The First Evidence of the Beneficial Effects of Se-Supplementation on In Vitro Cultivated Olive Tree Explants
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Plant Material and Growing Conditions
4.2. Se Treatment
- -
- viability (%): incidence of green and viable explants;
- -
- shoots (n): average number of shoots developed by each initial explant;
- -
- shoot length (mm): average length of developed shoots;
- -
- nodes (n): average number of nodes developed by each initial explant, reusable for further proliferation subculture (multiplication rate);
- -
- callus (%): incidence of explants that produced basal callus;
- -
- green fresh weight (mg): average fresh weight of developed vegetative organs (leaves, stems, buds);
- -
- callus fresh weight (mg): average fresh weight of callus masses that may have developed at the base of explants;
- -
- total dry weight (mg): average dry weight of the vegetative organs and callus, obtained by keeping the plant material in an oven for three days at 105 °C;
- -
- dry matter (%): average incidence of dry matter on total fresh weight, calculated for each comparison (calculated values).
4.3. Total Se Content of the Proliferated Explants
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Birringer, M.; Pilawa, S.; Flohé, L. Trends in selenium biochemistry. Nat. Prod. Rep. 2002, 19, 693–718. [Google Scholar] [CrossRef] [PubMed]
- Germ, L.M.; Stibilj, V.; Kreft, I. Metabolic importance of selenium for plants. Eur. J. Plant Sci. Biotechnol. 2007, 1, 91–97. [Google Scholar]
- Zhu, Y.G.; Pilon-Smits, E.A.H.; Zhao, F.J.; Williams, P.N.; Meharg, A.A. Selenium in higher plants: Understanding mechanisms for biofortification and phytoremediation. Trends Plant Sci. 2009, 14, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, S.J. Review of selenium toxicity in the aquatic food chain. Sci. Total Environ. 2004, 326, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, R.; Nasim, S.A.; Dhir, B.; Mujib, A. Selenium treatment alters phytochemical and biochemical activity of in vitro-grown tissues and organs of Allium sativum L. In Vitro Cell. Dev. Biol. Plant 2012, 48, 411–416. [Google Scholar] [CrossRef]
- Hartikainen, H.; Xue, T.; Piironen, V. Selenium as an antioxidant and prooxidant in ryegrass. Plant Soil 2000, 225, 193–200. [Google Scholar] [CrossRef]
- El-Ramady, H.; Abdalla, N.; Taha, H.S.; Alshaal, T.; El Henawy, A.; Salah, E.D.F.; Shams, M.S.; Youssef, S.M.; Shalaby, T.; Bayoumi, Y. Selenium and nano-selenium in plant nutrition. Environ. Chem. Lett. 2016, 14, 123–147. [Google Scholar] [CrossRef]
- Sors, T.G.; Ellis, D.R.; Gun, N.N.; Lahner, B.; Lee, S.; Leustek, T.; Pickering, I.J.; Salt, D.E. Analysis of sulfur and selenium assimilation in Astragalus plants with varying capacities to accumulate selenium. Plant J. 2005, 42, 785–797. [Google Scholar] [CrossRef]
- Iwaoka, M.; Ooka, R.; Nakazato, T.; Yoshida, S.; Oishi, S. Synthesis of selenocysteine and selenomethionine derivatives from sulfur-containing aminoacids. Chem. Biodivers. 2008, 5, 359–374. [Google Scholar] [CrossRef]
- Zhou, X.; Yang, J.; Kronzucker, H.J.; Shi, W. Selenium Biofortification and Interaction with Other Elements in Plants: A Review. Front. Plant Sci. 2020, 11, 586421. [Google Scholar] [CrossRef]
- Kuznetsov, V.; Kholodova, V.; Kuznetsov, V.; Yagodin, B.A. Selenium regulates the water status of plants exposed to drought. Dokl. Biol. Sci. 2003, 390, 266–268. [Google Scholar] [CrossRef]
- Malerba, M.; Cerana, R. Effect of Selenium on the Responses Induced by Heat Stress in Plant Cell Cultures. Plants 2018, 7, 64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pezzarossa, B.; Remorini, D.; Gentile, M.L.; Massai, R. Effects of foliar and fruit addition of sodium selenate on selenium accumulation and fruit quality. J. Sci. Food Agric. 2012, 92, 781–786. [Google Scholar] [CrossRef]
- Proietti, P.; Nasini, L.; Del Buono, D.; D’Amato, R.; Tedeschini, E.; Businelli, D. Se protects olive (Olea europaea L.) from drought stress. Sci. Hortic. 2013, 164, 165–171. [Google Scholar] [CrossRef]
- D’Amato, R.; De Feudis, M.; Hasuoka, P.E.; Regni, L.; Pacheco, P.H.; Onofri, A.; Businelli, D.; Proietti, P. The selenium supplementation influences olive tree production and oil stability against oxidation and can alleviate the water deficiency effects. Front. Plant Sci. 2018, 9, 1191. [Google Scholar] [CrossRef] [PubMed]
- Regni, L.; Palmerini, C.A.; Del Pino, A.M.; Businelli, D.; D’Amato, R.; Mairech, H.; Marmottini, F.; Micheli, M.; Pacheco, P.H.; Proietti, P. Effects of selenium supplementation on olive under salt stress conditions. Sci. Hortic. 2021, 278, 109866. [Google Scholar] [CrossRef]
- D’Amato, R.; Proietti, P.; Nasini, L.; Del Buono, D.; Tedeschini, E.; Businelli, D. Increase in the selenium content of extra virgin olive oil: Quantitative and qualitative implications. Grasas Aceites 2014, 65, e025. [Google Scholar]
- D’Amato, R.; Proietti, P.; Onofri, A.; Regni, L.; Esposto, S.; Servili, M.; Businelli, D.; Selvaggini, R. Biofortification (Se): Does it increase the content of phenolic compounds in virgin olive oil (VOO)? PLoS ONE 2017, 12, e0176580. [Google Scholar] [CrossRef] [PubMed]
- Tedeschini, E.; Proietti, P.; Timorato, V.; D’Amato, R.; Nasini, L.; Del Buono, D.; Frenguelli, G. Selenium as stressor and antioxidant affects pollen performance in Olea europaea. Flora Morphol. Distrib. Funct. Ecol. Plants 2015, 215, 16–22. [Google Scholar] [CrossRef] [Green Version]
- Del Pino, A.M.; Regni, L.; D’Amato, R.; Tedeschini, E.; Businelli, D.; Proietti, P.; Palmerini, C.A. Selenium-enriched pollen grains of Olea europaea L.: Ca2+ signalling and germination under oxidative stress. Front. Plant Sci. 2019, 10, 1611. [Google Scholar] [CrossRef]
- D’Amato, R.; Petrelli, M.; Proietti, P.; Onofri, A.; Regni, L.; Perugini, D.; Businelli, D. Determination of changes in the concentration and distribution of elements within olive drupes (cv. Leccino) from Se biofortified plants, using laser ablation inductively coupled plasma mass spectrometry. J. Sci. Food Agric. 2018, 98, 4971–4977. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, R.; Regni, L.; Falcinelli, B.; Mattioli, S.; Benincasa, P.; Dal Bosco, A.; Pacheco, P.; Proietti, P.; Troni, E.; Santi, C.; et al. Current Knowledge on Selenium Biofortification to Improve the Nutraceutical Profile of Food: A Comprehensive Review. J. Agric. Food Chem. 2020, 68, 4075–4097. [Google Scholar] [CrossRef]
- Kavand, S.; Kermani, M.J.; Haghnazari, A.; Khosravi, P.; Azimi, M.R. Micropropagation and medium-term conservation of Rosa pulverulenta. Acta Sci. Agron. 2011, 33, 297–301. [Google Scholar]
- Balla, I.; Mansvelt, L. Micropropagation of peach rootstocks and cultivars. Methods Mol. Biol. 2013, 11013, 137–148. [Google Scholar]
- Dobránszki, J.; Silva, J.A. Micropropagation of apple—A review. Biotechnol. Adv. 2010, 28, 462–488. [Google Scholar] [CrossRef]
- Mangal, M.; Sharma, D.; Sharma, M.; Kumar, S. In vitro regeneration in olive (Olea europaea L.), cv “Frantoio” from nodal segments. Indian J. Expt. Biol. 2014, 52, 12–916. [Google Scholar]
- Micheli, M.; El Behi, A.W.; Zakhour, D.; Yasin, M.; Standardi, A. In vitro proliferation of olive (‘Dolce Agogia’ and “Moraiolo”): Effect of different cytokinins. Acta Hort. 2010, 884, 587–590. [Google Scholar] [CrossRef]
- Breznik, B.; Germ, M.; Gaberščik, A.; Kreft, I. Combined effects of elevated UV-B radiation and the addition of selenium on common (Fagopyrum esculentum Moench) and tartary [Fagopyrum tataricum (L.) Gaertn.] buckwheat. Photosynthetica 2004, 21, 59–64. [Google Scholar] [CrossRef]
- Van Hoewyk, D. A tale of two toxicities: Malformed selenoproteins and oxidative stress both contribute to selenium stress in plants. Ann. Bot. 2013, 112, 965–972. [Google Scholar] [CrossRef] [Green Version]
- Dimkovikj, A.; Fisher, B.; Hutchison, K.; Van Hoewyk, D. Stuck between a ROS and a hard place: Analysis of the ubiquitin proteasome pathway in selenocysteine treated Brassica napus reveals different toxicities during selenium assimilation. J. Plant Physiol. 2015, 18, 50–54. [Google Scholar] [CrossRef]
- Van Hoewyk, D. Defects in endoplasmic reticulum-associated degradation (ERAD) increase selenate sensitivity in Arabidopsis. Plant. Signal. Behav. 2016, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolbert, Z.; Lehotai, N.; Molnár, A.; Feigl, G. “The roots” of selenium toxicity: A new concept. Plant. Signal. Behav. 2016, 11, e124193. [Google Scholar] [CrossRef] [Green Version]
- Fritz, W.P.; Spracklen, R.E.; Spiby, M.C.; Meacham, A.; Mead, M.; Harriman, L.J.; Trueman, B.M.; Smith, B.; Thomas, M.R. Interactions between selenium and sulphur nutrition in Arabidopsis thaliana. J. Exp. Bot. 2004, 55, 1927–1937. [Google Scholar]
- Wrobel, K.; Esperanza, M.G.; Barrientos, E.Y.; Escobosa, A.R.C.; Wrobel, K. Different approaches in metabolomic analysis of plants exposed to selenium: A comprehensive review. Acta Physiol. Plant. 2020, 42, 125. [Google Scholar] [CrossRef]
- Gupta, M.; Gupta, S. An Overview of Selenium Uptake, Metabolism, and Toxicity in Plants. Front. Plant Sci. 2017, 7, 2074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winkel, L.H.E.; Vriens, B.; Jones, G.D.; Schneider, L.S.; Pilon-Smits, E.A.H.; Banuelos, G.S. Selenium cycling across soil–plant–atmosphere interfaces: A critical review. Nutrients 2015, 7, 4199–4239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Germ, M.; Kreft, I.; Stibilj, V.; Urbanc-Berčič, O. Combined effects of selenium and drought on photosynthesis and mitochondrial respiration in potato. Plant. Physiol. Biochem. 2007, 45, 162–167. [Google Scholar] [CrossRef]
- Garcia-Ferrìz, L.; Ghorbel, R.; Ybarra, M.; Mari, A.; Belay, A.; Trujillo, I. Micropropagation of olive mature trees. Acta Hort. 2002, 586, 879–882. [Google Scholar] [CrossRef]
- Sgir, S.; Chatelet, P.; Ouazzani, N.; Dosba, F.; Belkoura, I. Micropropagation of eight moroccan and french olive cultivars. Hortscience 2005, 40, 193–196. [Google Scholar] [CrossRef]
- Ali, A.; Ahmad, T.; Abbasi, N.A.; Hafiz, I.A. Effect of different media and growth regulators on in vitro shoot proliferation of olive cultivar “Moraiolo”. Pak. J. Bot. 2009, 41, 783–795. [Google Scholar]
- Mendoza-de Gyves, E.; Mira, F.R.; Ruiu, F.; Rugini, E. Stimulation of node and lateral shoot formation in micropropagation of olive (Olea europaea L.) by using dikegulac. Plant Cell Tissue Organ. Cult. 2008, 92, 233–238. [Google Scholar] [CrossRef]
- Peixe, A.; Raposo, A.; Lourenco, R.; Cardoso, H.; Macedo, A. Coconut water and BAP successfully replaced zeatin in olive (Olea europaea L.) micropropagation. Sci. Hort. 2007, 113, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Demo, P.; Kuria, P.; Nyende, A.B.; Kahangi, E.M. Table sugar as an alternative low cost medium component for in vitro micropropagation of potato. Afr. J. Biotechnol. 2008, 7, 2578–2584. [Google Scholar]
- Rugini, E. In vitro propagation of some olive (Olea europea L.) cultivars with different root-ability, and medium development using analytic data from developing shoots and embryos. Sci. Hort. 1984, 24, 123–134. [Google Scholar] [CrossRef]
- US EPA Method 3052 Microwave Assisted Acid Digestion of Siliceous and Organically Based Matrices. Available online: https://www.epa.gov/hw-sw846/sw-846-test-method-3052-microwave-assisted-acid-digestion-siliceous-and-organically-based (accessed on 24 April 2021).
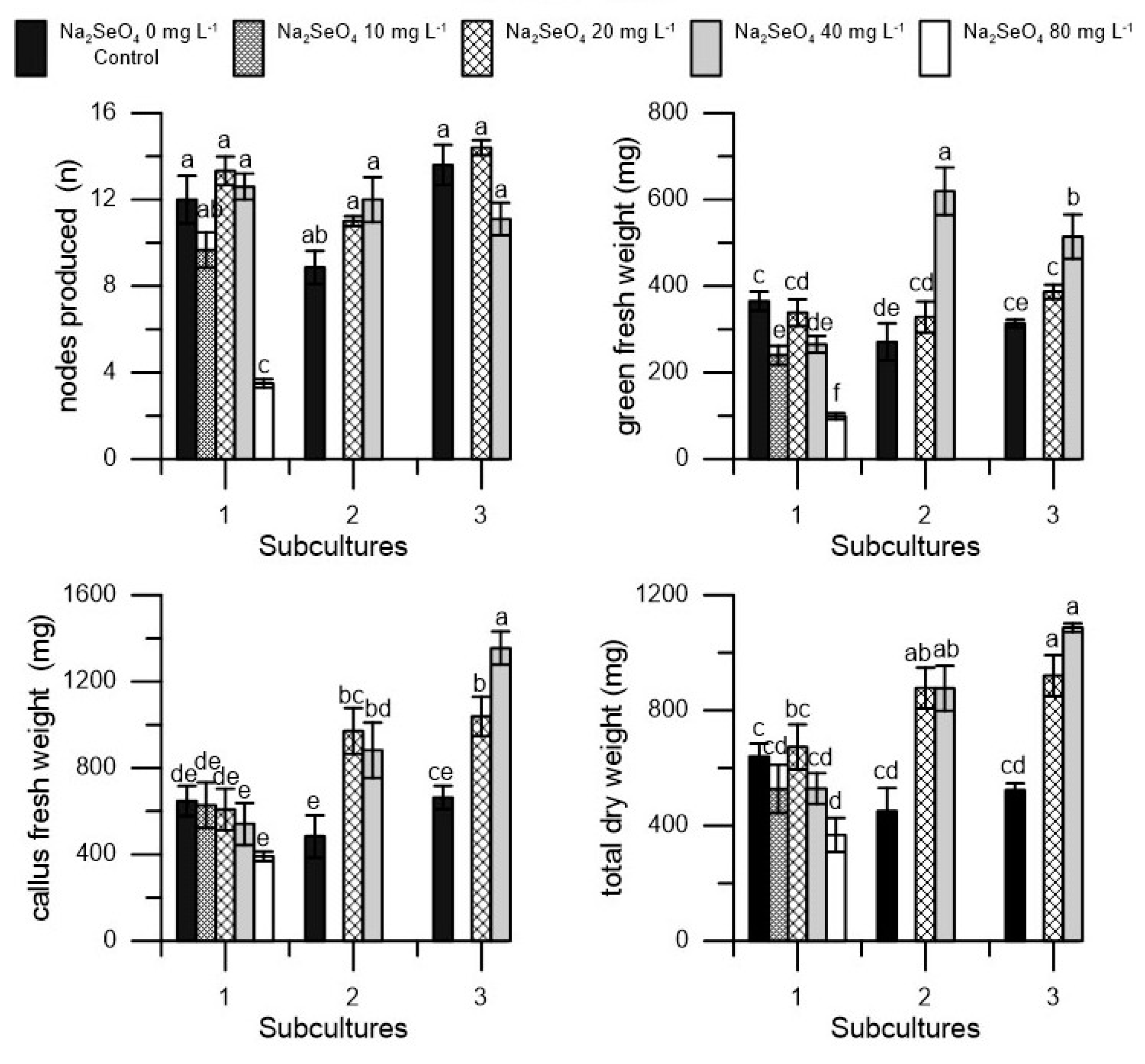
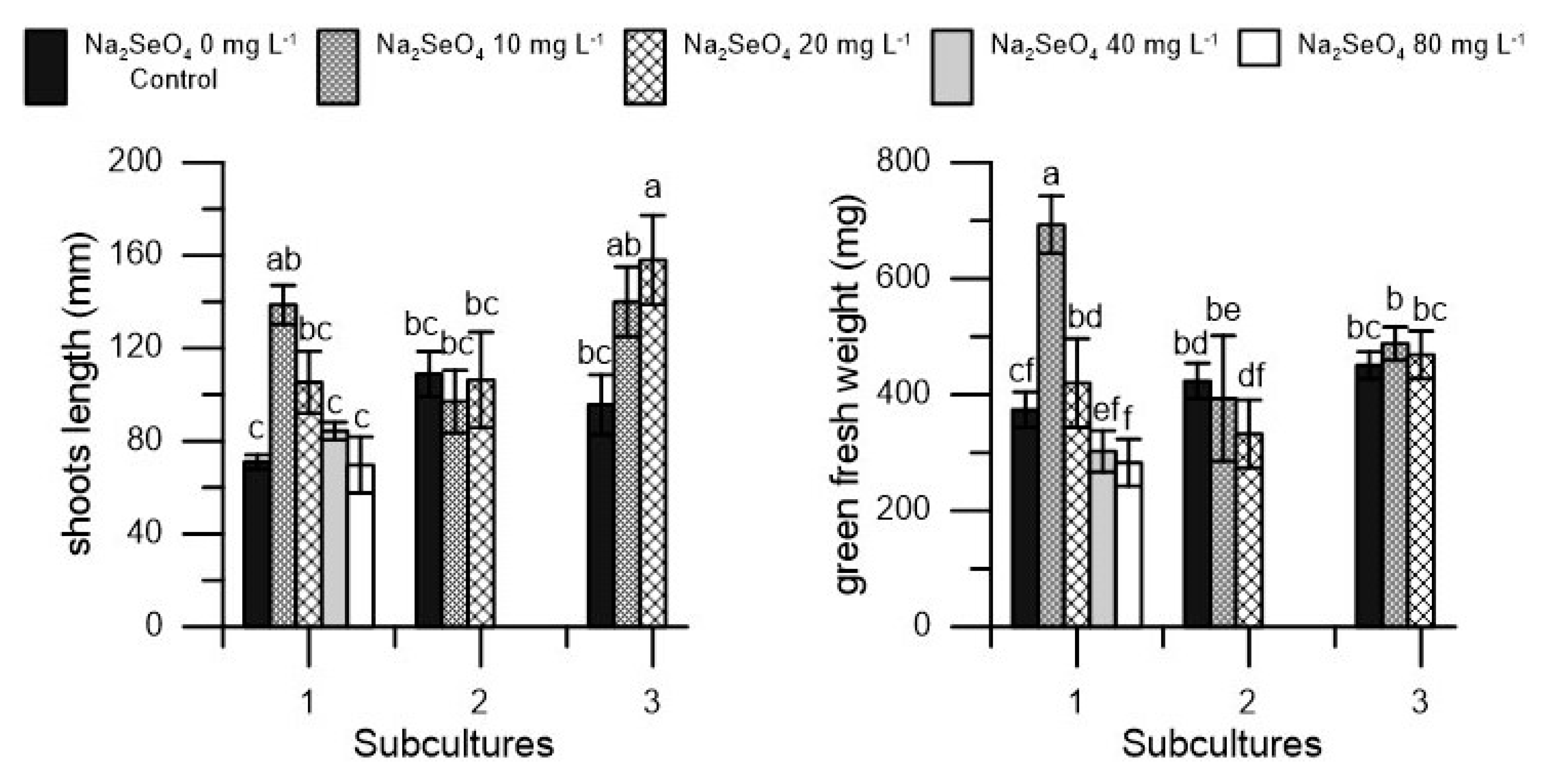
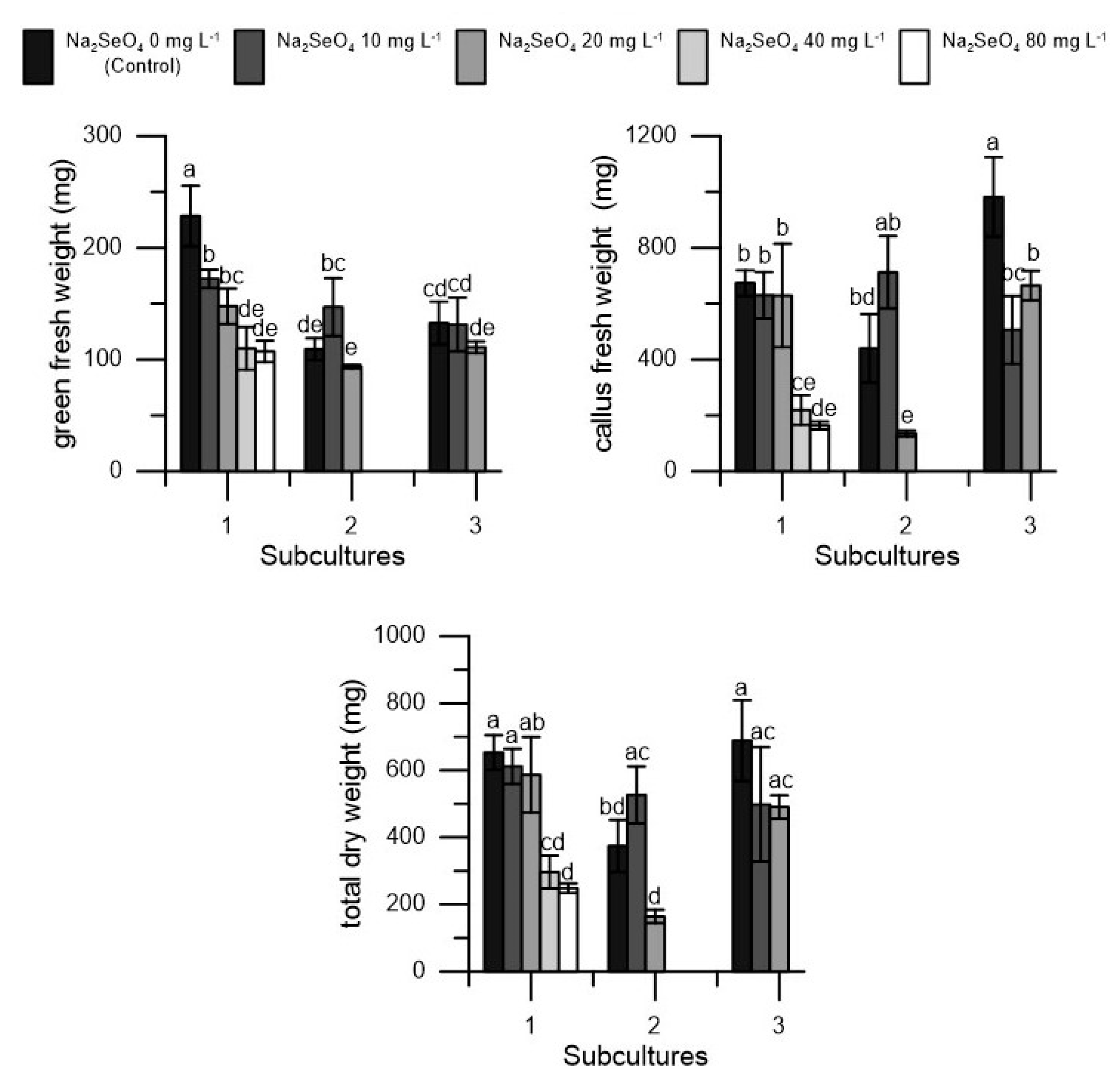
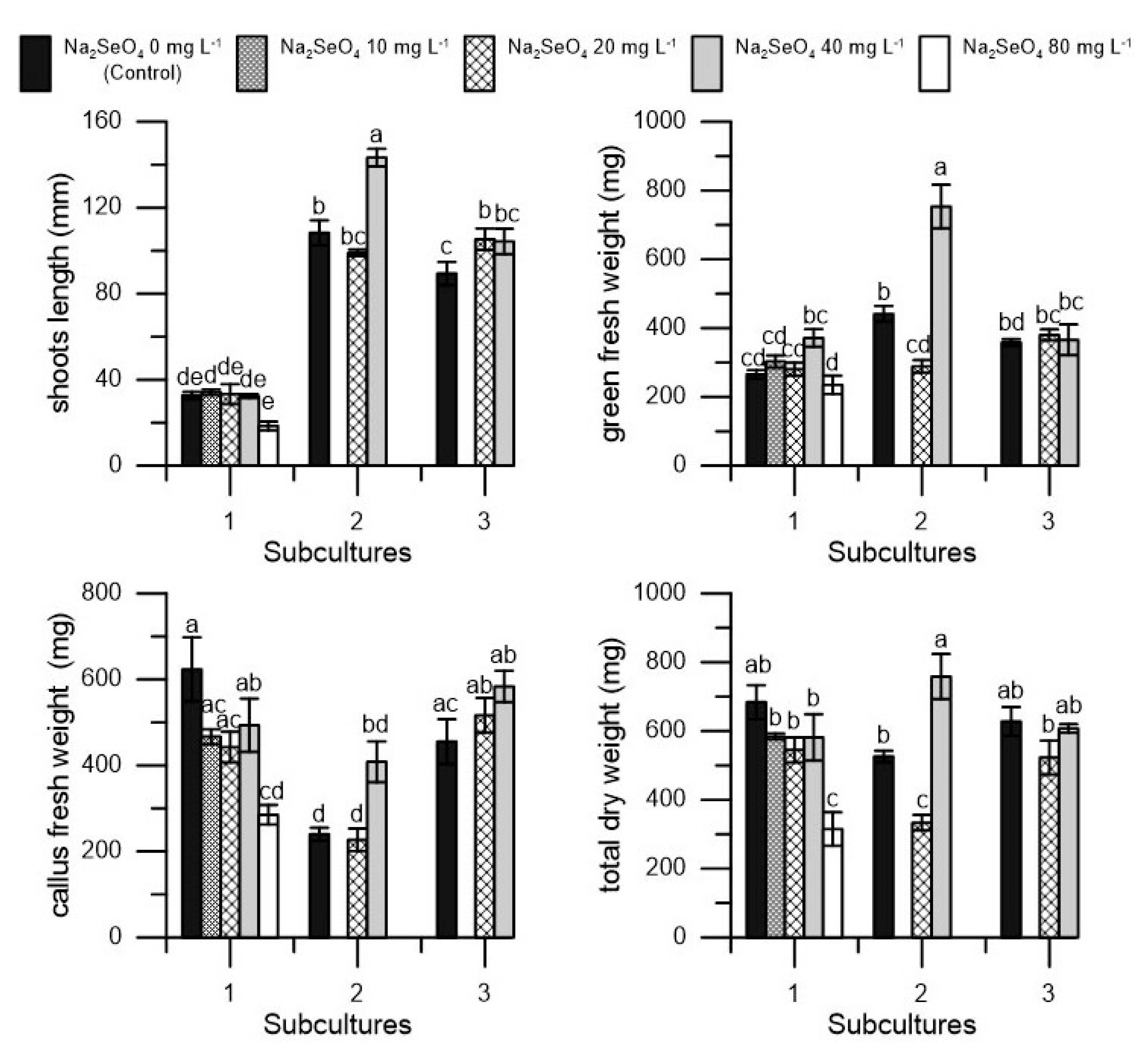
| Na2SeO4 | Vitality | Shoots | Shoots Length | Nodes | Green Fresh Weight | Callus Fresh Weight | Total Dry Weight | Dry Matter |
|---|---|---|---|---|---|---|---|---|
| (mg L−1) | (%) | (n) | (mm) | (n) | (mg) | (mg) | (mg) | (%) |
| 0 (control) | 100 a | 2.72 a | 62.68 a | 11.49 a | 316.50 ± 55.4 b | 597.94 a | 537.83 ± 99.8 b | 58.81 ± 7.3 b |
| 20 | 100 a | 2.93 a | 80.83 a | 12.91 a | 351.22 ± 43.8 ab | 872.22 a | 823.54 ± 130.4 a | 67.31 ± 2.7 a |
| 40 | 100 a | 2.80 a | 84.59 a | 11.90 a | 466.22 ± 90.1 a | 925.89 a | 830.66 ± 187.6 a | 59.67 ± 3.2 b |
| Na2SeO4 | Vitality | Shoots | Shoots Length | Nodes | Green Fresh Weight | Callus Fresh Weight | Total Dry Weight | Dry Matter |
|---|---|---|---|---|---|---|---|---|
| (mg L−1) | (%) | (n) | (mm) | (n) | (mg) | (mg) | (mg) | (%) |
| 0 (control) | 100 a | 3.29 a | 91.84 a | 11.71 a | 415.89 a | 567.11 a | 680.69 a | 69.25 a |
| 10 | 100 a | 3.80 a | 125.22 a | 13.69 a | 524.78 a | 643.33 a | 737.95 a | 63.17 a |
| 20 | 100 a | 3.67 a | 123.22 a | 14.13 a | 407.00 a | 533.57 a | 652.16 a | 69.33 a |
| Na2SeO4 | Vitality | Shoots | Shoots Length | Nodes | Green Fresh Weight | Callus Fresh Weight | Total Dry Weight | Dry Matter |
|---|---|---|---|---|---|---|---|---|
| (mg L−1) | (%) | (n) | (mm) | (n) | (mg) | (mg) | (mg) | (%) |
| 0 (control) | 100 a | 1.78 a | 49.11 a | 6.42 a | 156.78 a | 698.69 a | 572.23 a | 66.89 a |
| 10 | 100 a | 1.82 a | 58.22 a | 7.18 a | 150.22 a | 616.33 a | 545.47 a | 71.16 a |
| 20 | 100 a | 1.59 a | 50.72 a | 5.87 a | 117.44 a | 476.56 a | 413.73 a | 69.65 a |
| Na2SeO4 | Vitality | Shoots | Shoot Length | Nodes | Green Fresh Weight | Callus Fresh Weight | Total Dry Weight | Dry Matter |
|---|---|---|---|---|---|---|---|---|
| (mg L−1) | (%) | (n) | (mm) | (n) | (mg) | (mg) | (mg) | (%) |
| 0 (control) | 100 a | 3.42 a | 76.78 a | 13.71 a | 355.00 ± 56.5 b | 439.56 a | 612.58 ± 64.4 a | 77.09 a |
| 20 | 100 a | 2.69 a | 79.21 a | 11.71 a | 316.56 ± 39.1 b | 395.44 a | 466.97 ± 82.5 b | 65.59 a |
| 40 | 100 a | 3.92 a | 84.59 a | 12.80 a | 496.89 ± 157.4 a | 495.11 a | 649.16 ± 106.1 a | 65.44 a |
| µg Se kg−1 Plant DW | |||
|---|---|---|---|
| Subculture 1 | Subculture 2 | Subculture 3 | |
| Na2Se O4 (mg L−1) | ‘San Felice’ | ||
| 0 (control) | 0.92 ± 0.1 d | 2.65 ± 0.8 c | 1.12 ± 0.2 c |
| 10 | 20.97 ± 3.1 c | - | - |
| 20 | 31.40 ± 3.4 c | 24.79 ± 3.8 b | 30.48 ± 3.6 b |
| 40 | 57.87 ± 4.6 b | 53.84 ± 3.4 a | 54.27 ± 1.5 a |
| 80 | 95.54 ± 3.3 a | - | - |
| ‘Moraiolo’ | |||
| 0 (control) | 1.04 ± 0.2 d | 1.60 ± 0.4 b | 1.22 ± 0.3 b |
| 10 | 10.59 ± 2.1 c | 11.46 ± 2.6 a | 9.83 ± 1.9 a |
| 20 | 17.00 ± 2.3 c | 19.09 ± 1.4 a | 19.45 ± 5.5 a |
| 40 | 33.53 ± 6.8 b | - | - |
| 80 | 94.85 ± 10.4 a | - | - |
| ‘Frantoio’ | |||
| 0 (control) | 0.84 ± 0.3 e | 1.23 ± 0.4 c | 1.20 ± 0.3 c |
| 10 | 8.55 ± 1.4 d | 14.12 ± 2.5 b | 12.47 ± 1.8 b |
| 20 | 31.18 ± 4.2 c | 31.74 ± 3.1 a | 28.96 ± 5.8 a |
| 40 | 53.73 ± 2.1 b | - | - |
| 80 | 130.73 ± 14.6a | - | - |
| ‘Canino’ | |||
| 0 (control) | 0.99 ± 0.4 d | 1.17 ± 0.2 c | 1.15 ± 0.4 c |
| 10 | 19.84 ± 5.6 c | - | - |
| 20 | 29.02 ± 5.8 c | 32.53 ± 3.1 b | 28.74 ± 6.1 b |
| 40 | 63.87 ± 3.5 b | 55.90 ± 4.3 a | 58.36 ± 6.2 a |
| 80 | 114.21 ± 18.6 a | - | - |
| Cultivar | Na2SeO4 (mg L−1) | ||||
|---|---|---|---|---|---|
| 0 (Control) | 10 | 20 | 40 | 80 | |
| ‘San Felice’ | ✓ | - | ✓ | ✓ | - |
| ‘Moraiolo’ | ✓ | ✓ | ✓ | - | - |
| ‘Frantoio’ | ✓ | ✓ | ✓ | - | - |
| ‘Canino’ | ✓ | - | ✓ | ✓ | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Regni, L.; Micheli, M.; Del Pino, A.M.; Palmerini, C.A.; D’Amato, R.; Facchin, S.L.; Famiani, F.; Peruzzi, A.; Mairech, H.; Proietti, P. The First Evidence of the Beneficial Effects of Se-Supplementation on In Vitro Cultivated Olive Tree Explants. Plants 2021, 10, 1630. https://doi.org/10.3390/plants10081630
Regni L, Micheli M, Del Pino AM, Palmerini CA, D’Amato R, Facchin SL, Famiani F, Peruzzi A, Mairech H, Proietti P. The First Evidence of the Beneficial Effects of Se-Supplementation on In Vitro Cultivated Olive Tree Explants. Plants. 2021; 10(8):1630. https://doi.org/10.3390/plants10081630
Chicago/Turabian StyleRegni, Luca, Maurizio Micheli, Alberto Marco Del Pino, Carlo Alberto Palmerini, Roberto D’Amato, Simona Lucia Facchin, Franco Famiani, Alessandro Peruzzi, Hanene Mairech, and Primo Proietti. 2021. "The First Evidence of the Beneficial Effects of Se-Supplementation on In Vitro Cultivated Olive Tree Explants" Plants 10, no. 8: 1630. https://doi.org/10.3390/plants10081630
APA StyleRegni, L., Micheli, M., Del Pino, A. M., Palmerini, C. A., D’Amato, R., Facchin, S. L., Famiani, F., Peruzzi, A., Mairech, H., & Proietti, P. (2021). The First Evidence of the Beneficial Effects of Se-Supplementation on In Vitro Cultivated Olive Tree Explants. Plants, 10(8), 1630. https://doi.org/10.3390/plants10081630









