Resistance to Cucumber Green Mottle Mosaic Virus in Cucumis melo
Abstract
1. Introduction
2. Results
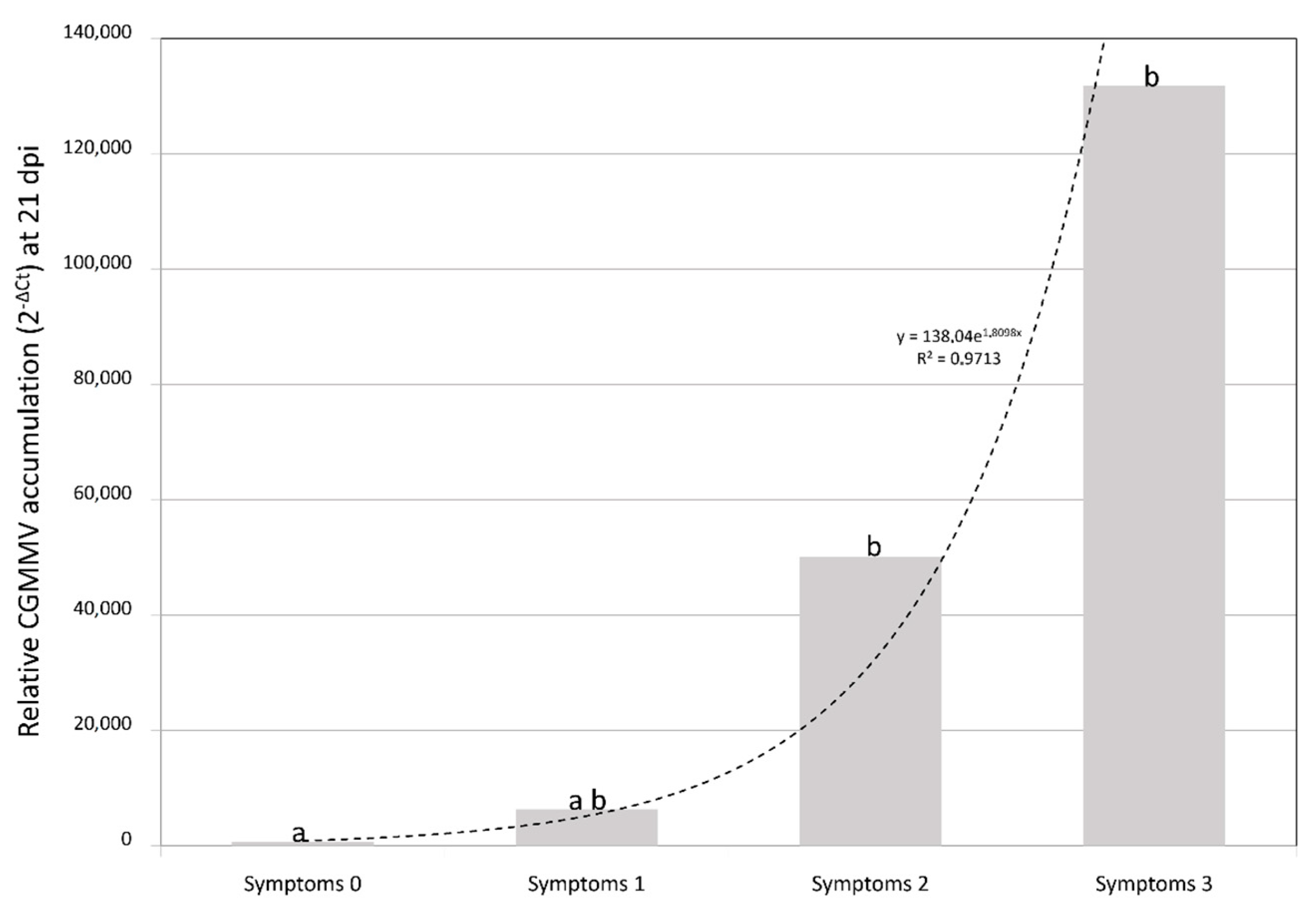
2.1. Symptom Expression
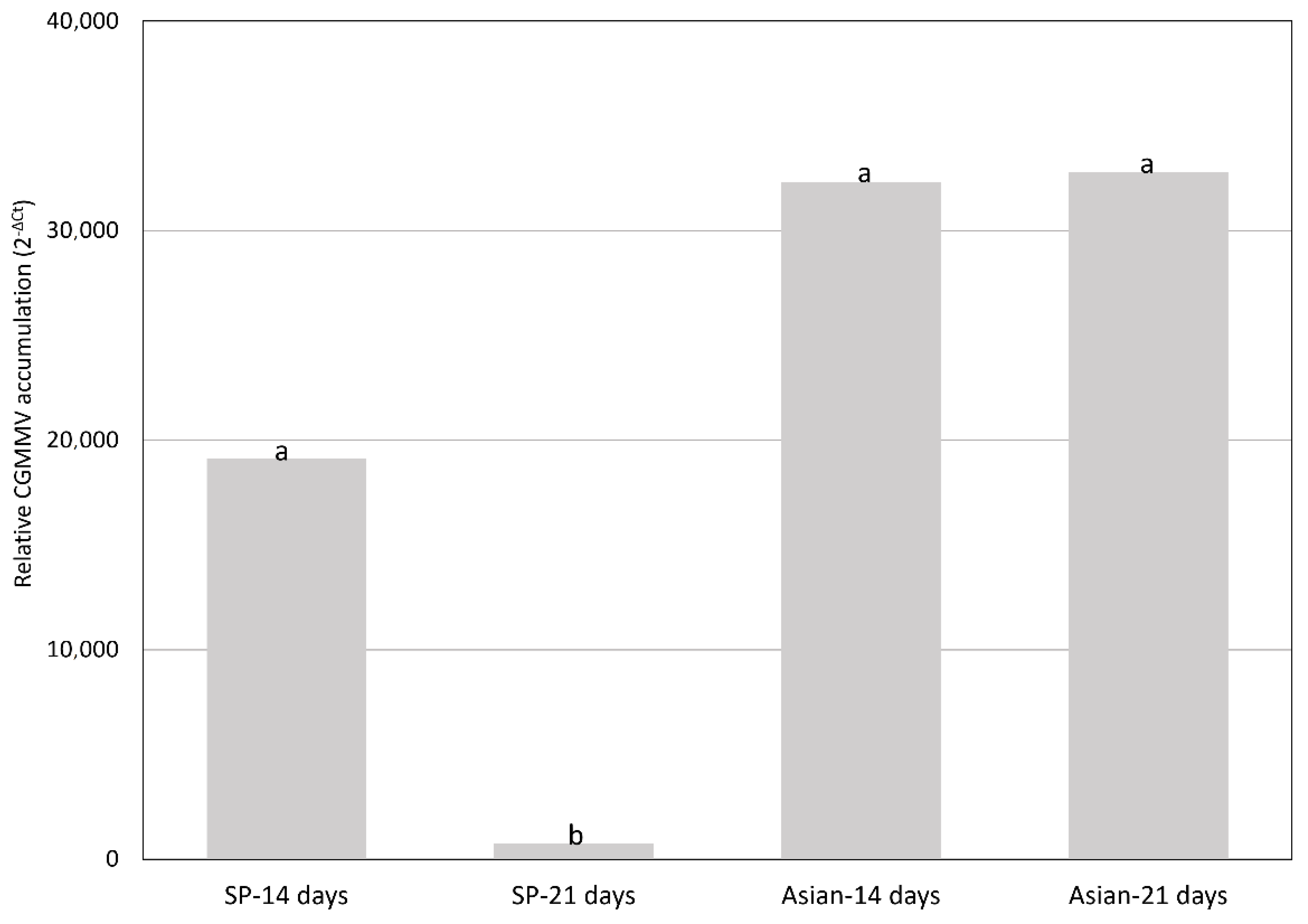
2.2. CGMMV Viral Loads
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Virus Sources for Mechanical Inoculation
4.3. Mechanical Inoculation
4.4. Evaluation of Symptoms and Quantification of the Virus
4.5. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lovisolo, O. Virus and viroid diseases of cucurbits. Acta Hortic 1980, 88, 33–82. [Google Scholar] [CrossRef]
- Adams, M.J.; Antoniw, J.M.; Kreuze, J. Virgaviridae: A new family of rod-shaped plant viruses. Arch. Virol. 2009, 154, 1967–1972. [Google Scholar] [CrossRef]
- Mandal, S.; Mandal, B.; Varma, A. Properties, diagnosis and management of Cucumber green mottle mosaic virus. Plant Viruses 2008, 2, 25–34. [Google Scholar]
- van Koot, Y.; van Dorst, H.J.M. Virusziekten van komkommer in Nederland (with a summary: Virus diseases of cucumber in the Netherlands). Tijdschr. Plantenziekten 1959, 65, 257–271. [Google Scholar]
- Dombrovsky, A.; Tran-Nguyen, L.T.T.; Jones, R.A.C. Cucumber green mottle mosaic virus: Rapidly increasing global distribution, etiology, epidemiology, and management. Ann. Rev. Phytopathol. 2017, 55, 231–256. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.W.; Luo, L.X.; Li, J.Q.; Liu, P.F.; Chen, X.Y.; Hao, J.J. Pollen and seed transmission of Cucumber green mottle mosaic virus in cucumber. Plant Pathol. 2014, 63, 72–77. [Google Scholar] [CrossRef]
- Ainsworth, G.C. Mosaic disease of cucumber. Ann. Appl. Biol. 1935, 22, 55–67. [Google Scholar] [CrossRef]
- Reingold, V.; Lachman, O.; Blaosov, E.; Dombrovsky, A. Seed disinfection treatments do not sufficiently eliminate the infectivity of Cucumber green mottle mosaic virus (CGMMV) on cucurbit seeds. Plant Pathol. 2015, 64, 245–255. [Google Scholar] [CrossRef]
- Reingold, V.; Lachman, O.; Belausov, E.; Koren, A.; Mor, N.; Dombrovsky, A. Epidemiological study of Cucumber green mottle mosaic virus in greenhouses enables reduction of disease damage in cucurbit production. Ann. Appl. Biol. 2016, 168, 29–40. [Google Scholar] [CrossRef]
- Crespo, O.; Janssen, D.; García, C.; Ruiz, L. Biological and molecular diversity of Cucumber green mottle mosaic virus in Spain. Plant Dis. 2017, 101, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Marco, C.F.; Aguilar, J.M.; Abad, J.; Gómez-Guillamón, M.L.; Aranda, M.A. Melon resistance to Cucurbit yellow stunting disorder virus in characterized by reduced virus accumulation. Phytopathology 2003, 93, 844–852. [Google Scholar] [CrossRef]
- Díaz-Pendón, J.A.; Fernández-Muñoz, R.; Gómez-Guillamón, M.L.; Moriones, E. Inheritance of resistance to Watermelon mosaic virus in Cucumis melo that impairs virus accumulation, symptom expression, and aphid transmission. Phytopathology 2005, 95, 840–846. [Google Scholar] [CrossRef]
- Martín-Hernández, A.M.; Picó, B. Natural resistances to viruses in cucurbits. Agronomy 2021, 11, 23. [Google Scholar] [CrossRef]
- FAOStat. 2020. Available online: www.fao.org/faostat (accessed on 20 March 2021).
- Ali, E.M.; Tabei, Y.; Kobayashi, K.; Yamaoka, N.; Nishiguchi, M. Molecular analysis of transgenic melon plants showing virus resistance conferred by direct repeat of movement gene of Cucumber green mottle mosaic virus. Plant Cell Rep. 2012, 31, 1371–1377. [Google Scholar] [CrossRef][Green Version]
- Rajamony, L.; More, T.A.; Seshadri, V.S.; Varma, A. Reaction of muskmelon collections to Cucumber green mottle mosaic virus. J. Phytopathol. 1990, 129, 237–244. [Google Scholar] [CrossRef]
- Pitrat, M. Disease resistance in melon and its modification by molecular breeding techniques. In Functional Genomics and Biotechnology in Solanaceae and Cucurbitaceae Crops; Ezura, H., Ariizumi, T., Garcia-Mas, J., Rose, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 175–197. [Google Scholar]
- Rajamony, L.; More, T.A.; Seshadri, V.S. Inheritance of resistance to Cucumber green mottle mosaic virus in muskmelon (Cucumis melo L.). Euphytica 1990, 47, 93–97. [Google Scholar]
- Pan, R.S.; More, T.A. Screening of melon (Cucumis melo L.) germplasm for multiple disease resistance. Euphytica 1996, 88, 125–128. [Google Scholar] [CrossRef]
- Sugiyama, M.; Ohara, T.; Sakata, Y. A new source of resistance to Cucumber green mottle mosaic virus in melon. J. Jpn. Soc. Hortic. Sci. 2006, 75, 469–475. [Google Scholar] [CrossRef]
- Sugiyama, M.; Ohara, T.; Sakata, Y. Inheritance of resistance to Cucumber green mottle mosaic virus in Cucumis melo L. ‘Chang Bougi’. J. Jpn. Soc. Hortic. Sci. 2007, 76, 316–318. [Google Scholar] [CrossRef]
- Flores-León, A.; García-Martínez, S.; González, V.; Garcés-Claver, A.; Martí, R.; Julián, C.; Sifres, A.; Pérez-de-Castro, A.; Díez, M.J.; López, C.; et al. Grafting snake melon [Cucumis melo L. subsp. melo Var. flexuosus (L.) Naudin] in organic farming: Effects on agronomic performance; resistance to pathogens; sugar, acid, and VOC profiles; and consumer acceptance. Front. Plant Sci. 2021, 12, 613845. [Google Scholar] [CrossRef] [PubMed]
- Pascual, L.; Yan, J.; Pujol, M.; Monforte, A.J.; Picó, B.; Martín-Hernández, A.M. CmVPS41 Is a general gatekeeper for resistance to Cucumber mosaic virus phloem entry in melon. Front. Plant Sci. 2019, 10, 1219. [Google Scholar] [CrossRef]
- Brotman, Y.; Normantovich, M.; Goldenberg, Z.; Zvirin, Z.; Kovalski, I.; Stovbun, N.; Doniger, T.; Bolger, A.M.; Troadec, C.; Bendahmane, A.; et al. Dual resistance of melon to Fusarium oxysporum Races 0 and 2 and to Papaya ring-spot virus is controlled by a pair of head-to-head-oriented NB-LRR genes of unusual architecture. Mol. Plant 2013, 6, 235–238. [Google Scholar] [CrossRef]
- Pitrat, M.; Lecoq, H. Two alleles for Watermelon Mosaic Virus 1 resistance in melon. Cucurbit Genet. Coop. Rep. 1983, 6, 52–53. [Google Scholar]
- Crespo, O.; Janssen, D.; Robles, C.; Ruiz, L. Resistance to Cucumber green mottle mosaic virus in Cucumis sativus. Euphytica 2018, 214, 201. [Google Scholar] [CrossRef]
- Giner, A.; Pascual, L.; Bourgeois, M.; Gyetvai, G.; Rios, P.; Picó, B.; Troadec, C.; Bendahmane, A.; Garcia-Mas, J.; Martín-Hernández, A.M. A mutation in the melon Vacuolar Protein Sorting 41prevents systemic infection of Cucumber mosaic virus. Sci. Rep. 2017, 7, 10471. [Google Scholar] [CrossRef]
- Díaz, J.A.; Mallor, C.; Soria, C.; Camero, R.; Garzo, E.; Fereres, A.; Alvarez, J.M.; Gómez-Guillamón, M.L.; Luis-Arteaga, M.; Moriones, E. Potential sources of resistance for melon to nonpersistently aphidborne viruses. Plant Dis. 2003, 87, 960–964. [Google Scholar] [CrossRef] [PubMed]
- Daryono, B.S.; Somowiyarjo, S.; Natsuaki, K.T. Biological and molecular characterization of melon-Infecting Kyuri green mottle mosaic virus in Indonesia. J. Phytopathol. 2005, 153, 588–595. [Google Scholar] [CrossRef]
- Den Nijs, A.P.M. Inheritance of resistance to Cucumber green mottle mosaic virus (Cgm) in Cucumis anguria L. Cucurbit Gen. Coop. Rep. 1982, 5, 57–58. [Google Scholar]
- Esteras, C.; Formisano, G.; Roig, C.; Díaz, A.; Blanca, J.; Garcia-Mas, J.; Gómez-Guillamón, M.L.; López-Sesé, A.I.; Lázaro, A.; Monforte, A.J.; et al. SNP genotyping in melons: Genetic variation, population structure, and linkage disequilibrium. Theor. Appl. Genet. 2013, 126, 1285–1303. [Google Scholar] [CrossRef] [PubMed]
- Leida, C.; Moser, C.; Esteras, C.; Sulpice, R.; Lunn, J.E.; de Langen, F.; Monforte, A.J.; Picó, B. Variability of candidate genes, genetic structure and association with sugar accumulation and climacteric behavior in a broad germplasm collection of melon (Cucumis melo L.). BMC Genet. 2015, 16, 28. [Google Scholar] [CrossRef] [PubMed]
- Gonzalo, M.J.; Díaz, A.; Dhillon, N.P.S.; Reddy, U.K.; Picó, B.; Monforte, A.J. Re-evaluation of the role of Indian germplasm as center of melon diversification based on genotyping-by-sequencing analysis. BMC Genom. 2019, 20, 448. [Google Scholar] [CrossRef] [PubMed]
- Karchi, Z.; Cohen, S.; Govers, A. Inheritance of resistance to Cucumber mosaic virus in melons. Phytopathology 1975, 65, 479–481. [Google Scholar] [CrossRef]
- Cáceres-Burbano, A.; Perpiña Martin, G.; Ferriol Molina, M.; Picó Sirvent, M.B.; Gisbert Domenech, M.C. New Cucumis rootstocks for melon: ’UPV-FA’ and ’UPV-FMy’. HortScience 2017, 52, 792–797. [Google Scholar] [CrossRef]
- Esteras, C.; Lunn, J.; Sulpice, R.; Blanca, J.; Garcia-Mas, J.; Pitrat, M.; Nuez, F.; Picó, B. Phenotyping a highly diverse core melon collection to be screened using Ecotilling. In Proceedings of the 8th Plant Genomics European Meeting, Lisbon, Portugal, 7–10 October 2009. [Google Scholar]
- Sáez, C.; Martínez, C.; Ferriol, M.; Manzano, S.; Velasco, L.; Jamilena, M.; López, C.; Picó, B. Resistance to Tomato leaf curl New Delhi virus in Cucurbita spp. Ann. Appl. Biol. 2016, 169, 91–105. [Google Scholar] [CrossRef]
- Bio-Rad Laboratories Inc. Real-Time PCR Applications Guide; Bio-Rad Laboratories Inc.: Hercules, CA, USA, 2006; p. 100. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta c (t)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]


| Accession ID | Accession Name | Country of Origin |
|---|---|---|
| C. melo subsp. Melo | ||
| Inodorus-ibericus Spanish landraces | ||
| BGV016451 | Amarillo Groc | Spain |
| BGV015753 | Blanco | Spain |
| BGV004871 | Tendral | Spain |
| BGV013188 | Pipa de oro | Spain |
| BGV003686 | Piñoncillo | Spain |
| BGV003692 | Blanco Redondo | Spain |
| BGV003718 | Mochuelo | Spain |
| BGV004884 | Melón Rochet | Spain |
| Ameri/adana/chandalak/casaba and other landraces from Europe and Asia | ||
| PI 125951 | 3584 | Afghanistan |
| BGV001367 | Nanatri | Georgia |
| PI 314427 | Koljoznitza | Georgia |
| CUM 259 | Apelsinaja | Russia |
| PI 276660 | VIR610-chandalak | Afghanistan |
| BGV001364 | Asli | Tunisia |
| BGV001365 | Tokash | Tajikistan |
| PI 169331 | Altimbas | Turkey |
| PI 506459 | Salgirskaja | Ukraine |
| BGV001362 | Kizil-uruk | Uzbekistan |
| PI 169305 | Kirkagac | Turkey |
| PI 476342 | Imljskaha | Kazakhstan |
| Am-BirUkr | Birjucekutskaja | Ukraine |
| In-HamiChi | Hami Melon | China |
| Am-OuzUzb2 | Ouzbeque | Uzbekistan |
| Am-SouiMor | Souilah | Morocco |
| Cantalupensis and reticulatus commercial cultivars and landraces from Europe, Asia, and America | ||
| Can-VedFran | Vedrantais | France |
| Can-NYIsr | Noy Israel | Israel |
| Can-NOFran | Nantais Oblong | France |
| Ames 26811 | PMR-45 | USA |
| La-OgenBul | Dvash Ha Ogen | Bulgaria |
| Flexuosus and Dudaim accessions | ||
| BGV004853 | Alficoz | Spain |
| PI 273438 | Queen Anne’s Pocket Melon | Georgia |
| C. melo subsp. agrestis | ||
| Momordica accessions and other landraces from India | ||
| PI 124112 | 2564 | India |
| PI 180280 | Kahkri | India |
| PI 381781 | Sm1 | India |
| PI 381789 | Sm9 | India |
| PI 271332 | Khira | India |
| Conomon, chinensis and makuwa accessions from Far East | ||
| PI 420176 | Ginsen makuwa | Japan |
| Con-FreeCJa | Freeman’s Cucumber | Japan |
| PI 161375 | Songwhan Charmi | Korea |
| Con-ShiroJa | Shiro Uri Okayama | Japan |
| Kachri and wild agrestis from Asia and Africa | ||
| Am-SarakIran | Sarakhs | Iran |
| PI 614521 | KSM 531 Kachri | India |
| PI 164493 | Kakru | India |
| PI 185111 | 15591 | Ghana |
| PI 164797 | 9227 | India |
| PI 532839 | Chibbar | India |
| PI 536476 | KLM 1733 | Maldives |
| Wild Cucumis spp. | ||
| BGV11135 | C. metuliferus | |
| BGV012786 | C. ficifolius | |
| BGV012795 | C. anguria | |
| BGV008535 | C. myriocarpus | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz, L.; López, C.; Picó, B.; Janssen, D. Resistance to Cucumber Green Mottle Mosaic Virus in Cucumis melo. Plants 2021, 10, 1077. https://doi.org/10.3390/plants10061077
Ruiz L, López C, Picó B, Janssen D. Resistance to Cucumber Green Mottle Mosaic Virus in Cucumis melo. Plants. 2021; 10(6):1077. https://doi.org/10.3390/plants10061077
Chicago/Turabian StyleRuiz, Leticia, Carmelo López, Belén Picó, and Dirk Janssen. 2021. "Resistance to Cucumber Green Mottle Mosaic Virus in Cucumis melo" Plants 10, no. 6: 1077. https://doi.org/10.3390/plants10061077
APA StyleRuiz, L., López, C., Picó, B., & Janssen, D. (2021). Resistance to Cucumber Green Mottle Mosaic Virus in Cucumis melo. Plants, 10(6), 1077. https://doi.org/10.3390/plants10061077








