Epizoochory in Parrots as an Overlooked Yet Widespread Plant–Animal Mutualism
Abstract
1. Introduction
2. Results
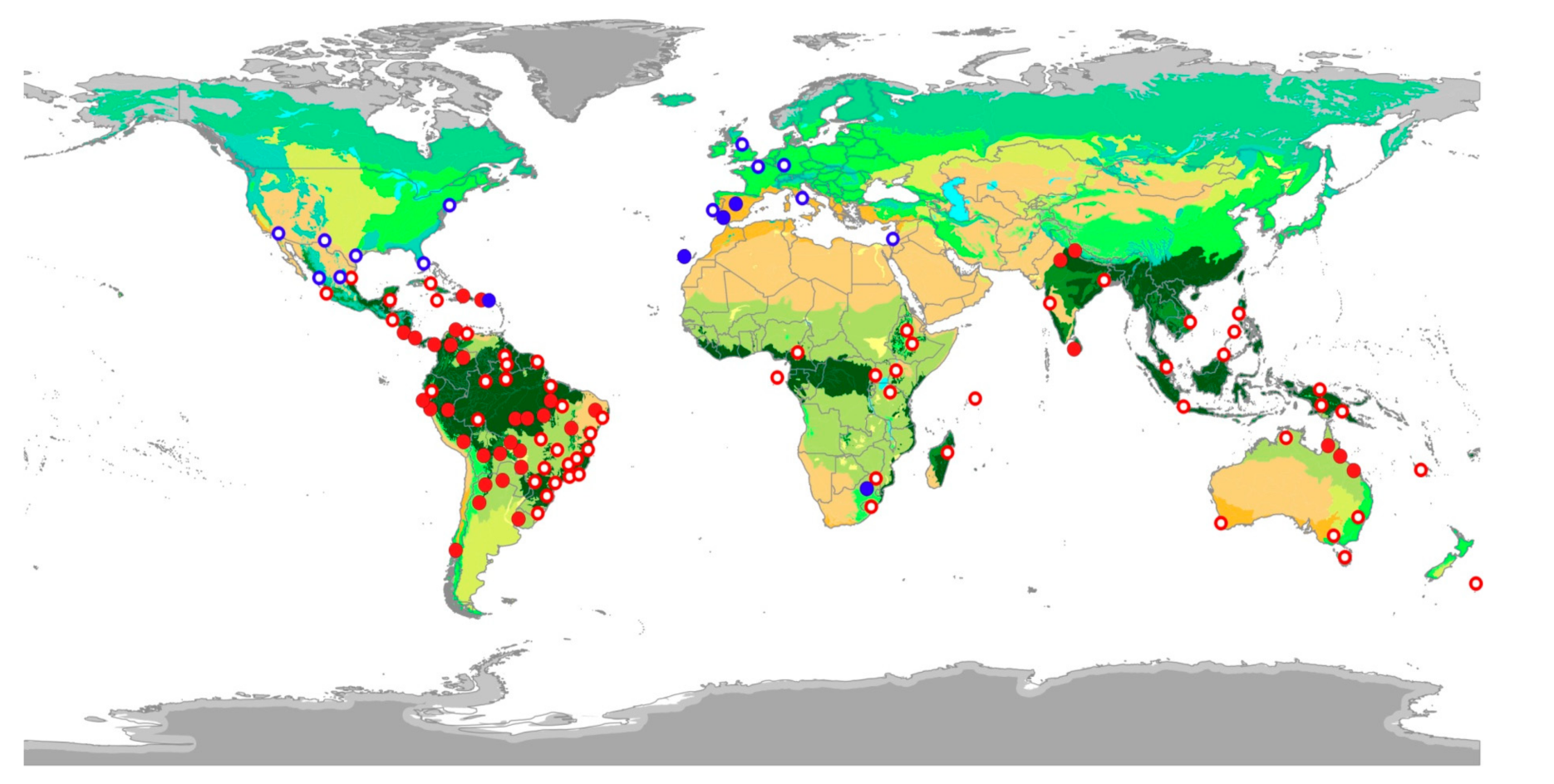
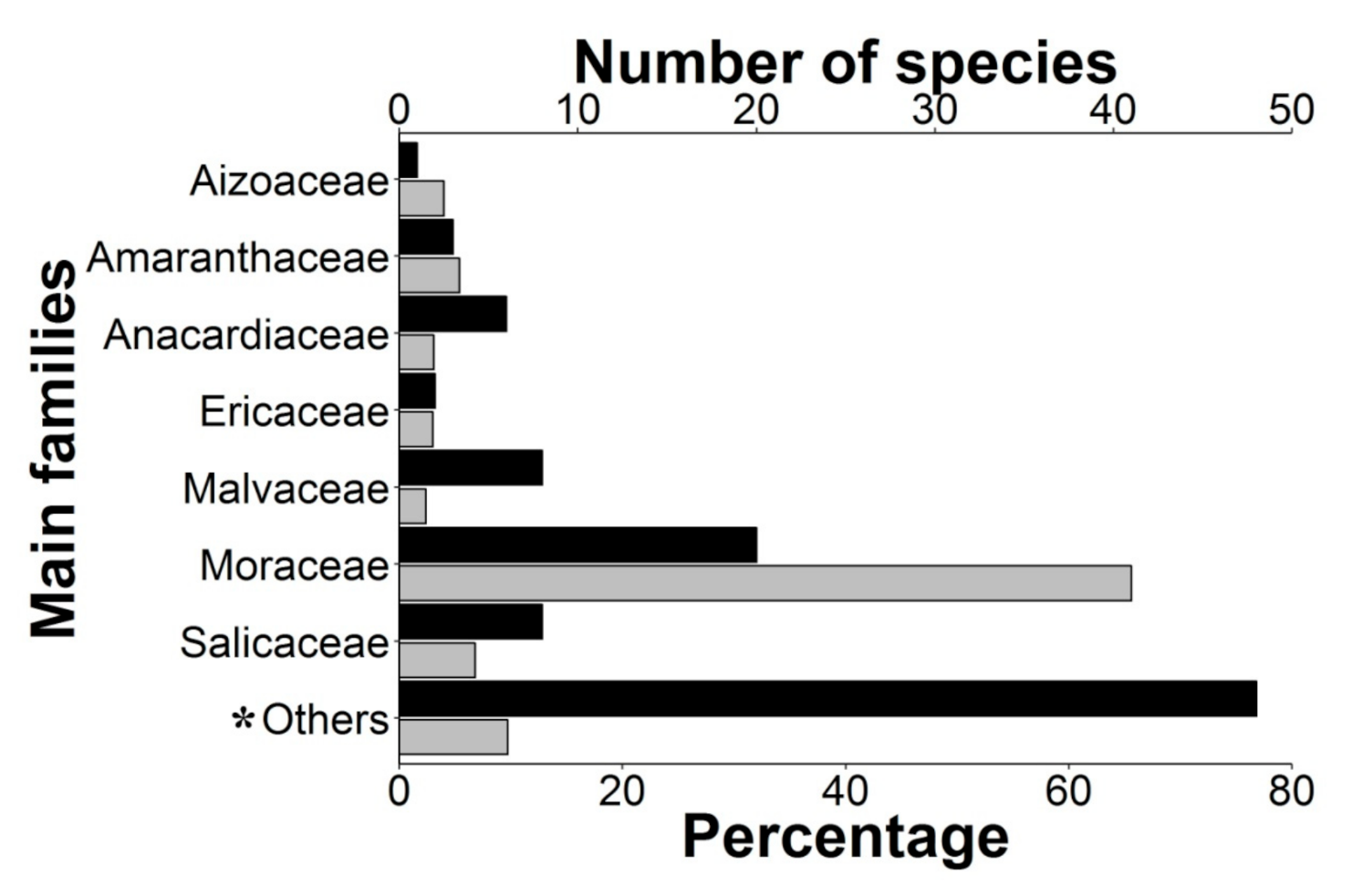
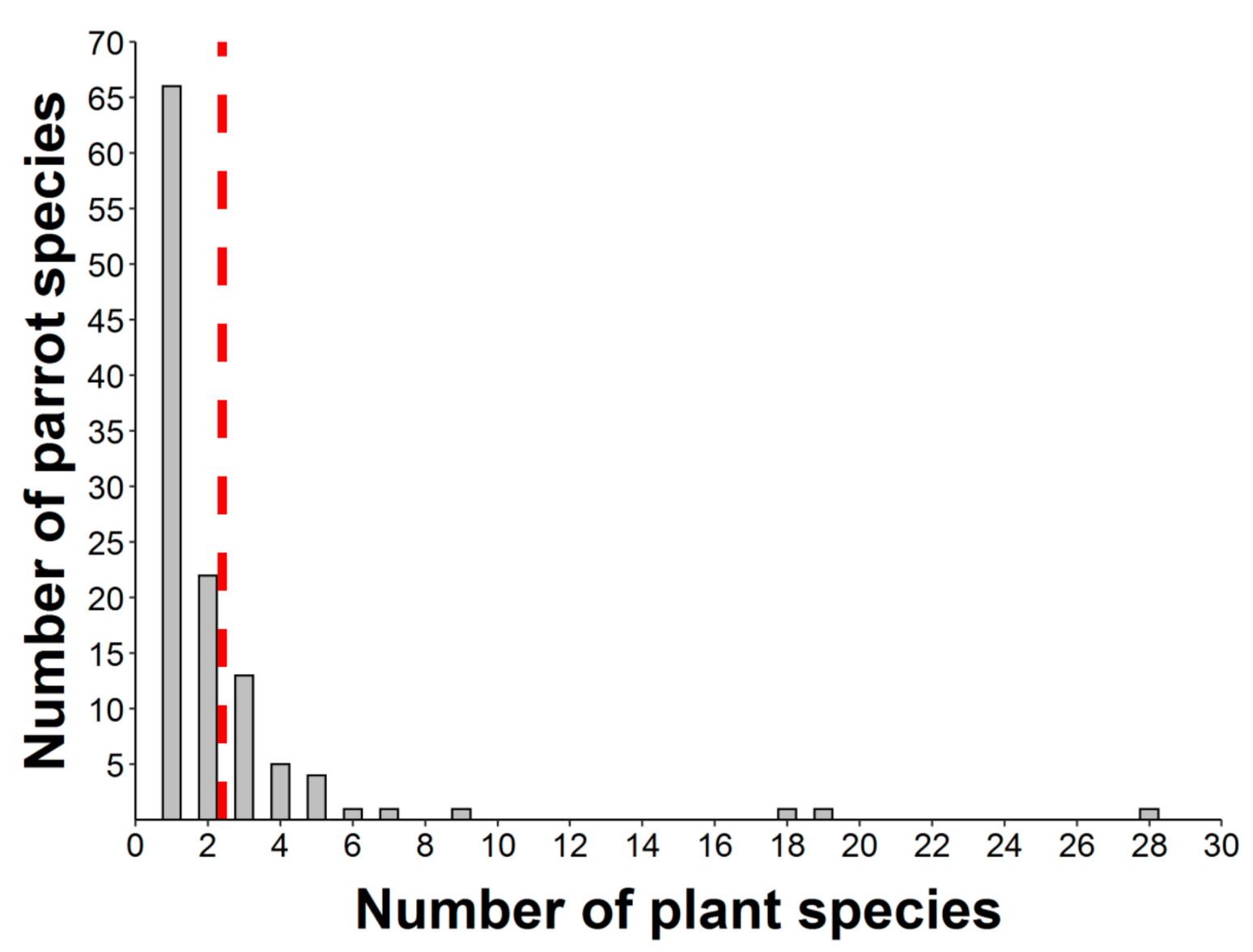
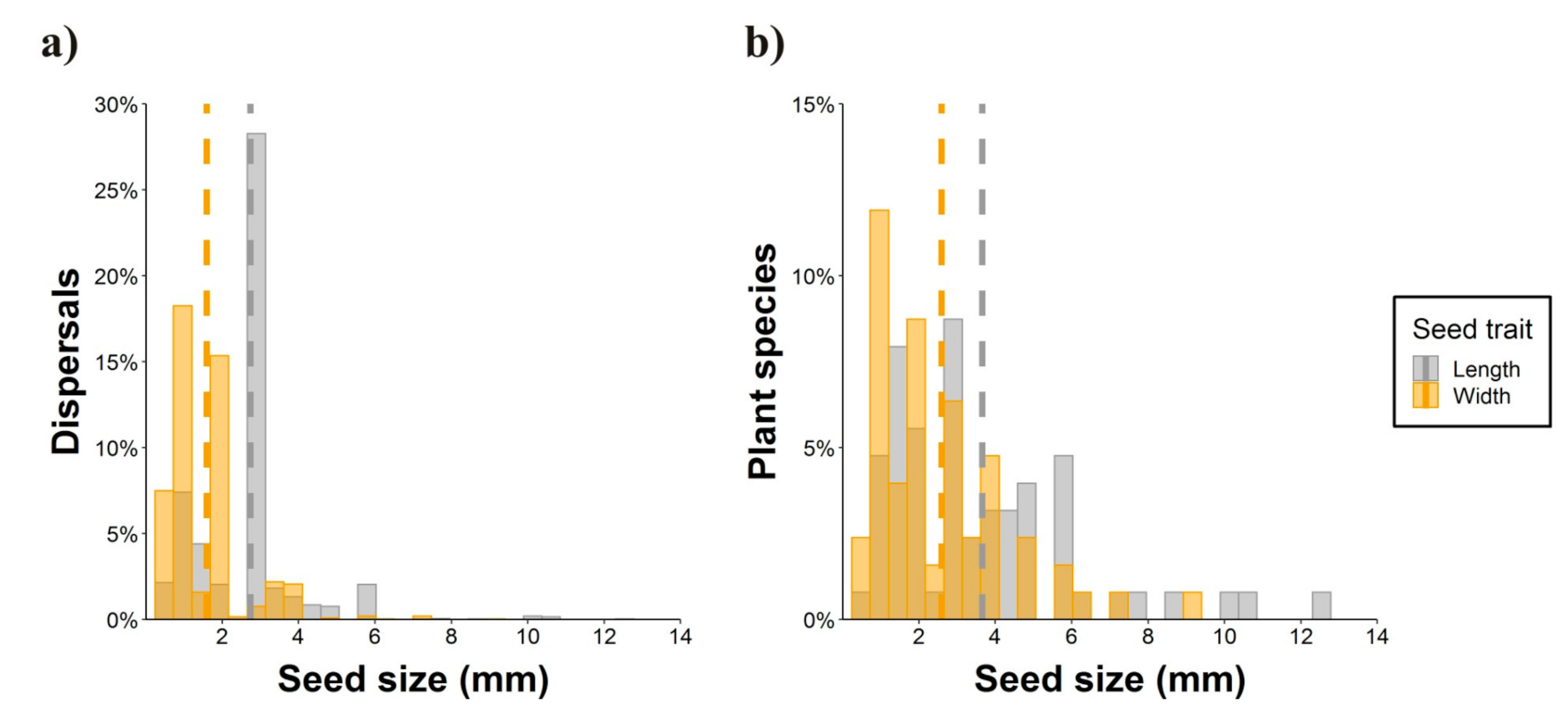
2.1. Epizoochory Events and Dispersal Behavior
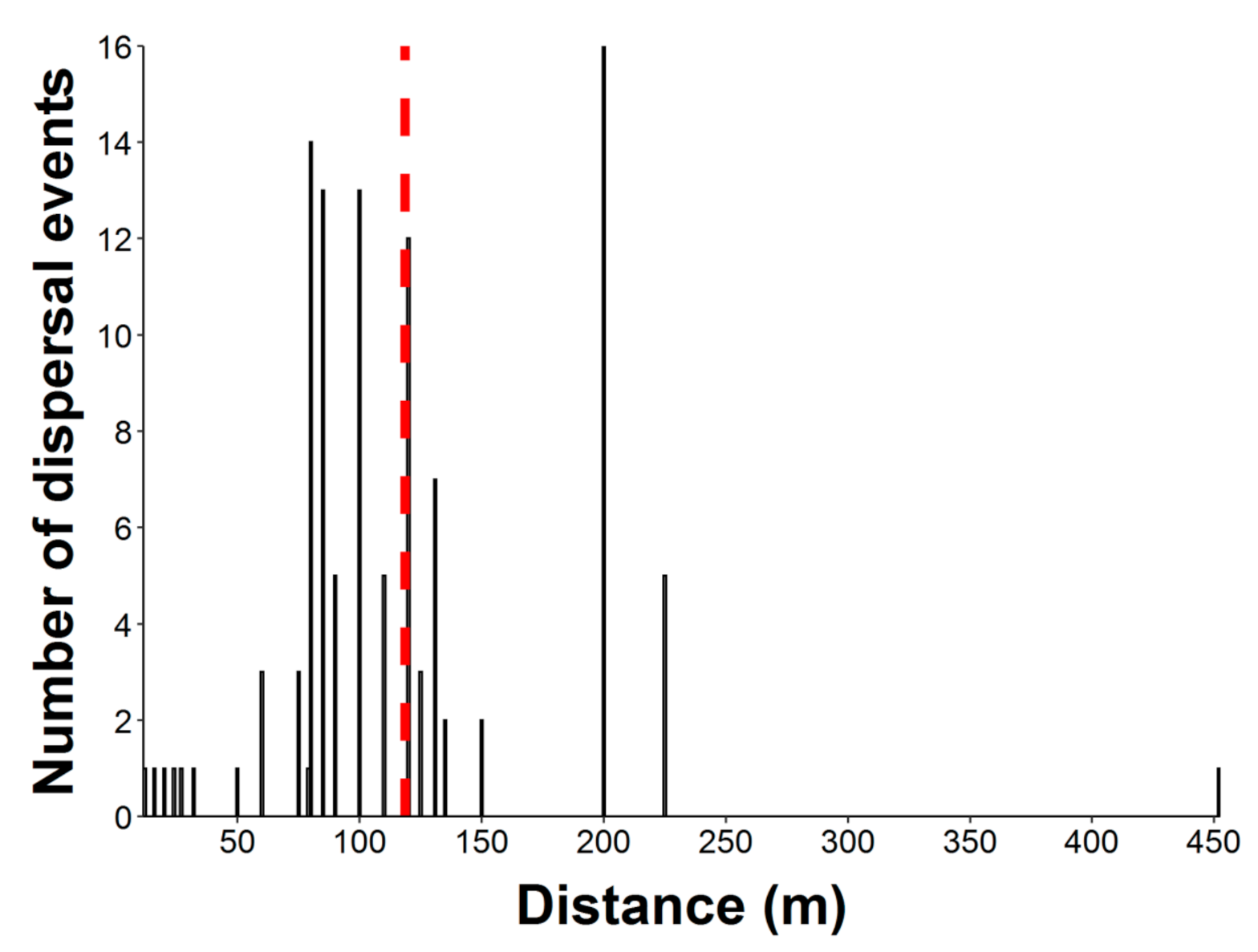
2.2. Seed Dispersal Distances
3. Discussion
3.1. Epizoochory in Parrots as a Widespread Mutualism
3.2. Ecological and Conservation Implications
4. Materials and Methods
4.1. Data Recording and Source
4.2. Seed Dispersal Distances
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bascompte, J.; Jordano, P. Plant-Animal Mutualistic Networks: The Architecture of Biodiversity. Annu. Rev. Ecol. Evol. Syst. 2007, 38, 567–593. [Google Scholar] [CrossRef]
- Jordano, P. Chasing Ecological Interactions. PLoS Biol. 2016, 14, e1002559. [Google Scholar] [CrossRef] [PubMed]
- Valiente-Banuet, A.; Aizen, M.A.; Alcántara, J.M.; Arroyo, J.; Cocucci, A.; Galetti, M.; García, M.B.; García, D.; Gómez, J.M.; Jordano, P.; et al. Beyond species loss: The extinction of ecological interactions in a changing world. Funct. Ecol. 2014, 29, 299–307. [Google Scholar] [CrossRef]
- Howe, H.F. Making dispersal syndromes and networks useful in tropical conservation and restoration. Glob. Ecol. Conserv. 2016, 6, 152–178. [Google Scholar] [CrossRef]
- Guimarães, P.R.; Pires, M.M.; Jordano, P.; Bascompte, J.; Thompson, J.N. Indirect effects drive coevolution in mutualistic networks. Nat. Cell Biol. 2017, 550, 511–514. [Google Scholar] [CrossRef]
- Traveset, A.; Willson, M.F. Effect of Birds and Bears on Seed Germination of Fleshy-Fruited Plants in Temperate Rainforests of Southeast Alaska. Oikos 1997, 80, 89. [Google Scholar] [CrossRef]
- Fleming, T.H.; Kress, W.J. The Ornaments of Life: Coevolution and Conservation in the Tropics, 1st ed.; University of Chicago Press: Chicago, IL, USA, 2013. [Google Scholar] [CrossRef]
- Wisz, M.S.; Pottier, J.; Kissling, W.D.; Pellissier, L.; Lenoir, J.; Damgaard, C.F.; Dormann, C.F.; Forchhammer, M.C.; Grytnes, J.; Guisan, A.; et al. The role of biotic interactions in shaping distributions and realised assemblages of species: Implications for species distribution modelling. Biol. Rev. 2012, 88, 15–30. [Google Scholar] [CrossRef]
- Baños-Villalba, A.; Blanco, G.; Díaz-Luque, J.A.; Dénes, F.V.; Hiraldo, F.; Tella, J.L. Seed dispersal by macaws shapes the landscape of an Amazonian ecosystem. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Hortal, J.; de Bello, F.; Diniz-Filho, J.A.F.; Lewinsohn, T.M.; Lobo, J.M.; Ladle, R.J. Seven Shortfalls that Beset Large-Scale Knowledge of Biodiversity. Annu. Rev. Ecol. Evol. Syst. 2015, 46, 523–549. [Google Scholar] [CrossRef]
- Sorensen, A.E. Seed dispersal by adhesion. Annu. Rev. Ecol. Evol. Syst. 1986, 17, 443–463. [Google Scholar] [CrossRef]
- Albert, A.; Mårell, A.; Picard, M.; Baltzinger, C. Using basic plant traits to predict ungulate seed dispersal potential. Ecography 2015, 38, 440–449. [Google Scholar] [CrossRef]
- Baltzinger, C.; Karimi, S.; Shukla, U. Plants on the Move: Hitch-Hiking with Ungulates Distributes Diaspores Across Landscapes. Front. Ecol. Evol. 2019, 7, 38. [Google Scholar] [CrossRef]
- Gorb, E.V.; Yang, S.; Priewe, J.; Gorb, S.N. The contact separation force of the fruit burrs from five plant taxa dispersing by epizoochory. Plant. Biosyst. Int. J. Deal. All Asp. Plant. Biol. 2019, 154, 38–48. [Google Scholar] [CrossRef]
- Van Leeuwen, C.H.A.; Tella, J.L.; Green, A.J. Editorial: Animal-Mediated Dispersal in Understudied Systems. Front. Ecol. Evol. 2020, 7, 508. [Google Scholar] [CrossRef]
- Van Leeuwen, C.H.A. Internal and External Dispersal of Plants by Animals: An Aquatic Perspective on Alien Interference. Front. Plant. Sci. 2018, 9, 153. [Google Scholar] [CrossRef]
- Figuerola, J.; Green, A.J. Dispersal of aquatic organisms by waterbirds: A review of past research and priorities for future studies. Freshw. Biol. 2002, 47, 483–494. [Google Scholar] [CrossRef]
- Heinken, T.; Schmidt, M.; von Oheimb, G.; Kriebitzsch, W.-U.; Ellenberg, H. Soil seed banks near rubbing trees indicate dispersal of plant species into forests by wild boar. Basic Appl. Ecol. 2006, 7, 31–44. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, H.; Zhang, Y.; Yao, H.; Yang, W.; Zhao, Y.; Ruan, X.; Xiang, Z. First evidence of epizoochorous seed dispersal by golden snub-nosed monkeys (Rhinopithecus roxellana) in temperate forest. Plant Ecol. 2018, 219, 417–427. [Google Scholar] [CrossRef]
- Liehrmann, O.; Jégoux, F.; Guilbert, M.-A.; Isselin-Nondedeu, F.; Saïd, S.; Locatelli, Y.; Baltzinger, C. Epizoochorous dispersal by ungulates depends on fur, grooming and social interactions. Ecol. Evol. 2018, 8, 1582–1594. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.M.; Ramos, J.A.; da Silva, L.P.; Timoteo, S.; Araújo, P.M.; Felgueiras, M.S.; Rosa, A.; Matos, C.; Encarnação, P.; Tenreiro, P.Q.; et al. Endozoochory largely outweighs epizoochory in migrating passerines. J. Avian Biol. 2013, 45, 59–64. [Google Scholar] [CrossRef]
- Choi, C.-Y.; Nam, H.-Y.; Chae, H.-Y. Exotic Seeds on the Feathers of Migratory Birds on a Stopover Island in Korea. J. Ecol. Environ. 2010, 33, 19–22. [Google Scholar] [CrossRef][Green Version]
- Nogales, M.; Heleno, R.; Traveset, A.; Vargas, P. Evidence for overlooked mechanisms of long-distance seed dispersal to and between oceanic islands. New Phytol. 2012, 194, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Toft, C.A.; Wright, T.F. Parrots of the Wild: A Natural History of the World’s Most Captivating Birds, 1st ed.; University of California Press: Los Angeles, CA, USA, 2015. [Google Scholar]
- Blanco, G.; Hiraldo, F.; Tella, J.L. Ecological functions of parrots: An integrative perspective from plant life cycle to ecosystem functioning. Emu Austral. Ornithol. 2017, 118, 36–49. [Google Scholar] [CrossRef]
- Blanco, G.; Hiraldo, F.; Rojas, A.; Dénes, F.V.; Tella, J.L. Parrots as key multilinkers in ecosystem structure and functioning. Ecol. Evol. 2015, 5, 4141–4160. [Google Scholar] [CrossRef]
- Montesinos-Navarro, A.; Hiraldo, F.; Tella, J.L.; Blanco, G. Network structure embracing mutualism–antagonism continuums increases community robustness. Nat. Ecol. Evol. 2017, 1, 1661–1669. [Google Scholar] [CrossRef] [PubMed]
- Tella, J.L.; Baños-Villalba, A.; Hernández-Brito, D.; Rojas, A.; Pacífico, E.; Díaz-Luque, J.A.; Carrete, M.; Blanco, G.; Hiraldo, F. Parrots as overlooked seed dispersers. Front. Ecol. Environ. 2015, 13, 338–339. [Google Scholar] [CrossRef]
- Tella, J.L.; Hiraldo, F.; Pacífico, E.; Díaz-Luque, J.A.; Dénes, F.V.; Fontoura, F.M.; Guedes, N.; Blanco, G. Conserving the Diversity of Ecological Interactions: The Role of Two Threatened Macaw Species as Legitimate Dispersers of “Megafaunal” Fruits. Diversity 2020, 12, 45. [Google Scholar] [CrossRef]
- Blanco, G.; Bravo, C.; Chamorro, D.; Lovas-Kiss, Á.; Hiraldo, F.; Tella, J.L. Herb endozoochory by cockatoos: Is ‘foliage the fruit’? Austral. Ecol. 2019, 45, 122–126. [Google Scholar] [CrossRef]
- Sebastián-González, E.; Hiraldo, F.; Blanco, G.; Hernández-Brito, D.; Romero-Vidal, P.; Carrete, M.; Gómez-Llanos, E.; Pacífico, E.C.; Díaz-Luque, J.A.; Dénes, F.V.; et al. The extent, frequency and ecological functions of food wasting by parrots. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Blanco, G.; Bravo, C.; Pacifico, E.C.; Chamorro, D.; Speziale, K.L.; Lambertucci, S.A.; Hiraldo, F.; Tella, J.L. Internal seed dispersal by parrots: An overview of a neglected mutualism. Peer J. 2016, 4, e1688. [Google Scholar] [CrossRef]
- Renton, K.; Salinas-Melgoza, A.; de Labra-Hernández, M.Á.; de la Parra-Martínez, S.M. Resource requirements of parrots: Nest site selectivity and dietary plasticity of Psittaciformes. J. Ornithol. 2015, 156, 73–90. [Google Scholar] [CrossRef]
- Couvreur, M.; Christiaen, B.; Verheyen, K.; Hermy, M. Large herbivores as mobile links between isolated nature reserves through adhesive seed dispersal. Appl. Veg. Sci. 2004, 7, 229–236. [Google Scholar] [CrossRef]
- Carlquist, S. The Biota of Long-Distance Dispersal. I. Principles of Dispersal and Evolution. Q. Rev. Biol. 1966, 41, 247–270. [Google Scholar] [CrossRef] [PubMed]
- Brunet, J. Plant colonization in heterogeneous landscapes: An 80-year perspective on restoration of broadleaved forest vegetation. J. Appl. Ecol. 2007, 44, 563–572. [Google Scholar] [CrossRef]
- Green, A.J.; Elmberg, J.; Lovas-Kiss, Á. Beyond Scatter-Hoarding and Frugivory: European Corvids as Overlooked Vectors for a Broad Range of Plants. Front. Ecol. Evol. 2019, 7, 133. [Google Scholar] [CrossRef]
- Traveset, A.; Robertson, A.W.; Rodríguez-Pérez, J. A review on the role of endozoochory in seed germination. In Seed Dispersal: Theory and Its Application in a Changing World, 1st ed.; Dennis, A.J., Green, R.J., Schupp, E.W., Westcott, D.A., Eds.; CABI Publishing: Wallingford, UK, 2007; Volume 4, pp. 78–103. [Google Scholar]
- Jordaan, L.A.; Johnson, S.D.; Downs, C.T. The role of avian frugivores in germination of seeds of fleshy-fruited invasive alien plants. Biol. Invasions 2011, 13, 1917–1930. [Google Scholar] [CrossRef]
- De Bie, T.; de Meester, L.; Brendonck, L.; Martens, K.; Goddeeris, B.; Ercken, D.; Hampel, H.; Denys, L.; Vanhecke, L.; van der Gucht, K.; et al. Body size and dispersal mode as key traits determining metacommunity structure of aquatic organisms. Ecol. Lett. 2012, 15, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, P.R.; Cardona, L.; Cárdenas, S.; Link, A. Oilbirds disperse large seeds at longer distance than extinct megafauna. Sci. Rep. 2021, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Whelan, C.J.; Wenny, D.G.; Marquis, R.J. Ecosystem Services Provided by Birds. Ann. N. Y. Acad. Sci. 2008, 1134, 25–60. [Google Scholar] [CrossRef] [PubMed]
- Cadotte, M.W.; Yasui, S.L.E.; Livingstone, S.; MacIvor, J.S. Are urban systems beneficial, detrimental, or indifferent for biological invasion? Biol. Invasions 2017, 19, 3489–3503. [Google Scholar] [CrossRef]
- Gaertner, M.; Wilson, J.R.U.; Cadotte, M.W.; MacIvor, J.S.; Zenni, R.D.; Richardson, D.M. Non-native species in urban environments: Patterns, processes, impacts and challenges. Biol. Invasions 2017, 19, 3461–3469. [Google Scholar] [CrossRef]
- Richardson, D.M.; Allsopp, N.; D’Antonio, C.M.; Milton, S.J.; Rejmánek, M. Plant invasions—The role of mutualisms. Biol. Rev. 2007, 75, 65–93. [Google Scholar] [CrossRef]
- Gosper, C.R.; Stansbury, C.D.; Vivian-Smith, G. Seed dispersal of fleshy-fruited invasive plants by birds: Contributing factors and management options. Divers. Distrib. 2005, 11, 549–558. [Google Scholar] [CrossRef]
- Buckley, Y.M.; Anderson, S.; Catterall, C.P.; Corlett, R.T.; Engel, T.; Gosper, C.R.; Nathan, R.; Richardson, D.M.; Setter, M.; Spiegel, O.; et al. Management of plant invasions mediated by frugivore interactions. J. Appl. Ecol. 2006, 43, 848–857. [Google Scholar] [CrossRef]
- Gelmi-Candusso, T.A.; Hämäläinen, A.M. Seeds and the City: The Interdependence of Zoochory and Ecosystem Dynamics in Urban Environments. Front. Ecol. Evol. 2019, 7, 41. [Google Scholar] [CrossRef]
- Cardador, L.; Lattuada, M.; Strubbe, D.; Tella, J.L.; Reino, L.; Figueira, R.; Carrete, M. Regional Bans on Wild-Bird Trade Modify Invasion Risks at a Global Scale. Conserv. Lett. 2017, 10, 717–725. [Google Scholar] [CrossRef]
- Menchetti, M.; Mori, E. Worldwide impact of alien parrots (Aves Psittaciformes) on native biodiversity and environment: A review. Ethol. Ecol. Evol. 2014, 26, 172–194. [Google Scholar] [CrossRef]
- Traveset, A.; Richardson, D.M. Mutualistic Interactions and Biological Invasions. Annu. Rev. Ecol. Evol. Syst. 2014, 45, 89–113. [Google Scholar] [CrossRef]
- Aizen, M.A.; Morales, C.L.; Morales, J.M. Invasive mutualists erode native pollination webs. PLoS Biol. 2018, 6, e31. [Google Scholar] [CrossRef]
- Molefe, K.L.; Tedder, M.J.; Thabethe, V.; Rushworth, I.; Downs, C.T. Role of native avian frugivores in germination facilitation and potential dispersal of invasive American bramble (Rubus cuneifolius) in South Africa. Biol. Invasions. 2020, 22, 1109–1120. [Google Scholar] [CrossRef]
- Ricciardi, A.; Hoopes, M.F.; Marchetti, M.P.; Lockwood, J.L. Progress toward understanding the ecological impacts of nonnative species. Ecol. Monogr. 2013, 83, 263–282. [Google Scholar] [CrossRef]
- Bravo, C.; Chamorro, D.; Hiraldo, F.; Speziale, K.; Lambertucci, S.A.; Tella, J.L.; Blanco, G. Physiological dormancy broken by endozoochory: Austral parakeets (Enicognathus ferrugineus) as legitimate dispersers of calafate (Berberis microphylla) in the Patagonian Andes. J. Plant. Ecol. 2020, 13, 538–544. [Google Scholar] [CrossRef]
- Blanco, G.; Tella, J.L.; Díaz-Luque, J.A.; Hiraldo, F. Multiple External Seed Dispersers Challenge the Megafaunal Syndrome Anachronism and the Surrogate Ecological Function of Livestock. Front. Ecol. Evol. 2019, 7, 328. [Google Scholar] [CrossRef]
- Luna, Á.; Romero-Vidal, P.; Hiraldo, F.; Tella, J.L. Cities may save some threatened species but not their ecological functions. Peer J. 2018, 6, e4908. [Google Scholar] [CrossRef] [PubMed]
- Tella, J.L.; Blanco, G.; Dénes, F.V.; Hiraldo, F. Overlooked Parrot Seed Dispersal in Australia and South America: Insights on the Evolution of Dispersal Syndromes and Seed Size in Araucaria Trees. Front. Ecol. Evol. 2019, 7, 82. [Google Scholar] [CrossRef]
- Couvreur, M.; Verheyen, K.; Vellend, M.; Lamoot, I.; Cosyns, E.; Hoffmann, M.; Hermy, M. Epizoochory by large herbivores: Merging data with models. Basic Appl. Ecol. 2008, 9, 204–212. [Google Scholar] [CrossRef]
- Montoya, D.; Zavala, M.A.; Rodríguez, M.A.; Purves, D.W. Animal Versus Wind Dispersal and the Robustness of Tree Species to Deforestation. Science 2008, 320, 1502–1504. [Google Scholar] [CrossRef] [PubMed]
- Tella, J.L.; Romero-Vidal, P.; Hiraldo, F.; Blanco, G.; Hernández-Brito, D.; Rosseto, F.; Toledo-González, B.; Pacífico, E.C.; Díaz-Luque, J.A.; Dénes, F.V.; et al. Roadside car surveys: Methodological constraints and solutions for estimating parrot abundances across the world. in preparation.

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Brito, D.; Romero-Vidal, P.; Hiraldo, F.; Blanco, G.; Díaz-Luque, J.A.; Barbosa, J.M.; Symes, C.T.; White, T.H.; Pacífico, E.C.; Sebastián-González, E.; et al. Epizoochory in Parrots as an Overlooked Yet Widespread Plant–Animal Mutualism. Plants 2021, 10, 760. https://doi.org/10.3390/plants10040760
Hernández-Brito D, Romero-Vidal P, Hiraldo F, Blanco G, Díaz-Luque JA, Barbosa JM, Symes CT, White TH, Pacífico EC, Sebastián-González E, et al. Epizoochory in Parrots as an Overlooked Yet Widespread Plant–Animal Mutualism. Plants. 2021; 10(4):760. https://doi.org/10.3390/plants10040760
Chicago/Turabian StyleHernández-Brito, Dailos, Pedro Romero-Vidal, Fernando Hiraldo, Guillermo Blanco, José A. Díaz-Luque, Jomar M. Barbosa, Craig T. Symes, Thomas H. White, Erica C. Pacífico, Esther Sebastián-González, and et al. 2021. "Epizoochory in Parrots as an Overlooked Yet Widespread Plant–Animal Mutualism" Plants 10, no. 4: 760. https://doi.org/10.3390/plants10040760
APA StyleHernández-Brito, D., Romero-Vidal, P., Hiraldo, F., Blanco, G., Díaz-Luque, J. A., Barbosa, J. M., Symes, C. T., White, T. H., Pacífico, E. C., Sebastián-González, E., Carrete, M., & Tella, J. L. (2021). Epizoochory in Parrots as an Overlooked Yet Widespread Plant–Animal Mutualism. Plants, 10(4), 760. https://doi.org/10.3390/plants10040760