Chilling and Freezing Temperature Stress Differently Influence Glucosinolates Content in Brassica oleracea var. acephala
Abstract
:1. Introduction
2. Results
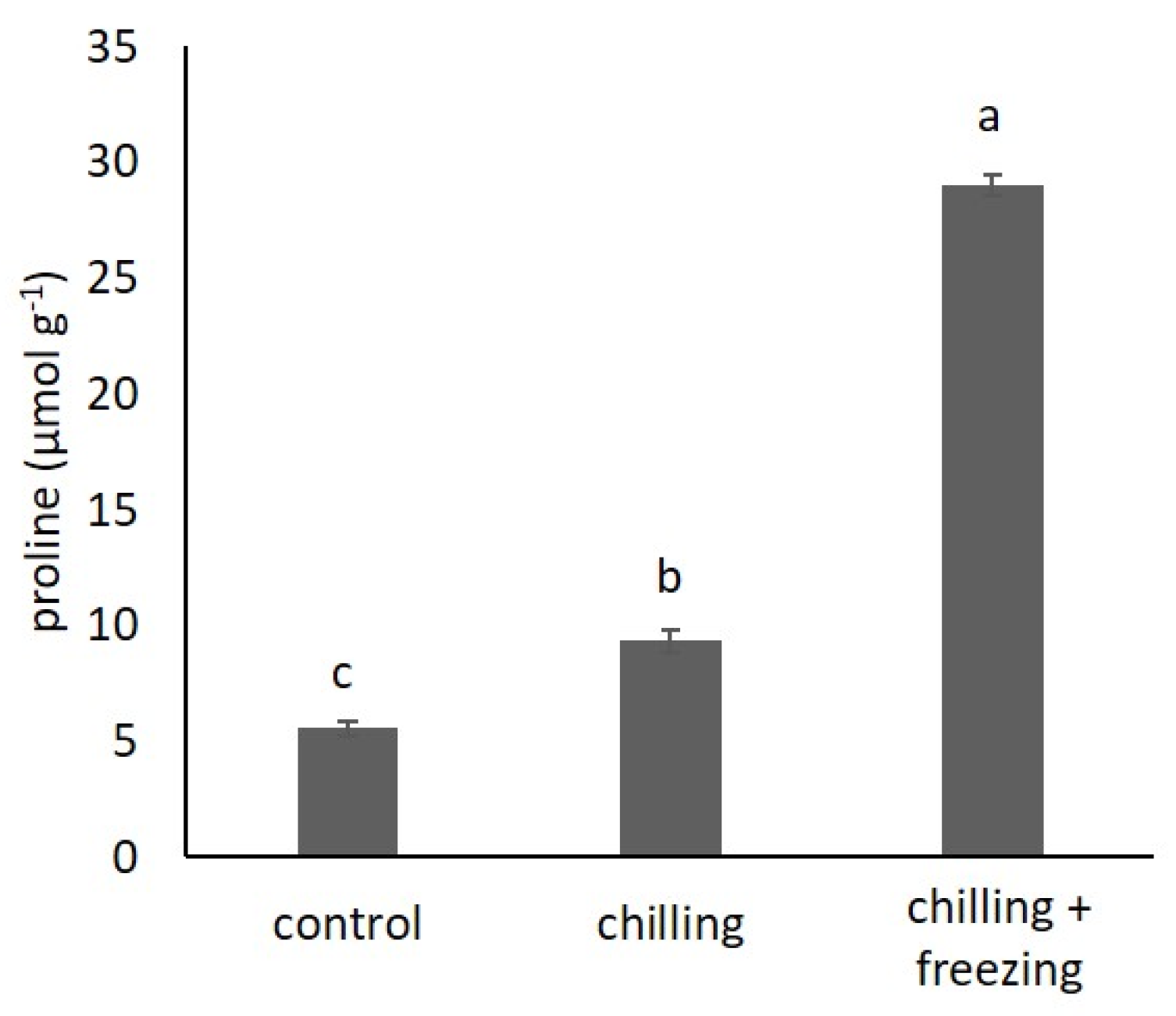
2.1. Proline Content
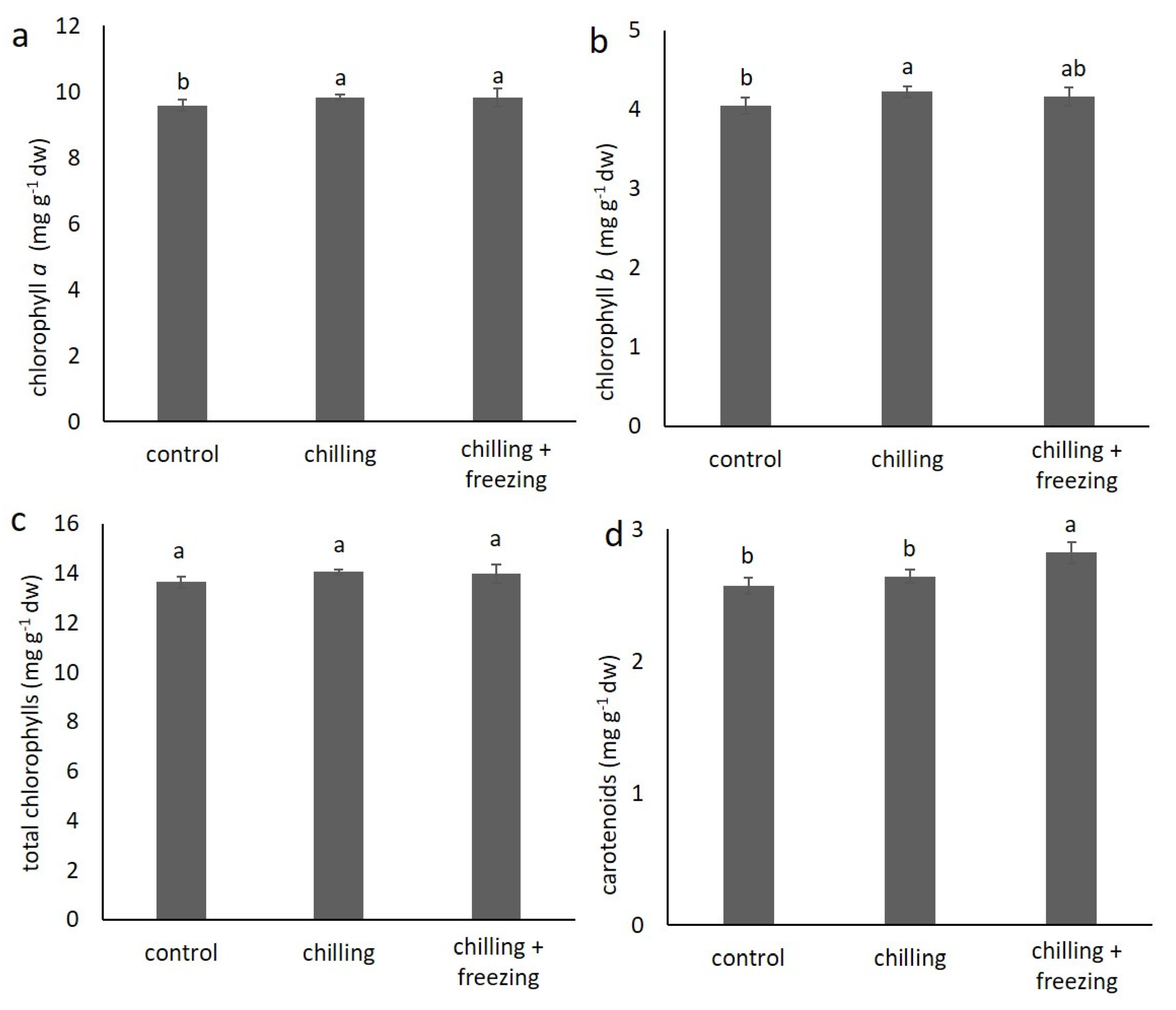
2.2. Contents of Chlorophylls and Carotenoids
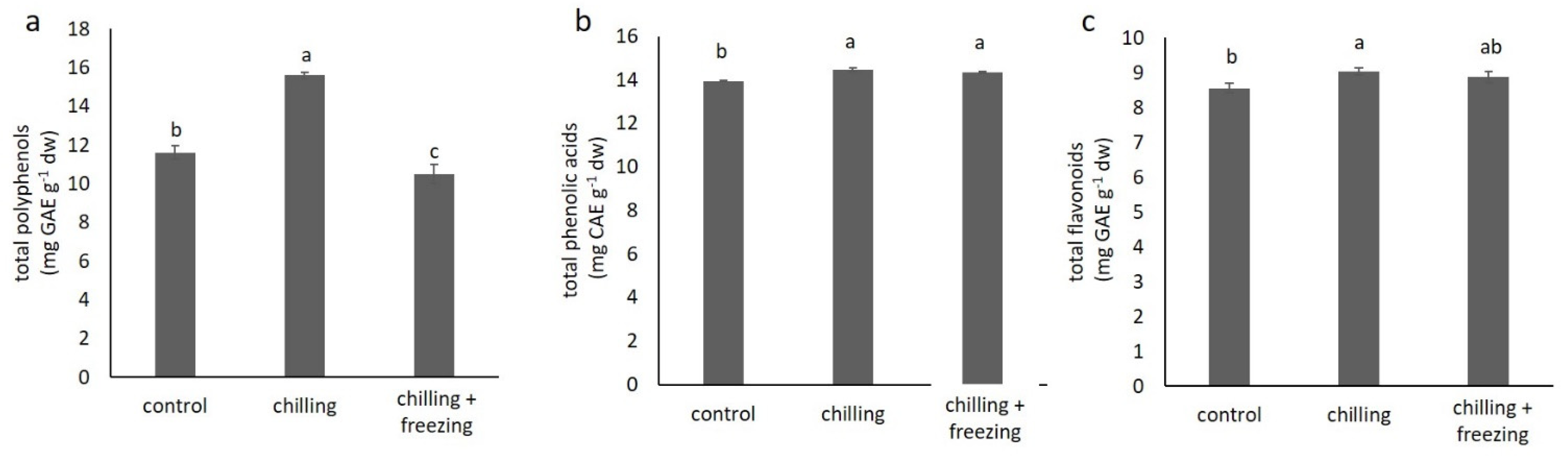
2.3. Polyphenolic Compound Contents
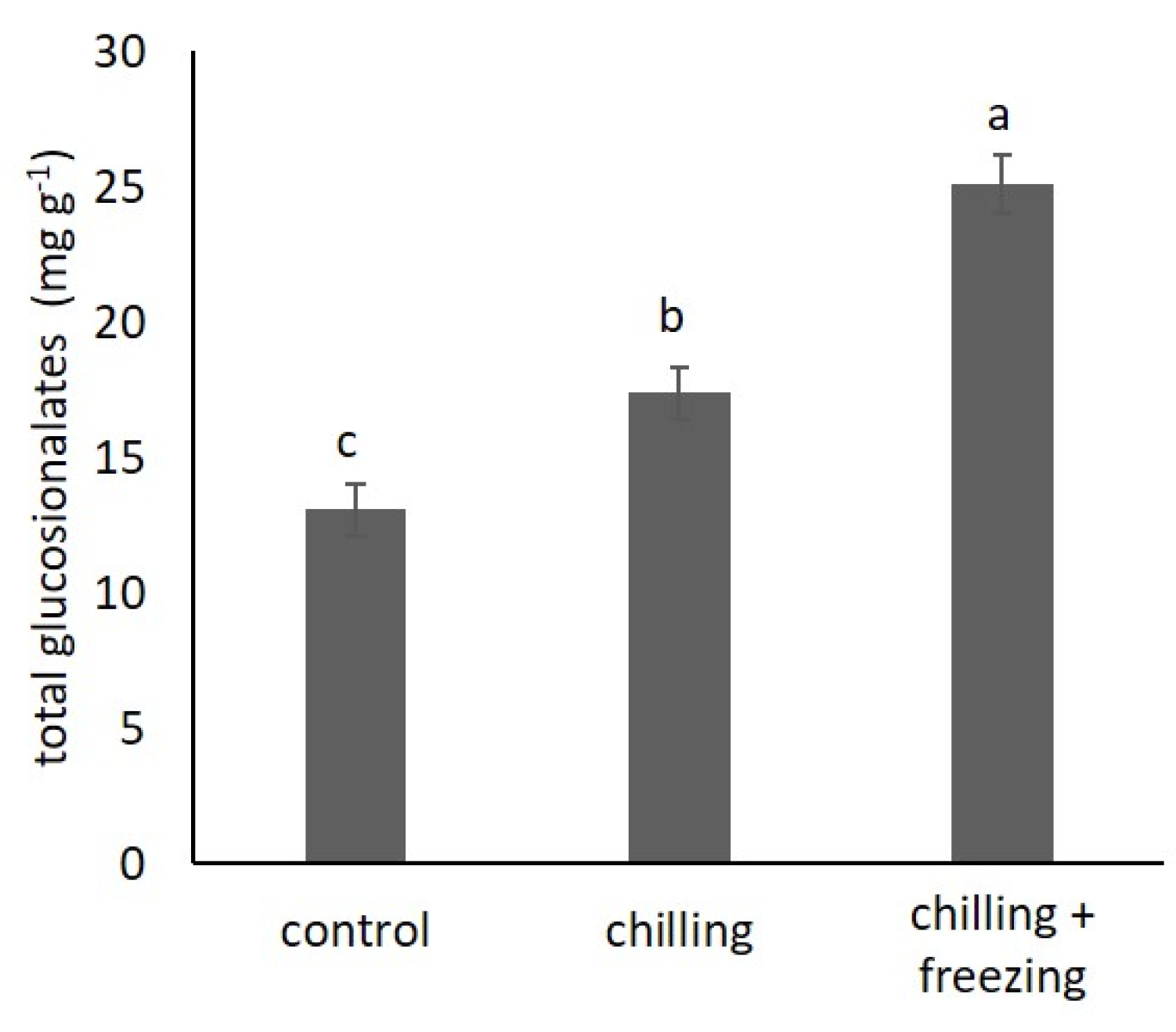
2.4. Glucosinolate Contents
3. Discussion
4. Materials and Methods
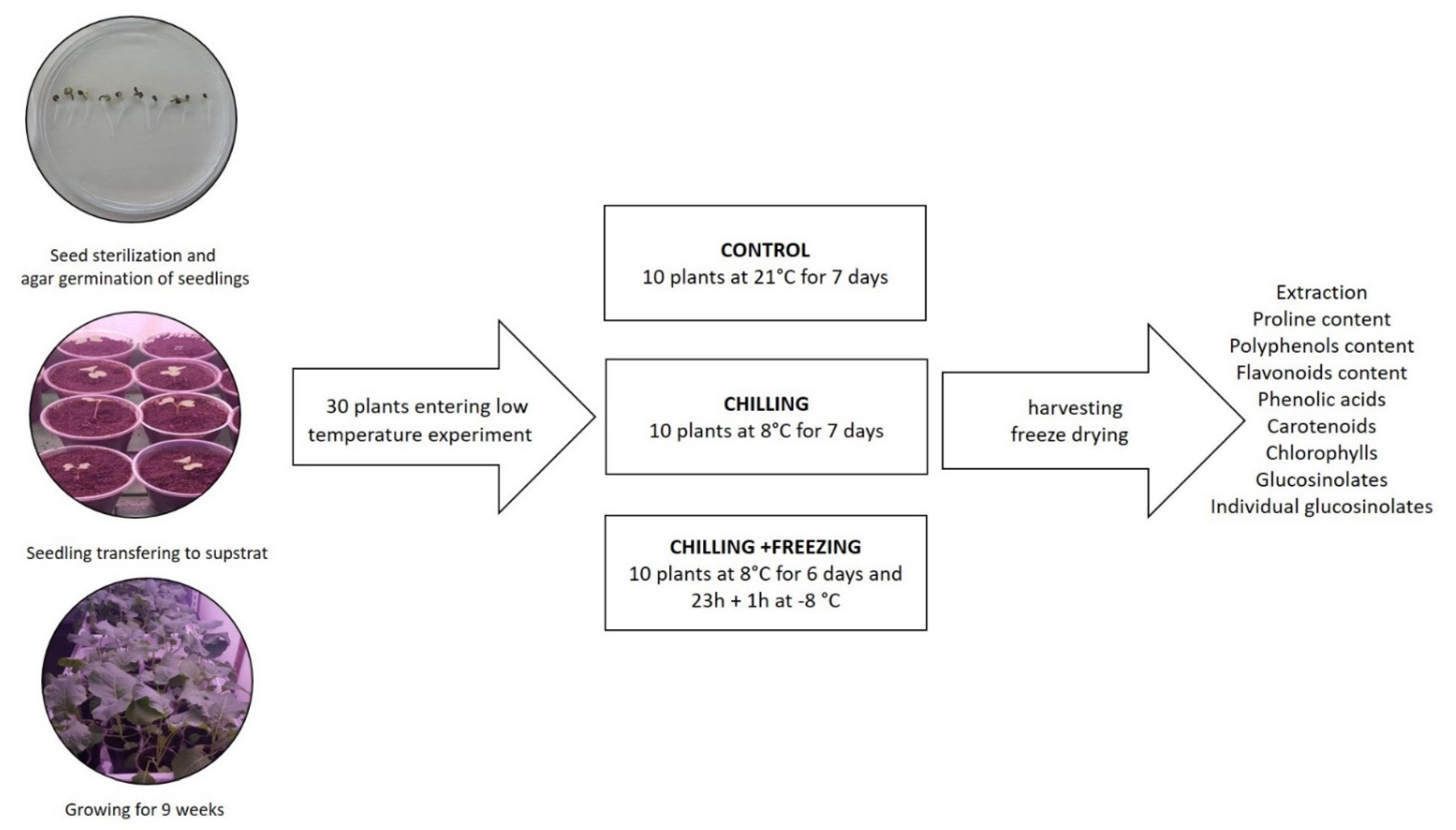
4.1. Plant Growth and Low Temperature Treatment
4.2. Determination of Proline Content
4.3. Determination of Total Phenol Content
4.4. Determination of Total Phenolic Acids
4.5. Determination of Total Flavonoids
4.6. Determination of Chlorophyll and Carotenoid Contents
4.7. Determination of Total and Individual Glucosinolates
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Šamec, D.; Urlić, B.; Salopek-Sondi, B. Kale (Brassica oleracea var. acephala) as a superfood: Review of the scientific evidence behind the statement. Crit. Rev. Food Sci. Nutr. 2019, 59, 2411–2422. [Google Scholar] [CrossRef]
- Šamec, D.; Salopek-Sondi, B. Cruciferous (Brassicaceae) vegetables. In Nonvitamin and Nonmineral Nutritional Supplements; Nabavi, S.M., Sanches Silva, T., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 195–202. [Google Scholar]
- Quirante-Moya, S.; García-Ibañez, P.; Quirante-Moya, F.; Villaño, D.; Moreno, D.A. The Role of Brassica Bioactives on Human Health: Are We Studying It the Right Way? Molecules 2020, 25, 1591. [Google Scholar] [CrossRef] [Green Version]
- Favela-González, K.M.; Hernández-Almanza, A.Y.; Fuente-Salcido, N.M. The value of bioactive compounds of cruciferous vegetables (Brassica) as antimicrobials and antioxidants: A review. J. Food Biochem. 2020, 44, e13414. [Google Scholar] [CrossRef]
- Šamec, D.; Linić, I.; Salopek-Sondi, B. Salinity Stress as an Elicitor for Phytochemicals and Minerals Accumulation in Selected Leafy Vegetables of Brassicaceae. Agronomy 2021, 11, 361. [Google Scholar] [CrossRef]
- Renna, M.; Stellacci, A.M.; Corbo, F.; Santamaria, P. The Use of a Nutrient Quality Score is Eective to Assess the Overall Nutritional Value of Three Brassica Microgreens. Foods 2020, 9, 1226. [Google Scholar] [CrossRef] [PubMed]
- Essoh, A.P.; Monteiro, F.; Pena, A.R.; Pais, M.S.; Moura, M.; Romeiras, M.M. Exploring glucosinolates diversity in Brassicaceae: A genomic and chemical assessment for deciphering abiotic stress tolerance. Plant Physiol. Biochem. 2020, 150, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Šamec, D.; Karalija, E.; Šola, I.; Vujčić-Bok, V.; Salopek-Sondi, B. The Role of Polyphenols in Abiotic Stress Response: The Influence of Molecular Structure. Plants 2021, 10, 118. [Google Scholar] [CrossRef]
- Pavlović, I.; Petrík, I.; Tarkowska, D.; Lepeduš, H.; Vujčić Bok, V.; Radić Brkanac, S.; Novák, O.; Salopek Sondi, B. Correlations between Phytohormones and Drought Tolerance in Selected Brassica Crops: Chinese Cabbage, White Cabbage and Kale. Int. J. Mol. Sci. 2018, 19, 2866. [Google Scholar] [CrossRef] [Green Version]
- Linić, I.; Šamec, D.; Grúz, J.; Vujčić, B.V.; Strnad, M.; Salopek Sondi, B. Involvement of Phenolic Acids in Short-Term Adaptation to Salinity Stress is Species-Specific among Brassicaceae. Plants 2019, 8, 155. [Google Scholar] [CrossRef] [Green Version]
- Pavlović, I.; Mlinarić, S.; Tarkowska, D.; Oklestkova, J.; Novak, O.; Lepeduš, H.; Vujčić Bok, V.; Radić Brkanac, S.; Strnad, M.; Salopek Sondi, B. Early Brassica crops responses to salinity stress: A Comparative Analysis between Chinese cabbage, White cabbage and Kale. Front. Plant Sci. 2019, 10, 450. [Google Scholar] [CrossRef] [Green Version]
- Soengas, P.; Rodríguez, V.M.; Velasco, P.; Cartea, M.E. Effect of Temperature Stress on Antioxidant Defenses in Brassica oleracea. ACS Omega 2018, 3, 5237–5243. [Google Scholar] [CrossRef]
- Podda, A.; Pollastri, S.; Bartolini, P.; Pisuttu, C.; Pellegrini, E.; Nali, C.; Cencetti, G.; Michelozzi, M.; Frassinetti, S.; Giorgetti, L.; et al. Drought stress modulates secondary metabolites in Brassica oleracea L. convar. acephala (DC) Alef, var. sabellica L. J. Sci. Food Agric. 2019, 99, 5533–5540. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.U.; Park, J.-I.; Jung, H.-J.; Hur, Y.; Nou, I.-S. Anthocyanin biosynthesis for cold and freezing stress tolerance and desirable color in Brassica rapa. Funct. Integr. Genom. 2015, 15, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Neugart, S.; Krumbein, A.; Zrenner, R. Influence of Light and Temperature on Gene Expression Leading to Accumulation of Specific Flavonol Glycosides and Hydroxycinnamic Acid Derivatives in Kale (Brassica oleracea var. sabellica). Front. Plant Sci. 2016, 7, 326. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mageney, V.; Baldermann, S.; Albach, D.C. Intraspecific Variation in Carotenoids of Brassica oleracea var. sabellica. J. Agric. Food Chem. 2016, 64, 3251–3257. [Google Scholar]
- Martínez-Ballesta, M.C.; Moreno, D.A.; Carvajal, M. The Physiological Importance of Glucosinolates on Plant Response to Abiotic Stress in Brassica. Int. J. Mol. Sci. 2013, 14, 11607–11625. [Google Scholar] [CrossRef] [Green Version]
- Steindal, A.L.H.; Rødven, R.; Hansen, E.; Mølmann, J. Effects of photoperiod, growth temperature and cold acclimatisation on glucosinolates, sugars and fatty acids in kale. Food Chem. 2015, 174, 44–51. [Google Scholar] [CrossRef]
- Jurkow, R.; Wurst, A.; Kalisz, A.; Agnieszka Sękara, A.; Cebula, S. Cold stress modifies bioactive compounds of kale cultivars during fall–winter harvests. Acta Agrobot. 2019, 72, 1761. [Google Scholar] [CrossRef] [Green Version]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls and carotenoids: Measurement and characterization by UV–VIS spectroscopy. In Current Protocols in Food Analytical Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2001; pp. F4.3.1–F4.3.8. [Google Scholar]
- Hayat, S.; Hayat, Q.; Alyemeni, M.N.; Wani, A.S.; Pichtel, J.; Ahmad, A. Role of proline under changing environments. Plant Signal. Behav. 2012, 7, 1456–1466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atici, O.; Demir, Y.; Kocaçalikan, I. Effects of Low Temperature on Winter Wheat and Cabbage Leaves. Biol Plant. 2003, 46, 603–606. [Google Scholar] [CrossRef]
- He, H.; Lei, Y.; Yi, Z.; Raza, A.; Zeng, L.; Yan, L.; Xiaoyu, D.; Yong, C.; Xiling, Z. Study on the mechanism of exogenous serotonin improving cold tolerance of rapeseed (Brassica napus L.) seedlings. Plant Growth Regul. 2021, 94, 161–170. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, F.; Yang, P.; Li, J.; Yan, G.; Hu, L. Responses of canola (Brassica napus L.) cultivars under contrasting temperature regimes during early seedling growth stage as revealed by multiple physiological criteria. Acta Physiol Plant. 2015, 37, 7. [Google Scholar] [CrossRef]
- Ensminger, I.; Busch, F.; Huner, N.P.A. Photostasis and cold acclimation: Sensing low temperature through photosynthesis. Phisiol. Plant. 2006, 126, 28–44. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Jajoo, A.; Oukarroum, A.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Łukasik, I.; Goltsev, V.; Ladle, R.J. Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol Plant. 2016, 38, 102. [Google Scholar] [CrossRef] [Green Version]
- Zietz, M.; Weckmüller, A.; Schmidt, S.; Rohn, S.; Schreiner, M.; Krumbein, A.; Kroh, L.W. Genotypic and climatic influence on the antioxidant activity of flavonoids in Kale (Brassica oleracea var. sabellica). J. Agric. Food Chem. 2010, 58, 2123–2130. [Google Scholar] [CrossRef]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of Phenylpropanoid pathway and the role of Polyphenols in Plants under Abiotic Stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef] [Green Version]
- Sarıkamış, G.; Çakır, A. Influence of low temperature on aliphatic and indole glucosinolates in broccoli (Brassica oleracea var. italica L.). Acta Hortic. 2016, 1145, 79–84. [Google Scholar] [CrossRef]
- Fahey, J.W.; Stephenson, K.K.; Wade, K.L.; Talalay, P. Urease from Helicobacter pylori is inactivated by sulforaphane and other isothiocyanates. Biochem. Biophys. Res. Commun. 2013, 435, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kapusta-Duch, J.; Kusznirewic, B.; Leszczynska, T.; Borczak, B. Effect of conventional cooking on changes in the contents of basic composition and glucosinolates in kale. Ecol. Chem. Eng. A 2016, 23, 465–480. [Google Scholar]
- Korus, A.; Slupski, J.; Gebczynski, P.; Bana, A. Effect of preliminary processing and method of preservation on the content of glucosinolates in kale (Brassica oleracea L. var. acephala) leaves. LWT Food Sci. Technol. 2014, 59, 1003–1008. [Google Scholar] [CrossRef]
- Cartea, M.E.; Velasco, P.; Obregon, S.; Padilla, G.; de Haro, A. Seasonal variation in glucosinolate content in Brassica oleracea crops grown in northwestern Spain. Phytochemistry 2008, 69, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Šamec, D.; Kruk, V.; Ivanišević, P. Influence of Seed Origin on Morphological Characteristics and Phytochemicals Levels in Brassica oleracea var. acephala. Agronomy 2019, 9, 502. [Google Scholar] [CrossRef] [Green Version]
- Šamec, D.; Bogović, M.; Vincek, D.; Martinčić, J.; Salopek-Sondi, B. Assessing the authenticity of the white cabbage (Brassica oleracea var. capitata f. alba) cv. ‘Varaždinski’ by molecular and phytochemical markers. Food Res. Int. 2014, 60, 266–272. [Google Scholar] [CrossRef]
- McMath, A. European Pharmacopoeia, 4th ed.; Council of Europe: Strasbourg, France, 2004; pp. 2377–2378. [Google Scholar]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents inmulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Aghajanzadeh, T.A.; Hawkesford, M.J.; De Kok, L.J. The significance of glucosinolates for sulfur storage in Brassicaceae seedlings. Front. Plant Sci. 2014, 5, 704. [Google Scholar] [CrossRef] [Green Version]
- Jakovljević, T.; Cvjetko, M.; Sedak, M.; Đokić, M.; Bilandžić, N.; Vorkapić-Furač, J.; Radojčić-Redovniković, I. Balance of glucosinolates content under Cd stress n two Brassica species. Plant Physiol. Biochem. 2013, 63, 99–106. [Google Scholar] [CrossRef] [PubMed]

| Chlorophyll a/ Chlorophyll b | Total Chlorophylls/ Total Carotenoids | |
|---|---|---|
| control | 2.37 ± 0.06 a | 5.31 ± 0.07 a |
| chilling | 2.33 ± 0.04 a | 5.32 ± 0.08 a |
| chilling + freezing | 2.36 ± 0.03 a | 4.96 ± 0.03 b |
| Control | Chilling | Chilling + Freezing | ||
|---|---|---|---|---|
| aliphatic | Glucoiberin | 1.87 ± 0.02 b | 2.75 ± 0.18 a | 1.13 ± 0.16 b |
| Progoitrin | 0.19 ± 0.06 b | 0.32 ± 0.04 a | nd | |
| Sinigrin | 0.57 ± 0.06 c | 0.82 ± 0.12 b | 1.18 ± 0.10 a | |
| Glucoraphanin | 0.25 ± 0.01 c | 0.85 ± 0.06 a | 0.41 ± 0.05 b | |
| Gluconapin | nd | nd | nd | |
| indolic | 4-hydroxyglucobrassicin | 0.18 ± 0.02 a | 0.20 ± 0.05 a | 0.15 ± 0.01 a |
| Glucobrassicin | 2.05 ± 0.36 b | 1.86 ± 0.16 b | 3.98 ± 0.18 a | |
| 4-methoxyglucobrassicin | 0.10 ± 0.01 a | 0.05 ± 0.02 a | 0.07 ± 0.06 a | |
| Neoglucobrasscin | 0.09 ± 0.03 b | 0.04 ± 0.01 c | 0.14 ± 0.02 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ljubej, V.; Radojčić Redovniković, I.; Salopek-Sondi, B.; Smolko, A.; Roje, S.; Šamec, D. Chilling and Freezing Temperature Stress Differently Influence Glucosinolates Content in Brassica oleracea var. acephala. Plants 2021, 10, 1305. https://doi.org/10.3390/plants10071305
Ljubej V, Radojčić Redovniković I, Salopek-Sondi B, Smolko A, Roje S, Šamec D. Chilling and Freezing Temperature Stress Differently Influence Glucosinolates Content in Brassica oleracea var. acephala. Plants. 2021; 10(7):1305. https://doi.org/10.3390/plants10071305
Chicago/Turabian StyleLjubej, Valentina, Ivana Radojčić Redovniković, Branka Salopek-Sondi, Ana Smolko, Sanja Roje, and Dunja Šamec. 2021. "Chilling and Freezing Temperature Stress Differently Influence Glucosinolates Content in Brassica oleracea var. acephala" Plants 10, no. 7: 1305. https://doi.org/10.3390/plants10071305
APA StyleLjubej, V., Radojčić Redovniković, I., Salopek-Sondi, B., Smolko, A., Roje, S., & Šamec, D. (2021). Chilling and Freezing Temperature Stress Differently Influence Glucosinolates Content in Brassica oleracea var. acephala. Plants, 10(7), 1305. https://doi.org/10.3390/plants10071305








