Antcins from Antrodia cinnamomea and Antrodia salmonea Inhibit Angiotensin-Converting Enzyme 2 (ACE2) in Epithelial Cells: Can Be Potential Candidates for the Development of SARS-CoV-2 Prophylactic Agents
Abstract
:1. Introduction
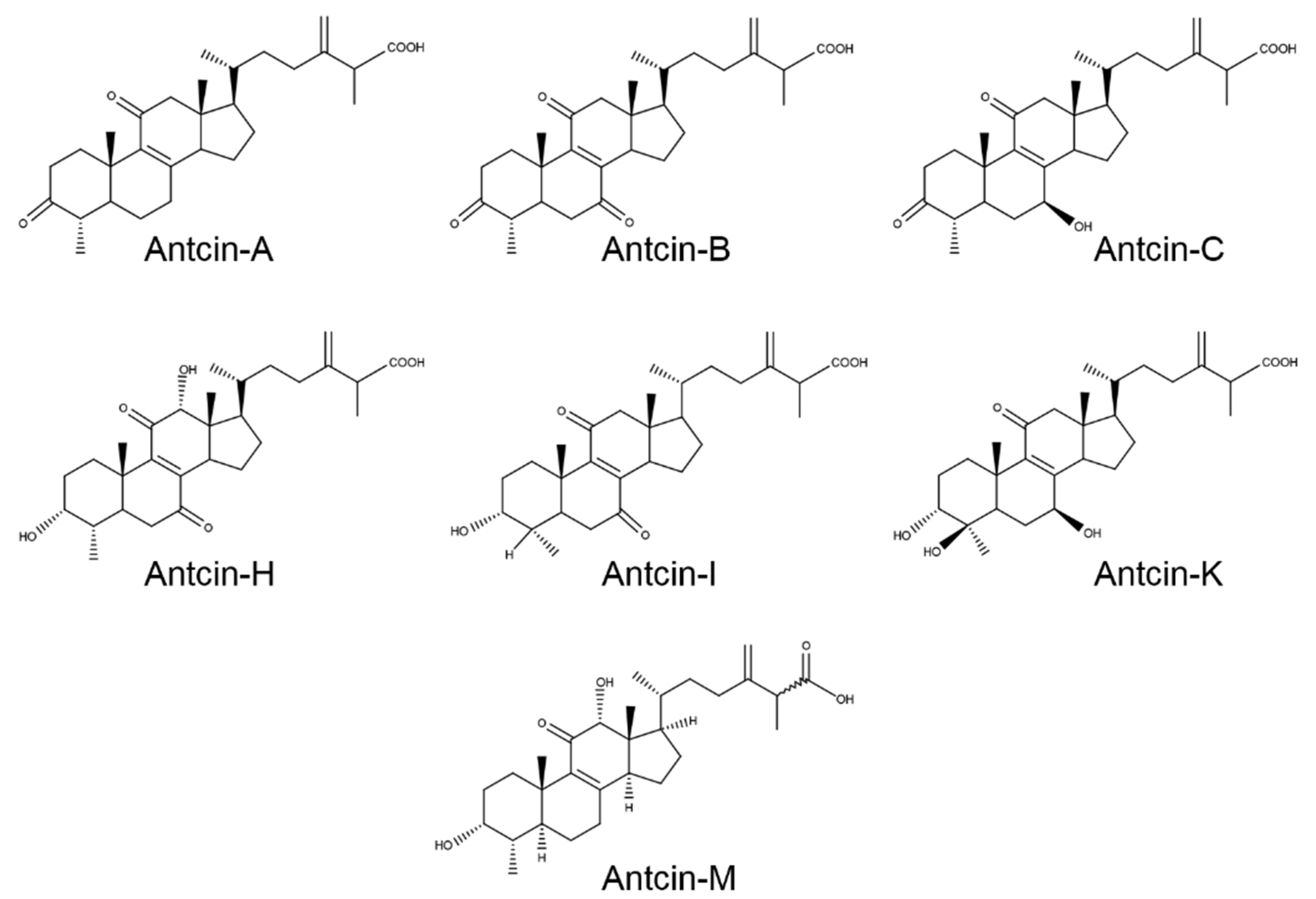
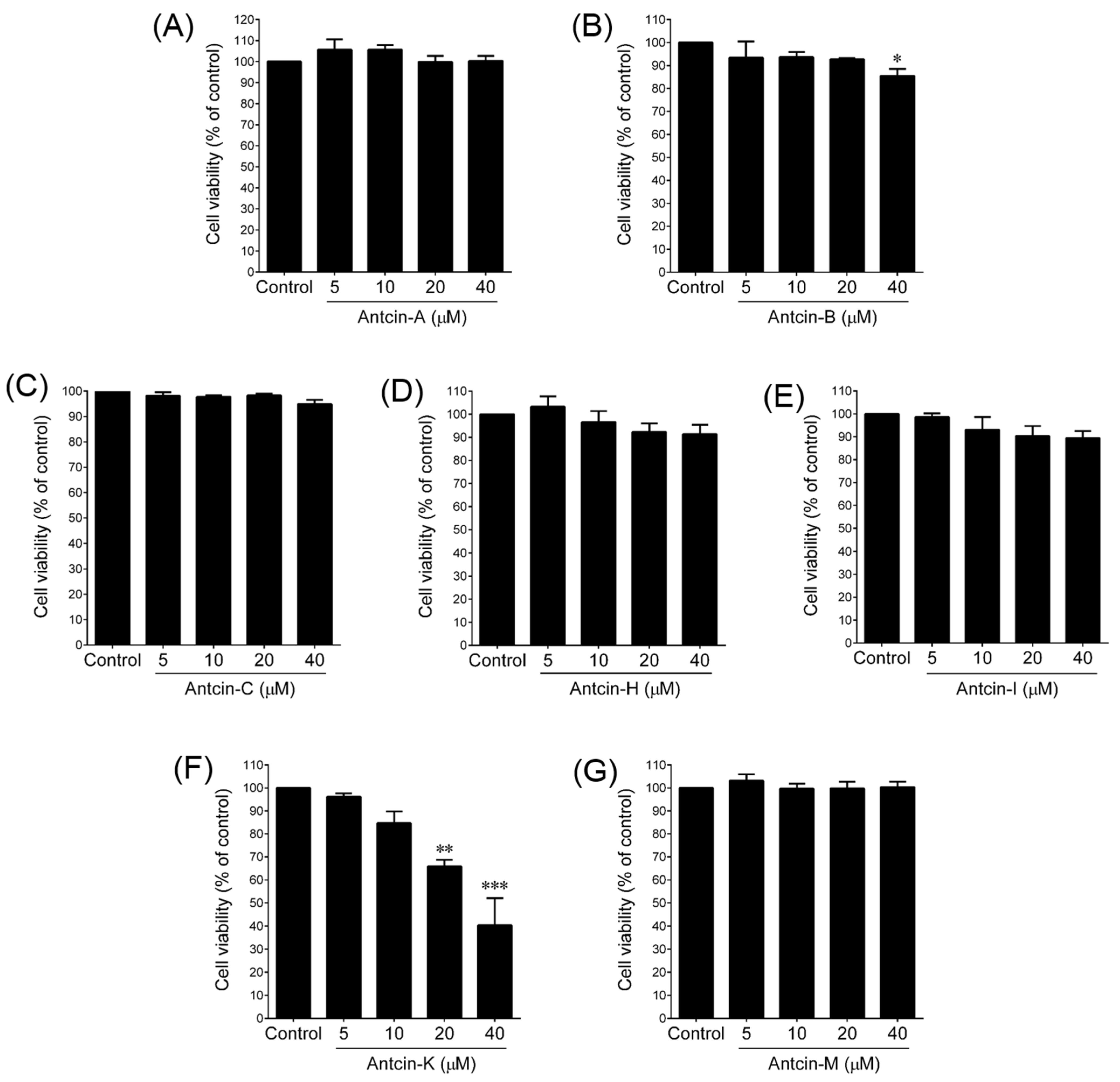
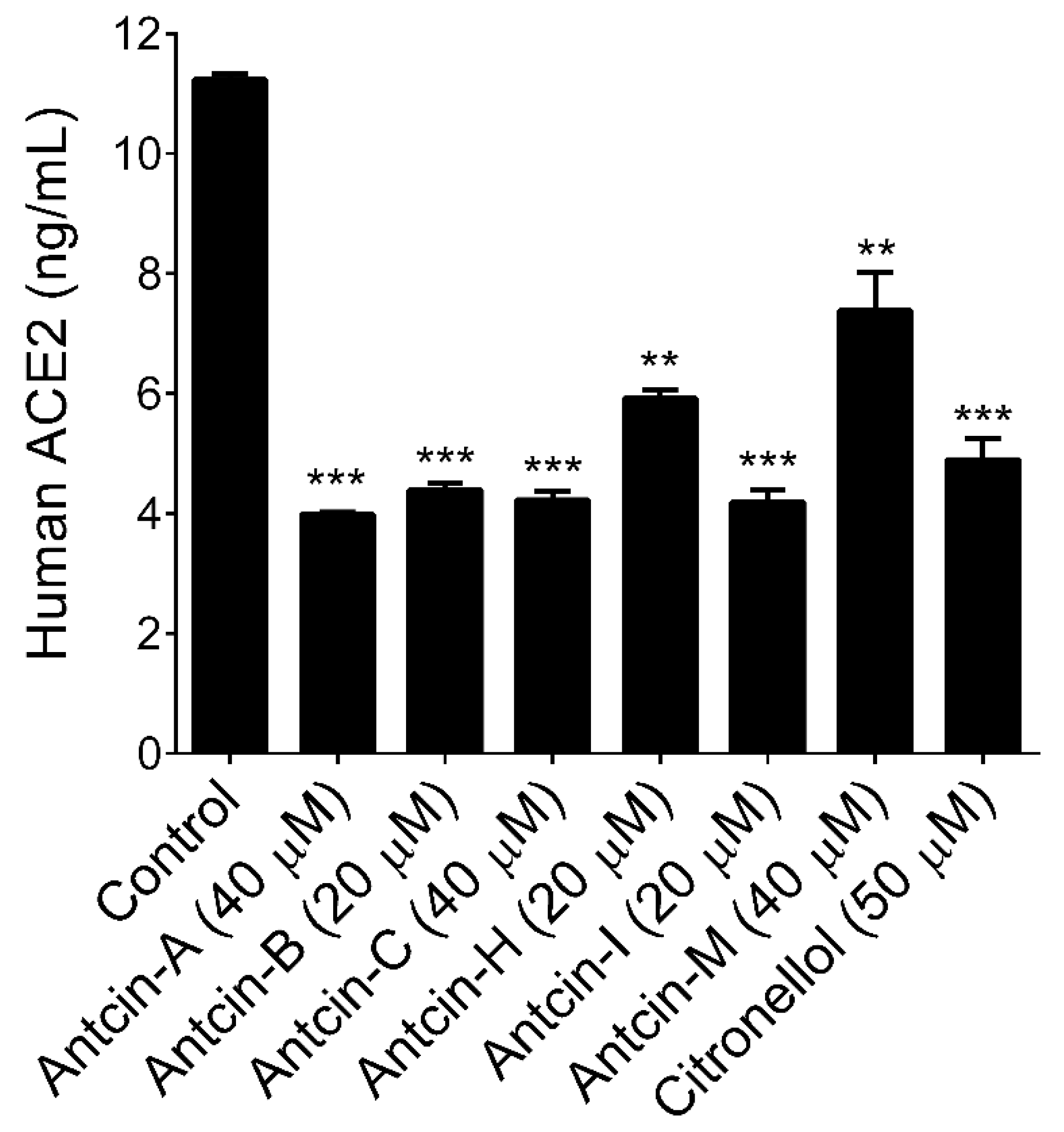
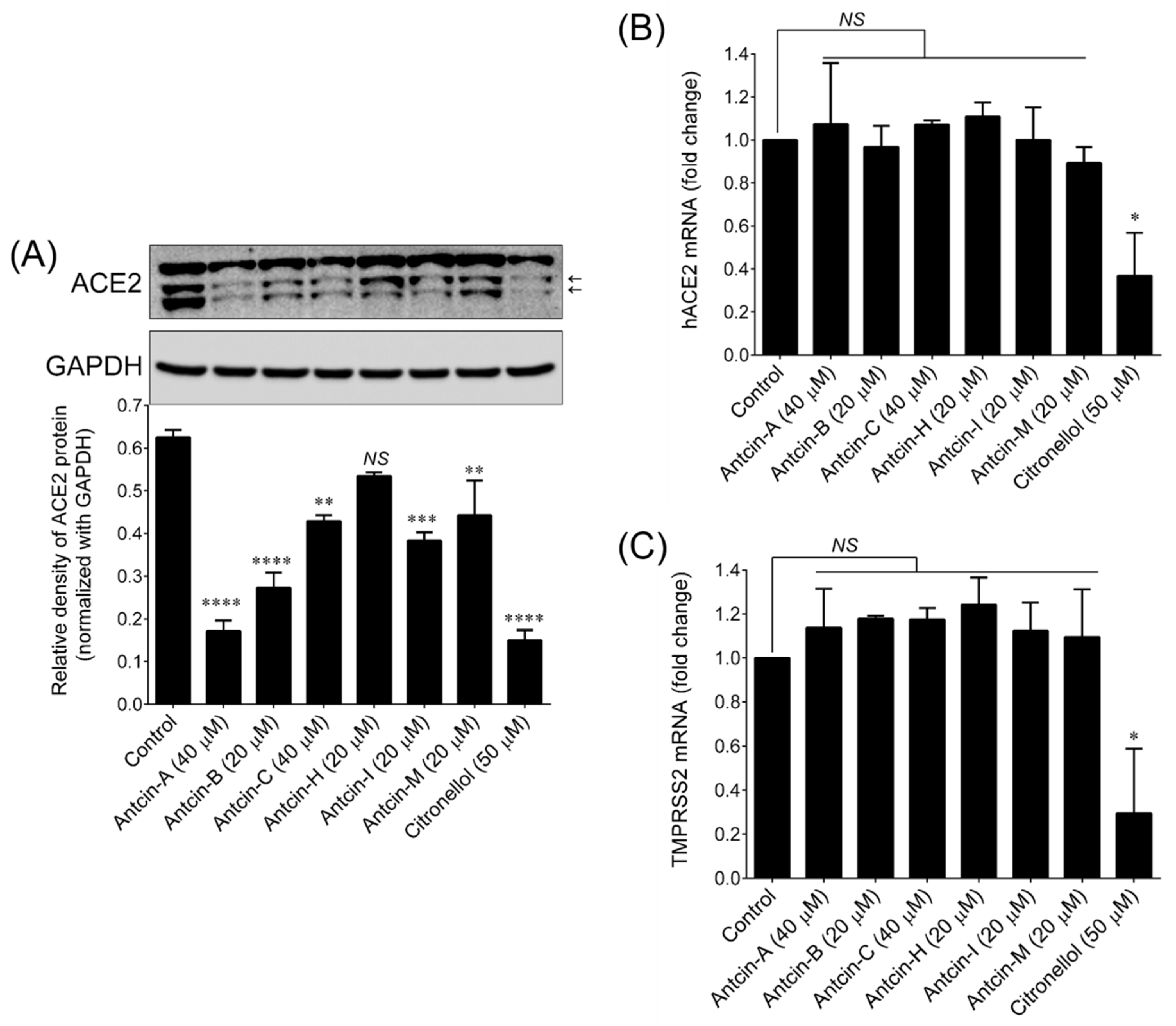
2. Results and Discussion
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Cell Culture and Sample Treatment
3.3. Cell Viability Assay
3.4. Determination of ACE2 Activity
3.5. Determination of ACE2 Protein
3.6. Quantitative Real-Time PCR
3.7. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Sample Availability
References
- Zaim, S.; Chong, J.H.; Sankaranarayanan, V.; Harky, A. COVID-19 and multiorgan response. Curr. Probl. Cardiol. 2020, 45, 100618. [Google Scholar] [CrossRef]
- Hsu, J. Covid-19: What now for remdesivir? BMJ 2020, 371, m4457. [Google Scholar] [CrossRef]
- Kumar, S.; Saxena, S.K. Structural and molecular perspectives of SARS-CoV-2. Methods 2021. [Google Scholar] [CrossRef]
- Lan, J.; Ge, J.; Yu, J.; Shan, S.; Zhou, H.; Fan, S.; Zhang, Q.; Shi, X.; Wang, Q.; Zhang, L.; et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 2020, 581, 215–220. [Google Scholar] [CrossRef] [Green Version]
- Ganjhu, R.K.; Mudgal, P.P.; Maity, H.; Dowarha, D.; Devadiga, S.; Nag, S.; Arunkumar, G. Herbal plants and plant preparations as remedial approach for viral diseases. Virus Dis. 2015, 26, 225–236. [Google Scholar] [CrossRef]
- Lin, L.T.; Hsu, W.C.; Lin, C.C. Antiviral natural products and herbal medicines. J. Trad. Complemet Med. 2014, 4, 24–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020, 181, 271–280.e278. [Google Scholar] [CrossRef] [PubMed]
- Cheke, R.S.; Narkhede, R.R.; Shinde, S.D.; Ambhore, J.P.; Jain, P.G. Natural product emerging as potential SARS spike glycoproteins-ACE2 inhibitors to combat COVID-19 attributed by in-silico investigations. Biointerface Res. Appl. Chem. 2021, 11, 10628–10639. [Google Scholar]
- Abubakar, M.B.; Usman, D.; El-Saber Batiha, G.; Cruz-Martins, N.; Malami, I.; Ibrahim, K.G.; Abubakar, B.; Bello, M.B.; Muhammad, A.; Gan, S.H.; et al. Natural products modulating angiotensin converting enzyme 2 (ACE2) as potential COVID-19 therapies. Front. Pharmacol. 2021, 12, 629935. [Google Scholar] [CrossRef] [PubMed]
- Joshi, G.; Sindhu, J.; Thakur, S.; Rana, A.; Sharma, G.; Mayank; Poduri, R. Recent efforts for drug identification from phytochemicals against SARS-CoV-2: Exploration of the chemical space to identify druggable leads. Food Chem. Toxicol. 2021, 152, 112160. [Google Scholar] [CrossRef]
- Antonio, A.d.S.; Wiedemann, L.S.M.; Veiga-Junior, V.F. Natural products’ role against COVID-19. RSC Adv. 2020, 10, 23379–23393. [Google Scholar] [CrossRef]
- Chiu, H.H. Phylogenetic analysis of Antrodia species and Antrodia camphorata inferred from internal transcribed spacer region. Antonie Leeuwenhoek 2007, 91, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.C.; El-Shazly, M.; Wu, T.Y.; Du, Y.C.; Chang, T.T.; Chen, C.F.; Hsu, Y.M.; Lai, K.H.; Chiu, C.P.; Chang, F.R.; et al. Recent research and development of Antrodia cinnamomea. Pharmacol. Ther. 2013, 139, 124–156. [Google Scholar] [CrossRef] [PubMed]
- Senthil Kumar, K.J.; Gokila Vani, M.; Chen, C.Y.; Hsiao, W.W.; Li, J.; Lin, Z.X.; Chu, F.H.; Yen, G.C.; Wang, S.Y. A mechanistic and empirical review of antcins, a new class of phytosterols of formosan fungi origin. J. Food Drug Anal. 2020, 28, 38–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.Y.; Chien, S.C.; Tsao, N.W.; Lai, C.S.; Wang, Y.Y.; Hsiao, W.W.; Chu, F.H.; Kuo, Y.H.; Wang, S.Y. Metabolite profiling and comparison of bioactivity in Antrodia cinnamomea and Antrodia salmonea fruiting bodies. Planta Med. 2016, 82, 244–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arquimedes Gasparotto, J.; Sara Emília Lima, T.; Francislaine Aparecida dos Reis, L.; Francielli, G.; Thaise, B.; Priscila de, S. Natural agents modulating ACE-2: A review of compounds with potential against SARS-CoV-2 infections. Curr. Pharm. Design 2021, 27, 1588–1596. [Google Scholar]
- Gopinath, K.; Jokinen, E.M.; Kurkinen, S.T.; Pentikäinen, O.T. Screening of natural products targeting SARS-CoV-2–ACE2 receptor interface – a mixmd based HTVS pipeline. Front. Chem. 2020, 8, 589769. [Google Scholar] [CrossRef]
- Muchtaridi, M.; Fauzi, M.; Khairul Ikram, N.K.; Mohd Gazzali, A.; Wahab, H.A. Natural flavonoids as potential angiotensin-converting enzyme 2 inhibitors for anti-SARS-CoV-2. Molecules 2020, 25, 3980. [Google Scholar] [CrossRef]
- Verma, S.; Twilley, D.; Esmear, T.; Oosthuizen, C.B.; Reid, A.-M.; Nel, M.; Lall, N. Anti-SARS-CoV natural products with the potential to inhibit SARS-CoV-2 (COVID-19). Front. Pharmacol. 2020, 11, 561334. [Google Scholar] [CrossRef]
- Lin, C.; Li, Y.; Zhang, Y.; Liu, Z.; Mu, X.; Gu, C.; Liu, J.; Li, Y.; Li, G.; Chen, J. Ceftazidime is a potential drug to inhibit SARS-CoV-2 infection in vitro by blocking spike protein–ACE2 interaction. Signal Transduct. Target. Ther. 2021, 6, 198. [Google Scholar] [CrossRef]
- Ivanov, V.; Goc, A.; Ivanova, S.; Niedzwiecki, A.; Rath, M. Inhibition of ACE2 expression by ascorbic acid alone and its combinations with other natural compounds. Infect. Dis. 2021, 14, 1178633721994605. [Google Scholar]
- Wu, C.Y.; Lin, Y.S.; Yang, Y.H.; Shu, L.H.; Cheng, Y.C.; Liu, H.T. Gb-2 inhibits ACE2 and TMPRSS2 expression: In vivo and in vitro studies. Biomed. Pharmacother. 2020, 132, 110816. [Google Scholar] [CrossRef] [PubMed]
- Warner, F.J.; Lew, R.A.; Smith, A.I.; Lambert, D.W.; Hooper, N.M.; Turner, A.J. Angiotensin-converting enzyme 2 (ACE2), but not ace, is preferentially localized to the apical surface of polarized kidney cells. J. Biol. Chem. 2005, 280, 39353–39362. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Zhong, L.; Deng, J.; Peng, J.; Dan, H.; Zeng, X.; Li, T.; Chen, Q. High expression of ACE2 receptor of 2019-nCOV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xuan, B.; Yan, Y.; Zhu, X.; Shen, C.; Zhao, G.; Ji, L.; Xu, D.; Xiong, H.; Yu, T.; et al. Profiling ACE2 expression in colon tissue of healthy adults and colorectal cancer patients by single-cell transcriptome analysis. MedRxiv 2020, 2020, 20023457. [Google Scholar]
- Vardhan, S.; Sahoo, S.K. In silico admet and molecular docking study on searching potential inhibitors from limonoids and triterpenoids for COVID-19. Comput. Biol. Med. 2020, 124, 103936. [Google Scholar] [CrossRef]
- Carino, A.; Moraca, F.; Fiorillo, B.; Marchianò, S.; Sepe, V.; Biagioli, M.; Finamore, C.; Bozza, S.; Francisci, D.; Distrutti, E.; et al. Hijacking SARS-CoV-2/ACE2 receptor interaction by natural and semi-synthetic steroidal agents acting on functional pockets on the receptor binding domain. Front. Chem. 2020, 8, 573885. [Google Scholar] [CrossRef]
- Li, H.; Cheng, C.; Li, S.; Wu, Y.; Liu, Z.; Liu, M.; Chen, J.; Zhong, Q.; Zhang, X.; Liu, S.; et al. Discovery and structural optimization of 3-o-β-chacotriosyl oleanane-type triterpenoids as potent entry inhibitors of SARS-CoV-2 virus infections. Eur. J. Med. Chem. 2021, 215, 113242. [Google Scholar] [CrossRef]
- Chen, Y.C.; Liu, Y.L.; Li, F.Y.; Chang, C.I.; Wang, S.Y.; Lee, K.Y.; Li, S.L.; Chen, Y.P.; Jinn, T.R.; Tzen, J.T. Antcin a, a steroid-like compound from Antrodia camphorata, exerts anti-inflammatory effect via mimicking glucocorticoids. Acta Pharmacol. Sin 2011, 32, 904–911. [Google Scholar] [CrossRef]
- Senthil Kumar, K.J.; Gokila Vani, M.; Wang, C.S.; Chen, C.C.; Chen, Y.C.; Lu, L.P.; Huang, C.H.; Lai, C.S.; Wang, S.Y. Geranium and lemon essential oils and their active compounds downregulate angiotensin-converting enzyme 2 (ACE2), a SARS-CoV-2 spike receptor-binding domain, in epithelial cells. Plants 2020, 9, 770. [Google Scholar] [CrossRef]
- Wang, G.Z.; Zhao, Q.; Liang, F.; Zhang, C.; Zhang, H.; Wang, J.; Chen, Z.Y.; Wu, R.; Yu, H.; Sun, B.B.; et al. Degradation of SARS-CoV-2 receptor ACE2 by tobacco carcinogen-induced Skp2 in lung epithelial cells. Front. Med. 2020, 15, 253–263. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Senthil Kumar, K.J.; Gokila Vani, M.; Hsieh, H.-W.; Lin, C.-C.; Wang, S.-Y. Antcins from Antrodia cinnamomea and Antrodia salmonea Inhibit Angiotensin-Converting Enzyme 2 (ACE2) in Epithelial Cells: Can Be Potential Candidates for the Development of SARS-CoV-2 Prophylactic Agents. Plants 2021, 10, 1736. https://doi.org/10.3390/plants10081736
Senthil Kumar KJ, Gokila Vani M, Hsieh H-W, Lin C-C, Wang S-Y. Antcins from Antrodia cinnamomea and Antrodia salmonea Inhibit Angiotensin-Converting Enzyme 2 (ACE2) in Epithelial Cells: Can Be Potential Candidates for the Development of SARS-CoV-2 Prophylactic Agents. Plants. 2021; 10(8):1736. https://doi.org/10.3390/plants10081736
Chicago/Turabian StyleSenthil Kumar, K. J., M. Gokila Vani, Han-Wen Hsieh, Chin-Chung Lin, and Sheng-Yang Wang. 2021. "Antcins from Antrodia cinnamomea and Antrodia salmonea Inhibit Angiotensin-Converting Enzyme 2 (ACE2) in Epithelial Cells: Can Be Potential Candidates for the Development of SARS-CoV-2 Prophylactic Agents" Plants 10, no. 8: 1736. https://doi.org/10.3390/plants10081736
APA StyleSenthil Kumar, K. J., Gokila Vani, M., Hsieh, H.-W., Lin, C.-C., & Wang, S.-Y. (2021). Antcins from Antrodia cinnamomea and Antrodia salmonea Inhibit Angiotensin-Converting Enzyme 2 (ACE2) in Epithelial Cells: Can Be Potential Candidates for the Development of SARS-CoV-2 Prophylactic Agents. Plants, 10(8), 1736. https://doi.org/10.3390/plants10081736







