The Interactive Effect of Elevated CO2 and Herbivores on the Nitrogen-Fixing Plant Alnus incana ssp. rugosa
Abstract
1. Introduction
2. Results
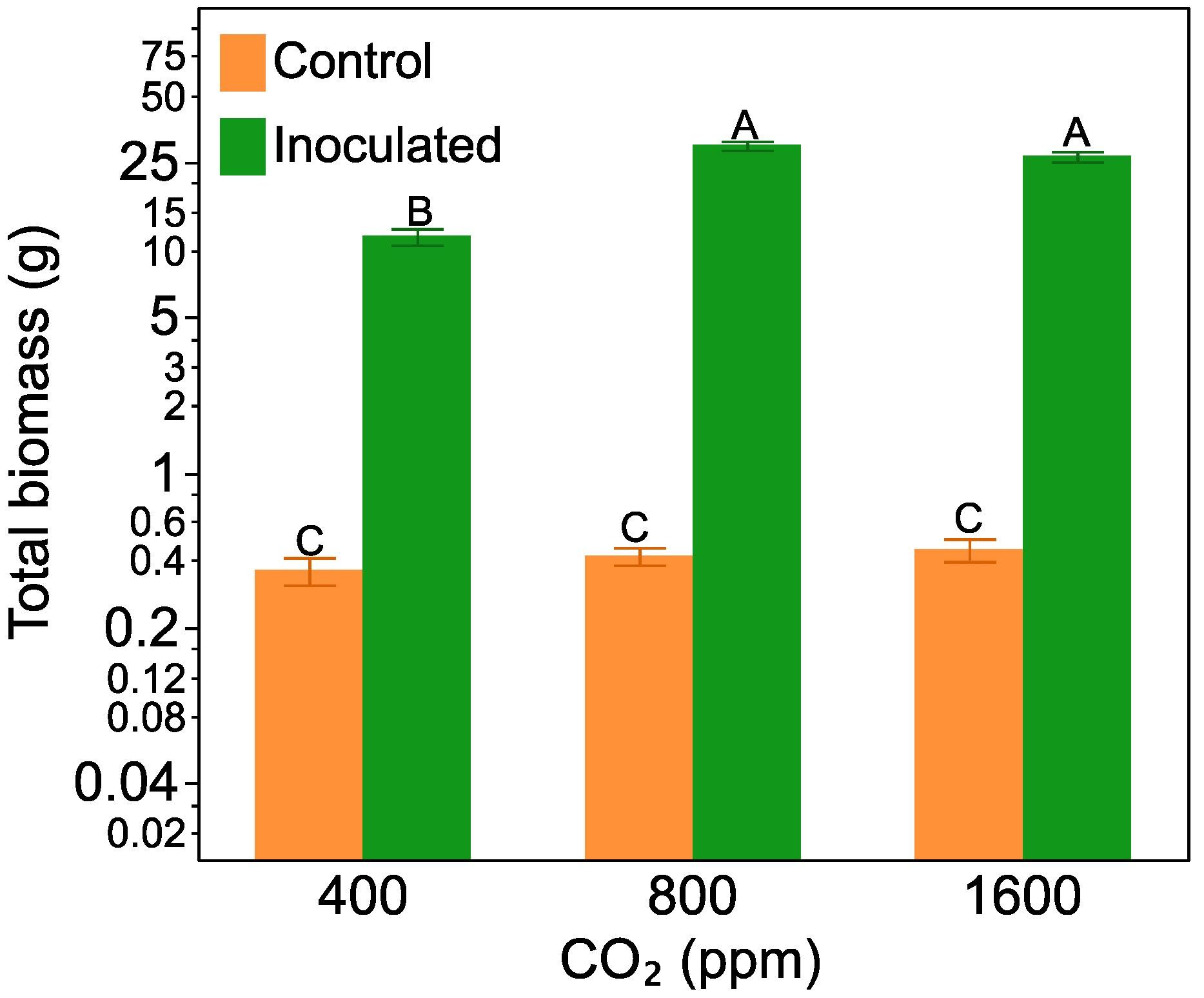
2.1. Plant Biomass
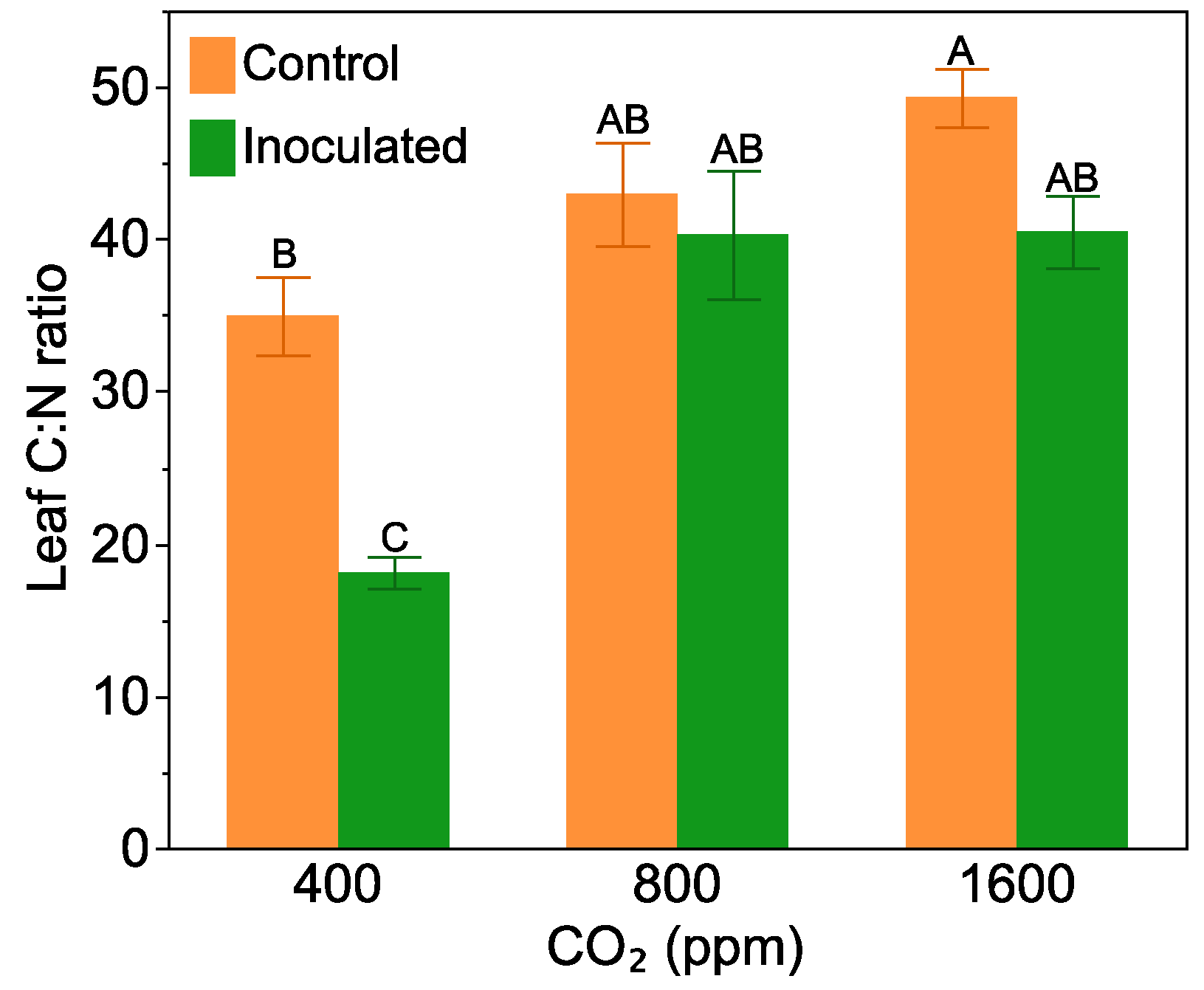
2.2. Leaf C:N Ratio
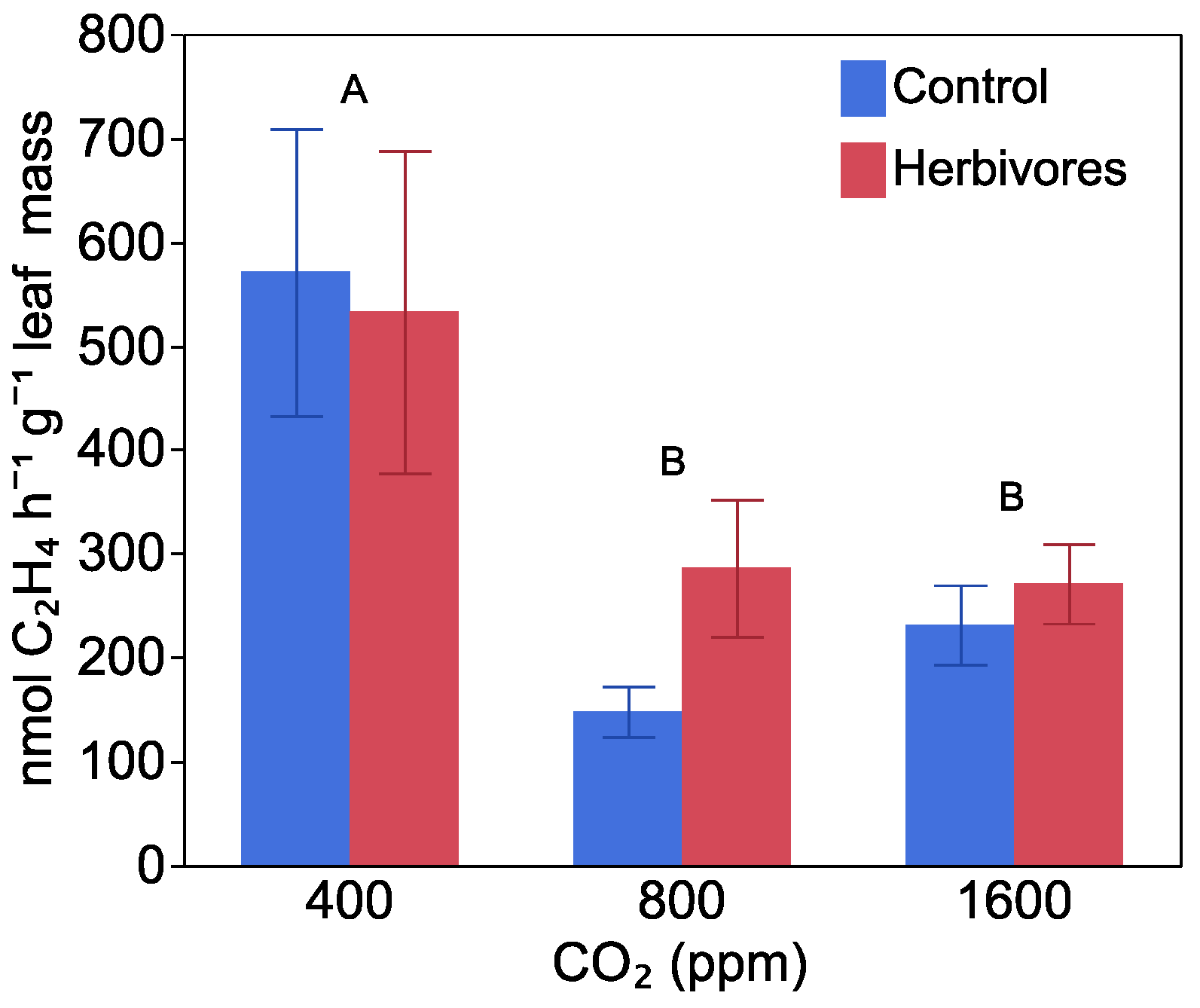
2.3. Nitrogen Fixation
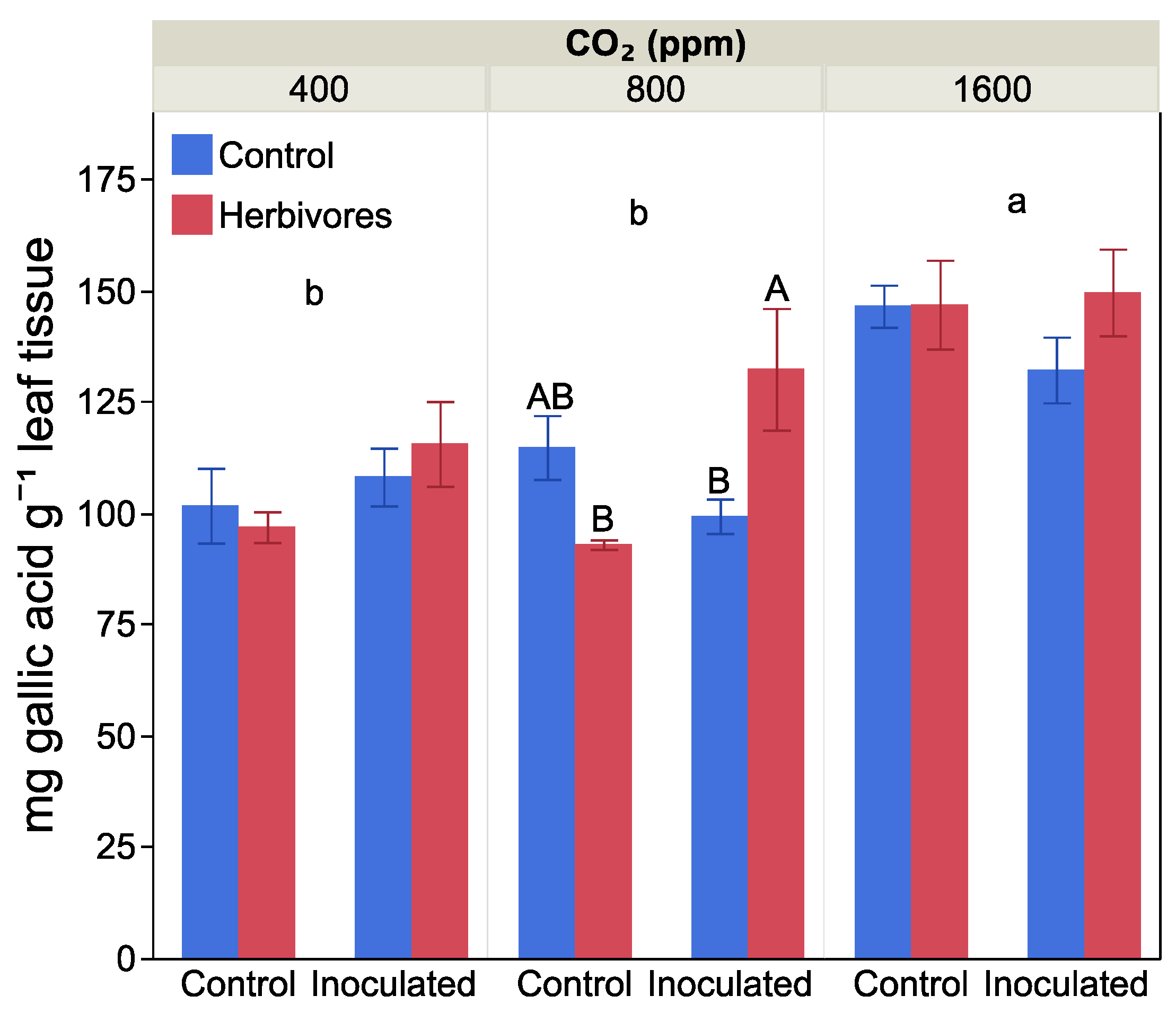
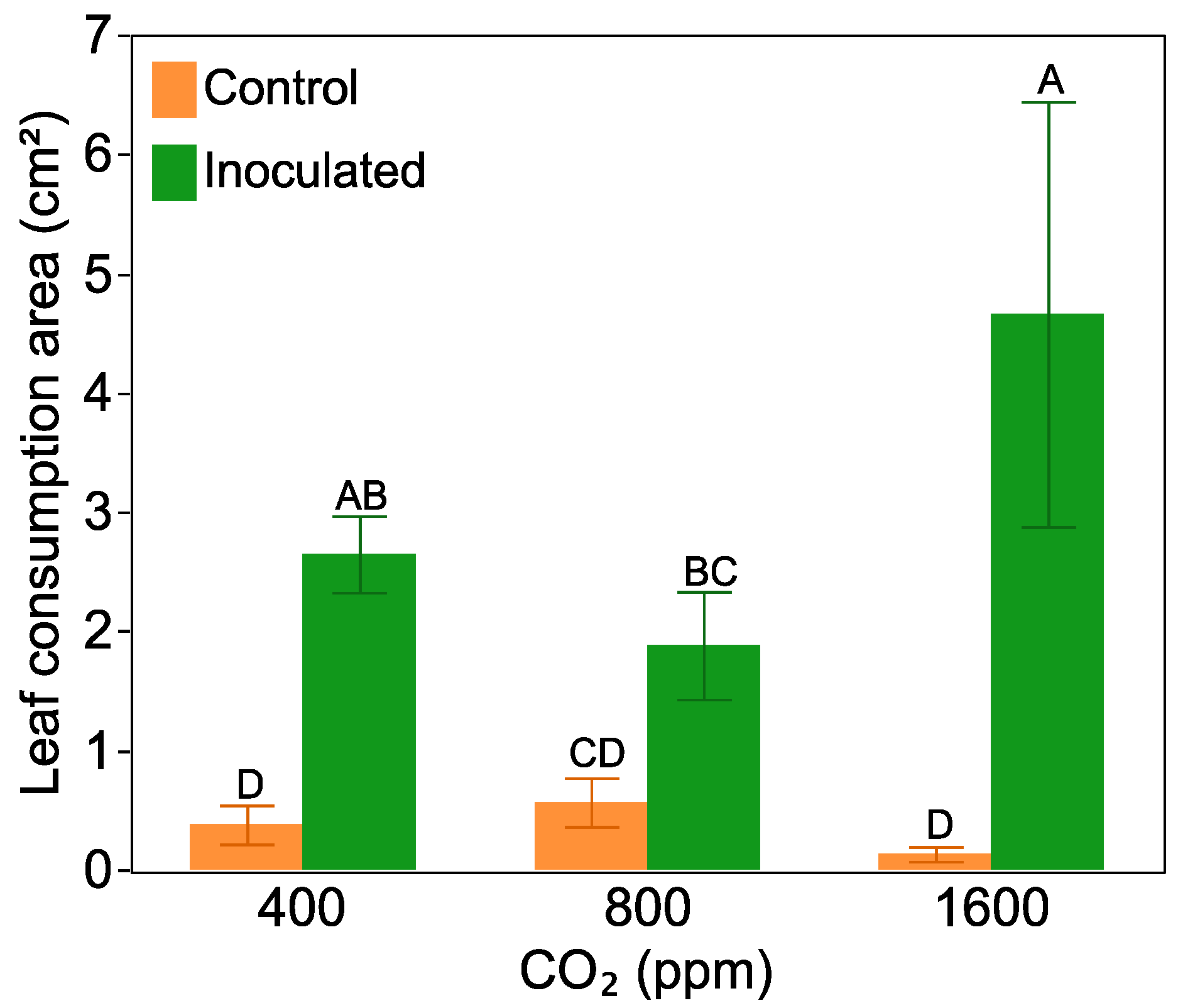
2.4. Leaf Damage and Antiherbivore Compounds
2.5. Herbivore Choice Experiment
3. Discussion
4. Materials and Methods
4.1. Plant Growth Condition and Treatments
4.2. Herbivore Choice Experiment
4.3. Herbivores Treatment
4.4. Nitrogen Fixation
4.5. C-Based Antiherbivore Compounds
4.6. N-Based Antiherbivore Compounds
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Steemans, P.; le Hérissé, A.; Melvin, J.; Miller, M.A.; Paris, F.; Verniers, J.; Wellman, C.H. Origin and radiation of the earliest vascular land plants. Science 2009, 324, 353. [Google Scholar] [CrossRef]
- Berner, R.A. Geocarbsulf: A Combined model for phanerozoic atmospheric O2 and CO2. Geochim. Cosmochim. Acta 2006, 70, 5653–5664. [Google Scholar] [CrossRef]
- Li, H.-L.; Wang, W.; Mortimer, P.E.; Li, R.-Q.; Li, D.-Z.; Hyde, K.D.; Xu, J.-C.; Soltis, D.E.; Chen, Z.-D. Large-scale phylogenetic analyses reveal multiple gains of actinorhizal nitrogen-fixing symbioses in angiosperms associated with climate change. Sci. Rep. 2015, 5, 14023. [Google Scholar] [CrossRef]
- Tajika, E. Climate change during the last 150 million years: Reconstruction from a carbon cycle model. Earth Planet. Sci. Lett. 1998, 160, 695–707. [Google Scholar] [CrossRef]
- Bice, K.L.; Norris, R.D. Possible atmospheric CO2 extremes of the Middle Cretaceous (late Albian–Turonian). Paleoceanography 2002, 17, 1017–1070. [Google Scholar] [CrossRef]
- Werner, G.D.A.; Cornwell, W.K.; Sprent, J.I.; Kattge, J.; Kiers, E.T. A single evolutionary innovation drives the deep evolution of symbiotic N2-fixation in angiosperms. Nat. Commun. 2014, 5, 4087. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.; Dyckmans, J.; Flessa, H. Effects of elevated carbon dioxide concentration on growth and N2 fixation of young Robinia pseudoacacia. Tree Physiol. 2004, 24, 323–330. [Google Scholar] [CrossRef][Green Version]
- Rogers, A.; Gibon, Y.; Stitt, M.; Morgan, P.B.; Bernacchi, C.J.; Ort, D.R.; Long, S.P. Increased C availability at elevated carbon dioxide concentration improves N assimilation in a legume. Plant Cell Environ. 2006, 29, 1651–1658. [Google Scholar] [CrossRef] [PubMed]
- Parvin, S.; Uddin, S.; Bourgault, M.; Roessner, U.; Tausz-Posch, S.; Armstrong, R.; O’Leary, G.; Fitzgerald, G.; Tausz, M. Water availability moderates N2 fixation benefit from elevated [CO2]: A 2-year free-air CO2 enrichment study on lentil (Lens culinaris MEDIK.) in a water limited agroecosystem. Plant Cell Environ. 2018, 41, 2418–2434. [Google Scholar] [CrossRef] [PubMed]
- Robinson, E.A.; Ryan, G.D.; Newman, J. A meta-analytical review of the effects of elevated CO2 on plant-arthropod interactions highlights the importance of interacting environmental and biological variables. New Phytol. 2012, 194, 321–336. [Google Scholar] [CrossRef]
- Chen, H.; Markham, J. Ancient CO2 levels favor nitrogen fixing plants over a broader range of soil N compared to present. Sci. Rep. 2021, 11, 3038. [Google Scholar] [CrossRef]
- Van Velzen, R.; Doyle, J.J.; Geurts, R. A resurrected scenario: Single gain and massive loss of nitrogen-fixing nodulation. Trends Plant Sci. 2019, 24, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Swensen, S.M. The evolution of actinorhizal symbioses: Evidence for multiple origins of the symbiotic association. Am. J. Bot. 1996, 83, 1503–1512. [Google Scholar] [CrossRef]
- Prior, S.A.; Runion, G.; Marble, S.; Rogers, H.H.; Gilliam, C.H.; Torbert, H. A review of elevated atmospheric CO2 effects on plant growth and water relations: Implications for horticulture. HortScience 2011, 46, 158–162. [Google Scholar] [CrossRef]
- Bugbee, B.; Spanarkel, B.; Johnson, S. CO2 crop growth enhancement and toxicity in wheat and rice. Adv. Space Res. 1994, 14, 257–267. [Google Scholar] [CrossRef]
- Li, Q.; Lai, L.; Du, H.; Cai, W.; Guan, T.; Zhang, X.; Jiang, L.; Zheng, Y.; Yu, Y.; Gao, Y.; et al. Elevated CO2 concentrations affect the growth patterns of dominant C3 and C4 shrub species differently in the Mu Us Sandy Land of Inner Mongolia. Botany 2017, 95, 869–877. [Google Scholar] [CrossRef]
- Coley, P.D. Possible effects of climate change on plant/herbivore interactions in moist tropical forests. Clim. Chang. 1998, 39, 455–472. [Google Scholar] [CrossRef]
- Coley, P.D.; Massa, M.; Lovelock, C.E.; Winter, K. Effects of elevated CO2 on foliar chemistry of saplings of nine species of tropical tree. Oecologia 2002, 133, 62–69. [Google Scholar] [CrossRef]
- Agrell, J.; Anderson, P.; Oleszek, W.; Stochmal, A.; Agrell, C. Elevated CO2 levels and herbivore damage alter host plant preferences. Oikos 2006, 112, 63–72. [Google Scholar] [CrossRef]
- Karowe, D.N. Are legume-feeding herbivores buffered against direct effects of elevated carbon dioxide on host plants? A test with the sulfur butterfly, Colias philodice. Glob. Chang. Biol. 2007, 13, 2045–2051. [Google Scholar] [CrossRef]
- DeLucia, E.H.; Nabity, P.D.; Zavala, J.A.; Berenbaum, M.R. Climate change: Resetting plant- insect interactions. Plant Physiol. 2012, 160, 1677–1685. [Google Scholar] [CrossRef] [PubMed]
- Mithöfer, A.; Boland, W. Plant defense against herbivores: Chemical aspects. Annu. Rev. Plant Biol. 2012, 63, 431–450. [Google Scholar] [CrossRef]
- Tscharntke, T.; Thiessen, S.; Dolch, R.; Boland, W. Herbivory, induced resistance, and interplant signal transfer in Alnus glutinosa. Biochem. Syst. Ecol. 2001, 29, 1025–1047. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Tilman, D. Responses of legumes to herbivores and nutrients during succession on a nitrogen-poor soil. Ecology 1995, 76, 2648–2655. [Google Scholar] [CrossRef]
- Kirschbaum, M.U.F. Does enhanced photosynthesis enhance growth? Lessons learned from CO2 Enrichment Studies. Plant Physiol. 2011, 155, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.-F.; Wang, Y.-L.; Huang, Z.-Z.; Zhu, X.-C.; Zhang, F.-J.; Chen, F.-D.; Fang, W.-M.; Teng, N.-J. Effects of CO2 enrichment on growth and development of Impatiens hawkeri. Sci. World J. 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Zheng, Y.; Li, F.; Hao, L.; Shedayi, A.A.; Guo, L.; Ma, C.; Huang, B.; Xu, M. The optimal CO2 concentrations for the growth of three perennial grass species. BMC Plant Biol. 2018, 18, 27. [Google Scholar] [CrossRef]
- De Graaff, M.A.; van Groenigen, K.J.; Six, J.; Hungate, B.; van Kessel, C. Interactions between plant growth and soil nutrient cycling under elevated CO2: A meta-analysis. Glob. Chang. Biol. 2006, 12, 2077–2091. [Google Scholar] [CrossRef]
- Van Groenigen, K.-J.; Six, J.; Hungate, B.A.; de Graaff, M.-A.; van Breemen, N.; van Kessel, C. Element interactions limit soil carbon storage. Proc. Natl. Acad. Sci. USA 2006, 103, 6571–6574. [Google Scholar] [CrossRef]
- Gifford, R.M.; Barrett, D.J.; Lutze, J.L. The effects of elevated [CO2] on the C:N and C:P mass ratios of plant tissues. Plant Soil. 2000, 224, 1–14. [Google Scholar] [CrossRef]
- Zhu, C.; Zhu, J.; Zeng, Q.; Liu, G.; Xie, Z.; Tang, H.; Cao, J.; Zhao, X. Elevated CO2 accelerates flag leaf senescence in wheat due to ear photosynthesis which causes greater ear nitrogen sink capacity and ear carbon sink limitation. Funct Plant Biol. 2009, 36, 291–299. [Google Scholar] [CrossRef] [PubMed]
- De la Mata, L.; Cabello, P.; de la Haba, P.; Agüera, E. Growth under elevated atmospheric CO2 concentration accelerates leaf senescence in sunflower (Helianthus annuus L.) plants. J. Plant Physiol. 2012, 169, 1392–1400. [Google Scholar] [CrossRef] [PubMed]
- West, J.B.; HilleRisLambers, J.; Lee, T.D.; Hobbie, S.E.; Reich, P.B. Legume species identity and soil nitrogen supply determine symbiotic nitrogen-fixation responses to elevated atmospheric [CO2]. New Phytol. 2005, 167, 523–530. [Google Scholar] [CrossRef]
- Zanetti, S.; Hartwig, U.A.; Luscher, A.; Hebeisen, T.; Frehner, M.; Fischer, B.U.; Hendrey, G.R.; Blum, H.; Nosberger, J. Stimulation of symbiotic N2 fixation in Trifolium repens L. under elevated atmospheric pCO2 in a grassland ecosystem. Plant Physiol. 1996, 112, 575–583. [Google Scholar] [CrossRef] [PubMed]
- Rogers, A.; Ainsworth, E.A.; Leakey, A.D.B. Will elevated carbon dioxide concentration amplify the benefits of nitrogen fixation in legumes? Plant Physiol. 2009, 151, 1009–1016. [Google Scholar] [CrossRef]
- Hungate, B.A.; Stiling, P.D.; Dijkstra, P.; Johnson, D.W.; Ketterer, M.E.; Hymus, G.J.; Hinkle, C.R.; Drake, B.G. CO2 elicits long-term decline in nitrogen fixation. Science 2004, 304, 1291. [Google Scholar] [CrossRef] [PubMed]
- Edwards, E.J.; McCaffery, S.; Evans, J.R. Phosphorus availability and elevated CO2 affect biological nitrogen fixation and nutrient fluxes in a clover-dominated sward. New Phytol. 2006, 169, 157–167. [Google Scholar] [CrossRef]
- Manzoni, S. Flexible carbon-use efficiency across litter types and during decomposition partly compensates nutrient imbalances—results from analytical stoichiometric models. Front. Microbiol. 2017, 8, 661. [Google Scholar] [CrossRef] [PubMed]
- War, A.R.; Paulraj, M.G.; Ahmad, T.; Buhroo, A.A.; Hussain, B.; Ignacimuthu, S.; Sharma, H.C. Mechanisms of plant defense against insect herbivores. Plant Signal. Behav. 2012, 7, 1306–1320. [Google Scholar] [CrossRef]
- Kaplan, I.; Halitschke, R.; Kessler, A.; Sardanelli, S.; Denno, R.F. Constitutive and induced defenses to herbivory in above-and belowground plant tissues. Ecology 2008, 89, 392–406. [Google Scholar] [CrossRef]
- Cervilla, L.M.; Blasco, B.; Rios, J.J.; Rosales, M.A.; Sánchez-Rodríguez, E.; Rubio-Wilhelmi, M.M.; Romero, L.; Ruiz, J.M. Parameters symptomatic for boron toxicity in leaves of tomato plants. J. Bot. 2012, 2012, 1–17. [Google Scholar] [CrossRef]
- Kovalikova, Z.; Kubes, J.; Skalicky, M.; Kuchtickova, N.; Maskova, L.; Tuma, J.; Vachova, P.; Hejnak, V. Changes in content of polyphenols and ascorbic acid in leaves of white cabbage after pest infestation. Molecules 2019, 24, 2622. [Google Scholar] [CrossRef]
- Fürstenberg-Hägg, J.; Zagrobelny, M.; Bak, S. Plant defense against insect herbivores. Int. J. Mol. Sci. 2013, 14, 10242–10297. [Google Scholar] [CrossRef]
- Luczynski, A.; Isman, M.B.; Raworth, D.A. Strawberry foliar phenolics and their relationship to development of the two spotted spider mite. J. Econ. Entomol. 1990, 83, 557–563. [Google Scholar] [CrossRef]
- Xu, H.; Xie, H.; Wu, S.; Wang, Z.; He, K. Effects of elevated CO2 and increased N fertilization on plant secondary metabolites and chewing insect fitness. Front. Plant Sci. 2019, 10, 739. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.S. Inducible direct plant defense against insect herbivores: A review. Insect Sci. 2008, 15, 101–114. [Google Scholar] [CrossRef]
- Taylor, B.N.; Ostrowsky, L.R. Nitrogen-fixing and non-fixing trees differ in leaf chemistry and defence but not herbivory in a lowland Costa Rican rain forest. J. Trop. Ecol. 2019, 35, 270–279. [Google Scholar] [CrossRef]
- Chen, H.; Renault, S.; Markham, J. The effect of Frankia and multiple ectomycorrhizal fungil species on Alnus growing in low fertility soil. Symbiosis 2020, 80, 207–215. [Google Scholar] [CrossRef]
- Markham, J.H.; Zekveld, C. Nitrogen fixation makes biomass allocation to roots independent of soil nitrogen supply. Can. J. Bot. 2007, 85, 787–793. [Google Scholar] [CrossRef]
- Murray, T.J.; Tissue, D.T.; Ellsworth, D.S.; Riegler, M. Interactive effects of pre-industrial, current and future CO2 and temperature on an insect herbivore of eucalyptus. Oecologia 2013, 171, 1025–1035. [Google Scholar] [CrossRef]
- Chen, H.; Markham, J. Using microcontrollers and sensors to build an inexpensive CO2 control system for growth chambers. APPS 2020, 8, 8–12. [Google Scholar] [CrossRef]
- Boddey, R.M.; Peoples, M.B.; Palmer, B.; Dart, P.J. Use of the 15N natural abundance technique to quantify biological nitrogen fixation by woody perennials. Nutr. Cycl. Agroecosyst. 2000, 57, 235–270. [Google Scholar] [CrossRef]
- Alinian, S.; Razmjoo, J.; Zeinali, H. Flavonoids, anthocynins, phenolics and essential oil produced in cumin (Cuminum cyminum L.) accessions under different irrigation regimes. Ind. Crops Prod. 2016, 81, 49–55. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Rocha, A.M.C.N.; Morais, A.M.M.B. Polyphenoloxidase activity and total phenolic content as related to browning of minimally processed ‘Jonagored’ apple. J. Sci. Food Agric. 2002, 82, 120–126. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Markham, J. The Interactive Effect of Elevated CO2 and Herbivores on the Nitrogen-Fixing Plant Alnus incana ssp. rugosa. Plants 2021, 10, 440. https://doi.org/10.3390/plants10030440
Chen H, Markham J. The Interactive Effect of Elevated CO2 and Herbivores on the Nitrogen-Fixing Plant Alnus incana ssp. rugosa. Plants. 2021; 10(3):440. https://doi.org/10.3390/plants10030440
Chicago/Turabian StyleChen, Haoran, and John Markham. 2021. "The Interactive Effect of Elevated CO2 and Herbivores on the Nitrogen-Fixing Plant Alnus incana ssp. rugosa" Plants 10, no. 3: 440. https://doi.org/10.3390/plants10030440
APA StyleChen, H., & Markham, J. (2021). The Interactive Effect of Elevated CO2 and Herbivores on the Nitrogen-Fixing Plant Alnus incana ssp. rugosa. Plants, 10(3), 440. https://doi.org/10.3390/plants10030440





