Efficiency of Herkogamy in Narcissus bulbocodium (Amaryllidaceae)
Abstract
1. Introduction
1.1. Plant Species
1.2. Study Area
2. Results
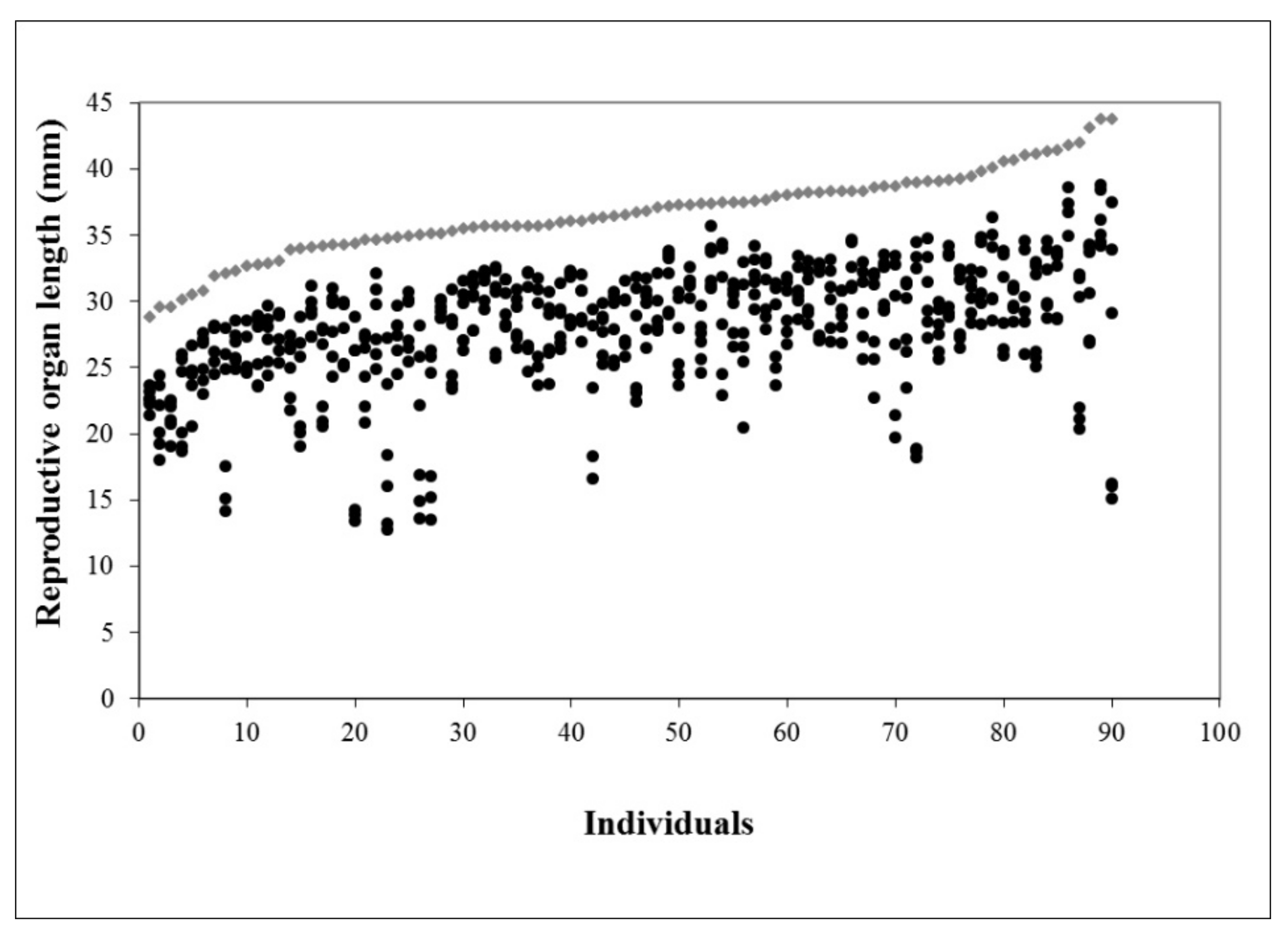
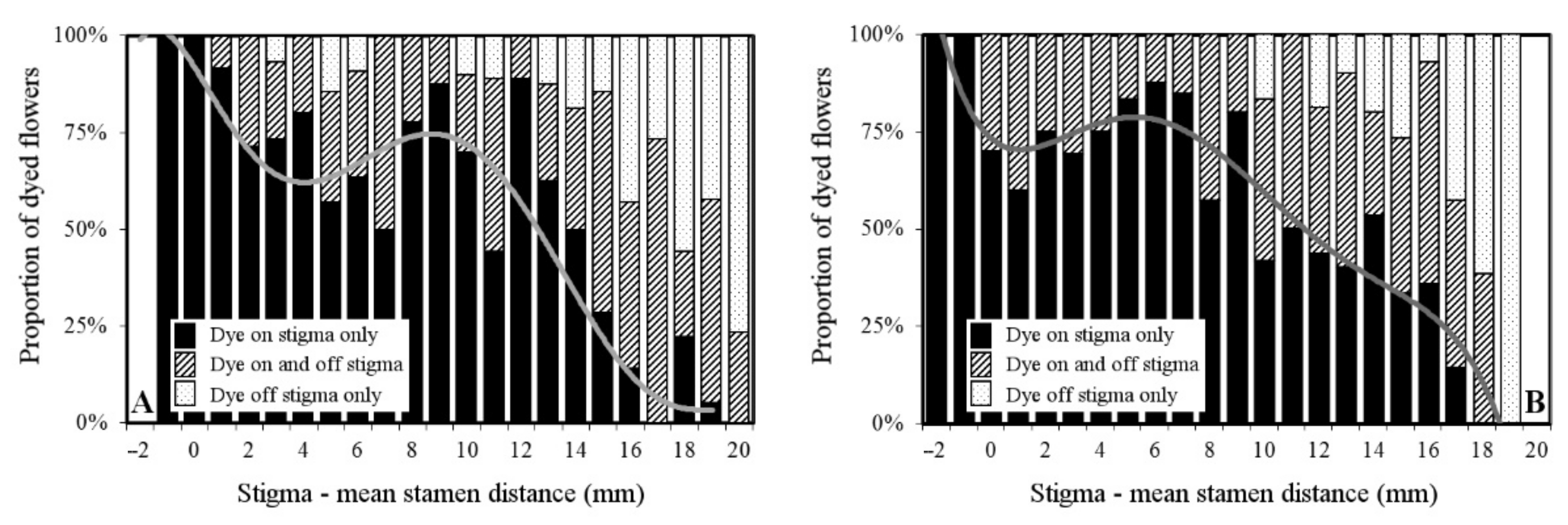
2.1. Floral Morphology and Female Fitness
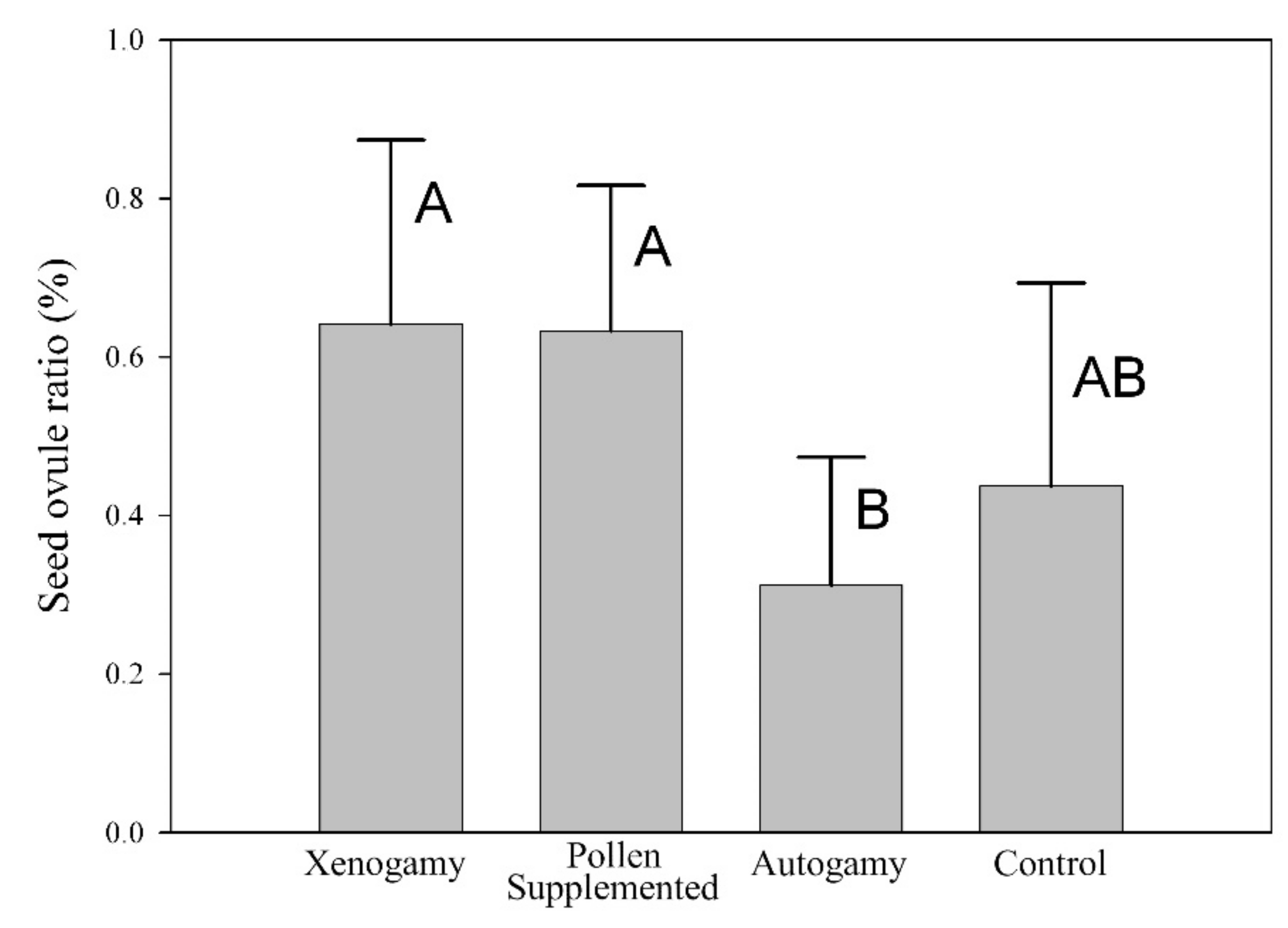
2.2. Mating System
2.3. Self-Interference and Herkogamy
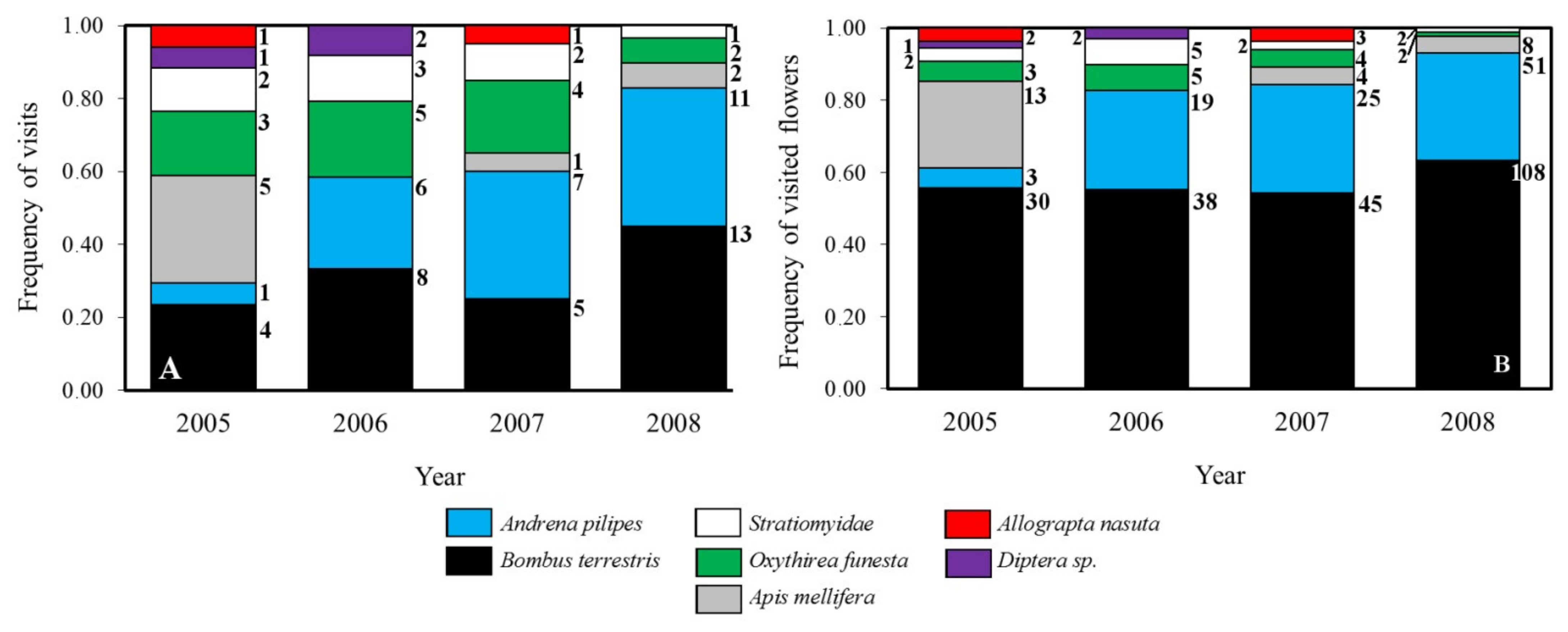
2.4. Pollinator’s Assemblage
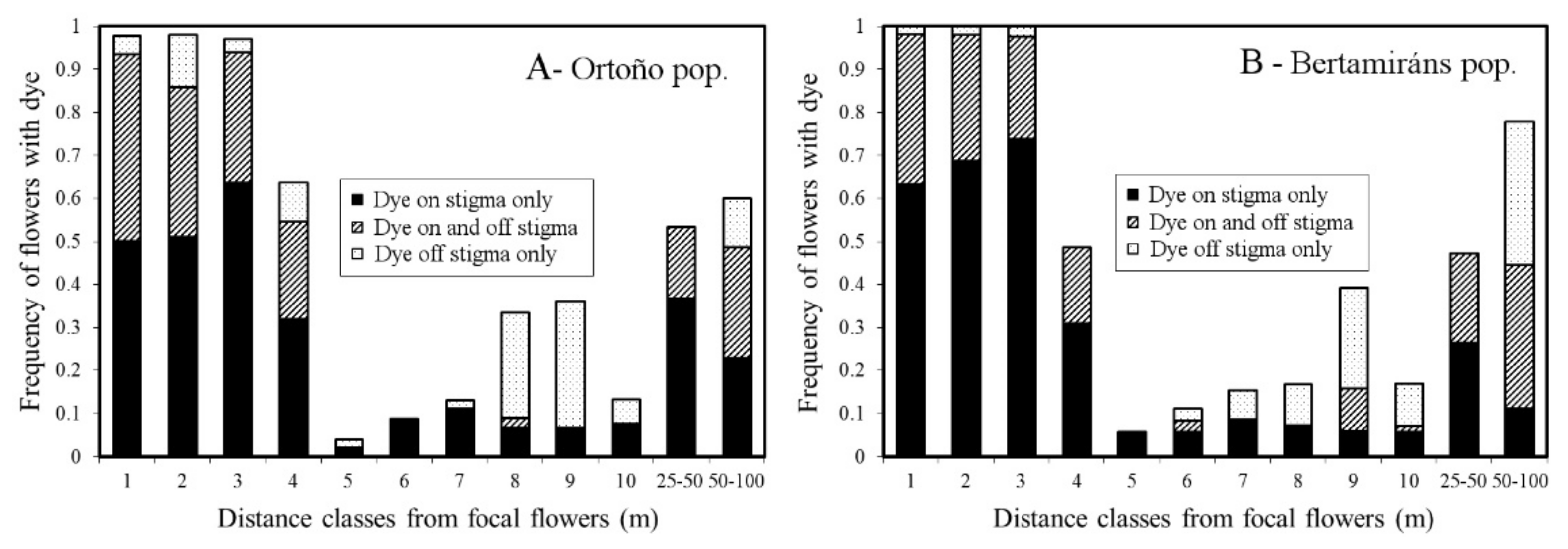
2.5. Pollen Dispersal Range and Precision
3. Discussion
3.1. Floral Morphology and Integration
3.2. Integration, Pollination, and Pollination Range
3.3. Mating System and Pollen Limitation
3.4. Herkogamy and Pollination
4. Materials and Methods
4.1. Floral Morphology and Female Fitness
4.2. Pollen Loads on Stigmas
4.3. Mating System
4.4. Self-Interference and Herkogamy
4.5. Pollinator’s Assemblage
4.6. Pollen Dispersal Range and Precision
4.7. Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Armbruster, W.S. Evolution of floral morphology and function: An integrative approach to adaptation, constraint, and compromise in Dalechampia (Euphorbiaceae). In Floral Biology. Studies on Floral Evolution in Animal-Pollinated Plants; Lloyd, D.G., Barrett, S.C.H., Eds.; Chapman and Hall: New York, NY, USA, 1996; pp. 241–272. [Google Scholar]
- Herrera, C.M. Floral traits and plant adaptation to insect pollinators: A devil’s advocate approach. In Floral Biology. Studies on Floral Evolution in Animal-Pollinated Plants; Lloyd, D.G., Barrett, S.C.H., Eds.; Chapman and Hall: New York, NY, USA, 1996; pp. 65–87. [Google Scholar]
- Medel, R.; Valiente, A.; Botto-Mahan, C.; Pohl, N.; Carvallo, G.; Pérez, F.; Navarro, L. The influence of insects and hummingbirds on the geographical variation of the flower phenotype in Mimulus luteus (Phrymaceae). Ecography 2007, 30, 812–818. [Google Scholar] [CrossRef]
- Berg, R.L. The ecological significance of correlation Pleiades. Evolution 1960, 14, 171–180. [Google Scholar] [CrossRef]
- Armbruster, W.S.; Di Stilio, V.S.; Tuxill, J.D.; Flores, T.C.; Runk, J.L.V. Covariance and decoupling of floral and vegetative traits in nine Neotropical plants: A re-evaluation of Berg’s correlation-pleiades concept. Am. J. Bot. 1999, 86, 39–55. [Google Scholar] [CrossRef]
- Cresswell, J.E. Stabilizing selection and the structural variability of flowers within species. Ann. Bot. 1998, 81, 463–473. [Google Scholar] [CrossRef]
- Cresswell, J.E. Manipulation of female architecture in flowers reveals a narrow optimum for pollen deposition. Ecology 2000, 81, 3244–3249. [Google Scholar] [CrossRef]
- Barrett, S.C.H. Sexual interference of the floral kind. Heredity 2002, 88, 154–159. [Google Scholar] [CrossRef]
- Cesaro, A.C.; Thompson, J.D. Darwin’s cross-promotion hypothesis and the evolution of stylar polymorphism. Ecol. Lett. 2004, 7, 1209–1215. [Google Scholar] [CrossRef]
- Charlesworth, D.; Charlesworth, B. Inbreeding depression and its evolutionary consequences. Annu. Rev. Ecol. Evol. Syst. 1987, 18, 237–268. [Google Scholar] [CrossRef]
- Cesaro, A.C.; Barrett, S.C.H.; Maurice, S.; Vaissiere, B.E.; Thompson, J.D. An experimental evaluation of self-interference in Narcissus assoanus: Functional and evolutionary implications. J. Evol. Biol. 2004, 17, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- Webb, C.J.; Lloyd, D.G. The avoidance of interference between the presentation of pollen and stigmas in angiosperms. II. Herkogamy. N. Z. J. Bot. 1986, 24, 163–178. [Google Scholar] [CrossRef]
- Armbruster, W.S.; Pélabon, C.; Hansen, T.F.; Bolstad, G.H. Macroevolutionary patterns of pollination accuracy: A comparison of three genera. New Phytol. 2009, 183, 600–617. [Google Scholar] [CrossRef]
- Medrano, M.; Herrera, C.M.; Barrett, S.C.H. Herkogamy and mating patterns in the self-compatible daffodil Narcissus longispathus. Ann. Bot. 2005, 95, 1105–1111. [Google Scholar] [CrossRef] [PubMed]
- Brunet, J.; Eckert, C.G. Effects of floral morphology and display on outcrossing in blue columbine, Aquilegia caerulea (Ranunculaceae). Funct. Ecol. 1998, 12, 596–606. [Google Scholar] [CrossRef]
- Karron, J.D.; Jackson, R.T.; Thumser, N.N.; Schlicht, S.L. Outcrossing rates of individual Mimulus rigens genets are correlated with anther-stigma separation. Heredity 1997, 79, 365–370. [Google Scholar]
- Aedo, C. Narcissus L. In Flora Ibérica; Aedo, C., Herrero, A., Eds.; Real Jardín Botánico de Madrid, CSIC: Madrid, Spain, 2005; Volume 20, pp. 340–397. [Google Scholar]
- INM (Instituto Nacional de Meteorología). Calendario Meteorológico 2005; Serie Monografías; Ministerio de Medio Ambiente: Madrid, Spain, 2006.
- Jules, E.S.; Rathcke, B.J. Mechanisms of reduced Trillium recruitment along edges of old-growth forest fragments. Conserv. Biol. 1999, 13, 784–793. [Google Scholar] [CrossRef]
- Campbell, D.R. Measurements of selection in a hermaphroditic plant: Variation in male and female pollination success. Evolution 1989, 43, 318–334. [Google Scholar] [CrossRef]
- Ordano, M.; Fornoni, J.; Boege, K.; Domínguez, C.A. The adaptive value of phenotypic floral integration. New Phytol. 2008, 179, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Ferrero, V.; Chapela, I.; Arroyo, J.; Navarro, L. Reciprocal style polymorphisms are not so easily categorized: The case of heterostyly in Lithodora and Glandora (Boraginaceae). Plant Biol. 2011, 13, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Herrera, C.M.; Cerdá, X.; García, M.B.; Guitián, J.; Medrano, M.; Rey, P.; Sánchez-Lafuente, A.M. Floral integration, phenotypic covariance structure and pollinator variation in bumblebee-pollinated Helleborus foetidus. J. Evol. Biol. 2002, 15, 108–121. [Google Scholar] [CrossRef]
- Garófalo, C.A. Bionomics of Bombus (fervodobombus) morio 2. Body size and length of life of workers. J. Apic. Res. 1978, 17, 130–136. [Google Scholar] [CrossRef]
- Wolf, S.; Moritz, R.F.A. Foraging distance in Bombus terrestris L. (Hymenoptera: Apidae). Apidologie 2008, 39, 419–427. [Google Scholar] [CrossRef]
- Cruden, R.W. Pollen-ovule ratios: A conservative indicator of breeding systems in flowering plants. Evolution 1977, 31, 32–46. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, D.G. Self- and cross-fertilization in plants. II. The selection of self-fertilization. Int. J. Plant Sci. 1992, 153, 370–380. [Google Scholar] [CrossRef]
- Navarro, L.; Guitián, J. The role of floral biology and breeding system on the reproductive success of the endemic P. viscosa Rothm. (Caryophyllaceae). Biol. Conserv. 2002, 103, 125–132. [Google Scholar] [CrossRef]
- Vale, A.; Rojas, D.; Álvarez, J.C.; Navarro, L. Breeding system and factors limiting fruit production in the nectarless orchid Broughtonia lindenii. Plant Biol. 2011, 13, 51–61. [Google Scholar] [CrossRef]
- Guitián, J.; Guitián, P.; Medrano, M.; Sánchez, J.M. Variation in floral morphology and individual fecundity in Erythronium dens-canis (Liliaceae). Ecography 1999, 22, 708–714. [Google Scholar] [CrossRef]
- Motten, A.F. Pollination ecology of the spring wildflower community of a temperate deciduous forest. Ecol. Monogr. 1986, 56, 21–42. [Google Scholar] [CrossRef]
- Ferrero, V.; Arroyo, J.; Vargas, P.; Thompson, J.D.; Navarro, L. Evolutionary transitions of style polymorphism in Lithodora (Boraginaceae). Persp. Plant Ecol. Evol. Syst. 2009, 11, 111–125. [Google Scholar] [CrossRef]
- Lloyd, D.G.; Webb, C.J. The evolution of heterostyly. In Evolution and Function of Heterostyly; Barrett, S.C.H., Ed.; Springer: Berlin, Germany, 1992; pp. 151–178. [Google Scholar]
- Lloyd, D.G.; Webb, C.J. The selection of heterostyly. In Evolution and Function of Heterostyly; Barrett, S.C.H., Ed.; Springer: Berlin, Germany, 1992; pp. 179–207. [Google Scholar]
- Eckert, C.G.; Schaefer, A. Does self-pollination provide reproductive assurance in Aquilegia canadensis (Ranunculaceae)? Amer. J. Bot. 1998, 85, 9191–9924. [Google Scholar] [CrossRef]
- Barrett, S.C.H. Evolution of sex: The evolution of plant sexual diversity. Nat. Rev. Genet. 2002, 3, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Kelly, L.M. A cladistic analysis of Asarum (Aristolochiaceae) and implications for the evolution of herkogamy. Am. J. Bot. 1997, 84, 1752–1765. [Google Scholar] [CrossRef]
- Dafni, A.; Maués, M.M. A rapid and simple procedure to determine stigma receptivity. Sex Plant Reprod. 1998, 11, 177–180. [Google Scholar] [CrossRef]
- Navarro, L. Is the dichogamy of Salvia verbenaca (Lamiaceae) an effective barrier to self-fertilization? Plant Syst. Evol. 1997, 207, 111–117. [Google Scholar] [CrossRef]
- Larrinaga, A.R.; Guitián, P.; Garrido, J.L.; Guitián, J. Floral morphology and reproductive success in herkogamous Narcissus cyclamineus (Amaryllidaceae). Plant Syst. Evol. 2009, 278, 149–157. [Google Scholar] [CrossRef][Green Version]
- Navarro, L.; Ayensa, G.; Ferrero, V.; Sánchez, J.M. The avoidance of self-interference in the endemic daffodil Narcissus cyclamineus (Amaryllidaceae). Plant Ecol. 2012, 213, 1813–1822. [Google Scholar] [CrossRef]
- Marques, I.; Fuertes Aguilar, J.; Martins-Louçao, M.A.; Moharrek, F.; Nieto Feliner, G. A three-genome five-gene comprehensive phylogeny of the bulbous genus Narcissus (Amaryllidaceae) challenges current classifications and reveals multiple hybridization events. Taxon 2017, 66, 832–854. [Google Scholar] [CrossRef]
- Cruden, R.W. Pollen grains: Why so many? Plant Syst. Evol. 2000, 222, 143–165. [Google Scholar] [CrossRef]
- Castro, S.; Silveira, P.; Navarro, L. How flower biology and breeding system affect the reproductive success of the narrow endemic Polygala vayredae Costa (Polygalaceae)? Bot. J. Linn. Soc. 2008, 157, 67–81. [Google Scholar] [CrossRef]
- Dafni, A.; Kevan, P.G.; Husband, B.C. Practical Pollination Biology; Enviroquest: Cambridge, ON, Canada, 2005. [Google Scholar]
- Herrera, C.M. Pollinator abundance, morphology, and flower visitation rate: Analysis of the “quantity” component in a plant-pollinator system. Oecologia 1989, 80, 241–248. [Google Scholar] [CrossRef]
- Waser, N.M.; Price, M.V. A comparison of pollen and fluorescent dye carry-over by natural pollinators of Ipomopsis aggregata (Polemoniaceae). Ecology 1982, 63, 1168–1172. [Google Scholar] [CrossRef]
- Adler, L.S.; Irwin, R.E. Comparison of pollen transfer dynamics by multiple floral visitors: Experiments with pollen and fluorescent dye. Ann. Bot. 2006, 97, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Rademaker, M.C.J.; de Jong, T.J.; Klinkhamer, P.G.L. Pollen dynamics of bumble-bee visitation on Echium vulgare. Funct. Ecol. 1997, 11, 554–563. [Google Scholar] [CrossRef]
- Waser, N.M. Comparative pollen and dye transfer by pollinators of Delphinium nelsonii. Funct. Ecol. 1988, 2, 41–48. [Google Scholar] [CrossRef]
- Thomson, J.D.; Price, M.V.; Waser, N.M.; Stratton, D.A. Comparative studies of pollen and fluorescent dye transport by bumble bees visiting Erythronium grandiflorum. Oecologia 1986, 69, 561–566. [Google Scholar] [CrossRef] [PubMed]
- R Development Core Team. R: A Language and Environment for Statistical Computing; v. 3.5.1, 2018; R Foundation for Statistical Computing: Vienna, Austria, 2018; ISBN 3-900051-07-0. Available online: http://www.R-project.org (accessed on 3 September 2018).

| Ovary Length (mm) | Corolla Length (mm) | Style Length (mm) | Stamen Length (mm) | Stamen Amplitude (mm) | Herkogamy (mm) | Stigma Exertion (mm) | Pollen (n) | Ovule (n) | P/O | |
|---|---|---|---|---|---|---|---|---|---|---|
| Min | 4.59 | 27.15 | 28.77 | 18.53 | 1.43 | 1.69 | −3.45 | 124398 | 97 | 832.9 |
| Max | 14.67 | 42.88 | 43.77 | 36.89 | 16.26 | 10.01 | 7.01 | 214202 | 169 | 1650.0 |
| Mean | 8.64 | 34.58 | 36.58 | 28.05 | 6.52 | 5.25 | 2.00 | 165089 | 131 | 1308.6 |
| CV (%) | 21.06 | 9.77 | 8.69 | 12.16 | 48.65 | 32.18 | 36.89 | 15.42 | 20.41 | 23.64 |
| Variable | Axis | ||
|---|---|---|---|
| I | II | III | |
| Corolla length | 0.90 | −0.24 | 0.37 |
| Style length | 0.90 | −0.34 | −0.19 |
| Stamen length | 0.86 | 0.42 | −0.20 |
| Stamen amplitude | −0.16 | −0.97 | −0.11 |
| % variance explained | 59.79 | 32.35 | 5.58 |
| Variable | b | β | t | p |
|---|---|---|---|---|
| Constant | 447.02 | 4.91 | <0.001 | |
| Herkogamy distance | −23.64 | −0.15 | −1.39 | 0.17 |
| Stigma exertion | −42.64 | −0.33 | −3.25 | 0.002 |
| Scores on PCA1 | 7.59 | 0.05 | 0.45 | 0.65 |
| Scores on PCA2 | 18.57 | 0.08 | 0.76 | 0.45 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro, L.; Ayensa, G.; Sánchez, J.M. Efficiency of Herkogamy in Narcissus bulbocodium (Amaryllidaceae). Plants 2021, 10, 648. https://doi.org/10.3390/plants10040648
Navarro L, Ayensa G, Sánchez JM. Efficiency of Herkogamy in Narcissus bulbocodium (Amaryllidaceae). Plants. 2021; 10(4):648. https://doi.org/10.3390/plants10040648
Chicago/Turabian StyleNavarro, Luis, Garbiñe Ayensa, and José María Sánchez. 2021. "Efficiency of Herkogamy in Narcissus bulbocodium (Amaryllidaceae)" Plants 10, no. 4: 648. https://doi.org/10.3390/plants10040648
APA StyleNavarro, L., Ayensa, G., & Sánchez, J. M. (2021). Efficiency of Herkogamy in Narcissus bulbocodium (Amaryllidaceae). Plants, 10(4), 648. https://doi.org/10.3390/plants10040648







