1991, 82, 231–236. [Google Scholar] [CrossRef]
- Nguyen, P.M.; Niemeyer, E.D. Effect of nitrogen fertilization on the phenolic composition and antioxidant properties of basil (Ocimum basilicum L.). J. Agric. Food Chem. 2008, 56, 8685–8691. [Google Scholar] [CrossRef]
- Pellegrini, N.; Re, R.; Yang, M.; Rice-Evans, C. Screening of dietary carotenoids and carotenoid-rich fruit extracts for antioxidant activities applying 2,2’-azinobis (3-ethylenebenzothiazoline-6-sulfonic acid) radical cation decolorization assay. Methods Enzymol. 1999, 299, 379–389. [Google Scholar]
- Quartacci, M.F.; Sgherri, C.; Frisenda, S. Biochar amendment affects phenolic composition and antioxidant capacity restoring the nutraceutical value of lettuce grown in a copper-contaminated soil. Sci. Hort. 2017, 215, 9–14. [Google Scholar] [CrossRef]
- Duling, D.R. Simulation of multiple isotropic spin trap EPR spectra. J. Magn. Reson. 1994, 104, 105–110. [Google Scholar] [CrossRef]
| Phenolic Acid | Control | Defoliated |
|---|---|---|
| Protocatechuic acid | 0.5 ± 0.09 b | 2.2 ± 0.5 a |
| Chlorogenic acid | 320.0 ± 8.8 b | 771.1 ± 11.0 a |
| Vanillic acid | 1.6 ± 0.4 b | 11.6 ± 0.9 a |
| Syringic acid | 4.8 ± 0.9 b | 7.5 ± 0.2 a |
| p-Coumaric acid | 2.4 ± 0.5 a | 0.3 ± 0.03 b |
| Ferulic acid | 0.6 ± 0.1 b | 1.3 ± 0.3 a |
| Total | 329.9 ± 12.0 b | 794.0 ± 12.7 a |
| Phenolic Acid | Control | Defoliated |
|---|---|---|
| Gallic acid | 85.1 ± 3.1 b | 130.0 ± 4.3 a |
| Protocatechuic acid | 1.2 ± 0.1 a | 0.9 ± 0.1 a |
| p-Hydroxybenzoic acid | 5.4 ± 0.5 a | 5.2 ± 0.1 a |
| Caffeic acid | 952.1 ± 12.6 b | 1211.0 ± 15.9 a |
| Syringic acid | 16.2 ± 1.9 a | 15.5 ± 1.0 a |
| p-Coumaric acid | 392.0 ± 9.1 b | 596.1 ± 10.3 a |
| Ferulic acid | 0.7 ± 0.1 a | 0.2 ± 0.1 b |
| Total | 1452.7 ± 15.86 a | 1958.9 ± 20.58 b |
| Flavonol | Control | Defoliated |
|---|---|---|
| Quercetin glucuronide | 134.77 ± 14.17 b | 275.16 ± 1.20 a |
| Quercetin glucoside | 130.06 ± 19.64 b | 198.46 ± 3.76 a |
| Kaempferol galactoside | 9.96 ± 2.73 b | 19.04 ± 0.54 a |
| Kaempferol glucuronide | 3.71 ± 0.51 b | 8.64 ± 0.56 a |
| Kaempferol glucoside | 36.06 ± 7.33 b | 66.76 ± 1.61 a |
| Isorhamnetin galactoside | 2.00 ± 0.60 a | 2.41 ± 0.08 a |
| Isorhamnetin glucoside | 10.32 ± 3.06 a | 12.29 ± 0.50 a |
| Isorhamnetin glucuronide | 0.21 ± 0.11 b | 0.72 ± 0.07 a |
| Total | 327.09 ± 18.76 b | 583.48 ± 4.88 a |
| Decay Rate Constant | No. of Antioxidants | |||
|---|---|---|---|---|
| kF | kI | FRA | IRA | |
| Control | 7.47 ± 0.09 b | nd | 14.00 ± 0.71 b | nd |
| Defoliated | 10.82 ± 0.11 a | 0.32 ± 0.04 | 20.67 ± 0.92 a | 65.54 ± 1.25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quartacci, M.F.; Sgherri, C.; Pinzino, C. Impact of Leaf Removal on Phenolics and Antioxidant Activity of Trebbiano Berries (Vitis vinifera L.). Plants 2022, 11, 1303. https://doi.org/10.3390/plants11101303
Quartacci MF, Sgherri C, Pinzino C. Impact of Leaf Removal on Phenolics and Antioxidant Activity of Trebbiano Berries (Vitis vinifera L.). Plants. 2022; 11(10):1303. https://doi.org/10.3390/plants11101303
Chicago/Turabian StyleQuartacci, Mike Frank, Cristina Sgherri, and Calogero Pinzino. 2022. "Impact of Leaf Removal on Phenolics and Antioxidant Activity of Trebbiano Berries (Vitis vinifera L.)" Plants 11, no. 10: 1303. https://doi.org/10.3390/plants11101303
APA StyleQuartacci, M. F., Sgherri, C., & Pinzino, C. (2022). Impact of Leaf Removal on Phenolics and Antioxidant Activity of Trebbiano Berries (Vitis vinifera L.). Plants, 11(10), 1303. https://doi.org/10.3390/plants11101303






